The Azurin-Derived Peptide CT-p19LC Exhibits Membrane-Active Properties and Induces Cancer Cell Death
Abstract
:1. Introduction
2. Materials and Methods
2.1. Azurin-Derived Peptides
2.2. Circular Dichroism Spectroscopy
2.3. Human Cancer Cell Lines and Cell Culture Conditions
2.4. MTT Cell Proliferation Assays
2.5. LDH Release Assays
2.6. Quantitative Cellular Interaction
2.7. CT-p19LC Cellular Uptake
2.8. Apoptosis Assay
2.9. Zeta Potential Measurements of Live A549, MCF-7, HeLa and HT-29 Cancer Cells and 16HBE14o- and MCF10A Non-Cancer Cells in the Presence of CT-p19LC
2.10. GP Determination for Membrane Order Evaluation
2.11. Statistical Analysis
3. Results and Discussion
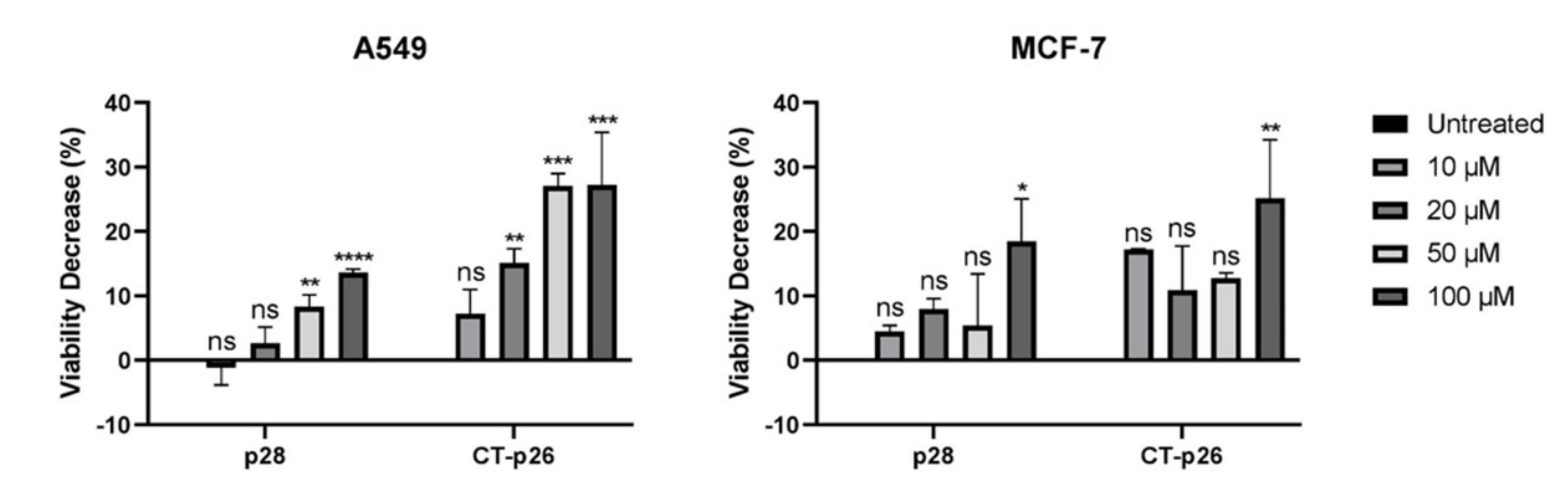
3.1. CT-p26 Peptide Effect on Cell Viability Confirms the Anticancer Potential of C-Terminal Azurin
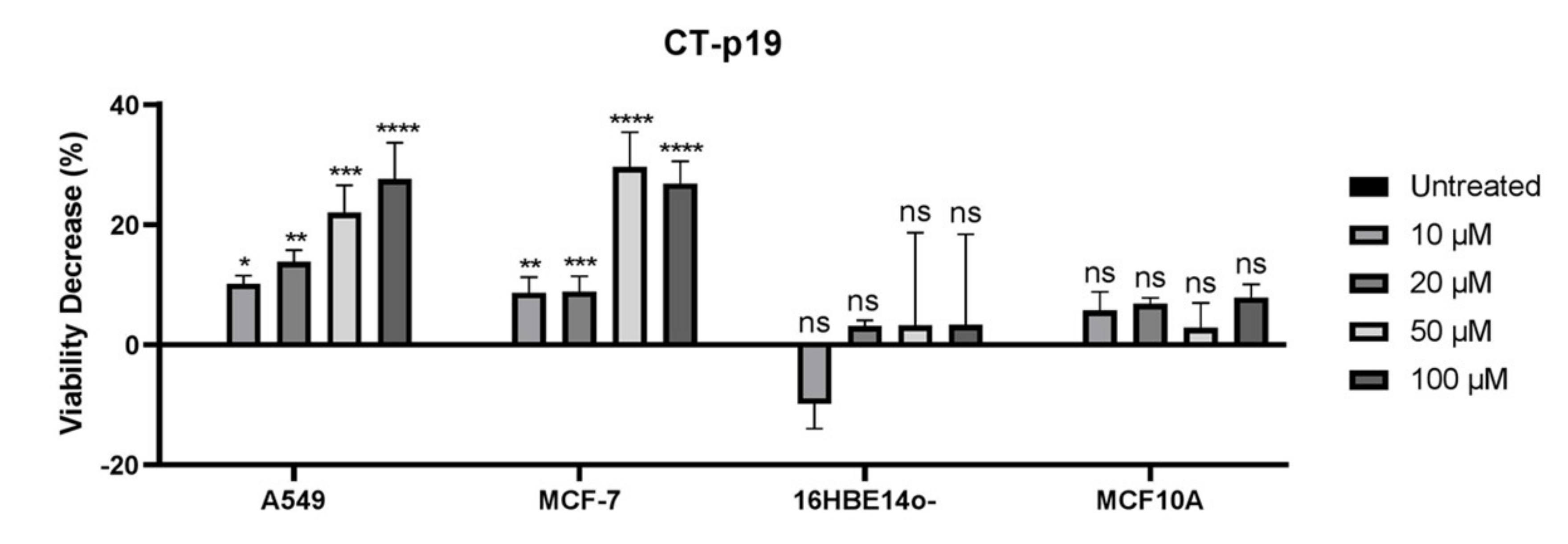
3.2. CT-p19 Peptide Decreases Cancer Cell Viability and Has Selective Property
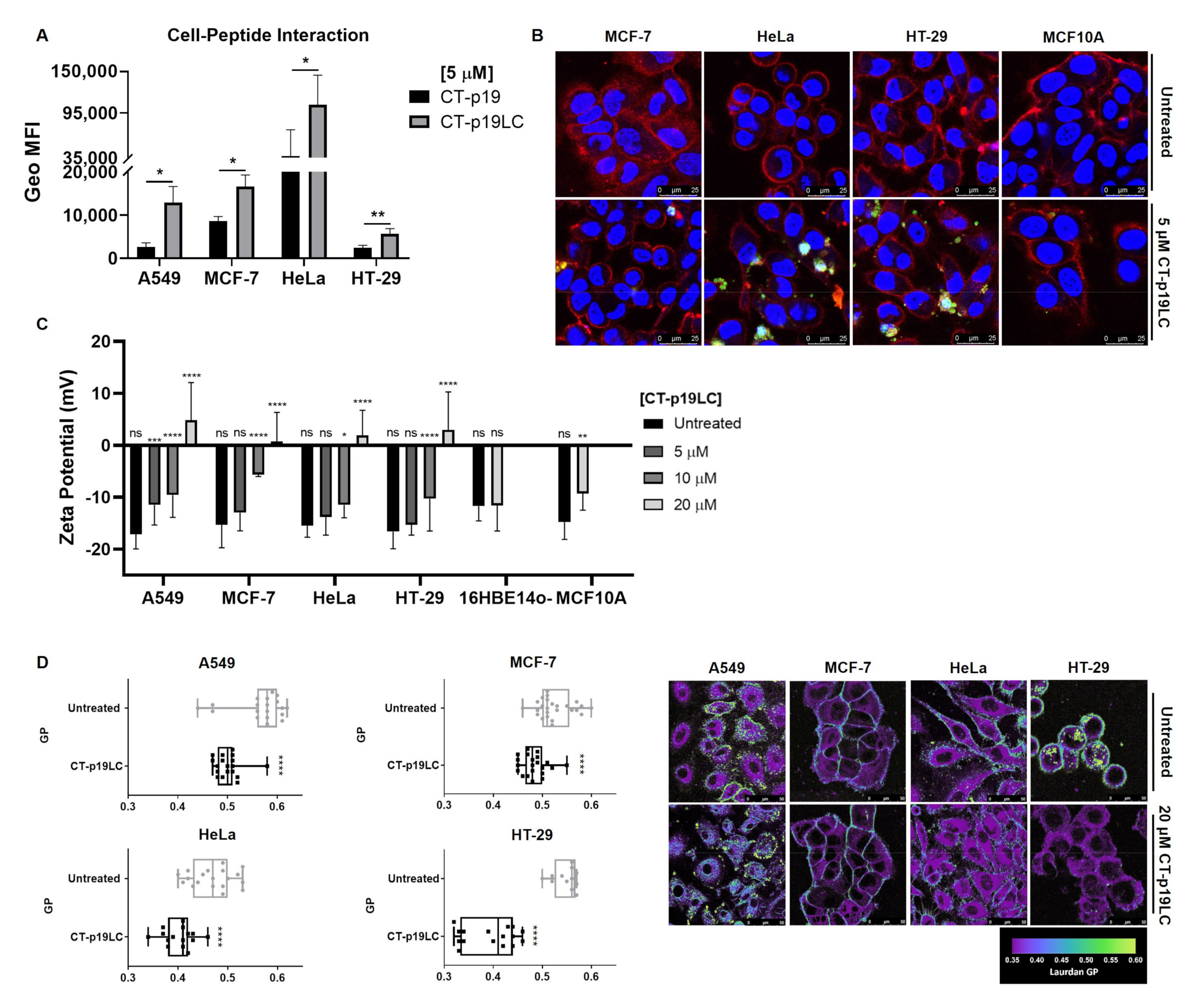
3.3. The Newly Designed CT-p19LC Peptide Reduces Proliferation and Induces Cell Death in Cancer Cell Lines
3.4. CT-p19LC Peptide Targets Cellular Plasma Membrane
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Gabernet, G.; Müller, A.T.; Hiss, J.A.; Schneider, G. Membranolytic anticancer peptides. Med. Chem. Commun. 2016, 7, 1–14. [Google Scholar] [CrossRef]
