Immunological Aspects Related to Viral Infections in Severe Asthma and the Role of Omalizumab
Abstract
1. Introduction
2. Methods
3. Immune Response to Respiratory Viruses and SARS-CoV-2
4. Severe Asthma and COVID-19: Epidemiological Data
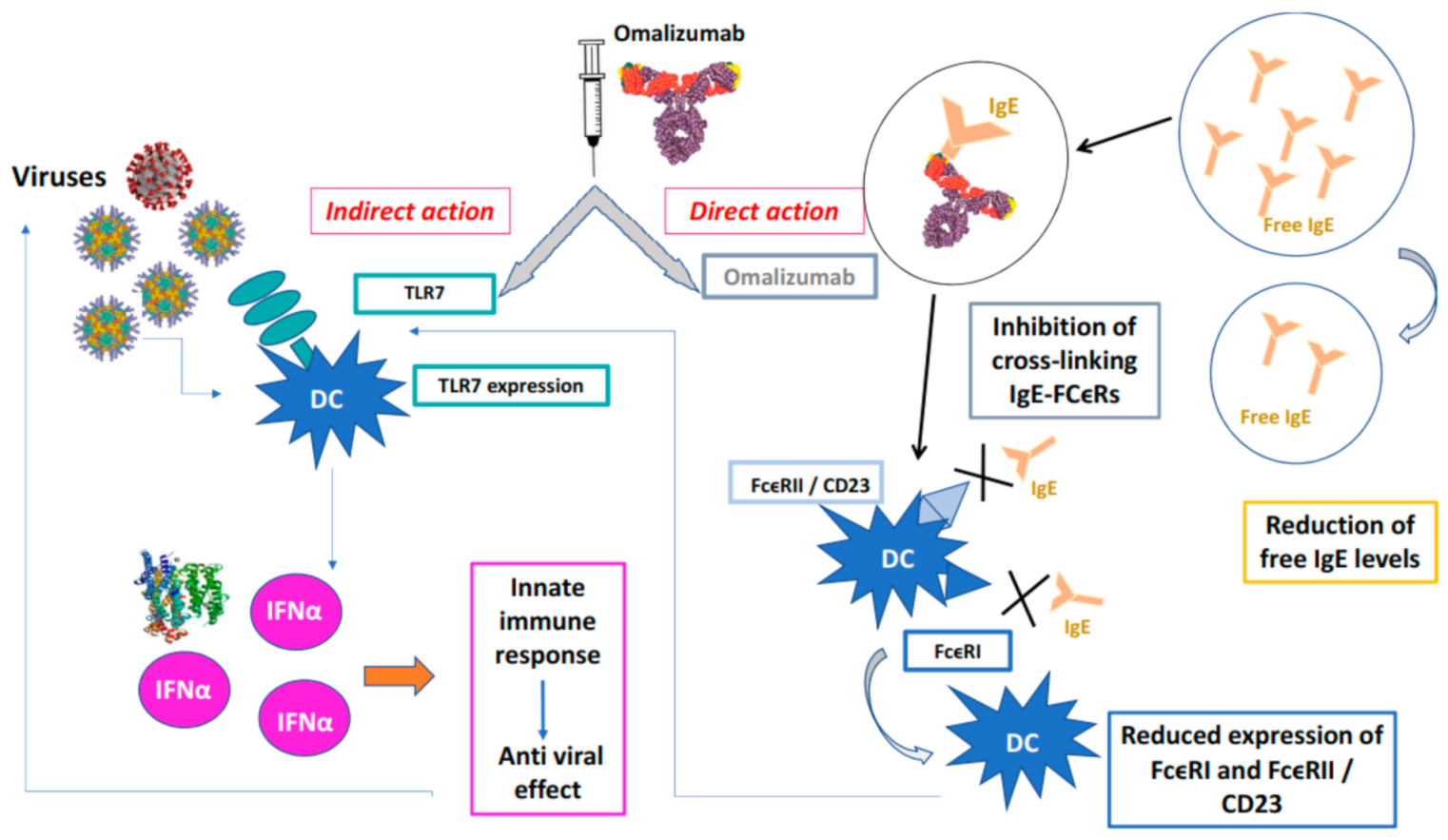
4.1. Impact of Monoclonal Antibodies for Severe Asthma on Viral Infections
4.2. Effect of Omalizumab in Patients with Severe Allergic Asthma and Respiratory Viral Infections
4.3. Omalizumab and Reduction of Virus-Induced Seasonal Exacerbations: Trial Data
4.3.1. Trial on Paediatric Patients: Focus on the PROSE Study
- 62% reduction in the overall time of taking salbutamol every 2 h (15 h vs. 30.8 h; p < 0.001).
