Exhaled Nitric Oxide Reflects the Immune Reactions of the Airways in Early Rheumatoid Arthritis
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Population
2.2. NO Analysis
2.3. Spirometry
2.4. Blood Analyses
2.5. Analyses of ACPA and RF
2.6. Analyses of Free SC, SC ACPAs, Anti-Rgp IgG and Total IgA, IgA1 and IgA2
2.7. Statistical Analysis
3. Results
3.1. Baseline Characteristics of the Study Population
3.2. NO in Relation to Clinical and Inflammation Markers
3.3. NO in Relation to Antibodies
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ACPA | anti-citrullinated protein/peptide antibody |
| AU | arbitrary unit |
| Anti-CCP2 | anti-cyclic citrullinated peptide version 2 |
| BMI | body mass index |
| CANO | alveolar NO |
| CawNO | NO content in the airway walls |
| CV | coefficient of variation |
| DAS28 | Disease Activity Score for 28 joints |
| DawNO | NO diffusion capacity over the airway wall |
| ESR | erythrocyte sedimentation rate |
| FENO,50 | fraction of exhaled nitric oxide at a flow of 50 mL/s |
| FEV1 | forced expiratory volume at 1 s |
| FVC | forced vital capacity |
| HAQ | Health Assessment Questionnaire |
| HMA | Högman–Meriläinen algorithm |
| Ig | immunoglobulin |
| mAb | monoclonal antibody |
| NO | nitric oxide |
| iNOS | nitric oxide synthase |
| NOx | nitrate/nitrite in serum |
| OD | optical density |
| PAD | peptidyl arginine deaminase |
| Poly Ig | polymeric immunoglobulin |
| ppb | parts per billion |
| RF | rheumatoid factor |
| Rgp | arginine gingipain |
| SC | secretory component |
| CRP | C-reactive protein |
| TNF | tumour necrosis factor |
References
- Scott, D.L.; Wolfe, F.; Huizinga, T.W. Rheumatoid arthritis. Lancet 2010, 376, 1094–1108. [Google Scholar] [CrossRef]
- Rönnelid, J.; Wick, M.C.; Lampa, J.; Lindblad, S.; Nordmark, B.; Klareskog, L.; van Vollenhoven, R. Longitudinal analysis of anti-citrullinated protein/peptide antibodies (anti-CP) during 5 year follow-up in early rheumatoid arthritis: Anti-CP status is a stable phenotype that predicts worse disease activity and greater radiological progression. Ann. Rheum. Dis. 2005, 64, 1744–1749. [Google Scholar] [CrossRef] [PubMed]
- Kastbom, A.; Strandberg, G.; Lindroos, A.; Skogh, T. Anti-CCP antibody test predicts the disease course during 3 years in early rheumatoid arthritis (the Swedish TIRA project). Ann. Rheum. Dis. 2004, 63, 1085–1089. [Google Scholar] [CrossRef]
- Malmström, V.; Catrina, A.I.; Klareskog, L. The immunopathogenesis of seropositive rheumatoid arthritis: From triggering to targeting. Nat. Rev. Immunol. 2017, 17, 60–75. [Google Scholar] [CrossRef] [PubMed]