- Hoskin, D.W.; Ramamoorthy, A. Studies on Anticancer Activities of Antimicrobial Peptides. Biochim. Biophys. Acta—Biomembr. 2008, 1778, 357–375. [Google Scholar] [CrossRef] [Green Version]
- Harris, F.; Dennison, S.R.; Singh, J.; Phoenix, D.A. On the Selectivity and Efficacy of Defense Peptides with Respect to Cancer Cells. Med. Res. Rev. 2013, 33, 190–234. [Google Scholar] [CrossRef]
- Gaspar, D.; Veiga, A.S.; Castanho, M.A.R.B. From Antimicrobial to Anticancer Peptides. A Review. Front. Microbiol. 2013, 4, 1–16. [Google Scholar] [CrossRef] [Green Version]
- Leuschner, C.; Hansel, W. Targeting Breast and Prostate Cancers Through Their Hormone Receptors. Biol. Reprod. 2005, 73, 860–865. [Google Scholar] [CrossRef] [Green Version]
- Bernardes, N.; Fialho, A.M. Perturbing the Dynamics and Organization of Cell Membrane Components: A New Paradigm for Cancer-Targeted Therapies. Int. J. Mol. Sci. 2018, 19, 3871. [Google Scholar] [CrossRef] [Green Version]
- Ehrenstein, G.; Lecar, H. Electrically gated ionic channels in lipid bilayers. Q. Rev. Biophys. 1977, 10, 1–34. [Google Scholar] [CrossRef] [PubMed]
- Hilchie, A.L.; Hoskin, D.W. Application of cationic antimicrobial peptides in cancer treatment: Laboratory investigations and clinical potential. In Proceedings of the Emerging Cancer Therapy: Microbial Approaches and Biotechnological Tools; Wiley: Hoboken, NJ, USA, 2010; pp. 309–332. [Google Scholar]
