Relationship between Metalloprotease-7 and -14 and Tissue Inhibitor of Metalloprotease 1 Expression by Mucosal Stromal Cells and Colorectal Cancer Development in Inflammatory Bowel Disease
Abstract
1. Introduction
2. Materials and Methods
2.1. Patient Selection, Patient Characteristics and Tissue Specimen Handling
2.2. Immunohistochemistry Analysis
2.3. Data Analysis and Statistical Methods
3. Results
3.1. Clinical–Pathological Characteristics of Patients
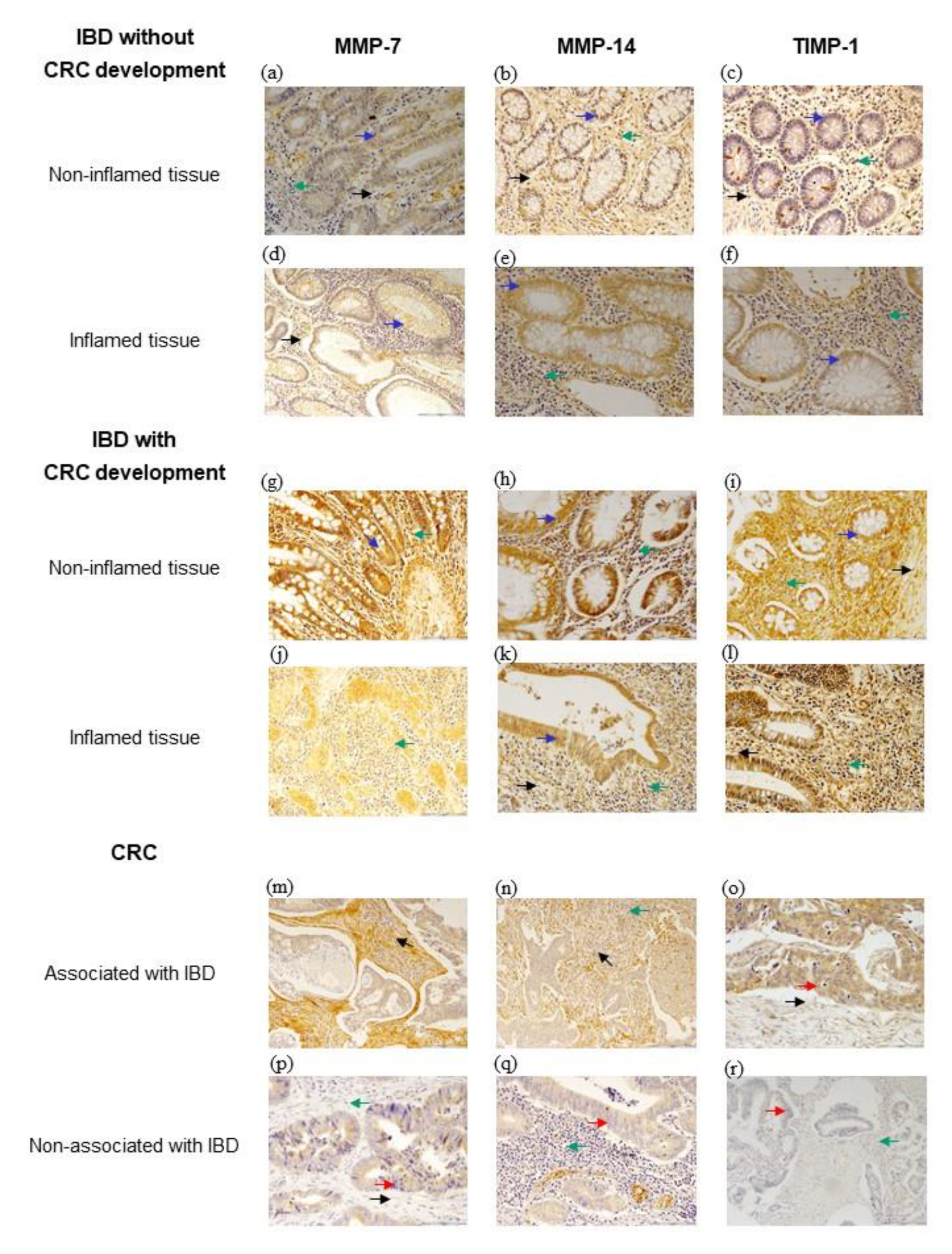
3.2. Immunohistochemical Expression of MMP-7, MMP-14 and TIMP-1
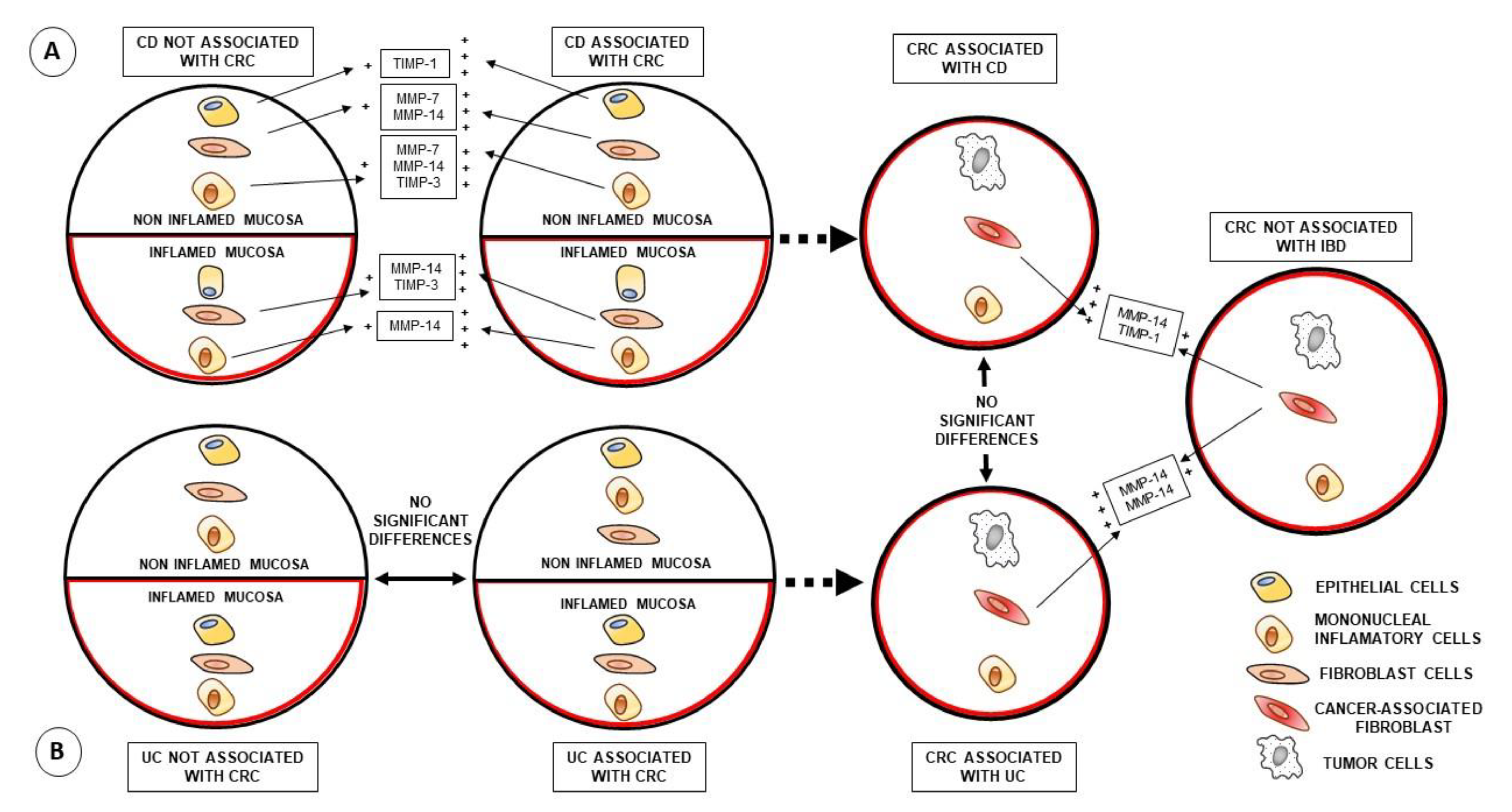
3.3. Comparison of MMP-7, MMP-14 and TIMP-1 Expression of CRC Associated or Not with IBD
3.4. Comparison of MMP-7, MMP-14 and TIMP-1 Expression of Inflamed Tissues from IBD Patients with or without CRC Development
3.5. Comparison of MMP-7, MMP-14 and TIMP-1 Expression of Non-Inflamed Tissues from IBD Patients with or without CRC Development
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Baragi, V.M.; Qiu, L.; Gunja-Smith, Z.; Woessner, J.F.J.; Lesch, C.A.; Guglietta, A. Role of metalloproteinases in the development and healing of acetic acid-induced gastric ulcer in rats. Scand. J. Gastroenterol. 1997, 32, 419–426. [Google Scholar] [CrossRef] [PubMed]
- Arthur, M.J. Fibrosis and altered matrix degradation. Digestion 1998, 59, 376–380. [Google Scholar] [CrossRef]
- Altadill, A.; Eiro, N.; Gonzalez, L.O.; Junquera, S.; Gonzalez-Quintana, J.M.; Sanchez, M.R.; Andicoechea, A.; Saro, C.; Rodrigo, L.; Vizoso, F.J. Comparative analysis of the expression of metalloproteases and their inhibitors in resected crohn’s disease and complicated diverticular disease. Inflamm. Bowel Dis. 2012, 18, 120–130. [Google Scholar] [CrossRef] [PubMed]
- von Lampe, B.; Barthel, B.; Coupland, S.E.; Riecken, E.O.; Rosewicz, S. Differential expression of matrix metalloproteinases and their tissue inhibitors in colon mucosa of patients with inflammatory bowel disease. Gut 2000, 47, 63–73. [Google Scholar] [CrossRef]
