Diazoxide for Neonatal Hyperinsulinemic Hypoglycemia and Pulmonary Hypertension
Abstract
1. Introduction
2. Pharmacology of Diazoxide
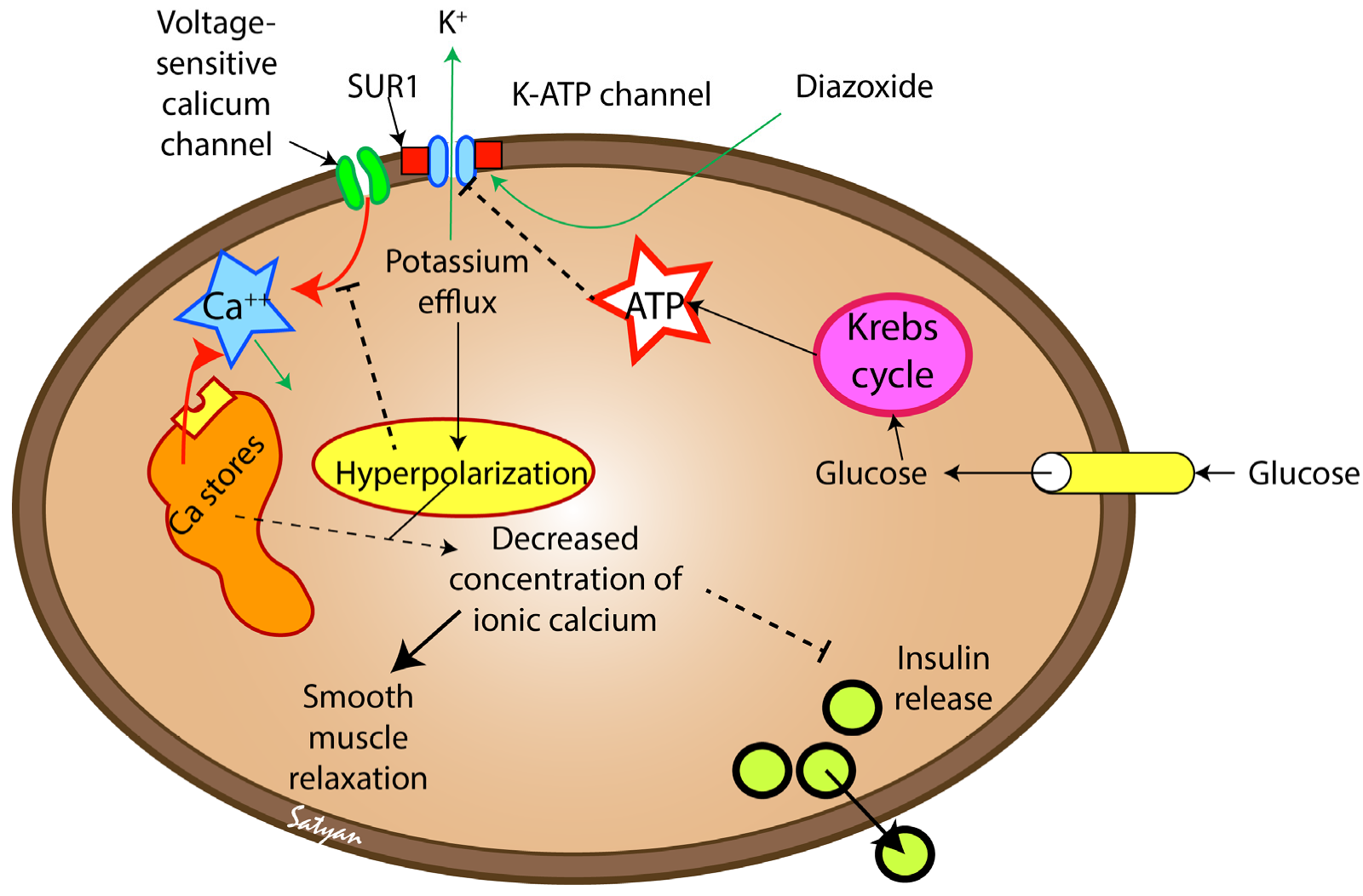
2.1. Mechanism of Action
2.2. Pharmacokinetics and Dosing of Diazoxide
3. Diazoxide Use in Hyperinsulinism
3.1. Hyperinsulinemic Hypoglycemia
3.2. Diazoxide in Hyperinsulinemia Hypoglycemia
3.3. Diazoxide Use in HH and Cardiovascular Complications
3.4. Diazoxide and Necrotizing Enterocolitis
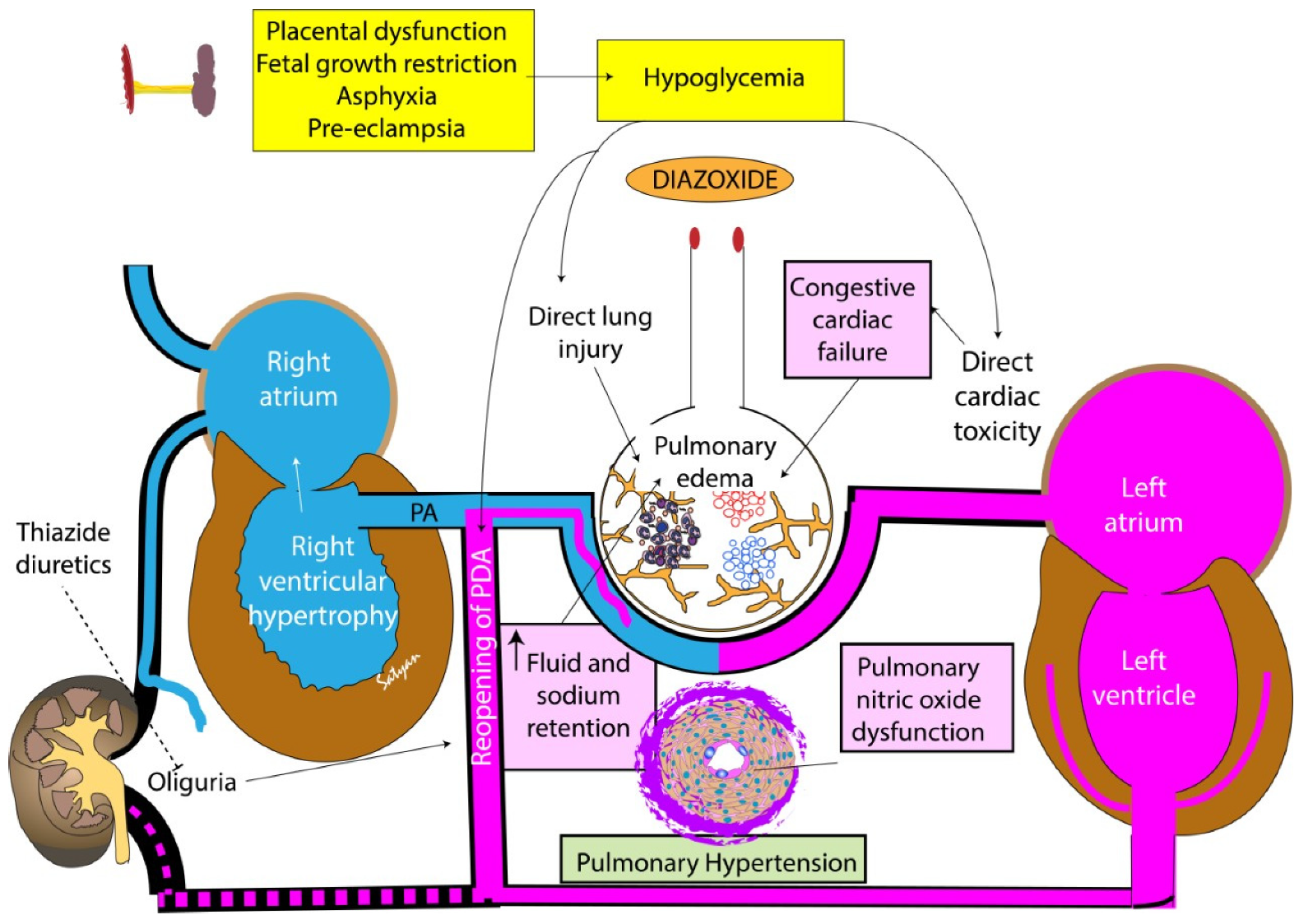
4. Pathogenesis of Pulmonary Hypertension and Heart Failure after Diazoxide Use
5. Other Adverse Effects of Diazoxide Use in Neonates
6. Case Series from a Single Center (University of California Davis NICU)
7. Current Evidence on Diazoxide Use in Neonates
8. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Güemes, M.; Hussain, K. Hyperinsulinemic hypoglycemia. Pediatr. Clin. 2015, 62, 1017–1036. [Google Scholar] [CrossRef] [PubMed]
- Thornton, P.S.; Stanley, C.A.; De Leon, D.D.; Harris, D.; Haymond, M.W.; Hussain, K.; Levitsky, L.L.; Murad, M.H.; Rozance, P.J.; Simmons, R.A.; et al. Recommendations from the Pediatric Endocrine Society for evaluation and management of persistent hypoglycemia in neonates, infants, and children. J. Pediatr. 2015, 167, 238–245. [Google Scholar] [CrossRef] [PubMed]
- Adamkin, D.H. Committee on Fetus and Newborn. Postnatal glucose homeostasis in late-preterm and term infants. Pediatrics 2011, 127, 575–579. [Google Scholar] [CrossRef] [PubMed]
