Immunity and Breast Cancer: Focus on Eosinophils
Abstract
1. Introduction
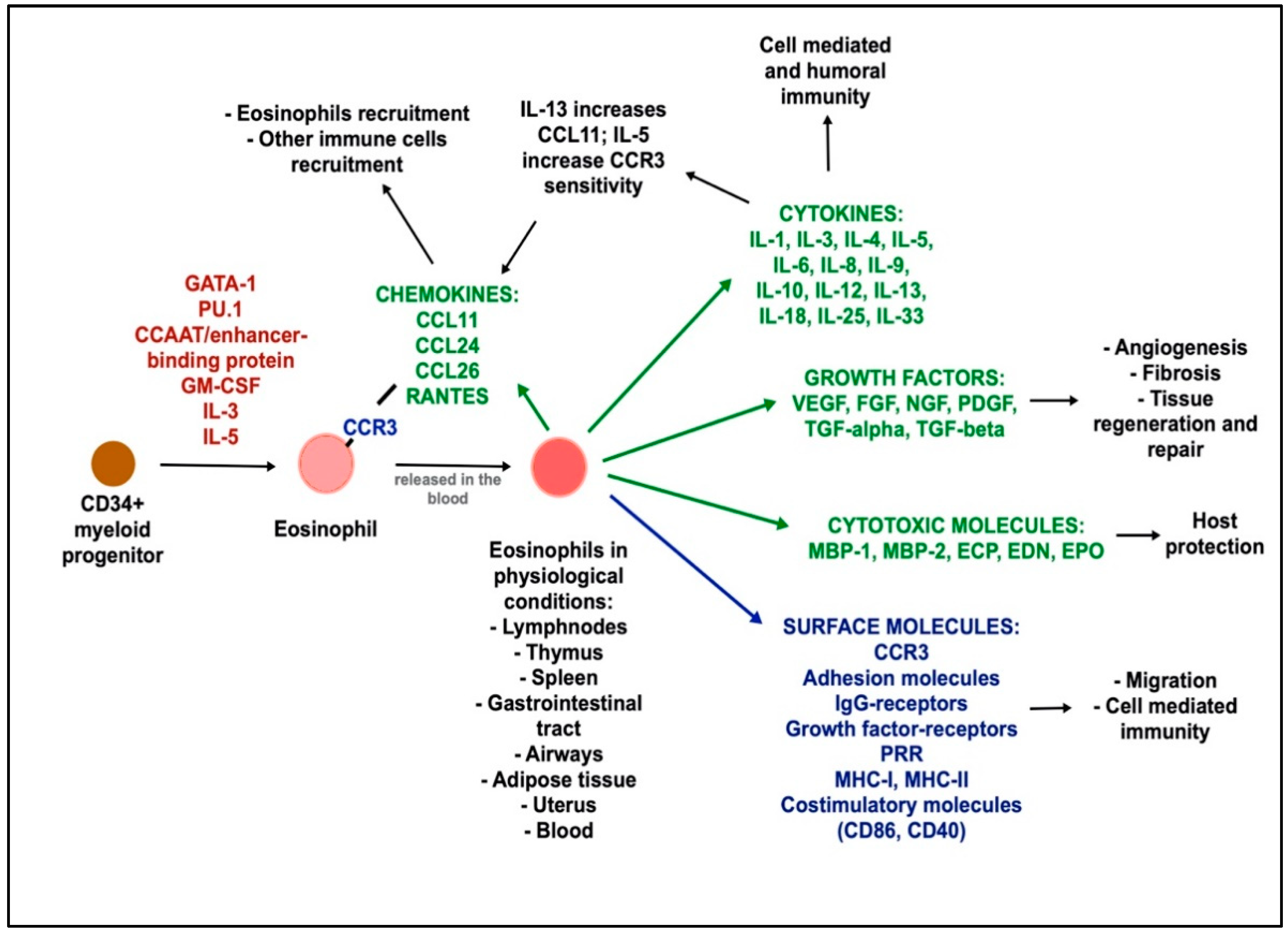
2. Eosinophil Biology and Functions
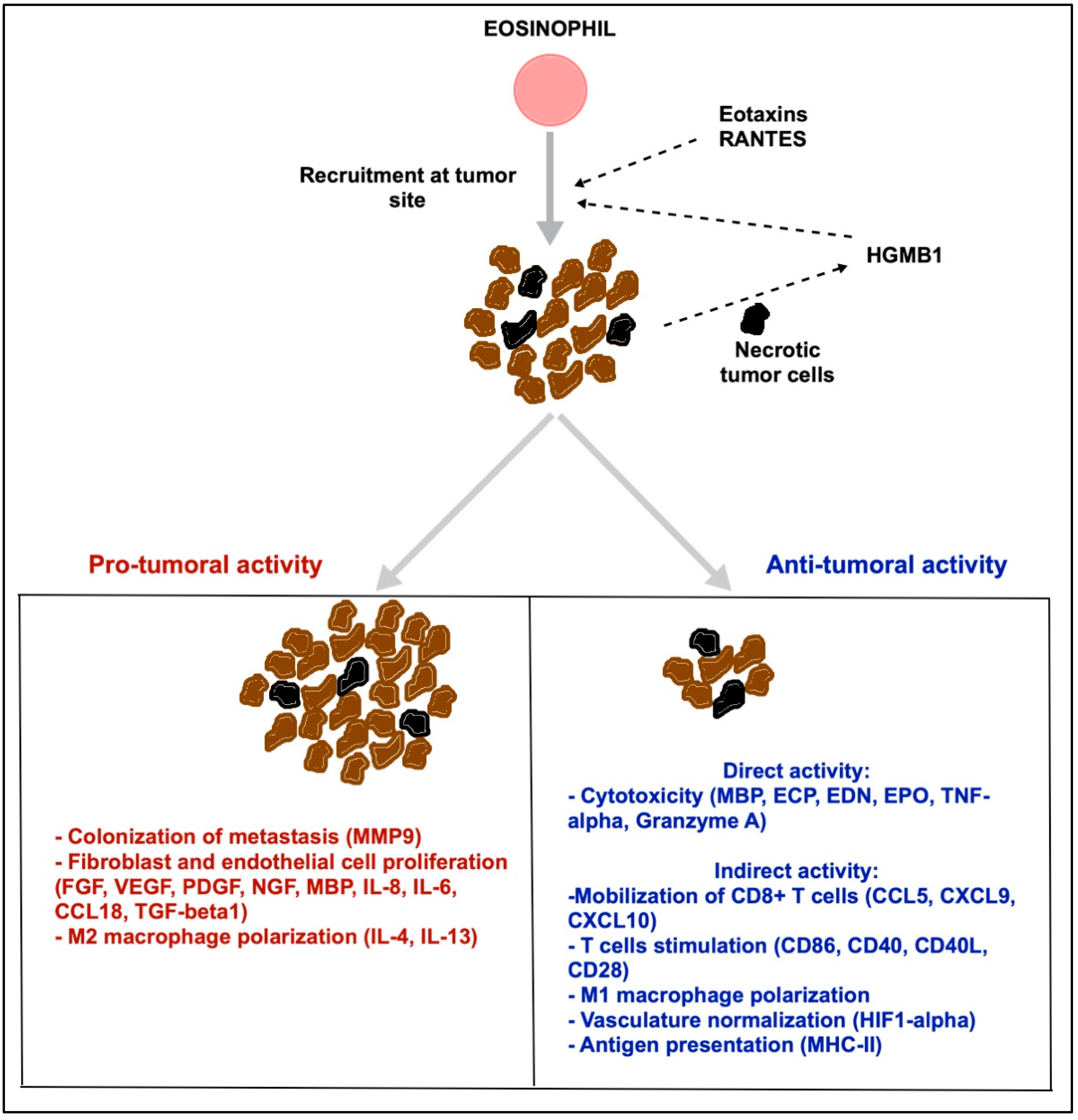
3. Eosinophils and Cancer
4. Eosinophils and Breast Cancer
4.1. Tumor-Associated Tissue Eosinophilia (TATE)
4.2. Preclinical Studies in Breast Cancer
4.3. Circulating Eosinophils in Breast Cancer
4.4. Eosinophil-Related Cytokines in Breast Cancer
4.5. Allergy and Breast Cancer
5. Discussion
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Bray, F.; Ferlay, J.; Soerjomataram, I.; Siegel, R.L.; Torre, A.L.; Jemal, A. Global cancer statistics 2018, GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2018, 68, 394–424. [Google Scholar] [CrossRef]
- Loibl, S.; Schneeweiss, A.; Huober, J.B.; Braun, M.; Rey, J.; Blohmer, J.U.; Furlanetto, J.; Zahm, D.M.; Hanusch, C.; Thomalla, J.; et al. Durvalumab improves long-term outcome in TNBC: Results from the phase II randomized GeparNUEVO study investigating neodjuvant durvalumab in addition to an anthracycline/taxane based neoadjuvant chemotherapy in early triple-negative breast cancer (TNBC). J. Clin. Oncol. 2021, 39 (Suppl. 15), 506. [Google Scholar] [CrossRef]
- Mittal, D.; Gubin, M.M.; Schreiber, R.D.; Smyth, M.J. New insights into cancer immunoediting and its three component phases—Elimination, equilibrium and escape. Curr. Opin. Immunol. 2014, 27, 16–25. [Google Scholar] [CrossRef] [PubMed]
- Ono, M.; Tsuda, H.; Shimizu, C.; Yamamoto, S.; Shibata, T.; Yamamoto, H.; Hirata, T.; Yonemori, K.; Ando, M.; Tamura, K.; et al. Tumor-infiltrating lymphocytes are correlated with response to neoadjuvant chemotherapy in triple-negative breast cancer. Breast Cancer Res. Treat. 2012, 132, 793–805. [Google Scholar] [CrossRef] [PubMed]
- Hwang, H.W.; Jung, H.; Hyeon, J.; Park, Y.H.; Ahn, J.S.; Im, Y.; Nam, S.J.; Kim, S.W.; Lee, J.E.; Yu, J.; et al. A nomogram to predict pathologic complete response (pCR) and the value of tumor-infiltrating lymphocytes (TILs) for prediction of response to neoadjuvant chemotherapy (NAC) in breast cancer patients. Breast Cancer Res. Treat. 2019, 173, 255–266. [Google Scholar] [CrossRef] [PubMed]
- Stanton, S.E.; Disis, M.L. Clinical significance of tumor-infiltrating lymphocytes in breast cancer. J. Immunother. Cancer 2016, 4, 59. [Google Scholar] [CrossRef]
- Salgado, R.; Denkert, C.; Campbell, C.; Savas, P.; Nuciforo, P.; Aura, C.; de Azambuja, E.; Eidtmann, H.; Ellis, C.E.; Baselga, J.; et al. Tumor-Infiltrating Lymphocytes and Associations with Pathological Complete Response and Event-Free Survival in HER2-Positive Early-Stage Breast Cancer Treated with Lapatinib and Trastuzumab: A Secondary Analysis of the NeoALTTO Trial. JAMA Oncol. 2015, 1, 448–454. [Google Scholar] [CrossRef]
- Denkert, C.; Loibl, S.; Noske, A.; Roller, M.; Müller, B.M.; Komor, M.; Budczies, J.; Darb-Esfahani, S.; Kronenwett, R.; Hanusch, C.; et al. Tumor-associated lymphocytes as an independent predictor of response to neoadjuvant chemotherapy in breast cancer. J. Clin. Oncol. 2010, 28, 105–113. [Google Scholar] [CrossRef]
- Denkert, C.; von Minckwitz, G.; Brase, J.C.; Sinn, B.V.; Gade, S.; Kronenwett, R.; Pfitzner, B.M.; Salat, C.; Loi, S.; Schmitt, W.D.; et al. Tumor-infiltrating lymphocytes and response to neoadjuvant chemotherapy with or without carboplatin in human epidermal growth factor receptor 2-positive and triple-negative primary breast cancers. J. Clin. Oncol. 2015, 33, 983–991. [Google Scholar] [CrossRef]
- Stanton, S.E.; Adams, S.; Disis, M.L. Variation in the Incidence and Magnitude of Tumor-Infiltrating Lymphocytes in Breast Cancer Subtypes: A Systematic Review. JAMA Oncol. 2016, 2, 1354–1360. [Google Scholar] [CrossRef]
- Khoury, T.; Nagrale, V.; Opyrchal, M.; Peng, X.; Wang, D.; Yao, S. Prognostic Significance of Stromal Versus Intratumoral Infiltrating Lymphocytes in Different Subtypes of Breast Cancer Treated with Cytotoxic Neoadjuvant Chemotherapy. Appl. Immunohistochem. Mol. Morphol. 2018, 26, 523–532. [Google Scholar] [CrossRef]
