Efficacy of Acetylcholinesterase Inhibitors on Cognitive Function in Alzheimer’s Disease. Review of Reviews
Abstract
:1. Introduction
2. Materials and Methods
2.1. Review Questions
2.2. Data Sources and Search Strategy
2.3. Exclusion Criteria
2.4. Screening Review (Review Selection) and Data Collection
2.5. Data Extraction and Data Measurement
2.6. Assessment of the Methodological Quality of the Study and Grading of the Evidence
2.7. Quality Analysis of Reviews
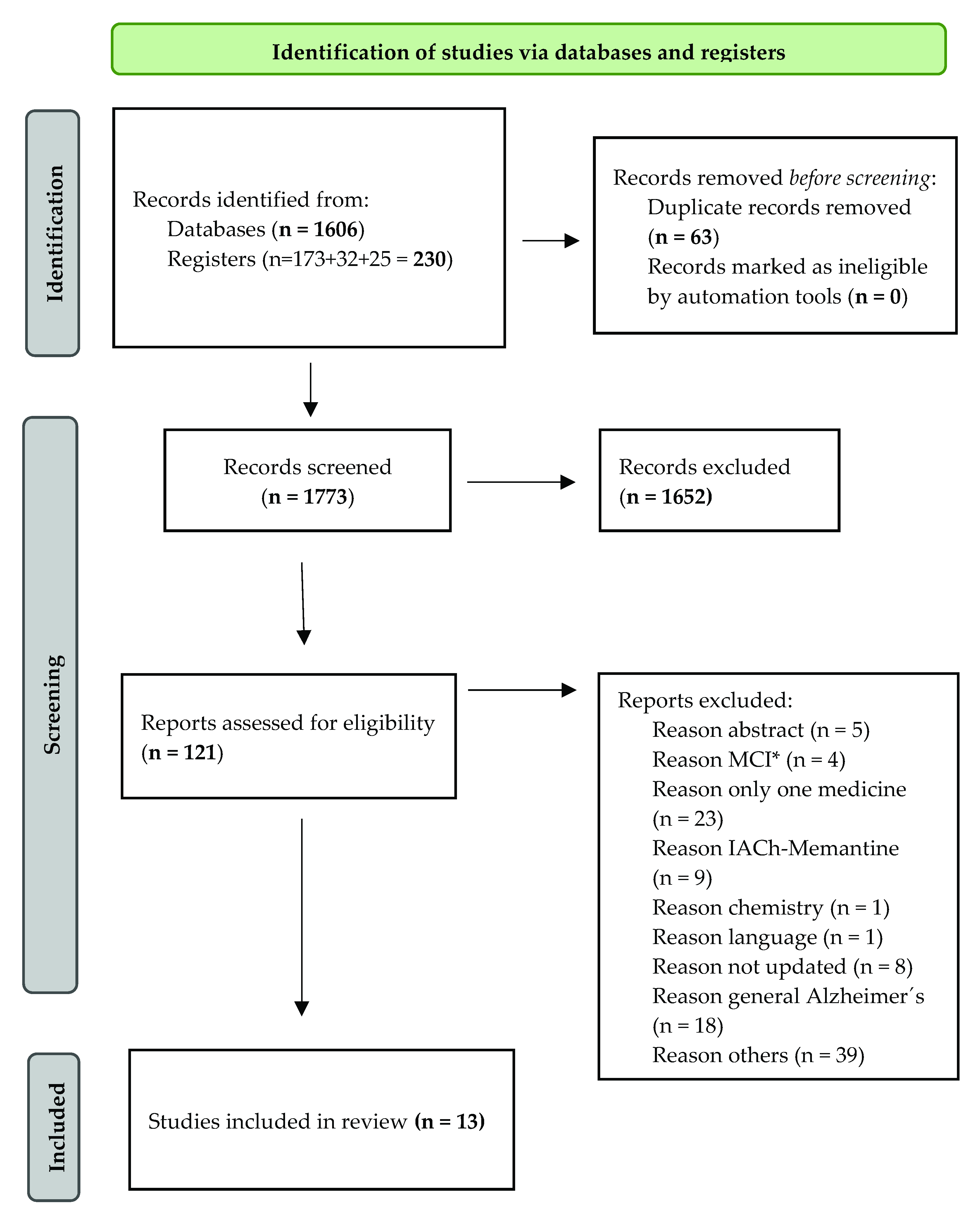
3. Results
3.1. Review Characteristics of the Reviews
3.2. Study Quality and Evidence Synthesis
4. Discussion
4.1. Limitations and Strengths
4.2. Implications for Practice
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- World Health, O. Towards a Dementia Plan: A WHO Guide; World Health Organization: Geneva, Switzerland, 2018. [Google Scholar]
- Prince, M.; Knapp, M.; Guerchet, M.M.; Prina, M.P.; Comas-Herrera, A.; Wittenberg, R.; Adelaja, B.; Hu, B.; King, D.; Rehill, A.; et al. Dementia UK: Update, 2nd ed.; © Alzheimer’s Society 2014. All rights reserved; Alzheimer’s Society: London, UK, 2014; ISBN 978-1-906647-31-5. [Google Scholar]
- Niu, H.; Álvarez-Álvarez, I.; Guillén-Grima, F.; Aguinaga-Ontoso, I. Prevalence and incidence of Alzheimer’s disease in Europe: A meta-analysis. Neurologia 2017, 32, 523–532. [Google Scholar] [CrossRef]
- Prince, M.; Bryce, R.; Albanese, E.; Wimo, A.; Ribeiro, W.; Ferri, C.P. The global prevalence of dementia: A systematic review and metaanalysis. Alzheimer’s Dement. J. Alzheimer’s Assoc. 2013, 9, 63–75.e62. [Google Scholar] [CrossRef]
- Global Burden of Disease Collaborative Network. Global Burden of Disease Study 2016 (GBD 2016) Incidence, Prevalence, and Years Lived with Disability 1990–2016; Institute for Health Metrics and Evaluation (IHME): Seattle, DC, USA, 2017. [Google Scholar]
- Álvarez Castillo, A.; Rodríguez Alfaro, J.M.; Salas Boza, A. Influencia de la enfermedad de Alzheimer en los sistemas de neurotransmisión sináptica. Rev. Med. Sinerg. 2020, 5, e442. [Google Scholar] [CrossRef]
- Gaugler, J.; Bryan James, T.J.; Reimer, J.; Weuve, J.; Alzheimer’s Association. 2021 Alzheimer’s Disease Facts and Figures; Alzheimer’s Dementia: Chicago, IL, USA, 2021; Volume 17. [Google Scholar]
- Alberca Serrano, R.; López Pousa, S. Enfermedad de Alzheimer y otras Demencias, 4th ed.; Médica Panamericana: Madrid, Spain, 2011; ISBN 9788498355345. [Google Scholar]
- González, M. Atlas of Biomarkers for Alzheimer’s Disease; Springer International Publishing: Berlin/Heidelberg, Germany, 2014. [Google Scholar]
- Sevigny, J.; Chiao, P.; Bussière, T.; Weinreb, P.H.; Williams, L.; Maier, M.; Dunstan, R.; Salloway, S.; Chen, T.; Ling, Y.; et al. The antibody aducanumab reduces Aβ plaques in Alzheimer’s disease. Nature 2016, 537, 50–56. [Google Scholar] [CrossRef] [PubMed]