- Gray, K.D.; Dudash, K.; Escobar, C.; Freel, C.; Harrison, T.; McMillan, C.; Puia-Dumitrescu, M.; Cotten, C.M.; Benjamin, R.; Clark, R.H.; et al. Prevalence and safety of diazoxide in the neonatal intensive care unit. J. Perinatol. 2018, 38, 1496–1502. [Google Scholar] [CrossRef]
- Keyes, M.L.; Healy, H.; Sparger, K.A.; Orth, L.E.; Geha, M.; Roumiantsev, S.; Matute, J.D. Necrotizing Enterocolitis in Neonates With Hyperinsulinemic Hypoglycemia Treated With Diazoxide. Pediatrics 2021, 147, e20193202. [Google Scholar] [CrossRef]
- Chen, S.C.; Dastamani, A.; Pintus, D.; Yau, D.; Aftab, S.; Bath, L.; Swinburne, C.; Hunter, L.; Giardini, A.; Christov, G.; et al. Diazoxide-induced pulmonary hypertension in hyperinsulinaemic hypoglycaemia: Recommendations from a multicentre study in the United Kingdom. Clin. Endocrinol. 2019, 91, 770–775. [Google Scholar] [CrossRef]
- Herrera, A.; Vajravelu, M.E.; Givler, S.; Mitteer, L.; Avitabile, C.M.; Lord, K.; De León, D.D. Prevalence of Adverse Events in Children With Congenital Hyperinsulinism Treated With Diazoxide. J. Clin. Endocrinol. Metab. 2018, 103, 4365–4372. [Google Scholar] [CrossRef]
- Timlin, M.R.; Black, A.B.; Delaney, H.M.; Matos, R.I.; Percival, C.S. Development of Pulmonary Hypertension During Treatment with Diazoxide: A Case Series and Literature Review. Pediatr. Cardiol. 2017, 38, 1247–1250. [Google Scholar] [CrossRef]
