Brain Metabolic Correlates of Persistent Olfactory Dysfunction after SARS-Cov2 Infection
Abstract
1. Introduction
2. Material and Methods
2.1. Patients
2.2. Olfactory Test
2.3. [18F]-FDG Brain PET Acquisition and Image Processing
2.4. Voxel-Wise Analysis of Hypometabolic Signature of Olfactory Dysfunction after SARS-CoV-2 Infection
2.5. Structural Connectivity of Regions of Hypometabolism in Patients with Olfactory Dysfunction
3. Results
3.1. Patients
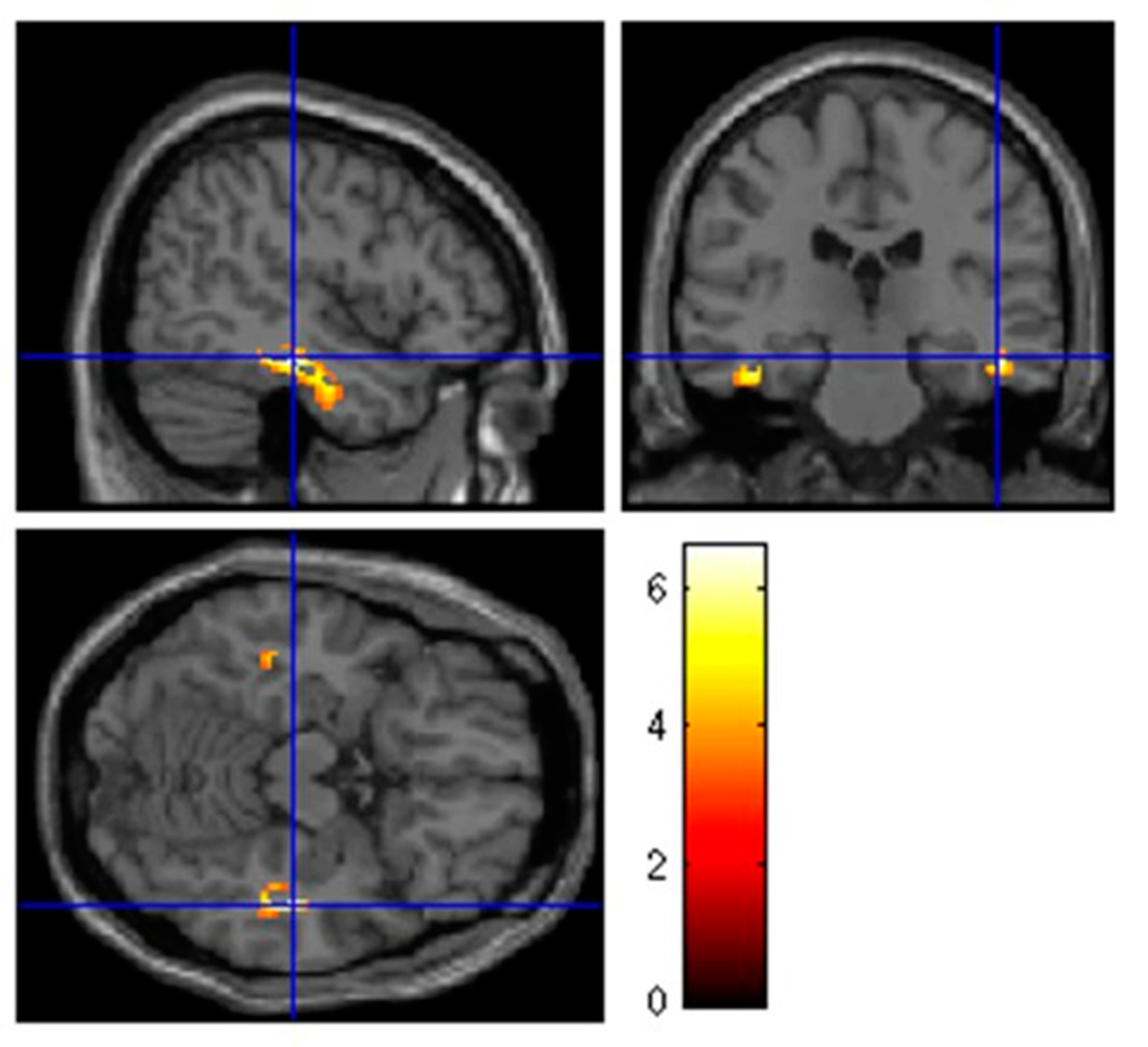
3.2. Hypometabolism in Patients with Isolated Persistent Hyposmia after SARS-CoV-2 Infection
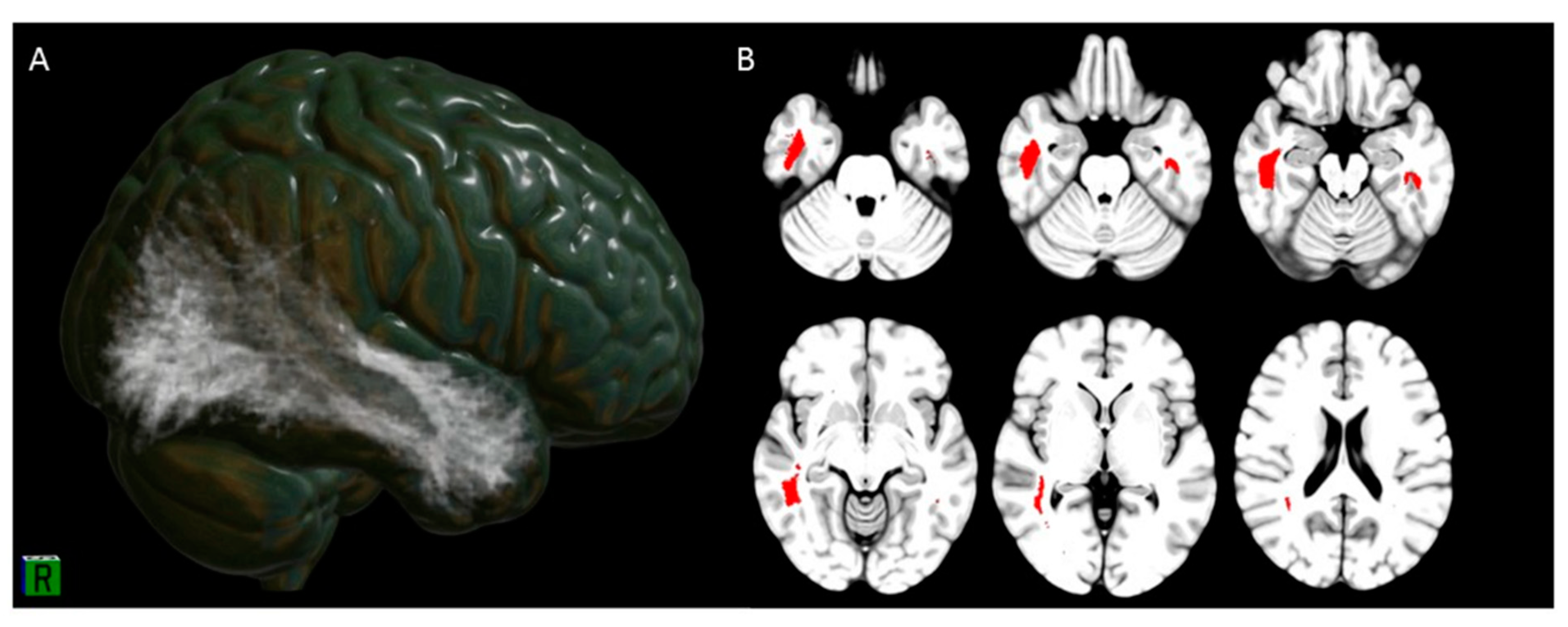
3.3. Hyposmia Clusters Tractography and Connectivity
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ellul, M.A.; Benjamin, L.; Singh, B.; Lant, S.; Michael, B.D.; Easton, A.; Kneen, R.; Defres, S.; Sejvar, J.; Solomon, T. Neurological associations of COVID-19. Lancet Neurol. 2020, 19, 767–783. [Google Scholar] [CrossRef]
- Lechien, J.R.; Chiesa-Estomba, C.M.; De Siati, D.R.; Horoi, M.; Le Bon, S.D.; Rodriguez, A.; Dequanter, D.; Blecic, S.; El Afia, F.; Distinguin, L.; et al. Olfactory and gustatory dysfunctions as a clinical presentation of mild-to-moderate forms of the coronavirus disease (COVID-19): A multicenter European study. Eur. Arch. Otorhinolaryngol. 2020, 277, 2251–2261. [Google Scholar] [CrossRef]
- Saussez, S.; Lechien, J.R.; Hopkins, C. Anosmia: An evolution of our understanding of its importance in COVID-19 and what questions remain to be answered. Eur. Arch. Otorhinolaryngol. 2020, 1–5. [Google Scholar] [CrossRef]
- Fotuhi, M.; Mian, A.; Meysami, S.; Raji, C.A. Neurobiology of COVID-19. J. Alzheimers. Dis. 2020, 76, 3–19. [Google Scholar] [CrossRef] [PubMed]
- Galougahi, M.K.; Ghorbani, J.; Bakhshayeshkaram, M.; Naeini, A.S.; Haseli, S. Olfactory Bulb Magnetic Resonance Imaging in SARS-CoV-2-Induced Anosmia: The First Report. Acad Radiol. 2020, 27, 892–893. [Google Scholar] [CrossRef] [PubMed]
- Politi, L.S.; Salsano, E.; Grimaldi, M. Magnetic Resonance Imaging Alteration of the Brain in a Patient With Coronavirus Disease 2019 (COVID-19) and Anosmia. JAMA Neurol. 2020, 77, 1028–1029. [Google Scholar] [CrossRef] [PubMed]