- Peng, G.L.; Li, L.; Guo, Y.W.; Yu, P.; Yin, X.; Wang, S.; Liu, C. CD8(+) cytotoxic and FoxP3(+) regulatory T lymphocytes serve as prognostic factors in breast cancer. Am. J. Transl. Res. 2019, 11, 5039–5053. [Google Scholar]
- Ladoire, S.; Arnould, L.; Apetoh, L.; Coudert, B.; Martin, F.; Chauffert, B.; Fumoleau, P.; Ghiringhelli, F. Pathologic complete response to neoadjuvant chemotherapy of breast carcinoma is associated with the disappearance of tumor-infiltrating foxp3+ regulatory T cells. Clin. Cancer Res. 2008, 14, 2413–2420. [Google Scholar] [CrossRef]
- Wagner, J.; Rapsomaniki, M.A.; Chevrier, S.; Anzeneder, T.; Langwieder, C.; Dykgers, A.; Rees, M.; Ramaswamy, A.; Muenst, S.; Soysal, S.D.; et al. A Single-Cell Atlas of the Tumor and Immune Ecosystem of Human Breast Cancer. Cell 2019, 177, 1330–1345.e18. [Google Scholar] [CrossRef] [PubMed]
- Salemme, V.; Centonze, G.; Cavallo, F.; Defilippi, P.; Conti, L. The Crosstalk Between Tumor Cells and the Immune Microenvironment in Breast Cancer: Implications for Immunotherapy. Front. Oncol. 2021, 11, 610303. [Google Scholar] [CrossRef] [PubMed]
- Ali, H.R.; Chlon, L.; Pharoah, P.D.; Markowetz, F.; Caldas, C. Patterns of Immune Infiltration in Breast Cancer and Their Clinical Implications: A Gene-Expression-Based Retrospective Study. PLoS Med. 2016, 13, e1002194. [Google Scholar] [CrossRef] [PubMed]
- Papatestas, A.E.; Lesnick, G.J.; Genkins, G.; Aufses Jr, A.H. The prognostic significance of peripheral lymphocyte counts in patients with breast carcinoma. Cancer 1976, 37, 164–168. [Google Scholar] [CrossRef]
- Vicente Conesa, M.A.; Garcia-Martinez, E.; Gonzalez Billalabeitia, E.; Benito, A.; Garcia Garcia, T.; Vicente Garcia, V.; Ayala de la Peña, F. Predictive value of peripheral blood lymphocyte count in breast cancer patients treated with primary chemotherapy. Breast 2012, 21, 468–474. [Google Scholar] [CrossRef] [PubMed]
- Ownby, H.E.; Roi, L.D.; Isenberg, R.R.; Brennan, M.J. Peripheral lymphocyte and eosinophil counts as indicators of prognosis in primary breast cancer. Cancer 1983, 52, 126–130. [Google Scholar] [CrossRef]
- Pattison, C.W.; Woods, K.L.; Morrison, J.M. Lymphocytopenia as an independent predictor of early recurrence in breast cancer. Br. J. Cancer 1987, 55, 75–76. [Google Scholar] [CrossRef] [PubMed]
- Koh, C.H.; Bhoo-Pathy, N.; Ng, K.L.; Jabir, R.S.; Tan, G.; See, M.; Jamaris, S.; Taib, N.A. Utility of pre-treatment neutrophil-lymphocyte ratio and platelet-lymphocyte ratio as prognostic factors in breast cancer. Br. J. Cancer 2015, 113, 150–158. [Google Scholar] [CrossRef]
- Simon, S.C.S.; Utikal, J.; Umansky, V. Opposing roles of eosinophils in cancer. Cancer Immunol. Immunother. 2019, 68, 823–833. [Google Scholar] [CrossRef]
- Marichal, T.; Mesnil, C.; Bureau, F. Homeostatic Eosinophils: Characteristics and Functions. Front. Med. 2017, 4, 101. [Google Scholar] [CrossRef]
- Lowe, D.; Jorizzo, J.; Hutt, M.S. Tumour-associated eosinophilia: A review. J. Clin. Pathol. 1981, 34, 1343–1348. [Google Scholar] [CrossRef]
- Galdiero, M.R.; Varricchi, G.; Seaf, M.; Marone, G.; Levi-Shaffer, F.; Marone, G. Bidirectional Mast Cell-Eosinophil Interactions in Inflammatory Disorders and Cancer. Front. Med. 2017, 4, 103. [Google Scholar] [CrossRef]
- Davis, B.P.; Rothenberg, M.E. Eosinophils and cancer. Cancer Immunol. Res. 2014, 2, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Gatault, S.; Legrand, F.; Delbeke, M.; Loiseau, S.; Capron, M. Involvement of eosinophils in the anti-tumor response. Cancer Immunol. Immunother. 2012, 61, 1527–1534. [Google Scholar] [CrossRef] [PubMed]
- Drissen, R.; Buza-Vidas, N.; Woll, P.; Thongjuea, S.; Gambardella, A.; Giustacchini, A.; Mancini, E.; Zriwil, A.; Lutteropp, M.; Grover, A.; et al. Distinct myeloid progenitor-differentiation pathways identified through single-cell RNA sequencing. Nat. Immunol. 2016, 17, 666–676. [Google Scholar] [CrossRef] [PubMed]
- Reichman, H.; Karo-Atar, D.; Munitz, A. Emerging Roles for Eosinophils in the Tumor Microenvironment. Trends Cancer 2016, 2, 664–675. [Google Scholar] [CrossRef]
- Sakkal, S.; Miller, S.; Apostolopoulos, V.; Nurgali, K. Eosinophils in Cancer: Favourable or Unfavourable? Curr. Med. Chem. 2016, 23, 650–666. [Google Scholar] [CrossRef]