- Di Sebastiano, P.; di Mola, F.F.; Artese, L.; Rossi, C.; Mascetta, G.; Pernthaler, H.; Innocenti, P. Beneficial effects of Batimastat (BB-94), a matrix metalloproteinase inhibitor, in rat experimental colitis. Digestion 2001, 63, 234–239. [Google Scholar] [CrossRef]
- Eiro, N.; Vizoso, F.J. Inflammation and cancer. World J. Gastrointest. Surg. 2012, 4, 62–72. [Google Scholar] [CrossRef]
- Vendramini-Costa, D.B.; Carvalho, J.E. Molecular link mechanisms between inflammation and cancer. Curr. Pharm. Des. 2012, 18, 3831–3852. [Google Scholar] [CrossRef]
- Eaden, J.A.; Abrams, K.R.; Mayberry, J.F. The risk of colorectal cancer in ulcerative colitis: A meta-analysis. Gut 2001, 48, 526–535. [Google Scholar] [CrossRef]
- Rubin, D.C.; Shaker, A.; Levin, M.S. Chronic intestinal inflammation: Inflammatory bowel disease and colitis-associated colon cancer. Front. Immunol. 2012, 3, 107. [Google Scholar] [CrossRef]
- Asano, T.; Tada, M.; Cheng, S.; Takemoto, N.; Kuramae, T.; Abe, M.; Takahashi, O.; Miyamoto, M.; Hamada, J.; Moriuchi, T.; et al. Prognostic values of matrix metalloproteinase family expression in human colorectal carcinoma. J. Surg. Res. 2008, 146, 32–42. [Google Scholar] [CrossRef]
- Pesta, M.; Topolcan, O.; Holubec, L.J.; Rupert, K.; Cerna, M.; Holubec, L.S.; Treska, V.; Finek, J.; Cerny, R. Clinicopathological assessment and quantitative estimation of the matrix metalloproteinases MMP-2 and MMP-7 and the inhibitors TIMP-1 and TIMP-2 in colorectal carcinoma tissue samples. Anticancer Res. 2007, 27, 1863–1867. [Google Scholar] [PubMed]
- Gonzalez, L.; Eiro, N.; Gonzalez, L.O.; Andicoechea, A.; Barbon, E.; Garcia-Muniz, J.L.; Vizoso, F.J. Effect of the expression of matrix metalloproteases and their tissue inhibitors on survival of patients with resectable colorectal cancer. Dig. Dis. Sci. 2012, 57, 2063–2071. [Google Scholar] [CrossRef] [PubMed]
- Lyall, M.S.; Dundas, S.R.; Curran, S.; Murray, G.I. Profiling markers of prognosis in colorectal cancer. Clin. Cancer Res. 2006, 12, 1184–1191. [Google Scholar] [CrossRef]
