Current Knowledge on the Use of Neuromonitoring in Thyroid Surgery
Abstract
1. Introduction
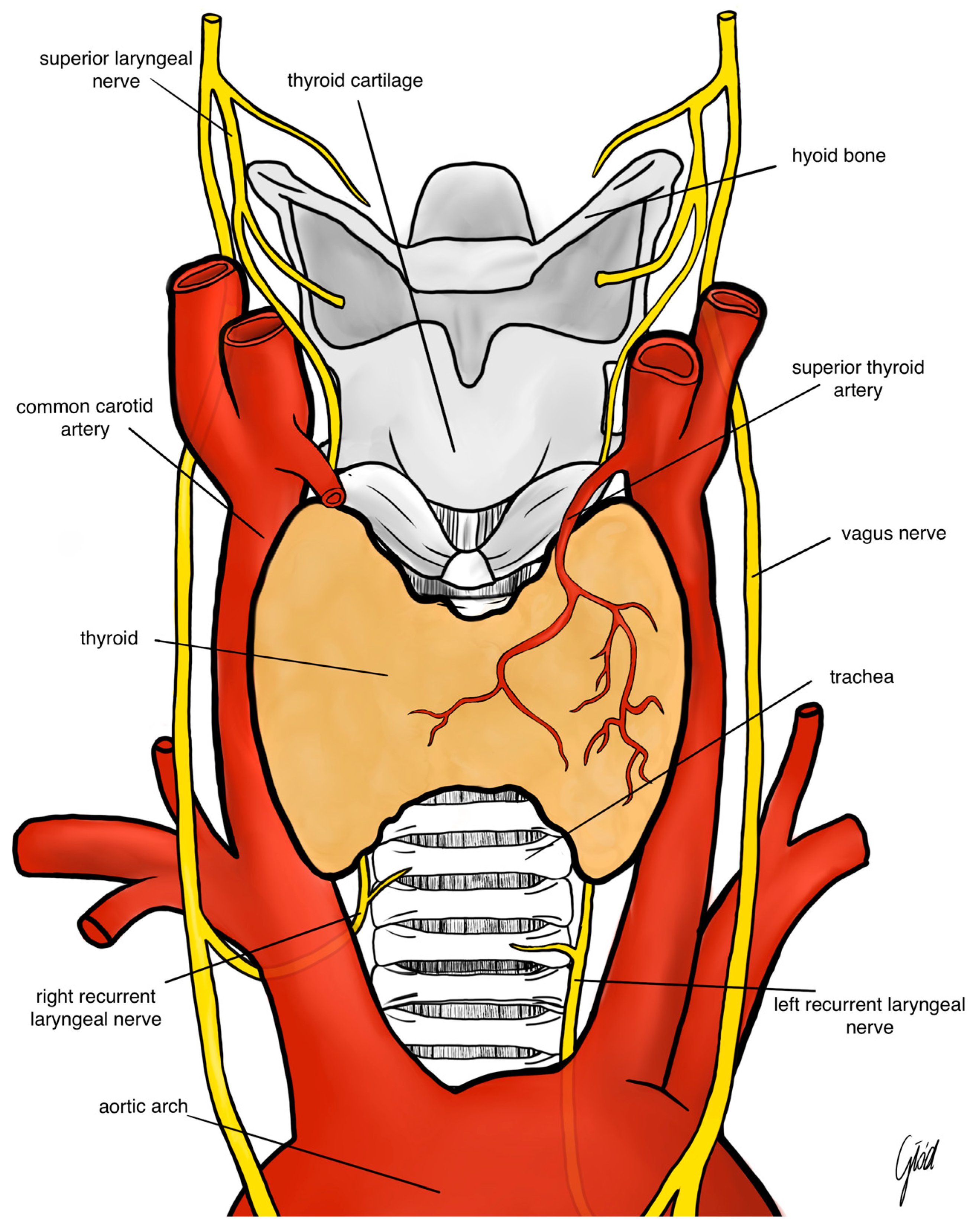
2. Anatomy of the Laryngeal Nerves
3. Mechanisms and Risk Factors of Nerve Injury
4. Consequences and Diagnosis
5. Monitoring of the Laryngeal Nerves
5.1. History
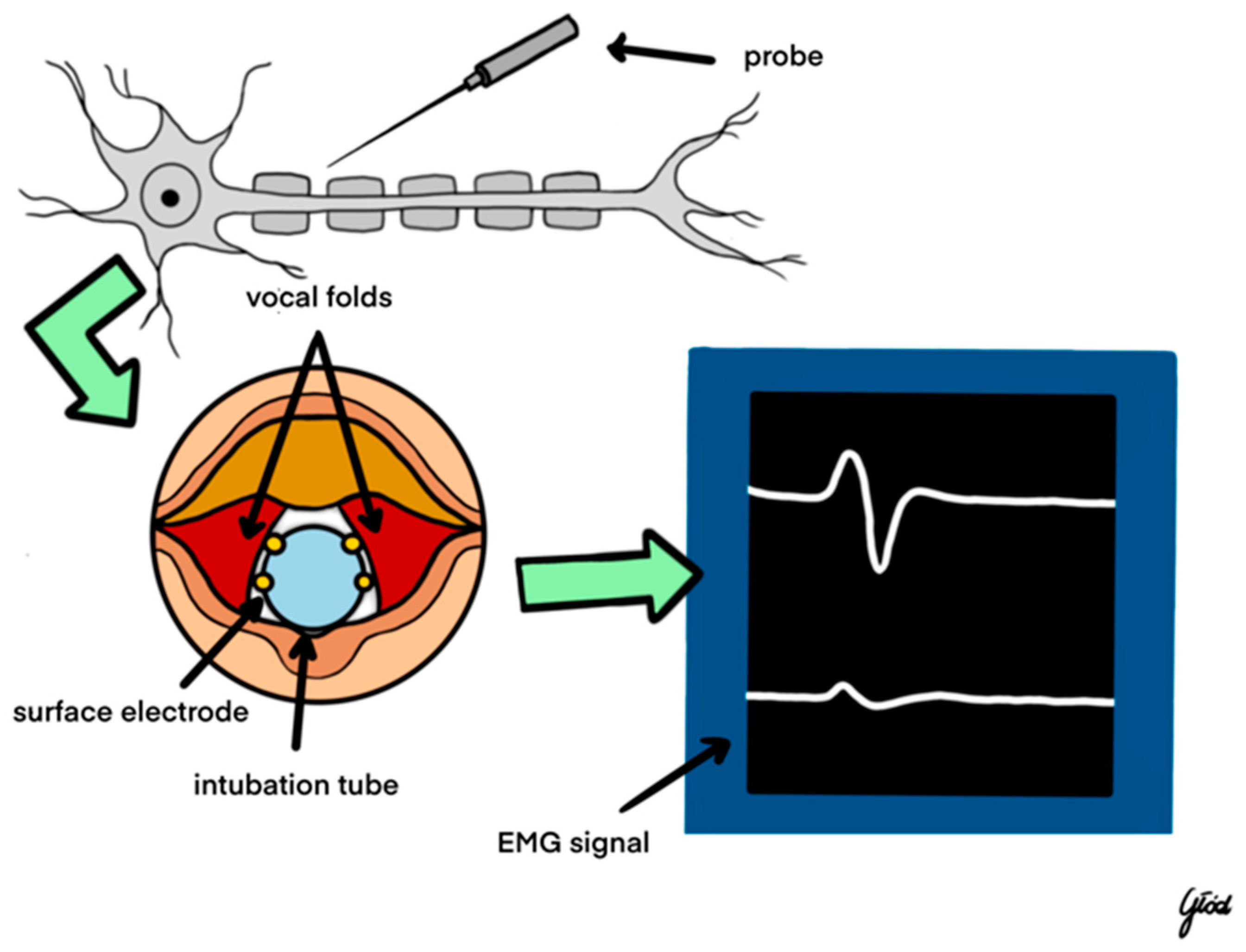
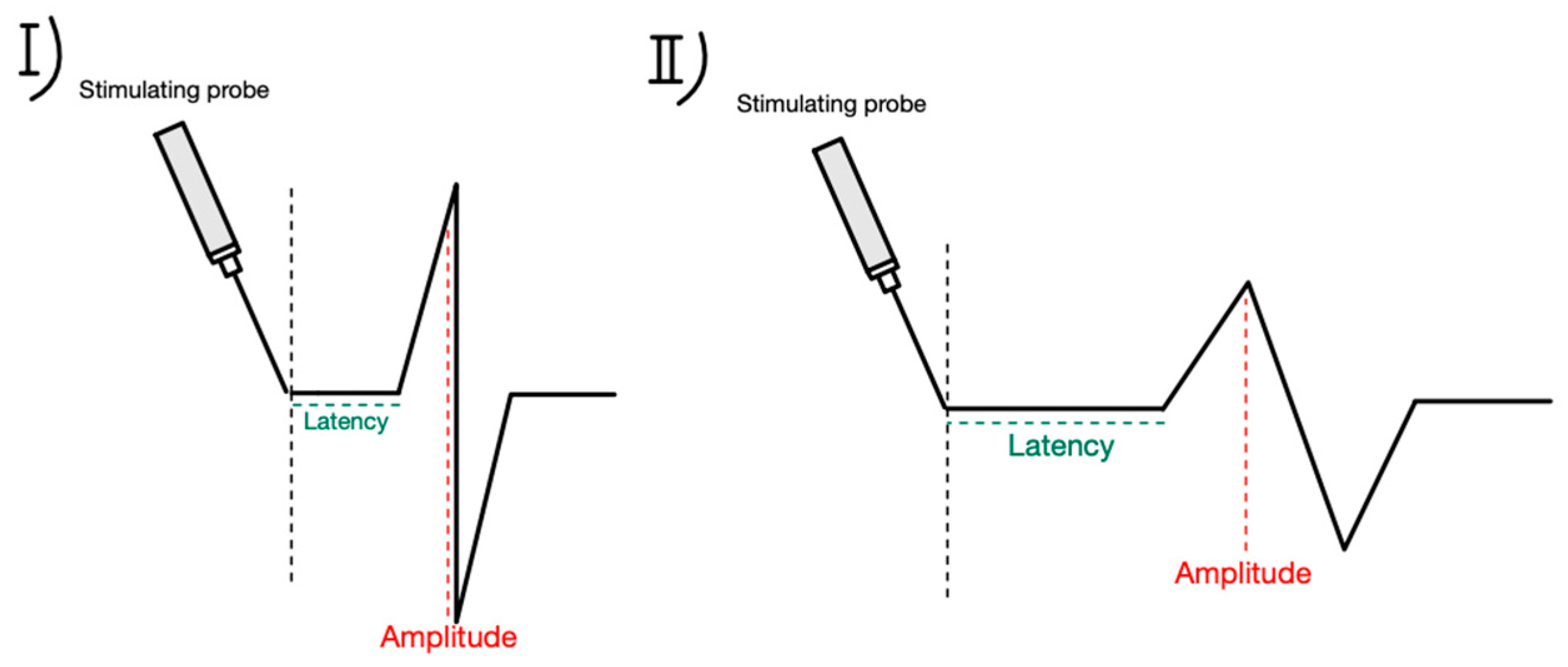
5.2. Technique
5.3. Types of IONM
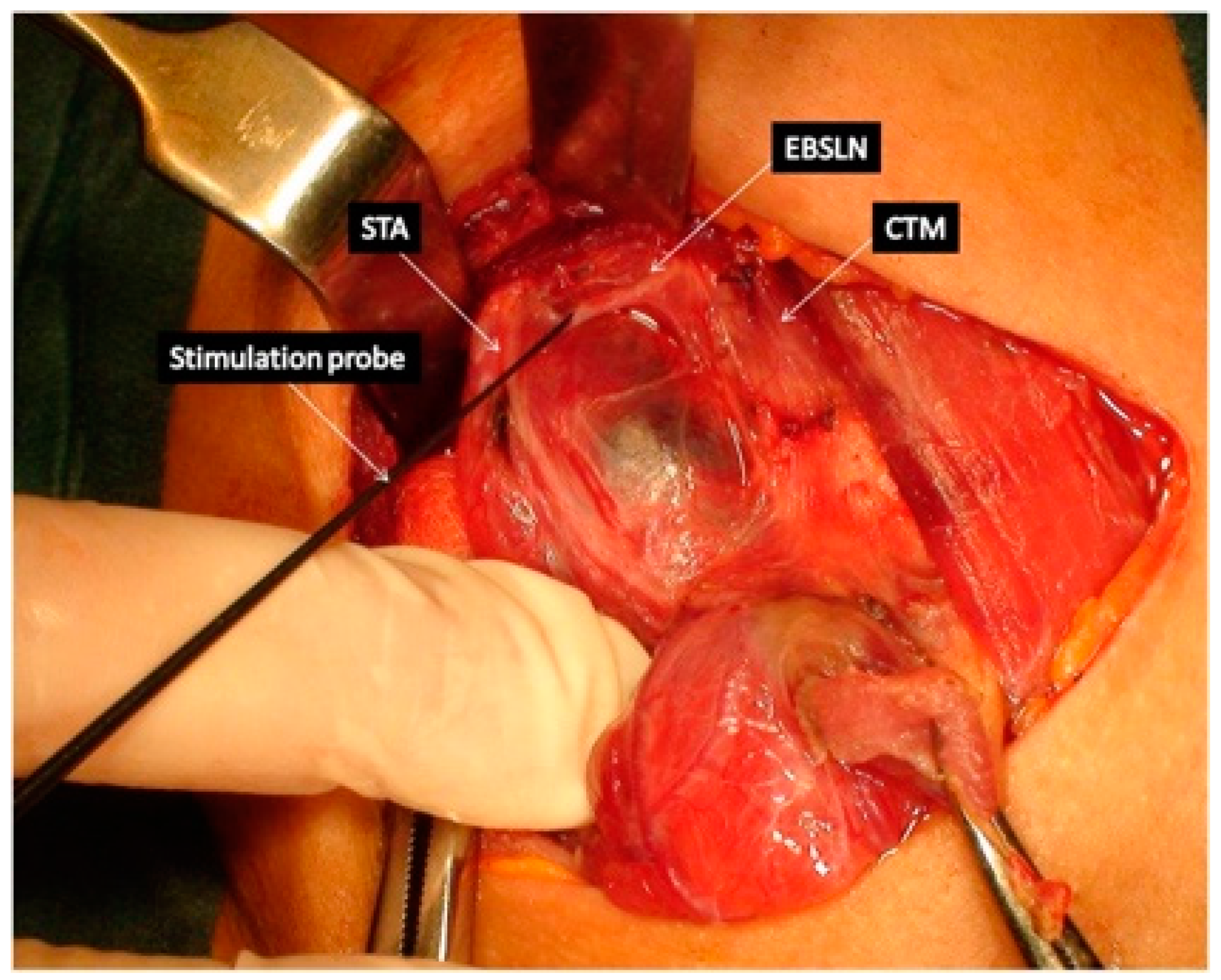
5.4. Use of IONM in EBSLN Assessment
6. Clinical Aspects of the Use of Neuromonitoring in Thyroid Surgery
6.1. Complete Removal of Thyroid Tissue
6.2. IONM in the Reduction of RLN Injury
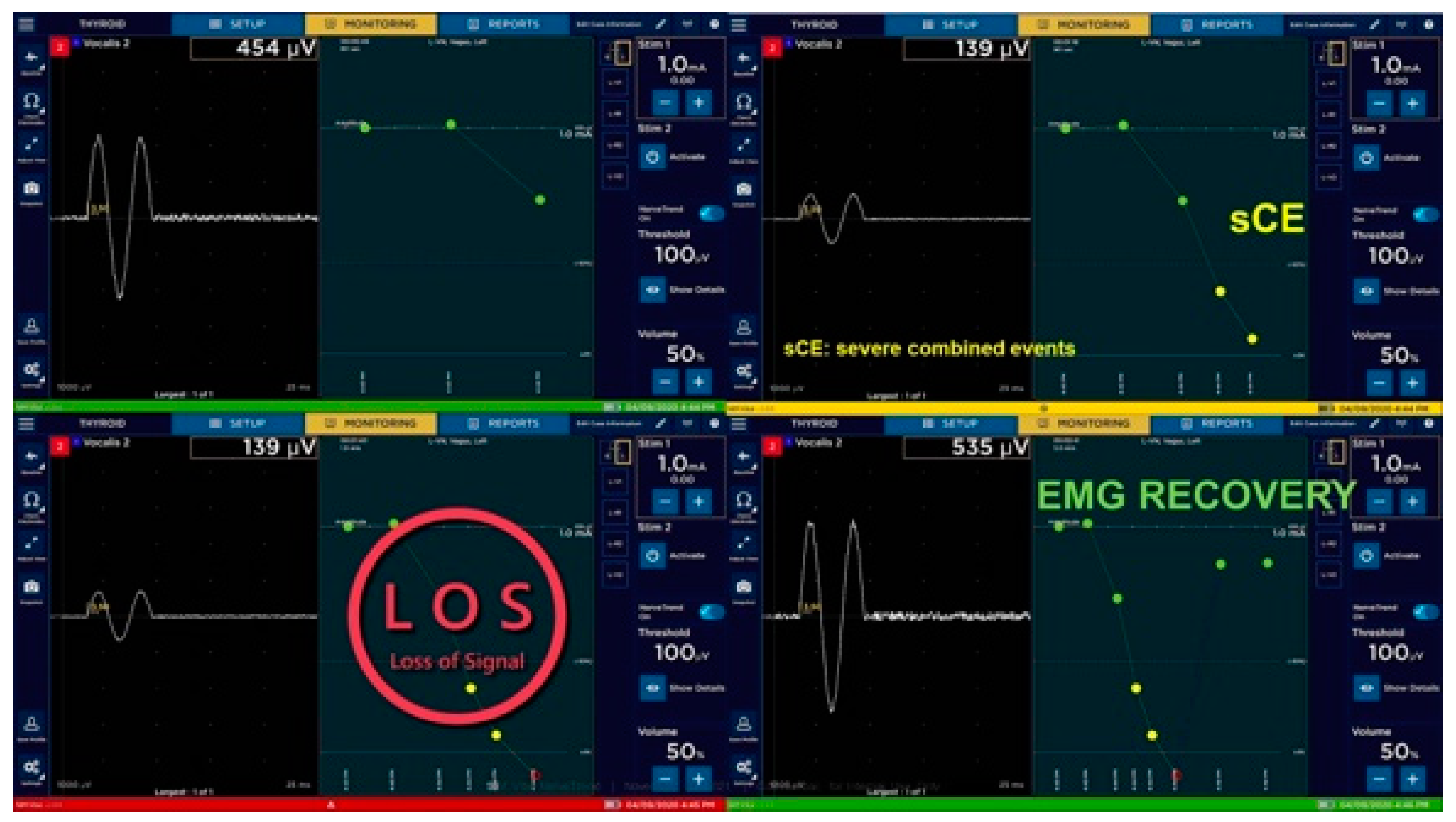
6.3. Loss of Signal during Thyroidectomy and Decision Making
6.4. Educational Value of Intraoperative Neuromonitoring: Research Studies
7. Medical and Legal Aspects of Neuromonitoring Use in Thyroid Surgery
8. Prospects for the Development of Neuromonitoring around the World
9. Safety of Intraoperative Neuromonitoring
10. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Agency for Healthcare Research and Quality. Statistical Brief 86. Healthcare Cost and Utilization Project (HCUP). 2010. Available online: http://www.hcup-us.ahrq.gov/reports/statbriefs/sb86.jsp (accessed on 4 June 2012).