- Yousefi, S.; Simon, D.; Stojkov, D.; Karsonova, A.; Karaulov, A.; Simon, H. In vivo evidence for extracellular DNA trap formation. Cell Death Dis. 2020, 11, 300. [Google Scholar] [CrossRef]
- Grisaru-Tal, S.; Itan, M.; Klion, A.D.; Munitz, A. A new dawn for eosinophils in the tumour microenvironment. Nat. Rev. Cancer 2020, 20, 594–607. [Google Scholar] [CrossRef] [PubMed]
- Mattei, F.; Andreoni, S.; Manone, G.; Gambardella, A.R.; Loffredo, S.; Varricchi, G.; Schiavoni, G. Eosinophils in the Tumor Microenvironment. Adv. Exp. Med. Biol. 2020, 1273, 1–28. [Google Scholar] [PubMed]
- Carretero, R.; Sektioglu, I.M.; Garbi, N.; Salgado, O.C.; Beckhove, P.; Hämmerling, G.J. Eosinophils orchestrate cancer rejection by normalizing tumor vessels and enhancing infiltration of CD8(+) T cells. Nat. Immunol. 2015, 16, 609–617. [Google Scholar] [CrossRef] [PubMed]
- Hollande, C.; Boussier, J.; Ziai, J.; Nozawa, T.; Bondet, V.; Phung, W.; Lu, B.; Darragh Duffy, D.; Paradis, V.; Mallet, V.; et al. Inhibition of the dipeptidyl peptidase DPP4 (CD26) reveals IL-33-dependent eosinophil-mediated control of tumor growth. Nat. Immunol. 2019, 20, 257–264. [Google Scholar] [CrossRef]
- Kienzl, M.; Hasenoehrl, C.; Valadez-Cosmes, P.; Maitz, K.; Sarsembayeva, A.; Sturm, E.; Heinemann, A.; Kargl, J.; Schicho, R. IL-33 reduces tumor growth in models of colorectal cancer with the help of eosinophils. Oncoimmunology 2020, 9, 1776059. [Google Scholar] [CrossRef] [PubMed]
- Tamura, N.; Ishii, N.; Nakazawa, M.; Nagoya, M.; Yoshinari, M.; Amano, T.; Nakazima, H.; Minami, M. Requirement of CD80 and CD86 molecules for antigen presentation by eosinophils. Scand J. Immunol. 1996, 44, 229–238. [Google Scholar] [CrossRef]
- Mattes, J.; Hulett, M.; Xie, W.; Hogan, S.; Rothenberg, M.E.; Foster, P.; Parish, C. Immunotherapy of cytotoxic T cell-resistant tumors by T helper 2 cells: An eotaxin and STAT6-dependent process. J. Exp. Med. 2003, 197, 387–393. [Google Scholar] [CrossRef] [PubMed]
- Zheng, X.; Zhang, N.; Qian, L.; Wang, X.; Fan, P.; Kuai, J.; Lin, S.; Liu, C.; Jiang, W.; Qin, S.; et al. CTLA4 blockade promotes vessel normalization in breast tumors via the accumulation of eosinophils. Int. J. Cancer 2020, 146, 1730–1740. [Google Scholar] [CrossRef]
- Arnold, I.C.; Artola-Boran, M.; Gurtner, A.; Bertram, K.; Bauer, M.; Frangez, Z.; Becher, B.; Kopf, M.; Yousefi, S.; Simon, H.; et al. The GM-CSF-IRF5 signaling axis in eosinophils promotes antitumor immunity through activation of type 1 T cell responses. J. Exp. Med. 2020, 217, e20190706. [Google Scholar] [CrossRef]
- Jacquelot, N.; Seillet, C.; Wang, M.; Pizzolla, A.; Yang Liao, Y.; Hediyeh-Zadeh, S.; Grisaru-Tal, S.; Louis, C.; Huang, O.; Schreuder, J.; et al. Blockade of the co-inhibitory molecule PD-1 unleashes ILC2-dependent antitumor immunity in melanoma. Nat. Immunol. 2021, 22, 851–864. [Google Scholar] [CrossRef] [PubMed]
- Lai, W.; Xie, H.; Liu, Y.; Zheng, F.; Zhang, Y.; Lei, Q.; Lv, L.; Dong, J.; Song, J.; Gao, X.; et al. Human pluripotent stem cell-derived eosinophils reveal potent cytotoxicity against solid tumors. Stem Cell Rep. 2021, 16, 1697–1704. [Google Scholar] [CrossRef] [PubMed]
- Cheng, J.N.; Luo, W.; Sun, C.; Jin, Z.; Zeng, X.; Alexander, P.B.; Gong, Z.; Xia, X.; Ding, X.; Xu, S.; et al. Radiation-induced eosinophils improve cytotoxic T lymphocyte recruitment and response to immunotherapy. Sci. Adv. 2021, 7, eabc7609. [Google Scholar] [CrossRef]
- Noffz, G.; Qin, Z.; Kopf, M.; Blankenstein, T. Neutrophils but not eosinophils are involved in growth suppression of IL-4-secreting tumors. J. Immunol. 1998, 160, 345–350. [Google Scholar]
- Hu, G.; Wang, S.; Zhong, K.; XU, F.; Huang, L.; Chen, W.; Cheng, P. Tumor-associated tissue eosinophilia predicts favorable clinical outcome in solid tumors: A meta-analysis. BMC Cancer 2020, 20, 454. [Google Scholar] [CrossRef]
- Alrawi, S.J.; Tan, D.; Stoler, D.L.; Dayton, M.; Anderson, G.R.; Mojica, P.; Douglas, W.; Hicks Jr, W.; Rigual, N.; Loree, T. Tissue eosinophilic infiltration: A useful marker for assessing stromal invasion, survival and locoregional recurrence in head and neck squamous neoplasia. Cancer J. 2005, 11, 217–225. [Google Scholar] [CrossRef] [PubMed]