- Egeblad, M.; Werb, Z. New functions for the matrix metalloproteinases in cancer progression. Nat. Rev. Cancer 2002, 2, 161–174. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Y.; Goldberg, I.D.; Shi, Y.E. Complex roles of tissue inhibitors of metalloproteinases in cancer. Oncogene 2002, 21, 2245–2252. [Google Scholar] [CrossRef] [PubMed]
- Curran, S.; Dundas, S.R.; Buxton, J.; Leeman, M.F.; Ramsay, R.; Murray, G.I. Matrix metalloproteinase/tissue inhibitors of matrix metalloproteinase phenotype identifies poor prognosis colorectal cancers. Clin. Cancer Res. 2004, 10, 8229–8234. [Google Scholar] [CrossRef]
- Hilska, M.; Roberts, P.J.; Collan, Y.U.; Laine, V.J.; Kossi, J.; Hirsimaki, P.; Rahkonen, O.; Laato, M. Prognostic significance of matrix metalloproteinases-1, -2, -7 and -13 and tissue inhibitors of metalloproteinases-1, -2, -3 and -4 in colorectal cancer. Int. J. Cancer 2007, 121, 714–723. [Google Scholar] [CrossRef]
- Rath, T.; Roderfeld, M.; Graf, J.; Wagner, S.; Vehr, A.K.; Dietrich, C.; Geier, A.; Roeb, E. Enhanced expression of MMP-7 and MMP-13 in inflammatory bowel disease: A precancerous potential? Inflamm. Bowel Dis. 2006, 12, 1025–1035. [Google Scholar] [CrossRef]
- González, L.O.; Corte, M.D.; Junquera, S.; González-Fernández, R.; del Casar, J.M.; García, C.; Andicoechea, A.; Vázquez, J.; Pérez-Fernández, R.; Vizoso, F.J. Expression and prognostic significance of metalloproteases and their inhibitors in luminal A and basal-like phenotypes of breast carcinoma. Hum. Pathol. 2009, 40, 1224–1233. [Google Scholar] [CrossRef]
- González, L.O.; Pidal, I.; Junquera, S.; Corte, M.D.; Vázquez, J.; Rodríguez, J.C.; Lamelas, M.L.; Merino, A.M.; García-Muñiz, J.L.; Vizoso, F.J. Overexpression of matrix metalloproteinases and their inhibitors in mononuclear inflammatory cells in breast cancer correlates with metastasis-relapse. Br. J. Cancer 2007, 97, 957–963. [Google Scholar] [CrossRef]
- Jensen, A.B.; Larsen, M.; Gislum, M.; Skriver, M.V.; Jepsen, P.; Norgaard, B.; Sorensen, H.T. Survival after colorectal cancer in patients with ulcerative colitis: A nationwide population-based Danish study. Am. J. Gastroenterol. 2006, 101, 1283–1287. [Google Scholar] [CrossRef]