- Rusinek, D.; Chmielik, E.; Krajewska, J.; Jarzab, M.; Oczko-Wojciechowska, M.; Czarniecka, A.; Jarzab, B. Current Advances in Thyroid Cancer Management. Are We Ready for the Epidemic Rise of Diagnoses? Int. J. Mol. Sci. 2017, 18, 1817. [Google Scholar] [CrossRef] [PubMed]
- Jeannon, J.P.; Orabi, A.A.; Bruch, G.A.; Abdalsalam, H.A.; Simo, R. Diagnosis of recurrent laryngeal nerve palsy after thyroidectomy: A systematic review. Int. J. Clin. Pract. 2009, 63, 624–629. [Google Scholar] [CrossRef]
- Chandrasekhar, S.S.; Randolph, G.W.; Seidman, M.D.; Rosenfeld, R.M.; Angelos, P.; Barkmeier-Kraemer, J.; Benninger, M.S.; Blumin, J.H.; Dennis, G.; Hanks, J.; et al. Clinical Practice Guideline: Improving voice outcomes after thyroid surgery. Otolaryngol. Head Neck Surg. 2013, 148, S1–S37. [Google Scholar]
- Randolph, G.W.; Dralle, H.; Abdullah, H.; International Intraoperative Monitoring Study Group. Electrophysiologic recurrent laryngeal nerve monitoring during thyroid and parathyroid surgery: International standards guideline statement. Laryngoscope 2011, 122, S1–S16. [Google Scholar] [CrossRef] [PubMed]
- Barczyński, M.; Randolph, G.W.; Cernea, C.R.; Dralle, H.; Dionigi, G.; Alesina, P.F.; Mihai, R.; Finck, R.; Finck, C.; Lombardi, D.; et al. External branch of the superior laryngeal nerve monitoring during thyroid and parathyroid surgery: International neural monitoring study Group standards guideline statement. Laryngoscope 2013, 123, S1–S14. [Google Scholar] [CrossRef] [PubMed]