- Catrina, A.I.; Ytterberg, A.J.; Reynisdottir, G.; Malmstrom, V.; Klareskog, L. Lungs, joints and immunity against citrullinated proteins in rheumatoid arthritis. Nat. Rev. Rheumatol. 2014, 10, 645–653. [Google Scholar] [CrossRef]
- Curran, A.M.; Naik, P.; Giles, J.T.; Darrah, E. PAD enzymes in rheumatoid arthritis: Pathogenic effectors and autoimmune targets. Nat. Rev. Rheumatol. 2020, 16, 301–315. [Google Scholar] [CrossRef]
- Brandtzaeg, P. Mucosal immunity: Induction, dissemination, and effector functions. Scand. J. Immunol. 2009, 70, 505–515. [Google Scholar] [CrossRef] [PubMed]
- Martinsson, K.; Ljungberg, K.R.; Ziegelasch, M.; Cedergren, J.; Eriksson, P.; Klimovich, V.; Reckner, Å.; Griazeva, I.; Sjöwall, C.; Samoylovich, M.; et al. Elevated free secretory component in early rheumatoid arthritis and prior to arthritis development in patients at increased risk. Rheumatology 2020, 59, 979–987. [Google Scholar] [CrossRef]
- de Molon, R.S.; Rossa, C., Jr.; Thurlings, R.M.; Cirelli, J.A.; Koenders, M.I. Linkage of Periodontitis and Rheumatoid Arthritis: Current Evidence and Potential Biological Interactions. Int. J. Mol. Sci. 2019, 20, 4541. [Google Scholar] [CrossRef]
- Kharlamova, N.; Jiang, X.; Sherina, N.; Potempa, B.; Israelsson, L.; Quirke, A.M.; Eriksson, K.; Yucel-Lindberg, T.; Venables, P.J.; Potempa, J.; et al. Antibodies to Porphyromonas gingivalis Indicate Interaction Between Oral Infection, Smoking, and Risk Genes in Rheumatoid Arthritis Etiology. Arthritis Rheumatol. 2016, 68, 604–613. [Google Scholar] [CrossRef]
- Mercado, F.; Marshall, R.I.; Klestov, A.C.; Bartold, P.M. Is there a relationship between rheumatoid arthritis and periodontal disease? J. Clin. Periodontol. 2000, 27, 267–272. [Google Scholar] [CrossRef] [PubMed]
- Ricciardolo, F.L.; Sterk, P.J.; Gaston, B.; Folkerts, G. Nitric oxide in health and disease of the respiratory system. Physiol. Rev. 2004, 84, 731–765. [Google Scholar] [CrossRef] [PubMed]
- Ricciardolo, F.L.; Di Stefano, A.; Sabatini, F.; Folkerts, G. Reactive nitrogen species in the respiratory tract. Eur. J. Pharmacol. 2006, 533, 240–252. [Google Scholar] [CrossRef] [PubMed]
- Farrell, A.J.; Blake, D.R.; Palmer, R.M.; Moncada, S. Increased concentrations of nitrite in synovial fluid and serum samples suggest increased nitric oxide synthesis in rheumatic diseases. Ann. Rheum. Dis. 1992, 51, 1219–1222. [Google Scholar] [CrossRef] [PubMed]
- Högman, M.; Holmkvist, T.; Wegener, T.; Emtner, M.; Andersson, M.; Hedenström, H.; Meriläinen, P. Extended NO analysis applied to patients with COPD, allergic asthma and allergic rhinitis. Respir. Med. 2002, 96, 24–30. [Google Scholar] [CrossRef] [PubMed]