- De Cosio, A.P.; Thornton, P. Current and Emerging Agents for the Treatment of Hypoglycemia in Patients with Congenital Hyperinsulinism. Paediatr. Drugs 2019, 21, 123–136. [Google Scholar] [CrossRef]
- Seltzer, H.S.; Allen, E.W. Hyperglycemia and inhibition of insulin secretion during administration of diazoxide and trichlormethiazide in man. Diabetes 1969, 18, 19–28. [Google Scholar] [CrossRef]
- Coetzee, W.A. Multiplicity of effectors of the cardioprotective agent, diazoxide. Pharmacol. Ther. 2013, 140, 167–175. [Google Scholar] [CrossRef] [PubMed]
- Kizu, R.; Nishimura, K.; Sato, R.; Kosaki, K.; Tanaka, T.; Tanigawara, Y.; Hasegawa, T. Population Pharmacokinetics of Diazoxide in Children with Hyperinsulinemic Hypoglycemia. Horm. Res. Paediatr. 2017, 88, 316–323. [Google Scholar] [CrossRef] [PubMed]
- Pruitt, A.W.; Faraj, B.A.; Dayton, P.G. Metabolism of diazoxide in man and experimental animals. J. Pharmacol. Exp. Ther. 1974, 188, 248–256. [Google Scholar] [PubMed]
- Drash, A.; Wolff, F. Drug Therapy in Leucine-Sensitive Hypoglycemia. Metabolism 1964, 13, 487–492. [Google Scholar] [CrossRef] [PubMed]
- Raisingani, M.; Brar, P.C. Characterization of the duration of treatment with diazoxide in infants with prolonged hyperinsulinism (PHI). J. Pediatr. Endocrinol. Metab. 2019, 32, 1241–1245. [Google Scholar] [CrossRef]
- Brar, P.C.; Heksch, R.; Cossen, K.; De Leon, D.D.; Kamboj, M.K.; Marks, S.D.; Marshall, B.A.; Miller, R.; Page, L.; Stanley, T.; et al. Management and Appropriate Use of Diazoxide in Infants and Children with Hyperinsulinism. J.Clin. Endocrinol. Metab. 2020, 105, 3750–3761. [Google Scholar] [CrossRef]