- Chiangjong, W.; Chutipongtanate, S.; Hongeng, S. Anticancer peptide: Physicochemical property, functional aspect and trend in clinical application (Review). Int. J. Oncol. 2020, 57, 678–696. [Google Scholar] [CrossRef] [PubMed]
- Teixeira, V.; Feio, M.J.; Bastos, M. Role of Lipids in the Interaction of Antimicrobial Peptides with Membranes. Prog. Lipid Res. 2012, 51, 149–177. [Google Scholar] [CrossRef] [PubMed]
- Hetz, C.; Bono, M.R.; Barros, L.F.; Lagos, R. Microcin E492, a Channel-Forming Bacteriocin from Klebsiella Pneumoniae, Induces Apoptosis in Some Human Cell Lines. Proc. Natl. Acad. Sci. USA 2002, 99, 2696–2701. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xie, M.; Liu, D.; Yang, Y. Anti-cancer peptides: Classification, mechanism of action, reconstruction and modification. Biol. Open 2020, 10, 200004. [Google Scholar] [CrossRef]
- Hilchie, A.L.; Hoskin, D.W.; Power Coombs, M.R. Anticancer Activities of Natural and Synthetic Peptides. Adv. Exp. Med. Biol. 2019, 1117, 131–147. [Google Scholar] [CrossRef] [PubMed]
- Pubmed, Keyword: Anticancer Peptides in Clinical Trials. 2021. Available online: https://pubmed.ncbi.nlm.nih.gov/?term=anticancer+peptide&filter=pubt.clinicaltrial&filter=years.2000-2021&sort=date (accessed on 13 March 2021).
