Cutaneous Squamous Cell Carcinoma: From Pathophysiology to Novel Therapeutic Approaches
Abstract
1. Introduction
2. Epidemiology
3. Risk Factors
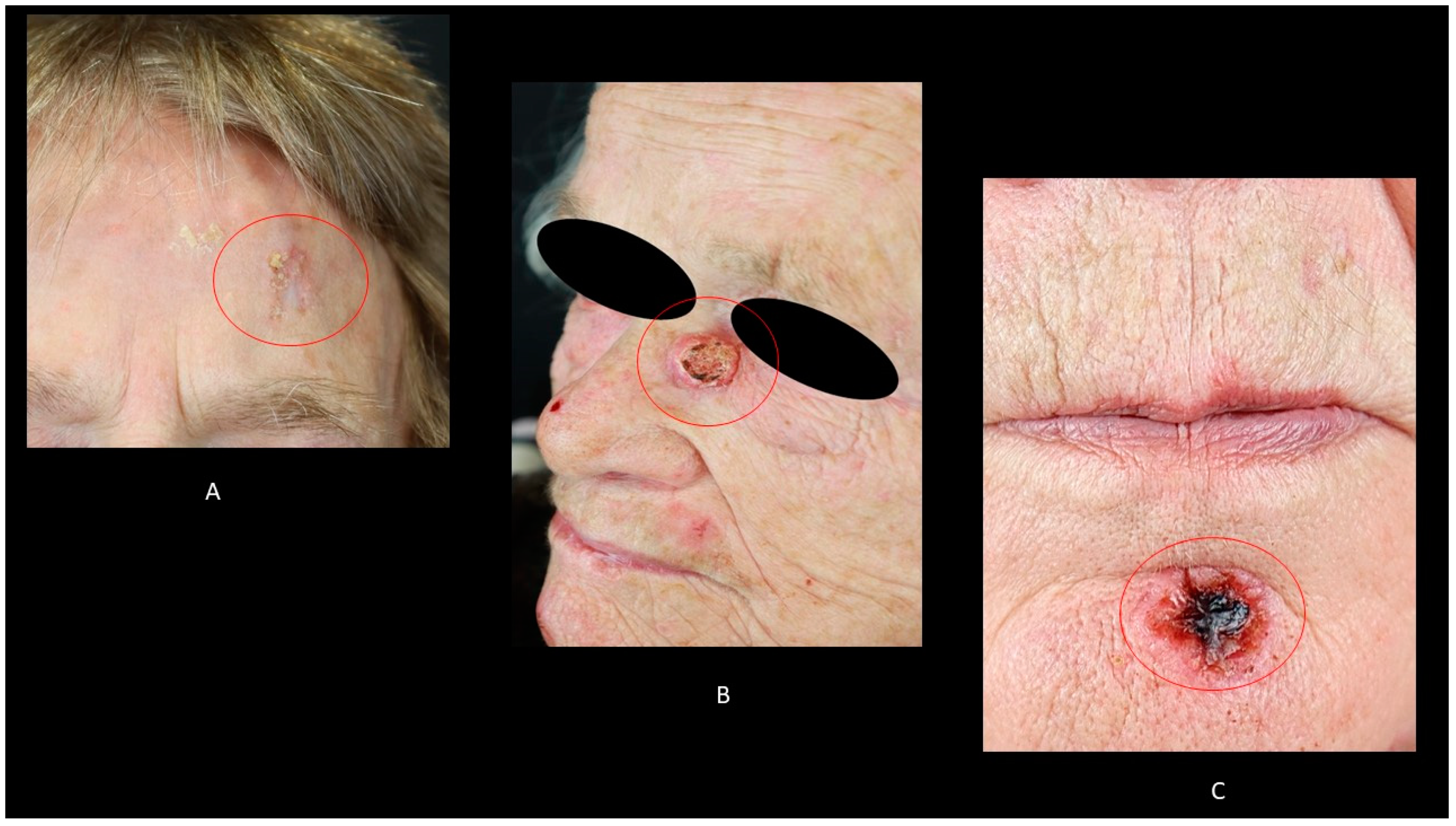
4. Clinical Features
4.1. Bowen’s Disease (BD)
4.2. Keratoacanthoma (KA)
4.3. Invasive cSCC
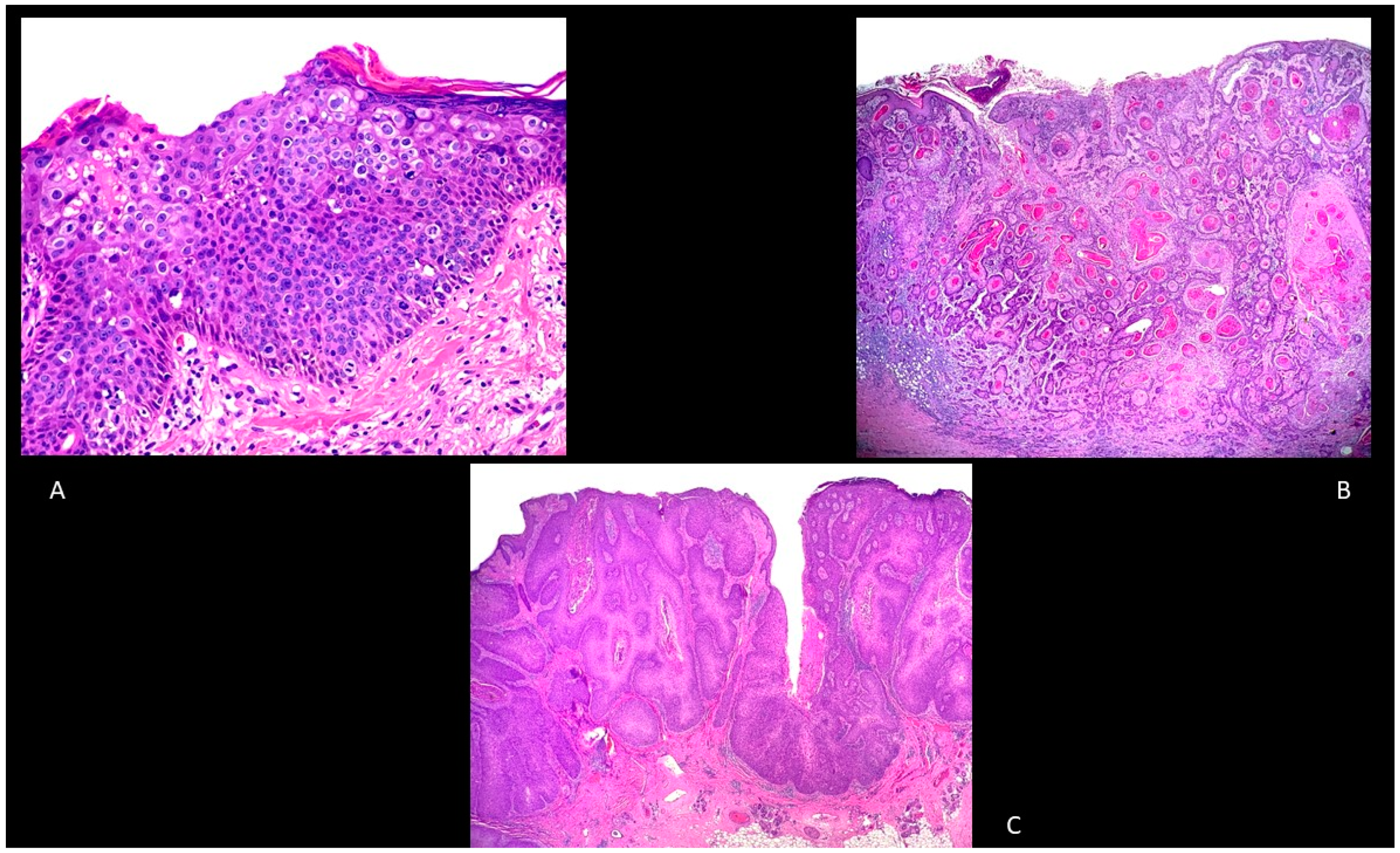
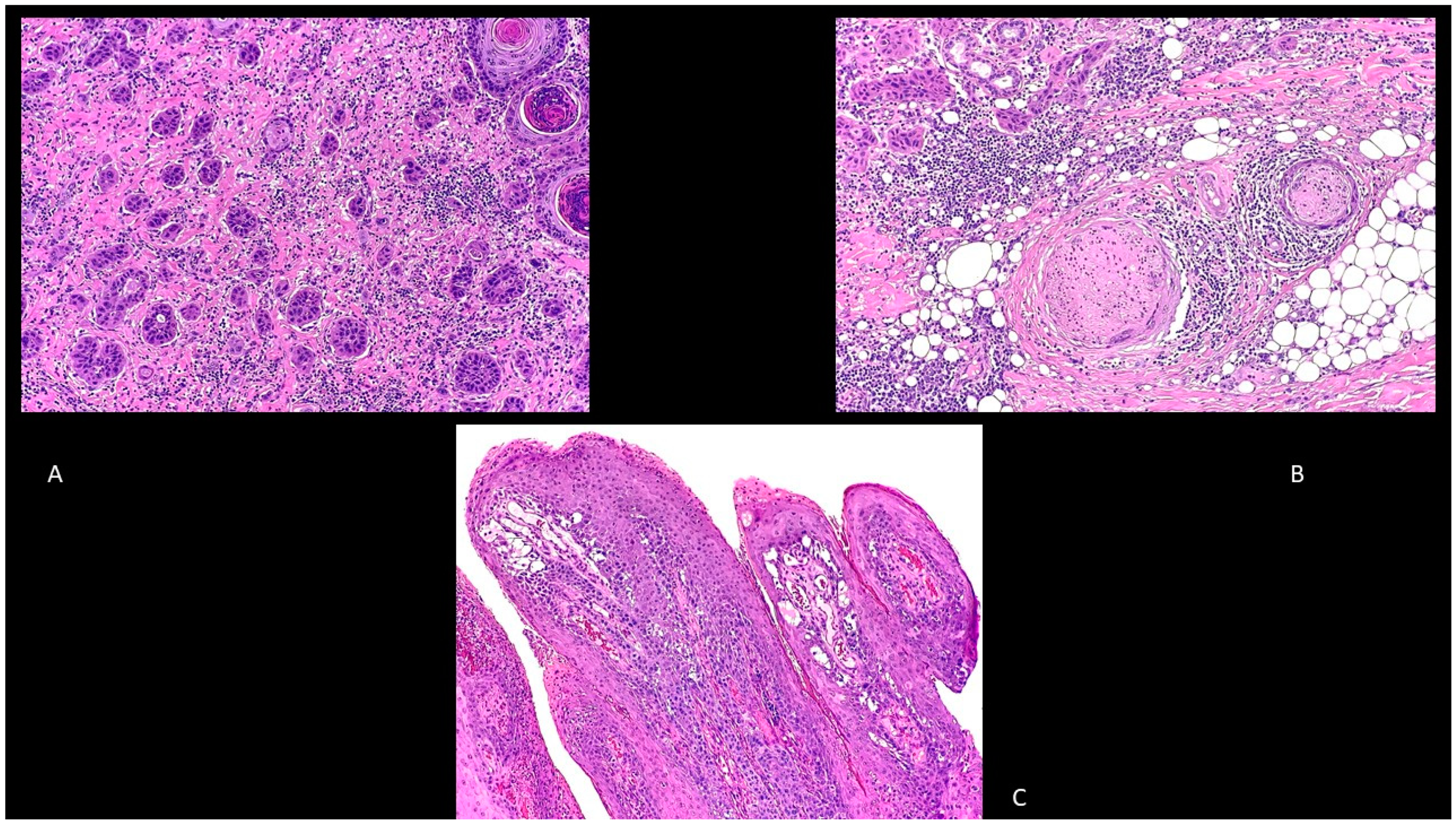
5. Histology
5.1. BD
5.2. Keratoacanthoma (KA)
5.3. Invasive cSCC
5.4. Other cSCC Variants
6. Dermoscopy
6.1. BD
6.2. Invasive cSCC
7. Reflectance Confocal Microscopy
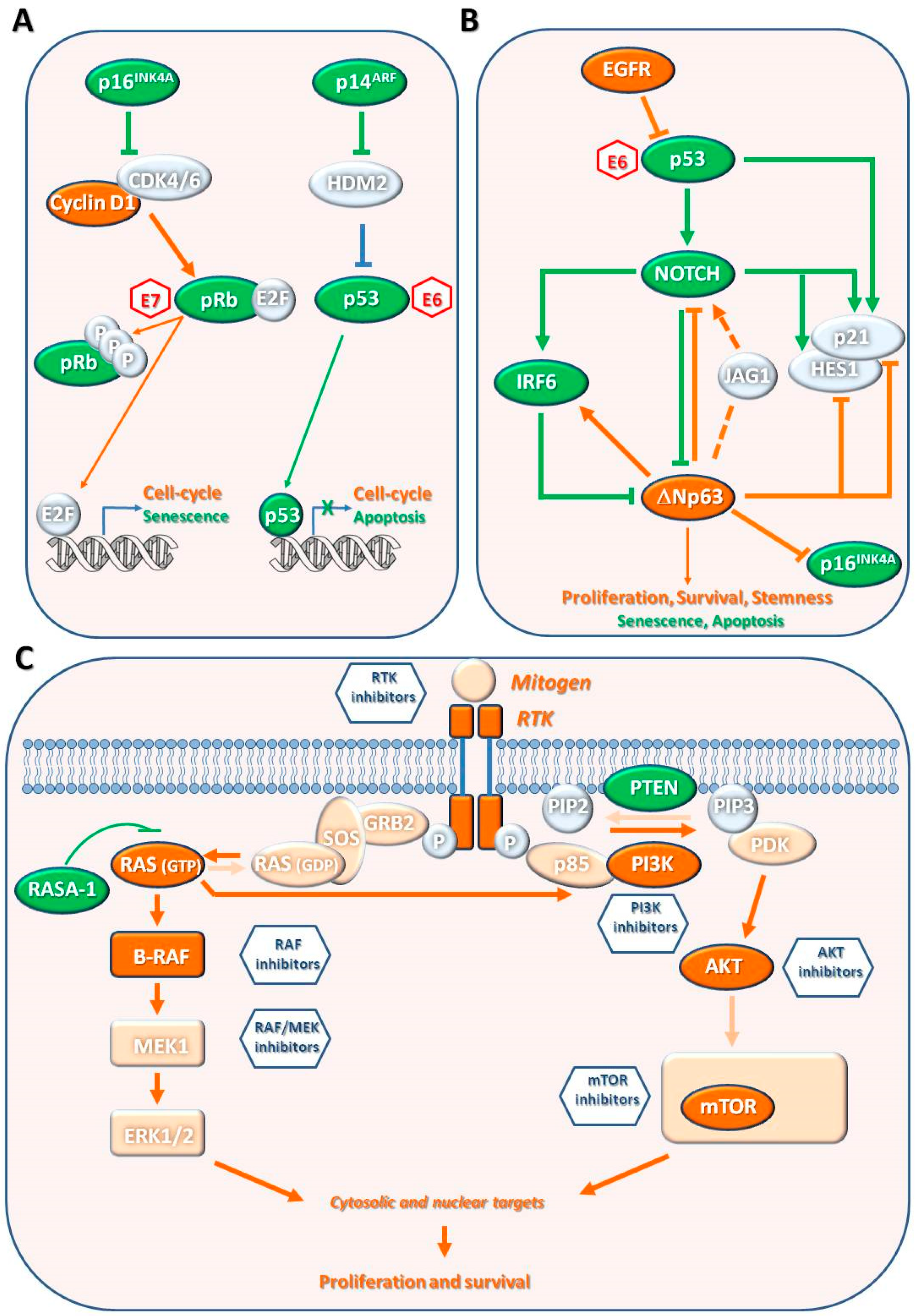
8. Pathogenesis of cSCC
8.1. Genetic Mutations
8.1.1. Cell Cycle Regulation, Apoptosis, and Senescence
TP53
CDKN2A Locus Gene and pRb Pathway
hTERT Promoter
8.1.2. Keratinocyte Differentiation
NOTCH1 and NOTCH2
TP63
RIPK4
8.1.3. Mitogenic/Survival Signaling Pathways
Epidermal Growth Factor Receptor
RAS-RAF-MEK-ERK Pathway
PI3K/AKT/mTOR Pathway
8.2. Epigenetic Modifications
8.3. Viral Pathogenesis
8.4. Tumor Microenvironment
9. Treatment of cSCC
9.1. Local Treatment
9.1.1. Surgery for cSCC
9.1.2. Standard Excision with Postoperative Margin Assessment
9.1.3. Micrographically Controlled Surgery
9.1.4. Surgery for Regional Nodal Disease
9.1.5. Treatment Alternatives to Surgery for Limited Cases of Low-Risk cSCC
Curettage and Electrodessication
Cryotherapy and PDT
Intralesional Cytostatic Drugs
9.2. Systemic Treatment, RT and Electrochemotherapy
9.2.1. Systemic Treatment
9.2.2. Radical RT
9.2.3. Adjuvant Therapy
9.2.4. Neoadjuvant Therapy
9.2.5. Electrochemotherapy
10. Prevention of cSCC
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Que, S.K.T.; Zwald, F.O.; Schmults, C.D. Cutaneous squamous cell carcinoma: Incidence, risk factors, diagnosis, and staging. J. Am. Acad. Dermatol. 2018, 78, 237–247. [Google Scholar] [CrossRef] [PubMed]
- Martincorena, I.; Roshan, A.; Gerstung, M.; Ellis, P.; van Loo, P.; McLaren, S.; Wedge, D.C.; Fullam, A.; Alexandrov, L.B.; Tubio, J.M.; et al. Tumor evolution. High burden and pervasive positive selection of somatic mutations in normal human skin. Science 2015, 348, 880–886. [Google Scholar] [CrossRef]
- Skulsky, S.L.; O’Sullivan, B.; McArdle, O.; Leader, M.; Roche, M.; Conlon, P.J.; O’Neill, J.P. Review of high-risk features of cutaneous squamous cell carcinoma and discrepancies between the American Joint Committee on Cancer and NCCN Clinical Practice Guidelines In Oncology. Head Neck 2017, 39, 578–594. [Google Scholar] [CrossRef]
- Kim, J.Y.S.; Kozlow, J.H.; Mittal, B.; Moyer, J.; Olenecki, T.; Rodgers, P. Guidelines of care for the management of cutaneous squamous cell carcinoma. J. Am. Acad. Dermatol. 2018, 78, 560–578. [Google Scholar] [CrossRef]
- Xiang, F.; Lucas, R.; Hales, S.; Neale, R. Incidence of nonmelanoma skin cancer in relation to ambient UV radiation in white populations, 1978-2012: Empirical relationships. JAMA Dermatol. 2014, 150, 1063–1071. [Google Scholar] [CrossRef]
- Brougham, N.D.L.S.; Dennett, E.R.; Cameron, R.; Tan, S.T. The incidence of metastasis from cutaneous squamous cell carcinoma and the impact of its risk factors. J. Surg. Oncol. 2012, 106, 811–815. [Google Scholar] [CrossRef] [PubMed]
- Nissinen, L.; Farshchian, M.; Riihilä, P.; Kähäri, V.-M. New perspectives on role of tumor microenvironment in progression of cutaneous squamous cell carcinoma. Cell Tissue Res. 2016, 365, 691–702. [Google Scholar] [CrossRef] [PubMed]
- Stratigos, A.J.; Garbe, C.; Dessinioti, C.; Lebbe, C.; Bataille, V.; Bastholt, L.; Dreno, B.; Concetta Fargnoli, M.; Forsea, A.M.; Frenard, C.; et al. European interdisciplinary guideline on invasive squamous cell carcinoma of the skin: Part 2. Treatment. Eur. J. Cancer 2020, 128, 83–102. [Google Scholar] [CrossRef] [PubMed]
- Trakatelli, M.; Ulrich, C.; Del Marmol, V.; Euvrard, S.; Euvard, S.; Stockfleth, E.; Abeni, D. Epidemiology of nonmelanoma skin cancer (NMSC) in Europe: Accurate and comparable data are needed for effective public health monitoring and interventions. Br. J. Dermatol. 2007, 156, 1–7. [Google Scholar] [CrossRef]
- Staples, M.P.; Elwood, M.; Burton, R.C.; Williams, J.L.; Marks, R.; Giles, G.G. Non-melanoma skin cancer in Australia: The 2002 national survey and trends since 1985. Med. J. Aust. 2006, 184, 6–10. [Google Scholar] [CrossRef] [PubMed]
- Andersson, E.M.; Paoli, J.; Wastensson, G. Incidence of cutaneous squamous cell carcinoma in coastal and inland areas of Western Sweden. Cancer Epidemiol. 2011, 35, e69–e74. [Google Scholar] [CrossRef]
- Brewster, D.H.; Bhatti, L.A.; Inglis, J.H.C.; Nairn, E.R.; Doherty, V.R. Recent trends in incidence of nonmelanoma skin cancers in the East of Scotland, 1992-2003. Br. J. Dermatol. 2007, 156, 1295–1300. [Google Scholar] [CrossRef] [PubMed]
- Katalinic, A.; Kunze, U.; Schäfer, T. Epidemiology of cutaneous melanoma and non-melanoma skin cancer in Schleswig-Holstein, Germany: Incidence, clinical subtypes, tumour stages and localization (epidemiology of skin cancer). Br. J. Dermatol. 2003, 149, 1200–1206. [Google Scholar] [CrossRef]
- Stratigos, A.J.; Garbe, C.; Dessinioti, C.; Lebbe, C.; Bataille, V.; Bastholt, L.; Dreno, B.; Fargnoli, M.C.; Forsea, A.M.; Frenard, C.; et al. European interdisciplinary guideline on invasive squamous cell carcinoma of the skin: Part 1. epidemiology, diagnostics and prevention. Eur. J. Cancer 2020, 128, 60–82. [Google Scholar] [CrossRef] [PubMed]
- Morris, K.L.; Luke, M.C.; Perna, F.M. Prevalence of Skin Cancer Examination among Users of Indoor Tanning Beds. JAMA Dermatol. 2018, 154, 840–842. [Google Scholar] [CrossRef] [PubMed]
- Green, A.C.; Olsen, C.M. Cutaneous squamous cell carcinoma: An epidemiological review. Br. J. Dermatol. 2017, 177, 373–381. [Google Scholar] [CrossRef] [PubMed]
- Fania, L.; Didona, D.; Morese, R.; Campana, I.; Coco, V.; Di Pietro, F.R.; Ricci, F.; Pallotta, S.; Candi, E.; Abeni, D.; et al. Basal Cell Carcinoma: From Pathophysiology to Novel Therapeutic Approaches. Biomedicines 2020, 8, 449. [Google Scholar] [CrossRef] [PubMed]
- Wehner, M.R.; Shive, M.L.; Chren, M.-M.; Han, J.; Qureshi, A.A.; Linos, E. Indoor tanning and non-melanoma skin cancer: Systematic review and meta-analysis. BMJ 2012, 345, e5909. [Google Scholar] [CrossRef] [PubMed]
- Stern, R.S. The risk of squamous cell and basal cell cancer associated with psoralen and ultraviolet a therapy: A 30-year prospective study. J. Am. Acad. Dermatol. 2012, 66, 553–562. [Google Scholar] [CrossRef]
- Chahoud, J.; Semaan, A.; Chen, Y.; Cao, M.; Rieber, A.G.; Rady, P.; Tyring, S.K. Association Between β-Genus Human Papillomavirus and Cutaneous Squamous Cell Carcinoma in Immunocompetent Individuals-A Meta-analysis. JAMA Dermatol. 2016, 152, 1354–1364. [Google Scholar] [CrossRef]
- Paradisi, A.; Waterboer, T.; Ricci, F.; Sampogna, F.; Pawlita, M.; Abeni, D. Concomitant seropositivity for HPV 16 and cutaneous HPV types increases the risk of recurrent squamous cell carcinoma of the skin. Eur. J. Dermatol. 2020, 30, 493–498. [Google Scholar] [CrossRef] [PubMed]
- Pirie, K.; Beral, V.; Heath, A.K.; Green, J.; Reeves, G.K.; Peto, R.; McBride, P.; Olsen, C.M.; Green, A.C. Heterogeneous relationships of squamous and basal cell carcinomas of the skin with smoking: The UK Million Women Study and meta-analysis of prospective studies. Br. J. Cancer 2018, 119, 114–120. [Google Scholar] [CrossRef] [PubMed]
