Progress in Investigating the Impact of Obesity on Male Reproductive Function
Abstract
1. Introduction
2. Classification and Distribution of Adipose Tissue
3. The Impact of Obesity on Male Fertility
3.1. The Impact of Obesity on Spermatogenesis and Sperm Quality
3.2. The Impact of Obesity on Erectile Dysfunction
3.3. The Impact of Male Obesity on Offspring
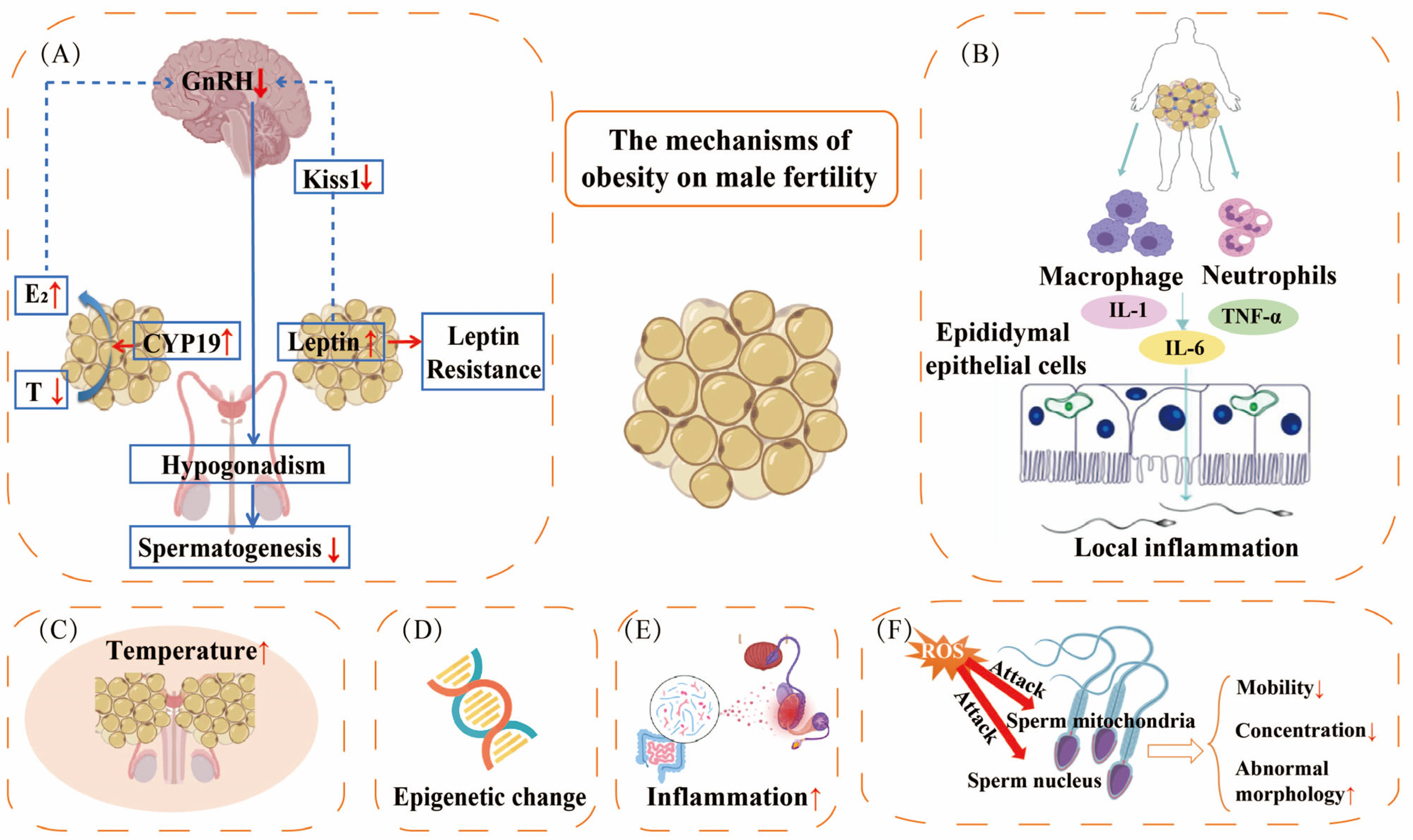
4. Mechanisms Through Which Obesity Impacts Male Fertility
4.1. Endocrine Disorder
4.2. Inflammation of the Reproductive System
4.3. Elevated Localized Heat in the Reproductive System
4.4. Epigenetic Modification
4.4.1. Impact of Obesity on Sperm DNA Methylation
4.4.2. Impact of Obesity on Spermatozoa Through Histone Modification
4.4.3. The Impact of Obesity on Sperm RNA
4.5. Intestinal Microbiota
4.6. Oxidative Stress
5. Intervention Measures
5.1. Weight Loss
5.2. Weight Loss Drugs
5.3. Regulate Intestinal Microbiota
5.4. Improve Oxidative Stress
6. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Leslie, S.W.; Soon-Sutton, T.L.; Khan, M.A.B. Male Infertility. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2024. [Google Scholar]
- Weidner, W.; Colpi, G.; Hargreave, T.; Papp, G.; Pomerol, J. EAU guidelines on male infertility. Eur. Urol. 2002, 42, 313–322. [Google Scholar] [CrossRef] [PubMed]
- Sciorio, R.; Greco, P.F.; Greco, E.; Tramontano, L.; Elshaer, F.M.; Fleming, S. Potential effects of environmental toxicants on sperm quality and potential risk for fertility in humans. Front. Endocrinol. 2025, 16, 1545593. [Google Scholar] [CrossRef] [PubMed]
- Cui, J.; Wang, C.; Zheng, Y.; Zhang, Y.; Luo, S.; Ren, Z.; Qin, X.; Zhang, M.; Gao, F.; Huang, H.; et al. Mechanisms and reversibility of nicotine-induced spermatogenesis impairment and DNA methylation changes. Commun. Biol. 2025, 8, 1053. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.K.; Choi, W.Y.; Lee, J.I.; Kim, T.J. Impact of conventional cigarette and electronic cigarette use on sperm quality and IVF/ICSI outcomes. Sci. Rep. 2025, 15, 23714. [Google Scholar] [CrossRef]
- Finelli, R.; Mottola, F.; Agarwal, A. Impact of alcohol consumption on male fertility potential: A narrative review. Int. J. Environ. Res. Public Health 2021, 19, 328. [Google Scholar] [CrossRef]
- Barbouni, K.; Jotautis, V.; Metallinou, D.; Diamanti, A.; Orovou, E.; Liepinaitienė, A.; Nikolaidis, P.; Karampas, G.; Sarantaki, A. When Weight Matters: How Obesity Impacts Reproductive Health and Pregnancy-A Systematic Review. Curr. Obes. Rep. 2025, 14, 1–24. [Google Scholar] [CrossRef]
- Khorasani, F.; Iranifard, E.; Roudsari, R.L. Gender differences in psychological status of infertile couples: A systematic review and meta-analysis. BMC Public Health 2025, 25, 2131. [Google Scholar] [CrossRef]
- Reddy, A.G.; Williams, P.L.; Souter, I.; Ford, J.B.; Dadd, R.; Abou-Ghayda, R.; Hauser, R.; Chavarro, J.E.; Mínguez-Alarcón, L. Perceived stress in relation to testicular function markers among men attending a fertility center. Fertil. Steril. 2025, 124, 62–70. [Google Scholar] [CrossRef]
- McPherson, N.O.; Lane, M. Male obesity and subfertility, is it really about increased adiposity? Asian J. Androl. 2015, 17, 450. [Google Scholar] [CrossRef]
- GBD 2015 Obesity Collaborators. Health effects of overweight and obesity in 195 countries over 25 years. N. Engl. J. Med. 2017, 377, 13–27. [Google Scholar] [CrossRef]
- WHO. World Health Organization Obesity and Overweight. Available online: https://www.who.int/news-room/fact-sheets/detail/obesity-and-overweight (accessed on 22 August 2025).