- De Paz, D.; Chang, K.P.; Kao, H.K.; Lao, W.W.; Huang, Y.; Chang, Y.; Huang, Y. Clinical Implications of Tumor-Associated Tissue Eosinophilia in Tongue Squamous Cell Carcinoma. Laryngoscope 2019, 129, 1123–1129. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, D.T.; Biassi, T.P.; Faustino, S.E.; Carvalho, A.L.; Landman, G.; Kowalski, L.P. Eosinophils may predict occult lymph node metastasis in early oral cancer. Clin. Oral. Investig. 2012, 16, 1523–1528. [Google Scholar] [CrossRef]
- van Driel, W.J.; Hogendoorn, P.C.; Jansen, F.W.; Zwinderman, A.H.; Trimbos, J.B.; Fleuren, G.J. Tumor-associated eosinophilic infiltrate of cervical cancer is indicative for a less effective immune response. Hum. Pathol. 1996, 27, 904–911. [Google Scholar] [CrossRef]
- Mascitti, M.; Togni, L.; Rubini, C.; Troiano, G.; Lo Muzio, L.; Santarelli, A. Tumour-associated tissue eosinophilia (TATE) in oral squamous cell carcinoma: A comprehensive review. Histol. Histopathol. 2021, 36, 113–122. [Google Scholar]
- Samoszuk, M.K.; Nguyen, V.; Gluzman, I.; Pham, J.H. Occult deposition of eosinophil peroxidase in a subset of human breast carcinomas. Am. J. Pathol. 1996, 148, 701–706. [Google Scholar] [PubMed]
- Chouliaras, K.; Tokumaru, Y.; Asaoka, M.; Oshi, M.; Attwood, K.M.; Yoshida, K.; Ishikawa, T.; Takabe, K. Prevalence and clinical relevance of tumor-associated tissue eosinophilia (TATE) in breast cancer. Surgery 2020, 169, 1234–1239. [Google Scholar] [CrossRef]
- Grisaru-Tal, S.; Itan, M.; Grass, D.G.; Torres-Roca, J.; Eschrich, S.A.; Gordon, Y.; Dolitzk, A.; Hazut, I.; Avlas, S.; Jacobsen, E.A.; et al. Primary tumors from mucosal barrier organs drive unique eosinophil infiltration patterns and clinical associations. Oncoimmunology 2020, 10, 1859732. [Google Scholar] [CrossRef] [PubMed]
- Voorwerk, L.; Garner, H.; Blomberg, O.S.; Spagnuolo, L.; Chalabi, M.; van Dyk, E.; Isaeva, O.I.; Bakker, N.; Klaver, C.; Duijst, M.; et al. LBA10 Critical role of eosinophils during response to immune checkpoint blockade in breast cancer and other cancer types. Ann. Oncology. 2020, 31, S1142. [Google Scholar] [CrossRef]
- Maeda, T.; Kitazoe, M.; Tada, H.; de Llorens, R.; Salomon, D.S.; Ueda, M.; Yamada, H.; Seno, M. Growth inhibition of mammalian cells by eosinophil cationic protein. Eur. J. Biochem. 2002, 269, 307–316. [Google Scholar] [CrossRef] [PubMed]
- Furbert-Harris, P.M.; Laniyan, I.; Harris, D.; Dunston, G.M.; Vaughn, T.; Abdelnaby, A.; Parish-Gause, D.; Oredipe, O.A. Activated eosinophils infiltrate MCF-7 breast multicellular tumor spheroids. Anticancer Res. 2003, 23, 71–78. [Google Scholar]
- Qi, L.; Zhang, Q.; Miao, Y.; Kang, W.; Tian, Z.; Xu, D.; Xiao, W.; Fang, F. Interleukin-33 activates and recruits natural killer cells to inhibit pulmonary metastatic cancer development. Int. J. Cancer 2020, 146, 1421–1434. [Google Scholar] [CrossRef]
- Benatar, T.; Cao, M.Y.; Lee, Y.; Lightfoot, J.; Feng, N.; Gu, X.; Lee, V.; Jin, H.; Wang, M.; Wright, J.A.; et al. IL-17E, a proinflammatory cytokine, has antitumor efficacy against several tumor types in vivo. Cancer Immunol. Immunother. 2010, 59, 805–817. [Google Scholar] [CrossRef] [PubMed]
- Jovanovic, I.P.; Pejnovic, N.N.; Radosavljevic, G.D.; Pantic, J.M.; Milovanovic, M.Z.; Arsenijevic, N.N.; Lukic, M.L. Interleukin-33/ST2 axis promotes breast cancer growth and metastases by facilitating intratumoral accumulation of immunosuppressive and innate lymphoid cells. Int. J. Cancer 2014, 134, 1669–1682. [Google Scholar] [CrossRef]
- Xiao, P.; Wan, X.; Cui, B.; Liu, Y.; Qiu, C.; Rong, J.; Zheng, M.; Song, Y.; Chen, L.; He, J.; et al. Interleukin 33 in tumor microenvironment is crucial for the accumulation and function of myeloid-derived suppressor cells. Oncoimmunology 2016, 5, e1063772. [Google Scholar] [CrossRef]
- Shani, O.; Vorobyov, T.; Monteran, L.; Dor Lavie, D.; Cohen, N.; Raz, Y.; Tsarfaty, G.; Avivi, C.; Barshack, I.; Erez, N. Fibroblast-Derived IL33 Facilitates Breast Cancer Metastasis by Modifying the Immune Microenvironment and Driving Type 2 Immunity. Cancer Res. 2020, 80, 5317–5329. [Google Scholar] [CrossRef] [PubMed]