- Yoshida, K.; Kawai, M.; Marumo, C.; Kanazawa, H.; Matsukura, T.; Kusuda, S.; Yorifuji, T.; Heike, T. High prevalence of severe circulatory complications with diazoxide in premature infants. Neonatology 2014, 105, 166–171. [Google Scholar] [CrossRef]
- Menni, F.; de Lonlay, P.; Sevin, C.; Touati, G.; Peigné, C.; Barbier, V.; Nihoul-Fékété, C.; Saudubray, J.M.; Robert, J.J. Neurologic outcomes of 90 neonates and infants with persistent hyperinsulinemic hypoglycemia. Pediatrics 2001, 107, 476–479. [Google Scholar] [CrossRef]
- Fafoula, O.; Alkhayyat, H.; Hussain, K. Prolonged hyperinsulinaemic hypoglycaemia in newborns with intrauterine growth retardation. Arch. Dis. Child. Fetal Neonatal Ed. 2006, 91, F467. [Google Scholar] [CrossRef]
- Hoe, F.M.; Thornton, P.S.; Wanner, L.A.; Steinkrauss, L.; Simmons, R.A.; Stanley, C.A. Clinical features and insulin regulation in infants with a syndrome of prolonged neonatal hyperinsulinism. J. Pediatr. 2006, 148, 207–212. [Google Scholar] [CrossRef]
- Demirbilek, H.; Hussain, K. Congenital hyperinsulinism: Diagnosis and treatment update. J. Clin. Res. Pediatr. Endocrinol. 2017, 9 (Suppl. 2), 69. [Google Scholar] [CrossRef] [PubMed]
- Med, D. Label: Proglycem—Diazoxide Suspension. Drug Label Information 2022. Available online: https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=b16c7832-2fd9-49af-b923-1dc0d91fd6e2 (accessed on 28 November 2022).
