Sex-Specific Patterns and Predictors of Reverse Left Ventricular Remodeling and Outcomes in STEMI Patients with LVEF ≤ 50% After Successful Primary Angioplasty
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Echocardiographic Assessment
2.3. Speckle-Tracking Echocardiography and Myocardial Work Indices
2.4. Clinical Follow-Up and Endpoints
2.5. Statistical Analysis
3. Results
3.1. Study Population
3.2. Echocardiographic Findings
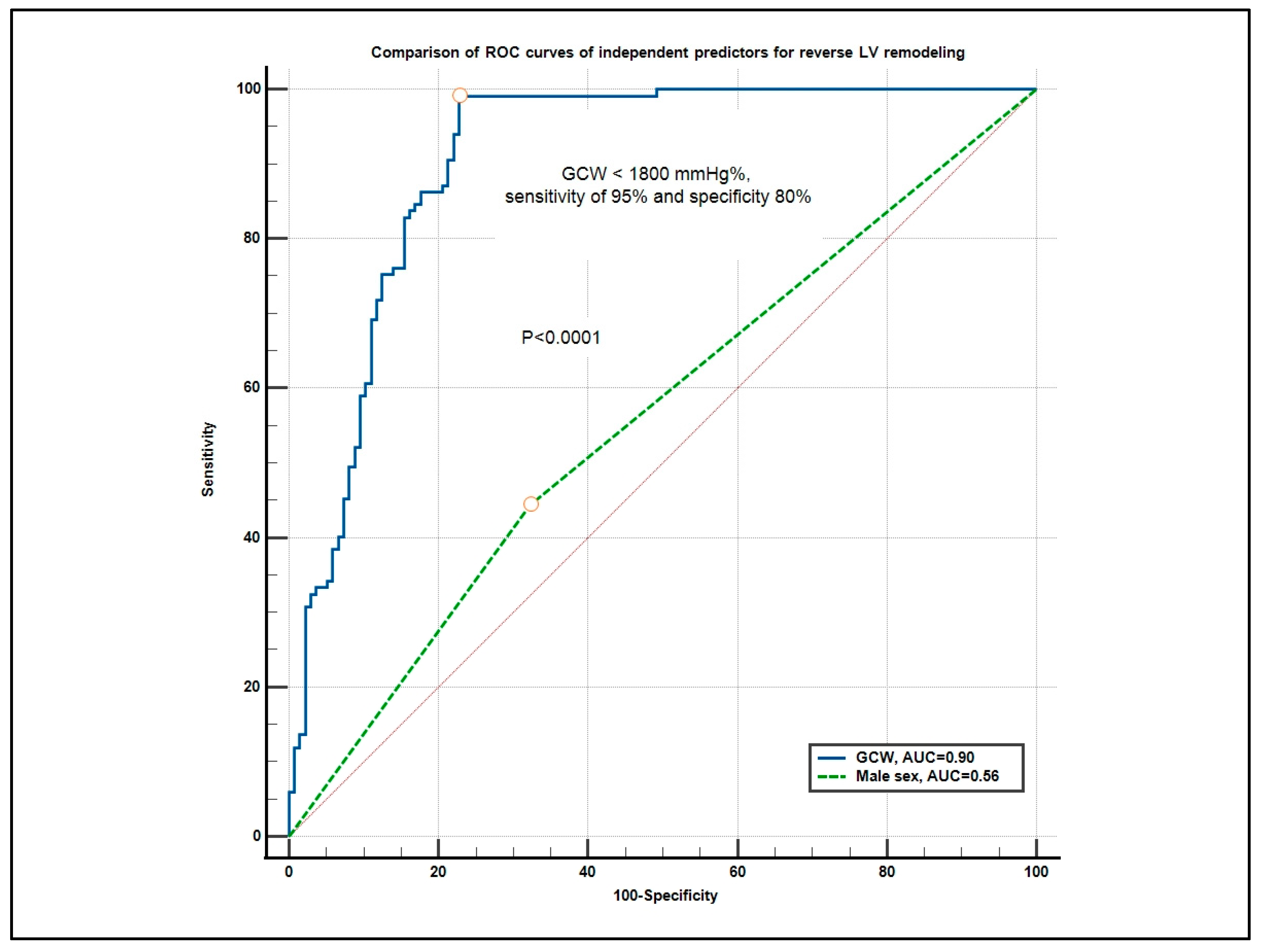
3.3. Predictors of LV Reverse Remodeling
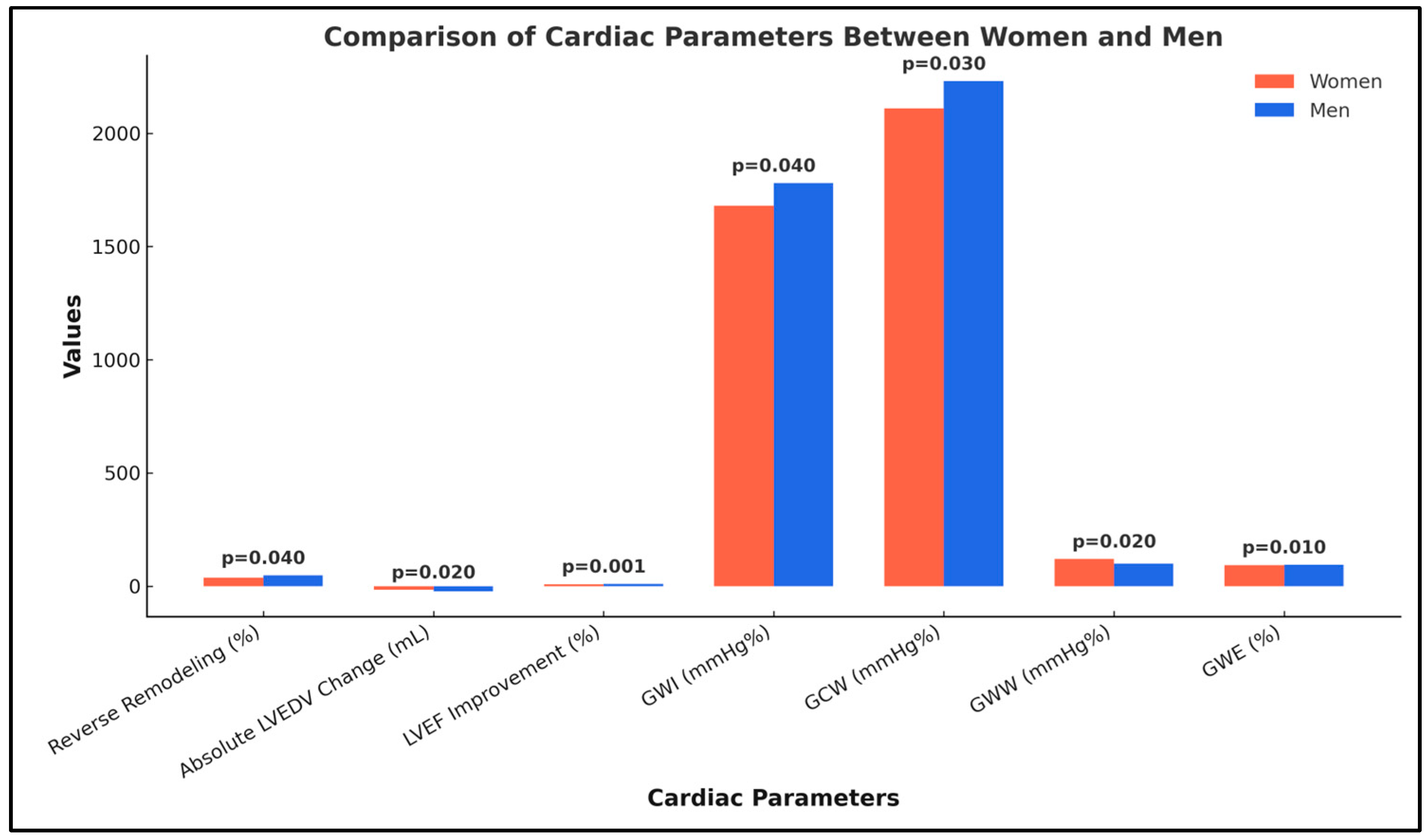
3.4. Sex-Specific Patterns of Reverse LV Remodeling
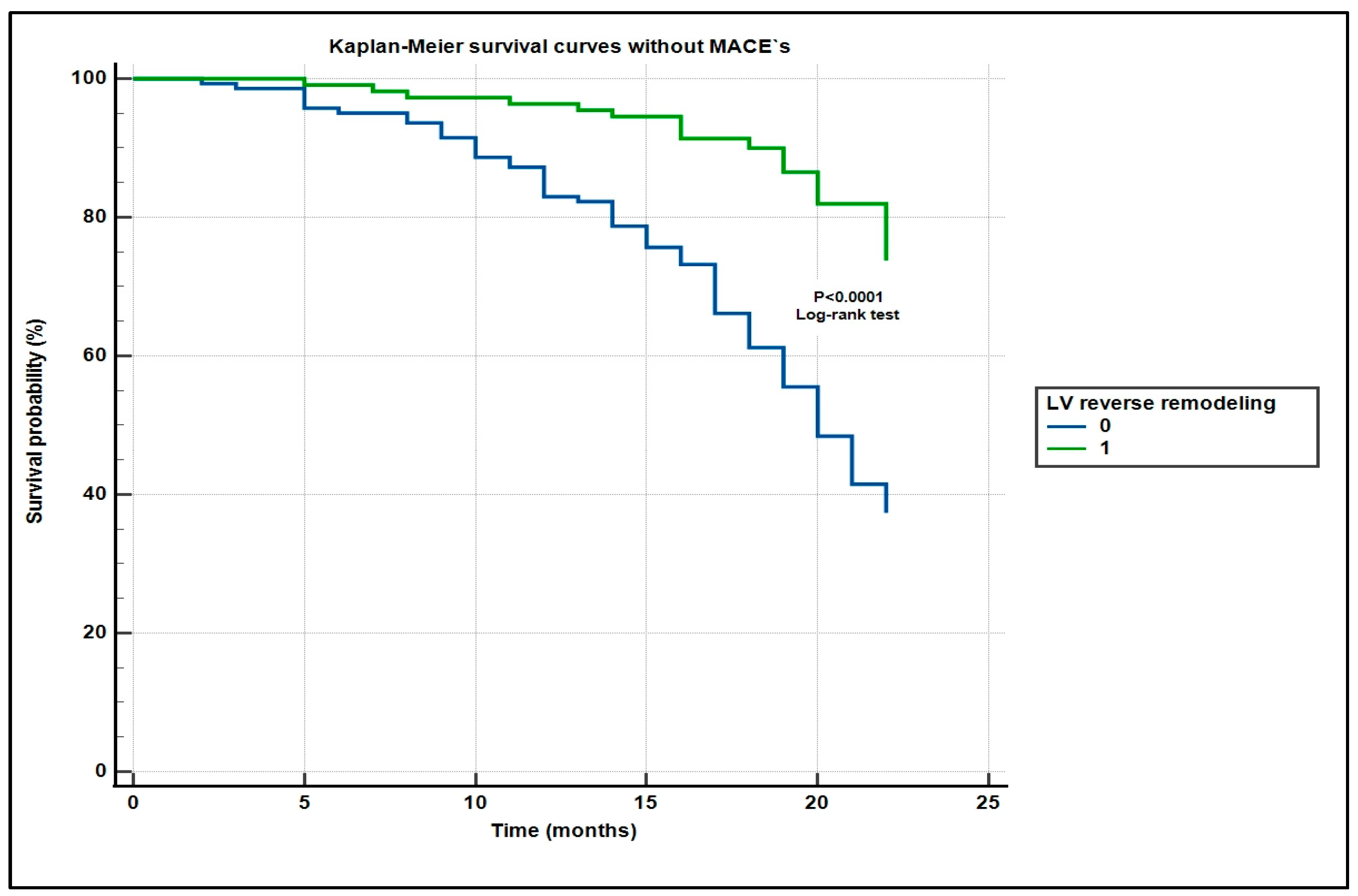
3.5. Clinical Outcomes
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kristensen, S.D.; Aboyans, V. 2017 ESC Guidelines for the management of acute myocardial infarction in patients presenting with ST-segment elevation. Eur. Heart J. 2018, 39, 119–177. [Google Scholar] [CrossRef]
- O’gara, P.T.; Kushner, F.G.; Ascheim, D.D.; Casey, D.E.; Chung, M.K.; De Lemos, J.A.; Ettinger, S.M.; Fang, J.C.; Fesmire, F.M.; Franklin, B.A. 2013 ACCF/AHA guideline for the management of ST-elevation myocardial infarction: A report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines. J. Am. Coll. Cardiol. 2013, 61, e78–e140. [Google Scholar] [CrossRef] [PubMed]
- Pfeffer, M.A.; Braunwald, E.J.C. Ventricular remodeling after myocardial infarction. Experimental observations and clinical implications. Circulation 1990, 81, 1161–1172. [Google Scholar] [CrossRef] [PubMed]
- Bolognese, L.; Cerisano, G. Early predictors of left ventricular remodeling after acute myocardial infarction. Am. Heart J. 1999, 138, S79–S83. [Google Scholar] [CrossRef] [PubMed]
- White, H.D.; Norris, R.M.; Brown, M.A.; Brandt, P.W.; Whitlock, R.M.; Wild, C.J. Left ventricular end-systolic volume as the major determinant of survival after recovery from myocardial infarction. Circulation 1987, 76, 44–51. [Google Scholar] [CrossRef] [PubMed]
- Vaccarino, V.; Parsons, L.; Every, N.R.; Barron, H.V.; Krumholz, H.M. Sex-based differences in early mortality after myocardial infarction. N. Engl. J. Med. 1999, 341, 217–225. [Google Scholar] [CrossRef] [PubMed]
- Maas, A.H.; Appelman, Y.E. Gender differences in coronary heart disease. Neth. Heart J. 2010, 18, 598–603. [Google Scholar] [CrossRef] [PubMed]
