Recent Advances in Hydrogels: Ophthalmic Applications in Cell Delivery, Vitreous Substitutes, and Ocular Adhesives
Abstract
1. Introduction
2. Hydrogel Types and Classifications
3. Hydrogels Used in Cell and Stem Cell Delivery
4. Hydrogels Used as Vitreous Substitutes
5. Hydrogels Used as Ocular Adhesives in Corneal Wounds and Incisions
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| BDDE | 1,4-Butanediol diglycidyl ether |
| MTT | 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl-2H-tetrazolium bromide |
| AMD | age-related macular degeneration |
| ADA | alginate dialdehyde |
| CGP | β-glycerophosphate-bound chitosan |
| BM-MSC | bone marrow mesenchymal stem cells |
| CMCTS | carboxymethyl chitosan |
| CS | chitosan |
| CPX | ciprofloxacin |
| CCI | clear corneal incisions |
| CT | cross-linking time |
| FD | degree of methacrylation |
| DMEK | Descemet membrane endothelial keratoplasty |
| Alg-DA | dopamine-modified alginate |
| dsDNA | double stranded DNA |
| CE | European Conformity |
| ECM | extracellular matrix |
| FDA | Food and Drug Administration |
| FT-IR | Fourier-transform infrared spectroscopy |
| GE | gelatin |
| GelMA | gelatin methacryloyl |
| GELGYM | gelatin-glycidyl-methacrylate hydrogel |
| gel-HA | gelatin-hyaluronic acid |
| Gtn-HPA | gelatin-hydroxyphenyl propionic acid |
| GelCORE + MC | GelCORE loaded with micelles |
| GG | Gellan Gum |
| DFG | Gellan gum grafted with dopamine |
| AuNRs | gold nanorods |
| hRPCs | human RPCs |
| HAA | hyaluronan modified with aldehyde |
| HKA | hyaluronan modified with ketone |
| HPCTS | hydroxypropyl chitosan |
| ISO | International Organization for Standardization |
| IOL | intraocular lens |
| IOP | Intraocular pressure |
| HA-MA | methacrylated hyaluronic acid |
| D-MA | methacrylated hydroxyl dendrimer |
| NIR | near infrared |
| VC | N-vinylcaprolactam |
| OHA | oxidized hyaluronic acid |
| PEGOA₄ | PEG-tetraoxyamine |
| C3F8 | perfluorocarbon |
| PBS | phosphate buffered saline |
| PTT | photothermal therapy |
| iPSC | pluripotent stem cells |
| PCL | poly(3-caprolactone) |
| PASA | poly(aspartic acid) |
| PEGDA | poly(ethylene glycol) diacrylate |
| PEGMA | poly(ethylene glycol) methacrylate |
| PGS | poly(glycerol-sebacate) |
| PHIS | poly(histidine) |
| PGA | poly(L-glutamic acid) |
| PLLA/PLGA | poly(L-lactic acid)/poly(lactic-co-glycolic acid) |
| poly(MAM-co-MAA-co-BMAC | poly(methacrylamide-co-methacrylate-co-bis(methylacryloyl-cystamine)) |
| PNIPAM | poly(N-isopropylacrylamide) |
| PHBHx | poly(PHBHx/PEG/PPG urethane) (PHxEP), poly(ε-caprolactone) (PCL), poly[(R)-3-hydroxybutyrate-(R)-3-hydroxyhexanoate] |
| PDA | polydopamine |
| PEG | polyethylene glycol |
| PPG | polypropylene glycol |
| RD | retinal degenerative diseases |
| RGCs | retinal ganglion cells |
| RPE | retinal pigment epithelium |
| RPC | retinal progenitor cell |
| RP | retinitis pigmentosa |
| SF6 | Sulfur hexafluoride |
| SIA | surgical induced astigmatism |
| TEA | triethanolamine |
| UV | ultraviolet |
| UM | uveal melanoma |
| %wt | weight percent |
References
- Steinmetz, J.D.; Bourne, R.R.A.; Briant, P.S.; Flaxman, S.R.; Taylor, H.R.B.; Jonas, J.B.; Abdoli, A.A.; Abrha, W.A.; Abualhasan, A.; Abu-Gharbieh, E.G.; et al. Causes of Blindness and Vision Impairment in 2020 and Trends over 30 Years, and Prevalence of Avoidable Blindness in Relation to VISION 2020: The Right to Sight: An Analysis for the Global Burden of Disease Study. Lancet Glob. Health 2021, 9, e144–e160. [Google Scholar] [CrossRef]
- Bourne, R.; Steinmetz, J.D.; Flaxman, S.; Briant, P.S.; Taylor, H.R.; Resnikoff, S.; Casson, R.J.; Abdoli, A.; Abu-Gharbieh, E.; Afshin, A.; et al. Trends in Prevalence of Blindness and Distance and near Vision Impairment over 30 Years: An Analysis for the Global Burden of Disease Study. Lancet Glob. Health 2021, 9, e130–e143. [Google Scholar] [CrossRef]
- Fricke, T.R.; Tahhan, N.; Resnikoff, S.; Papas, E.; Burnett, A.; Ho, S.M.; Naduvilath, T.; Naidoo, K.S. Global Prevalence of Presbyopia and Vision Impairment from Uncorrected Presbyopia. Ophthalmology 2018, 125, 1492–1499. [Google Scholar] [CrossRef] [PubMed]
- National Academies of Sciences, Engineering, and Medicine. Committee on Public Health Approaches to Reduce Vision Impairment and Promote Eye Health; Board on Population Health and Public Health, Practice; Health and Medicine Division. In Making Eye Health a Population Health Imperative: Vision for Tomorrow; Teutsch, S.M., McCoy, M.A., Woodbury, R.B., Welp, A., Eds.; National Academies Press: Washington, DC, USA, 2016; p. 23471. ISBN 978-0-309-43998-5. [Google Scholar]
- Patel, A. Ocular Drug Delivery Systems: An Overview. World J. Pharmacol. 2013, 2, 47. [Google Scholar] [CrossRef] [PubMed]
- Gaudana, R.; Ananthula, H.K.; Parenky, A.; Mitra, A.K. Ocular Drug Delivery. AAPS J. 2010, 12, 348–360. [Google Scholar] [CrossRef] [PubMed]