- Watanabe, T.; Konishi, T.; Kishimoto, J.; Kotake, K.; Muto, T.; Sugihara, K. Japanese Society for Cancer of the Colon and Rectum. Ulcerative colitis-associated colorectal cancer shows a poorer survival than sporadic colorectal cancer: A nationwide Japanese study. Inflamm. Bowel Dis. 2011, 17, 802–808. [Google Scholar] [CrossRef]
- Nam, J.; Perera, P.; Liu, J.; Rath, B.; Deschner, J.; Gassner, R.; Butterfield, T.A.; Agarwal, S. Sequential alterations in catabolic and anabolic gene expression parallel pathological changes during progression of monoiodoacetate-induced arthritis. PLoS ONE 2011, 6, e24320. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Lee, E.J.; Gawel, D.R.; Lilja, S.; Schafer, S.; Zhang, H.; Benson, M. Meta-Analysis of Expression Profiling Data Indicates Need for Combinatorial Biomarkers in Pediatric Ulcerative Colitis. J. Immunol. Res. 2020, 2020, 8279619. [Google Scholar] [CrossRef] [PubMed]
- Fang, Y.J.; Lu, Z.H.; Wang, G.Q.; Pan, Z.Z.; Zhou, Z.W.; Yun, J.P.; Zhang, M.F.; Wan, D.S. Elevated expressions of MMP7, TROP2, and survivin are associated with survival, disease recurrence, and liver metastasis of colon cancer. Int. J. Colorectal. Dis. 2009, 24, 875–884. [Google Scholar] [CrossRef] [PubMed]
- Fuksiewicz, M.; Kotowicz, B.; Rutkowski, A.; Kowalska, M. The matrix metalloproteinase-7 and pro-enzyme of metalloproteinase-1 as a potential marker for patients with rectal cancer without distant metastasis. Tumour Biol. 2015, 36, 3629–3635. [Google Scholar] [CrossRef]
- McGuire, J.K.; Li, Q.; Parks, W.C. Matrilysin (matrix metalloproteinase-7) mediates E-cadherin ectodomain shedding in injured lung epithelium. Am. J. Pathol. 2003, 162, 1831–1843. [Google Scholar] [CrossRef]
- Ougolkov, A.V.; Yamashita, K.; Mai, M.; Minamoto, T. Oncogenic beta-catenin and MMP-7 (matrilysin) cosegregate in late-stage clinical colon cancer. Gastroenterology 2002, 122, 60–71. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Ii, M.; Yamamoto, H.; Adachi, Y.; Maruyama, Y.; Shinomura, Y. Role of matrix metalloproteinase-7 (matrilysin) in human cancer invasion, apoptosis, growth, and angiogenesis. Exp. Biol. Med. (Maywood) 2006, 231, 20–27. [Google Scholar] [CrossRef]