- Birks, J. Cholinesterase inhibitors for Alzheimer’s disease. Cochrane Database Syst. Rev. 2006, Cd005593. [Google Scholar] [CrossRef]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. PLoS Med. 2021, 18, e1003583. [Google Scholar] [CrossRef] [PubMed]
- Inglis, F. The tolerability and safety of cholinesterase inhibitors in the treatment of dementia. Int. J. Clin. Pract. 2002, 127, 45–63. [Google Scholar]
- Higgins, J.P.; Altman, D.G.; Gøtzsche, P.C.; Jüni, P.; Moher, D.; Oxman, A.D.; Savovic, J.; Schulz, K.F.; Weeks, L.; Sterne, J.A. The Cochrane Collaboration’s tool for assessing risk of bias in randomised trials. BMJ 2011, 343, d5928. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Whiting, P.; Savović, J.; Higgins, J.P.; Caldwell, D.M.; Reeves, B.C.; Shea, B.; Davies, P.; Kleijnen, J.; Churchill, R. ROBIS: A new tool to assess risk of bias in systematic reviews was developed. J. Clin. Epidemiol. 2016, 69, 225–234. [Google Scholar] [CrossRef] [Green Version]
- Taylor, R.S.; Elston, J. The use of surrogate outcomes in model-based cost-effectiveness analyses: A survey of UK Health Technology Assessment reports. Health Technol. Assess. 2009, 13, 1–50. [Google Scholar] [CrossRef]
- Urrútia, G.; Bonfill, X. PRISMA declaration: A proposal to improve the publication of systematic reviews and meta-analyses. Med. Clin. 2010, 135, 507–511. [Google Scholar] [CrossRef] [PubMed]
- Blanco-Silvente, L.; Castells, X.; Saez, M.; Barceló, M.A.; Garre-Olmo, J.; Vilalta-Franch, J.; Capellà, D. Discontinuation, Efficacy, and Safety of Cholinesterase Inhibitors for Alzheimer’s Disease: A Meta-Analysis and Meta-Regression of 43 Randomized Clinical Trials Enrolling 16 106 Patients. Int. J. Neuropsychopharmacol. 2017, 20, 519–528. [Google Scholar] [CrossRef] [PubMed]
- Bond, M.; Rogers, G.; Peters, J.; Anderson, R.; Hoyle, M.; Miners, A.; Moxham, T.; Davis, S.; Thokala, P.; Wailoo, A.; et al. The effectiveness and cost-effectiveness of donepezil, galantamine, rivastigmine and memantine for the treatment of Alzheimer’s disease (review of Technology Appraisal No. 111): A systematic review and economic model. Health Technol. Assess. 2012, 16, 1–470. [Google Scholar] [CrossRef] [PubMed]
- Clegg, A.; Bryant, J.; Nicholson, T.; McIntyre, L.; De Broe, S.; Gerard, K.; Waugh, N. Clinical and cost-effectiveness of donepezil, rivastigmine, and galantamine for Alzheimer’s disease. A systematic review. Int. J. Technol. Assess. Health Care 2002, 18, 497–507. [Google Scholar] [CrossRef] [PubMed]
- Grimmer, T.; Kurz, A. Effects of cholinesterase inhibitors on behavioural disturbances in Alzheimer’s disease—A systematic review. Drugs Aging 2006, 23, 957–967. [Google Scholar] [CrossRef] [PubMed]
- Hansen, R.A.; Gartlehner, G.; Webb, A.P.; Morgan, L.C.; Moore, C.G.; Jonas, D.E. Efficacy and safety of donepezil, galantamine, and rivastigmine for the treatment of Alzheimer’s disease: A systematic review and meta-analysis. Clin. Interv. Aging 2008, 3, 211–225. [Google Scholar] [PubMed]