- 42% reduction in hospital stay (34.5 h vs. 58.5 h; p < 0.001).
- It is the only pharmacological strategy for asthma or intervention with evidence of efficacy in reducing autumn exacerbations in children with allergic asthma [55].
- It is particularly effective in patients with severe allergic asthma, for whom there are limited therapeutic options and who are more exposed to the risk of exacerbations [55].
- There is no evidence that omalizumab is associated with adverse events greater than placebo, except for injection site reactions [56].
4.3.2. Trial on Adult and Adolescent Patients
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Busse, W.W.; Lemanske, R.F., Jr.; Gern, J.E. Role of viral respiratory infections in asthma and asthma exacerbations. Lancet 2010, 376, 826–834. [Google Scholar] [CrossRef]
- Global Initiative for Asthma. Global Strategy for Asthma Management and Prevention. 2020. Available online: www.ginasthma.org (accessed on 1 March 2021).
- Adeli, M.; El-Shareif, T.; Hendaus, M.A. Asthma exacerbation related to viral infections: An up to date summary. J. Fam. Med. Prim. Care 2019, 8, 2753–2759. [Google Scholar] [CrossRef] [PubMed]
- Cardet, J.C.; Casale, T.B. New insights into the utility of omalizumab. J. Allergy Clin. Immunol. 2019, 143, 923–926. [Google Scholar] [CrossRef] [PubMed]
- Gill, M.A.; Liu, A.H.; Calatroni, A.; Krouse, R.Z.; Shao, B.; Schiltz, A.; Gern, J.E.; Togias, A.; Busse, W.W. Enhanced plasmacytoid dendritic cell antiviral responses after omalizumab. J. Allergy Clin. Immunol. 2018, 14, 1735–1743. [Google Scholar] [CrossRef]
- Novak, N.; Cabanillas, B. Viruses and asthma: The role of common respiratory viruses in asthma and its potential meaning for SARS-CoV-2. Immunology 2020, 161, 83–93. [Google Scholar] [CrossRef]
- Corren, J.; Casale, T.; Haselkorn, T.; Yang, M.; Iqbal, A.; Ortiz, B.; Busse, W. Effect of omalizumab on seasonal exacerbations in adolescents and adults with moderate-to-severe allergic asthma. Ann. Allergy Asthma Immunol. Suppl. 2018, 121, 56–57. [Google Scholar] [CrossRef]
- Bai, T.R.; Vonk, J.M.; Postma, D.S.; Boezen, H.M. Severe exacerbations predict excess lung function decline in asthma. Eur. Respir. J. 2007, 30, 452–456. [Google Scholar] [CrossRef]
- World Health Organisation. Global Surveillance, Prevention and Control of Chronic Respiratory Diseases: A Comprehensive Approach. 2007. Available online: https://www.who.int/gard/publications/GARD_Manual/en/ (accessed on 3 March 2021).