- Shah, T.; Palaskas, N.; Ahmed, A. An update on gender disparities in coronary heart disease care. Curr. Atheroscler. Rep. 2016, 18, 28. [Google Scholar] [CrossRef] [PubMed]
- Regitz-Zagrosek, V. Therapeutic implications of the gender-specific aspects of cardiovascular disease. Nat. Rev. Drug Discov. 2006, 5, 425–439. [Google Scholar] [CrossRef] [PubMed]
- Thygesen, K.; Alpert, J.S.; Jaffe, A.S.; Chaitman, B.R.; Bax, J.J.; Morrow, D.A.; White, H.D.; Group, E.S.D. Fourth universal definition of myocardial infarction (2018). Eur. Heart J. 2018, 40, 237–269. [Google Scholar] [CrossRef] [PubMed]
- Beek, A.M.; Kühl, H.P.; Bondarenko, O.; Twisk, J.W.; Hofman, M.B.; Van Dockum, W.G.; Visser, C.A.; Van Rossum, A.C. Delayed contrast-enhanced magnetic resonance imaging for the prediction of regional functional improvement after acute myocardial infarction. J. Am. Coll. Cardiol. 2003, 42, 895–901. [Google Scholar] [CrossRef] [PubMed]
- Wu, K.C.; Zerhouni, E.A.; Judd, R.M.; Lugo-Olivieri, C.H.; Barouch, L.A.; Schulman, S.P.; Blumenthal, R.S.; Lima, J.A.J.C. Prognostic significance of microvascular obstruction by magnetic resonance imaging in patients with acute myocardial infarction. Circulation 1998, 97, 765–772. [Google Scholar] [CrossRef] [PubMed]
- Huttin, O.; Coiro, S.; Selton-Suty, C.; Juilliere, Y.; Donal, E.; Magne, J.; Sadoul, N.; Zannad, F.; Rossignol, P.; Girerd, N.J. Prediction of left ventricular remodeling after a myocardial infarction: Role of myocardial deformation: A systematic review and meta-analysis. PLoS ONE 2016, 11, e0168349. [Google Scholar] [CrossRef] [PubMed]
- Meimoun, P.; Abdani, S.; Stracchi, V.; Elmkies, F.; Boulanger, J.; Botoro, T.; Zemir, H.; Clerc, J.J. Usefulness of noninvasive myocardial work to predict left ventricular recovery and acute complications after acute anterior myocardial infarction treated by percutaneous coronary intervention. J. Am. Soc. Echocardiogr. 2020, 33, 1180–1190. [Google Scholar] [CrossRef] [PubMed]
- Vaccarino, V.; Horwitz, R.I.; Meehan, T.P.; Petrillo, M.K.; Radford, M.J.; Krumholz, H.M. Sex differences in mortality after myocardial infarction: Evidence for a sex-age interaction. Arch. Intern. Med. 1998, 158, 2054–2062. [Google Scholar] [CrossRef] [PubMed]