- Panagopoulos, V.; Leach, D.A.; Zinonos, I.; Ponomarev, V.; Licari, G.; Liapis, V.; Ingman, W.I.; Anderson, P.; DeNichilo, M.O.; Evdokiou, A. Inflammatory peroxidases promote breast cancer progression in mice via regulation of the tumour microenvironment. Int. J. Oncol. 2017, 50, 1191–1200. [Google Scholar] [CrossRef]
- Delyon, J.; Mateus, C.; Lefeuvre, D.; Lanoy, E.; Zitvogel, L.; Chaput, N.; Roy, S.; Eggermont, A.M.M.; Routier, E.; Robert, C. Experience in daily practice with ipilimumab for the treatment of patients with metastatic melanoma: An early increase in lymphocyte and eosinophil counts is associated with improved survival. Ann. Oncol. 2013, 24, 1697–1703. [Google Scholar] [CrossRef] [PubMed]
- Ferrucci, P.F.; Gandini, S.; Cocorocchio, E.; Pala, L.; Baldini, F.; Mosconi, M.; Antonini Cappellini, G.C.; Albertazzi, E.; Martinoli, C. Baseline relative eosinophil count as a predictive biomarker for ipilimumab treatment in advanced melanoma. Oncotarget 2017, 8, 79809–79815. [Google Scholar] [CrossRef]
- Weide, B.; Martens, A.; Hassel, J.C.; Berking, C.; Postow, M.A.; Bisschop, K.; Simeone, E.; Johanna Mangana, J.; Schilling, B.; Di Giacomo, A.M.; et al. Baseline Biomarkers for Outcome of Melanoma Patients Treated with Pembrolizumab. Clin. Cancer Res. 2016, 22, 5487–5496. [Google Scholar] [CrossRef]
- Sibille, A.; Henket, M.; Corhay, J.L.; Louis, R.; Duysinx, B. Clinical benefit to programmed death-1 inhibition for non-small-cell lung cancer is associated with higher blood eosinophil levels. Acta Oncol. 2020, 59, 257–259. [Google Scholar] [CrossRef] [PubMed]
- Temiz, M.Z.; Colakerol, A.; Ulus, I.; Kilic, E.; Paslanmaz, F.; Sahin, S.; Yuruk, E.; Kandirali, E.; Semercioz, A.; Muslumanoglu, A.Y. Prediction of non-muscle-invasive bladder cancer recurrence during intravesical BCG immunotherapy by use of peripheral blood eosinophil count and percentage: A preliminary report. Cancer Immunol. Immunother. 2021, 70, 245–252. [Google Scholar] [CrossRef]
- Moreira, A.; Erdmann, M.; Uslu, U.; Vass, V.; Schuler, G.; Schuler-Thurner, B. Blood Eosinophilia is an on-Treatment Biomarker in Patients with Solid Tumors Undergoing Dendritic Cell Vaccination with Autologous Tumor-RNA. Pharmaceutics 2020, 12, 210. [Google Scholar] [CrossRef]
- Sheng, L.; Chen, X.; Wang, Q.; Lyu, S.; Li, P. Interferon-α2b enhances survival and modulates transcriptional profiles and the immune response in melanoma patients treated with dendritic cell vaccines. Biomed. Pharmacother. Fr. 2020, 125, 109966. [Google Scholar] [CrossRef]
- Verhaart, S.L.; Abu-Ghanem, Y.; Mulder, S.F.; Oosting, S.; Van Der Veldt, A.; Osanto, S.; Aarts, M.J.B.; Houtsma, D.; Peters, F.P.J.; Groenewegen, G.; et al. Real-world Data of Nivolumab for Patients with Advanced Renal Cell Carcinoma in the Netherlands: An Analysis of Toxicity, Efficacy, and Predictive Markers. Clin. Genitourin. Cancer 2021, 19, e1–e274. [Google Scholar] [CrossRef] [PubMed]
- Gündüz, S.; Göksu, S.S.; Arslan, D.; Murat Tatli, A.; Uysal, M.; Gündüz, U.R.; Sevinç, M.M.; Coşkun, H.S.; Bozcuk, H.; Mutlu, H.; et al. Factors affecting disease-free survival in patients with human epidermal growth factor receptor 2-positive breast cancer who receive adjuvant trastuzumab. Mol. Clin. Oncol. 2015, 3, 1109–1112. [Google Scholar] [CrossRef][Green Version]
- Onesti, C.E.; Josse, C.; Poncin, A.; Frères, P.; Poulet, C.; Bours, V.; Jerusalem, G. Predictive and prognostic role of peripheral blood eosinophil count in triple-negative and hormone receptor-negative/HER2-positive breast cancer patients undergoing neoadjuvant treatment. Oncotarget 2018, 9, 33719–33733. [Google Scholar] [CrossRef]
- Onesti, C.E.; Josse, C.; Boulet, D.; Thiry, J.; Beaumecker, B.; Bours, V.; Jerusalem, G. Blood eosinophilic relative count is prognostic for breast cancer and associated with the presence of tumor at diagnosis and at time of relapse. Oncoimmunology 2020, 9, 1761176. [Google Scholar] [CrossRef]
- Zenan, H.; Zixiong, L.; Zhicheng, Y.; Mei, H.; Xiongbin, Y.; Tiantian, W.; Min, D.; Renbin, L.; Changchang, J. Clinical prognostic evaluation of immunocytes in different molecular subtypes of breast cancer. J. Cell Physiol. 2019, 234, 20584–20602. [Google Scholar] [CrossRef]