- Willoughby, C.E.; Ponzin, D.; Ferrari, S.; Lobo, A.; Landau, K.; Omidi, Y. Anatomy and Physiology of the Human Eye: Effects of Mucopolysaccharidoses Disease on Structure and Function-a Review: Anatomy and Physiology of the Eye. Clin. Exp. Ophthalmol. 2010, 38, 2–11. [Google Scholar] [CrossRef]
- Souto, E.B.; Dias-Ferreira, J.; López-Machado, A.; Ettcheto, M.; Cano, A.; Camins Espuny, A.; Espina, M.; Garcia, M.L.; Sánchez-López, E. Advanced Formulation Approaches for Ocular Drug Delivery: State-Of-The-Art and Recent Patents. Pharmaceutics 2019, 11, 460. [Google Scholar] [CrossRef]
- Smolle, M.; Keller, C.; Pinggera, G.; Deibl, M.; Rieder, J.; Lirk, P. Clear Hydro-Gel, Compared to Ointment, Provides Improved Eye Comfort after Brief Surgery. Can. J. Anesth./J. Canadien d’Anesthésie 2004, 51, 126–129. [Google Scholar] [CrossRef]
- Varela-Fernández, R.; Díaz-Tomé, V.; Luaces-Rodríguez, A.; Conde-Penedo, A.; García-Otero, X.; Luzardo-Álvarez, A.; Fernández-Ferreiro, A.; Otero-Espinar, F. Drug Delivery to the Posterior Segment of the Eye: Biopharmaceutic and Pharmacokinetic Considerations. Pharmaceutics 2020, 12, 269. [Google Scholar] [CrossRef]
- Cox, J.T.; Eliott, D.; Sobrin, L. Inflammatory Complications of Intravitreal Anti-VEGF Injections. J. Clin. Med. 2021, 10, 981. [Google Scholar] [CrossRef]
- Sampat, K.M.; Garg, S.J. Complications of Intravitreal Injections. Curr. Opin. Ophthalmol. 2010, 21, 178–183. [Google Scholar] [CrossRef] [PubMed]
- Shikari, H.; Silva, P.S.; Sun, J.K. Complications of Intravitreal Injections in Patients with Diabetes. Semin. Ophthalmol. 2014, 29, 276–289. [Google Scholar] [CrossRef] [PubMed]
- Kang-Mieler, J.J.; Rudeen, K.M.; Liu, W.; Mieler, W.F. Advances in Ocular Drug Delivery Systems. Eye 2020, 34, 1371–1379. [Google Scholar] [CrossRef] [PubMed]
- Allen, D.; Vasavada, A. Cataract and Surgery for Cataract. BMJ 2006, 333, 128–132. [Google Scholar] [CrossRef]
- Davis, G. The Evolution of Cataract Surgery. Mo. Med. 2016, 113, 5. [Google Scholar]
- Henry, C.R.; Flynn Jr, H.W.; Miller, D.; Schefler, A.C.; Forster, R.K.; Alfonso, E.C. Delayed-Onset Endophthalmitis Associated with Corneal Suture Infections. J. Ophthalmic Inflamm. Infect. 2013, 3, 51. [Google Scholar] [CrossRef]
- Churchill, A.J.; Hillman, J.S. Post-Operative Astigmatism Control by Selective Suture Removal. Eye 1996, 10, 103–106. [Google Scholar] [CrossRef]
- Satitpitakul, V.; Uramphorn, N.; Kasetsuwan, N. Factors Predicting Change in Corneal Astigmatism Following Suture Removal in Post-Penetrating Keratoplasty Patients. Clin. Ophtalmol. 2019, 13, 1593–1597. [Google Scholar] [CrossRef] [PubMed]
- Lee, B.J.; Smith, S.D.; Jeng, B.H. Suture-Related Corneal Infections after Clear Corneal Cataract Surgery. J. Cataract Refract. Surg. 2009, 35, 939–942. [Google Scholar] [CrossRef]
- Stanford, M.R.; Fenech, T.; Hunter, P.A. Timing of Removal of Sutures in Control of Post-Operative Astigmatism. Eye 1993, 7, 143–147. [Google Scholar] [CrossRef]
- Danjoux, J.P.; Reck, A.C. Corneal Sutures: Is Routine Removal Really Necessary? Eye 1994, 8, 339–342. [Google Scholar] [CrossRef][Green Version]
- Wang, W.; Lo, A. Diabetic Retinopathy: Pathophysiology and Treatments. Int. J. Mol. Sci. 2018, 19, 1816. [Google Scholar] [CrossRef]
- Duh, E.J.; Sun, J.K.; Stitt, A.W. Diabetic Retinopathy: Current Understanding, Mechanisms, and Treatment Strategies. JCI Insight 2017, 2, e93751. [Google Scholar] [CrossRef]
- Corcóstegui, B.; Durán, S.; González-Albarrán, M.O.; Hernández, C.; Ruiz-Moreno, J.M.; Salvador, J.; Udaondo, P.; Simó, R. Update on Diagnosis and Treatment of Diabetic Retinopathy: A Consensus Guideline of the Working Group of Ocular Health (Spanish Society of Diabetes and Spanish Vitreous and Retina Society). J. Ophthalmol. 2017, 2017, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Donati, S.; Caprani, S.M.; Airaghi, G.; Vinciguerra, R.; Bartalena, L.; Testa, F.; Mariotti, C.; Porta, G.; Simonelli, F.; Azzolini, C. Vitreous Substitutes: The Present and the Future. BioMed Res. Int. 2014, 2014, 351804. [Google Scholar] [CrossRef]
- Yadav, I.; Purohit, S.D.; Singh, H.; Bhushan, S.; Yadav, M.K.; Velpandian, T.; Chawla, R.; Hazra, S.; Mishra, N.C. Vitreous Substitutes: An Overview of the Properties, Importance, and Development. J. Biomed. Mater. Res. 2021, 109, 1156–1176. [Google Scholar] [CrossRef]
- Mondelo-García, C.; Bandín-Vilar, E.; García-Quintanilla, L.; Castro-Balado, A.; del Amo, E.M.; Gil-Martínez, M.; Blanco-Teijeiro, M.J.; González-Barcia, M.; Zarra-Ferro, I.; Fernández-Ferreiro, A.; et al. Current Situation and Challenges in Vitreous Substitutes. Macromol. Biosci. 2021, 21, 2100066. [Google Scholar] [CrossRef] [PubMed]
- Gaddam, S.; Periasamy, R.; Gangaraju, R. Adult Stem Cell Therapeutics in Diabetic Retinopathy. Int. J. Mol. Sci. 2019, 20, 4876. [Google Scholar] [CrossRef] [PubMed]
- Kirchhof, S.; Goepferich, A.M.; Brandl, F.P. Hydrogels in Ophthalmic Applications. Eur. J. Pharm. Biopharm. 2015, 95, 227–238. [Google Scholar] [CrossRef] [PubMed]