- Cenko, E.; Yoon, J.; Kedev, S.; Stankovic, G.; Vasiljevic, Z.; Krljanac, G.; Kalpak, O.; Ricci, B.; Miličić, D.; Manfrini, O.J. Sex differences in outcomes after STEMI: Effect modification by treatment strategy and age. Eur. Heart J. 2018, 178, 632–639. [Google Scholar] [CrossRef] [PubMed]
- Abdu, F.A.; Liu, L.; Mohammed, A.-Q.; Yin, G.; Xu, B.; Zhang, W.; Xu, S.; Lv, X.; Fan, R.; Feng, C.J.E.J.o.I.M. Prognostic impact of coronary microvascular dysfunction in patients with myocardial infarction with non-obstructive coronary arteries. Circulation 2021, 92, 79–85. [Google Scholar] [CrossRef] [PubMed]
- Frankel, D.S.; Mountantonakis, S.E.; Zado, E.S.; Anter, E.; Bala, R.; Cooper, J.M.; Deo, R.; Dixit, S.; Epstein, A.E.; Garcia, F.C. Noninvasive programmed ventricular stimulation early after ventricular tachycardia ablation to predict risk of late recurrence. J. Am. Coll. Cardiol. 2012, 59, 1529–1535. [Google Scholar] [CrossRef] [PubMed]
- Chen, P.; Aurich, M.; Greiner, S.; Maliandi, G.; Müller-Hennessen, M.; Giannitsis, E.; Meder, B.; Frey, N.; Pleger, S.; Mereles, D.J. Prognostic relevance of global work index and global constructive work in patients with non-ischemic dilated cardiomyopathy. Int. J. Cardiovasc. Imaging 2024, 40, 1575–1584. [Google Scholar] [CrossRef] [PubMed]
- Ilardi, F.; D’Andrea, A.; D’Ascenzi, F.; Bandera, F.; Benfari, G.; Esposito, R.; Malagoli, A.; Mandoli, G.E.; Santoro, C.; Russo, V.J. Myocardial work by echocardiography: Principles and applications in clinical practice. J. Clin Med. 2021, 10, 4521. [Google Scholar] [CrossRef] [PubMed]
- Frantz, S.; Hundertmark, M.J.; Schulz-Menger, J.; Bengel, F.M.; Bauersachs, J.J. Left ventricular remodelling post-myocardial infarction: Pathophysiology, imaging, and novel therapies. Eur. Heart J. 2022, 43, 2549–2561. [Google Scholar] [CrossRef] [PubMed]
- Stanga, L.C.; Vaduba, D.; Grigoras, M.L.; Nussbaum, L.A.; Gurgus, D.; Strat, L.; Zamfir, A.S.; Poroch, V.; Folescu, R.J.R.C. Nosocomial infections distribution and impact in medical units. Rev. Chim. 2019, 70, 2265–2268. [Google Scholar] [CrossRef]
- Iacob, M.S.; Kundnani, N.R.; Sharma, A.; Meche, V.; Ciobotaru, P.; Bedreag, O.; Sandesc, D.; Dragan, S.R.; Papurica, M.; Stanga, L.C.J.L. Multifactorial Risk Stratification in Patients with Heart Failure, Chronic Kidney Disease, and Atrial Fibrillation: A Comprehensive Analysis. Life 2025, 15, 786. [Google Scholar] [CrossRef] [PubMed]