- Haug, B.E.; Camilio, K.A.; Eliassen, L.T.; Stensen, W.; Svendsen, J.S.; Berg, K.; Mortensen, B.; Serin, G.; Mirjolet, J.-F.; Bichat, F.; et al. Discovery of a 9-Mer Cationic Peptide (LTX-315) as a Potential First in Class Oncolytic Peptide. J. Med. Chem. 2016, 59, 2918–2927. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wali, A.F.; Majid, S.; Rasool, S.; Shehada, S.B.; Abdulkareem, S.K.; Firdous, A.; Beigh, S.; Shakeel, S.; Mushtaq, S.; Akbar, I.; et al. Natural products against cancer: Review on phytochemicals from marine sources in preventing cancer. Saudi Pharm. J. 2019, 27, 767–777. [Google Scholar] [CrossRef] [PubMed]
- Yamada, T.; Christov, K.; Shilkaitis, A.; Bratescu, L.; Green, A.; Santini, S.; Bizzarri, A.R.; Cannistraro, S.; Gupta, T.K.D.; Beattie, C.W. P28, A First in Class Peptide Inhibitor of Cop1 Binding to P53. Br. J. Cancer 2013, 108, 2495–2504. [Google Scholar] [CrossRef] [Green Version]
- Lulla, R.R.; Goldman, S.; Yamada, T.; Beattie, C.W.; Bressler, L.; Pacini, M.; Pollack, I.F.; Fisher, P.G.; Packer, R.J.; Dunkel, I.J.; et al. Phase I Trial of P28 (NSC745104), a Non-HDM2-Mediated Peptide Inhibitor of P53 Ubiquitination in Pediatric Patients with Recurrent or Progressive Central Nervous System Tumors: A Pediatric Brain Tumor Consortium Study. Neuro. Oncol. 2016, 18, 1319–1325. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Warso, M.A.; Richards, J.M.; Mehta, D.; Christov, K.; Schaeffer, C.; Rae Bressler, L.; Yamada, T.; Majumdar, D.; Kennedy, S.A.; Beattie, C.W.; et al. A First-in-Class, First-in-Human, Phase I Trial of P28, a Non-HDM2-Mediated Peptide Inhibitor of P53 Ubiquitination in Patients with Advanced Solid Tumours. Br. J. Cancer 2013, 108, 1061–1070. [Google Scholar] [CrossRef]
- Chakrabarty, A.M.; Bernardes, N.; Fialho, A.M. Bacterial Proteins and Peptides in Cancer Therapy: Today and Tomorrow. Bioengineered 2014, 5, 234–242. [Google Scholar] [CrossRef] [Green Version]
- Yamada, T.; Das Gupta, T.K.; Beattie, C.W. P28-Mediated Activation of P53 in G2-M Phase of the Cell Cycle Enhances the Efficacy of DNA Damaging and Antimitotic Chemotherapy. Cancer Res. 2016, 76, 2354–2365. [Google Scholar] [CrossRef] [Green Version]
- Bernardes, N.; Ribeiro, A.S.; Abreu, S.; Vieira, A.F.; Carreto, L.; Santos, M.; Seruca, R.; Paredes, J.; Fialho, A.M. High-Throughput Molecular Profiling of a P-Cadherin Overexpressing Breast Cancer Model Reveals New Targets for the Anti-Cancer Bacterial Protein Azurin. Int. J. Biochem. Cell Biol. 2014, 50, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Bernardes, N.; Ribeiro, A.S.; Abreu, S.; Mota, B.; Matos, R.G.; Arraiano, C.M.; Seruca, R.; Paredes, J.; Fialho, A.M. The bacterial protein azurin impairs invasion and FAK/Src signaling in P-cadherin-overexpressing breast cancer cell models. PLoS ONE 2013, 8, e69023. [Google Scholar] [CrossRef] [Green Version]
- Mehta, R.R.; Yamada, T.; Taylor, B.N.; Christov, K.; King, M.L.; Majumdar, D.; Lekmine, F.; Tiruppathi, C.; Shilkaitis, A.; Bratescu, L.; et al. A Cell Penetrating Peptide Derived from Azurin Inhibits Angiogenesis and Tumor Growth by Inhibiting Phosphorylation of VEGFR-2, FAK Akt. Angiogenesis 2011, 14, 355–369. [Google Scholar] [CrossRef] [PubMed]