- Wang, K.; Han, Z. Injectable Hydrogels for Ophthalmic Applications. J. Control. Release 2017, 268, 212–224. [Google Scholar] [CrossRef]
- Parhi, R. Cross-Linked Hydrogel for Pharmaceutical Applications: A Review. Adv. Pharm. Bull. 2017, 7, 515–530. [Google Scholar] [CrossRef]
- Thakur, V.K.; Thakur, M.K. (Eds.) Hydrogels: Recent Advances; Gels Horizons: From Science to Smart Materials; Springer: Singapore, 2018; ISBN 978-981-10-6076-2. [Google Scholar]
- Lynch, C.R.; Kondiah, P.P.D.; Choonara, Y.E.; du Toit, L.C.; Ally, N.; Pillay, V. Hydrogel Biomaterials for Application in Ocular Drug Delivery. Front. Bioeng. Biotechnol. 2020, 8, 228. [Google Scholar] [CrossRef]
- Trujillo-de Santiago, G.; Sharifi, R.; Yue, K.; Sani, E.S.; Kashaf, S.S.; Alvarez, M.M.; Leijten, J.; Khademhosseini, A.; Dana, R.; Annabi, N. Ocular Adhesives: Design, Chemistry, Crosslinking Mechanisms, and Applications. Biomaterials 2019, 197, 345–367. [Google Scholar] [CrossRef] [PubMed]
- Swindle-Reilly, K.E.; Reilly, M.A.; Ravi, N. Current concepts in the design of hydrogels as vitreous substitutes. In Biomaterials and Regenerative Medicine in Ophthalmology; Elsevier: Amsterdam, The Netherlands, 2016; pp. 101–130. ISBN 978-0-08-100147-9. [Google Scholar]
- Mantha, S.; Pillai, S.; Khayambashi, P.; Upadhyay, A.; Zhang, Y.; Tao, O.; Pham, H.M.; Tran, S.D. Smart Hydrogels in Tissue Engineering and Regenerative Medicine. Materials 2019, 12, 3323. [Google Scholar] [CrossRef]
- Rim, M.A.; Choi, J.H.; Park, A.; Youn, J.; Lee, S.; Kim, N.E.; Song, J.E.; Khang, G. Characterization of Gelatin/Gellan Gum/Glycol Chitosan Ternary Hydrogel for Retinal Pigment Epithelial Tissue Reconstruction Materials. ACS Appl. Bio Mater. 2020, 3, 6079–6087. [Google Scholar] [CrossRef]
- Bhattacharjee, P.; Ahearne, M. Significance of Crosslinking Approaches in the Development of Next Generation Hydrogels for Corneal Tissue Engineering. Pharmaceutics 2021, 13, 319. [Google Scholar] [CrossRef] [PubMed]
- Patenaude, M.; Smeets, N.M.B.; Hoare, T. Designing Injectable, Covalently Cross-Linked Hydrogels for Biomedical Applications. Macromol. Rapid Commun. 2014, 35, 598–617. [Google Scholar] [CrossRef]
- Trombino, S.; Servidio, C.; Curcio, F.; Cassano, R. Strategies for Hyaluronic Acid-Based Hydrogel Design in Drug Delivery. Pharmaceutics 2019, 11, 407. [Google Scholar] [CrossRef]
- Catoira, M.C.; Fusaro, L.; Di Francesco, D.; Ramella, M.; Boccafoschi, F. Overview of Natural Hydrogels for Regenerative Medicine Applications. J. Mater. Sci.: Mater. Med. 2019, 30, 115. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Z.; Vizetto-Duarte, C.; Moay, Z.K.; Setyawati, M.I.; Rakshit, M.; Kathawala, M.H.; Ng, K.W. Composite Hydrogels in Three-Dimensional in Vitro Models. Front. Bioeng. Biotechnol. 2020, 8, 611. [Google Scholar] [CrossRef]
- Aurand, E.R.; Lampe, K.J.; Bjugstad, K.B. Defining and Designing Polymers and Hydrogels for Neural Tissue Engineering. Neurosci. Res. 2012, 72, 199–213. [Google Scholar] [CrossRef]
- Madduma-Bandarage, U.S.K.; Madihally, S.V. Synthetic Hydrogels: Synthesis, Novel Trends, and Applications. J. Appl. Polym. Sci. 2021, 138, 50376. [Google Scholar] [CrossRef]
- Ayatullah Hosne Asif, A.K.M.; Rahman, M.; Sarker, P.; Hasan, M.Z.; Paul, D. Hydrogel Fibre: Future Material of Interest for Biomedical Applications. J. Text. Sci. Technol. 2019, 5, 92–107. [Google Scholar] [CrossRef][Green Version]
- Xue, K.; Liu, Z.; Jiang, L.; Kai, D.; Li, Z.; Su, X.; Loh, X.J. A New Highly Transparent Injectable PHA-Based Thermogelling Vitreous Substitute. Biomater. Sci. 2020, 8, 926–936. [Google Scholar] [CrossRef]
- Fathi, M.; Barar, J.; Aghanejad, A.; Omidi, Y. Hydrogels for Ocular Drug Delivery and Tissue Engineering. Bioimpacts 2015, 5, 159–164. [Google Scholar] [CrossRef]
- Liu, X.-W.; Zhu, S.; Wu, S.-R.; Wang, P.; Han, G.-Z. Response Behavior of Ion-Sensitive Hydrogel Based on Crown Ether. Colloids Surf. A Physicochem. Eng. Asp. 2013, 417, 140–145. [Google Scholar] [CrossRef]
- Huang, H.; Qi, X.; Chen, Y.; Wu, Z. Thermo-Sensitive Hydrogels for Delivering Biotherapeutic Molecules: A Review. Saudi Pharm. J. 2019, 27, 990–999. [Google Scholar] [CrossRef] [PubMed]
- Rizwan, M.; Yahya, R.; Hassan, A.; Yar, M.; Azzahari, A.; Selvanathan, V.; Sonsudin, F.; Abouloula, C. PH Sensitive Hydrogels in Drug Delivery: Brief History, Properties, Swelling, and Release Mechanism, Material Selection and Applications. Polymers 2017, 9, 137. [Google Scholar] [CrossRef] [PubMed]
- Ofridam, F.; Tarhini, M.; Lebaz, N.; Gagnière, É.; Mangin, D.; Elaissari, A. PH-Sensitive Polymers: Classification and Some Fine Potential Applications. Polym. Adv. Technol. 2021, 32, 1455–1484. [Google Scholar] [CrossRef]
- Kubota, T.; Kurashina, Y.; Zhao, J.; Ando, K.; Onoe, H. Ultrasound-Triggered on-Demand Drug Delivery Using Hydrogel Microbeads with Release Enhancer. Mater. Des. 2021, 203, 109580. [Google Scholar] [CrossRef]