- Schneider, R.; Randolph, G.W.; Dionigi, G.; Wu, C.W.; Barczynski, M.; Chiang, F.Y.; Al-Quaryshi, Z.; Angelos, P.; Brauckhoff, K.; Cernea, C.R.; et al. International neural monitoring study group guideline 2018 part I: Staging bilateral thyroid surgery with monitoring loss of signal. Laryngoscope 2018, 128, S1–S17. [Google Scholar] [CrossRef]
- Wu, C.W.; Dionigi, G.; Barczynski, M.; Chiang, F.Y.; Dralle, H.; Schneider, R.; Al-Quaryshi, Z.; Angelos, P.; Brauckhoff, K.; Brooks, J.A.; et al. International neuromonitoring study group guidelines 2018: Part II: Optimal recurrent laryngeal nerve management for invasive thyroid cancer-incorporation of surgical, laryngeal, and neural electrophysiologic data. Laryngoscope 2018, 128, S18–S27. [Google Scholar] [CrossRef]
- Pardal-Refoyo, J.L.; Parente-Arias, P.; Arroyo-Domingo, M.M.; Maza-Solano, J.M.; Granell-Navarro, J.; Martínez-Salazar, J.M.; Moreno-Luna, R.; Vargas-Yglesias, E. Recommendations on the use of neuromonitoring in thyroid and parathyroid surgery. Acta Otorrinolaringol. Esp. 2018, 69, 231–242. [Google Scholar] [CrossRef]