- Dusingize, J.C.; Olsen, C.M.; Pandeya, N.P.; Subramaniam, P.; Thompson, B.S.; Neale, R.E.; Green, A.C.; Whiteman, D.C. Cigarette Smoking and the Risks of Basal Cell Carcinoma and Squamous Cell Carcinoma. J. Invest. Dermatol. 2017, 137, 1700–1708. [Google Scholar] [CrossRef] [PubMed]
- Garrett, G.L.; Blanc, P.D.; Boscardin, J.; Lloyd, A.A.; Ahmed, R.L.; Anthony, T.; Bibee, K.; Breithaupt, A.; Cannon, J.; Chen, A.; et al. Incidence of and Risk Factors for Skin Cancer in Organ Transplant Recipients in the United States. JAMA Dermatol. 2017, 153, 296–303. [Google Scholar] [CrossRef] [PubMed]
- Omland, S.H.; Ahlström, M.G.; Gerstoft, J.; Pedersen, G.; Mohey, R.; Pedersen, C.; Kronborg, G.; Larsen, C.S.; Kvinesdal, B.; Gniadecki, R.; et al. Risk of skin cancer in patients with HIV: A Danish nationwide cohort study. J. Am. Acad. Dermatol. 2018, 79, 689–695. [Google Scholar] [CrossRef] [PubMed]
- Omland, S.H.; Gniadecki, R.; Hædersdal, M.; Helweg-Larsen, J.; Omland, L.H. Skin Cancer Risk in Hematopoietic Stem-Cell Transplant Recipients Compared With Background Population and Renal Transplant Recipients: A Population-Based Cohort Study. JAMA Dermatol. 2016, 152, 177–183. [Google Scholar] [CrossRef]
- Fania, L.; Abeni, D.; Esposito, I.; Spagnoletti, G.; Citterio, F.; Romagnoli, J.; Castriota, M.; Ricci, F.; Moro, F.; Perino, F.; et al. Behavioral and demographic factors associated with occurrence of non-melanoma skin cancer in organ transplant recipients. G. Ital. Dermatol. Venereol. Organo Uff. Soc. Ital. Dermatol. Sifilogr. 2020, 155, 669–675. [Google Scholar] [CrossRef]
- Wu, J.H.; Cohen, D.N.; Rady, P.L.; Tyring, S.K. BRAF inhibitor-associated cutaneous squamous cell carcinoma: New mechanistic insight, emerging evidence for viral involvement and perspectives on clinical management. Br. J. Dermatol. 2017, 177, 914–923. [Google Scholar] [CrossRef] [PubMed]
- Peng, L.; Wang, Y.; Hong, Y.; Ye, X.; Shi, P.; Zhang, J.; Zhao, Q. Incidence and relative risk of cutaneous squamous cell carcinoma with single-agent BRAF inhibitor and dual BRAF/MEK inhibitors in cancer patients: A meta-analysis. Oncotarget 2017, 8, 83280–83291. [Google Scholar] [CrossRef]
- Nagarajan, P.; Asgari, M.M.; Green, A.C.; Guhan, S.M.; Arron, S.T.; Proby, C.M.; Rollison, D.E.; Harwood, C.A.; Toland, A.E. Keratinocyte Carcinomas: Current Concepts and Future Research Priorities. Clin. Cancer Res. 2019, 25, 2379–2391. [Google Scholar] [CrossRef]
- Mohan, S.V.; Chang, J.; Li, S.; Henry, A.S.; Wood, D.J.; Chang, A.L.S. Increased Risk of Cutaneous Squamous Cell Carcinoma After Vismodegib Therapy for Basal Cell Carcinoma. JAMA Dermatol. 2016, 152, 527–532. [Google Scholar] [CrossRef] [PubMed]
- Gandini, S.; Palli, D.; Spadola, G.; Bendinelli, B.; Cocorocchio, E.; Stanganelli, I.; Miligi, L.; Masala, G.; Caini, S. Anti-hypertensive drugs and skin cancer risk: A review of the literature and meta-analysis. Crit. Rev. Oncol. Hematol. 2018, 122, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Tang, H.; Fu, S.; Zhai, S.; Song, Y.; Asgari, M.M.; Han, J. Use of antihypertensive drugs and risk of keratinocyte carcinoma: A meta-analysis of observational studies. Pharmacoepidemiol. Drug Saf. 2018, 27, 279–288. [Google Scholar] [CrossRef] [PubMed]
- Flohil, S.C.; van der Leest, R.J.T.; Arends, L.R.; Vries, E.; Nijsten, T. Risk of subsequent cutaneous malignancy in patients with prior keratinocyte carcinoma: A systematic review and meta-analysis. Eur. J. Cancer 2013, 49, 2365–2375. [Google Scholar] [CrossRef] [PubMed]
- Ricci, F.; Paradisi, A.; Fania, L.; Pallotta, S.; Di Lella, G.; Sobrino, L.; Panebianco, A.; Abeni, D. High melanoma risk in non-melanoma skin cancer patients under age 40: A large retrospective cohort study. G. Ital. Dermatol. Venereol. Organo Uff. Soc. Ital. Dermatol. Sifilogr. 2019. [Google Scholar] [CrossRef]
- Ashford, B.G.; Clark, J.; Gupta, R.; Iyer, N.G.; Yu, B.; Ranson, M. Reviewing the genetic alterations in high-risk cutaneous squamous cell carcinoma: A search for prognostic markers and therapeutic targets. Head Neck 2017, 39, 1462–1469. [Google Scholar] [CrossRef]
- Kossard, S.; Rosen, R. Cutaneous Bowen’s disease. J. Am. Acad. Dermatol. 1992, 27, 406–410. [Google Scholar] [CrossRef]
- Kallini, J.R.; Hamed, N.; Khachemoune, A. Squamous cell carcinoma of the skin: Epidemiology, classification, management, and novel trends. Int. J. Dermatol. 2015, 54, 130–140. [Google Scholar] [CrossRef]
- Schwartz, R.A. Keratoacanthoma. J. Am. Acad. Dermatol. 1994, 30, 1–19. [Google Scholar] [CrossRef]
- Paolino, G.; Donati, M.; Didona, D.; Mercuri, S.R.; Cantisani, C. Histology of Non-Melanoma Skin Cancers: An Update. Biomedicines 2017, 5, 71. [Google Scholar] [CrossRef] [PubMed]
- Esther, R.J.; Lamps, L.; Schwartz, H.S. Marjolin ulcers: Secondary carcinomas in chronic wounds. J. South. Orthop. Assoc. 1999, 8, 181–187. [Google Scholar]
- Miller, S.J. The National Comprehensive Cancer Network (NCCN) guidelines of care for nonmelanoma skin cancers. Dermatol. Surg. 2000, 26, 289–292. [Google Scholar] [CrossRef]
- Kempf, W.; Hantschke, M.; Kutzner, H.; Burgdorf, W. Dermatopathologie; Springer: Berlin/Heidelberg, Germany, 2015; ISBN 9783662464083. [Google Scholar]
- Zalaudek, I.; Argenziano, G. Dermoscopy of actinic keratosis, intraepidermal carcinoma and squamous cell carcinoma. Curr. Probl. Dermatol. 2015, 46, 70–76. [Google Scholar] [CrossRef]
- Pan, Y.; Chamberlain, A.J.; Bailey, M.; Chong, A.H.; Haskett, M.; Kelly, J.W. Dermatoscopy aids in the diagnosis of the solitary red scaly patch or plaque-features distinguishing superficial basal cell carcinoma, intraepidermal carcinoma, and psoriasis. J. Am. Acad. Dermatol. 2008, 59, 268–274. [Google Scholar] [CrossRef] [PubMed]
- Zalaudek, I.; Argenziano, G.; Leinweber, B.; Citarella, L.; Hofmann-Wellenhof, R.; Malvehy, J.; Puig, S.; Pizzichetta, M.A.; Thomas, L.; Soyer, H.P.; et al. Dermoscopy of Bowen’s disease. Br. J. Dermatol. 2004, 150, 1112–1116. [Google Scholar] [CrossRef]
- Mun, J.-H.; Kim, S.-H.; Jung, D.-S.; Ko, H.-C.; Kwon, K.-S.; Kim, M.-B. Dermoscopic features of Bowen’s disease in Asians. J. Eur. Acad. Dermatol. Venereol. 2010, 24, 805–810. [Google Scholar] [CrossRef] [PubMed]
- Payapvipapong, K.; Tanaka, M. Dermoscopic classification of Bowen’s disease. Australas. J. Dermatol. 2015, 56, 32–35. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Lin, J.; Fang, S.; Han, S.; Song, Z. What’s new in dermoscopy of Bowen’s disease: Two new dermoscopic signs and its differential diagnosis. Int. J. Dermatol. 2017, 56, 1022–1025. [Google Scholar] [CrossRef]
- Zalaudek, I.; Giacomel, J.; Schmid, K.; Bondino, S.; Rosendahl, C.; Cavicchini, S.; Tourlaki, A.; Gasparini, S.; Bourne, P.; Keir, J.; et al. Dermatoscopy of facial actinic keratosis, intraepidermal carcinoma, and invasive squamous cell carcinoma: A progression model. J. Am. Acad. Dermatol. 2012, 66, 589–597. [Google Scholar] [CrossRef] [PubMed]
- Rosendahl, C.; Cameron, A.; Argenziano, G.; Zalaudek, I.; Tschandl, P.; Kittler, H. Dermoscopy of squamous cell carcinoma and keratoacanthoma. Arch. Dermatol. 2012, 148, 1386–1392. [Google Scholar] [CrossRef] [PubMed]
- Pyne, J.; Sapkota, D.; Wong, J.C. Squamous cell carcinoma: Variation in dermatoscopic vascular features between well and non-well differentiated tumors. Dermatol. Pract. Concept. 2012, 2, 204a05. [Google Scholar] [CrossRef]