- Yamada, T.; Mehta, R.R.; Lekmine, F.; Christov, K.; King, M.L.; Majumdar, D.; Shilkaitis, A.; Green, A.; Bratescu, L.; Beattie, C.W.; et al. A Peptide Fragment of Azurin Induces a P53-Mediated Cell Cycle Arrest in Human Breast Cancer Cells. Mol. Cancer Ther. 2009, 8, 2947–2958. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bernardes, N.; Abreu, S.; Carvalho, F.A.; Fernandes, F.; Santos, N.; Fialho, A.M. Modulation of Membrane Properties of Lung Cancer Cells by Azurin Enhances the Sensitivity to EGFR-Targeted Therapy and Decreased. β 1 Integrin.— Mediat. Adhesion. Cell Cycle 2016, 15, 1415–1424. [Google Scholar] [CrossRef] [Green Version]
- Taylor, B.N.; Mehta, R.R.; Yamada, T.; Lekmine, F.; Christov, K.; Chakrabarty, A.M.; Green, A.; Bratescu, L.; Shilkaitis, A.; Beattie, C.W.; et al. Noncationic peptides obtained from azurin preferentially enter cancer cells. Cancer Res. 2009, 69, 537–546. [Google Scholar] [CrossRef] [Green Version]
- Chaudhari, A.; Mahfouz, M.; Fialho, A.M.; Yamada, T.; Granja, A.T.; Zhu, Y.; Hashimoto, W.; Schlarb-Ridley, B.; Cho, W.; Das Gupta, T.K.; et al. Cupredoxin—Cancer Interrelationship: Azurin Binding with EphB2, Interference in EphB2 Tyrosine Phosphorylation, and Inhibition of Cancer Growth. Biochemistry 2007, 46, 1799–1810. [Google Scholar] [CrossRef]
- Micewicz, E.D.; Jung, C.; Shaue, D.; Ewa, D.; Luong, H.; McBride, W.H.; Ruchala, P. Small Azurin Derived Peptide Targets Ephrin Receptors for Radiotherapy. Int. J. Pept. Res. Ther. 2011, 17, 247–257. [Google Scholar] [CrossRef]
- Bernardes, N.; Garizo, A.R.; Pinto, S.N.; Caniço, B.; Perdigão, C.; Fernandes, F.; Fialho, A.M. Azurin Interaction with the Lipid Raft Components Ganglioside GM-1 and Caveolin-1 Increases Membrane Fluidity and Sensitivity to Anti-Cancer Drugs. Cell Cycle 2018, 17, 1649–1666. [Google Scholar] [CrossRef]
- Coelho, L.F. Anticancer Activity of CT-p19LC, a Synthetic Peptide Derived from the Bacterial Protein Azurin. Master’s Thesis, Instituto Superior Técnico, University of Lisbon, Lisbon, Portugal, 2017. [Google Scholar]
- Whitmore, L.; Wallace, B.A. DICHROWEB, an Online Server for Protein Secondary Structure Analyses from Circular Dichroism Spectroscopic Data. Nucleic Acids Res. 2004, 32, W668–W673. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cozens, A.L.; Yezzi, M.J.; Kunzelmann, K.; Ohrui, T.; Chin, L.; Eng, K.; Finkbeiner, W.E.; Widdicombe, J.H.; Gruenert, D. CFfR Expression and Chloride Secretion in Polarized Immortal Human Bronchial Epithelial Cells. Am. J. Respir. Cell. Mol. Biol. 1994, 10, 38–47. [Google Scholar] [CrossRef] [PubMed]
- Pinto, S.N.; Silva, L.C.; de Almeida, R.F.M.; Prieto, M. Membrane Domain Formation, Interdigitation, and Morphological Alterations Induced by the Very Long Chain Asymmetric C24:1 Ceramide. Biophys. J. 2008, 95, 2867–2879. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Owen, D.M.; Rentero, C.; Magenau, A.; Abu-Siniyeh, A.; Gaus, K. Quantitative Imaging of Membrane Lipid Order in Cells and Organisms. Nat. Protoc. 2012, 7, 24–35. [Google Scholar] [CrossRef]