- Pelaia, C.; Calabrese, C.; Terracciano, R.; de Blasio, F.; Vatrella, A.; Pelaia, G. Omalizumab, the first available antibody for biological treatment of severe asthma: More than a decade of real-life effectiveness. Ther. Adv. Respir. Dis. 2018, 12. [Google Scholar] [CrossRef]
- Channappanavar, R.; Fehr, A.R.; Vijay, R.; Mack, M.; Zhao, J.; Meyerholz, D.K.; Perlman, S. Dysregulated Type I interferon and inflammatory monocyte-macrophage responses cause lethal Pneumonia in SARS-CoV-Infected Mice. Cell Host Microbe 2016, 19, 181–193. [Google Scholar] [CrossRef]
- Gonzales-van Horn, S.R.; Farrar, J.D. Interferon at the crossroads of allergy and viral infections. J. Leukoc. Biol. 2015, 98, 185–194. [Google Scholar] [CrossRef]
- Gern, J.E. How rhinovirus infections cause exacerbations of asthma. Clin. Exp. Allergy 2015, 45, 32–42. [Google Scholar] [CrossRef]
- Teach, S.J.; Gill, M.A.; Togias, A.; Sorkness, C.A.; Arbes, S.J., Jr.; Calatroni, A.; Wildfire, J.J.; Gergen, P.J.; Cohen, R.T.; Pongracic, J.A.; et al. Preseasonal treatment with either omalizumab or an inhaled corticosteroid boost to prevent fall asthma exacerbations. J. Allergy Clin. Immunol. 2015, 136, 1476–1485. [Google Scholar] [CrossRef]
- Cameron, M.J.; Ran, L.; Xu, L.; Danesh, A.; Bermejo-Martin, J.F.; Cameron, C.M.; Muller, M.P.; Gold, W.L.; Richardson, S.E.; Poutanen, S.M.; et al. Interferon-mediated immunopathological events are associated with atypical innate and adaptive immune responses in patients with severe acute respiratory syndrome. J. Virol. 2007, 81, 8692–8706. [Google Scholar] [CrossRef]
- Carli, G.; Cecchi, L.; Stebbing, J.; Parronchi, P.; Farsi, A. Is asthma protective against COVID-19? Allergy 2021, 76, 866–868. [Google Scholar] [CrossRef]
- Huang, C.; Wang, Y.; Li, X.; Ren, L.; Zhao, J.; Hu, Y.; Zhang, L.; Fan, G.; Xu, J.; Gu, X.; et al. Clinical features of patients infected with 201nonovel coronavirus in Wuhan, China. Lancet 2020, 395, 497–506. [Google Scholar] [CrossRef]
- Zhang, Y.; Xiao, M.; Zhang, S.; Xia, P.; Cao, W.; Jiang, W.; Chen, H.; Ding, X.; Zhao, H.; Zhang, H.; et al. Coagulopathy and Antiphospholipid Antibodies in Patients with Covid-19. N. Engl. J. Med. 2020, 382, e38. [Google Scholar] [CrossRef]
- Zhang, J.J.; Dong, X.; Cao, Y.Y.; Yuan, Y.D.; Yang, Y.B.; Yan, Y.Q.; Akdis, C.A.; Gao, Y.D. Clinical characteristics of 140 patients infected with SARS-CoV-2 in Wuhan, China. Allergy 2020, 75, 1730–1741. [Google Scholar] [CrossRef]
- Dastoli, S.; Bennardo, L.; Patruno, C.; Nisticò, S.P. Are erythema multiforme and urticaria related to a better outcome of COVID-19? Dermatol. Ther. 2020, 33, e13681. [Google Scholar] [CrossRef]
- Flores-Torres, A.S.; Salinas-Carmona, M.C.; Salinas, E.; Rosas-Taraco, A.G. Eosinophils and respiratory viruses. Viral. Immunol. 2019, 32, 198–207. [Google Scholar] [CrossRef]
- Lindsley, A.W.; Schwartz, J.T.; Rothenberg, M.E. Eosinophil responses during COVID-19 infections and coronavirus vaccination. J. Allergy Clin. Immunol. 2020, 146. [Google Scholar] [CrossRef]
- Renner, A.; Marth, K.; Patocka, K.; Idzko, M.; Pohl, W. COVID-19 in two severe asthmatics receiving benralizumab: Busting the eosinophilia myth. ERJ Open Res. 2020, 6, 00457–2020. [Google Scholar] [CrossRef]
- Du, Y.; Tu, L.; Zhu, P.; Mu, M.; Wang, R.; Yang, P.; Wang, X.; Hu, C.; Ping, R.; Hu, P.; et al. Clinical features of 85 fatal cases of COVID-19 from Wuhan: A retrospective observational study. Am. J. Respir. Crit. Care Med. 2020, 201, 1372–1379. [Google Scholar] [CrossRef]