- van Driel, B.; Nollet, E.J. Tracing triggers of cardiac remodelling and heart failure. Neth. Heart J. 2021, 29, 357–358. [Google Scholar] [CrossRef] [PubMed]
- Daliu, P.; Bogdan, I.; Rosca, O.; Licker, M.; Stanga, L.C.; Hogea, E.; Vaduva, D.B.; Muntean, D.J.B. Fungal Pulmonary Coinfections in COVID-19: Microbiological Assessment, Inflammatory Profiles, and Clinical Outcomes. Biomedicines 2025, 13, 864. [Google Scholar] [CrossRef] [PubMed]
- Vaccarino, V.; Badimon, L.; Corti, R.; De Wit, C.; Dorobantu, M.; Hall, A.; Koller, A.; Marzilli, M.; Pries, A.; Bugiardini, R.J. Ischaemic heart disease in women: Are there sex differences in pathophysiology and risk factors? Position paper from the working group on coronary pathophysiology and microcirculation of the European Society of Cardiology. Cardiovasc. Res. 2011, 90, 9–17. [Google Scholar] [CrossRef] [PubMed]
- De Luca, G.; Suryapranata, H.; Ottervanger, J.P.; Antman, E.M.J.C. Time delay to treatment and mortality in primary angioplasty for acute myocardial infarction: Every minute of delay counts. Circulation 2004, 109, 1223–1225. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Zhao, H.; Wan, F.; Shen, X.-d.; Ding, S.; Pu, J.J. Inhomogeneous Distribution of Regional Myocardial Work Efficiency Predicts Early Left Ventricular Remodeling After Acute Anterior Myocardial Infarction Treated with Primary Percutaneous Intervention. Front. Cardiovasc. Med. 2022, 9, 922567. [Google Scholar] [CrossRef] [PubMed]
- Lustosa, R.P.; Butcher, S.C.; van der Bijl, P.; El Mahdiui, M.; Montero-Cabezas, J.M.; Kostyukevich, M.V.; Rocha De Lorenzo, A.; Knuuti, J.; Ajmone Marsan, N.; Bax, J.J. Global left ventricular myocardial work efficiency and long-term prognosis in patients after ST-segment–elevation myocardial infarction. Circ. Cardiovasc. Imaging 2021, 14, e012072. [Google Scholar] [CrossRef] [PubMed]
- Butcher, S.C.; Lustosa, R.P.; Abou, R.; Marsan, N.A.; Bax, J.J.; Delgado, V.J. Prognostic implications of left ventricular myocardial work index in patients with ST-segment elevation myocardial infarction and reduced left ventricular ejection fraction. Eur. Heart J. Cardiovasc. Imaging 2022, 23, 699–707. [Google Scholar] [CrossRef] [PubMed]