- Takahashi, M.; Takahashi, K.; Matsumoto, S.; Takashima, T.; Asano, Y.; Morisaki, T.; Kashiwagi, S.; Noda, S.; Onoda, N.; Ohira, M.; et al. Low Eosinophil Percentages as a New Predictive Marker for Infusion Reactions Due to Trastuzumab. Anticancer. Res. 2020, 40, 4047–4051. [Google Scholar] [CrossRef] [PubMed]
- Beer, L.A.; Kossenkov, A.V.; Liu, Q.; Prak, E.L.; Domchek, S.; Speicher, D.W.; Ky, B. Baseline Immunoglobulin E Levels as a Marker of Doxorubicin- and Trastuzumab-Associated Cardiac Dysfunction. Circ. Res. 2016, 119, 1135–1144. [Google Scholar] [CrossRef]
- Taweesedt, P.T.; Nordstrom, C.W.; Stoeckel, J.; Dumic, I. Pulmonary Manifestations of Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS) Syndrome: A Systematic Review. Biomed. Res. Int. 2019, 2019, 7863815. [Google Scholar] [CrossRef] [PubMed]
- Occhipinti, M.; Falcone, R.; Onesti, C.E.; Marchetti, P. Hyperprogressive Disease and Early Hypereosinophilia After Anti-PD-1 Treatment: A Case Report. Drug Saf. Case Rep. 2018, 5, 12. [Google Scholar] [CrossRef] [PubMed]
- Dehqanzada, Z.A.; Storrer, C.E.; Hueman, M.T.; Foley, R.J.; Harris, K.A.; Jama, Y.H.; Shriver, C.D.; Ponniah, S.; Peoples, G.E. Assessing serum cytokine profiles in breast cancer patients receiving a HER2/neu vaccine using Luminex technology. Oncol. Rep. 2007, 17, 687–694. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez, R.M.; Daly, D.S.; Tan, R.; Marks, J.R.; Zangar, R.C. Plasma biomarker profiles differ depending on breast cancer subtype but RANTES is consistently increased. Cancer Epidemiol. Biomark. Prev. 2011, 20, 1543–1551. [Google Scholar] [CrossRef]
- Lv, M.; Xiaoping, X.; Cai, H.; Li, D.; Wang, J.; Fu, X.; Yu, F.; Sun, M.; Lv, Z. Cytokines as prognstic tool in breast carcinoma. Front. Biosci. 2011, 16, 2515–2526. [Google Scholar] [CrossRef]
- Mortimer, J.E.; Waliany, S.; Dieli-Conwright, C.M.; Patel, S.K.; Hurria, A.; Chao, J.; Tiep, B.; Behrendt, C.E. Objective physical and mental markers of self-reported fatigue in women undergoing (neo)adjuvant chemotherapy for early-stage breast cancer. Cancer 2017, 123, 1810–1816. [Google Scholar] [CrossRef]
- Yu, L.R.; Cao, Z.; Makhoul, I.; Daniels, J.R.; Klimberg, S.; Wei, J.Y.; Bai, J.P.; Li, J.; Lathrop, J.T.; Beger, R.D.; et al. Immune response proteins as predictive biomarkers of doxorubicin-induced cardiotoxicity in breast cancer patients. Exp. Biol. Med. 2018, 243, 248–255. [Google Scholar] [CrossRef] [PubMed]
- König, A.; Vilsmaier, T.; Rack, B.; Klaus Friese, K.; Janni, W.; Jeschke, U.; Andergassen, U.; Trapp, E.; Jückstock, J.; Jäge, B.; et al. Determination of Interleukin-4, -5, -6, -8 and -13 in Serum of Patients with Breast Cancer Before Treatment and its Correlation to Circulating Tumor Cells. Anticancer. Res. 2016, 36, 3123–3130. [Google Scholar] [PubMed]
- Little, A.C.; Pathanjeli, P.; Wu, Z.; Bao, L.; Goo, L.E.; Yates, J.A.; Oliver, C.R.; Soellner, M.B.; Merajver, S.D. IL-4/IL-13 Stimulated Macrophages Enhance Breast Cancer Invasion via Rho-GTPase Regulation of Synergistic VEGF/CCL-18 Signaling. Front. Oncol. 2019, 9, 456. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, A.; Leland, P.; Joshi, B.H.; Puri, R.K. Targeting of IL-4 and IL-13 receptors for cancer therapy. Cytokine 2015, 75, 79–88. [Google Scholar] [CrossRef]
- Onesti, C.E.J.C.; Boulet, D.; Bours, V.; Jerusalem, G. High Plasmatic level of IL-4 and IL-13 are associated with recurrence and worse survival in breast cancer. Tumori J. 2020, 106, 118. [Google Scholar]
- Turner, M.C.; Chen, Y.; Krewski, D.; Ghadirian, P. An overview of the association between allergy and cancer. Int. J. Cancer 2006, 118, 3124–3132. [Google Scholar] [CrossRef]
- Vojtechova, P.; Martin, R.M. The association of atopic diseases with breast, prostate, and colorectal cancers: A meta-analysis. Cancer Causes Control 2009, 20, 1091–1105. [Google Scholar] [CrossRef]
- Bożek, A.; Jarzab, J.; Mielnik, M.; Bogacz, A.; Kozlowska, R.; Mangold, D. Can atopy have a protective effect against cancer? PLoS ONE 2020, 15, e0226950. [Google Scholar] [CrossRef]