- Hassani, M.; Leijte, G.; Bruse, N.; Kox, M.; Pickkers, P.; Vrisekoop, N.; Koenderman, L. Differentiation and activation of eosinophils in the human bone marrow during experimental human endotoxemia. J. Leukoc. Biol. 2020, 108, 1665–1671. [Google Scholar] [CrossRef]
- Butterfield, J.H. Treatment of hypereosinophilic syndromes with prednisone, hydroxyurea, and interferon. Immunol. Allergy Clin. N. Am. 2007, 27, 493–518. [Google Scholar] [CrossRef]
- Matucci, A.; Caminati, M.; Vivarelli, E.; Vianello, A.; Micheletto, C.; Menzella, F.; Crisafulli, E.; Passalacqua, G.; Bagnasco, D.; Lombardi, C.; et al. COVID-19 in severe asthmatic patients during ongoing treatment with biologicals targeting type 2 inflammation: Results from a multicenter Italian survey. Allergy 2020. [Google Scholar] [CrossRef]
- Licari, A.; Castagnoli, R.; Votto, M.; Brambilla, I.; Ciprandi, G.; Marseglia, G.L. Biologic Use in Allergic and Asthmatic Children and Adolescents During the COVID-19 Pandemic. Pediatric Allergy Immunol. Pulmonol. 2020, 33, 155–158. [Google Scholar] [CrossRef]
- Criado, P.R.; Pagliari, C.; Criado, R.F.J.; Marques, G.F.; Belda, W., Jr. What the physicians should know about mast cells, dendritic cells, urticaria, and omalizumab during COVID-19 or asymptomatic infections due to SARS-CoV-2? Dermatol. Ther. 2020, 33, e14068. [Google Scholar] [CrossRef]
- Heffler, E.; Blasi, F.; Latorre, M.; Menzella, F.; Paggiaro, P.; Pelaia, G.; Senna, G.; Canonica, G.W. SANI Network. The Severe Asthma Network in Italy: Findings and Perspectives. J. Allergy Clin. Immunol. Pract. 2019, 7, 1462–1468. [Google Scholar] [CrossRef]
- Liu, S.; Zhi, Y.; Ying, S. COVID-19 and Asthma: Reflection During the Pandemic. Clin. Rev. Allerg. Immunol. 2020, 59, 78–88. [Google Scholar] [CrossRef]
- Morais-Almeida, M.; Aguiar, R.; Martin, B.; Ansotegui, I.J.; Ebisawa, M.; Arruda, L.K.; Caminati, M.; Canonica, G.W.; Carr, T.; Chupp, G.; et al. COVID-19, asthma, and biological therapies: What we need to know. World Allergy Organ. J. 2020, 13, 100126. [Google Scholar] [CrossRef]
- Riggioni, C.; Comberiati, P.; Giovannini, M.; Agache, I.; Akdis, M.; Alves-Correia, M.; Antó, J.M.; Arcolaci, A.; Azkur, A.K.; Azkur, D.; et al. A compendium answering 150 questions on COVID-19 and SARS-CoV-2. Allergy 2020, 75, 2503–2541. [Google Scholar] [CrossRef] [PubMed]
- Vultaggio, A.; Agache, I.; Akdis, C.A.; Akdis, M.; Bavbek, S.; Bossios, A.; Bousquet, J.; Boyman, O.; Chaker, A.M.; Chan, S.; et al. Considerations on biologicals for patients with allergic disease in times of the COVID-19 pandemic: An EAACI statement. Allergy 2020, 75, 2764–2774. [Google Scholar] [CrossRef] [PubMed]
- Piñeros, Y.S.S.; Bal, S.M.; van de Pol, M.A.; Dierdorp, B.S.; Dekker, T.; Dijkhuis, A.; Brinkman, P.; van der Sluijs, K.F.; Zwinderman, A.H.; Majoor, C.J.; et al. Anti-IL-5 in Mild Asthma Alters Rhinovirus-induced Macrophage, B-Cell, and Neutrophil Responses (MATERIAL). A Placebo-controlled, Double-Blind Study. Am. J. Respir. Crit. Care Med. 2019, 199, 508–517. [Google Scholar] [CrossRef] [PubMed]
- Renner, A.; Marth, K.; Patocka, K.; Pohl, W. COVID-19 in a severe eosinophilic asthmatic receiving benralizumab—A case study. J. Asthma 2020, 18. [Google Scholar] [CrossRef]
- Abdelmaksoud, A.; Goldust, M.; Vestita, M. Omalizumab and COVID19 Treatment: Could It Help? Dermatol. Ther. 2020, 33, e13792. [Google Scholar] [CrossRef]
