ReCODE: A Personalized, Targeted, Multi-Factorial Therapeutic Program for Reversal of Cognitive Decline
Abstract
:1. Introduction
2. Methods
2.1. Study Design and Participant Enrollment
2.2. The ReCODE Program
2.3. Statistical Analysis
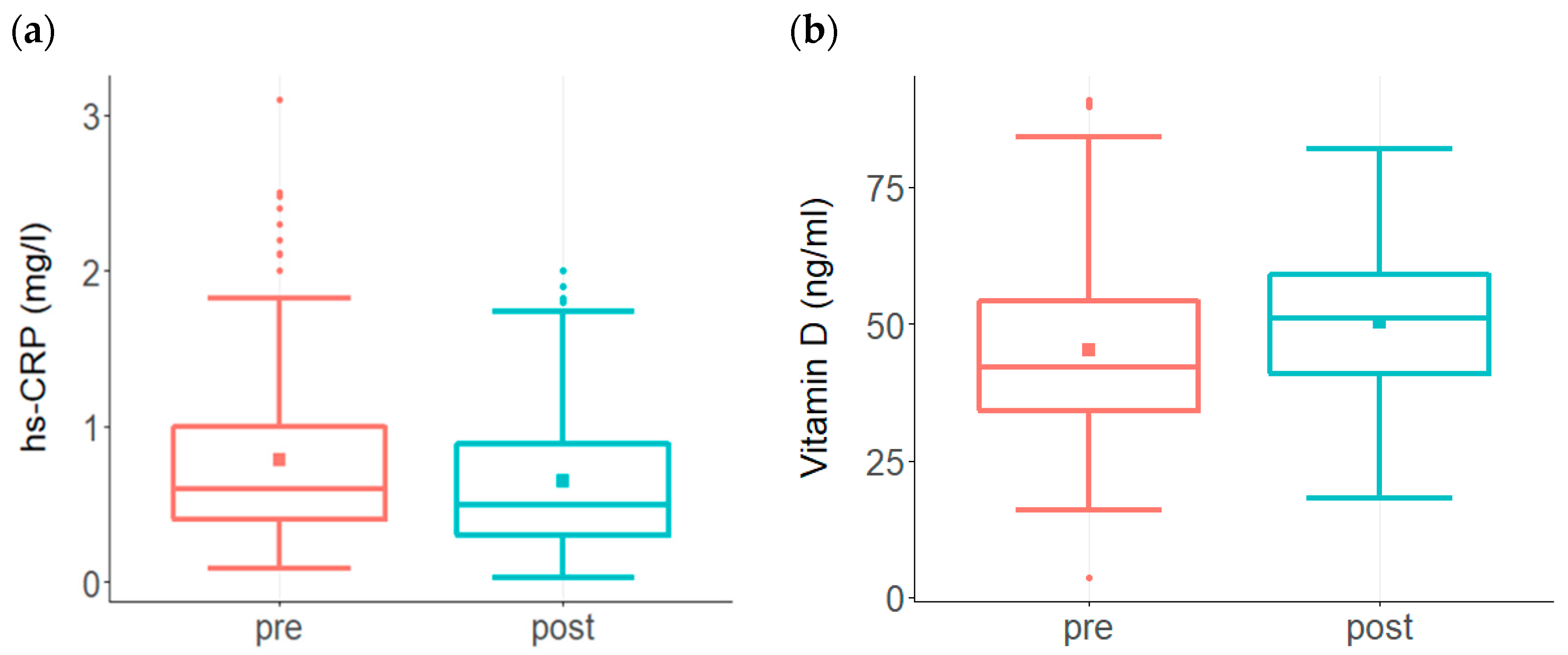
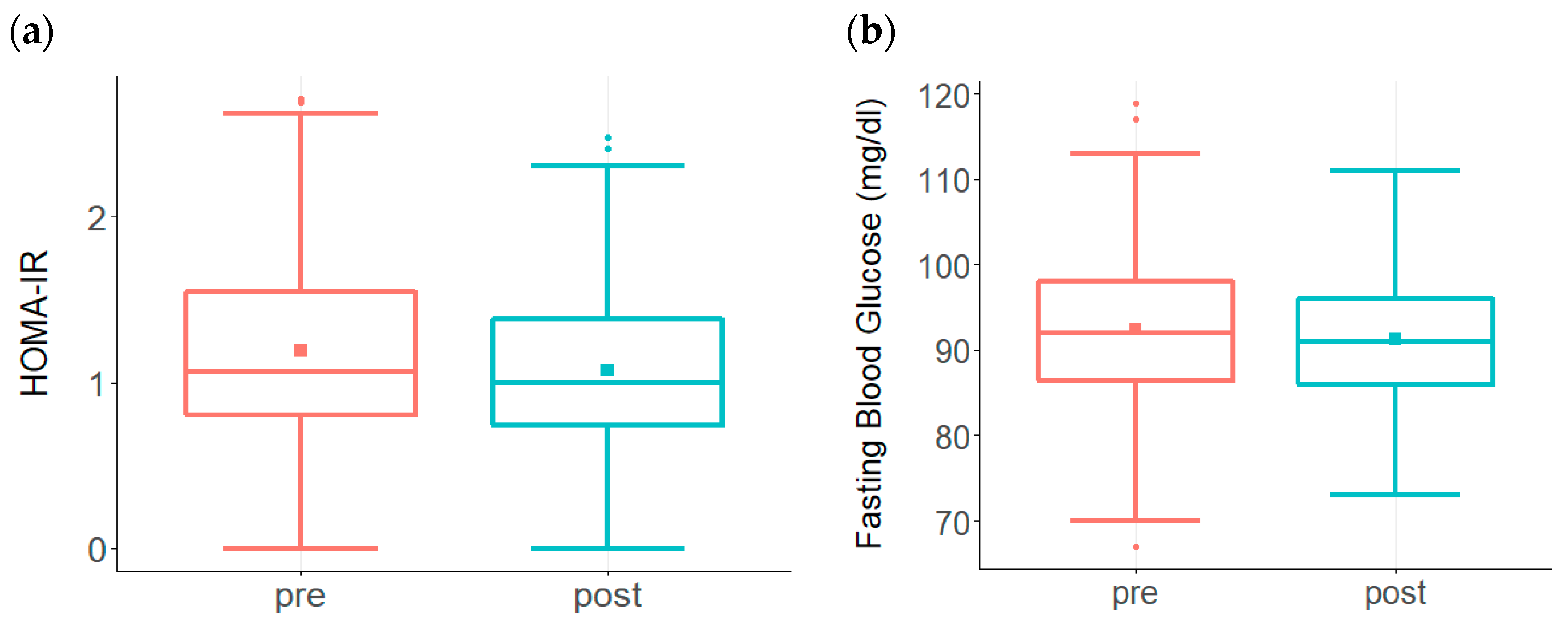
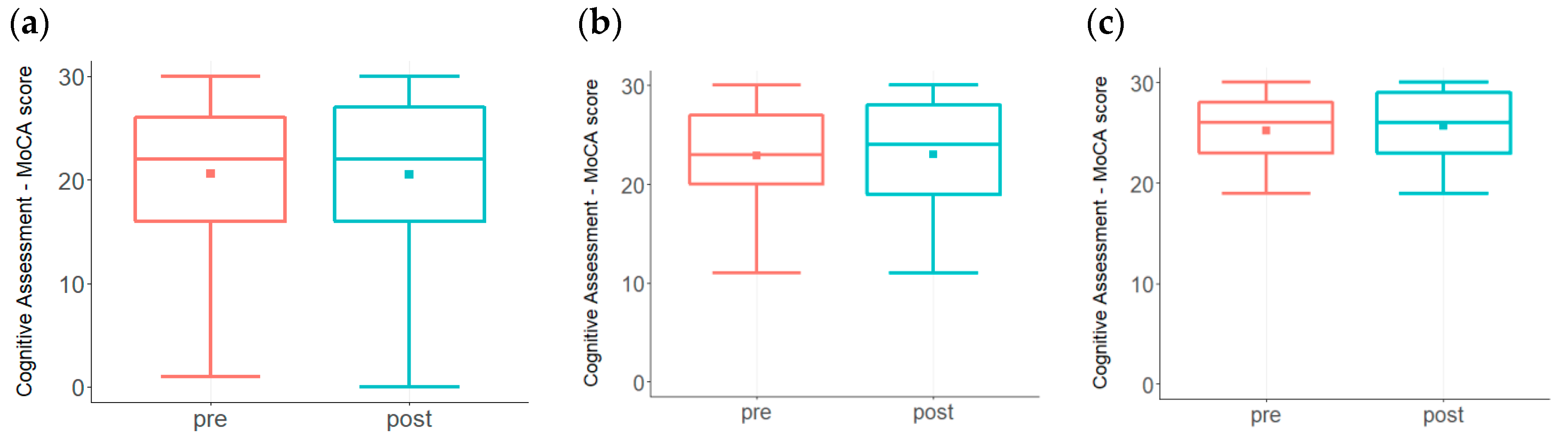
3. Results
4. Discussion & Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- 2021 Alzheimer’s disease facts and figures. J. Alzheimer’s Assoc. 2021, 17, 327–406. [CrossRef]
- El-Hayek, Y.H.; Wiley, R.E.; Khoury, C.P.; Daya, R.P.; Ballard, C.; Evans, A.R.; Karran, M.; Molinuevo, J.L.; Norton, M.; Atri, A. Tip of the Iceberg: Assessing the Global Socioeconomic Costs of Alzheimer’s Disease and Related Dementias and Strategic Implications for Stakeholders. J. Alzheimer’s Dis. 2019, 70, 323–341. [Google Scholar] [CrossRef] [Green Version]
- James, B.D.; Leurgans, S.E.; Hebert, L.E.; Scherr, P.A.; Yaffe, K.; Bennett, D.A. Contribution of Alzheimer disease to mortality in the United States. Neurology 2014, 82, 1045–1050. [Google Scholar] [CrossRef] [Green Version]
- Calabro, M.; Rinaldi, C.; Santoro, G.; Crisafulli, C. The biological pathways of Alzheimer disease: A review. AIMS Neurosci. 2021, 8, 86–132. [Google Scholar] [CrossRef] [PubMed]
- Peng, Y.; Gao, P.; Shi, L.; Chen, L.; Liu, J.; Long, J. Central and Peripheral Metabolic Defects Contribute to the Pathogenesis of Alzheimer’s Disease: Targeting Mitochondria for Diagnosis and Prevention. Antioxid. Redox Signal. 2020, 32, 1188–1236. [Google Scholar] [CrossRef] [PubMed]
- Merlo, S.; Spampinato, S.; Canonico, P.L.; Copani, A.; Sortino, M.A. Alzheimer’s disease: Brain expression of a metabolic disorder? Trends Endocrinol. Metab. 2010, 21, 537–544. [Google Scholar] [CrossRef] [PubMed]
- Ishii, M.; Iadecola, C. Metabolic and Non-Cognitive Manifestations of Alzheimer’s Disease: The Hypothalamus as Both Culprit and Target of Pathology. Cell Metab. 2015, 22, 761–776. [Google Scholar] [CrossRef] [Green Version]