- Wang, S.; Chen, B.; Ouyang, L.; Wang, D.; Tan, J.; Qiao, Y.; Ge, S.; Ruan, J.; Zhuang, A.; Liu, X.; et al. A Novel Stimuli-Responsive Injectable Antibacterial Hydrogel to Achieve Synergetic Photothermal/Gene-Targeted Therapy towards Uveal Melanoma. Adv. Sci. 2021, 2004721. [Google Scholar] [CrossRef] [PubMed]
- Zeng, J.; Shi, D.; Gu, Y.; Kaneko, T.; Zhang, L.; Zhang, H.; Kaneko, D.; Chen, M. Injectable and Near-Infrared-Responsive Hydrogels Encapsulating Dopamine-Stabilized Gold Nanorods with Long Photothermal Activity Controlled for Tumor Therapy. Biomacromolecules 2019, 20, 3375–3384. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.P.; Lokhande, G.; Singh, K.A.; Jaiswal, M.K.; Rajput, S.; Gaharwar, A.K. Light-Triggered In Situ Gelation of Hydrogels Using 2D Molybdenum Disulfide (MoS2) Nanoassemblies as Crosslink Epicenter. Adv. Mater. 2021, 33, 2101238. [Google Scholar] [CrossRef]
- Liu, Z.; Liu, J.; Cui, X.; Wang, X.; Zhang, L.; Tang, P. Recent Advances on Magnetic Sensitive Hydrogels in Tissue Engineering. Front. Chem. 2020, 8, 124. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Zuo, X.; Li, S.; Sun, M.; Xie, H.; Zhang, K.; Zhou, J.; Che, L.; Ma, J.; Jia, Z.; et al. Synergistic Therapy of Magnetism-Responsive Hydrogel for Soft Tissue Injuries. Bioact. Mater. 2019, 4, 160–166. [Google Scholar] [CrossRef] [PubMed]
- Patel, H.; Panchal, D.R.; Patel, U.; Brahmbhatt, T.; Suthar, M. Matrix Type Drug Delivery System: A Review. J. Pharm. Sci. Biosci. Res. 2011, 1, 9. [Google Scholar]
- Wert, K.J.; Lin, J.H.; Tsang, S.H. General Pathophysiology in Retinal Degeneration. In Developments in Ophthalmology; Casaroli-Marano, R.P., Zarbin, M.A., Eds.; S. KARGER AG: Basel, Switzerland, 2014; Volume 53, pp. 33–43. ISBN 978-3-318-02584-2. [Google Scholar]
- Kador, K.E.; Goldberg, J.L. Scaffolds and Stem Cells: Delivery of Cell Transplants for Retinal Degenerations. Expert Rev. Ophthalmol. 2012, 7, 459–470. [Google Scholar] [CrossRef]
- Wang, Y.; Tang, Z.; Gu, P. Stem/Progenitor Cell-Based Transplantation for Retinal Degeneration: A Review of Clinical Trials. Cell Death Dis. 2020, 11, 793. [Google Scholar] [CrossRef]
- Tomita, M.; Lavik, E.; Klassen, H.; Zahir, T.; Langer, R.; Young, M.J. Biodegradable Polymer Composite Grafts Promote the Survival and Differentiation of Retinal Progenitor Cells. Stem Cells 2005, 23, 1579–1588. [Google Scholar] [CrossRef]
- Kim, H.S.; Kim, D.; Jeong, Y.W.; Choi, M.J.; Lee, G.W.; Thangavelu, M.; Song, J.E.; Khang, G. Engineering Retinal Pigment Epithelial Cells Regeneration for Transplantation in Regenerative Medicine Using PEG/Gellan Gum Hydrogels. Int. J. Biol. Macromol. 2019, 130, 220–228. [Google Scholar] [CrossRef]
- Mazumder, M.A.J.; Fitzpatrick, S.D.; Muirhead, B.; Sheardown, H. Cell-Adhesive Thermogelling PNIPAAm/Hyaluronic Acid Cell Delivery Hydrogels for Potential Application as Minimally Invasive Retinal Therapeutics. J. Biomed. Mater. Res. 2012, 100A, 1877–1887. [Google Scholar] [CrossRef] [PubMed]
- Park, J.; Baranov, P.; Aydin, A.; Abdelgawad, H.; Singh, D.; Niu, W.; Kurisawa, M.; Spector, M.; Young, M.J. In Situ Cross-Linking Hydrogel as a Vehicle for Retinal Progenitor Cell Transplantation. Cell Transplant. 2019, 28, 596–606. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.H. Injectable Hydrogels Delivering Therapeutic Agents for Disease Treatment and Tissue Engineering. Biomater. Res. 2018, 22, 27. [Google Scholar] [CrossRef] [PubMed]
- Cai, X.; Conley, S.M.; Naash, M.I. RPE65: Role in the Visual Cycle, Human Retinal Disease, and Gene Therapy. Ophthalmic Genet. 2009, 30, 57–62. [Google Scholar] [CrossRef]
- Liu, T.; Jenwitheesuk, E.; Teller, D.C.; Samudrala, R. Structural Insights into the Cellular Retinaldehyde-Binding Protein (CRALBP). Proteins 2005, 61, 412–422. [Google Scholar] [CrossRef]
- Choi, S. Encyclopedia of Signaling Molecules; Springer International Publishing: Cham, Germany, 2018; ISBN 978-3-319-67198-7. [Google Scholar]
- Wen, B.; Li, S.; Li, H.; Chen, Y.; Ma, X.; Wang, J.; Lu, F.; Qu, J.; Hou, L. Microphthalmia-Associated Transcription Factor Regulates the Visual Cycle Genes Rlbp1 and Rdh5 in the Retinal Pigment Epithelium. Sci. Rep. 2016, 6, 21208. [Google Scholar] [CrossRef]
- Campochiaro, P.A.; Jerdan, J.A.; Glaser, B.M. The Extracellular Matrix of Human Retinal Pigment Epithelial Cells In Vivo and Its Synthesis In Vitro. Investig. Ophthalmol. Vis. Sci. 1986, 27, 7. [Google Scholar]
- Lee, W.; Choi, J.H.; Lee, J.; Youn, J.; Kim, W.; Jeon, G.; Lee, S.W.; Song, J.E.; Khang, G. Dopamine-Functionalized Gellan Gum Hydrogel as a Candidate Biomaterial for a Retinal Pigment Epithelium Cell Delivery System. ACS Appl. Bio Mater. 2021, 4, 1771–1782. [Google Scholar] [CrossRef]
- Murphy, C.M.; O’Brien, F.J. Understanding the Effect of Mean Pore Size on Cell Activity in Collagen-Glycosaminoglycan Scaffolds. Cell Adhes. Migr. 2010, 4, 377–381. [Google Scholar] [CrossRef]
- Murphy, C.M.; Haugh, M.G.; O’Brien, F.J. The Effect of Mean Pore Size on Cell Attachment, Proliferation and Migration in Collagen–Glycosaminoglycan Scaffolds for Bone Tissue Engineering. Biomaterials 2010, 31, 461–466. [Google Scholar] [CrossRef] [PubMed]