- Rosen, E.D.; Spiegelman, B.M. What we talk about when we talk about fat. Cell 2014, 156, 20–44. [Google Scholar] [CrossRef]
- Powell-Wiley, T.M.; Poirier, P.; Burke, L.E.; Després, J.-P.; Gordon-Larsen, P.; Lavie, C.J.; Lear, S.A.; Ndumele, C.E.; Neeland, I.J.; Sanders, P.; et al. Obesity and cardiovascular disease: A scientific statement from the American Heart Association. Circulation 2021, 143, e984–e1010. [Google Scholar] [CrossRef] [PubMed]
- Kwak, J.; Shin, D. Gene–Nutrient Interactions in Obesity: COBLL1 Genetic Variants Interact with Dietary Fat Intake to Modulate the Incidence of Obesity. Int. J. Mol. Sci. 2023, 24, 3758. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Chen, L.; Xuan, Y.; Zhang, L.; Tian, W.; Zhu, Y.; Wang, J.; Wang, X.; Qiu, J.; Yu, J.; et al. Iron overload in hypothalamic AgRP neurons contributes to obesity and related metabolic disorders. Cell Rep. 2024, 43, 113900. [Google Scholar] [CrossRef] [PubMed]
- Cordeiro, B.; Ahn, J.J.; Gawde, S.; Ucciferri, C.; Alvarez-Sanchez, N.; Revelo, X.S.; Stickle, N.; Massey, K.; Brooks, D.G.; Guthridge, J.M.; et al. Obesity intensifies sex-specific interferon signaling to selectively worsen central nervous system autoimmunity in females. Cell Metab. 2024, 36, 2298–2314.e11. [Google Scholar] [CrossRef]
- İncedal Irgat, S.; Bakirhan, H. The effect of obesity on human reproductive health and foetal life. Hum. Fertil. 2022, 25, 860–871. [Google Scholar] [CrossRef]
- Kozopas, N.M.; Chornenka, O.I.; Vorobets, M.Z.; Lapovets, L.Y.; Maksymyuk, H.V. Body mass index and sperm quality: Is there a relationship? J. Hum. Reprod. Sci. 2020, 13, 110–113. [Google Scholar] [CrossRef]
- Nikolic, A.Z.; Dragojevic-Dikic, S.; Kocic, J.; Babic, U.; Joksimovic, A.; Radakovic-Cosic, J.; Vladimir, G.; Danijela, S.; Stefan, D.; Aleksandra, P.; et al. Influence of male body mass index on semen analysis parameters and in vitro f20ertilization outcomes. Medicine 2024, 103, e38949. [Google Scholar] [CrossRef]
- Kurowska, P.; Mlyczyńska, E.; Dawid, M.; Respekta, N.; Pich, K.; Serra, L.; Dupont, J.; Rak, A. Endocrine disruptor chemicals, adipokines and reproductive functions. Endocrine 2022, 78, 205–218. [Google Scholar] [CrossRef]
- Ho, J.H.; Adam, S.; Azmi, S.; Ferdousi, M.; Liu, Y.; Kalteniece, A.; Dhage, S.S.; Keevil, B.G.; Syed, A.A.; Ammori, B.J.; et al. Male sexual dysfunction in obesity: The role of sex hormones and small fibre neuropathy. PLoS ONE 2019, 14, e0221992. [Google Scholar] [CrossRef]
- Elmas, M.A.; Ozakpinar, O.B.; Kolgazi, M.; Sener, G.; Arbak, S.; Ercan, F. Exercise improves testicular morphology and oxidative stress parameters in rats with testicular damage induced by a high-fat diet. Andrologia 2022, 54, e14600. [Google Scholar] [CrossRef]
- Lalrinzuali, S.; Khushboo, M.; Dinata, R.; Bhanushree, B.; Nisa, N.; Bidanchi, R.M.; Laskar, S.-A.; Manikandan, B.; Abinash, G.; Pori, B.; et al. Long-term consumption of fermented pork fat-based diets differing in calorie, fat content, and fatty acid levels mediates oxidative stress, inflammation, redox imbalance, germ cell apoptosis, disruption of steroidogenesis, and testicular dysfunction in Wistar rats. Environ. Sci. Pollut. Res. 2023, 30, 52446–52471. [Google Scholar]
- Bakos, H.W.; Mitchell, M.; Setchell, B.P.; Lane, M. The effect of paternal diet-induced obesity on sperm function and fertilization in a mouse model. Int. J. Androl. 2011, 34, 402–410. [Google Scholar] [CrossRef]
- Craig, J.R.; Jenkins, T.G.; Carrell, D.T.; Hotaling, J.M. Obesity, male infertility, and the sperm epigenome. Fertil. Steril. 2017, 107, 848–859. [Google Scholar] [CrossRef] [PubMed]
- Jokinen, R.; Pirnes-Karhu, S.; Pietiläinen, K.H.; Pirinen, E. Adipose tissue NAD+-homeostasis, sirtuins and poly (ADP-ribose) polymerases-important players in mitochondrial metabolism and metabolic health. Redox Biol. 2017, 12, 246–263. [Google Scholar] [CrossRef] [PubMed]
- He, N.; Liu, M.; Wu, Y. Adipose tissue and hematopoiesis: Friend or foe? J. Clin. Lab. Anal. 2023, 37, e24872. [Google Scholar] [CrossRef] [PubMed]
- Kawai, T.; Autieri, M.V.; Scalia, R. Adipose tissue inflammation and metabolic dysfunction in obesity. Am. J. Physiol.-Cell Physiol. 2021, 320, C375–C391. [Google Scholar] [CrossRef]
- Choubey, M.; Ranjan, A.; Krishna, A. Adiponectin/AdipoRs signaling as a key player in testicular aging and associated metabolic disorders. In Vitamins and Hormones; Academic Press: Cambridge, MA, USA, 2021; Volume 115, pp. 611–634. [Google Scholar]
- Fazeli, S.A.; Nourollahi, S.; Alirezaei, A.; Mirhashemi, S.; Davarian, A.; Hosseini, I. Perirenal adipose tissue: Clinical implication and therapeutic interventions. Indian J. Nephrol. 2024, 34, 573–582. [Google Scholar] [CrossRef]