- Feng, A.L.; Puram, S.V.; Singer, M.C.; Modi, R.; Kamani, D.; Randolph, G.W. Increased prevalence of neural monitoring during thyroidectomy: Global surgical survey. Laryngoscope 2020, 130, 1097–1104. [Google Scholar] [CrossRef]
- Bartsch, D.K.; Dotzenrath, C.; Vorländer, C.; Zielke, A.; Weber, T.; Buhr, H.J.; Klinger, C.; Lorenz, K.; The StuDoQ/Thyroid Study Group TSS. Current Practice of Surgery for Benign Goitre—An Analysis of the Prospective DGAV StuDoQ|Thyroid Registry. J. Clin. Med. 2019, 8, 477. [Google Scholar] [CrossRef]
- Randolph, G. The Recurrent and Superior Laryngeal Nerve; Springer: New York, NY, USA, 2016. [Google Scholar] [CrossRef]
- Wojtczak, B.; Kaliszewski, K.; Sutkowski, K.; Bolanowski, M.; Barczyński, M. A functional assessment of anatomical variants of the recurrent laryngeal nerve during thyroidectomies using neuromonitoring. Endocrine 2018, 59, 82–89. [Google Scholar] [CrossRef] [PubMed]
- Fundakowski, C.E.; Hales, N.W.; Agrawal, N.; Barczyński, M.; Camacho, P.M.; Hartl, D.M.; Kandil, E.; Liddy, W.E.; McKenzie, T.J.; Morris, J.C. Surgical management of the recurrent laryngeal nerve in thyroidectomy: American Head and Neck Society Consensus Statement. Head Neck 2018, 40, 663–675. [Google Scholar] [CrossRef] [PubMed]
- Bevan, K.; Griffiths, M.V.; Morgan, M.H. Cricothyroid muscle paralysis: Its recognition and diagnosis. J. Laryngol. Otol. 1989, 103, 191–195. [Google Scholar] [CrossRef]