- Timóteo, A.T.; Branco, L.M.; Galrinho, A.; Rio, P.; Papoila, A.L.; Alves, M.; Ferreira, R.C. Global left ventricular myocardial work index and medium-term adverse cardiovascular events after ST-elevation myocardial infarction. Int. J. Cardiol. 2024, 399, 131781. [Google Scholar] [CrossRef] [PubMed]
- Dekleva, M.; Djuric, T.; Djordjevic, A.; Soldatovic, I.; Stankovic, A.; Suzic Lazic, J.; Zivkovic, M.J.B. Sex-Related Differences in Heart Failure Development in Patients After First Myocardial Infarction: The Role of Galectin-3. Biomedicines 2024, 12, 2661. [Google Scholar] [CrossRef] [PubMed]
- Vyas, R.; Changal, K.H.; Bhuta, S.; Pasadyn, V.; Katterle, K.; Niedoba, M.J.; Vora, K.; Dharmakumar, R.; Gupta, R.J. Impact of intramyocardial hemorrhage on clinical outcomes in ST-elevation myocardial infarction: A systematic review and meta-analysis. J. Soc. Cardiovasc. Angiogr. Interv. 2022, 1, 100444. [Google Scholar] [CrossRef] [PubMed]

| Women n = 91 | Men n = 162 | All Patients n = 253 | p-Value | |
|---|---|---|---|---|
| Mean age | 69.9 ± 10.2 | 64.4 ± 12.3 | 66.4 ± 13.3 | <0.0001 |
| Diabetes mellitus | 28 (30.7) | 47 (29.6) | 75 (29) | 0.86 |
| Systemic hypertension | 81 (89.0) | 121 (75.0) | 195 (77) | <0.01 |
| Hypercholesterolemia | 70 (76.9) | 113 (70.3) | 183 (72.3) | 0.26 |
| Smoking history | 25 (27.4) | 90 (55.9) | 115 (45.4) | <0.0001 |
| Obesity | 23 (25.2) | 39 (24.3) | 62 (24.5) | 0.87 |
| SBP (mmHg) | 126.0 ± 30.9 | 134.2 ± 23.5 | 130.4 ± 26.7 | 0.03 |
| DBP (mmHg) | 72.9 ± 18.9 | 78.3 ± 13.8 | 75.9 ± 16.1 | 0.01 |
| Heart rate (beats/min) | 80.9 ± 22.7 | 80.0 ± 16.7 | 80.2 ± 19.1 | 0.74 |
| Chronic renal failure | 19 (20.8) | 24 (15.7) | 43 (16.9) | 0.31 |
| Killip class I (%) II (%) III (%) IV (%) | 16 (17.7) 33 (36.3) 17 (18.6) 25 (27.4) | 41 (25.6) 78 (48.0) 27 (16.6) 16 (9.8) | 57 (22.5) 111 (43.8) 44 (17.3) 41 (16.2) | 0.14 0.07 0.10 <0.01 |
| Peak CPK-MB (IU/L), median (25th, 75th percentile) | 207 (80–453) | 176 (66–337) | 183 (77–382) | 0.09 |
| Time from symptom onset to reperfusion (min) | 211 (171–261) | 187 (126–237) | 196 (142–246) | 0.15 |
| NT-proBNP (ng/L) median (25th, 75th percentile) | 300.0 (30–396) | 200 (61.5–450.0) | 200 (70–890) | <0.01 |
| eGFR (mL/min/1.73 m2) | 41.1 ± 20.2 | 55.1 ± 23.9 | 50.7 ± 23.4 | <0.0001 |