- Lommatzsch, M.; Stoll, P.; Virchow, J.C. COVID-19 in a patient with severe asthma treated with Omalizumab. Allergy 2020, 75, 2705–2708. [Google Scholar] [CrossRef]
- Menzella, F.; Bertolini, F.; Biava, M.; Galeone, C.; Scelfo, C.; Caminati, M. Severe refractory asthma: Current treatment options and ongoing research. Drugs Context 2018, 7, 212561. [Google Scholar] [CrossRef]
- Pelaia, G.; Vatrella, A.; Busceti, M.T.; Gallelli, L.; Terracciano, R.; Maselli, R. Anti-IgE therapy with omalizumab for severe asthma: Current concepts and potential developments. Curr. Drug. Targets 2015, 16, 171–178. [Google Scholar] [CrossRef]
- Rodrigo, G.J.; Neffen, H.; Castro-Rodriguez, J.A. Efficacy and safety of subcutaneous omalizumab vs placebo as add-on therapy to corticosteroids for children and adults with asthma: A systematic review. Chest 2011, 139, 28–35. [Google Scholar] [CrossRef]
- Normansell, R.; Walker, S.; Milan, S.J.; Walters, E.H.; Nair, P. Omalizumab for asthma in adults and children. Cochrane Database Syst. Rev. 2014, 1, CD003559. [Google Scholar] [CrossRef]
- Tabatabaian, F.; Ledford, D.K. Omalizumab for severe asthma: Toward personalized treatment based on biomarker profile and clinical history. J. Asthma Allergy. 2018, 11, 53–61. [Google Scholar] [CrossRef]
- Cabrejos, S.; Moreira, A.; Ramirez, A.; Quirce, S.; Campos, G.S.; Davila, I.; Campo, P. FENOMA study: Achieving full control in patients with severe allergic asthma. J. Asthma Allergy 2020, 13, 159–166. [Google Scholar] [CrossRef]
- Menzella, F.; Galeone, C.; Formisano, D.; Castagnetti, C.; Ruggiero, P.; Simonazzi, A.; Zucchi, L. Real-life Efficacy of Omalizumab After 9 Years of Follow-up. Allergy Asthma Immunol. Res. 2017, 9, 368–372. [Google Scholar] [CrossRef][Green Version]
- Available online: https://www.ema.europa.eu/en/documents/product-information/xolair-epar-product-information_en.pdf (accessed on 1 March 2021).
- Esquivel, A.; Busse, W.W.; Calatroni, A.; Togias, A.G.; Grindle, K.G.; Bochkov, Y.A.; Gruchalla, R.S.; Kattan, M.; Kercsmar, C.M.; Hershey, G.K.; et al. Effects of Omalizumab on Rhinovirus Infections, Illnesses, and Exacerbations of Asthma. Am. J. Respir. Crit. Care Med. 2017, 196, 985–992. [Google Scholar] [CrossRef]
- Holguin, F.; Cardet, J.C.; Chung, K.F.; Diver, S.; Ferreira, D.S.; Fitzpatrick, A.; Gaga, M.; Kellermeyer, L.; Khurana, S.; Knight, S.; et al. Management of severe asthma: A European Respiratory Society/American Thoracic Society guideline. Eur. Respir. J. 2020, 55, 1900588.37. [Google Scholar] [CrossRef]
- Menzella, F.; Galeone, C.; Bertolini, F.; Castagnetti, C.; Facciolongo, N. Innovative treatments for severe refractory asthma: How to choose the right option for the right patient? J. Asthma Allergy 2017, 10, 237–247. [Google Scholar] [CrossRef]
- Casale, T.B.; Stokes, J. Anti-IgE therapy: Clinical utility beyond asthma. J. Allergy Clin. Immunol. 2009. [Google Scholar] [CrossRef]
- Kantor, D.B.; Stenquist, N.; McDonald, M.C.; Schultz, B.J.; Hauptman, M.; Smallwood, C.D.; Nelson, K.A.; Perzanowski, M.S.; Matsui, E.C.; Phipatanakul, W.; et al. Rhinovirus and serum IgE are associated with acute asthma exacerbation severity in children. J. Allergy Clin. Immunol. 2016, 138, 1467–1471. [Google Scholar] [CrossRef]
- López-Abente, J.; Benito-Villalvilla, C.; Jaumont, X.; Pfister, P.; Tassinari, P.; Palomares, O. Omalizumab restores the ability of human plasmacytoid dendritic cells to induce Foxp3+Tregs. Eur. Respir. J. 2021, 57, 2000751. [Google Scholar] [CrossRef]