- Park, H.; Guo, X.; Temenoff, J.S.; Tabata, Y.; Caplan, A.I.; Kasper, F.K.; Mikos, A.G. Effect of Swelling Ratio of Injectable Hydrogel Composites on Chondrogenic Differentiation of Encapsulated Rabbit Marrow Mesenchymal Stem Cells In Vitro. Biomacromolecules 2009, 10, 541–546. [Google Scholar] [CrossRef] [PubMed]
- Mallakpour, S.; Zadehnazari, A. New Organosoluble, Thermally Stable, and Nanostructured Poly(Amide-Imide)s with Dopamine Pendant Groups: Microwave-Assisted Synthesis and Characterization. Int. J. Polym. Anal. Charact. 2012, 17, 408–416. [Google Scholar] [CrossRef]
- Han, L.; Wang, M.; Li, P.; Gan, D.; Yan, L.; Xu, J.; Wang, K.; Fang, L.; Chan, C.W.; Zhang, H.; et al. Mussel-Inspired Tissue-Adhesive Hydrogel Based on the Polydopamine–Chondroitin Sulfate Complex for Growth-Factor-Free Cartilage Regeneration. ACS Appl. Mater. Interfaces 2018, 10, 28015–28026. [Google Scholar] [CrossRef] [PubMed]
- Jiang, F.; Tang, Z.; Zhang, Y.; Ju, Y.; Gao, H.; Sun, N.; Liu, F.; Gu, P.; Zhang, W. Enhanced Proliferation and Differentiation of Retinal Progenitor Cells through a Self-Healing Injectable Hydrogel. Biomater. Sci. 2019, 7, 2335–2347. [Google Scholar] [CrossRef]
- Zhang, D.; Shen, B.; Zhang, Y.; Ni, N.; Wang, Y.; Fan, X.; Sun, H.; Gu, P. Betacellulin Regulates the Proliferation and Differentiation of Retinal Progenitor Cells in Vitro. J. Cell. Mol. Med. 2018, 22, 330–345. [Google Scholar] [CrossRef] [PubMed]
- Ji, J.; Zhang, D.; Wei, W.; Shen, B.; Zhang, Y.; Wang, Y.; Tang, Z.; Ni, N.; Sun, H.; Liu, J.; et al. Decellularized Matrix of Adipose-Derived Mesenchymal Stromal Cells Enhanced Retinal Progenitor Cell Proliferation via the Akt/Erk Pathway and Neuronal Differentiation. Cytotherapy 2018, 20, 74–86. [Google Scholar] [CrossRef]
- Tang, Z.; Jiang, F.; Zhang, Y.; Zhang, Y.; Yang, Y.; Huang, X.; Wang, Y.; Zhang, D.; Ni, N.; Liu, F.; et al. Mussel-Inspired Injectable Hydrogel and Its Counterpart for Actuating Proliferation and Neuronal Differentiation of Retinal Progenitor Cells. Biomaterials 2019, 194, 57–72. [Google Scholar] [CrossRef]
- Colombe Dromel, P.; Singh, D.; Alexander-Katz, A.; Kurisawa, M.; Spector, M.; Young, M. Injectable Gelatin Hydroxyphenyl Propionic Acid Hydrogel Protects Human Retinal Progenitor Cells (HRPCs) from Shear Stress Applied during Small-Bore Needle Injection. Appl. Mater. Today 2020, 19, 100602. [Google Scholar] [CrossRef]
- Foster, W.J. Vitreous Substitutes. Expert Rev. Ophthalmol. 2008, 3, 211–218. [Google Scholar] [CrossRef]
- Alovisi, C.; Panico, C.; de Sanctis, U.; Eandi, C.M. Vitreous Substitutes: Old and New Materials in Vitreoretinal Surgery. J. Ophthalmol. 2017, 2017, 1–6. [Google Scholar] [CrossRef]
- Mateo-Montoya, A.; de Smet, M.D. Air as Tamponade for Retinal Detachments. Eur. J. Ophthalmol. 2014, 24, 242–246. [Google Scholar] [CrossRef]
- Kanclerz, P.; Grzybowski, A. Complications Associated with the Use of Expandable Gases in Vitrectomy. J. Ophthalmol. 2018, 2018, 8606494. [Google Scholar] [CrossRef]
- Yu, Q.; Liu, K.; Su, L.; Xia, X.; Xu, X. Perfluorocarbon Liquid: Its Application in Vitreoretinal Surgery and Related Ocular Inflammation. BioMed Res. Int. 2014, 2014, 250323. [Google Scholar] [CrossRef]
- Jiang, X.; Peng, Y.; Yang, C.; Liu, W.; Han, B. The Feasibility Study of an in Situ Marine Polysaccharide-Based Hydrogel as the Vitreous Substitute: In Situ Marine Polysaccharide-Based Hydrogel. J. Biomed. Mater. Res. 2018, 106, 1997–2006. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Chi, J.; Jiang, Z.; Hu, H.; Yang, C.; Liu, W.; Han, B. A Self-Healing and Injectable Hydrogel Based on Water-Soluble Chitosan and Hyaluronic Acid for Vitreous Substitute. Carbohydr. Polym. 2021, 256, 117519. [Google Scholar] [CrossRef]
- Uesugi, K.; Sakaguchi, H.; Hayashida, Y.; Hayashi, R.; Baba, K.; Suganuma, Y.; Yokoi, H.; Tsujikawa, M.; Nishida, K. A Self-Assembling Peptide Gel as a Vitreous Substitute: A Rabbit Study. Investig. Ophthalmol. Vis. Sci. 2017, 58, 4068. [Google Scholar] [CrossRef]
- Laradji, A.; Shui, Y.-B.; Karakocak, B.B.; Evans, L.; Hamilton, P.; Ravi, N. Bioinspired Thermosensitive Hydrogel as a Vitreous Substitute: Synthesis, Properties, and Progress of Animal Studies. Materials 2020, 13, 1337. [Google Scholar] [CrossRef]
- Liu, Z.; Liow, S.S.; Lai, S.L.; Alli-Shaik, A.; Holder, G.E.; Parikh, B.H.; Krishnakumar, S.; Li, Z.; Tan, M.J.; Gunaratne, J.; et al. Retinal-Detachment Repair and Vitreous-like-Body Reformation via a Thermogelling Polymer Endotamponade. Nat. Biomed. Eng. 2019, 3, 598–610. [Google Scholar] [CrossRef] [PubMed]
- Tram, N.K.; Maxwell, C.J.; Swindle-Reilly, K.E. Macro- and Microscale Properties of the Vitreous Humor to Inform Substitute Design and Intravitreal Biotransport. Curr. Eye Res. 2021, 46, 429–444. [Google Scholar] [CrossRef] [PubMed]