- Högman, M. Extended NO analysis applied to patients with known altered values of exhaled NO. In Disease Markers in Exhaled Breath Basic Mechanism and Clinical Application; Marczin, N., Yacoub, M.H., Eds.; NATO Science Series: Series I: Life Behavioural Sciences 346; IOS Press: Amsterdam, The Netherlands, 2002; pp. 187–190. [Google Scholar]
- Tiev, K.P.; Le-Dong, N.N.; Duong-Quy, S.; Hua-Huy, T.; Cabane, J.; Dinh-Xuan, A.T. Exhaled nitric oxide, but not serum nitrite and nitrate, is a marker of interstitial lung disease in systemic sclerosis. Nitric Oxide 2009, 20, 200–206. [Google Scholar] [CrossRef] [PubMed]
- Thornadtsson, A.; Lind, A.; Weitoft, T.; Högman, M. Altered levels of exhaled nitric oxide in rheumatoid arthritis. Nitric Oxide 2018, 76, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Weitoft, T.; Lind, A.; Larsson, A.; Rönnelid, J.; Högman, M. Exhaled nitric oxide in early rheumatoid arthritis and effects of methotrexate treatment. Sci. Rep. 2022, 12, 6489. [Google Scholar] [CrossRef] [PubMed]
- Aletaha, D.; Neogi, T.; Silman, A.J.; Funovits, J.; Felson, D.T.; Bingham, C.O., 3rd; Birnbaum, N.S.; Burmester, G.R.; Bykerk, V.P.; Cohen, M.D.; et al. 2010 Rheumatoid arthritis classification criteria: An American College of Rheumatology/European League Against Rheumatism collaborative initiative. Ann. Rheum. Dis. 2010, 69, 1580–1588. [Google Scholar] [CrossRef] [PubMed]
- Prevoo, M.L.; van t’Hof, M.A.; Kuper, H.H.; van Leeuwen, M.A.; van de Putte, L.B.; van Riel, P.L. Modified disease activity scores that include twenty-eight-joint counts. Development and validation in a prospective longitudinal study of patients with rheumatoid arthritis. Arthritis Rheum. 1995, 38, 44–48. [Google Scholar] [CrossRef]
- Ekdahl, C.; Eberhardt, K.; Andersson, S.I.; Svensson, B. Assessing disability in patients with rheumatoid arthritis. Scand. J. Rheumatol. 1988, 17, 263–271. [Google Scholar] [CrossRef] [PubMed]
- The American Thoracic Society (ATS); The European Respiratory Society (ERS). ATS/ERS recommendations for standardized procedures for the online and offline measurement of exhaled lower respiratory nitric oxide and nasal nitric oxide, 2005. Am. J. Respir. Crit. Care Med. 2005, 171, 912–930. [Google Scholar] [CrossRef] [PubMed]
- Horváth, I.; Barnes, P.J.; Loukides, S.; Sterk, P.J.; Högman, M.; Olin, A.-C.; Amann, A.; Antus, B.; Baraldi, E.; Bikov, A.; et al. A European Respiratory Society technical standard: Exhaled biomarkers in lung disease. Eur. Respir. J. 2017, 49, 1600965. [Google Scholar] [CrossRef]
- Hedenström, H.; Malmberg, P.; Agarwal, K. Reference values for lung function tests in female. Regression equations with smoking variables. Bull. Eur. Physiopathol. Respir. 1985, 21, 551–557. [Google Scholar] [PubMed]
