A Nomogram Built on Clinical Factors and CT Attenuation Scores for Predicting Treatment Response of Acute Myeloid Leukemia Patients
Abstract
1. Introduction
2. Material and Methods
2.1. Ethics Statement
2.2. Patients
2.3. Clinical Data
2.4. CT Scanning
2.5. Image Evaluation
2.6. Statistical Analysis
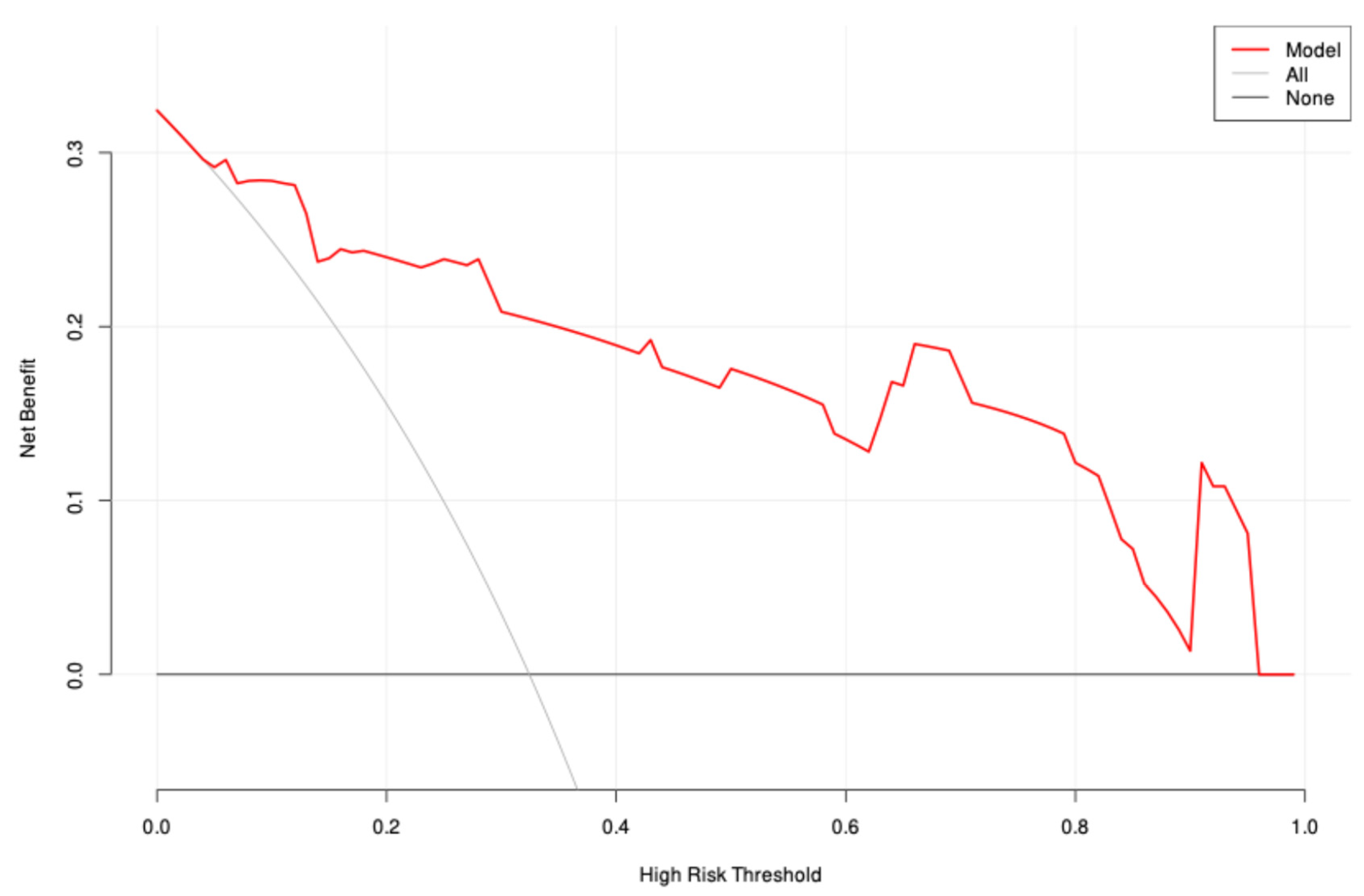
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| AML | Acute myeloid leukemia |
| AUC | Area under the curve |
| BMB | bone marrow blast |
| CR | Complete remission |
| DCA | Decision curve analysis |
| HU | Hounsfield units |
| Hb | Hemoglobin |
| NCR | Non-complete remission |
| NE | Neutrophil |
| Plt | Platelets |
| ROC | Receiver operating characteristic |
| WBC | White blood cell |
References
- Döhner, H.; Weisdorf, D.J.; Bloomfield, C.D. Acute Myeloid Leukemia. N. Engl. J. Med. 2015, 373, 1136–1152. [Google Scholar] [CrossRef] [PubMed]
- De Kouchkovsky, I.; Abdul-Hay, M. Acute myeloid leukemia: A comprehensive review and 2016 update. Blood Cancer J. 2016, 6, e441. [Google Scholar] [CrossRef] [PubMed]
- Short, N.J.; Konopleva, M.; Kadia, T.M.; Borthakur, G.; Ravandi, F.; DiNardo, C.D.; Daver, N. Advances in the Treatment of Acute Myeloid Leukemia: New Drugs and New Challenges. Cancer Discov. 2020, 10, 506–525. [Google Scholar] [CrossRef] [PubMed]
- Liu, H. Emerging agents and regimens for AML. J. Hematol. Oncol. 2021, 14, 49. [Google Scholar] [CrossRef]
- Greiner, J.; Götz, M.; Wais, V. Increasing Role of Targeted Immunotherapies in the Treatment of AML. Int. J. Mol. Sci. 2022, 23, 3304. [Google Scholar] [CrossRef]
- Abaza, Y.; McMahon, C.; Garcia, J.S. Advancements and Challenges in the Treatment of AML. Am. Soc. Clin. Oncol. Educ. Book 2024, 44, e438662. [Google Scholar] [CrossRef]
- Pullarkat, V.; Aldoss, I. Prognostic and therapeutic implications of early treatment response assessment in acute myeloid leukemia. Crit. Rev. Oncol. Hematol. 2015, 95, 38–45. [Google Scholar] [CrossRef]
- Vanhelleputte, P.; Nijs, K.; Delforge, M.; Evers, G.; Vanderschueren, S. Pain during bone marrow aspiration: Prevalence and prevention. J. Pain. Symptom Manag. 2003, 26, 860–866. [Google Scholar] [CrossRef]
- Agool, A.; Slart, R.H.J.A.; Kluin, P.M.; de Wolf, J.T.M.; Dierckx, R.A.J.O.; Vellenga, E. F-18 FLT PET: A noninvasive diagnostic tool for visualization of the bone marrow compartment in patients with aplastic anemia: A pilot study. Clin. Nucl. Med. 2011, 36, 286–289. [Google Scholar] [CrossRef]