- Ito, T.K.; Ishii, G.; Chiba, H.; Ochiai, A. The VEGF angiogenic switch of fibroblasts is regulated by MMP-7 from cancer cells. Oncogene 2007, 26, 7194–7203. [Google Scholar] [CrossRef]
- Hemers, E.; Duval, C.; McCaig, C.; Handley, M.; Dockray, G.J.; Varro, A. Insulin-like growth factor binding protein-5 is a target of matrix metalloproteinase-7: Implications for epithelial-mesenchymal signaling. Cancer Res. 2005, 65, 7363–7369. [Google Scholar] [CrossRef] [PubMed]
- Bhowmick, N.A.; Neilson, E.G.; Moses, H.L. Stromal fibroblasts in cancer initiation and progression. Nature 2004, 432, 332–337. [Google Scholar] [CrossRef] [PubMed]
- Bai, Y.P.; Shang, K.; Chen, H.; Ding, F.; Wang, Z.; Liang, C.; Xu, Y.; Sun, M.H.; Li, Y.Y. FGF-1/-3/FGFR4 signaling in cancer-associated fibroblasts promotes tumor progression in colon cancer through Erk and MMP-7. Cancer Sci. 2015, 106, 1278–1287. [Google Scholar] [CrossRef]
- Kanazawa, A.; Oshima, T.; Yoshihara, K.; Tamura, S.; Yamada, T.; Inagaki, D.; Sato, T.; Yamamoto, N.; Shiozawa, M.; Morinaga, S.; et al. Relation of MT1-MMP gene expression to outcomes in colorectal cancer. J. Surg. Oncol. 2010, 102, 571–575. [Google Scholar] [CrossRef]
- Wurtz, S.O.; Schrohl, A.S.; Mouridsen, H.; Brunner, N. TIMP-1 as a tumor marker in breast cancer--an update. Acta Oncol. 2008, 47, 580–590. [Google Scholar] [CrossRef]
- Gustavson, M.D.; Crawford, H.C.; Fingleton, B.; Matrisian, L.M. Tcf binding sequence and position determines beta-catenin and Lef-1 responsiveness of MMP-7 promoters. Mol. Carcinog. 2004, 41, 125–139. [Google Scholar] [CrossRef]
- Kitamura, T.; Biyajima, K.; Aoki, M.; Oshima, M.; Taketo, M.M. Matrix metalloproteinase 7 is required for tumor formation, but dispensable for invasion and fibrosis in SMAD4-deficient intestinal adenocarcinomas. Lab. Investig. 2009, 89, 98–105. [Google Scholar] [CrossRef]
- Chen, L.; Ke, X. MMP7 as a potential biomarker of colon cancer and its prognostic value by bioinformatics analysis. Medicine (Baltimore) 2021, 100, e24953. [Google Scholar] [CrossRef]