- Discher, D.E.; Smith, L.; Cho, S.; Colasurdo, M.; García, A.J.; Safran, S. Matrix Mechanosensing: From Scaling Concepts in ’Omics Data to Mechanisms in the Nucleus, Regeneration, and Cancer. Annu. Rev. Biophys. 2017, 46, 295–315. [Google Scholar] [CrossRef]
- Hytönen, V.P.; Wehrle-Haller, B. Mechanosensing in Cell–Matrix Adhesions–Converting Tension into Chemical Signals. Exp. Cell Res. 2016, 343, 35–41. [Google Scholar] [CrossRef] [PubMed]
- Cai, P.; Chen, X. Hydrogels for Artificial Vitreous: From Prolonged Substitution to Elicited Regeneration. ACS Mater. Lett. 2019, 1, 285–289. [Google Scholar] [CrossRef]
- Baker, A.E.G.; Cui, H.; Ballios, B.G.; Ing, S.; Yan, P.; Wolfer, J.; Wright, T.; Dang, M.; Gan, N.Y.; Cooke, M.J.; et al. Stable Oxime-Crosslinked Hyaluronan-Based Hydrogel as a Biomimetic Vitreous Substitute. Biomaterials 2021, 271, 120750. [Google Scholar] [CrossRef]
- Su, W.-Y.; Chen, K.-H.; Chen, Y.-C.; Lee, Y.-H.; Tseng, C.-L.; Lin, F.-H. An Injectable Oxidated Hyaluronic Acid/Adipic Acid Dihydrazide Hydrogel as a Vitreous Substitute. J. Biomater. Sci. Polym. Ed. 2011, 22, 1777–1797. [Google Scholar] [CrossRef]
- Barth, H.; Crafoord, S.; Andréasson, S.; Ghosh, F. A Cross-Linked Hyaluronic Acid Hydrogel (Healaflow®) as a Novel Vitreous Substitute. Graefe’s Arch. Clin. Exp. Ophthalmol. 2016, 254, 697–703. [Google Scholar] [CrossRef]
- Malkoch, M.; Vestberg, R.; Gupta, N.; Mespouille, L.; Dubois, P.; Mason, A.F.; Hedrick, J.L.; Liao, Q.; Frank, C.W.; Kingsbury, K.; et al. Synthesis of Well-Defined Hydrogel Networks Using Click Chemistry. Chem. Commun. 2006, 2774–2776. [Google Scholar] [CrossRef]
- Murali, K.; Kang, D.; Nazari, H.; Scianmarello, N.; Cadenas, E.; Tai, Y.-C.; Kashani, A.; Humayun, M. Spatial Variations in Vitreous Oxygen Consumption. PLoS ONE 2016, 11, e0149961. [Google Scholar] [CrossRef] [PubMed]
- Siegfried, C.J.; Shui, Y.-B. Intraocular Oxygen and Antioxidant Status: New Insights on the Effect of Vitrectomy and Glaucoma Pathogenesis. Am. J. Ophthalmol. 2019, 203, 12–25. [Google Scholar] [CrossRef] [PubMed]
- Holekamp, N.M.; Shui, Y.-B.; Beebe, D.C. Vitrectomy Surgery Increases Oxygen Exposure to the Lens: A Possible Mechanism for Nuclear Cataract Formation. Am. J. Ophthalmol. 2005, 139, 302–310. [Google Scholar] [CrossRef] [PubMed]
- Tram, N.K.; Jiang, P.; Torres-Flores, T.C.; Jacobs, K.M.; Chandler, H.L.; Swindle-Reilly, K.E. A Hydrogel Vitreous Substitute That Releases Antioxidant. Macromol. Biosci. 2020, 20, 1900305. [Google Scholar] [CrossRef]
- Tram, N.K.; McLean, R.M.; Swindle-Reilly, K.E. Glutathione Improves the Antioxidant Activity of Vitamin C in Human Lens and Retinal Epithelial Cells: Implications for Vitreous Substitutes. Curr. Eye Res. 2021, 46, 470–481. [Google Scholar] [CrossRef]
- Ankamah, E.; Sebag, J.; Ng, E.; Nolan, J.M. Vitreous Antioxidants, Degeneration, and Vitreo-Retinopathy: Exploring the Links. Antioxidants 2019, 9, 7. [Google Scholar] [CrossRef] [PubMed]
- Masket, S.; Hovanesian, J.A.; Levenson, J.; Tyson, F.; Flynn, W.; Endl, M.; Majmudar, P.A.; Modi, S.; Chu, R.; Raizman, M.B.; et al. Hydrogel Sealant versus Sutures to Prevent Fluid Egress after Cataract Surgery. J. Cataract Refract. Surg. 2014, 40, 2057–2066. [Google Scholar] [CrossRef] [PubMed]
- Ciapetti, G.; Cenni, E.; Marram, D.; Toni, A.; Pizzoferrato, A. Cytotoxicity Testing of Cyanoacrylates Using Direct Contact Assay on Cell Cultures. Biomaterials 1994, 15, 5. [Google Scholar] [CrossRef]
- Chen, W.-L.; Lin, C.-T.; Hsieh, C.-Y.; Tu, I.-H.; Chen, W.Y.W.; Hu, F.-R. Comparison of the Bacteriostatic Effects, Corneal Cytotoxicity, and the Ability to Seal Corneal Incisions Among Three Different Tissue Adhesives. Cornea 2007, 26, 1228–1234. [Google Scholar] [CrossRef] [PubMed]
- Panda, A.; Kumar, S.; Kumar, A.; Bansal, R.; Bhartiya, S. Fibrin Glue in Ophthalmology. Indian J. Ophthalmol. 2009, 57, 9. [Google Scholar] [CrossRef]
- Oelker, A.M.; Grinstaff, M.W. Ophthalmic Adhesives: A Materials Chemistry Perspective. J. Mater. Chem. 2008, 18, 2521. [Google Scholar] [CrossRef]
- Grinstaff, M.W. Designing Hydrogel Adhesives for Corneal Wound Repair. Biomaterials 2007, 28, 5205–5214. [Google Scholar] [CrossRef]
- Chenault, H.K.; Bhatia, S.K.; DiMaio, W.G.; Vincent, G.L.; Camacho, W.; Behrens, A. Sealing and Healing of Clear Corneal Incisions with an Improved Dextran Aldehyde-PEG Amine Tissue Adhesive. Curr. Eye Res. 2011, 36, 997–1004. [Google Scholar] [CrossRef]
- Oelker, A.M.; Berlin, J.A.; Wathier, M.; Grinstaff, M.W. Synthesis and Characterization of Dendron Cross-Linked PEG Hydrogels as Corneal Adhesives. Biomacromolecules 2011, 12, 1658–1665. [Google Scholar] [CrossRef] [PubMed]
- Chae, J.J.; Mulreany, D.G.; Guo, Q.; Lu, Q.; Choi, J.S.; Strehin, I.; Espinoza, F.A.; Schein, O.; Trexler, M.M.; Bower, K.S.; et al. Application of a Collagen-Based Membrane and Chondroitin Sulfate-Based Hydrogel Adhesive for the Potential Repair of Severe Ocular Surface Injuries. Mil. Med. 2014, 179, 686–694. [Google Scholar] [CrossRef] [PubMed]