- Kanda, A.; Yun, Y.; Bui, D.V.; Nguyen, L.M.; Kobayashi, Y.; Suzuki, K.; Mitani, A.; Sawada, S.; Hamada, S.; Asako, M.; et al. The multiple functions and subpopulations of eosinophils in tissues under steady-state and pathological conditions. Allergol. Int. 2021, 70, 9–18. [Google Scholar] [CrossRef]
- Prin, L.; Capron, M.; Tonnel, A.B.; Bletry, O.; Capron, A. Heterogeneity of human peripheral blood eosinophils: Variability in cell density and cytotoxic ability in relation to the level and the origin of hypereosinophilia. Int. Arch. Allergy Appl. Immunol. 1983, 72, 336–346. [Google Scholar] [CrossRef] [PubMed]
- Mesnil, C.; Raulier, S.; Paulissen, G.; Xiao, X.; Birrell, M.A.; Pirottin, D.; Janss, T.; Starkl, P.; Ramery, E.; Henket, M.; et al. Lung-resident eosinophils represent a distinct regulatory eosinophil subset. J. Clin. Investig. 2016, 126, 3279–3295. [Google Scholar] [CrossRef]
- Johansson, M.W. Activation states of blood eosinophils in asthma. Clin. Exp. Allergy 2014, 44, 482–498. [Google Scholar] [CrossRef] [PubMed]
- Metcalfe, D.D.; Pawankar, R.; Ackerman, S.J.; Akin, C.; Clayton, F.; Falcone, F.H.; Gleich, G.J.; Irani, A.; Johansson, M.W.; Klion, A.D.; et al. Biomarkers of the involvement of mast cells, basophils and eosinophils in asthma and allergic diseases. World Allergy Organ. J. 2016, 9, 7. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.Y.; Jarjour, N.N.; Busse, W.W.; Kelly, E.A.B. Chemokine receptor expression on human eosinophils from peripheral blood and bronchoalveolar lavage fluid after segmental antigen challenge. J. Allergy Clin. Immunol. 2003, 112, 556–562. [Google Scholar] [CrossRef]
- Liu, L.Y.; Sedgwick, J.B.; Bates, M.E.; Vrtis, R.F.; Gern, J.E.; Kita, H.; Jarjour, N.N.; Busse, W.W.; Kelly, E.A.B. Decreased expression of membrane IL-5 receptor alpha on human eosinophils: II. IL-5 down-modulates its receptor via a proteinase-mediated process. J. Immunol. 2002, 169, 6459–6466. [Google Scholar] [CrossRef] [PubMed]
- Mengelers, H.J.; Maikoe, T.; Hooibrink, B.; Kuypers, T.W.; Kreukniet, J.; Lammers, J.W.; Koenderman, L. Down modulation of L-Selectin expression on eosinophils recovered from bronchoalveolar lavage fluid after allergen provocation. Clin. Exp. Allergy 1993, 23, 196–204. [Google Scholar] [CrossRef]
- Hansel, T.T.; Braunstein, J.B.; Walker, C.; Blaser, K.; Bruijnzeel, P.L.; Virchow Jr, J.C.; Virchow Sr, C. Sputum eosinophils from asthmatics express ICAM-1 and HLA-DR. Clin. Exp. Immunol. 1991, 86, 271–277. [Google Scholar] [CrossRef] [PubMed]
- Hansel, T.T.; De Vries, I.J.; Carballido, J.M.; Braun, R.K.; Carballido-Perrig, N.; Rihs, S.; Blaser, K.; Walker, C. Induction and function of eosinophil intercellular adhesion molecule-1 and HLA-DR. J. Immunol. 1992, 149, 2130–2136. [Google Scholar]
- Lucey, D.R.; Dorsky, D.I.; Nicholson-Weller, A.; Weller, P.F. Human eosinophils express CD4 protein and bind human immunodeficiency virus 1 gp120. J. Exp. Med. 1989, 169, 327–332. [Google Scholar] [CrossRef] [PubMed]
- Rand, T.H.; Silberstein, D.S.; Kornfeld, H.; Weller, P.F. Human eosinophils express functional interleukin 2 receptors. J. Clin. Investig. 1991, 88, 825–832. [Google Scholar] [CrossRef]
- Riedel, D.; Lindemann, A.; Brach, M.; Mertelsmann, R.; Herrmann, F. Granulocyte-macrophage colony-stimulating factor and interleukin-3 induce surface expression of interleukin-2 receptor p55-chain and CD4 by human eosinophils. Immunology 1990, 70, 258–261. [Google Scholar] [PubMed]
- Woerly, G.; Roger, N.; Loiseau, S.; Dombrowicz, D.; Capron, A.; Capron, M. Expression of CD28 and CD86 by human eosinophils and role in the secretion of type 1 cytokines (interleukin 2 and interferon gamma): Inhibition by immunoglobulin a complexes. J. Exp. Med. 1999, 190, 487–495. [Google Scholar] [CrossRef] [PubMed]
- Rosenberg, H.F.; Domachowske, J.B. Eosinophils, ribonucleases and host defense: Solving the puzzle. Immunol. Res. 1999, 20, 261–274. [Google Scholar] [CrossRef] [PubMed]
| Experimental Model | Results | Role | Reference |
|---|---|---|---|
| Breast cancer mice model | DPP4-inhibitors induced intratumoral accumulation of CCL11, resulting in eosinophilic chemotaxis and reduced tumor growth. | Anti-tumoral | Hollande et al. [35] |