- Jansson, S.; Tisell, L.E.; Hagne, I.; Sanner, E.; Stenborg, R.; Svensson, P. Partial laryngeal nerve lesions before and after thyroid surgery. World J. Surg. 1988, 12, 522–527. [Google Scholar] [CrossRef]
- Teitelbaum, B.J.; Wenig, B.L. Superior laryngeal nerve injury from thyroid surgery. Head Neck 1995, 17, 36–40. [Google Scholar] [CrossRef]
- Cernea, C.R.; Ferraz, A.R.; Nishio, S.; Dutra, A., Jr.; Hojaij, F.C.; dos Santos, L.R. Surgical anatomy of the external branch of the superior laryngeal nerve. Head Neck 1992, 14, 380–383. [Google Scholar] [CrossRef]
- Lahey, F.H. Routine dissection and demonstration recurrent laryngeal nerve in subtotal thyroidectomy. Surg. Gynecol. Obstet. 1938, 66, 775–777. [Google Scholar]
- Shedd, D.P.; Burget, G.C. Identification of the recurrent laryngeal nerve. Arch. Surg. 1966, 92, 861–864. [Google Scholar] [CrossRef]
- Riddell, V. Thyroidectomy: Prevention of bilateral recurrent nerve palsy. Results of identification of the nerve over 23 consecutive years (1946–1969) with a description of an additional safety measure. Br. J. Surg. 1970, 57, 111. [Google Scholar]
- Gavilán, J.; Gavilan, C. Recurrent laryngeal nerve. Identification during thyroid and parathyroid surgery. Arch. Otolaryngol. Head Neck Surg. 1986, 112, 1286–1288. [Google Scholar] [CrossRef] [PubMed]