- Hyde, C.; Peters, J.; Bond, M.; Rogers, G.; Hoyle, M.; Anderson, R.; Jeffreys, M.; Davis, S.; Thokala, P.; Moxham, T. Evolution of the evidence on the effectiveness and cost-effectiveness of acetylcholinesterase inhibitors and memantine for Alzheimer’s disease: Systematic review and economic model. Age Ageing 2013, 42, 14–20. [Google Scholar] [CrossRef] [Green Version]
- Kaduszkiewicz, H.; Zimmermann, T.; Beck-Bornholdt, H.P.; van den Bussche, H. Cholinesterase inhibitors for patients with Alzheimer’s disease: Systematic review of randomised clinical trials. BMJ 2005, 331, 321–327. [Google Scholar] [CrossRef] [Green Version]
- Kobayashi, H.; Ohnishi, T.; Nakagawa, R.; Yoshizawa, K. The comparative efficacy and safety of cholinesterase inhibitors in patients with mild-to-moderate Alzheimer’s disease: A Bayesian network meta-analysis. Int. J. Geriatr. Psychiatry 2016, 31, 892–904. [Google Scholar] [CrossRef] [Green Version]
- Livingston, G.; Katona, C. How useful are cholinesterase inhibitors in the treatment of Alzheimer’s disease? A number needed to treat analysis. Int. J. Geriatr. Psychiatry 2000, 15, 203–207. [Google Scholar] [CrossRef]
- Ritchie, C.W.; Ames, D.; Clayton, T.; Lai, R. Metaanalysis of randomized trials of the efficacy and safety of donepezil, galantamine, and rivastigmine for the treatment of Alzheimer disease. Am. J. Geriatr. Psychiatry 2004, 12, 358–369. [Google Scholar] [CrossRef]
- Takeda, A.; Loveman, E.; Clegg, A.; Kirby, J.; Picot, J.; Payne, E.; Green, C. A systematic review of the clinical effectiveness of donepezil, rivastigmine and galantamine on cognition, quality of life and adverse events in Alzheimer’s disease. Int. J. Geriatr. Psychiatry 2006, 21, 17–28. [Google Scholar] [CrossRef] [PubMed]
- Trinh, N.H.; Hoblyn, J.; Mohanty, S.; Yaffe, K. Efficacy of cholinesterase inhibitors in the treatment of neuropsychiatric symptoms and functional impairment in Alzheimer disease: A meta-analysis. JAMA 2003, 289, 210–216. [Google Scholar] [CrossRef] [PubMed]
- O.M.S. CIE-10. Trastornos Mentales y del Comportamiento; Décima Revisión de la Clasificación Internacional de las Enfermedades; Descripciones Clínicas y pautas para el diagnóstico; Organización Mundial de la Salud: Geneva, Switzerland, 1992. [Google Scholar]
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, 3rd ed.; American Psychiatric Association: Washington, DC, USA, 1987. [Google Scholar]
- McKhann, G.; Drachman, D.; Folstein, M.; Katzman, R.; Price, D.; Stadlan, E.M. Clinical diagnosis of Alzheimer’s disease: Report of the NINCDS-ADRDA Work Group under the auspices of Department of Health and Human Services Task Force on Alzheimer’s Disease. Neurology 1984, 34, 939–944. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Blanco-Silvente, L.; Castells, X.; Garre-Olmo, J.; Vilalta-Franch, J.; Saez, M.; Barceló, M.; Capellà, D. Study of the strength of the evidence and the redundancy of the research on pharmacological treatment for Alzheimer’s disease: A cumulative meta-analysis and trial sequential analysis. Eur. J. Clin. Pharmacol. 2019, 75, 1659–1667. [Google Scholar] [CrossRef] [PubMed]