- Liao, H.Y.; Da, C.M.; Liao, B.; Zhang, H.H. Roles of matrix metalloproteinase-7 (MMP-7) in cancer. Clin. Biochem. 2021. [Google Scholar] [CrossRef] [PubMed]
- Andersen, N.N.; Jess, T. Has the risk of colorectal cancer in inflammatory bowel disease decreased? World J. Gastroenterol. 2013, 19, 7561–7568. [Google Scholar] [CrossRef]
- Mooiweer, E.; van der Meulen-de Jong, A.E.; Ponsioen, C.Y.; van der Woude, C.J.; van Bodegraven, A.A.; Jansen, J.M.; Mahmmod, N.; Kremer, W.; Siersema, P.D.; Oldenburg, B.; et al. Incidence of Interval Colorectal Cancer Among Inflammatory Bowel Disease Patients Undergoing Regular Colonoscopic Surveillance. Clin. Gastroenterol. Hepatol. 2015, 13, 1656–1661. [Google Scholar] [CrossRef] [PubMed]
| Characteristics | CRCs Not Associated with IBD N (%) | CRCs Associated with IBD N (%) | p-Value |
|---|---|---|---|
| All patients | 86 (100) | 23 (100) | |
| Age (years) | 0.070 | ||
| ≤64 | 43 (50) | 17 (73.9) | |
| >64 | 43 (50) | 6 (26.1) | |
| Gender | 0.922 | ||
| Male | 49 (56.9) | 14 (60.9) | |
| Female | 37 (43) | 9 (39.1) | |
| Tumor location | 0.399 | ||
| Right | 13 (15.1) | 5 (21.7) | |
| Transverse | 2 (2.3) | 2 (8.7) | |
| Left | 34 (39.5) | 7 (30.4) | |
| Rectum | 37 (43) | 9 (39.1) | |
| Histological grade | 0.438 | ||
| Well differentiated (I) | 16 (18.6) | 5 (21.7) | |
| Moderately differentiated (II) | 65 (75.5) | 15 (65.2) | |
| Poorly differentiated (III) | 5 (5.8) | 3 (13.0) | |
| Duke’s stage | 0.869 | ||
| A | 6 (6.9) | 1 (4.8) | |
| B | 49 (56.9) | 12 (50.5) | |
| C | 17 (19.7) | 6 (27.3) | |
| D | 14 (16.3) | 3 (13.0) |
| Clinical Characteristics | IBD Patients without CRCs Development N (%) | IBD Patients with CRCs Development N (%) | p-Value |
|---|---|---|---|
| All patients | 47 (100) | 23 (100) | |
| Age (years) | 0.615 | ||
| ≤38 | 27 (57.4) | 11 (47.8) | |
| >38 | 20 (42.6) | 12 (52.2) | |
| Gender | 1 | ||
| Male | 28 (59.6) | 14 (60.9) | |
| Female | 19 (40.4) | 9 (39.1) | |
| Immunosuppressive treatment | 0.304 | ||
| No | 29 (67.4) | 19 (82.6) | |
| Yes | 14 (32.6) | 4 (17.4) | |
| All patients | 47 (100) | 23 (100) | |
| Age (years) | 0.615 | ||