- Sinclair, C.F.; Kamani, D.; Gregory, W. The evolution and progress of standard procedures for intraoperative nerve monitoring. Ann. Thyroid 2019, 4. [Google Scholar] [CrossRef]
- Wojtczak, B.; Sutkowski, K.; Kaliszewski, K.; Barczyński, M.; Bolanowski, M. Thyroid reoperation using intraoperative neuromonitoring. Endocrine 2017, 58, 458–466. [Google Scholar] [CrossRef]
- Barczyński, M.; Konturek, A. Clinical validation of NerveTrend versus conventional i-IONM mode of NIM Vital in prevention of recurrent laryngeal nerve events during bilateral thyroid surgery: A randomized controlled trial. Head Neck 2023, 46, 492–502. [Google Scholar] [CrossRef] [PubMed]
- Schneider, R.; Machens, A.; Randolph, G.; Kamani, D.; Lorenz, K.; Dralle, H. Impact of continuous intraoperative vagus stimulation on intraoperative decision making in favor of or against bilateral surgery in benign goiter. Best Pract. Res. Clin. Endocrinol. Metab. 2019, 33, 101285. [Google Scholar] [CrossRef]
- Stankovic, P.; Wittlinger, J.; Georgiew, R.; Dominas, N.; Hoch, S.; Wilhelm, T. Continuous intraoperative neuromonitoring (cIONM) in head and neck surgery—A review. HNO 2020, 68, 86–92. [Google Scholar] [CrossRef]