- Jadad, A.R.; Moore, R.A.; Carroll, D.; Jenkinson, C.; Reynolds, D.J.; Gavaghan, D.J.; McQuay, H.J. Assessing the quality of reports of randomized clinical trials: Is blinding necessary? Control. Clin. Trials 1996, 17, 1–12. [Google Scholar] [CrossRef]
- Harris, R.P.; Helfand, M.; Woolf, S.H.; Lohr, K.N.; Mulrow, C.D.; Teutsch, S.M.; Atkins, D. REPRINT OF: Current Methods of the U.S. Preventive Services Task Force: A Review of the Process. Am. J. Prev. Med. 2020, 58, 316–331. [Google Scholar] [CrossRef]
- Egger, M.; Davey Smith, G.; Schneider, M.; Minder, C. Bias in meta-analysis detected by a simple, graphical test. BMJ 1997, 315, 629–634. [Google Scholar] [CrossRef] [Green Version]
- Jorge López-Álvarez, L.F.A.-O. Nuevos criterios diagnósticos de la demencia y la enfermedad de Alzheimer: Una visión desde la psicogeriatría. Psicogeriatría 2015, 5, 3–14. [Google Scholar]
- Lagakos, S.W. Surrogate markers in AIDS clinical trials: Conceptual basis, validation, and uncertainties. Clin. Infect. Dis. Off. Publ. Infect. Dis. Soc. Am. 1993, 16 (Suppl. 1), S22–S25. [Google Scholar] [CrossRef]
- Weeks, G.; George, J.; Maclure, K.; Stewart, D. Non-medical prescribing versus medical prescribing for acute and chronic disease management in primary and secondary care. Cochrane Database Syst. Rev. 2016, 11, Cd011227. [Google Scholar] [CrossRef] [Green Version]
- National Institute for Health and Clinical Excellence. Donepezil, Galantamine, Rivastigmine (Review) and Memantine for the Treatment of Alzheimer’s Disease (Amended); NICE technology appraisal guidance 111; National Institute for Health and Clinical Excellence: London, UK, 2009. [Google Scholar]
- Rédaction, P. Médicaments de la maladie d’Alzheimer: Enfin non remboursables en France! Rev. Prescrire 2018, 38, 1–2. [Google Scholar]
- Guyatt, G.H.; Oxman, A.D.; Kunz, R.; Woodcock, J.; Brozek, J.; Helfand, M.; Alonso-Coello, P.; Falck-Ytter, Y.; Jaeschke, R.; Vist, G.; et al. GRADE guidelines: 8. Rating the quality of evidenc—Indirectness. J. Clin. Epidemiol. 2011, 64, 1303–1310. [Google Scholar] [CrossRef]
- Raskind, M.A.; Peskind, E.R.; Truyen, L.; Kershaw, P.; Damaraju, C.V. The Cognitive Benefits of Galantamine Are Sustained for at Least 36 Months: A Long-term Extension Trial. Arch. Neurol. 2004, 61, 252–256. [Google Scholar] [CrossRef] [Green Version]
- Hashimoto, M.; Kazui, H.; Matsumoto, K.; Nakano, Y.; Yasuda, M.; Mori, E. Does Donepezil Treatment Slow the Progression of Hippocampal Atrophy in Patients With Alzheimer’s Disease? Am. J. Psychiatry 2005, 162, 676–682. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Yu, J.T.; Wang, H.F.; Meng, X.F.; Wang, C.; Tan, C.C.; Tan, L. Pharmacological treatment of neuropsychiatric symptoms in Alzheimer’s disease: A systematic review and meta-analysis. J. Neurol. Neurosurg. Psychiatry 2015, 86, 101–109. [Google Scholar] [CrossRef]