- Hu, S.; Xu, Z.; Yan, J.; Liu, M.; Sun, B.; Li, W.; Sang, Y. The treatment effect of diazoxide on 44 patients with congenital hyperinsulinism. J. Pediatr. Endocrinol. Metab. 2012, 25, 1119–1122. [Google Scholar] [CrossRef] [PubMed]
- Palladino, A.A.; Stanley, C.A. A specialized team approach to diagnosis and medical versus surgical treatment of infants with congenital hyperinsulinism. Semin. Pediatr. Surg. 2011, 20, 32–37. [Google Scholar] [CrossRef] [PubMed]
- Laing, D.; Hanning, S.M.; Harding, J.E.; Mravicich, L.C.; McKinlay, C.J. Diazoxide for the Treatment of Transitional Neonatal Hypoglycemia: A Systematic Review. J. Neonatol. 2021, 35, 203–208. [Google Scholar] [CrossRef]
- Balachandran, B.; Mukhopadhyay, K.; Sachdeva, N.; Walia, R.; Attri, S.V. Randomised controlled trial of diazoxide for small for gestational age neonates with hyperinsulinaemic hypoglycaemia provided early hypoglycaemic control without adverse effects. Acta Paediatr. 2018, 107, 990–995. [Google Scholar] [CrossRef] [PubMed]
- Davidov, A.S.; Elkon-Tamir, E.; Haham, A.; Shefer, G.; Weintrob, N.; Oren, A.; Lebenthal, Y.; Mandel, D.; Eyal, O. Higher C-peptide levels and glucose requirements may identify neonates with transient hyperinsulinism hypoglycemia who will benefit from diazoxide treatment. Eur. J. Pediatr. 2020, 179, 597–602. [Google Scholar] [CrossRef]
- Aynsley-Green, A.; Hussain, K.; Hall, J.; Saudubray, J.M.; Nihoul-Fékété, C.; De Lonlay-Debeney, P.; Brunelle, F.; Otonkoski, T.; Thornton, P.; Lindley, K.J. Practical management of hyperinsulinism in infancy. Arch. Dis. Child. Fetal Neonatal. Ed. 2000, 82, F98–F107. [Google Scholar] [CrossRef]
- Arnoux, J.B.; Verkarre, V.; Saint-Martin, C.; Montravers, F.; Brassier, A.; Valayannopoulos, V.; Brunelle, F.; Fournet, J.C.; Robert, J.J.; Aigrain, Y.; et al. Congenital hyperinsulinism: Current trends in diagnosis and therapy. Orphanet. J. Rare Dis. 2011, 6, 63. [Google Scholar] [CrossRef]
- Kapoor, R.R.; Flanagan, S.E.; James, C.; Shield, J.; Ellard, S.; Hussain, K. Hyperinsulinaemic hypoglycaemia. Arch. Dis. Child. 2009, 94, 450–457. [Google Scholar] [CrossRef]
- Thornton, P.; Truong, L.; Reynolds, C.; Hamby, T.; Nedrelow, J. Rate of Serious Adverse Events Associated with Diazoxide Treatment of Patients with Hyperinsulinism. Horm. Res. Paediatr. 2019, 91, 25–32. [Google Scholar] [CrossRef]
- FDA. FDA Drug Safety Communication. Available online: https://www.fda.gov/drugs/drug-safety-and-availability/fda-drug-safety-communication-fda-warns-about-serious-lung-condition-infants-and-newborns-treated (accessed on 12 September 2022).