- Hedenström, H.; Malmberg, P.; Fridriksson, H.V. Reference values for pulmonary function test in men: Regression equations which include tobacco smoking variables. Upsala J. Med. Sci. 1986, 91, 299–310. [Google Scholar] [CrossRef] [PubMed]
- Griazeva, I.V.; Samoĭlovich, M.P.; Klimovich, B.V.; Pavlova, M.S.; Vartanian, N.L.; Kirienko, A.N.; Klimovich, V.B. Monoclonal antibodies against human secretory component: Epitope specificity and utility for immunoanalysis. Zhurnal Mikrobiol. Epidemiol. Immunobiol. 2010, 4, 54–59. [Google Scholar]
- Malinovschi, A.; Janson, C.; Holmkvist, T.; Norbäck, D.; Meriläinen, P.; Högman, M. Effect of smoking on exhaled nitric oxide and flow-independent nitric oxide exchange parameters. Eur. Respir. J. 2006, 28, 339–345. [Google Scholar] [CrossRef] [PubMed]
- Tarbiah, N.; Todd, I.; Tighe, P.J.; Fairclough, L.C. Cigarette smoking differentially affects immunoglobulin class levels in serum and saliva: An investigation and review. Basic Clin. Pharmacol. Toxicol. 2019, 125, 474–483. [Google Scholar] [CrossRef] [PubMed]
- Matson, S.M.; Demoruelle, M.K.; Castro, M. Airway Disease in Rheumatoid Arthritis. Ann. Am. Thorac. Soc. 2022, 19, 343–352. [Google Scholar] [CrossRef]
- Jonsson, T.; Thorsteinsson, J.; Valdimarsson, H. Does smoking stimulate rheumatoid factor production in non-rheumatic individuals? APMIS 1998, 106, 970–974. [Google Scholar] [CrossRef]
- Manfredsdottir, V.F.; Vikingsdottir, T.; Jonsson, T.; Geirsson, A.J.; Kjartansson, O.; Heimisdottir, M.; Sigurdardottir, S.L.; Valdimarsson, H.; Vikingsson, A. The effects of tobacco smoking and rheumatoid factor seropositivity on disease activity and joint damage in early rheumatoid arthritis. Rheumatology 2006, 45, 734–740. [Google Scholar] [CrossRef] [PubMed]
- Abhishek, A.; Butt, S.; Gadsby, K.; Zhang, W.; Deighton, C.M. Anti-TNF-alpha agents are less effective for the treatment of rheumatoid arthritis in current smokers. J. Clin. Rheumatol. 2010, 16, 15–18. [Google Scholar] [CrossRef]
- Bobbio-Pallavicini, F.; Caporali, R.; Alpini, C.; Avalle, S.; Epis, O.M.; Klersy, C.; Montecucco, C. High IgA rheumatoid factor levels are associated with poor clinical response to tumour necrosis factor alpha inhibitors in rheumatoid arthritis. Ann. Rheum. Dis. 2007, 66, 302–307. [Google Scholar] [CrossRef] [PubMed]
- Otten, H.G.; Daha, M.R.; Maarl, M.G.J.; Hoogendoorn, L.I.; Beem, E.M.; Rooy, H.H.; Breedveld, F.C. IgA rheumatoid factor in mucosal fluids and serum of patients with rheumatoid arthritis: Immunological aspects and clinical significance. Clin. Exp. Immunol. 1992, 90, 256–259. [Google Scholar] [CrossRef] [PubMed]