- Bredesen, D.E. Reversal of cognitive decline: A novel therapeutic program. Aging (Albany NY) 2014, 6, 707–717. [Google Scholar] [CrossRef] [Green Version]
- Theendakara, V.; Peters-Libeu, C.A.; Spilman, P.; Poksay, K.S.; Bredesen, D.E.; Rao, R.V. Direct Transcriptional Effects of Apolipoprotein E. J. Neurosci. 2016, 36, 685–700. [Google Scholar] [CrossRef] [Green Version]
- Folch, J.; Petrov, D.; Ettcheto, M.; Abad, S.; Sanchez-Lopez, E.; Garcia, M.L.; Olloquequi, J.; Beas-Zarate, C.; Auladell, C.; Camins, A. Current Research Therapeutic Strategies for Alzheimer’s Disease Treatment. Neural Plast. 2016, 2016, 8501693. [Google Scholar] [CrossRef] [Green Version]
- Ngandu, T.; Lehtisalo, J.; Solomon, A.; Levalahti, E.; Ahtiluoto, S.; Antikainen, R.; Backman, L.; Hanninen, T.; Jula, A.; Laatikainen, T.; et al. A 2 year multidomain intervention of diet, exercise, cognitive training, and vascular risk monitoring versus control to prevent cognitive decline in at-risk elderly people (FINGER): A randomised controlled trial. Lancet 2015, 385, 2255–2263. [Google Scholar] [CrossRef]
- Schechter, G.; Azad, G.K.; Rao, R.; McKeany, A.; Matulaitis, M.; Kalos, D.M.; Kennedy, B.K. A Comprehensive, Multi-Modal Strategy to Mitigate Alzheimer’s Disease Risk Factors Improves Aspects of Metabolism and Offsets Cognitive Decline in Individuals with Cognitive Impairment. J. Alzheimer’s Dis. Rep. 2020, 4, 223–230. [Google Scholar] [CrossRef]
- Bredesen, D.E.; Sharlin, K.; Jenkins, D.; Okuno, M.; Youngberg, W.; Cohen, S.H.; Stefani, A.; Brown, R.L.; Conger, S.; Tanio, C.; et al. Reversal of Cognitive Decline: 100 Patients. J. Alzheimer’s Dis. Parkinsonism 2018, 8, 1–6. [Google Scholar] [CrossRef]
- Toups, K.; Hathaway, A.; Gordon, D.; Chung, H.; Raji, C.; Boyd, A.; Hill, B.D.; Hausman-Cohen, S.; Attarha, M.; Chwa, W.J.; et al. Precision Medicine Approach to Alzheimer’s Disease: Successful Proof-of-Concept Trial. medRxiv 2021. [Google Scholar] [CrossRef]