- Bekelman, J.E.; Li, Y.; Gross, C.P. Scope and impact of financial conflicts of interest in biomedical research: A systematic review. JAMA 2003, 289, 454–465. [Google Scholar] [CrossRef] [PubMed]
- Lundh, A.; Lexchin, J.; Mintzes, B.; Schroll, J.B.; Bero, L. Industry sponsorship and research outcome. Cochrane Database Syst. Rev. 2017, 2, Mr000033. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lockhart, I.; Orme, M.; Mitchell, S.J.D.; Extra, G.C.D. The Efficacy of Licensed-Indication Use of Donepezil and Memantine Monotherapies for Treating Behavioural and Psychological Symptoms of Dementia in Patients with Alzheimer’s Disease: Systematic Review and Meta-Analysis. Dement. Geriatr. Cogn. Disord. Extra 2011, 1, 212–227. [Google Scholar] [CrossRef] [PubMed]
- Matsunaga, S.; Kishi, T.; Iwata, N. Combination Therapy with Cholinesterase Inhibitors and Memantine for Alzheimer’s Disease: A Systematic Review and Meta-Analysis. Int. J. Neuropsychopharmacol. 2015, 18, pyu115. [Google Scholar] [CrossRef]
- Glinz, D.; Gloy, V.; Monsch, A.; Kressig, R.; Patel, C.; McCord, K.; Ademi, Z.; Tomonaga, Y.; Schwenkglenks, M.; Bucher, H.; et al. Acetylcholinesterase inhibitors combined with memantine for moderate to severe Alzheimer’s disease: A meta-analysis. Swiss Med. Wkly. 2019, 149, w20093. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hogan, D.B.; Goldlist, B.; Naglie, G.; Patterson, C. Comparison studies of cholinesterase inhibitors for Alzheimer’s disease. Lancet Neurol. 2004, 3, 622–626. [Google Scholar] [CrossRef]
- Borm, G.F.; Donders, A.R. Updating meta-analyses leads to larger type I errors than publication bias. J. Clin. Epidemiol. 2009, 62, 825–830.e10. [Google Scholar] [CrossRef] [PubMed]
- Brok, J.; Thorlund, K.; Wetterslev, J.; Gluud, C. Apparently conclusive meta-analyses may be inconclusive—Trial sequential analysis adjustment of random error risk due to repetitive testing of accumulating data in apparently conclusive neonatal meta-analyses. Int. J. Epidemiol. 2009, 38, 287–298. [Google Scholar] [CrossRef]
- Garegnani, L.I.; Arancibia, M.; Madrid, E.; Franco, J.V.A. Clinical trials with sequential analysis that were early-stopped: How to interpret them? Medwave 2020, 20, e7930. [Google Scholar] [CrossRef]
- Leinonen, A.; Koponen, M.; Hartikainen, S. Systematic Review: Representativeness of Participants in RCTs of Acetylcholinesterase Inhibitors. PLoS ONE 2015, 10, e0124500. [Google Scholar] [CrossRef]
- Reisberg, B.; Ferris, S.H.; Franssen, E.H.; Shulman, E.; Monteiro, I.; Sclan, S.G.; Steinberg, G.; Kluger, A.; Torossian, C.; de Leon, M.J.; et al. Mortality and temporal course of probable Alzheimer’s disease: A 5-year prospective study. Int. Psychogeriatr. 1996, 8, 291–311. [Google Scholar] [CrossRef] [PubMed]