- Calladine, D.; Ward, M.; Packard, R. Adherent Ocular Bandage for Clear Corneal Incisions Used in Cataract Surgery. J. Cataract Refract. Surg. 2010, 36, 1839–1848. [Google Scholar] [CrossRef] [PubMed]
- Masket, S.; Hovanesian, J.; Raizman, M.; Wee, D.; Fram, N. Use of a Calibrated Force Gauge in Clear Corneal Cataract Surgery to Quantify Point-Pressure Manipulation. J. Cataract Refract. Surg. 2013, 39, 511–518. [Google Scholar] [CrossRef]
- Chee, S.-P. Clear Corneal Incision Leakage after Phacoemulsification—Detection Using Povidone Iodine 5%. Int. Ophthalmol. 2007, 26, 175–179. [Google Scholar] [CrossRef] [PubMed]
- Nallasamy, N.; Grove, K.E.; Legault, G.L.; Daluvoy, M.B.; Kim, T. Hydrogel Ocular Sealant for Clear Corneal Incisions in Cataract Surgery. J. Cataract Refract. Surg. 2017, 43, 1010–1014. [Google Scholar] [CrossRef]
- Shehata, A.E.M.; Kambhampati, S.P.; Wang, J.; Soiberman, U.S. Burst Pressures in Eyes with Clear Corneal Incisions Treated with ReSure Glue. J. Ophthalmol. 2021, 2021, 6691489. [Google Scholar] [CrossRef]
- Fredell, M.; Hamill, M. The Use of Polyethylene Glycol Hydrogel Tissue Adhesive for Corneal Incision Closure Following Descemet Membrane Endothelial Keratoplasty (DMEK). Eye (London) 2019, 33, 858–859. [Google Scholar] [CrossRef] [PubMed]
- Potvin, R.; Matossian, C.; Makari, S. Cataract Surgery and Methods of Wound Closure: A Review. Clin. Ophthalmol. 2015, 9, 921–928. [Google Scholar] [CrossRef]
- Uy, H.S.; Kenyon, K.R. Surgical Outcomes after Application of a Liquid Adhesive Ocular Bandage to Clear Corneal Incisions during Cataract Surgery. J. Cataract Refract. Surg. 2013, 39, 1668–1674. [Google Scholar] [CrossRef]
- Kenyon, K.R.; Qiao, L.; Lee, E. Hydrogel Liquid Ocular Bandage (OcuSeal®) Is an Effective Microbial Barrier. Investig. Ophthalmol. Vis. Sci. 2014, 55, 2547. [Google Scholar]
- Kambhampati, S.P.P.; Lin, H.P.; Sharma, R.; Saragnese, D.; Cleland, J.L.; Yiu, S.C.; Rangaramanujam, K. OcuPairTM: A Novel Dendrimer-Hyaluronic Acid Hydrogel Sealant for Temporary Corneal Wound Stabilization. Investig. Ophthalmol. Vis. Sci. 2020, 61, 369. [Google Scholar]
- Rose, J.; Pacelli, S.; Haj, A.; Dua, H.; Hopkinson, A.; White, L.; Rose, F. Gelatin-Based Materials in Ocular Tissue Engineering. Materials 2014, 7, 3106–3135. [Google Scholar] [CrossRef]
- Shirzaei Sani, E.; Kheirkhah, A.; Rana, D.; Sun, Z.; Foulsham, W.; Sheikhi, A.; Khademhosseini, A.; Dana, R.; Annabi, N. Sutureless Repair of Corneal Injuries Using Naturally Derived Bioadhesive Hydrogels. Sci. Adv. 2019, 5, eaav1281. [Google Scholar] [CrossRef]
- Noshadi, I.; Hong, S.; Sullivan, K.E.; Shirzaei Sani, E.; Portillo-Lara, R.; Tamayol, A.; Shin, S.R.; Gao, A.E.; Stoppel, W.L.; Black III, L.D.; et al. In Vitro and in Vivo Analysis of Visible Light Crosslinkable Gelatin Methacryloyl (GelMA) Hydrogels. Biomater. Sci. 2017, 5, 2093–2105. [Google Scholar] [CrossRef]
- Bahney, C.; Lujan, T.; Hsu, C.; Bottlang, M.; West, J.; Johnstone, B. Visible Light Photoinitiation of Mesenchymal Stem Cell-Laden Bioresponsive Hydrogels. Eur. Cells Mater. 2011, 22, 43–55. [Google Scholar] [CrossRef] [PubMed]
- Annabi, N.; Yue, K.; Tamayol, A.; Khademhosseini, A. Elastic Sealants for Surgical Applications. Eur. J. Pharm. Biopharm. 2015, 95, 27–39. [Google Scholar] [CrossRef] [PubMed]
- Hamba, N.; Gerbi, A.; Tesfaye, S. Histopathological Effects of Ultraviolet Radiation Exposure on the Ocular Structures in Animal Studies –Literature Review. Transl. Res. Anat. 2021, 22, 100086. [Google Scholar] [CrossRef]
- Behar-Cohen, F.; Baillet, G.; de Ayguavives, T.; Ortega Garcia, P.; Krutmann, J.; Peña-García, P.; Reme, C.; Wolffsohn, J.S. Ultraviolet Damage to the Eye Revisited: Eye-Sun Protection Factor (E-SPF®), a New Ultraviolet Protection Label for Eyewear. Clin. Ophthalmol. 2013, 8, 87. [Google Scholar] [CrossRef] [PubMed]
- Khalil, I.A.; Saleh, B.; Ibrahim, D.M.; Jumelle, C.; Yung, A.; Dana, R.; Annabi, N. Ciprofloxacin-Loaded Bioadhesive Hydrogels for Ocular Applications. Biomater. Sci. 2020, 8, 5196–5209. [Google Scholar] [CrossRef] [PubMed]
- Sharifi, S.; Islam, M.M.; Sharifi, H.; Islam, R.; Koza, D.; Reyes-Ortega, F.; Alba-Molina, D.; Nilsson, P.H.; Dohlman, C.H.; Mollnes, T.E.; et al. Tuning Gelatin-Based Hydrogel towards Bioadhesive Ocular Tissue Engineering Applications. Bioact. Mater. 2021, 6, 3947–3961. [Google Scholar] [CrossRef] [PubMed]
- Maddula, S.; Davis, D.K.; Ness, P.J.; Burrow, M.K.; Olson, R.J. Comparison of Wound Strength with and without a Hydrogel Liquid Ocular Bandage in Human Cadaver Eyes. J. Cataract Refract. Surg. 2010, 36, 1775–1778. [Google Scholar] [CrossRef] [PubMed]
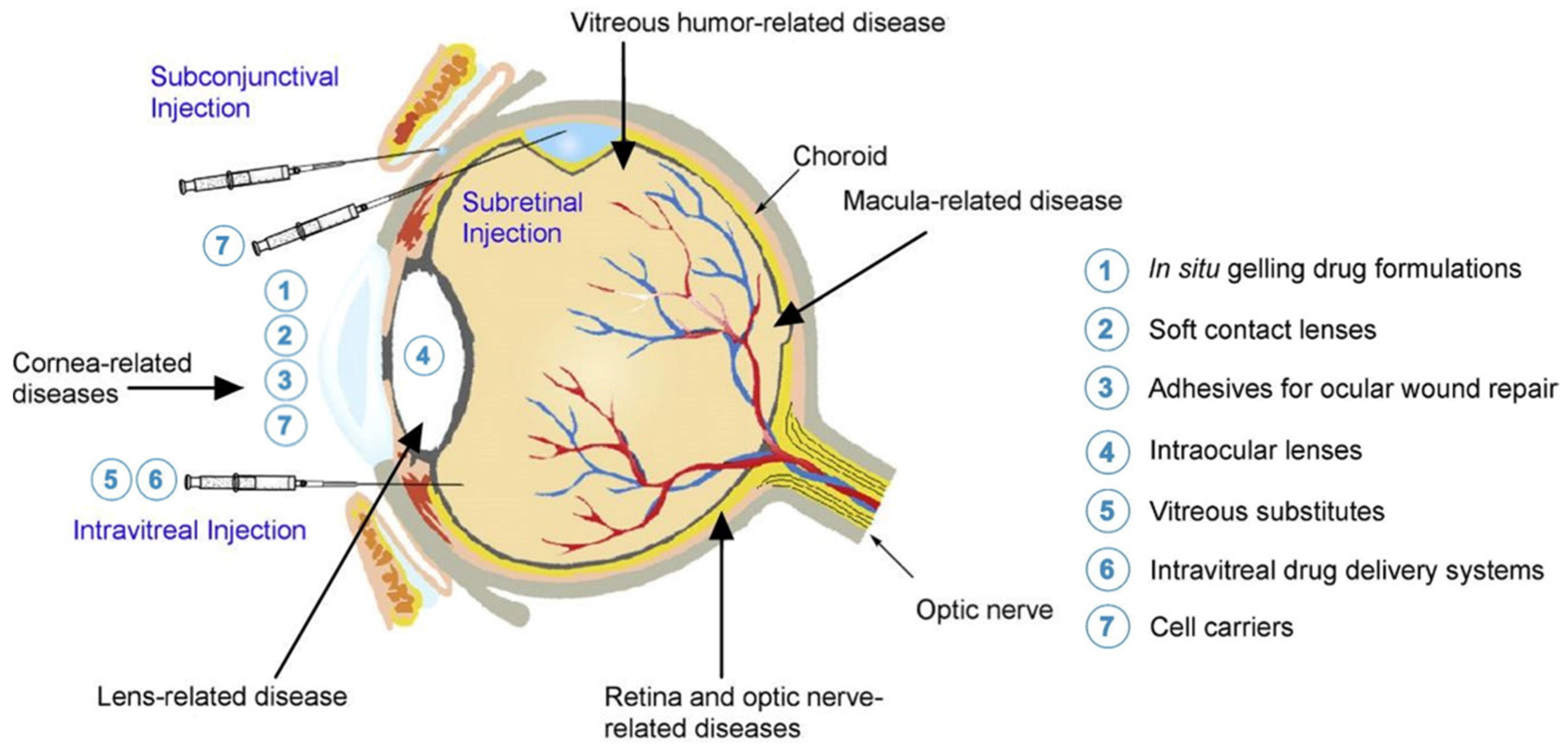
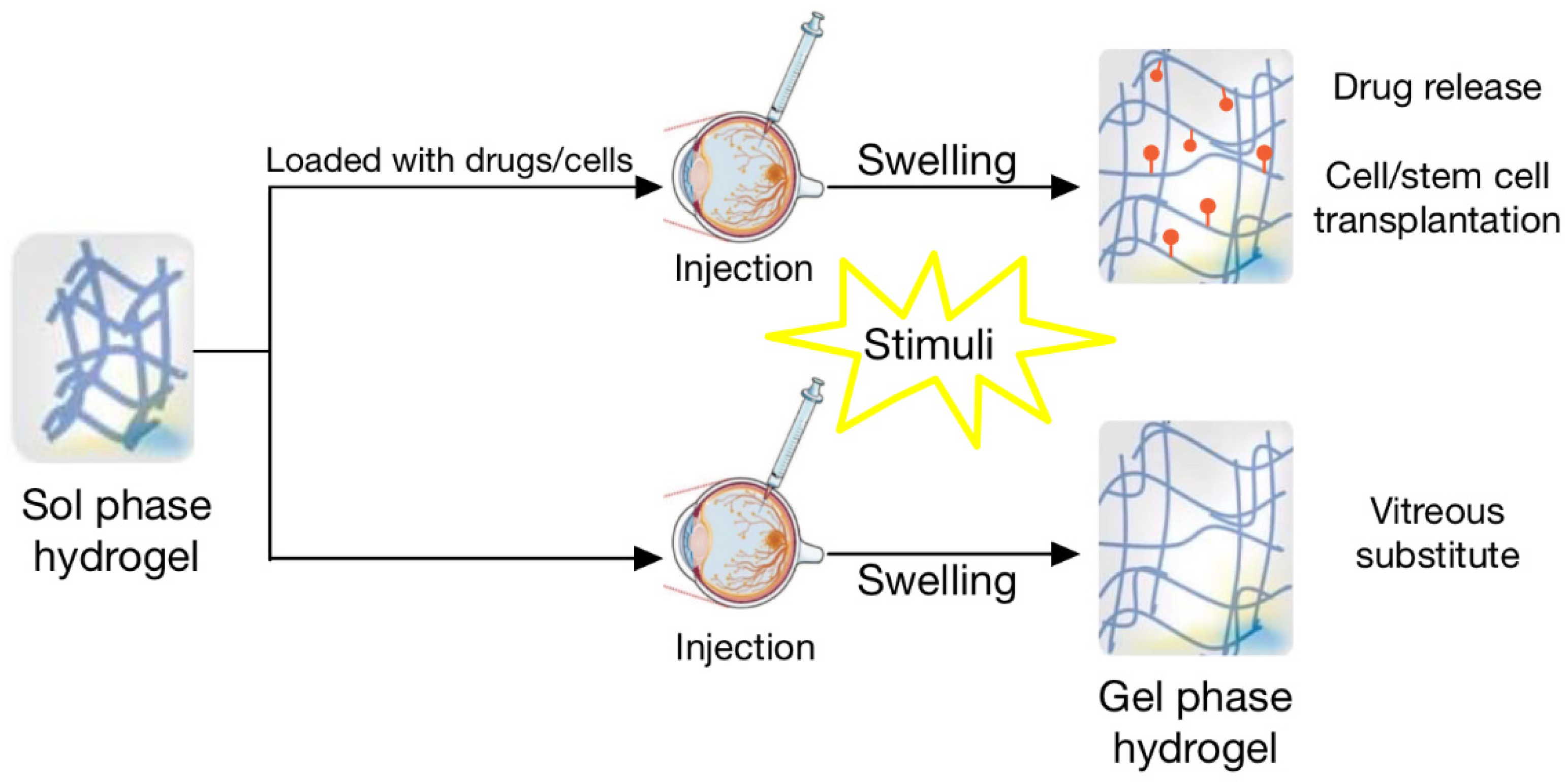
| Type of Hydrogel | Polymer(s) | Retinal Cell Type | In Vitro Cytocompatibility | In Vivo Biocompatibility | Notes |
|---|---|---|---|---|---|
| PEG/GG [64] | Polyethylene glycol and gellan gum | RPE | Yes—Live/Dead assay, MTT analysis, and gene expression analysis | Not conducted | None |