- Bredesen, D.E.; Amos, E.C.; Canick, J.; Ackerley, M.; Raji, C.; Fiala, M.; Ahdidan, J. Reversal of cognitive decline in Alzheimer’s disease. Aging (Albany NY) 2016, 8, 1250–1258. [Google Scholar] [CrossRef] [Green Version]
- Theendakara, V.; Patent, A.; Peters Libeu, C.A.; Philpot, B.; Flores, S.; Descamps, O.; Poksay, K.S.; Zhang, Q.; Cailing, G.; Hart, M.; et al. Neuroprotective Sirtuin ratio reversed by ApoE4. Proc. Natl. Acad. Sci. USA 2013, 110, 18303–18308. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Theendakara, V.; Peters-Libeu, C.A.; Bredesen, D.E.; Rao, R.V. Transcriptional Effects of ApoE4: Relevance to Alzheimer’s Disease. Mol. Neurobiol. 2017. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, R.; Schmidt, H.; Curb, J.D.; Masaki, K.; White, L.R.; Launer, L.J. Early inflammation and dementia: A 25-year follow-up of the Honolulu-Asia Aging Study. Ann. Neurol. 2002, 52, 168–174. [Google Scholar] [CrossRef]
- Noble, J.M.; Manly, J.J.; Schupf, N.; Tang, M.X.; Mayeux, R.; Luchsinger, J.A. Association of C-reactive protein with cognitive impairment. Arch. Neurol. 2010, 67, 87–92. [Google Scholar] [CrossRef]
- Luan, Y.Y.; Yao, Y.M. The Clinical Significance and Potential Role of C-Reactive Protein in Chronic Inflammatory and Neurodegenerative Diseases. Front. Immunol. 2018, 9, 1302. [Google Scholar] [CrossRef] [Green Version]
- Bredesen, D.E. Metabolic profiling distinguishes three subtypes of Alzheimer’s disease. Aging (Albany NY) 2015, 7, 595–600. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chai, B.; Gao, F.; Wu, R.; Dong, T.; Gu, C.; Lin, Q.; Zhang, Y. Vitamin D deficiency as a risk factor for dementia and Alzheimer’s disease: An updated meta-analysis. BMC Neurol. 2019, 19, 284. [Google Scholar] [CrossRef] [PubMed]