- Arrigan, M.; Smyth, L.; Harmon, M.; Flynn, C.; Sheehy, N. Imaging findings in recurrent extramedullary leukaemias. Cancer Imaging 2013, 13, 26–35. [Google Scholar] [CrossRef]
- Scialpi, M.; Reginelli, A.; D’Andrea, A.; Gravante, S.; Falcone, G.; Baccari, P.; Manganaro, L.; Palumbo, B.; Cappabianca, S. Pancreatic tumors imaging: An update. Int. J. Surg. 2016, 28 (Suppl. S1), S142–S155. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Li, Y.; Zhao, Y.; Qiao, J. CT and MRI of superficial solid tumors. Quant. Imaging Med. Surg. 2018, 8, 232–251. [Google Scholar] [CrossRef]
- Murthy, V.; Sonni, I.; Jariwala, N.; Juarez, R.; Reiter, R.E.; Raman, S.S.; Hope, T.A. The Role of PSMA PET/CT and PET/MRI in the Initial Staging of Prostate Cancer. Eur. Urol. Focus 2021, 7, 258–266. [Google Scholar] [CrossRef] [PubMed]
- Han, E.J.; Lee, B.-H.; Kim, J.-A.; Park, Y.H.; Choi, W.H. Early assessment of response to induction therapy in acute myeloid leukemia using 18F-FLT PET/CT. EJNMMI Res. 2017, 7, 75. [Google Scholar] [CrossRef] [PubMed]
- Tian, X.; Niu, J.; Li, W.; Zhou, X.J.; Wu, W.; Li, X.; Wang, J.; Wang, H. Fluid-attenuated inversion recovery diffusion-weighted imaging (DWI) for evaluating chemotherapy response in patients with acute leukemia: Comparison with conventional DWI. Indian. J. Cancer 2022, 59, 230–235. [Google Scholar] [CrossRef]
- Bian, W.; Zhang, J.; Huang, Q.; Niu, W.; Li, J.; Song, X.; Cui, S.; Zheng, Q.; Niu, J.; Zhou, X.J. Quantitative tumor burden imaging parameters of the spleen at MRI for predicting treatment response in patients with acute leukemia. Heliyon 2023, 9, e20348. [Google Scholar] [CrossRef]
- Kumasaka, S.; Kubota, J.; Kumasaka, Y.; Naito, C.; Tsushima, Y. A novel use of CT attenuation value: Increased bone marrow density in patients with acute myeloid leukemia. Acta Radiol. 2023, 64, 1573–1578. [Google Scholar] [CrossRef]
- Cheson, B.D.; Bennett, J.M.; Kopecky, K.J.; Büchner, T.; Willman, C.L.; Estey, E.H.; Schiffer, C.A.; Doehner, H.; Tallman, M.S.; Lister, T.A.; et al. Revised recommendations of the International Working Group for Diagnosis, Standardization of Response Criteria, Treatment Outcomes, and Reporting Standards for Therapeutic Trials in Acute Myeloid Leukemia. J. Clin. Oncol. 2003, 21, 4642–4649. [Google Scholar] [CrossRef]
- Robertson, F.; Leander, P.; Ekberg, O. Radiology of the spleen. Eur. Radiol. 2001, 11, 80–95. [Google Scholar] [CrossRef]