| Ge/GG/Cs [38] | Gelatin, gellan gum, and glycol chitosan | RPE | Yes—Live/Dead staining and gene expression analysis | Not conducted | Inclusion of Cs lead to higher proliferation rates |
| DFG [73] | Gellan gum grafted with dopamine | RPE | Yes—Live/Dead staining and gene expression analysis | Not conducted | Dopamine confers superior hydrogel injectability and a favorable microenvironment |
| CS-Odex [79] | Chitosan hydrochloride and oxidized dextran | RPC | Yes—Live/Dead assay and inflammatory and apoptotic factor expression analysis | Yes—H&E staining and Masson’s trichrome staining | Hydrogel is able to self-heal. CS increases preferential differentiation towards retinal neurons. |
| Gtn-HPA [66] | Gelatin-hydroxyphenyl propionic acid | RPC | Yes—Live/Dead assay | Yes—immunohistochemistry and anti-leukocyte staining | Gtn-HPA lowers proliferative potential and transplants show persistent retinal detachment |
| gel-HA [82] | Thiolated gelatin and methacrylated hyaluronic acid | RPC | Yes—Live/dead staining, inflammatory and apoptotic factor expression levels, and cell adhesion analysis | Not conducted | Improved cell proliferation |
| gel-HA-PDA [82] | Thiolated gelatin, methacrylated hyaluronic acid, and polydopamine | RPC | Yes—Live/dead staining, inflammatory and apoptotic factor expression levels, and cell adhesion analysis | Yes—H&E staining and Masson’s trichrome staining | Improved neuronal differentiation and cell migration and adhesion |
| Type of Hydrogel | Polymers | In Vitro Cytocompatibility | In Vivo Biocompatibility | Notes |
|---|---|---|---|---|
| HPCTS-ADA [89] | Hydroxypropyl chitosan and alginate dialdehyde | Yes—MTT assay | No—electroretinogram and histopathologic anaylsis | None |
| CMCTS-OHA [90] | Oxidized hyaluronic acid and carboxymethyl chitosan | Yes—MTT assay | Yes—H&E staining | Self-healing properties |
| PanaceaGel SPG-178 [91] | 13 amino acid peptide (RLDLRLALRLDLR) | Yes—Live/Dead staining | Yes—slit lamp examination, fundoscopy, electroretinography, histopathology | Self-assembling properties prevent damage from injection |
| poly(MAM-co-MAA-co-BMAC [92] | Thiolated gellan and poly(methacrylamide-co-methac -rylate-co-bis(methylacryloyl-cystamine)) | Yes—ECIS and CellTiter-Glo Luminescent Cell Viability end-point assay | Yes—electroretinography, optical coherence tomography, and H&E staining | Thermoresponsive |
| PHxEP [47] | Poly[(R)-3-hydroxybutyrate-(R)-3-hydroxyhexanoate], polyethylene glycol, and polypropylene glycol | Yes—MTT assay | Yes—histopathological examination | Thermoresponsive |
| EPC [93] | Poly(ε-caprolactone), polyethylene glycol, and polypropylene glycol | Not conducted | Yes—slit-lamp examinations, fundus evaluation, electroretinography, H&E staining | Degrades and regenerates a vitreous-like body |
| HA-oxime [98] | Hyaluronan modified with aldehyde or ketone and PEG-tetraoxyamine | Yes—Live/Dead assay | Yes—H&E examination | Click chemistry cross-linking system obviates the need for cross-linking agents |
| PEGDA [105] | Poly(ethylene glycol) diacrylate | Yes—CellTiter-Glo luminescent cell viability assay | Not conducted | Can be loaded with vitamin C to protect against oxidative damage |
| PEGDA-co-PEGMA [105] | Poly(ethylene glycol) methacrylate and poly(ethylene glycol) diacrylate | Yes—CellTiter-Glo luminescent cell viability assay | Not conducted | Can be loaded with vitamin C to protect against oxidative damage |
| Type of Hydrogel | Polymer | Corneal Incision Length (mm) | Burst Pressure (kPa) |
|---|---|---|---|
| ReSure® Sealant [121] | Polyethylene glycol | <3.5 | 12.4 |
| OcuSeal™ [136] | Poly(glycerol succinic acid) and PEG-aldehyde | <2.8 | 26.4 |
| OcuPairTM [126] | Methacrylated hydroxyl dendrimer and methacrylated hyaluronic acid | <6 | 9.3 |
| GelCORE [128] | Methacrylated gelatin | <3 | 30.1 |
| GELGYM [135] | Glicydlmethacrylated Gelatin | <4 | 26.7 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lin, K.T.; Wang, A.; Nguyen, A.B.; Iyer, J.; Tran, S.D. Recent Advances in Hydrogels: Ophthalmic Applications in Cell Delivery, Vitreous Substitutes, and Ocular Adhesives. Biomedicines 2021, 9, 1203. https://doi.org/10.3390/biomedicines9091203
Lin KT, Wang A, Nguyen AB, Iyer J, Tran SD. Recent Advances in Hydrogels: Ophthalmic Applications in Cell Delivery, Vitreous Substitutes, and Ocular Adhesives. Biomedicines. 2021; 9(9):1203. https://doi.org/10.3390/biomedicines9091203
Chicago/Turabian StyleLin, Kenny T., Athena Wang, Alexandra B. Nguyen, Janaki Iyer, and Simon D. Tran. 2021. "Recent Advances in Hydrogels: Ophthalmic Applications in Cell Delivery, Vitreous Substitutes, and Ocular Adhesives" Biomedicines 9, no. 9: 1203. https://doi.org/10.3390/biomedicines9091203
APA StyleLin, K. T., Wang, A., Nguyen, A. B., Iyer, J., & Tran, S. D. (2021). Recent Advances in Hydrogels: Ophthalmic Applications in Cell Delivery, Vitreous Substitutes, and Ocular Adhesives. Biomedicines, 9(9), 1203. https://doi.org/10.3390/biomedicines9091203