- Littlejohns, T.J.; Henley, W.E.; Lang, I.A.; Annweiler, C.; Beauchet, O.; Chaves, P.H.; Fried, L.; Kestenbaum, B.R.; Kuller, L.H.; Langa, K.M.; et al. Vitamin D and the risk of dementia and Alzheimer disease. Neurology 2014, 83, 920–928. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ferreira, L.S.S.; Fernandes, C.S.; Vieira, M.N.N.; De Felice, F.G. Insulin Resistance in Alzheimer’s Disease. Front. Neurosci. 2018, 12, 830. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- de la Monte, S.M. Insulin resistance and Alzheimer’s disease. BMB Rep. 2009, 42, 475–481. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, T.T.; Ta, Q.T.H.; Nguyen, T.K.O.; Nguyen, T.T.D.; Giau, V.V. Type 3 Diabetes and Its Role Implications in Alzheimer’s Disease. Int. J. Mol. Sci. 2020, 21, 3165. [Google Scholar] [CrossRef]
- Hoscheidt, S.M.; Starks, E.J.; Oh, J.M.; Zetterberg, H.; Blennow, K.; Krause, R.A.; Gleason, C.E.; Puglielli, L.; Atwood, C.S.; Carlsson, C.M.; et al. Insulin Resistance is Associated with Increased Levels of Cerebrospinal Fluid Biomarkers of Alzheimer’s Disease and Reduced Memory Function in At-Risk Healthy Middle-Aged Adults. J. Alzheimers Dis. 2016, 52, 1373–1383. [Google Scholar] [CrossRef] [Green Version]
- Morris, J.C. Mild cognitive impairment and preclinical Alzheimer’s disease. Geriatrics 2005, 9–14. [Google Scholar]
- Kelley, B.J.; Petersen, R.C. Alzheimer’s disease and mild cognitive impairment. Neurol. Clin. 2007, 25, 577–609. [Google Scholar] [CrossRef] [Green Version]
- Bredesen, D.E. Inhalational Alzheimer’s disease: An unrecognized-and treatable-epidemic. Aging (Albany NY) 2016, 8, 304–313. [Google Scholar] [CrossRef]
- Smith, T.; Gildeh, N.; Holmes, C. The Montreal Cognitive Assessment: Validity and utility in a memory clinic setting. Can. J. Psychiatry 2007, 52, 329–332. [Google Scholar] [CrossRef] [PubMed]
- Krishnan, K.; Rossetti, H.; Hynan, L.S.; Carter, K.; Falkowski, J.; Lacritz, L.; Cullum, C.M.; Weiner, M. Changes in Montreal Cognitive Assessment Scores Over Time. Assessment 2017, 24, 772–777. [Google Scholar] [CrossRef] [PubMed]
| Categories | Parameter |
|---|---|