| Breast cancer mice model | IL33 suppress the development of metastasis via recruitment and activation of NK cells. | Anti-tumoral | Qi et al. [57] |
| Breast cancer mice model | Anti-CTLA4 therapy increases TATE, which correlates with tumor vessel normalization and anti-CTLA4 efficacy. | Anti-tumoral | Zheng et al. [39] |
| Different mice models including breast cancer | Administration of IL-17E has antitumor activity by inducing production of IL-5 and eosinophil expansion. A higher efficacy was observed when IL-17E was used in combination with other anticancer treatments. | Anti-tumoral | Benatar et al. [58] |
| Different mice models including breast cancer | Response to cisplatine plus immune checkpoint inhibitors was lost with concomitant depletion of eosinophils. Blood eosinophils increased during treatment in mice responding to immunotherapy. | Anti-tumoral | Voorwerk et al. [54] |
| Different mice models including breast cancer | Injection of eosinophils derived from pluripotent stem cells reduces the growth of MDA-MB-231 breast cancer. | Anti-tumoral | Lai et al. [42] |
| Breast cancer mice model | IL-33 accelerates breast cancer progression and development of lung and liver metastases by inducing neovascularisation, facilitating expansion of immune suppressor cells within tumor (MDSCs, ILCs, T regs, macrophages M2) and by diminishing antitumor NK cells activity. | Pro-tumoral | Jovanovic at al. [59] |
| Breast cancer mice model | IL-33 in tumor microenvironment reduces the apoptosis and sustains the survival of MDSCs and augments their immunosuppressive ability. | Pro-tumoral | Xiao et al. [60] |
| Breast cancer mice model | IL-33 gene up-regulation in CAF associated with lung metastases. Inhibition of IL-33 reduces lung metastases. | Pro-tumoral | Shani et al. [61] |
| Breast cancer mice model | MPO and EPO increase primary tumor growth and promote metastases through promoting collagen deposition, fibroblastes migration and angiogenesis. | Pro-tumoral | Panagopoulos et al. [62] |
| Population | Number of Patients | Conclusions | Role | Reference |
|---|---|---|---|---|
| HER2+ breast cancer receiving adjuvant trastuzumab | 62 | Positive association between low baseline eosinophil count (≤70/mm³) and better disease-free survival rate. | Pro-tumoral | Gunduz et al. [71] |
| Breast cancer, all subtypes | 419 | Positive association between high baseline eosinophil count (≥55/mm³) and lower recurrence rate. | Anti-tumoral | Ownby et al. [19] |
| TNBC and HR-/HER2+ breast cancers receiving neoadjuvant chemotherapy | 112 | Positive association between baseline and post-surgery REC with pCR and survival rate. Increase in relative circulating eosinophil count after surgery, that remain stable for patients who do not experience relapse. | Anti-tumoral | Onesti et al. [72] |
| Breast cancer, all subtypes | 930 | Baseline REC ≥1.5% associated with better survival. Increase in REC after surgery, that remain stable for patients who do not experience relapse until 10 years of follow-up. | Anti-tumoral | Onesti et al. [73] |
| Breast cancer, all subtypes | 601 | No association between survival and eosinophil count. | No association | Zenan et al. [74] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Poncin, A.; Onesti, C.E.; Josse, C.; Boulet, D.; Thiry, J.; Bours, V.; Jerusalem, G. Immunity and Breast Cancer: Focus on Eosinophils. Biomedicines 2021, 9, 1087. https://doi.org/10.3390/biomedicines9091087
Poncin A, Onesti CE, Josse C, Boulet D, Thiry J, Bours V, Jerusalem G. Immunity and Breast Cancer: Focus on Eosinophils. Biomedicines. 2021; 9(9):1087. https://doi.org/10.3390/biomedicines9091087
Chicago/Turabian StylePoncin, Aurélie, Concetta Elisa Onesti, Claire Josse, Delphine Boulet, Jérôme Thiry, Vincent Bours, and Guy Jerusalem. 2021. "Immunity and Breast Cancer: Focus on Eosinophils" Biomedicines 9, no. 9: 1087. https://doi.org/10.3390/biomedicines9091087
APA StylePoncin, A., Onesti, C. E., Josse, C., Boulet, D., Thiry, J., Bours, V., & Jerusalem, G. (2021). Immunity and Breast Cancer: Focus on Eosinophils. Biomedicines, 9(9), 1087. https://doi.org/10.3390/biomedicines9091087