- Annunziata, S.; Bauckneht, M.; Albano, D.; Argiroffi, G.; Calabrò, D.; Abenavoli, E.; Linguanti, F.; Laudicella, R.; Young Committee of the Italian Association of Nuclear Medicine (AIMN). Impact of the COVID-19 pandemic in nuclear medicine departments: Preliminary report of the first international survey. Eur. J. Nucl. Med. Mol. Imaging 2020, 47, 2090–2099. [Google Scholar] [CrossRef] [PubMed]
- Guedj, E.; Million, M.; Dudouet, P.; Tissot-Dupont, H.; Bregeon, F.; Cammilleri, S.; Raoult, D. 18F-FDG brain PET hypometabolism in post-SARS-CoV-2 infection: Substrate for persistent/delayed disorders? Eur. J. Nucl. Med. Mol. Imaging 2020, 1–4. [Google Scholar] [CrossRef]
- Delorme, C.; Paccoud, O.; Kas, A.; Hesters, A.; Bombois, S.; Shambrook, P.; Boullet, A.; Doukhi, D.; Le Guennec, L.; Godefroy, N.; et al. COVID-19-related encephalopathy: A case series with brain FDG-positron-emission tomography/computed tomography findings. Eur. J. Neurol. 2020, 27, 2651–2657. [Google Scholar] [CrossRef]
- Karimi-Galougahi, M.; Yousefi-Koma, A.; Bakhshayeshkaram, M.; Raad, N.; Haseli, S. 18FDG PET/CT Scan Reveals Hypoactive Orbitofrontal Cortex in Anosmia of COVID-19. Acad Radiol. 2020, 27, 1042–1043. [Google Scholar] [CrossRef] [PubMed]
- Morbelli, S.; Ekmekcioglu, O.; Barthel, H.; Albert, N.L.; Boellaard, R.; Cecchin, D.; Guedj, E.; Lammertsma, A.A.; Law, I.; Penuelas, I.; et al. EANM Neuroimaging Committee. COVID-19 and the brain: Impact on nuclear medicine in neurology. Eur. J. Nucl. Med. Mol. Imaging 2020, 47, 2487–2492. [Google Scholar] [CrossRef] [PubMed]
- Meeting the challenge of long COVID. Nat. Med. 2020, 26, 1803. [CrossRef] [PubMed]
- The Lancet. Facing up to long COVID. Lancet 2020, 396, 1861. [Google Scholar] [CrossRef]
- Briner, H.R.; Simmen, D. Smell diskettes as screening test of olfaction. Rhinology 1999, 37, 145–148. [Google Scholar] [PubMed]
- Varrone, A.; Asenbaum, S.; Vander Borght, T.; Booij, J.; Nobili, F.; Någren, K.; Darcourt, J.; Kapucu, O.L.; Tatsch, K.; Bartenstein, P.; et al. EANM procedure guidelines for PET brain imaging using [18F]FDG, version 2. Eur. J. Nucl. Med. Mol. Imaging 2009, 36, 2103–2110. [Google Scholar] [CrossRef]