- Yélamos, O.; Braun, R.P.; Liopyris, K.; Wolner, Z.J.; Kerl, K.; Gerami, P.; Marghoob, A.A. Dermoscopy and dermatopathology correlates of cutaneous neoplasms. J. Am. Acad. Dermatol. 2019, 80, 341–363. [Google Scholar] [CrossRef] [PubMed]
- Manfredini, M.; Longo, C.; Ferrari, B.; Piana, S.; Benati, E.; Casari, A.; Pellacani, G.; Moscarella, E. Dermoscopic and reflectance confocal microscopy features of cutaneous squamous cell carcinoma. J. Eur. Acad. Dermatol. Venereol. 2017, 31, 1828–1833. [Google Scholar] [CrossRef] [PubMed]
- Lallas, A.; Pyne, J.; Kyrgidis, A.; Andreani, S.; Argenziano, G.; Cavaller, A.; Giacomel, J.; Longo, C.; Malvestiti, A.; Moscarella, E.; et al. The clinical and dermoscopic features of invasive cutaneous squamous cell carcinoma depend on the histopathological grade of differentiation. Br. J. Dermatol. 2015, 172, 1308–1315. [Google Scholar] [CrossRef] [PubMed]
- Rishpon, A.; Kim, N.; Scope, A.; Porges, L.; Oliviero, M.C.; Braun, R.P.; Marghoob, A.A.; Fox, C.A.; Rabinovitz, H.S. Reflectance confocal microscopy criteria for squamous cell carcinomas and actinic keratoses. Arch. Dermatol. 2009, 145, 766–772. [Google Scholar] [CrossRef] [PubMed]
- Aghassi, D.; Anderson, R.R.; González, S. Confocal laser microscopic imaging of actinic keratoses in vivo: A preliminary report. J. Am. Acad. Dermatol. 2000, 43, 42–48. [Google Scholar] [CrossRef]
- Incel, P.; Gurel, M.S.; Erdemir, A.V. Vascular patterns of nonpigmented tumoral skin lesions: Confocal perspectives. Skin Res. Technol. Off. J. Int. Soc. Bioeng. Skin Int. Soc. Dig. Imag. Skin Int. Soc. Skin Imag. 2015, 21, 333–339. [Google Scholar] [CrossRef]
- Peppelman, M.; Nguyen, K.P.; Hoogedoorn, L.; van Erp, P.E.J.; Gerritsen, M.-J.P. Reflectance confocal microscopy: Non-invasive distinction between actinic keratosis and squamous cell carcinoma. J. Eur. Acad. Dermatol. Venereol. 2015, 29, 1302–1309. [Google Scholar] [CrossRef] [PubMed]
- Ahlgrimm-Siess, V.; Cao, T.; Oliviero, M.; Hofmann-Wellenhof, R.; Rabinovitz, H.S.; Scope, A. The vasculature of nonmelanocytic skin tumors on reflectance confocal microscopy: Vascular features of squamous cell carcinoma in situ. Arch. Dermatol. 2011, 147, 264. [Google Scholar] [CrossRef]
- Skobe, M.; Rockwell, P.; Goldstein, N.; Vosseler, S.; Fusenig, N.E. Halting angiogenesis suppresses carcinoma cell invasion. Nat. Med. 1997, 3, 1222–1227. [Google Scholar] [CrossRef]
- Nguyen, K.P.; Peppelman, M.; Hoogedoorn, L.; van Erp, P.E.J.; Gerritsen, M.-J.P. The current role of in vivo reflectance confocal microscopy within the continuum of actinic keratosis and squamous cell carcinoma: A systematic review. Eur. J. Dermatol. 2016, 26, 549–565. [Google Scholar] [CrossRef] [PubMed]
- Corchado-Cobos, R.; García-Sancha, N.; González-Sarmiento, R.; Pérez-Losada, J.; Cañueto, J. Cutaneous Squamous Cell Carcinoma: From Biology to Therapy. Int. J. Mol. Sci. 2020, 21, 2956. [Google Scholar] [CrossRef] [PubMed]
- Lane, D.P. Cancer. p53, guardian of the genome. Nature 1992, 358, 15–16. [Google Scholar] [CrossRef] [PubMed]
- Campos, M.A.; Lopes, J.M.; Soares, P. The genetics of cutaneous squamous cell carcinogenesis. Eur. J. Dermatol. 2018, 28, 597–605. [Google Scholar] [CrossRef] [PubMed]
- Di Nardo, L.; Pellegrini, C.; Di Stefani, A.; Del Regno, L.; Sollena, P.; Piccerillo, A.; Longo, C.; Garbe, C.; Fargnoli, M.C.; Peris, K. Molecular genetics of cutaneous squamous cell carcinoma: Perspective for treatment strategies. J. Eur. Acad. Dermatol. Venereol. 2020, 34, 932–941. [Google Scholar] [CrossRef] [PubMed]
- Li, G.; Tron, V.; Ho, V. Induction of squamous cell carcinoma in p53-deficient mice after ultraviolet irradiation. J. Invest. Dermatol. 1998, 110, 72–75. [Google Scholar] [CrossRef] [PubMed]
- Brash, D.E.; Rudolph, J.A.; Simon, J.A.; Lin, A.; McKenna, G.J.; Baden, H.P.; Halperin, A.J.; Pontén, J. A role for sunlight in skin cancer: UV-induced p53 mutations in squamous cell carcinoma. Proc. Natl. Acad. Sci. USA 1991, 88, 10124–10128. [Google Scholar] [CrossRef] [PubMed]
- Ziegler, A.; Jonason, A.S.; Leffell, D.J.; Simon, J.A.; Sharma, H.W.; Kimmelman, J.; Remington, L.; Jacks, T.; Brash, D.E. Sunburn and p53 in the onset of skin cancer. Nature 1994, 372, 773–776. [Google Scholar] [CrossRef]
- Brash, D.E.; Ziegler, A.; Jonason, A.S.; Simon, J.A.; Kunala, S.; Leffell, D.J. Sunlight and sunburn in human skin cancer: p53, apoptosis, and tumor promotion. J. Investig. Dermatol. Symp. Proc. 1996, 1, 136–142. [Google Scholar] [PubMed]
- Li, Y.Y.; Hanna, G.J.; Laga, A.C.; Haddad, R.I.; Lorch, J.H.; Hammerman, P.S. Genomic analysis of metastatic cutaneous squamous cell carcinoma. Clin. Cancer Res. 2015, 21, 1447–1456. [Google Scholar] [CrossRef]
- Yilmaz, A.S.; Ozer, H.G.; Gillespie, J.L.; Allain, D.C.; Bernhardt, M.N.; Furlan, K.C.; Castro, L.T.F.; Peters, S.B.; Nagarajan, P.; Kang, S.Y.; et al. Differential mutation frequencies in metastatic cutaneous squamous cell carcinomas versus primary tumors. Cancer 2017, 123, 1184–1193. [Google Scholar] [CrossRef]
- D’Arcangelo, D.; Tinaburri, L.; Dellambra, E. The Role of p16INK4a Pathway in Human Epidermal Stem Cell Self-Renewal, Aging and Cancer. Int. J. Mol. Sci. 2017, 18, 1591. [Google Scholar] [CrossRef]
- Sherr, C.J. The INK4a/ARF network in tumour suppression. Nat. Rev. Mol. Cell Biol. 2001, 2, 731–737. [Google Scholar] [CrossRef] [PubMed]
- Brown, V.L.; Harwood, C.A.; Crook, T.; Cronin, J.G.; Kelsell, D.P.; Proby, C.M. p16INK4a and p14ARF tumor suppressor genes are commonly inactivated in cutaneous squamous cell carcinoma. J. Invest. Dermatol. 2004, 122, 1284–1292. [Google Scholar] [CrossRef] [PubMed]
- Inman, G.J.; Wang, J.; Nagano, A.; Alexandrov, L.B.; Purdie, K.J.; Taylor, R.G.; Sherwood, V.; Thomson, J.; Hogan, S.; Spender, L.C.; et al. The genomic landscape of cutaneous SCC reveals drivers and a novel azathioprine associated mutational signature. Nat. Commun. 2018, 9, 3667. [Google Scholar] [CrossRef] [PubMed]
- Küsters-Vandevelde, H.V.N.; van Leeuwen, A.; Verdijk, M.A.J.; de Koning, M.N.C.; Quint, W.G.V.; Melchers, W.J.G.; Ligtenberg, M.J.L.; Blokx, W.A.M. CDKN2A but not TP53 mutations nor HPV presence predict poor outcome in metastatic squamous cell carcinoma of the skin. Int. J. Cancer 2010, 126, 2123–2132. [Google Scholar] [CrossRef] [PubMed]
- Bito, T.; Ueda, M.; Ahmed, N.U.; Nagano, T.; Ichihashi, M. Cyclin D and retinoblastoma gene product expression in actinic keratosis and cutaneous squamous cell carcinoma in relation to p53 expression. J. Cutan. Pathol. 1995, 22, 427–434. [Google Scholar] [CrossRef]
- Shen, Y.; Xu, J.; Jin, J.; Tang, H.; Liang, J. Cyclin D1 expression in Bowen’s disease and cutaneous squamous cell carcinoma. Mol. Clin. Oncol. 2014, 2, 545–548. [Google Scholar] [CrossRef] [PubMed]
- Huang, K.; Huang, C.; Shan, K.; Chen, J.; Li, H. Significance of PC cell-derived growth factor and cyclin D1 expression in cutaneous squamous cell carcinoma. Clin. Exp. Dermatol. 2012, 37, 411–417. [Google Scholar] [CrossRef] [PubMed]
- Saawarn, S.; Astekar, M.; Saawarn, N.; Dhakar, N.; Gomateshwar Sagari, S. Cyclin d1 expression and its correlation with histopathological differentiation in oral squamous cell carcinoma. Sci. World J. 2012, 2012, 978327. [Google Scholar] [CrossRef]