| Culprit vessel LAD (%) LCX (%) RCA (%) | 50 (55) 30 (33) 10 (11) | 92 (57) 52 (32) 18 (13) | 142 (56.1) 82 (32.4) 28 (11) | 0.76 0.87 0.64 |
| Coronary artery disease 1-vessel (%) 2-vessel (%) 3-vessel (%) | 41 (45.0) 17 (18.6) 23 (25.2) | 75 (46.0) 34 (21.7) 29 (18.4) | 116 (45.8) 51 (20.1) 52 (20.5) | 0.88 0.56 0.21 |
| Medication at discharge ACEI or ARB Beta-blocker Calcim antagonists Statins | 34 (70.9) 69 (76) 23 (25.0) 75 (82.0) | 131 (81) 128 (79) 37 (23) 136 (84) | 165 (65.2) 197 (77.8) 60 (23.7) 211 (83.3) | 0.06 0.58 0.72 0.68 |
| Variable | Reverse Remodeling (n = 110) | No Reverse Remodeling (n = 143) | p-Value |
|---|---|---|---|
| Age, years | 63.2 ± 11.8 | 68.9 ± 11.4 | 0.001 |
| Female sex, n (%) | 32 (29) | 59 (41) | 0.048 |
| Diabetes mellitus, n (%) | 29 (26) | 46 (32) | 0.29 |
| Hypertension, n (%) | 80 (73) | 115 (80) | 0.19 |
| Killip class ≥ III, n (%) | 25 (23) | 60 (42) | 0.002 |
| NT-proBNP, ng/L (median, IQR) | 180 (70–380) | 260 (90–460) | 0.004 |
| eGFR, mL·min−1·1.73 m−2 | 54.3 ± 22.1 | 47.8 ± 24.6 | 0.03 |
| Symptom-to-reperfusion, min (median, IQR) | 188 (140–238) | 204 (150–252) | 0.08 |
| Parameter | Reverse LV Remodeling n = 110 | No Reverse LV Remodeling n = 143 | All STEMI Patients n = 253 | p-Value |
|---|---|---|---|---|
| Baseline | ||||
| LVEF (%) | 42.5 ± 8.3 | 41.3 ± 5.5 | 41.27 ± 6.86 | 0.19 |
| LVEDV (mL) | 123 ± 27 | 131 ± 29 | 127.5 ± 28.1 | 0.67 |
| LVESV (mL) | 44 ± 8.2 | 42 ± 9.5 | 42.88 ± 8.96 | 0.13 |
| Stroke volume index (mL/m2) | 42 ± 10.5 | 40.3 ± 11.2 | 41.0 ± 10.9 | 0.21 |
| E/A ratio | 1.10 ± 0.42 | 1.06 ± 0.48 | 1.08 ± 0.455 | 0.48 |
| WMSI | 2.21 ± 0.23 | 2.27 ± 0.24 | 2.24 ± 0.236 | 0.04 |
| GLS (%) | 18.6 ± 3.6 | 16.8 ± 3.4 | 17.6 ± 3.5 | <0.0001 |
| GWI (mmHg%) | 1885 ± 467 | 1527 ± 141 | 1682.43 ± 325.51 | <0.0001 |
| GWE (%) | 89.8 ± 6.4 | 77.5 ± 4.2 | 82.9 ± 5.3 | <0.0001 |
| GWW (mmHg%) | 198.5 ± 57 | 226 ± 20 | 214.13 ± 40.45 | 0.001 |
| GCW (mmHg%) | 2114 ± 196 | 1625 ± 147 | 1837.2 ± 170.01 | <0.0001 |
| After 6 months | ||||
| LVEF (%) | 61.2 ± 7.3 | 46.5 ± 3.2 | 52.91 ± 5.38 | <0.0001 |
| LVEDV (mL) | 104.3 ± 22.9 | 115.3 ± 25.5 | 110.5 ± 24.4 | <0.01 |
| LVESV (mL) | 47.1 ± 13.8 | 51.9 ± 12.2 | 49.8 ± 2.9 | <0.01 |
| Stroke volume index (mL/m2) | 46.5 ± 5.4 | 43.3 ±7.3 | 44.7 ± 6.5 | <0.001 |
| E/A ratio | 1.06 ± 0.30 | 1.04 ± 0.25 | 1.05 ± 0.27 | 0.56 |