- Takeda, Y.; Harada, Y.; Yoshikawa, T.; Dai, P. Mitochondrial Energy Metabolism in the Regulation of Thermogenic Brown Fats and Human Metabolic Diseases. Int. J. Mol. Sci. 2023, 24, 1352. [Google Scholar] [CrossRef]
- Chouchani, E.T.; Kajimura, S. Metabolic adaptation and maladaptation in adipose tissue. Nat. Metab. 2019, 1, 189–200. [Google Scholar] [CrossRef]
- González-García, I.; Tena-Sempere, M.; López, M. Estradiol regulation of brown adipose tissue thermogenesis. Sex Gend. Factors Affect. Metab. Homeost. Diabetes Obes. 2017, 1043, 315–335. [Google Scholar]
- White, U. Adipose tissue expansion in obesity, health, and disease. Front. Cell Dev. Biol. 2023, 11, 1188844. [Google Scholar] [CrossRef] [PubMed]
- Reyes-Farias, M.; Fos-Domenech, J.; Serra, D.; Herrero, L.; Sánchez-Infantes, D. White adipose tissue dysfunction in obesity and aging. Biochem. Pharmacol. 2021, 192, 114723. [Google Scholar] [CrossRef] [PubMed]
- Stanford, K.I.; Goodyear, L.J. The therapeutic potential of brown adipose tissue. Hepatobiliary Surg. Nutr. 2013, 2, 286–287. [Google Scholar] [PubMed]
- Lee, P.; Werner, C.D.; Kebebew, E.; Celi, F.S. Functional thermogenic beige adipogenesis is inducible in human neck fat. Int. J. Obes. 2014, 38, 170–176. [Google Scholar] [CrossRef]
- Cheng, L.; Wang, J.; Dai, H.; Duan, Y.; An, Y.; Shi, L.; Lv, Y.; Li, H.; Wang, C.; Ma, Q.; et al. Brown and beige adipose tissue: A novel therapeutic strategy for obesity and type 2 diabetes mellitus. Adipocyte 2021, 10, 48–65. [Google Scholar] [CrossRef]
- Rabiee, A. Beige fat maintenance; toward a sustained metabolic health. Front. Endocrinol. 2020, 11, 634. [Google Scholar] [CrossRef]
- Gurung, P.; Yetiskul, E.; Jialal, I. Physiology, male reproductive system. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2021. [Google Scholar] [PubMed]
- Zhang, L.F.; Ma, F.; Liu, J.F.; Feng, Y. Impact of high temperature on sperm function: An update. Natl. J. Androl. 2019, 25, 843–847. [Google Scholar]
- Chang, E.; Varghese, M.; Singer, K. Gender and Sex Differences in Adipose Tissue. Curr. Diabetes Rep. 2018, 18, 69. [Google Scholar] [CrossRef]
- Blaak, E. Gender differences in fat metabolism. Curr. Opin. Clin. Nutr. Metab. Care 2001, 4, 499–502. [Google Scholar] [CrossRef]
- Chusyd, D.E.; Wang, D.; Huffman, D.M.; Nagy, T.R. Relationships between rodent white adipose fat pads and human white adipose fat depots. Front. Nutr. 2016, 3, 10. [Google Scholar] [CrossRef]
- Altintas, M.M.; Rossetti, M.A.; Nayer, B.; Puig, A.; Zagallo, P.; Ortega, L.M.; Johnson, K.B.; McNamara, G.; Reiser, J.; Mendez, A.J. Apoptosis, mastocytosis, and diminished adipocytokine gene expression accompany reduced epididymal fat mass in long-standing diet-induced obese mice. Lipids Health Dis. 2011, 10, 198. [Google Scholar] [CrossRef]
- Chu, Y.; Huddleston, G.G.; Clancy, A.N.; Harris, R.B.; Bartness, T.J. Epididymal fat is necessary for spermatogenesis, but not testosterone production or copulatory behavior. Endocrinology 2010, 151, 5669–5679. [Google Scholar] [CrossRef]
- Crean, A.J.; Senior, A.M. High-fat diets reduce male reproductive success in animal models: A systematic review and meta-analysis. Obes Rev 2019, 20, 921–933. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Lin, Y.; Ou, C.; Xu, R.; Liu, T.; Zhong, Z.; Liu, L.; Zheng, Y.; Hou, S.; Lv, Z.; et al. Association between body mass index and semen quality: A systematic review and meta-analysis. Int. J. Obes. 2024, 48, 1383–1401. [Google Scholar] [CrossRef] [PubMed]
- Lu, J.C.; Jing, J.; Chen, L.; Ge, Y.F.; Feng, R.X.; Liang, Y.J.; Yao, B. Analysis of human sperm DNA fragmentation index (DFI) related factors: A report of 1010 subfertile men in China. Reprod. Biol. Endocrinol. 2018, 16, 23. [Google Scholar] [CrossRef] [PubMed]
- Manfrevola, F.; Chioccarelli, T.; Mele, V.G.; Porreca, V.; Mattia, M.; Cimini, D.; D’agostino, A.; Cobellis, G.; Fasano, S.; Schiraldi, C.; et al. Novel insights into circRNA saga coming from spermatozoa and epididymis of HFD mice. Int. J. Mol. Sci. 2023, 24, 6865. [Google Scholar] [CrossRef]
- Jing, J.; Peng, Y.; Fan, W.; Han, S.; Peng, Q.; Xue, C.; Qin, X.; Liu, Y.; Ding, Z. Obesity-induced oxidative stress and mitochondrial dysfunction negatively affect sperm quality. FEBS Open Bio 2023, 13, 763–778. [Google Scholar] [CrossRef]
- Oliveira, P.F.; Sousa, M.; Silva, B.M.; Monteiro, M.P.; Alves, M.G. Obesity, energy balance and spermatogenesis. Reproduction 2017, 153, R173–R185. [Google Scholar] [CrossRef]