- Liang, S.B.; Furihata, M.; Takeuchi, T.; Iwata, J.; Chen, B.K.; Sonobe, H.; Ohtsuki, Y. Overexpression of cyclin D1 in nonmelanocytic skin cancer. Virchows Arch. 2000, 436, 370–376. [Google Scholar] [CrossRef]
- Chiu, C.P.; Harley, C.B. Replicative senescence and cell immortality: The role of telomeres and telomerase. Proceedings of the Society for Experimental Biology and Medicine. Soc. Exp. Biol. Med. 1997, 214, 99–106. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.K.; Shaffer, P.M.; Slaughter, R.S.; McCroskey, R.P.; Abbott, M.T. Stoichiometry of the pyrimidine deoxyribonucleoside 2’-hydroxylase reaction and of the conversions of 5-hydroxymethyluracil to 5-formyluracil and of the latter to uracil-5-carboxylic acid. Biochemistry 1972, 11, 2172–2176. [Google Scholar] [CrossRef] [PubMed]
- Scott, G.A.; Laughlin, T.S.; Rothberg, P.G. Mutations of the TERT promoter are common in basal cell carcinoma and squamous cell carcinoma. Mod. Pathol. 2014, 27, 516–523. [Google Scholar] [CrossRef]
- Lefort, K.; Mandinova, A.; Ostano, P.; Kolev, V.; Calpini, V.; Kolfschoten, I.; Devgan, V.; Lieb, J.; Raffoul, W.; Hohl, D.; et al. Notch1 is a p53 target gene involved in human keratinocyte tumor suppression through negative regulation of ROCK1/2 and MRCKalpha kinases. Genes Dev. 2007, 21, 562–577. [Google Scholar] [CrossRef] [PubMed]
- Moretti, F.; Marinari, B.; Lo Iacono, N.; Botti, E.; Giunta, A.; Spallone, G.; Garaffo, G.; Vernersson-Lindahl, E.; Merlo, G.; Mills, A.A.; et al. A regulatory feedback loop involving p63 and IRF6 links the pathogenesis of 2 genetically different human ectodermal dysplasias. J. Clin. Invest. 2010, 120, 1570–1577. [Google Scholar] [CrossRef] [PubMed]
- Stransky, N.; Egloff, A.M.; Tward, A.D.; Kostic, A.D.; Cibulskis, K.; Sivachenko, A.; Kryukov, G.V.; Lawrence, M.S.; Sougnez, C.; McKenna, A.; et al. The mutational landscape of head and neck squamous cell carcinoma. Science 2011, 333, 1157–1160. [Google Scholar] [CrossRef] [PubMed]
- Nicolas, M.; Wolfer, A.; Raj, K.; Kummer, J.A.; Mill, P.; van Noort, M.; Hui, C.; Clevers, H.; Dotto, G.P.; Radtke, F. Notch1 functions as a tumor suppressor in mouse skin. Nat. Genet. 2003, 33, 416–421. [Google Scholar] [CrossRef] [PubMed]
- Wang, N.J.; Sanborn, Z.; Arnett, K.L.; Bayston, L.J.; Liao, W.; Proby, C.M.; Leigh, I.M.; Collisson, E.A.; Gordon, P.B.; Jakkula, L.; et al. Loss-of-function mutations in Notch receptors in cutaneous and lung squamous cell carcinoma. Proc. Natl. Acad. Sci. USA 2011, 108, 17761–17766. [Google Scholar] [CrossRef] [PubMed]
- South, A.P.; Purdie, K.J.; Watt, S.A.; Haldenby, S.; den Breems, N.; Dimon, M.; Arron, S.T.; Kluk, M.J.; Aster, J.C.; McHugh, A.; et al. NOTCH1 mutations occur early during cutaneous squamous cell carcinogenesis. J. Invest. Dermatol. 2014, 134, 2630–2638. [Google Scholar] [CrossRef]
- Smirnov, A.; Anemona, L.; Novelli, F.; Piro, C.M.; Annicchiarico-Petruzzelli, M.; Melino, G.; Candi, E. p63 Is a Promising Marker in the Diagnosis of Unusual Skin Cancer. Int. J. Mol. Sci. 2019, 20, 5781. [Google Scholar] [CrossRef] [PubMed]
- Duijf, P.H.G.; Vanmolkot, K.R.J.; Propping, P.; Friedl, W.; Krieger, E.; McKeon, F.; Dötsch, V.; Brunner, H.G.; van Bokhoven, H. Gain-of-function mutation in ADULT syndrome reveals the presence of a second transactivation domain in p63. Hum. Mol. Genet. 2002, 11, 799–804. [Google Scholar] [CrossRef] [PubMed]
- Romano, R.-A.; Smalley, K.; Magraw, C.; Serna, V.A.; Kurita, T.; Raghavan, S.; Sinha, S. ΔNp63 knockout mice reveal its indispensable role as a master regulator of epithelial development and differentiation. Development 2012, 139, 772–782. [Google Scholar] [CrossRef] [PubMed]
- Sen, G.L.; Boxer, L.D.; Webster, D.E.; Bussat, R.T.; Qu, K.; Zarnegar, B.J.; Johnston, D.; Siprashvili, Z.; Khavari, P.A. ZNF750 is a p63 target gene that induces KLF4 to drive terminal epidermal differentiation. Dev. Cell 2012, 22, 669–677. [Google Scholar] [CrossRef] [PubMed]
- Reis-Filho, J.S.; Torio, B.; Albergaria, A.; Schmitt, F.C. p63 expression in normal skin and usual cutaneous carcinomas. J. Cutan. Pathol. 2002, 29, 517–523. [Google Scholar] [CrossRef]
- Ha, L.; Ponnamperuma, R.M.; Jay, S.; Ricci, M.S.; Weinberg, W.C. Dysregulated ΔNp63α inhibits expression of Ink4a/arf, blocks senescence, and promotes malignant conversion of keratinocytes. PLoS ONE 2011, 6, e21877. [Google Scholar] [CrossRef]
- Gatti, V.; Fierro, C.; Annicchiarico-Petruzzelli, M.; Melino, G.; Peschiaroli, A. ΔNp63 in squamous cell carcinoma: Defining the oncogenic routes affecting epigenetic landscape and tumour microenvironment. Mol. Oncol. 2019, 13, 981–1001. [Google Scholar] [CrossRef] [PubMed]
- Pickering, C.R.; Zhou, J.H.; Lee, J.J.; Drummond, J.A.; Peng, S.A.; Saade, R.E.; Tsai, K.Y.; Curry, J.L.; Tetzlaff, M.T.; Lai, S.Y.; et al. Mutational landscape of aggressive cutaneous squamous cell carcinoma. Clin. Cancer Res. 2014, 20, 6582–6592. [Google Scholar] [CrossRef] [PubMed]
- Mauerer, A.; Herschberger, E.; Dietmaier, W.; Landthaler, M.; Hafner, C. Low incidence of EGFR and HRAS mutations in cutaneous squamous cell carcinomas of a German cohort. Exp. Dermatol. 2011, 20, 848–850. [Google Scholar] [CrossRef] [PubMed]
- Fogarty, G.B.; Conus, N.M.; Chu, J.; McArthur, G. Characterization of the expression and activation of the epidermal growth factor receptor in squamous cell carcinoma of the skin. Br. J. Dermatol. 2007, 156, 92–98. [Google Scholar] [CrossRef]
- Cañueto, J.; Cardeñoso, E.; García, J.L.; Santos-Briz, Á.; Castellanos-Martín, A.; Fernández-López, E.; Blanco Gómez, A.; Pérez-Losada, J.; Román-Curto, C. Epidermal growth factor receptor expression is associated with poor outcome in cutaneous squamous cell carcinoma. Br. J. Dermatol. 2017, 176, 1279–1287. [Google Scholar] [CrossRef]
- Nicholson, R.; Gee, J.; Harper, M. EGFR and cancer prognosis. Eur. J. Cancer 2001, 37, 9–15. [Google Scholar] [CrossRef]
- Oberholzer, P.A.; Kee, D.; Dziunycz, P.; Sucker, A.; Kamsukom, N.; Jones, R.; Roden, C.; Chalk, C.J.; Ardlie, K.; Palescandolo, E.; et al. RAS mutations are associated with the development of cutaneous squamous cell tumors in patients treated with RAF inhibitors. J. Clin. Oncol. 2012, 30, 316–321. [Google Scholar] [CrossRef] [PubMed]
- Su, F.; Viros, A.; Milagre, C.; Trunzer, K.; Bollag, G.; Spleiss, O.; Reis-Filho, J.S.; Kong, X.; Koya, R.C.; Flaherty, K.T.; et al. RAS mutations in cutaneous squamous-cell carcinomas in patients treated with BRAF inhibitors. N. Engl. J. Med. 2012, 366, 207–215. [Google Scholar] [CrossRef] [PubMed]
- Chrisostomidis, C.; Konofaos, P.; Karypidis, D.; Lazaris, A.; Kostakis, A.; Papadopoulos, O. The impact of Ets-1 oncoprotein and human endoglin (CD105) on the recurrence of non-melanoma skin cancers. Int. J. Dermatol. 2015, 54, 989–995. [Google Scholar] [CrossRef] [PubMed]
- Keehn, C.A.; Smoller, B.R.; Morgan, M.B. Ets-1 immunohistochemical expression in non-melanoma skin carcinoma. J. Cutan. Pathol. 2004, 31, 8–13. [Google Scholar] [CrossRef] [PubMed]
- Saxton, R.A.; Sabatini, D.M. mTOR Signaling in Growth, Metabolism, and Disease. Cell 2017, 168, 960–976. [Google Scholar] [CrossRef] [PubMed]
- Courtney, K.D.; Corcoran, R.B.; Engelman, J.A. The PI3K pathway as drug target in human cancer. J. Clin. Oncol. 2010, 28, 1075–1083. [Google Scholar] [CrossRef] [PubMed]