| WMSI | 1.96 ± 0.3 | 2.17 ± 0.4 | 2.08 ± 0.36 | <0.0001 |
| GLS (%) | −20.0 ± 2.6 | −19.1 ± 3.2 | −19.4 ± 2.6 | 0.024 |
| GWI (mmHg%) | 1936 ± 153 | 1865 ± 233 | 1896.1 ± 202.2 | <0.01 |
| GWE (%) | 91.4 ± 4.4 | 83.7 ± 5.6 | 87.0 ± 5.1 | <0.0001 |
| GWW (mmHg%) | 179.7 ± 53 | 208.0 ± 62 | 195.7 ± 58.3 | 0.0001 |
| GCW (mmHg%) | 2203 ± 236 | 2082 ± 304 | 2135.5 ± 276.4 | 0.0015 |
| Univariate Logistic Regression | Odds Ratio | 95% CI | p-Value |
|---|---|---|---|
| Male sex | 0.59 | 0.35–0.99 | 0.04 |
| Baseline WSMI | 1.25 | 1.18–1.32 | <0.0001 |
| Baseline GLS | 2.02 | 1.70–2.40 | <0.0001 |
| Baseline GWI (mmHg%) | 1.007 | 1.005–1.009 | <0.0001 |
| Baseline GCW (mmHg%) | 1.01 | 1.011–1.018 | <0.0001 |
| Baseline GWE (%) | 0.72 | 0.66–0.79 | 0.01 |
| Baseline GWW (mmHg%) | 1.06 | 1.42–1.78 | <0.0001 |
| Multivariate logistic regression | Odds Ratio | 95% CI | p-value |
| Baseline GWE (mmHg%) | 1.64 | 1.45–1.85 | <0.0001 |
| Female sex | 0.30 | 0.14–0.65 | <0.0001 |
| Variable | Adjusted OR | 95% CI | p-Value |
|---|---|---|---|
| Global work efficiency, per 1% | 1.57 | 1.38–1.79 | <0.001 |
| Female sex (reference = male) | 0.34 | 0.18–0.63 | <0.001 |
| Age, per year | 0.95 | 0.92–0.99 | 0.01 |
| Killip class III-IV | 0.54 | 0.30–0.97 | 0.04 |
| eGFR, per 10 mL·min−1·1.73 m−2 | 1.11 | 1.01–1.23 | 0.03 |
| Residual SYNTAX > 8 | 0.88 | 0.46–1.70 | 0.7 |
| Full guideline medical therapy | 1.12 | 0.57–2.19 | 0.75 |
| Parameter | Women (n = 91) | Men (n = 162) | All Patients (n = 253) | p-Value |
|---|---|---|---|---|
| Reverse remodeling (≥15% LVEDV reduction), n (%) | 34 (37%) | 76 (47%) | 110 (43%) | 0.04 |
| Absolute LVEDV change (mL), median (IQR) | −16.0 (−25.0 to −8.0) | −22.5 (−35.0 to −12.0) | −20.2 (−31.4 to −10.6) | 0.02 |
| LVEF improvement (%), mean ± SD | +7.2 ± 5.4 | +9.1 ± 5.7 | +8.4 ± 5.6 | 0.001 |
| Global work index (GWI, mmHg%) | 1680 ± 410 | 1780 ± 450 | 1744.7 ± 436.2 | 0.04 |
| Global constructive work (GCW, mmHg%) | 2110 ± 390 | 2230 ± 420 | 2187.1 ± 409.4 | 0.03 |
| Global wasted work (GWW, mmHg%) | 120 ± 50 | 100 ± 45 | 107.2 ± 46.9 | 0.02 |
| Parameter | Difference (Men–Women) | 95% CI | p-Value |
|---|---|---|---|
| Reverse remodeling (%) | +10 | −3.0% to +22.1% | 0.04 |
| Absolute LVEDV change (mL) | −6.5 | −17.6 to −16.3 | 0.02 |