- Komninos, D.; Ramos, L.; Van der Heijden, G.W.; Morrison, M.C.; Kleemann, R.; Van Herwaarden, A.E.; Kiliaan, A.J.; Arnoldussen, I.A.C. High fat diet-induced obesity prolongs critical stages of the spermatogenic cycle in a Ldlr−/−. Leiden mouse model. Sci. Rep. 2022, 12, 430. [Google Scholar] [CrossRef]
- Raad, G.; Azouri, J.; Rizk, K.; Zeidan, N.S.; Azouri, J.; Grandjean, V.; Hazzouri, M.; Drevet, J.R. Adverse effects of paternal obesity on the motile spermatozoa quality. PLoS ONE 2019, 14, e0211837. [Google Scholar] [CrossRef]
- Aleksandra, R.; Aleksandra, S.; Iwona, R. Erectile Dysfunction in Relation to Metabolic Disorders and the Concentration of Sex Hormones in Aging Men. Int. J. Environ. Res. Public Health 2022, 19, 7576. [Google Scholar] [CrossRef]
- Xu, M.; Zhou, H.; Zhang, R.; Pan, Y.; Liu, X. Correlation between visceral adiposity index and erectile dysfunction in American adult males: A cross-sectional study based on NHANES. Front. Endocrinol. 2023, 14, 1301284. [Google Scholar] [CrossRef]
- Huang, L.; Liu, H.; Li, L.; Wang, S.; Sun, G. Correlation between visceral fat metabolism score and erectile dysfunction: A cross-sectional study from NHANES 2001-2004. Front. Endocrinol. 2023, 14, 1283545. [Google Scholar] [CrossRef]
- Deng, C.Y.; Ke, X.P.; Guo, X.G. Investigating a novel surrogate indicator of adipose accumulation in relation to erectile dysfunction. Lipids Health Dis. 2024, 23, 139. [Google Scholar] [CrossRef] [PubMed]
- Moon, K.H.; Park, S.Y.; Kim, Y.W. Obesity and erectile dysfunction: From bench to clinical implication. World J. Men’s Health 2019, 37, 138–147. [Google Scholar] [CrossRef] [PubMed]
- Kaya, E.; Sikka, S.C.; Gur, S. A comprehensive review of metabolic syndrome affecting erectile dysfunction. J. Sex. Med. 2015, 12, 856–875. [Google Scholar] [CrossRef] [PubMed]
- Pizzol, D.; Smith, L.; Fontana, L.; Caruso, M.G.; Bertoldo, A.; Demurtas, J.; McDermott, D.; Garolla, A.; Grabovac, I. Associations between body mass index, waist circumference and erectile dys-function: A systematic review and META-analysis. Rev. Endocr. Metab. Disord. 2020, 21, 657–666. [Google Scholar] [CrossRef]
- Hannan, J.L.; Maio, M.T.; Komolova, M.; Adams, M.A. Beneficial impact of exercise and obesity interventions on erectile function and its risk factors. J. Sex. Med. 2009, 6 (Suppl. S3), 254–261. [Google Scholar] [CrossRef]
- Miñambres, I.; Sardà, H.; Urgell, E.; Genua, I.; Ramos, A.; Fernández-Ananin, S.; Balagué, C.; Sánchez-Quesada, J.L.; Bassas, L.; Pérez, A. Obesity Surgery Improves Hypogonadism and Sexual Function in Men without Effects in Sperm Quality. J. Clin. Med. 2022, 11, 5126. [Google Scholar] [CrossRef]
- Sertorio, M.N.; Estadella, D.; Ribeiro, D.A.; Pisani, L.P. Could parental high-fat intake program the reproductive health of male offspring? A review. Crit. Rev. Food Sci. Nutr. 2021, 63, 2074–2081. [Google Scholar] [CrossRef]
- Binder, N.K.; Hannan, N.J.; Gardner, D.K.; Kappen, C. Paternal diet-induced obesity retards early mouse embryo development, mitochondrial activity and pregnancy health. PLoS ONE 2012, 7, e52304. [Google Scholar] [CrossRef] [PubMed]
- Bernhardt, L.; Dittrich, M.; El-Merahbi, R.; Saliba, A.-E.; Müller, T.; Sumara, G.; Vogel, J.; Nichols-Burns, S.; Mitchell, M.; Haaf, T.; et al. A genome-wide transcriptomic analysis of embryos fathered by obese males in a murine model of diet-induced obesity. Sci. Rep. 2021, 11, 1979. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Ma, Y.; Zhu, C.; Li, Y.; Cao, H.; Wu, Z.; Jin, T.; Wang, Y.; Chen, S.; Dong, W. Paternal obesity induces subfertility in male offspring by modulating the oxidative stress-related transcriptional network. Int. J. Obes. 2024, 48, 1318–1331. [Google Scholar] [CrossRef] [PubMed]
- Di Vincenzo, A.; Busetto, L.; Vettor, R.; Rossato, M. Obesity, male reproductive function and bariatric surgery. Front. Endocrinol. 2018, 9, 769. [Google Scholar] [CrossRef]
- Bellastella, G.; Menafra, D.; Puliani, G.; Colao, A.; Savastano, S. How much does obesity affect the male reproductive function? Int. J. Obes. Suppl. 2019, 9, 50–64. [Google Scholar] [CrossRef]
- Michalakis, K.; Mintziori, G.; Kaprara, A.; Tarlatzis, B.C.; Goulis, D.G. The complex interaction between obesity, metabolic syndrome and reproductive axis: A narrative review. Metabolism 2013, 62, 457–478. [Google Scholar] [CrossRef]
- Mintziori, G.; Nigdelis, M.P.; Mathew, H.; Mousiolis, A.; Goulis, D.G.; Mantzoros, C.S. The effect of excess body fat on female and male reproduction. Metabolism 2020, 107, 154193. [Google Scholar] [CrossRef]
- Cardoso, A.M.; Alves, M.G.; Mathur, P.P.; Oliveira, P.F.; Cavaco, J.E.; Rato, L. Obesogens and male fertility. Obes. Rev. 2017, 18, 109–125. [Google Scholar] [CrossRef]