- Einspahr, J.G.; Calvert, V.; Alberts, D.S.; Curiel-Lewandrowski, C.; Warneke, J.; Krouse, R.; Stratton, S.P.; Liotta, L.; Longo, C.; Pellacani, G.; et al. Functional protein pathway activation mapping of the progression of normal skin to squamous cell carcinoma. Cancer Prev. Res. 2012, 5, 403–413. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Paredes, M.; Esteller, M. Cancer epigenetics reaches mainstream oncology. Nat. Med. 2011, 17, 330–339. [Google Scholar] [CrossRef] [PubMed]
- Vandiver, A.R.; Irizarry, R.A.; Hansen, K.D.; Garza, L.A.; Runarsson, A.; Li, X.; Chien, A.L.; Wang, T.S.; Leung, S.G.; Kang, S.; et al. Age and sun exposure-related widespread genomic blocks of hypomethylation in nonmalignant skin. Genome Biol. 2015, 16, 80. [Google Scholar] [CrossRef]
- Murao, K.; Kubo, Y.; Ohtani, N.; Hara, E.; Arase, S. Epigenetic abnormalities in cutaneous squamous cell carcinomas: Frequent inactivation of the RB1/p16 and p53 pathways. Br. J. Dermatol. 2006, 155, 999–1005. [Google Scholar] [CrossRef]
- Sandoval, J.; Esteller, M. Cancer epigenomics: Beyond genomics. Curr. Opin. Genet. Dev. 2012, 22, 50–55. [Google Scholar] [CrossRef] [PubMed]
- Darr, O.A.; Colacino, J.A.; Tang, A.L.; McHugh, J.B.; Bellile, E.L.; Bradford, C.R.; Prince, M.P.; Chepeha, D.B.; Rozek, L.S.; Moyer, J.S. Epigenetic alterations in metastatic cutaneous carcinoma. Head Neck 2015, 37, 994–1001. [Google Scholar] [CrossRef] [PubMed]
- Chiles, M.C.; Ai, L.; Zuo, C.; Fan, C.-Y.; Smoller, B.R. E-cadherin promoter hypermethylation in preneoplastic and neoplastic skin lesions. Mod. Pathol. 2003, 16, 1014–1018. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, N.; Odajima, T.; Ogi, K.; Ikeda, T.; Satoh, M. Expression of E-cadherin, alpha-catenin, and beta-catenin in the process of lymph node metastasis in oral squamous cell carcinoma. Br. J. Cancer 2003, 89, 557–563. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Paredes, M.; Bormann, F.; Raddatz, G.; Gutekunst, J.; Lucena-Porcel, C.; Köhler, F.; Wurzer, E.; Schmidt, K.; Gallinat, S.; Wenck, H.; et al. Methylation profiling identifies two subclasses of squamous cell carcinoma related to distinct cells of origin. Nat. Commun. 2018, 9, 577. [Google Scholar] [CrossRef] [PubMed]
- Hervás-Marín, D.; Higgins, F.; Sanmartín, O.; López-Guerrero, J.A.; Bañó, M.C.; Igual, J.C.; Quilis, I.; Sandoval, J. Genome wide DNA methylation profiling identifies specific epigenetic features in high-risk cutaneous squamous cell carcinoma. PLoS ONE 2019, 14, e0223341. [Google Scholar] [CrossRef]
- Kouzarides, T. Chromatin modifications and their function. Cell 2007, 128, 693–705. [Google Scholar] [CrossRef]
- Eckert, R.L.; Adhikary, G.; Rorke, E.A.; Chew, Y.C.; Balasubramanian, S. Polycomb group proteins are key regulators of keratinocyte function. J. Invest. Dermatol. 2011, 131, 295–301. [Google Scholar] [CrossRef] [PubMed]
- Ezhkova, E.; Pasolli, H.A.; Parker, J.S.; Stokes, N.; Su, I.; Hannon, G.; Tarakhovsky, A.; Fuchs, E. Ezh2 orchestrates gene expression for the stepwise differentiation of tissue-specific stem cells. Cell 2009, 136, 1122–1135. [Google Scholar] [CrossRef] [PubMed]
- Hernández-Ruiz, E.; Toll, A.; García-Diez, I.; Andrades, E.; Ferrandiz-Pulido, C.; Masferrer, E.; Yébenes, M.; Jaka, A.; Gimeno, J.; Gimeno, R.; et al. The Polycomb proteins RING1B and EZH2 repress the tumoral pro-inflammatory function in metastasizing primary cutaneous squamous cell carcinoma. Carcinogenesis 2018, 39, 503–513. [Google Scholar] [CrossRef] [PubMed]
- Tampa, M.; Mitran, C.I.; Mitran, M.I.; Nicolae, I.; Dumitru, A.; Matei, C.; Manolescu, L.; Popa, G.L.; Caruntu, C.; Georgescu, S.R. The Role of Beta HPV Types and HPV-Associated Inflammatory Processes in Cutaneous Squamous Cell Carcinoma. J. Immunol. Res. 2020, 2020, 5701639. [Google Scholar] [CrossRef] [PubMed]
- McLaughlin-Drubin, M.E.; Münger, K. Oncogenic activities of human papillomaviruses. Virus Res. 2009, 143, 195–208. [Google Scholar] [CrossRef] [PubMed]
- Paradisi, A.; Waterboer, T.; Sampogna, F.; Tabolli, S.; Simoni, S.; Pawlita, M.; Abeni, D. Seropositivity for human papillomavirus and incidence of subsequent squamous cell and basal cell carcinomas of the skin in patients with a previous nonmelanoma skin cancer. Br. J. Dermatol. 2011, 165, 782–791. [Google Scholar] [CrossRef] [PubMed]
- Astori, G.; Lavergne, D.; Benton, C.; Höckmayr, B.; Egawa, K.; Garbe, C.; de Villiers, E.M. Human papillomaviruses are commonly found in normal skin of immunocompetent hosts. J. Invest. Dermatol. 1998, 110, 752–755. [Google Scholar] [CrossRef] [PubMed]
- Vasiljević, N.; Hazard, K.; Dillner, J.; Forslund, O. Four novel human betapapillomaviruses of species 2 preferentially found in actinic keratosis. J. Gener. Virol. 2008, 89, 2467–2474. [Google Scholar] [CrossRef] [PubMed]
- Toll, A.; Lloveras, B.; Masferrer, E.; Ferrándiz-Pulido, C.; García-Patos, V.; Gheit, T.; Pujol, R.M.; Tommasino, M. Human beta papillomavirus DNA study in primary cutaneous squamous cell carcinomas and their corresponding metastases. Arch. Dermatol. Res. 2014, 306, 93–95. [Google Scholar] [CrossRef]
- Albrengues, J.; Bertero, T.; Grasset, E.; Bonan, S.; Maiel, M.; Bourget, I.; Philippe, C.; Herraiz Serrano, C.; Benamar, S.; Croce, O.; et al. Epigenetic switch drives the conversion of fibroblasts into proinvasive cancer-associated fibroblasts. Nat. Commun. 2015, 6, 10204. [Google Scholar] [CrossRef]
- Campisi, J. Aging, cellular senescence, and cancer. Annu. Rev. Physiol. 2013, 75, 685–705. [Google Scholar] [CrossRef] [PubMed]
- Dotto, G.P. Multifocal epithelial tumors and field cancerization: Stroma as a primary determinant. J. Clin. Invest. 2014, 124, 1446–1453. [Google Scholar] [CrossRef]
- Sasaki, K.; Sugai, T.; Ishida, K.; Osakabe, M.; Amano, H.; Kimura, H.; Sakuraba, M.; Kashiwa, K.; Kobayashi, S. Analysis of cancer-associated fibroblasts and the epithelial-mesenchymal transition in cutaneous basal cell carcinoma, squamous cell carcinoma, and malignant melanoma. Hum. Pathol. 2018, 79, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Tinaburri, L.; Valente, C.; Teson, M.; Minafò, Y.A.; Cordisco, S.; Guerra, L.; Dellambra, E. The Secretome of Aged Fibroblasts Promotes EMT-Like Phenotype in Primary Keratinocytes from Elderly Donors through BDNF-TrkB Axis. J. Invest. Dermatol. 2020. [Google Scholar] [CrossRef] [PubMed]
- Allen, C.T.; Ricker, J.L.; Chen, Z.; van Waes, C. Role of activated nuclear factor-kappaB in the pathogenesis and therapy of squamous cell carcinoma of the head and neck. Head Neck 2007, 29, 959–971. [Google Scholar] [CrossRef] [PubMed]
- Suarez-Carmona, M.; Hubert, P.; Gonzalez, A.; Duray, A.; Roncarati, P.; Erpicum, C.; Boniver, J.; Castronovo, V.; Noel, A.; Saussez, S.; et al. ΔNp63 isoform-mediated β-defensin family up-regulation is associated with (lymph)angiogenesis and poor prognosis in patients with squamous cell carcinoma. Oncotarget 2014, 5, 1856–1868. [Google Scholar] [CrossRef] [PubMed]
- Brantsch, K.D.; Meisner, C.; Schönfisch, B.; Trilling, B.; Wehner-Caroli, J.; Röcken, M.; Breuninger, H. Analysis of risk factors determining prognosis of cutaneous squamous-cell carcinoma: A prospective study. Lancet Oncol. 2008, 9, 713–720. [Google Scholar] [CrossRef]
- Chren, M.-M.; Linos, E.; Torres, J.S.; Stuart, S.E.; Parvataneni, R.; Boscardin, W.J. Tumor recurrence 5 years after treatment of cutaneous basal cell carcinoma and squamous cell carcinoma. J. Invest. Dermatol. 2013, 133, 1188–1196. [Google Scholar] [CrossRef] [PubMed]