- Gerardin, M.; Denizot, S.; Texier, R.; Jolliet, P. Pulmonary Hypertension in new-borns treated with diazoxide: About two cases. In Fundamental & Clinical Pharmacology; Wiley-Blackwell Publishing, Inc.: Malden, MA, USA, 2010. [Google Scholar]
- Demirel, F.; Unal, S.; Çetin, I.I.; Esen, I.; Araslı, A. Pulmonary hypertension and reopening of the ductus arteriosus in an infant treated with diazoxide. J. Pediatr. Endocrinol. Metab. 2011, 24, 603–605. [Google Scholar] [CrossRef]
- Yildizdas, D.; Erdem, S.; Küçükosmanoglu, O.; Yilmaz, M.; Yüksel, B. Pulmonary hypertension, heart failure and neutropenia due to diazoxide therapy. Adv. Ther. 2008, 25, 515–519. [Google Scholar] [CrossRef]
- Desai, J.; Key, L.; Swindall, A.; Gaston, K.; Talati, A.J. The danger of diazoxide in the neonatal intensive care unit. Ther. Adv. Drug Saf. 2021, 12, 20420986211011338. [Google Scholar] [CrossRef]
- Prado, L.A.; Castro, M.; Weisz, D.E.; Jain, A.; Belik, J. Necrotising enterocolitis in newborns receiving diazoxide. Arch. Dis. Child. Fetal Neonatal. Ed. 2021, 106, 306–310. [Google Scholar] [CrossRef]
- Theodorou, C.M.; Hirose, S. Necrotizing enterocolitis following diazoxide therapy for persistent neonatal hypoglycemia. J. Pediatr. Surg. Case Rep. 2020, 52, 101356. [Google Scholar] [CrossRef]
- Klinke, W.P.; Gilbert, J.A. Diazoxide in primary pulmonary hypertension. N. Engl. J. Med. 1980, 302, 91–92. [Google Scholar] [CrossRef]
- Buch, J.; Wennevold, A. Hazards of diazoxide in pulmonary hypertension. Br. Heart J. 1981, 46, 401–403. [Google Scholar] [CrossRef]
- Farahini, H.; Ajami, M.; Mirzay Razaz, J.; Azad, N.; Soleimani, M.; Ayatollahi, S.A.; Abotaleb, N.; Peyrovi, H.; Pazoki-Toroudi, H. Nitric Oxide is Necessary for Diazoxide Protection Against Ischemic Injury in Skeletal Muscle. Iran. J. Pharm. Res. 2012, 11, 375–381. [Google Scholar]
- Nebesio, T.D.; Hoover, W.C.; Caldwell, R.L.; Nitu, M.E.; Eugster, E.A. Development of pulmonary hypertension in an infant treated with diazoxide. J. Pediatr. Endocrinol. Metab. 2007, 20, 939–944. [Google Scholar] [CrossRef]
- Silvani, P.; Camporesi, A.; Mandelli, A.; Wolfler, A.; Salvo, I. A case of severe diazoxide toxicity. Paediatr. Anaesth. 2004, 14, 607–609. [Google Scholar] [CrossRef]
- Fine, L.G.; Weber, H. Effect of diazoxide on renal handling of sodium in the rat. Clin. Sci. Mol. Med. 1975, 49, 277–282. [Google Scholar] [CrossRef] [PubMed]
- Tas, E.; Mahmood, B.; Garibaldi, L.; Sperling, M. Liver injury may increase the risk of diazoxide toxicity: A case report. Eur. J. Pediatr. 2015, 174, 403–406. [Google Scholar] [CrossRef] [PubMed]
- Godinho, F.; Lewin, R.; Park, J.; Losa, I. Case of raised creatinine in a newborn with congenital hyperinsulinism: Diazoxide induced acute kidney injury. In Endocrine Abstracts; Bioscientifica: Bristol, UK, 2015. [Google Scholar]
- Hastings, L.A.; Preddy, J.; McCready, M.; Neville, K.; Verge, C.F. Pericardial Effusion Associated with Diazoxide Treatment for Congenital Hyperinsulinism. Horm. Res. Paediatr. 2020, 93, 206–211. [Google Scholar] [CrossRef] [PubMed]
- Maffre, I.; Vincenti, M.; Dalla Vale, F.; Amouroux, C.; Werner, O.; Meilhac, A.; de Barry, G.; Amedro, P. Diazoxide Causality Assessment of a Pericardial Effusion in a Child with Kabuki Syndrome. J. Clin. Res. Pediatr. Endocrinol. 2019, 11, 218–219. [Google Scholar] [CrossRef]