- Vanderhoek, M.; Juckett, M.B.; Perlman, S.B.; Nickles, R.J.; Jeraj, R. Early assessment of treatment response in patients with AML using [(18)F]FLT PET imaging. Leuk. Res. 2011, 35, 310–316. [Google Scholar] [CrossRef]
- Li, J.; Li, W.; Niu, J.; Song, X.; Wu, W.; Gong, T.; Zheng, R.; Ting-Fang Shih, T.; Li, W.; Zhou, X.J. Intravoxel Incoherent Motion Diffusion-weighted MRI of Infiltrated Marrow for Predicting Overall Survival in Newly Diagnosed Acute Myeloid Leukemia. Radiology 2020, 295, 155–161. [Google Scholar] [CrossRef] [PubMed]
- McQuilten, Z.K.; Busija, L.; Seymour, J.F.; Stanworth, S.; Wood, E.M.; Kenealy, M.; Weinkove, R.; Australasian Leukaemia and Lymphoma Group (ALLG). Hemoglobin is a key determinant of quality of life before and during azacitidine-based therapy for myelodysplasia and low blast count acute myeloid leukemia. Leuk. Lymphoma 2022, 63, 676–683. [Google Scholar] [CrossRef] [PubMed]
- Didi, I.; Alliot, J.M.; Dumas, P.Y.; Vergez, F.; Tavitian, S.; Largeaud, L.; Bidet, A.; Rieu, J.B.; Luquet, I.; Lechevalier, N.; et al. Artificial intelligence-based prediction models for acute myeloid leukemia using real-life data: A DATAML registry study. Leuk. Res. 2024, 136, 107437. [Google Scholar] [CrossRef] [PubMed]





| Variables | Total (n = 74) | CR (n = 24) | NCR (n = 50) | p |
|---|---|---|---|---|
| Age, M (Q1, Q3) | 70.50 (54.25, 80.00) | 68.50 (58.75, 78.50) | 72.00 (54.00, 81.00) | 0.959 |
| Gender, n (%) | 0.135 | |||
| Male | 28 (37.84) | 12 (50.00) | 16 (32.00) | |
| Female | 46 (62.16) | 12 (50.00) | 34 (68.00) | |
| WBC (×109/L), M (Q1, Q3) | 10.36 (2.51, 44.54) | 32.74 (4.25, 69.65) | 7.35 (2.50, 32.75) | 0.120 |
| Hb (g/L), Mean ± SD | 87.77 ± 24.74 | 99.00 ± 24.94 | 82.38 ± 23.00 | 0.006 * |
| Plt (×109/L), M (Q1, Q3) | 44.50 (25.25, 87.50) | 74.00 (41.75, 118.25) | 41.50 (23.50, 66.25) | 0.010 * |
| NE (×109/L), M (Q1, Q3) | 20.00 (8.00, 40.00) | 20.50 (11.00, 37.00) | 19.00 (7.00, 40.75) | 0.540 |
| BMB (%), M (Q1, Q3) | 46.00 (28.75, 80.00) | 65.00 (30.75, 83.50) | 40.25 (28.38, 74.88) | 0.241 |
| Spleen Length, M (Q1, Q3) | 97.07 (84.86, 115.19) | 102.49 (83.27, 122.87) | 96.96 (86.19, 111.58) | 0.665 |
| Spleen Width, M (Q1, Q3) | 36.92 (30.91, 43.45) | 37.62 (32.40, 43.72) | 36.56 (30.83, 43.20) | 0.673 |
| CT (HU), M (Q1, Q3) | 131.13 (105.57, 173.06) | 177.62 (158.42, 233.25) | 113.86 (92.10, 140.50) | <0.001 * |
| Variables | Univariate * | Multivariable * | |||||
|---|---|---|---|---|---|---|---|