- Teerds, K.J.; de Rooij, D.G.; Keijer, J. Functional relationship between obesity and male reproduction: From humans to animal models. Hum. Reprod. Update 2011, 17, 667–683. [Google Scholar] [CrossRef]
- Wolfe, A.; Hussain, M.A. The emerging role(s) for kisspeptin in metabolism in mammals. Front. Endocrinol. 2018, 9, 184. [Google Scholar] [CrossRef]
- Hotamisligil, G.S. Inflammation and metabolic disorders. Nature 2006, 444, 860–867. [Google Scholar] [CrossRef]
- Wang, J.; Zhang, S.; Hu, L.; Wang, Y.; Liu, K.; Le, J.; Tan, Y.; Li, T.; Xue, H.; Wei, Y.; et al. Pyrroloquinoline quinone inhibits PCSK9-NLRP3 mediated pyroptosis of Leydig cells in obese mice. Cell Death Dis. 2023, 14, 723. [Google Scholar] [CrossRef]
- Hu, Q.; Lu, Y.; Hu, F.; He, S.; Xu, X.; Niu, Y.; Zhang, H.; Li, X.; Su, Q. Resistant dextrin reduces obesity and attenuates adipose tissue inflammation in high-fat diet-fed mice. Int. J. Med. Sci. 2020, 17, 2611–2621. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Ding, Z. Obesity, a serious etiologic factor for male subfertility in modern society. Reproduction 2017, 154, R123–R131. [Google Scholar] [CrossRef] [PubMed]
- Mu, Y.; Yin, T.-L.; Zhang, Y.; Yang, J.; Wu, Y.-T. Diet-induced obesity impairs spermatogenesis: The critical role of NLRP3 in Sertoli cells. Inflamm. Regen. 2022, 42, 2. [Google Scholar] [CrossRef] [PubMed]
- Schjenken, J.E.; Moldenhauer, L.M.; Sharkey, D.J.; Chan, H.Y.; Chin, P.Y.; Fullston, T.; McPherson, N.O.; Robertson, S.A. High-fat diet alters male seminal plasma composition to impair female immune adaptation for pregnancy in mice. Endocrinology 2021, 162, bqab123. [Google Scholar] [CrossRef]
- Chaudhuri, G.R.; Das, A.; Kesh, S.B.; Bhattacharya, K.; Dutta, S.; Sengupta, P.; Syamal, A.K. Obesity and male infertility: Multifaceted reproductive disruption. Middle East Fertil. Soc. J. 2022, 27, 8. [Google Scholar] [CrossRef]
- Lainez, N.M.; Jonak, C.R.; Nair, M.G.; Ethell, I.M.; Wilson, E.H.; Carson, M.J.; Coss, D. Diet-induced obesity elicits macrophage infiltration and reduction in spine density in the hypothalami of male but not female mice. Front. Immunol. 2018, 9, 1992. [Google Scholar] [CrossRef]
- Leisegang, K.; Sengupta, P.; Agarwal, A.; Henkel, R. Obesity and male infertility: Mechanisms and management. Andrologia 2021, 53, e13617. [Google Scholar] [CrossRef]
- Ruiz-Valderrama, L.; Posadas-Rodríguez, J.; Bonilla-Jaime, H.; Tarragó-Castellanos, M.D.R.; González-Márquez, H.; Arrieta-Cruz, I.; González-Núñez, L.; Salame-Méndez, A.; Rodríguez-Tobón, A.; Morales-Méndez, J.G.; et al. Sperm dysfunction in the testes and epididymides due to overweight and obesity is not caused by oxidative stress. Int. J. Endocrinol. 2022, 2022, 3734572. [Google Scholar] [CrossRef]
- Garolla, A.; Torino, M.; Miola, P.; Caretta, N.; Pizzol, D.; Menegazzo, M.; Bertoldo, A.; Foresta, C. Twenty-four-hour monitoring of scrotal temperature in obese men and men with a varicocele as a mirror of spermatogenic function. Hum. Reprod. 2015, 30, 1006–1013. [Google Scholar] [CrossRef] [PubMed]
- Hoang-Thi, A.P.; Dang-Thi, A.T.; Phan-Van, S.; Nguyen-Ba, T.; Truong-Thi, P.L.; Le-Minh, T.; Nguyen-Vu, Q.-H.; Nguyen-Thanh, T. The Impact of High Ambient Temperature on Human Sperm Parameters: A Meta-Analysis. Iran. J. Public Health 2022, 51, 710–723. [Google Scholar] [CrossRef] [PubMed]
- Francis, S.; Yelumalai, S.; Jones, C.; Coward, K. Aberrant protamine content in sperm and consequential implications for infertility treatment. Hum. Fertil. 2014, 17, 80–89. [Google Scholar] [CrossRef] [PubMed]
- Crujeiras, A.B.; Casanueva, F.F. Obesity and the reproductive system disorders: Epigenetics as a potential bridge. Hum. Reprod. Update 2015, 21, 249–261. [Google Scholar] [CrossRef]
- Fofana, M.; Li, Z.; Li, H.; Li, W.; Wu, L.; Lu, L.; Liu, Q. Decreased ubiquitination and acetylation of histones 3 and 4 are associated with obesity-induced disorders of spermatogenesis in mice. Toxics 2024, 12, 296. [Google Scholar] [CrossRef]
- Portha, B.; Grandjean, V.; Movassat, J.J.N. Mother or father: Who is in the front line? Mechanisms underlying the non-genomic transmission of obesity/diabetes via the maternal or the paternal line. Nutrients 2019, 11, 233. [Google Scholar] [CrossRef]
- Crisóstomo, L.; Videira, R.A.; Jarak, I.; Starčević, K.; Mašek, T.; Rato, L.; Raposo, J.F.; Batterham, R.L.; Oliveira, P.F.; Alves, M.G. Inherited Metabolic Memory of High-Fat Diet Impairs Testicular Fatty Acid Content and Sperm Parameters. Mol. Nutr. Food Res. 2022, 66, 2100680. [Google Scholar] [CrossRef]
- Panera, N.; Mandato, C.; Crudele, A.; Bertrando, S.; Vajro, P.; Alisi, A. Genetics, epigenetics and transgenerational transmission of obesity in children. Front. Endocrinol. 2022, 13, 1006008. [Google Scholar] [CrossRef]