| General Health | BMI |
| Assessments | MoCA score |
| Inflammation | A/G Ratio, IL-6, TNF-Alpha, hs-CRP |
| Methylation | Folate, Homocysteine, Vitamin B12, Vitamin B6 |
| Hormones | Cortisol, DHEA-Sulfate, Estradiol, T3, T4, TSH, Testosterone, Pregnenolone, Progesterone, Vitamin D |
| Metals | Potassium, Magnesium, Copper, Zinc |
| Others | Lipid panel, Chemistry panel, Toxins (Organics and Heavy Metals), Cytoprotection, Antioxidants, Mycotoxins |
| Total Subjects | n = 255 |
|---|---|
| Males | 111 |
| Females | 144 |
| Average age | 73 |
| ApoE4-absent | 89 |
| ApoE4-1 copy | 104 |
| ApoE4-2 copies | 40 |
| Parameter | Target Range |
|---|---|
| MoCA | 26–30 |
| hs-CRP | <0.9 mg/L |
| Vitamin D | 40–80 ng/mL |
| HOMA-IR | ≤1.2 |
| Fasting Glucose | 70–90 mg/dL |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rao, R.V.; Kumar, S.; Gregory, J.; Coward, C.; Okada, S.; Lipa, W.; Kelly, L.; Bredesen, D.E. ReCODE: A Personalized, Targeted, Multi-Factorial Therapeutic Program for Reversal of Cognitive Decline. Biomedicines 2021, 9, 1348. https://doi.org/10.3390/biomedicines9101348
Rao RV, Kumar S, Gregory J, Coward C, Okada S, Lipa W, Kelly L, Bredesen DE. ReCODE: A Personalized, Targeted, Multi-Factorial Therapeutic Program for Reversal of Cognitive Decline. Biomedicines. 2021; 9(10):1348. https://doi.org/10.3390/biomedicines9101348
Chicago/Turabian StyleRao, Rammohan V, Sharanya Kumar, Julie Gregory, Christine Coward, Sho Okada, William Lipa, Lance Kelly, and Dale E Bredesen. 2021. "ReCODE: A Personalized, Targeted, Multi-Factorial Therapeutic Program for Reversal of Cognitive Decline" Biomedicines 9, no. 10: 1348. https://doi.org/10.3390/biomedicines9101348
APA StyleRao, R. V., Kumar, S., Gregory, J., Coward, C., Okada, S., Lipa, W., Kelly, L., & Bredesen, D. E. (2021). ReCODE: A Personalized, Targeted, Multi-Factorial Therapeutic Program for Reversal of Cognitive Decline. Biomedicines, 9(10), 1348. https://doi.org/10.3390/biomedicines9101348