- Marcolongo, R., Jr.; Carcassi, A.; Frullini, F.; Bianco, G.; Bravi, A. Levels of serum immunoglobulins in patients with rheumatoid arthritis. Ann. Rheum. Dis. 1967, 26, 412–418. [Google Scholar] [CrossRef] [PubMed]
- Veys, E.M.; Claessens, H.E. Serum levels of IgG, IgM, and IgA in rheumatoid arthritis. Ann. Rheum. Dis. 1968, 27, 431–440. [Google Scholar] [CrossRef] [PubMed]
- Aho, K.; Heliovaara, M.; Knekt, P.; Reunanen, A.; Aromaa, A.; Leino, A.; Kurki, P.; Heikkila, R.; Palosuo, T. Serum immunoglobulins and the risk of rheumatoid arthritis. Ann. Rheum. Dis. 1997, 56, 351–356. [Google Scholar] [CrossRef] [PubMed]
- He, Y.; Zha, Q.; Liu, D.; Lu, A. Relations between serum IgA level and cartilage erosion in 436 cases of rheumatoid arthritis. Immunol. Invest. 2007, 36, 285–291. [Google Scholar] [CrossRef] [PubMed]
- Blackburn, J.B.; Schaff, J.A.; Gutor, S.; Du, R.-H.; Nichols, D.; Sherrill, T.; Gutierrez, A.J.; Xin, M.K.; Wickersham, N.; Zhang, Y.; et al. Secretory cells are the primary source of pIgR in small airways. Am. J. Respir. Cell Mol. Biol. 2022, 67, 334–345. [Google Scholar] [CrossRef]
- Pilette, C.; Ouadrhiri, Y.; Godding, V.; Vaerman, J.P.; Sibille, Y. Lung mucosal immunity: Immunoglobulin-A revisited. Eur. Respir. J. 2001, 18, 571–588. [Google Scholar] [CrossRef]
- Klareskog, L.; Stolt, P.; Lundberg, K.; Kallberg, H.; Silva, C.; Grunewald, J.; Ronnelid, J.; Harris, H.E.; Ulfgren, A.; Dahlqvist, S.R.; et al. A new model for an etiology of RA; Smoking may trigger HLA-DR (SE)—Restricted immune reactions to autoantigens modified by citrullination. Arthritis Rheum. 2006, 54, 38–46. [Google Scholar] [CrossRef] [PubMed]
- Herlitz-Cifuentes, H.; Vejar, C.; Flores, A.; Jara, P.; Bustos, P.; Castro, I.; Poblete, E.; Saez, K.; Opazo, M.; Gajardo, J.; et al. Plasma from patients with rheumatoid arthritis reduces nitric oxide synthesis and induces reactive oxygen species in a cell-based biosensor. Biosensors 2019, 9, 32. [Google Scholar] [CrossRef]
- Chandrasekharan, U.M.; Wang, Z.; Wu, Y.; Tang, W.H.W.; Hazen, S.L.; Wang, S.; Husni, M.E. Elevated levels of plasma symmetric dimethylarginine and increased arginase activity as potential indicators of cardiovascular comorbidity in rheumatoid arthritis. Arthritis Res. Ther. 2018, 20, 123. [Google Scholar] [CrossRef] [PubMed]
- Pertsinidou, E.; Manivel, V.A.; Klareskog, L.; Alfredsson, L.; Mathsson, L.; Hansson, M.; Cornillet, M.; Serre, G.; Holmdahl, R.; Skriner, K.; et al. In early rheumatoid arthritis anti-citrullinated peptide antibodies associate with lower number of affected joints, and Rheumatoid factor associates with systemic inflammation. Ann. Rheum. Dis. 2024, 83, 277–287. [Google Scholar] [CrossRef] [PubMed]