- Friston, K.J.; Holmes, A.P.; Worsley, K.J.; Poline, J.P.; Frith, C.D.; Frackowiak, R.S. Statistical parametric maps in functional imaging: A general linear approach. Hum. Brain Mapp. 1994, 2, 189–210. [Google Scholar] [CrossRef]
- Morbelli, S.; Bauckneht, M.; Arnaldi, D.; Picco, A.; Pardini, M.; Brugnolo, A.; Buschiazzo, A.; Pagani, M.; Girtler, N.; Nieri, A.; et al. 18F-FDG PET diagnostic and prognostic patterns do not overlap in Alzheimer’s disease (AD) patients at the mild cognitive impairment (MCI) stage. Eur. J. Nucl. Med. Mol. Imaging 2017, 44, 2073–2083. [Google Scholar] [CrossRef]
- Foulon, C.; Cerliani, L.; Kinkingnehun, S.; Levy, R.; Rosso, C.; Urbanski, M.; Volle, E.; Thiebaut de Schotten, M. Advanced lesion symptom mapping analyses and implementation as BCBtoolkit. Gigascience 2018, 7, 1–17. [Google Scholar] [CrossRef]
- Rojkova, K.; Volle, E.; Urbanski, M.; Humbert, F.; Dell’Acqua, F.; Thiebaut de Schotten, M. Atlasing the frontal lobe connections and their variability due to age and education: A spherical deconvolution tractography study. Brain Struct. Funct. 2016, 221, 1751–1766. [Google Scholar] [CrossRef]
- Thiebaut de Schotten, M.; Dell’Acqua, F.; Ratiu, P.; Leslie, A.; Howells, H.; Cabanis, E.; Iba-Zizen, M.T.; Plaisant, O.; Simmons, A.; Dronkers, N.F.; et al. From Phineas Gage and Monsieur Leborgne to H.M.: Revisiting Disconnection Syndromes. Cereb. Cortex. 2015, 25, 4812–4827. [Google Scholar] [CrossRef]
- Massa, F.; Grisanti, S.; Brugnolo, A.; Doglione, E.; Orso, B.; Morbelli, S.; Bauckneht, M.; Origone, P.; Filippi, L.; Arnaldi, D.; et al. The role of anterior prefrontal cortex in prospective memory: An exploratory FDG-PET study in early Alzheimer’s disease. Neurobiol. Aging 2020, 96, 117–127. [Google Scholar] [CrossRef]
- Meng, X.; Deng, Y.; Dai, Z.; Meng, Z. COVID-19 and anosmia: A review based on up-to-date knowledge. Am. J. Otolaryngol. 2020, 41, 102581. [Google Scholar] [CrossRef]