- Nordström, P.; Religa, D.; Wimo, A.; Winblad, B.; Eriksdotter, M. The use of cholinesterase inhibitors and the risk of myocardial infarction and death: A nationwide cohort study in subjects with Alzheimer’s disease. Eur. Heart J. 2013, 34, 2585–2591. [Google Scholar] [CrossRef] [PubMed]
- Kaushik, V.; Smith, S.T.; Mikobi, E.; Raji, M.A. Acetylcholinesterase Inhibitors: Beneficial Effects on Comorbidities in Patients With Alzheimer’s Disease. Am. J. Alzheimer’s Dis. Other Dement. 2017, 33, 73–85. [Google Scholar] [CrossRef]
- Molnar, F.J.; Man-Son-Hing, M.; Fergusson, D. Systematic review of measures of clinical significance employed in randomized controlled trials of drugs for dementia. J. Am. Geriatr. Soc. 2009, 57, 536–546. [Google Scholar] [CrossRef]
- Ebrahem, A.S.; Oremus, M. A pharmacoeconomic evaluation of cholinesterase inhibitors and memantine for the treatment of Alzheimer’s disease. Expert Opin. Pharmacother. 2018, 19, 1245–1259. [Google Scholar] [CrossRef]
- Satyanarayana, M. Aducanumab: A fate undecided. C EN Glob. Enterp. 2020, 98, 34. [Google Scholar] [CrossRef]
- Bautista-Aguilera, Ó.M.; Ismaili, L.; Iriepa, I.; Diez-Iriepa, D.; Chabchoub, F.; Marco-Contelles, J.; Pérez, M. Tacrines as Therapeutic Agents for Alzheimer’s Disease. V. Recent Developments. Chem. Rec. 2021, 21, 162–174. [Google Scholar] [CrossRef]
- Walker, A.C.; Bhargava, R.; Vaziriyan-Sani, A.S.; Pourciau, C.; Donahue, E.T.; Dove, A.S.; Gebhardt, M.J.; Ellward, G.L.; Romeo, T.; Czyż, D.M. Colonization of the Caenorhabditis elegans gut with human enteric bacterial pathogens leads to proteostasis disruption that is rescued by butyrate. PLoS Pathog. 2021, 17, e1009510. [Google Scholar] [CrossRef]
- Wilmanski, T.; Diener, C.; Rappaport, N.; Patwardhan, S.; Wiedrick, J.; Lapidus, J.; Earls, J.C.; Zimmer, A.; Glusman, G.; Robinson, M.; et al. Gut Microbiome Pattern Reflects Healthy Aging and Predicts Extended Survival in Humans. bioRxiv 2020. [Google Scholar] [CrossRef] [Green Version]
- De la Monte, S.M. Type 3 diabetes is sporadic Alzheimer׳s disease: Mini-review. Eur. Neuropsychopharmacol. J. Eur. Coll. Neuropsychopharmacol. 2014, 24, 1954–1960. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Oset-Gasque, M.J.; Marco-Contelles, J. Alzheimer’s Disease, the “One-Molecule, One-Target” Paradigm, and the Multitarget Directed Ligand Approach. ACS Chem. Neurosci. 2018, 9, 401–403. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Del Pino, J.; Marco-Contelles, J.; López-Muñoz, F.; Romero, A.; Ramos, E. Neuroinflammation Signaling Modulated by ASS234, a Multitarget Small Molecule for Alzheimer’s Disease Therapy. ACS Chem. Neurosci. 2018, 9, 2880–2885. [Google Scholar] [CrossRef] [PubMed]
 , if all answers are yes or probably yes. High:
, if all answers are yes or probably yes. High:  , if any is no or probably no. Not clear:
, if any is no or probably no. Not clear:  , insufficient data].