- Gannon, J.R.; Emery, B.R.; Jenkins, T.G.; Carrell, D.T. The sperm epigenome: Implications for the embryo. Genet. Damage Hum. Spermatozoa 2014, 791, 53–66. [Google Scholar]
- McPherson, N.O.; Fullston, T.; Aitken, R.J.; Lane, M. Paternal obesity, interventions, and mechanistic pathways to impaired health in offspring. Ann. Nutr. Metab. 2014, 64, 231–238. [Google Scholar] [CrossRef]
- Fullston, T.; Teague, E.M.C.O.; Palmer, N.O.; DeBlasio, M.J.; Mitchell, M.; Corbett, M.; Print, C.G.; Owens, J.A.; Lane, M. Paternal obesity initiates metabolic disturbances in two generations of mice with incomplete penetrance to the F2 generation and alters the transcriptional profile of testis and sperm microRNA content. FASEB J. 2013, 27, 4226–4243. [Google Scholar] [CrossRef]
- Keyhan, S.; Burke, E.; Schrott, R.; Huang, Z.; Grenier, C.; Price, T.; Raburn, D.; Corcoran, D.L.; Soubry, A.; Hoyo, C.; et al. Male obesity impacts DNA methylation reprogramming in sperm. Clin. Epigenet. 2021, 13, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Donkin, I.; Versteyhe, S.; Ingerslev, L.R.; Qian, K.; Mechta, M.; Nordkap, L.; Mortensen, B.; Appel, E.V.R.; Jørgensen, N.; Kristiansen, V.B.; et al. Obesity and bariatric surgery drive epigenetic variation of spermatozoa in humans. Cell Metab. 2016, 23, 369–378. [Google Scholar] [CrossRef] [PubMed]
- Wu, H.Y.; Cheng, Y.; Jin, L.Y.; Zhou, Y.; Pang, H.Y.; Zhu, H.; Yan, C.-C.; Yan, Y.-S.; Yu, J.-E.; Sheng, J.-Z.; et al. Paternal obesity impairs hepatic gluconeogenesis of offspring by altering Igf2/H19 DNA methylation. Mol. Cell. Endocrinol. 2021, 529, 111264. [Google Scholar] [CrossRef]
- Mahmoud, A.M. An overview of epigenetics in obesity: The role of lifestyle and therapeutic interventions. Int. J. Mol. Sci. 2022, 23, 1341. [Google Scholar] [CrossRef] [PubMed]
- Deshpande, S.S.S.; Nemani, H.; Balasinor, N.H. Diet-induced-and genetic-obesity differentially alters male germline histones. Reproduction 2021, 162, 411–425. [Google Scholar] [CrossRef]
- Deshpande, S.S.; Bera, P.; Khambata, K.; Balasinor, N.H. Paternal obesity induces epigenetic aberrations and gene expression changes in placenta and fetus. Mol. Reprod. Dev. 2023, 90, 109–126. [Google Scholar] [CrossRef]
- Pepin, A.-S.; Lafleur, C.; Lambrot, R.; Dumeaux, V.; Kimmins, S. Sperm histone H3 lysine 4 tri-methylation serves as a metabolic sensor of paternal obesity and is associated with the inheritance of metabolic dysfunction. Mol. Metab. 2022, 59, 101463. [Google Scholar] [CrossRef]
- Terashima, M.; Barbour, S.; Ren, J.; Yu, W.; Han, Y.; Muegge, K. Effect of high fat diet on paternal sperm histone distribution and male offspring liver gene expression. Epigenetics 2015, 10, 861–871. [Google Scholar] [CrossRef]
- Zhang, Y.; Shi, J.; Rassoulzadegan, M.; Tuorto, F.; Chen, Q. Sperm RNA code programmes the metabolic health of offspring. Nat. Rev. Endocrinol. 2019, 15, 489–498. [Google Scholar] [CrossRef]
- Zhou, Y.; Wu, H.; Huang, H. Epigenetic effects of male obesity on sperm and offspring. J. Bio-X Res. 2018, 1, 105–110. [Google Scholar] [CrossRef]
- Ou, X.H.; Zhu, C.C.; Sun, S.C. Effects of obesity and diabetes on the epigenetic modification of mammalian gametes. J. Cell. Physiol. 2019, 234, 7847–7855. [Google Scholar] [CrossRef] [PubMed]
- Fontelles, C.C.; Carney, E.; Clarke, J.; Nguyen, N.M.; Yin, C.; Jin, L.; Cruz, M.I.; Ong, T.P.; Hilakivi-Clarke, L.; de Assis, S. Paternal overweight is associated with increased breast cancer risk in daughters in a mouse model. Sci. Rep. 2016, 6, 28602. [Google Scholar] [CrossRef] [PubMed]
- Crisostomo, L.; Bourgery, M.; Rato, L.; Raposo, J.F.; Batterham, R.L.; Kotaja, N.; Alves, M.G. Testicular “inherited metabolic memory” of ancestral high-fat diet is associated with sperm sncRNA content. Biomedicines 2022, 10, 909. [Google Scholar] [CrossRef] [PubMed]
- Lin, T.; Zhang, S.; Zhou, Y.; Wu, L.; Liu, X.; Huang, H. Small RNA perspective of physical exercise-related improvement of male reproductive dysfunction due to obesity. Front. Endocrinol. 2022, 13, 1038449. [Google Scholar] [CrossRef]
- Fullston, T.; Ohlsson-Teague, E.M.C.; Print, C.G.; Sandeman, L.Y.; Lane, M. Sperm microRNA content is altered in a mouse model of male obesity, but the same suite of microRNAs are not altered in offspring’s sperm. PLoS ONE 2016, 11, e0166076. [Google Scholar] [CrossRef]
- López, P.; Castro, A.; Flórez, M.; Miranda, K.; Aranda, P.; Sánchez-González, C.; Llopis, J.; Arredondo, M. miR-155 and miR-122 expression of spermatozoa in obese subjects. Front. Genet. 2018, 9, 175. [Google Scholar] [CrossRef]