- Koduri, G.; Solomon, J.J. Identification, Monitoring, and Management of Rheumatoid Arthritis-Associated Interstitial Lung Disease. Arthritis Rheumatol. 2023, 75, 2067–2077. [Google Scholar] [CrossRef]
| All (n = 44) | Non-Smokers (n = 32) | Current Smokers (n = 12) | p-Value | |
|---|---|---|---|---|
| Age (years) | 60 ± 14 | 59 ± 16 | 62 ± 9 | 0.521 |
| Sex (% female) | 59% | 66% | 42% | 0.150 |
| BMI | 28 ± 5 | 28 ± 5 | 28 ± 4 | 0.665 |
| Symptom duration (months) | 4 (2, 8) | 5 (3, 8) | 3 (2,11) | 0.195 |
| DAS28 | 4.47 ± 1.06 | 4.40 ± 1.19 | 4.65 ± 0.59 | 0.495 |
| - Swollen joints | 4 (2, 6) | 3 (2, 5) | 5 (3, 8) | 0.153 |
| - Tender joints | 4 (2, 5) | 4 (2, 6) | 4 (1, 5) | 0.866 |
| - Global health | 45 ± 24 | 42 ± 25 | 50 ± 21 | 0.478 |
| - ESR | 24 (12, 42) | 20 (12, 40) | 30 (13, 42) | 0.679 |
| CRP | 7.9 (3.2, 20) | 8.1 (2.6, 23) | 7.1 (3.3, 14) | 0.576 |
| NOx | 2.3 (1.8, 3.1) | 2.4 (1.6, 3.1) | 2.1 (1.3, 3.0) | 0.243 |
| HAQ | 0.92 ± 0.46 | 0.89 ± 0.47 | 0.98 ± 0.45 | 0.591 |
| Immunological markers | ||||
| IgA anti-CCP2 AU/mL | 10.5 (3.1, 23) | 6.8 (3.6, 21) | 15.5 (2.4, 28) | 0.668 |
| IgG anti-CCP2 AU/mL | 299 (93, 600) | 288 (122, 600) | 527 (56, 600) | 0.706 |
| IgA RF AU/mL | 26 (12, 94) | 25 (10, 56) | 56 (25, 136) | 0.059 |
| IgM RF IU/mL | 62 (20, 179) | 49 (18, 131) | 165 (34, 250) | 0.100 |
| Anti-Rgp AU/mL | 361 (174, 727) | 348 (168, 766) | 369 (175, 563) | 0.969 |
| SC | 0 (0, 30) | 0 (0, 13) | 24 (0, 47) | 0.035 |
| SC ACPAs | 29 (12, 130) | 24 (11, 62) | 119 (16, 217) | 0.082 |
| Total IgA g/L | 4.72 (3.77, 6.07) | 4.76 (3.78, 6.31) | 4.72 (3.85, 6.07) | 0.645 |
| Total IgA1, g/L | 4.15 (2.74, 7.18) | 4.15 (2.75, 7.18) | 4.32 (2.56, 9.25) | 1.000 |
| Total IgA2, g/L | 0.81 (0.48, 1.25) | 0.78 (0.45, 1.23) | 0.90 (0.54, 1.66) | 0.377 |
| NO analysis | ||||
| FENO,50 ppb | 16 (10, 24) | 19 (13, 25) | 10 (5, 16) | 0.002 |
| CANO ppb | 1.6 (1.0, 2.2) | 1.9 (1.2, 2.3) | 1.0 (0.4, 1.3) | 0.004 |
| CawNO ppb (n = 34) | 55 (24, 106) | 64 (33, 115) | 24 (20, 75) | 0.086 |
| DawNO mL/s (n = 34) | 17 (8, 30) | 16 (9, 31) | 19 (7, 29) | 0.809 |
| Lung function | ||||
| FEV1 % predicted | 84 ± 15 | 87 ± 15 | 78 ± 15 | 0.446 |
| FVC % predicted | 86 ± 11 | 87 ± 11 | 84 ± 13 | 0.071 |
| FENO,50 (n = 44) | CawNO (n = 34) | DawNO (n = 34) | CANO (n = 44) | |||||
|---|---|---|---|---|---|---|---|---|
| Rho | p-Value | Rho | p-Value | Rho | p-Value | Rho | p-Value | |
| DAS28 | ||||||||
| Total score | −0.248 | 0.109 | −0.327 | 0.063 | 0.150 | 0.406 | 0.169 | 0.279 |