- Diodato, A.; De Brimont, M.R.; Yim, Y.S.; Derian, N.; Perrin, S.; Pouch, J.; Klatzmann, D.; Garel, S.; Choi, G.B.; Fleischmann, A. Molecular signatures of neural connectivity in the olfactory cortex. Nat. Commun. 2016, 7, 12238. [Google Scholar] [CrossRef]
- Antczak, J.; Popp, R.; Hajak, G.; Zulley, J.; Marienhagen, J.; Geisler, P. Positron emission tomography findings in obstructive sleep apnea patients with residual sleepiness treated with continuous positive airway pressure. J. Physiol. Pharmacol. 2007, 5 (Suppl. 5), 25–35. [Google Scholar]
- Kas, A.; Soret, M.; Pyatigoskaya, N.; Habert, M.O.; Hesters, A.; Le Guennec, L.; Paccoud, O.; Bombois, S.; Delorme, C.; on the behalf of CoCo-Neurosciences study group and COVID SMIT PSL study group. The cerebral network of COVID-19-related encephalopathy: A longitudinal voxel-based 18F-FDG-PET study. Eur. J. Nucl. Med. Mol. Imaging 2021, 15, 1–15. [Google Scholar]
- Guedj, E.; Campion, J.Y.; Dudouet, P.; Kaphan, E.; Bregeon, F.; Tissot-Dupont, H.; Guis, S.; Barthelemy, F.; Habert, P.; Ceccaldi, M.; et al. 18 F-FDG brain PET hypometabolism in patients with long COVID. Eur. J. Nucl. Med. Mol. Imaging 2021, 26, 1–11. [Google Scholar]
- Sollini, M.; Morbelli, S.; Ciccarelli, M.; Cecconi, M.; Aghemo, A.; Morelli, P.; Chiola, S.; Gelardi, F.; Chiti, A. Long Covid hallmarks on [18F]FDG-PET/CT: A case-control study. Eur. J. Nucl. Med. Mol. Imaging 2021, 7, 1–11. [Google Scholar] [CrossRef]
- Almeria, M.; Cejudo, J.C.; Sotoca, J.; Deus, J.; Krupinski, J. Cognitive profile following COVID-19 infection: Clinical predictors leading to neuropsychological impairment. Brain Behav. Immun. Health 2020, 100163. [Google Scholar] [CrossRef] [PubMed]
- Aqrabawi, A.J.; Kim, J.C. Olfactory memory representations are stored in the anterior olfactory nucleus. Nat. Commun. 2020, 11, 1246. [Google Scholar] [CrossRef] [PubMed]
- Christopher, L.; Koshimori, Y.; Lang, A.E.; Criaud, M.; Strafella, A.P. Uncovering the role of the insula in non-motor symptoms of Parkinson’s disease. Brain 2014, 137, 2143–2154. [Google Scholar] [CrossRef]