- Fogal, B.; McClaskey, C.; Yan, S.; Yan, H.; Rivkees, S.A. Diazoxide promotes oligodendrocyte precursor cell proliferation and myelination. PLoS ONE 2010, 5, e10906. [Google Scholar] [CrossRef]
- Domoki, F.; Kis, B.; Nagy, K.; Farkas, E.; Busija, D.W.; Bari, F. Diazoxide preserves hypercapnia-induced arteriolar vasodilation after global cerebral ischemia in piglets. Am. J. Physiol. Heart Circ. Physiol. 2005, 289, H368–H373. [Google Scholar] [CrossRef]
- Katakam, P.V.; Dutta, S.; Sure, V.N.; Grovenburg, S.M.; Gordon, A.O.; Peterson, N.R.; Rutkai, I.; Busija, D.W. Depolarization of mitochondria in neurons promotes activation of nitric oxide synthase and generation of nitric oxide. Am. J. Physiol. Heart Circ. Physiol. 2016, 310, H1097–H1106. [Google Scholar] [CrossRef]
- Lenzsér, G.; Kis, B.; Bari, F.; Busija, D.W. Diazoxide preconditioning attenuates global cerebral ischemia-induced blood-brain barrier permeability. Brain Res. 2005, 1051, 72–80. [Google Scholar] [CrossRef]
- Chen, Y.; Zeng, H.; Liu, H. MiR-21 participates in the neuroprotection of diazoxide against hypoxic-ischemia encephalopathy by targeting PDCD4. Brain Inj. 2022, 36, 876–885. [Google Scholar] [CrossRef]
| Characteristic | Pulmonary Hypertension (n = 4) | No Pulmonary Hypertension (n = 17) | p-Value |
|---|---|---|---|
| Type of HH Transient Permanent | 4 (100%) 0 | 16 (94%) 1 (6%) | |
| Gestational Age (weeks) Mean (SD) | 31.25 (3.9) | 35.29 (3.3) | p = 0.043 |
| Birthweight (g) Mean (SD) | 1320 (911) | 1965 (672) | p = 0.12 |
| IUGR Yes Yes, with AEDF/REDF * | 3 (75%) 2 (50%) | 9 (53%) 1 (6%) | p = 0.6 |
| Age at Diazoxide Start <7 days 7–14 days >14 days | 1/4 (25%) 1/4 (25%) 2/4 (50%) | 5/17 (30%) 5/17 (30%) 7/17 (40%) | p = 0.9 |
| Starting Diazoxide Dose 10 mg/kg/d 5–9 mg/kg/d Less than 5 mg/kg/d | 3 (75%) 1 (25%) 0 | 4 (24%) 12 (70%) 1 (6%) | p = 0.08 |
| Prior Echocardiography Echo prior to diazoxide Mild Septal Flattening TR jet estimate > 45 mmHg | 4 (100%) 2 (50%) 1 (25%) | 11 (65%) 1 (6%) 1 ** (6%) | p = 0.07 |
| Respiratory Support at Diazoxide Start Room Air Respiratory Support Nasal Cannula CPAP Invasive Mechanical Ventilation | 1 (25%) 3 (75%) 1 2 0 | 16 (94% 1 (6%) 0 0 1 ** | p = 0.01 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Newman-Lindsay, S.; Lakshminrusimha, S.; Sankaran, D. Diazoxide for Neonatal Hyperinsulinemic Hypoglycemia and Pulmonary Hypertension. Children 2023, 10, 5. https://doi.org/10.3390/children10010005
Newman-Lindsay S, Lakshminrusimha S, Sankaran D. Diazoxide for Neonatal Hyperinsulinemic Hypoglycemia and Pulmonary Hypertension. Children. 2023; 10(1):5. https://doi.org/10.3390/children10010005
Chicago/Turabian StyleNewman-Lindsay, Shoshana, Satyan Lakshminrusimha, and Deepika Sankaran. 2023. "Diazoxide for Neonatal Hyperinsulinemic Hypoglycemia and Pulmonary Hypertension" Children 10, no. 1: 5. https://doi.org/10.3390/children10010005
APA StyleNewman-Lindsay, S., Lakshminrusimha, S., & Sankaran, D. (2023). Diazoxide for Neonatal Hyperinsulinemic Hypoglycemia and Pulmonary Hypertension. Children, 10(1), 5. https://doi.org/10.3390/children10010005