- Leibovitch, I.; Huilgol, S.C.; Selva, D.; Hill, D.; Richards, S.; Paver, R. Cutaneous squamous cell carcinoma treated with Mohs micrographic surgery in Australia I. Experience over 10 years. J. Am. Acad. Dermatol. 2005, 53, 253–260. [Google Scholar] [CrossRef] [PubMed]
- Marrazzo, G.; Zitelli, J.A.; Brodland, D. Clinical outcomes in high-risk squamous cell carcinoma patients treated with Mohs micrographic surgery alone. J. Am. Acad. Dermatol. 2019, 80, 633–638. [Google Scholar] [CrossRef] [PubMed]
- Brodland, D.G.; Zitelli, J.A. Surgical margins for excision of primary cutaneous squamous cell carcinoma. J. Am. Acad. Dermatol. 1992, 27, 241–248. [Google Scholar] [CrossRef]
- Jambusaria-Pahlajani, A.; Miller, C.J.; Quon, H.; Smith, N.; Klein, R.Q.; Schmults, C.D. Surgical monotherapy versus surgery plus adjuvant radiotherapy in high-risk cutaneous squamous cell carcinoma: A systematic review of outcomes. Dermatol. Surg. 2009, 35, 574–585. [Google Scholar] [CrossRef] [PubMed]
- Rowe, D.E.; Carroll, R.J.; Day, C.L. Prognostic factors for local recurrence, metastasis, and survival rates in squamous cell carcinoma of the skin, ear, and lip. J. Am. Acad. Dermatol. 1992, 26, 976–990. [Google Scholar] [CrossRef]
- Griffiths, R.W.; Feeley, K.; Suvarna, S.K. Audit of clinical and histological prognostic factors in primary invasive squamous cell carcinoma of the skin: Assessment in a minimum 5 year follow-up study after conventional excisional surgery. Br. J. Plast. Surg. 2002, 55, 287–292. [Google Scholar] [CrossRef] [PubMed]
- Motaparthi, K.; Kapil, J.P.; Velazquez, E.F. Cutaneous Squamous Cell Carcinoma: Review of the Eighth Edition of the American Joint Committee on Cancer Staging Guidelines, Prognostic Factors, and Histopathologic Variants. Adv. Anat. Pathol. 2017, 24, 171–194. [Google Scholar] [CrossRef] [PubMed]
- Motley, R.; Kersey, P.; Lawrence, C. Multiprofessional guidelines for the management of the patient with primary cutaneous squamous cell carcinoma. Br. J. Plast. Surg. 2003, 56, 85–91. [Google Scholar] [CrossRef]
- Bonerandi, J.J.; Beauvillain, C.; Caquant, L.; Chassagne, J.F.; Chaussade, V.; Clavère, P.; Desouches, C.; Garnier, F.; Grolleau, J.L.; Grossin, M.; et al. Guidelines for the diagnosis and treatment of cutaneous squamous cell carcinoma and precursor lesions. J. Eur. Acad. Dermatol. Venereol. 2011, 25 (Suppl. 5), 1–51. [Google Scholar] [CrossRef]
- Sapijaszko, M.; Zloty, D.; Bourcier, M.; Poulin, Y.; Janiszewski, P.; Ashkenas, J. Non-melanoma Skin Cancer in Canada Chapter 5: Management of Squamous Cell Carcinoma. J. Cutan. Med. Surg. 2015, 19, 249–259. [Google Scholar] [CrossRef]
- Chu, M.B.; Slutsky, J.B.; Dhandha, M.M.; Beal, B.T.; Armbrecht, E.S.; Walker, R.J.; Varvares, M.A.; Fosko, S.W. Evaluation of the definitions of "high-risk" cutaneous squamous cell carcinoma using the american joint committee on cancer staging criteria and national comprehensive cancer network guidelines. J. Skin Cancer 2014, 2014, 154340. [Google Scholar] [CrossRef]
- Stratigos, A.; Garbe, C.; Lebbe, C.; Malvehy, J.; Del Marmol, V.; Pehamberger, H.; Peris, K.; Becker, J.C.; Zalaudek, I.; Saiag, P.; et al. Diagnosis and treatment of invasive squamous cell carcinoma of the skin: European consensus-based interdisciplinary guideline. Eur. J. Cancer 2015, 51, 1989–2007. [Google Scholar] [CrossRef]
- Löser, C.R.; Rompel, R.; Möhrle, M.; Häfner, H.-M.; Kunte, C.; Hassel, J.; Hohenleutner, U.; Podda, M.; Sebastian, G.; Hafner, J.; et al. S1 guideline: Microscopically controlled surgery (MCS). J. Dtsch. Dermatol. Ges. 2015, 13, 942–951. [Google Scholar] [CrossRef]
- van Lee, C.B.; Roorda, B.M.; Wakkee, M.; Voorham, Q.; Mooyaart, A.L.; de Vijlder, H.C.; Nijsten, T.; van den Bos, R.R. Recurrence rates of cutaneous squamous cell carcinoma of the head and neck after Mohs micrographic surgery vs. standard excision: A retrospective cohort study. Br. J. Dermatol. 2019, 181, 338–343. [Google Scholar] [CrossRef]
- Veness, M.J.; Morgan, G.J.; Palme, C.E.; Gebski, V. Surgery and adjuvant radiotherapy in patients with cutaneous head and neck squamous cell carcinoma metastatic to lymph nodes: Combined treatment should be considered best practice. Laryngoscope 2005, 115, 870–875. [Google Scholar] [CrossRef] [PubMed]
- Xiao, Y.; Yuan, S.; Liu, F.; Liu, B.; Zhu, J.; He, W.; Li, W.; Kan, Q. Comparison between wait-and-see policy and elective neck dissection in clinically N0 cutaneous squamous cell carcinoma of head and neck. Medicine 2018, 97, e10782. [Google Scholar] [CrossRef] [PubMed]
- Cannon, R.B.; Dundar, Y.; Thomas, A.; Monroe, M.M.; Buchmann, L.O.; Witt, B.L.; Sowder, A.M.; Hunt, J.P. Elective Neck Dissection for Head and Neck Cutaneous Squamous Cell Carcinoma with Skull Base Invasion. Otolaryngol. Head Neck Surg. Off. J. Am. Acad. Otolaryngol. Head Neck Surg. 2017, 156, 671–676. [Google Scholar] [CrossRef] [PubMed]
- Morton, C.; Szeimies, R.-M.; Sidoroff, A.; Wennberg, A.-M.; Basset-Seguin, N.; Calzavara-Pinton, P.; Gilaberte, Y.; Hofbauer, G.; Hunger, R.; Karrer, S.; et al. European Dermatology Forum Guidelines on topical photodynamic therapy. Eur. J. Dermatol. 2015, 25, 296–311. [Google Scholar] [CrossRef] [PubMed]
- Patel, R.; Chang, A.L.S. Immune Checkpoint Inhibitors for Treating Advanced Cutaneous Squamous Cell Carcinoma. Am. J. Clin. Dermatol. 2019, 20, 477–482. [Google Scholar] [CrossRef]
- Migden, M.R.; Rischin, D.; Schmults, C.D.; Guminski, A.; Hauschild, A.; Lewis, K.D.; Chung, C.H.; Hernandez-Aya, L.; Lim, A.M.; Chang, A.L.S.; et al. PD-1 Blockade with Cemiplimab in Advanced Cutaneous Squamous-Cell Carcinoma. N. Engl. J. Med. 2018, 379, 341–351. [Google Scholar] [CrossRef] [PubMed]
- Lee, A.; Duggan, S.; Deeks, E.D. Correction to: Cemiplimab: A Review in Advanced Cutaneous Squamous Cell Carcinoma. Drugs 2020, 80, 939. [Google Scholar] [CrossRef] [PubMed]
- Migden, M.R.; Khushalani, N.I.; Chang, A.L.S.; Lewis, K.D.; Schmults, C.D.; Hernandez-Aya, L.; Meier, F.; Schadendorf, D.; Guminski, A.; Hauschild, A.; et al. Cemiplimab in locally advanced cutaneous squamous cell carcinoma: Results from an open-label, phase 2, single-arm trial. Lancet Oncol. 2020, 21, 294–305. [Google Scholar] [CrossRef]
- Rischin, D.; Migden, M.R.; Lim, A.M.; Schmults, C.D.; Khushalani, N.I.; Hughes, B.G.M.; Schadendorf, D.; Dunn, L.A.; Hernandez-Aya, L.; Chang, A.L.S.; et al. Phase 2 study of cemiplimab in patients with metastatic cutaneous squamous cell carcinoma: Primary analysis of fixed-dosing, long-term outcome of weight-based dosing. J. Immunother. Cancer 2020, 8. [Google Scholar] [CrossRef]
- Ribero, S.; Stucci, L.S.; Daniels, G.A.; Borradori, L. Drug therapy of advanced cutaneous squamous cell carcinoma: Is there any evidence? Curr. Opin. Oncol. 2017, 29, 129–135. [Google Scholar] [CrossRef] [PubMed]
- Montaudié, H.; Viotti, J.; Combemale, P.; Dutriaux, C.; Dupin, N.; Robert, C.; Mortier, L.; Kaphan, R.; Duval-Modeste, A.-B.; Dalle, S.; et al. Cetuximab is efficient and safe in patients with advanced cutaneous squamous cell carcinoma: A retrospective, multicentre study. Oncotarget 2020, 11, 378–385. [Google Scholar] [CrossRef] [PubMed]