| ≤38 | 27 (57.4) | 11 (47.8) | |
| >38 | 20 (42.6) | 12 (52.2) | |
| Gender | 1 | ||
| Male | 28 (59.6) | 14 (60.9) | |
| Female | 19 (40.4) | 9 (39.1) | |
| Immunosuppressive treatment | 0.304 | ||
| No | 29 (67.4) | 19 (82.6) | |
| Yes | 14 (32.6) | 4 (17.4) | |
| All patients | 47 (100) | 23 (100) |
| Factors | CRCs Not Associated with IBD N (%) | CRCs Associated with CD N (%) | CRC Associated with UC N (%) |
|---|---|---|---|
| MMP-7 | 86 (100) | 9 (100) | 13 (100) |
| Tumor cells | 67 (77.9) | 7 (77.8) | 1 (7.7) |
| Fibroblasts | 33 (38.4) | 5 (55.6) | 9 (69.2) * |
| MICs | 19 (22.1) | 3 (33.3) | 5 (38.5) |
| MMP-14 | 86 (100) | 6 (100) | 8 (100) |
| Tumor cells | 71 (82.6) | 5 (83.3) | 6 (75) |
| Fibroblasts | 13 (15.1) | 4 (66.7) ** | 6 (75) *** |
| MICs | 5 (5.8) | 1 (16.7) | 1 (12.5) |
| TIMP-1 | 86 (100) | 6 (100) | 8 (100) |
| Tumor cells | 69 (80.2) | 6 (100) | 0 (0) |
| Fibroblasts | 16 (18.6) | 4 (66.7) ** | 2 (25) |
| MICs | 27 (31.4) | 1 (16.7) | 1(12.5) |
| Factors | IBD without CRC Development N (%) | IBD with CRC Development N (%) | ||
|---|---|---|---|---|
| CD | UC | CD | UC | |
| MMP-7 | 29 (100) | 16 (100) | 8 (100) | 7 (100) |
| Epithelial cells | 22 (75.9) | 9 (56.3) | 6 (75) | 4 (57.1) |
| Fibroblasts | 13 (44.8) | 7 (43.8) | 5 (62.5) | 3 (42.9) |
| MICs | 15 (51.7) | 3 (18.8) | 5 (62.5) | 3 (42.9) |
| MMP-14 | 26 (100) | 5 (100) | 7 (100) | 4 (100) |
| Epithelial cells | 12 (46.2) | 4 (80) | 5 (71.4) | 3 (75) |
| Fibroblasts | 5 (19.2) | 4 (80) | 5 (71.4) * | 2 (50) |
| MICs | 3 (11.5) | 3 (60) | 4 (57.1) * | 0 (0) |
| TIMP-1 | 27 (100) | 15 (100) | 7 (100) | 5 (100) |
| Epithelial cells | 10 (37) | 7 (46.7) | 3 (42.9) | 1 (20) |
| Fibroblasts | 0 | 2 (13.3) | 2 (28.6) ** | 0 |
| MICs | 3(11.1) | 3 (20) | 0 | 0 |
| Factors | IBD without CRC Development N (%) | IBD with CRC Development N (%) | ||
|---|---|---|---|---|
| CD | UC | CD | UC | |
| MMP-7 | 31 (100) | 16 (100) | 9 (100) | 11 (100) |
| Epithelial cells | 17 (54.8) | 12 (75) | 7 (77.8) | 10 (90.9) |
| Fibroblasts | 4 (12.9) | 12 (75) | 6 (66.7) ** | 10 (90.9) |
| MICs | 5 (16.1) | 6 (37.5) | 5 (55.6) * | 9 (81.8) |