- Yamada, T.; Fialho, A.M.; Punj, V.; Bratescu, L.; Das Gupta, T.K.; Chakrabarty, A.M. Internalization of bacterial redox protein azurin in mammalian cells: Entry domain and specificity. Cell. Microbiol. 2005, 7, 1418–1431. [Google Scholar] [CrossRef] [PubMed]
- Freire, J.M.; Domingues, M.M.; Matos, J.; Melo, M.N.; Veiga, A.S.; Santos, N.C.; Castanho, M.A.R.B. Using zeta-potential measurements to quantify peptide partition to lipid membranes. Eur. Biophys. J. 2011, 40, 481–487. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Domingues, M.M.; Santiago, P.S.; Castanho, M.A.; Santos, N.C. What can light scattering spectroscopy do for membrane-active peptide studies? J. Pept. Sci. 2008, 14, 394–400. [Google Scholar] [CrossRef] [PubMed]
- Pinto, S.N.; Fernandes, F.; Fedorov, A.; Futerman, A.H.; Silva, L.C.; Prieto, M. A Combined Fluorescence Spectroscopy, Confocal and 2-Photon Microscopy Approach to Re-Evaluate the Properties of Sphingolipid Domains. BBA—Biomembr. 2013, 1828, 2099–2110. [Google Scholar] [CrossRef] [PubMed] [Green Version]

| PROTEIN/ PEPTIDE | STRUCTURE | AMINO ACID SEQUENCE | AZURIN POSITION | HYDROPHOBICITY | CHARGE | ISOELECTRIC POINT | WATER SOLUBILITY | SMV SCORE | VIABILITY DECREASE AFTER 100 μM OF PROTEIN/ PEPTIDE TREATMENT | REFERENCES |
|---|---|---|---|---|---|---|---|---|---|---|
| Azurin |  | 128 aa | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | 20–40% | [30] |
| p28 |  | LSTAADMQGVVTDGMASGLDKDYLKPDD | 50–77 aa | n.a. | n.a. | n.a. | n.a. | n.a. | 0–25% | [21] |
| CT-p26 |  | VTFDVSKLKEGEQYMFFCTFPGHSAL | 95–120 aa | −0.03 | −0.5 | 5.3 | Poor | 0.76 | n.a. | [31] |
| CT-p19 | n.a. | VSKLKEGEQYMFFCTFPGH | 99–117 aa | −0.08 | 0.5 | 7.0 | Poor | 0.90 | n.a. | [31] |
| CT-p19LC | n.a. | VSKLRKGEKYMFFCTFPGH | n.a. | −0.16 | 3.5 | 10.0 | Good | 0.99 | n.a. | [31] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Garizo, A.R.; Coelho, L.F.; Pinto, S.; Dias, T.P.; Fernandes, F.; Bernardes, N.; Fialho, A.M. The Azurin-Derived Peptide CT-p19LC Exhibits Membrane-Active Properties and Induces Cancer Cell Death. Biomedicines 2021, 9, 1194. https://doi.org/10.3390/biomedicines9091194
Garizo AR, Coelho LF, Pinto S, Dias TP, Fernandes F, Bernardes N, Fialho AM. The Azurin-Derived Peptide CT-p19LC Exhibits Membrane-Active Properties and Induces Cancer Cell Death. Biomedicines. 2021; 9(9):1194. https://doi.org/10.3390/biomedicines9091194
Chicago/Turabian StyleGarizo, Ana Rita, Lígia F. Coelho, Sandra Pinto, Tiago P. Dias, Fábio Fernandes, Nuno Bernardes, and Arsénio M. Fialho. 2021. "The Azurin-Derived Peptide CT-p19LC Exhibits Membrane-Active Properties and Induces Cancer Cell Death" Biomedicines 9, no. 9: 1194. https://doi.org/10.3390/biomedicines9091194
APA StyleGarizo, A. R., Coelho, L. F., Pinto, S., Dias, T. P., Fernandes, F., Bernardes, N., & Fialho, A. M. (2021). The Azurin-Derived Peptide CT-p19LC Exhibits Membrane-Active Properties and Induces Cancer Cell Death. Biomedicines, 9(9), 1194. https://doi.org/10.3390/biomedicines9091194