- Yamaya, M. Virus infection-induced bronchial asthma exacerbation. Pulm. Med. 2012, 2012, 834826. [Google Scholar] [CrossRef]
- Efthimiou, J.; Poll, C.; Barnes, P.J. Dual mechanism of action of T2 inhibitor therapies in virally induced exacerbations of asthma: Evidence for a beneficial counter-regulation. Eur. Respir. J. 2019, 54, 1802390. [Google Scholar] [CrossRef] [PubMed]
- Holgate, S.; Casale, T.; Wenzel, S.; Bousquet, J.; Deniz, Y.; Reisner, C. The anti-inflammatory effects of omalizumab confirm the central role of IgE in allergic Inflammation. J. Allergy Clin. Immunol. 2005, 115, 459–465. [Google Scholar] [CrossRef] [PubMed]
- Leru, P.M. Biomarkers in asthma-interpretation and utily in current asthma management. Curr. Respir. Med. Rev. Spec. Issue 2021. [Google Scholar] [CrossRef]
- Kantor, D.B.; McDonald, M.C.; Stenquist, N.; Schultz, B.J.; Smallwood, C.D.; Nelson, K.A.; Phipatanakul, W.; Hirschhorn, J.N. Omalizumab Is Associated with Reduced Acute Severity of Rhinovirus-triggered Asthma Exacerbation. Am. J. Respir. Crit. Care Med. 2016, 194, 1552–1555. [Google Scholar] [CrossRef] [PubMed]
| Patient | Recommendation | Administration |
|---|---|---|
| Uninfected patients | Continued with terapy | Home-use |
| Infected patients | Maintained or temporarily suspended terapy | Home-use |
| Author | Study Population | Study Design | Observations/Results |
|---|---|---|---|
| Teach et al. [14] J. Allergy Clin. Immunol. 2015 | 453 asthmatic children aged 6 to 17 years with 1 or more recent exacerbations. | A 3-arm, randomized, double-blind, double placebo-controlled, multicenter clinical trial. | Adding omalizumab before return to school reduces fall asthma exacerbations, particularly among those with a recent exacerbation. |
| Novak, N et al. [6] Immunology. 2020 | 161 patients aged 6–17 years with allergic asthma. | A prospective, observational cohort study. | Treatment with omalizumab resulted in a greater reduction in the severity of HRV-induced exacerbations than in those treated with ICS. |
| Trial on adult and adolescent patients: | |||
| Wark, PAB et al. [52] Am. J Respir. Crit. Care Med. 2018 | 10 adult patients with severe allergic asthma not controlled by maximally dosed ICS/LABA therapy. | A prospective, observational cohort study. | Omalizumab treatment improves the innate antiviral response to influenza A and HRV by increasing the production of IFN-α and IFN-λ. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Menzella, F.; Ghidoni, G.; Galeone, C.; Capobelli, S.; Scelfo, C.; Facciolongo, N.C. Immunological Aspects Related to Viral Infections in Severe Asthma and the Role of Omalizumab. Biomedicines 2021, 9, 348. https://doi.org/10.3390/biomedicines9040348
Menzella F, Ghidoni G, Galeone C, Capobelli S, Scelfo C, Facciolongo NC. Immunological Aspects Related to Viral Infections in Severe Asthma and the Role of Omalizumab. Biomedicines. 2021; 9(4):348. https://doi.org/10.3390/biomedicines9040348
Chicago/Turabian StyleMenzella, Francesco, Giulia Ghidoni, Carla Galeone, Silvia Capobelli, Chiara Scelfo, and Nicola Cosimo Facciolongo. 2021. "Immunological Aspects Related to Viral Infections in Severe Asthma and the Role of Omalizumab" Biomedicines 9, no. 4: 348. https://doi.org/10.3390/biomedicines9040348
APA StyleMenzella, F., Ghidoni, G., Galeone, C., Capobelli, S., Scelfo, C., & Facciolongo, N. C. (2021). Immunological Aspects Related to Viral Infections in Severe Asthma and the Role of Omalizumab. Biomedicines, 9(4), 348. https://doi.org/10.3390/biomedicines9040348