- Wang, C.Y.; Liu, H.Y. Epigenetic mechanism of obesity-induced male infertility. Zhonghua Nan Ke Xue = Natl. J. Androl. 2019, 25, 1036–1039. [Google Scholar]
- Grande, G.; Graziani, A.; De Toni, L.; Garolla, A.; Ferlin, A. Male tract microbiota and male infertility. Cells 2024, 13, 1275. [Google Scholar] [CrossRef]
- Hao, Y.; Feng, Y.; Yan, X.; Chen, L.; Ma, X.; Tang, X.; Zhong, R.; Sun, Z.; Agarwal, M.; Zhang, H.; et al. Gut microbiota-testis axis: FMT mitigates high-fat diet-diminished male fertility via improving systemic and testicular metabolome. Microbiol. Spectr. 2022, 10, e00028-22. [Google Scholar] [CrossRef]
- Ding, N.; Zhang, X.; Zhang, X.D.; Jing, J.; Liu, S.S.; Mu, Y.P.; Peng, L.L.; Yan, Y.J.; Xiao, G.M.; Bi, X.Y.; et al. Impairment of spermatogenesis and sperm motility by the high-fat diet-induced dysbiosis of gut microbes. Gut 2020, 69, 1608–1619. [Google Scholar] [CrossRef]
- Martinot, E.; Thirouard, L.; Holota, H.; Monrose, M.; Garcia, M.; Beaudoin, C.; Volle, D.H. Intestinal microbiota defines the GUT-TESTIS axis. Gut 2022, 71, 844–845. [Google Scholar] [CrossRef] [PubMed]
- Fijak, M.; Meinhardt, A. The testis in immune privilege. Immunol. Rev. 2006, 213, 66–81. [Google Scholar] [CrossRef] [PubMed]
- Javurek, A.B.; Spollen, W.G.; Ali, A.M.M.; Johnson, S.A.; Lubahn, D.B.; Bivens, N.J.; Bromert, K.H.; Ellersieck, M.R.; Givan, S.A.; Rosenfeld, C.S. Discovery of a novel seminal fluid microbiome and influence of estrogen receptor alpha genetic status. Sci. Rep. 2016, 6, 23027. [Google Scholar] [PubMed]
- Wang, S.; Sun, J.; Wang, J.; Ping, Z.; Liu, L. Does obesity based on body mass index affect semen quality?—A meta-analysis and systematic review from the general population rather than the infertile population. Andrologia 2021, 53, e14099. [Google Scholar] [CrossRef]
- Walters, J.L.; De Iuliis, G.N.; Dun, M.D.; Aitken, R.J.; McLaughlin, E.A.; Nixon, B.; Bromfield, E.G. Pharmacological inhibition of arachidonate 15-lipoxygenase protects human spermatozoa against oxidative stress. Biol. Reprod. 2018, 98, 784–794. [Google Scholar] [CrossRef]
- Almabhouh, F.A.; Mokhtar, A.H.M.; Malik, I.A.; Aziz, N.A.A.A.; Durairajanayagam, D.; Singh, H.J. Leptin and reproductive dysfunction in obese men. Andrologia 2020, 52, e13433. [Google Scholar] [CrossRef]
- Muñoz, E.; Fuentes, F.; Felmer, R.; Arias, M.E.; Yeste, M. Effects of reactive oxygen and nitrogen species on male fertility. Antioxid. Redox Signal. 2024, 40, 802–836. [Google Scholar] [CrossRef]
- Kaltsas, A.; Markou, E.; Kyrgiafini, M.-A.; Zikopoulos, A.; Symeonidis, E.N.; Dimitriadis, F.; Zachariou, A.; Sofikitis, N.; Chrisofos, M. Oxidative-stress-mediated epigenetic dysregulation in spermatogenesis: Implications for male infertility and offspring health. Genes 2025, 16, 93. [Google Scholar] [CrossRef]
- De Mello, A.H.; Costa, A.B.; Engel, J.D.G.; Rezin, G.T. Mitochondrial dysfunction in obesity. Life Sci. 2018, 192, 26–32. [Google Scholar] [CrossRef]
- Moustakli, E.; Zikopoulos, A.; Skentou, C.; Bouba, I.; Tsirka, G.; Stavros, S.; Vrachnis, D.; Vrachnis, N.; Potiris, A.; Georgiou, I.; et al. Sperm mitochondrial content and mitochondrial DNA to nuclear DNA ratio are associated with body mass index and progressive motility. Biomedicines 2023, 11, 3014. [Google Scholar] [CrossRef]
- Moustakli, E.; Zikopoulos, A.; Skentou, C.; Dafopoulos, S.; Stavros, S.; Dafopoulos, K.; Drakakis, P.; Georgiou, I.; Zachariou, A. Association of obesity with telomere length in human sperm. J. Clin. Med. 2024, 13, 2150. [Google Scholar] [CrossRef] [PubMed]
- Raee, P.; Shams Mofarahe, Z.; Nazarian, H.; Abdollahifar, M.A.; Ghaffari Novin, M.; Aghamiri, S.; Ghaffari Novin, M. Male obesity is associated with sperm telomere shortening and aberrant mRNA expression of autophagy-related genes. Basic Clin. Androl. 2023, 33, 13. [Google Scholar] [CrossRef] [PubMed]
- Santi, D.; Greco, C.; Barbonetti, A.; Simoni, M.; Maggi, M.; Corona, G. Weight loss as therapeutic option to restore fertility in obese men: A meta-analytic study. World J. Men’s Health 2024, 43, 333–343. [Google Scholar] [CrossRef] [PubMed]
- Sharma, A.; Papanikolaou, N.; Sherif, S.A.; Dimakopolou, A.; Thaventhiran, T.; Go, C.; Entwistle, O.H.; Brown, A.; Luo, R.; Jha, R.; et al. Improvements in sperm motility following low-or high-intensity dietary interventions in men with obesity. J. Clin. Endocrinol. Metab. 2024, 109, 449–460. [Google Scholar] [CrossRef]
- Salonia, A.; Capogrosso, P.; Boeri, L.; Cocci, A.; Corona, G.; Dinkelman-Smit, M.; Falcone, M.; Jensen, C.F.; Gül, M.; Kalkanli, A.; et al. European association of urology guidelines on male sexual and reproductive health: 2025 update on male hypogonadism, erectile dysfunction, premature ejaculation, and peyronie’s disease. Eur. Urol. 2025, 87, 601–616. [Google Scholar] [CrossRef]