- Haghshomar, M.; Dolatshahi, M.; Ghazi Sherbaf, F.; Sanjari Moghaddam, H.; Shirin Shandiz, M.; Aarabi, M.H. Disruption of Inferior Longitudinal Fasciculus Microstructure in Parkinson’s Disease: A Systematic Review of Diffusion Tensor Imaging Studies. Front. Neurol. 2018, 26, 598. [Google Scholar] [CrossRef]
- Gongvatana, A.; Schweinsburg, B.C.; Taylor, M.J.; Theilmann, R.J.; Letendre, S.L.; Alhassoon, O.M.; Jacobus, J.; Woods, S.P.; Jernigan, T.L.; Ellis, R.J.; et al. White matter tract injury and cognitive impairment in human immunodeficiency virus-infected individuals. J. Neurovirol. 2009, 15, 187–195. [Google Scholar] [CrossRef] [PubMed]
- Bladowska, J.; Zimny, A.; Knysz, B.; Małyszczak, K.; Kołtowska, A.; Szewczyk, P.; Gąsiorowski, J.; Furdal, M.; Sąsiadek, M.J. Evaluation of early cerebral metabolic, perfusion and microstructural changes in HCV-positive patients: A pilot study. J. Hepatol. 2013, 59, 651–657. [Google Scholar] [CrossRef] [PubMed]
- Kandemirli, S.G.; Altundag, A.; Yildirim, D.; Tekcan Sanli, D.E.; Saatci, O. Olfactory Bulb MRI and Paranasal Sinus CT Findings in Persistent COVID-19 Anosmia. Acad Radiol. 2021, 28, 28–35. [Google Scholar] [CrossRef] [PubMed]
- Laurendon, T.; Radulesco, T.; Mugnier, J.; Gérault, M.; Chagnaud, C.; El Ahmadi, A.A.; Varoquaux, A. Bilateral transient olfactory bulb edema during COVID-19-related anosmia. Neurology 2020, 95, 224–225. [Google Scholar] [CrossRef] [PubMed]
- Magistretti, P.J.; Pellerin, L. Astrocytes Couple Synaptic Activity to Glucose Utilization in the Brain. News Physiol. Sci. 1999, 14, 177–182. [Google Scholar] [CrossRef]
- Passali, G.C.; Bentivoglio, A.R. Comment to the article “Olfactory and gustatory dysfunctions as a clinical presentation of mild-to-moderate forms of the coronavirus disease (COVID-19): A multicenter European study”. Eur. Arch. Otorhinolaryngol. 2020, 277, 2391–2392. [Google Scholar] [CrossRef] [PubMed]

| Characteristics | SARS-CoV-2 Patients with Hyposmia (n = 14) * |
|---|---|
| Age (years) | 64.4 ± 10.9 (range 51–79) |
| Sex | |
| Male | 7/14 |
| Female | 7/14 |
| Time Since Diagnosis of SARS-CoV-2 infection (weeks) † | 8.3 ± 2.1 (range 4–14) |