- William, W.N.; Feng, L.; Ferrarotto, R.; Ginsberg, L.; Kies, M.; Lippman, S.; Glisson, B.; Kim, E.S. Gefitinib for patients with incurable cutaneous squamous cell carcinoma: A single-arm phase II clinical trial. J. Am. Acad. Dermatol. 2017, 77, 1110–1113.e2. [Google Scholar] [CrossRef]
- Brewster, A.M.; Lee, J.J.; Clayman, G.L.; Clifford, J.L.; Reyes, M.J.T.N.; Zhou, X.; Sabichi, A.L.; Strom, S.S.; Collins, R.; Meyers, C.A.; et al. Randomized trial of adjuvant 13-cis-retinoic acid and interferon alfa for patients with aggressive skin squamous cell carcinoma. J. Clin. Oncol. 2007, 25, 1974–1978. [Google Scholar] [CrossRef] [PubMed]
- Goyal, U.; Prabhakar, N.K.; Davuluri, R.; Morrison, C.M.; Yi, S.K. Role of Concurrent Systemic Therapy with Adjuvant Radiation Therapy for Locally Advanced Cutaneous Head and Neck Squamous Cell Carcinoma. Cureus 2017, 9, e1784. [Google Scholar] [CrossRef] [PubMed]
- Gellrich, F.F.; Hüning, S.; Beissert, S.; Eigentler, T.; Stockfleth, E.; Gutzmer, R.; Meier, F. Medical treatment of advanced cutaneous squamous-cell carcinoma. J. Eur. Acad. Dermatol. Venereol. 2019, 33, 38–43. [Google Scholar] [CrossRef] [PubMed]
- Behshad, R.; Garcia-Zuazaga, J.; Bordeaux, J.S. Systemic treatment of locally advanced nonmetastatic cutaneous squamous cell carcinoma: A review of the literature. Br. J. Dermatol. 2011, 165, 1169–1177. [Google Scholar] [CrossRef] [PubMed]
- Trodello, C.; Pepper, J.-P.; Wong, M.; Wysong, A. Cisplatin and Cetuximab Treatment for Metastatic Cutaneous Squamous Cell Carcinoma: A Systematic Review. Dermatol. Surg. 2017, 43, 40–49. [Google Scholar] [CrossRef]
- Bertino, G.; Sersa, G.; de Terlizzi, F.; Occhini, A.; Plaschke, C.C.; Groselj, A.; Langdon, C.; Grau, J.J.; McCaul, J.A.; Heuveling, D.; et al. European Research on Electrochemotherapy in Head and Neck Cancer (EURECA) project: Results of the treatment of skin cancer. Eur. J. Cancer 2016, 63, 41–52. [Google Scholar] [CrossRef] [PubMed]
- Lansbury, L.; Bath-Hextall, F.; Perkins, W.; Stanton, W.; Leonardi-Bee, J. Interventions for non-metastatic squamous cell carcinoma of the skin: Systematic review and pooled analysis of observational studies. BMJ 2013, 347, f6153. [Google Scholar] [CrossRef] [PubMed]
- Porceddu, S.V. Prognostic factors and the role of adjuvant radiation therapy in non-melanoma skin cancer of the head and neck. Am. Soc. Clin. Oncol. Educ. Book 2015, e513–e518. [Google Scholar] [CrossRef] [PubMed]
- Babington, S.; Veness, M.J.; Cakir, B.; Gebski, V.J.; Morgan, G.J. Squamous cell carcinoma of the lip: Is there a role for adjuvant radiotherapy in improving local control following incomplete or inadequate excision? ANZ J. Surg. 2003, 73, 621–625. [Google Scholar] [CrossRef]
- Likhacheva, A.; Awan, M.; Barker, C.A.; Bhatnagar, A.; Bradfield, L.; Brady, M.S.; Buzurovic, I.; Geiger, J.L.; Parvathaneni, U.; Zaky, S.; et al. Definitive and Postoperative Radiation Therapy for Basal and Squamous Cell Cancers of the Skin: Executive Summary of an American Society for Radiation Oncology Clinical Practice Guideline. Pract. Radiat. Oncol. 2020, 10, 8–20. [Google Scholar] [CrossRef] [PubMed]
- Cuperus, E.; Leguit, R.; Albregts, M.; Toonstra, J. Post radiation skin tumors: Basal cell carcinomas, squamous cell carcinomas and angiosarcomas. A review of this late effect of radiotherapy. Eur. J. Dermatol. 2013, 23, 749–757. [Google Scholar] [CrossRef]
- Cassarino, D.S.; Derienzo, D.P.; Barr, R.J. Cutaneous squamous cell carcinoma: A comprehensive clinicopathologic classification. Part one. J. Cutan. Pathol. 2006, 33, 191–206. [Google Scholar] [CrossRef] [PubMed]
- Que, S.K.T.; Zwald, F.O.; Schmults, C.D. Cutaneous squamous cell carcinoma: Management of advanced and high-stage tumors. J. Am. Acad. Dermatol. 2018, 78, 249–261. [Google Scholar] [CrossRef]
- Concu, R.; Cordeiro, M.N.D.S. Cetuximab and the Head and Neck Squamous Cell Cancer. Curr. Top. Med. Chem. 2018, 18, 192–198. [Google Scholar] [CrossRef]
- Maubec, E. Update of the Management of Cutaneous Squamous-cell Carcinoma. Acta Derm. Venereol. 2020, 100, adv00143. [Google Scholar] [CrossRef] [PubMed]
- Cañueto, J.; Jaka, A.; Toll, A. Utilidad de la radioterapia en adyuvancia en el carcinoma epidermoide cutáneo. Actas Dermosifiliogr. 2018, 109, 476–484. [Google Scholar] [CrossRef]
- Harris, B.N.; Pipkorn, P.; Nguyen, K.N.B.; Jackson, R.S.; Rao, S.; Moore, M.G.; Farwell, D.G.; Bewley, A.F. Association of Adjuvant Radiation Therapy With Survival in Patients With Advanced Cutaneous Squamous Cell Carcinoma of the Head and Neck. JAMA Otolaryngol. Head Neck Surg. 2019, 145, 153–158. [Google Scholar] [CrossRef] [PubMed]
- Varra, V.; Smile, T.D.; Geiger, J.L.; Koyfman, S.A. Recent and Emerging Therapies for Cutaneous Squamous Cell Carcinomas of the Head and Neck. Curr. Treat. Options Oncol. 2020, 21, 37. [Google Scholar] [CrossRef] [PubMed]
- Gehl, J.; Sersa, G.; Matthiessen, L.W.; Muir, T.; Soden, D.; Occhini, A.; Quaglino, P.; Curatolo, P.; Campana, L.G.; Kunte, C.; et al. Updated standard operating procedures for electrochemotherapy of cutaneous tumours and skin metastases. Acta Oncol. 2018, 57, 874–882. [Google Scholar] [CrossRef] [PubMed]
- Testori, A.; Tosti, G.; Martinoli, C.; Spadola, G.; Cataldo, F.; Verrecchia, F.; Baldini, F.; Mosconi, M.; Soteldo, J.; Tedeschi, I.; et al. Electrochemotherapy for cutaneous and subcutaneous tumor lesions: A novel therapeutic approach. Dermatol. Ther. 2010, 23, 651–661. [Google Scholar] [CrossRef]
- Di Monta, G.; Caracò, C.; Simeone, E.; Grimaldi, A.M.; Marone, U.; Di Marzo, M.; Vanella, V.; Festino, L.; Palla, M.; Mori, S.; et al. Electrochemotherapy efficacy evaluation for treatment of locally advanced stage III cutaneous squamous cell carcinoma: A 22-cases retrospective analysis. J. Transl. Med. 2017, 15, 82. [Google Scholar] [CrossRef]
- Seyed Jafari, S.M.; Jabbary Lak, F.; Gazdhar, A.; Shafighi, M.; Borradori, L.; Hunger, R.E. Application of electrochemotherapy in the management of primary and metastatic cutaneous malignant tumours: A systematic review and meta-analysis. Eur. J. Dermatol. 2018, 28, 287–313. [Google Scholar] [CrossRef]
- Saraiya, M.; Glanz, K.; Briss, P.A.; Nichols, P.; White, C.; Das, D.; Smith, S.J.; Tannor, B.; Hutchinson, A.B.; Wilson, K.M.; et al. Interventions to prevent skin cancer by reducing exposure to ultraviolet radiation: A systematic review. Am. J. Prev. Med. 2004, 27, 422–466. [Google Scholar] [CrossRef] [PubMed]
- Gordon, L.G.; Rowell, D. Health system costs of skin cancer and cost-effectiveness of skin cancer prevention and screening: A systematic review. Eur. J. Cancer Prevent. Off. J. Eur. Cancer Prevent. Organis. 2015, 24, 141–149. [Google Scholar] [CrossRef]
- Schmitt, J.; Haufe, E.; Trautmann, F.; Schulze, H.-J.; Elsner, P.; Drexler, H.; Bauer, A.; Letzel, S.; John, S.M.; Fartasch, M.; et al. Is ultraviolet exposure acquired at work the most important risk factor for cutaneous squamous cell carcinoma? Results of the population-based case-control study FB-181. Br. J. Dermatol. 2018, 178, 462–472. [Google Scholar] [CrossRef] [PubMed]
- Strickland, P.T.; Vitasa, B.C.; West, S.K.; Rosenthal, F.S.; Emmett, E.A.; Taylor, H.R. Quantitative carcinogenesis in man: Solar ultraviolet B dose dependence of skin cancer in Maryland watermen. J. Natl. Cancer Inst. 1989, 81, 1910–1913. [Google Scholar] [CrossRef]