- Morrison, A.E.; Papamargaritis, D.; Jayasena, C.N.; Potdar, N.; Yates, T.; Biddle, G.J.; Hamza, M.; Davies, M.J. Can obesity pharmacotherapy be used to manage male infertility? Nat. Rev. Endocrinol. 2025, 21, 518–520. [Google Scholar] [CrossRef]
- Guo, B.; Li, J.; Ma, Y.; Zhao, Y.; Liu, J. Efficacy and safety of letrozole or anastrozole in the treatment of male infertility with low testosterone-estradiol ratio: A meta-analysis and systematic review. Andrology 2022, 10, 894–909. [Google Scholar] [CrossRef]
- Liu, S.; Kuei, M.L.S.; Saffari, S.E.; Zheng, J.; Yeun, T.T.; Leng, J.P.W.; Viardot-Foucault, V.; Nadarajah, S.; Chan, J.K.Y.; Hao, T.H. Do men with normal testosterone–oestradiol ratios benefit from letrozole for the treatment of male infertility? Reprod. Biomed. Online 2019, 38, 39–45. [Google Scholar] [CrossRef]
- Korani, M. Aromatase inhibitors in male: A literature review. Med. Clínica Práctica 2023, 6, 100356. [Google Scholar] [CrossRef]
- Crosnoe, L.E.; Grober, E.; Ohl, D.; Kim, E.D. Exogenous testosterone: A preventable cause of male infertility. Transl. Androl. Urol. 2013, 2, 106. [Google Scholar]
- Corona, G.; Rastrelli, G.; Sparano, C.; Vignozzi, L.; Sforza, A.; Maggi, M. Pharmacological management of testosterone deficiency in men current advances and future directions. Expert Rev. Clin. Pharmacol. 2024, 17, 665–681. [Google Scholar] [CrossRef] [PubMed]
- Zhang, T.; Sun, P.; Geng, Q.; Fan, H.; Gong, Y.; Hu, Y.; Shan, L.; Sun, Y.; Shen, W.; Zhou, Y. Disrupted spermatogenesis in a metabolic syndrome model: The role of vitamin A metabolism in the gut–testis axis. Gut 2022, 71, 78–87. [Google Scholar] [CrossRef]
- Liu, D.; Han, X.; Zou, W.; Yang, Z.; Peng, J.; Li, Y.; Liu, Y.; Jia, M.; Liu, W.; Li, H.; et al. Probiotics Combined with Metformin Improves Sperm Parameters in Obese Male Mice through Modulation of Intestinal Microbiota Equilibrium. Reprod. Sci. 2025, 32, 116–130. [Google Scholar] [CrossRef] [PubMed]
- Ferramosca, A.; Zara, V. Diet and male fertility: The impact of nutrients and antioxidants on sperm energetic metabolism. Int. J. Mol. Sci. 2022, 23, 2542. [Google Scholar] [CrossRef] [PubMed]
- Heriberto, V.M.L.; Patrick, M.S.; Elihu, C.P.; Bravo, G.; Julian, A.C.D. Whole tomato lipidic extract improved sperm quality in obese rats induced by a high-carbohydrate diet. Andrology 2025, 13, 955–967. [Google Scholar] [CrossRef]
- El-Wakf, A.M.; El-Habibi, E.S.M.; Ali, D.A.; Abd El-Ghany, E.; Elmougy, R. Marjoram and sage oils protect against testicular apoptosis, suppressed Ki-67 expression and cell cycle arrest as a therapy for male infertility in the obese rats. J. Food Biochem. 2020, 44, e13080. [Google Scholar] [CrossRef]

| WAT | BAT | |
|---|---|---|
| Cellular structure | Unilocular lipid droplets, relatively sparse mitochondria [27]. | Multilocular lipid droplets, and more mitochondria [32]. |
| Distribution | Thighs, buttocks, lower abdomen, and pubic area, and visceral fat [28]. | Cervical, perirenal, suprarenal, and cardiothoracic areas, particularly surrounding the aorta and mediastinal structures [31]. |
| Main functions | Energy storage [4]. | Heat production (non-shivering thermogenesis) [33]. |
| Relationship with obesity | Dysregulation of WAT expansion and function [35], excessive fat accumulation [36]. | Increase energy expenditure and protect against obesity [37]. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kang, Y.; Li, P.; Yuan, S.; Fu, S.; Zhang, X.; Zhang, J.; Dong, C.; Xiong, R.; Zhao, H.; Huang, D. Progress in Investigating the Impact of Obesity on Male Reproductive Function. Biomedicines 2025, 13, 2054. https://doi.org/10.3390/biomedicines13092054
Kang Y, Li P, Yuan S, Fu S, Zhang X, Zhang J, Dong C, Xiong R, Zhao H, Huang D. Progress in Investigating the Impact of Obesity on Male Reproductive Function. Biomedicines. 2025; 13(9):2054. https://doi.org/10.3390/biomedicines13092054
Chicago/Turabian StyleKang, Yafei, Peiling Li, Suying Yuan, Sen Fu, Xue Zhang, Jiaxing Zhang, Chenle Dong, Renhui Xiong, Hu Zhao, and Donghui Huang. 2025. "Progress in Investigating the Impact of Obesity on Male Reproductive Function" Biomedicines 13, no. 9: 2054. https://doi.org/10.3390/biomedicines13092054
APA StyleKang, Y., Li, P., Yuan, S., Fu, S., Zhang, X., Zhang, J., Dong, C., Xiong, R., Zhao, H., & Huang, D. (2025). Progress in Investigating the Impact of Obesity on Male Reproductive Function. Biomedicines, 13(9), 2054. https://doi.org/10.3390/biomedicines13092054