| Time Since first negative swab after proven SARS-CoV-2 infection (weeks) | 4.0 ± 1.9 (range 1–7) |
| Olfactory test (number of correct answers) | |
| 6/8 | 2 |
| 5/8 | 2 |
| 4/8 | 5 |
| 3/8 | 2 |
| 2/8 | 3 |
| Cluster Level | Peak Level | |||||||
|---|---|---|---|---|---|---|---|---|
| Cluster Extent | Corrected p-Value | Cortical Region | Maximum Zscore | Talairach Coordinates | Cortical Region | BA | ||
| 260 | 0.032 | |||||||
| R-limbic | 5.68 | 45 | −26 | −9 | Parahippocampal Gyrus | 36 | ||
| R-Temporal | 3.45 | 45 | −22 | −11 | Fusiform Gyrus | 20 | ||
| R-Temporal | 3.41 | 45 | −33 | −9 | Fusiform Gyrus | 37 | ||
| 155 | 0.034 | L-Limbic | 5.15 | −39 | −29 | −13 | Parahippocampal Gyrus | 36 |
| L-sublobar | 3.36 | −44 | −37 | 17 | Insula | 13 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Donegani, M.I.; Miceli, A.; Pardini, M.; Bauckneht, M.; Chiola, S.; Pennone, M.; Marini, C.; Massa, F.; Raffa, S.; Ferrarazzo, G.; et al. Brain Metabolic Correlates of Persistent Olfactory Dysfunction after SARS-Cov2 Infection. Biomedicines 2021, 9, 287. https://doi.org/10.3390/biomedicines9030287
Donegani MI, Miceli A, Pardini M, Bauckneht M, Chiola S, Pennone M, Marini C, Massa F, Raffa S, Ferrarazzo G, et al. Brain Metabolic Correlates of Persistent Olfactory Dysfunction after SARS-Cov2 Infection. Biomedicines. 2021; 9(3):287. https://doi.org/10.3390/biomedicines9030287
Chicago/Turabian StyleDonegani, Maria Isabella, Alberto Miceli, Matteo Pardini, Matteo Bauckneht, Silvia Chiola, Michele Pennone, Cecilia Marini, Federico Massa, Stefano Raffa, Giulia Ferrarazzo, and et al. 2021. "Brain Metabolic Correlates of Persistent Olfactory Dysfunction after SARS-Cov2 Infection" Biomedicines 9, no. 3: 287. https://doi.org/10.3390/biomedicines9030287
APA StyleDonegani, M. I., Miceli, A., Pardini, M., Bauckneht, M., Chiola, S., Pennone, M., Marini, C., Massa, F., Raffa, S., Ferrarazzo, G., Arnaldi, D., Sambuceti, G., Nobili, F., & Morbelli, S. (2021). Brain Metabolic Correlates of Persistent Olfactory Dysfunction after SARS-Cov2 Infection. Biomedicines, 9(3), 287. https://doi.org/10.3390/biomedicines9030287