| MMP-14 | 27 (100) | 15 (100) | 6 (100) | 8 (100) |
| Epithelial cells | 6 (22.2) | 9 (60) | 4 (66.7) | 1 (12.5) |
| Fibroblasts | 1 (3.7) | 8 (53.3) | 3 (50) * | 1 (12.5) |
| MICs | 0 | 2 (13.3) | 2 (33.3) * | 0 |
| TIMP-1 | 27 (100) | 15 (100) | 6 (100) | 8 (100) |
| Epithelial cells | 10 (37) | 9 (60) | 6 (100) * | 1 (12.5) |
| Fibroblasts | 1 (3.7) | 2 (13.3) | 1 (16.7) | 0 |
| MICs | 2 (7.4) | 0 | 4 (66.7) ** | 0 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Altadill, A.; Eiro, N.; González, L.O.; Andicoechea, A.; Fernández-Francos, S.; Rodrigo, L.; García-Muñiz, J.L.; Vizoso, F.J. Relationship between Metalloprotease-7 and -14 and Tissue Inhibitor of Metalloprotease 1 Expression by Mucosal Stromal Cells and Colorectal Cancer Development in Inflammatory Bowel Disease. Biomedicines 2021, 9, 495. https://doi.org/10.3390/biomedicines9050495
Altadill A, Eiro N, González LO, Andicoechea A, Fernández-Francos S, Rodrigo L, García-Muñiz JL, Vizoso FJ. Relationship between Metalloprotease-7 and -14 and Tissue Inhibitor of Metalloprotease 1 Expression by Mucosal Stromal Cells and Colorectal Cancer Development in Inflammatory Bowel Disease. Biomedicines. 2021; 9(5):495. https://doi.org/10.3390/biomedicines9050495
Chicago/Turabian StyleAltadill, Antonio, Noemi Eiro, Luis O. González, Alejandro Andicoechea, Silvia Fernández-Francos, Luis Rodrigo, José Luis García-Muñiz, and Francisco J. Vizoso. 2021. "Relationship between Metalloprotease-7 and -14 and Tissue Inhibitor of Metalloprotease 1 Expression by Mucosal Stromal Cells and Colorectal Cancer Development in Inflammatory Bowel Disease" Biomedicines 9, no. 5: 495. https://doi.org/10.3390/biomedicines9050495
APA StyleAltadill, A., Eiro, N., González, L. O., Andicoechea, A., Fernández-Francos, S., Rodrigo, L., García-Muñiz, J. L., & Vizoso, F. J. (2021). Relationship between Metalloprotease-7 and -14 and Tissue Inhibitor of Metalloprotease 1 Expression by Mucosal Stromal Cells and Colorectal Cancer Development in Inflammatory Bowel Disease. Biomedicines, 9(5), 495. https://doi.org/10.3390/biomedicines9050495