, insufficient data].
 , if all answers are yes or probably yes. High:
, if all answers are yes or probably yes. High:  , if any is no or probably no. Not clear:
, if any is no or probably no. Not clear:  , insufficient data].
, insufficient data].| Review ID | ROBIS Domain | Risk of Bias in the Review | |||
|---|---|---|---|---|---|
| Domain 1: Study Eligibility Criteria | Domain 2: Identification and Selection of Studies | Domain 3: Data Collection and Study Evaluation Domain | 4: Synthesis and Results | ||
| Birks, JS. 2016 [11] |  |  |  |  |  HIGH HIGH |
| Blanco-Silvente, L. et al. 2017 [18] |  |  |  |  |  LOW LOW |
| Bond, M. et al. 2012 [19] |  |  |  |  |  LOW LOW |
| Clegg, A. et al. 2002 [20] |  |  |  |  |  HIGH HIGH |
| Grimmer, T. et al. 2006 [21] |  |  |  | 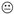 | 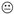 NOT CLEAR NOT CLEAR |
| Hansen, R. A. et al. 2008 [22] |  |  |  |  |  LOW LOW |
| Hyde, C. et al. 2013 [23] |  |  |  |  |  LOW LOW |
| Kaduszkiewicz. H. et al. 2005 [24] |  |  |  | 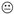 | 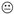 NOT CLEAR NOT CLEAR |
| Kobayashi, H. et al. 2016 [25] |  |  |  |  |  LOW LOW |
| Livingston, G. et al. 2000 [26] |  |  |  |  |  LOW LOW |
| Ritchie, C. W. et al. 2004 [27] |  |  |  |  |  LOW LOW |
| Takeda, A. et al. 2006 [28] |  |  |  |  |  LOW LOW |
| Trinh N. et al. 2003 [29] |  |  |  |  |  LOW LOW |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Moreta, M.P.-G.; Burgos-Alonso, N.; Torrecilla, M.; Marco-Contelles, J.; Bruzos-Cidón, C. Efficacy of Acetylcholinesterase Inhibitors on Cognitive Function in Alzheimer’s Disease. Review of Reviews. Biomedicines 2021, 9, 1689. https://doi.org/10.3390/biomedicines9111689
Moreta MP-G, Burgos-Alonso N, Torrecilla M, Marco-Contelles J, Bruzos-Cidón C. Efficacy of Acetylcholinesterase Inhibitors on Cognitive Function in Alzheimer’s Disease. Review of Reviews. Biomedicines. 2021; 9(11):1689. https://doi.org/10.3390/biomedicines9111689
Chicago/Turabian StyleMoreta, Marta Pérez-Gómez, Natalia Burgos-Alonso, María Torrecilla, José Marco-Contelles, and Cristina Bruzos-Cidón. 2021. "Efficacy of Acetylcholinesterase Inhibitors on Cognitive Function in Alzheimer’s Disease. Review of Reviews" Biomedicines 9, no. 11: 1689. https://doi.org/10.3390/biomedicines9111689
APA StyleMoreta, M. P.-G., Burgos-Alonso, N., Torrecilla, M., Marco-Contelles, J., & Bruzos-Cidón, C. (2021). Efficacy of Acetylcholinesterase Inhibitors on Cognitive Function in Alzheimer’s Disease. Review of Reviews. Biomedicines, 9(11), 1689. https://doi.org/10.3390/biomedicines9111689