| β | OR (95%CI) | p | β | OR (95%CI) | p | ||
| Age | 0.00 | 1.00 (0.97~1.03) | 0.866 | ||||
| Gender | −0.75 | 0.47 (0.17~1.27) | 0.138 | ||||
| WBC(×109/L) | 0.00 | 1.00 (1.00~1.01) | 0.274 | ||||
| Hb(g/L) | 0.03 | 1.03 (1.01~1.05) | 0.009 * | 0.03 | 1.03 (1.00~1.06) | 0.094 | |
| Plt(×109/L) | 0.01 | 1.01 (1.01~1.02) | 0.014 * | 0.02 | 1.02 (1.01~1.03) | 0.033 * | |
| NE(×109/L) | 0.00 | 1.00 (0.98~1.03) | 0.838 | ||||
| Bone marrow blasts(%) | 0.01 | 1.01 (0.99~1.03) | 0.191 | ||||
| Spleen Length | 0.01 | 1.01 (0.99~1.03) | 0.410 | ||||
| Spleen Width | 0.01 | 1.01 (0.97~1.05) | 0.732 | ||||
| CT(HU) | 0.03 | 1.03 (1.02~1.05) | <0.001 * | 0 | 0.03 | 1.04 (1.02~1.05) | <0.001 * |
| Model | AUC (95%CI) | Accuracy (95%CI) | Sensitivity (95%CI) | Specificity (95%CI) | PPV (95%CI) | NPV (95%CI) | Cut-Off |
|---|---|---|---|---|---|---|---|
| Nomogram | 0.912 (0.842–0.983) | 0.865 (0.765–0.933) | 0.880 (0.790–0.970) | 0.833 (0.684–0.982) | 0.917 (0.838–0.995) | 0.769 (0.607–0.931) | 0.337 |
| Clinical | 0.762 (0.639–0.885) | 0.743 (0.628–0.838) | 0.780 (0.665–0.895) | 0.667 (0.478–0.855) | 0.830 (0.722–0.937) | 0.593 (0.407–0.778) | 0.337 |
| CT | 0.882 (0.804–0.960) | 0.851 (0.750–0.923) | 0.880 (0.790–0.970) | 0.792 (0.629–0.954) | 0.898 (0.813–0.983) | 0.760 (0.593–0.927) | 155.9 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, L.; Lu, W.; Xiong, L.; Qi, H.; Gale, R.P.; Yin, B. A Nomogram Built on Clinical Factors and CT Attenuation Scores for Predicting Treatment Response of Acute Myeloid Leukemia Patients. Biomedicines 2025, 13, 198. https://doi.org/10.3390/biomedicines13010198
Liu L, Lu W, Xiong L, Qi H, Gale RP, Yin B. A Nomogram Built on Clinical Factors and CT Attenuation Scores for Predicting Treatment Response of Acute Myeloid Leukemia Patients. Biomedicines. 2025; 13(1):198. https://doi.org/10.3390/biomedicines13010198
Chicago/Turabian StyleLiu, Linna, Wenzheng Lu, Li Xiong, Han Qi, Robert Peter Gale, and Bin Yin. 2025. "A Nomogram Built on Clinical Factors and CT Attenuation Scores for Predicting Treatment Response of Acute Myeloid Leukemia Patients" Biomedicines 13, no. 1: 198. https://doi.org/10.3390/biomedicines13010198
APA StyleLiu, L., Lu, W., Xiong, L., Qi, H., Gale, R. P., & Yin, B. (2025). A Nomogram Built on Clinical Factors and CT Attenuation Scores for Predicting Treatment Response of Acute Myeloid Leukemia Patients. Biomedicines, 13(1), 198. https://doi.org/10.3390/biomedicines13010198