| LVEF improvement (%) | +1.9 | +0.5 to +3.3 | 0.001 |
| Global work index (GWI, mmHg%) | +100 | +0.3 to +199.7 | 0.04 |
| Global constructive work (GCW, mmHg%) | +120 | +17.1 to +222.9 | 0.03 |
| Global wasted work (GWW, mmHg%) | −20 | −42.8 to +2.8 | 0.02 |
| Global work efficiency (GWE, %) | +1.5 | +0.65 to +2.35 | 0.01 |
| Group | MACEs, n (%) | p-Value |
|---|---|---|
| All patients (n = 253) | 45 (18%) | — |
| Reverse remodeling | 11 (10) | |
| No reverse remodeling | 34 (24) | <0.01 |
| Women (n = 91) | ||
| Reverse remodeling | 3 (9) | |
| No reverse remodeling | 17 (29) | 0.02 |
| Men (n = 162) | ||
| Reverse remodeling | 6 (8) | |
| No reverse remodeling | 19 (22) | 0.016 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Buz, B.-F.; Arnautu, S.-F.; Tomescu, M.-C.; Andor, M.; Crisan, S.; Gaita, D.; Vacarescu, C.; Luca, C.-T.; Mornos, C.; Cozma, D.; et al. Sex-Specific Patterns and Predictors of Reverse Left Ventricular Remodeling and Outcomes in STEMI Patients with LVEF ≤ 50% After Successful Primary Angioplasty. Biomedicines 2025, 13, 1782. https://doi.org/10.3390/biomedicines13071782
Buz B-F, Arnautu S-F, Tomescu M-C, Andor M, Crisan S, Gaita D, Vacarescu C, Luca C-T, Mornos C, Cozma D, et al. Sex-Specific Patterns and Predictors of Reverse Left Ventricular Remodeling and Outcomes in STEMI Patients with LVEF ≤ 50% After Successful Primary Angioplasty. Biomedicines. 2025; 13(7):1782. https://doi.org/10.3390/biomedicines13071782
Chicago/Turabian StyleBuz, Bogdan-Flaviu, Sergiu-Florin Arnautu, Mirela-Cleopatra Tomescu, Minodora Andor, Simina Crisan, Dan Gaita, Cristina Vacarescu, Constantin-Tudor Luca, Cristian Mornos, Dragos Cozma, and et al. 2025. "Sex-Specific Patterns and Predictors of Reverse Left Ventricular Remodeling and Outcomes in STEMI Patients with LVEF ≤ 50% After Successful Primary Angioplasty" Biomedicines 13, no. 7: 1782. https://doi.org/10.3390/biomedicines13071782
APA StyleBuz, B.-F., Arnautu, S.-F., Tomescu, M.-C., Andor, M., Crisan, S., Gaita, D., Vacarescu, C., Luca, C.-T., Mornos, C., Cozma, D., & Arnăutu, D.-A. (2025). Sex-Specific Patterns and Predictors of Reverse Left Ventricular Remodeling and Outcomes in STEMI Patients with LVEF ≤ 50% After Successful Primary Angioplasty. Biomedicines, 13(7), 1782. https://doi.org/10.3390/biomedicines13071782