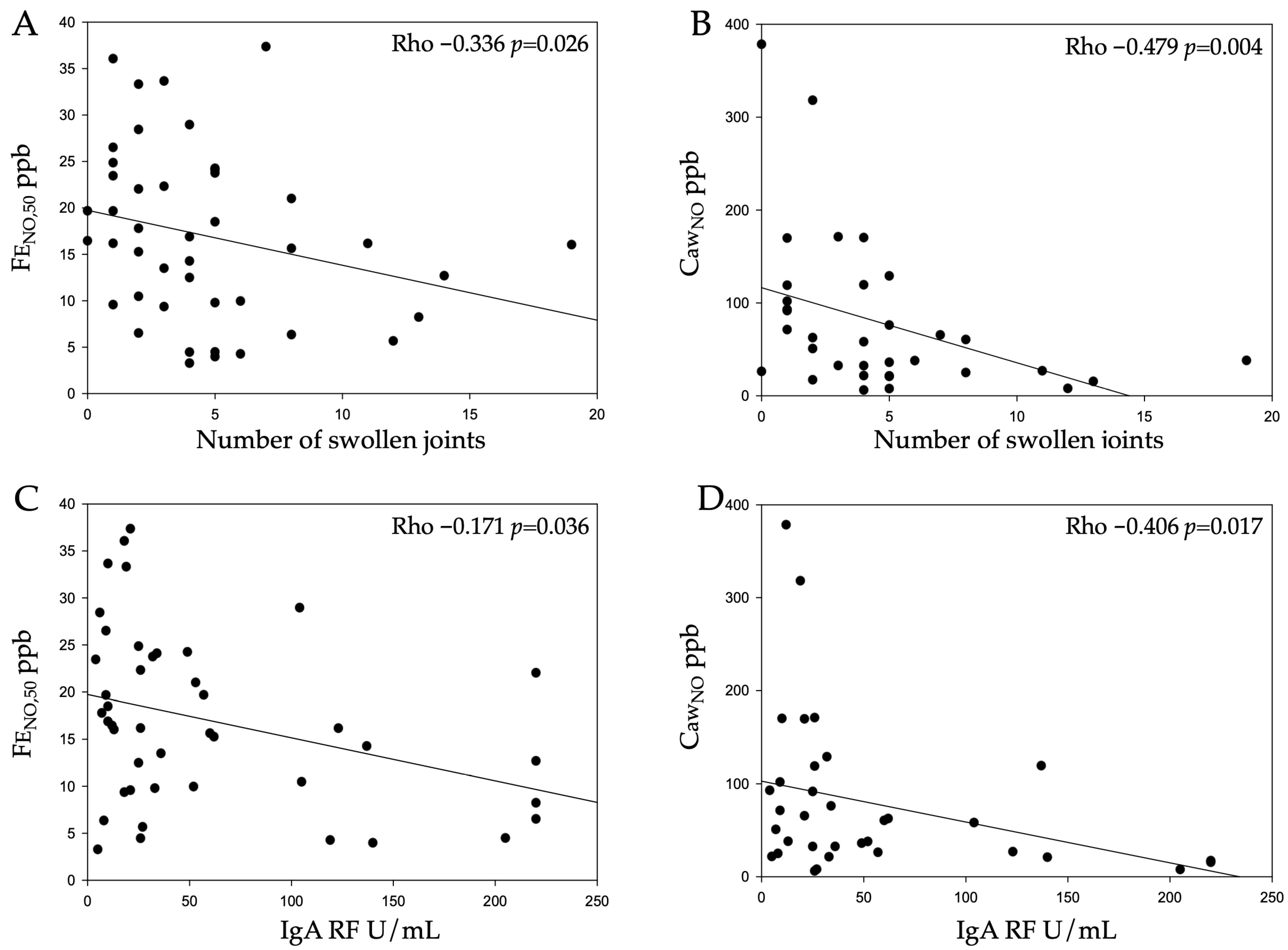
| Number of swollen joints | −0.336 | 0.026 | −0.479 | 0.004 * | 0.385 | 0.025 | 0.023 | 0.882 |
| Number of tender joints | −0.171 | 0.268 | −0.140 | 0.430 | −0.076 | 0.669 | −0.025 | 0.870 |
| Global Health | −0.130 | 0.406 | −0.023 | 0.901 | −0.129 | 0.473 | −0.068 | 0.666 |
| ESR | −0.001 | 0.995 | −0.111 | 0.532 | 0.167 | 0.346 | 0.285 | 0.064 |
| CRP | 0.030 | 0.848 | −0.136 | 0.442 | 0.172 | 0.332 | 0.362 | 0.016 |
| Immunological markers | ||||||||
| IgA anti-CCP2 | −0.113 | 0.465 | −0.180 | 0.309 | 0.138 | 0.435 | 0.063 | 0.686 |
| IgG anti-CCP2 | −0.148 | 0.311 | −0.181 | 0.305 | 0.107 | 0.547 | −0.027 | 0.860 |
| IgA RF | −0.171 | 0.036 | −0.406 | 0.017 | 0.357 | 0.037 | −0.001 | 0.994 |
| IgM RF | −0.173 | 0.262 | −0.151 | 0.394 | 0.001 | 0.994 | 0.039 | 0.804 |
| IgG anti-Rgp | −0.063 | 0.683 | −0.044 | 0.803 | 0.027 | 0.878 | 0.015 | 0.922 |
| SC | −0.173 | 0.266 | −0.007 | 0.971 | −0.160 | 0.366 | −0.345 | 0.023 |
| SC ACPA | −0.255 | 0.103 | −0.050 | 0.783 | −0.093 | 0.608 | −0.131 | 0.408 |
| Total IgA | 0.184 | 0.231 | 0.087 | 0.624 | 0.141 | 0.427 | 0.071 | 0.646 |
| Total IgA1 | 0.270 | 0.076 | −0.035 | 0.844 | 0.242 | 0.168 | 0.128 | 0.406 |
| Total IgA2 | 0.062 | 0.689 | −0.169 | 0.339 | 0.130 | 0.462 | −0.047 | 0.759 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Weitoft, T.; Rönnelid, J.; Lind, A.; de Vries, C.; Larsson, A.; Potempa, B.; Potempa, J.; Kastbom, A.; Martinsson, K.; Lundberg, K.; et al. Exhaled Nitric Oxide Reflects the Immune Reactions of the Airways in Early Rheumatoid Arthritis. Biomedicines 2024, 12, 964. https://doi.org/10.3390/biomedicines12050964
Weitoft T, Rönnelid J, Lind A, de Vries C, Larsson A, Potempa B, Potempa J, Kastbom A, Martinsson K, Lundberg K, et al. Exhaled Nitric Oxide Reflects the Immune Reactions of the Airways in Early Rheumatoid Arthritis. Biomedicines. 2024; 12(5):964. https://doi.org/10.3390/biomedicines12050964
Chicago/Turabian StyleWeitoft, Tomas, Johan Rönnelid, Anders Lind, Charlotte de Vries, Anders Larsson, Barbara Potempa, Jan Potempa, Alf Kastbom, Klara Martinsson, Karin Lundberg, and et al. 2024. "Exhaled Nitric Oxide Reflects the Immune Reactions of the Airways in Early Rheumatoid Arthritis" Biomedicines 12, no. 5: 964. https://doi.org/10.3390/biomedicines12050964
APA StyleWeitoft, T., Rönnelid, J., Lind, A., de Vries, C., Larsson, A., Potempa, B., Potempa, J., Kastbom, A., Martinsson, K., Lundberg, K., & Högman, M. (2024). Exhaled Nitric Oxide Reflects the Immune Reactions of the Airways in Early Rheumatoid Arthritis. Biomedicines, 12(5), 964. https://doi.org/10.3390/biomedicines12050964