- Rosso, S.; Zanetti, R.; Martinez, C.; Tormo, M.J.; Schraub, S.; Sancho-Garnier, H.; Franceschi, S.; Gafà, L.; Perea, E.; Navarro, C.; et al. The multicentre south European study ‘Helios’. II: Different sun exposure patterns in the aetiology of basal cell and squamous cell carcinomas of the skin. Br. J. Cancer 1996, 73, 1447–1454. [Google Scholar] [CrossRef]
- Schmitt, J.; Seidler, A.; Diepgen, T.L.; Bauer, A. Occupational ultraviolet light exposure increases the risk for the development of cutaneous squamous cell carcinoma: A systematic review and meta-analysis. Br. J. Dermatol. 2011, 164, 291–307. [Google Scholar] [CrossRef]
- Henrikson, N.B.; Morrison, C.C.; Blasi, P.R.; Nguyen, M.; Shibuya, K.C.; Patnode, C.D. Behavioral Counseling for Skin Cancer Prevention: Evidence Report and Systematic Review for the US Preventive Services Task Force. JAMA 2018, 319, 1143–1157. [Google Scholar] [CrossRef] [PubMed]
- Green, A.; Williams, G.; Nèale, R.; Hart, V.; Leslie, D.; Parsons, P.; Marks, G.C.; Gaffney, P.; Battistutta, D.; Frost, C.; et al. Daily sunscreen application and betacarotene supplementation in prevention of basal-cell and squamous-cell carcinomas of the skin: A randomised controlled trial. Lancet 1999, 354, 723–729. [Google Scholar] [CrossRef]
- Roberts, L.K.; Beasley, D.G. Commercial sunscreen lotions prevent ultraviolet-radiation-induced immune suppression of contact hypersensitivity. J. Invest. Dermatol. 1995, 105, 339–344. [Google Scholar] [CrossRef] [PubMed]
- Neale, R.; Russell, A.; Muller, H.K.; Green, A. Sun exposure, sunscreen and their effects on epidermal Langerhans cells. Photochem. Photobiol. 1997, 66, 260–264. [Google Scholar] [CrossRef]
- Nijsten, T.E.; Stern, R.S. Oral retinoid use reduces cutaneous squamous cell carcinoma risk in patients with psoriasis treated with psoralen-UVA: A nested cohort study. J. Am. Acad. Dermatol. 2003, 49, 644–650. [Google Scholar] [CrossRef]
- Bavinck, J.N.; Tieben, L.M.; van der Woude, F.J.; Tegzess, A.M.; Hermans, J.; ter Schegget, J.; Vermeer, B.J. Prevention of skin cancer and reduction of keratotic skin lesions during acitretin therapy in renal transplant recipients: A double-blind, placebo-controlled study. J. Clin. Oncol. 1995, 13, 1933–1938. [Google Scholar] [CrossRef] [PubMed]
- Kraemer, K.H.; DiGiovanna, J.J.; Moshell, A.N.; Tarone, R.E.; Peck, G.L. Prevention of skin cancer in xeroderma pigmentosum with the use of oral isotretinoin. N. Engl. J. Med. 1988, 318, 1633–1637. [Google Scholar] [CrossRef]
- McKenna, D.B.; Murphy, G.M. Skin cancer chemoprophylaxis in renal transplant recipients: 5 years of experience using low-dose acitretin. Br. J. Dermatol. 1999, 140, 656–660. [Google Scholar] [CrossRef]
- George, R.; Weightman, W.; Russ, G.R.; Bannister, K.M.; Mathew, T.H. Acitretin for chemoprevention of non-melanoma skin cancers in renal transplant recipients. Australas. J. Dermatol. 2002, 43, 269–273. [Google Scholar] [CrossRef]
- Chen, K.; Craig, J.C.; Shumack, S. Oral retinoids for the prevention of skin cancers in solid organ transplant recipients: A systematic review of randomized controlled trials. Br. J. Dermatol. 2005, 152, 518–523. [Google Scholar] [CrossRef] [PubMed]
- Chen, A.C.; Martin, A.J.; Choy, B.; Fernández-Peñas, P.; Dalziell, R.A.; McKenzie, C.A.; Scolyer, R.A.; Dhillon, H.M.; Vardy, J.L.; Kricker, A.; et al. A Phase 3 Randomized Trial of Nicotinamide for Skin-Cancer Chemoprevention. N. Engl. J. Med. 2015, 373, 1618–1626. [Google Scholar] [CrossRef]
- Chen, A.C.; Martin, A.J.; Damian, D.L. Nicotinamide for Skin-Cancer Chemoprevention. N. Engl. J. Med. 2016, 374, 789–790. [Google Scholar] [CrossRef]
- Sidaway, P. Skin cancer: Nicotinamide reduces new skin cancer risk. Nat. Rev. Clin. Oncol. 2016, 13, 4. [Google Scholar] [CrossRef] [PubMed]
- Fania, L.; Mazzanti, C.; Campione, E.; Candi, E.; Abeni, D.; Dellambra, E. Role of Nicotinamide in Genomic Stability and Skin Cancer Chemoprevention. Int. J. Mol. Sci. 2019, 20, 5946. [Google Scholar] [CrossRef] [PubMed]
- Johannesdottir, S.A.; Chang, E.T.; Mehnert, F.; Schmidt, M.; Olesen, A.B.; Sørensen, H.T. Nonsteroidal anti-inflammatory drugs and the risk of skin cancer: A population-based case-control study. Cancer 2012, 118, 4768–4776. [Google Scholar] [CrossRef] [PubMed]
- Muranushi, C.; Olsen, C.M.; Pandeya, N.; Green, A.C. Aspirin and nonsteroidal anti-inflammatory drugs can prevent cutaneous squamous cell carcinoma: A systematic review and meta-analysis. J. Invest. Dermatol. 2015, 135, 975–983. [Google Scholar] [CrossRef] [PubMed]
- Reinau, D.; Surber, C.; Jick, S.S.; Meier, C.R. Nonsteroidal anti-inflammatory drugs and the risk of nonmelanoma skin cancer. Int. J. Cancer 2015, 137, 144–153. [Google Scholar] [CrossRef] [PubMed]
- Torti, D.C.; Christensen, B.C.; Storm, C.A.; Fortuny, J.; Perry, A.E.; Zens, M.S.; Stukel, T.; Spencer, S.K.; Nelson, H.H.; Karagas, M.R. Analgesic and nonsteroidal anti-inflammatory use in relation to nonmelanoma skin cancer: A population-based case-control study. J. Am. Acad. Dermatol. 2011, 65, 304–312. [Google Scholar] [CrossRef]


| Clinical Risk Factors for Recurrence | |
|---|---|
| Size and location of lesion * | ≥20 mm on area L ≥10 mm on area M ≥6 mm on area H |
| Undefined borders | |
| Recurrent lesion | |
| SOTRs ** | |
| Previous radiotherapy or site of chronic inflammation | |
| Rapidly growing lesion | |
| Neurological symptoms | |
| Pathological risk factors for recurrence | |
| Not clearly differentiated lesion | |
| Adenoid, adenosquamous, or desmoplastic subtypes | |
| Clark level ≥IV | |
| Modified Breslow thickness ≥4 mm | |
| Perineural and/or vascular involvement |
| Nonpigmented Bowen’s Disease | Pigmented Bowen’s Disease | Invasive Squamous Cell Carcinoma |
|---|---|---|
| Dotted and glomerular vessels | Brown to gray globules/dots | Keratin/scales |
| Scaly white-to-yellow surfaces | Structureless pigmentation | Blood spots |
| Brown structureless areas | White circles | |
| Double-edge sign | White structureless areas | |
| Hairpin and linear-irregular vessels | ||
| Perivascular white halos | ||
| Ulceration |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fania, L.; Didona, D.; Di Pietro, F.R.; Verkhovskaia, S.; Morese, R.; Paolino, G.; Donati, M.; Ricci, F.; Coco, V.; Ricci, F.; et al. Cutaneous Squamous Cell Carcinoma: From Pathophysiology to Novel Therapeutic Approaches. Biomedicines 2021, 9, 171. https://doi.org/10.3390/biomedicines9020171
Fania L, Didona D, Di Pietro FR, Verkhovskaia S, Morese R, Paolino G, Donati M, Ricci F, Coco V, Ricci F, et al. Cutaneous Squamous Cell Carcinoma: From Pathophysiology to Novel Therapeutic Approaches. Biomedicines. 2021; 9(2):171. https://doi.org/10.3390/biomedicines9020171
Chicago/Turabian StyleFania, Luca, Dario Didona, Francesca Romana Di Pietro, Sofia Verkhovskaia, Roberto Morese, Giovanni Paolino, Michele Donati, Francesca Ricci, Valeria Coco, Francesco Ricci, and et al. 2021. "Cutaneous Squamous Cell Carcinoma: From Pathophysiology to Novel Therapeutic Approaches" Biomedicines 9, no. 2: 171. https://doi.org/10.3390/biomedicines9020171
APA StyleFania, L., Didona, D., Di Pietro, F. R., Verkhovskaia, S., Morese, R., Paolino, G., Donati, M., Ricci, F., Coco, V., Ricci, F., Candi, E., Abeni, D., & Dellambra, E. (2021). Cutaneous Squamous Cell Carcinoma: From Pathophysiology to Novel Therapeutic Approaches. Biomedicines, 9(2), 171. https://doi.org/10.3390/biomedicines9020171