- Lamadé, W.; Meyding-Lamadé, U.; Buchhold, C.; Brauer, M.; Brandner, R.; Uttenweiler, V.; Motsch, J.; Klar, E.; Herfarth, C. First continuous nerve monitoring in thyroid gland surgery. Chirurg 2000, 71, 551–557. [Google Scholar]
- Phelan, E.; Schneider, R.; Lorenz, K.; Dralle, H.; Kamani, D.; Potenza, A.; Sritharan, N.; Shin, J.; Randolph, G.W. Continuous vagal IONM prevents recurrent laryngeal nerve paralysis by revealing initial EMG changes of impending neuropraxic injury: A prospective, multicenter study. Laryngoscope 2014, 124, 1498–1505. [Google Scholar] [CrossRef]
- Schneider, R.; Randolph, G.W.; Sekulla, C.; Phelan, E.; Thanh, P.N.; Bucher, M.; Machens, A.; Dralle, H.; Lorenz, K. Continuous intraoperative vagus nerve stimulation for identification of imminent recurrent laryngeal nerve injury. Head Neck 2013, 35, 1591–1598. [Google Scholar] [CrossRef]
- Kark, A.E.; Kissin, M.W.; Auerbach, R.; Meikle, M. Voice changes after thyroidectomy: Role of the external laryngeal nerve. Br. Med. J. 1984, 289, 1412–1415. [Google Scholar] [CrossRef]
- Lekacos, N.L.; Miligos, N.D.; Tzardis, P.J.; Majiatis, S.; Patoulis, J. The superior laryngeal nerve in thyroidectomy. Am. Surg. 1987, 53, 610–612. [Google Scholar]
- Lynch, J.; Parameswaran, R. Management of unilateral recurrent laryngeal nerve injury after thyroid surgery: A review. Head Neck 2017, 39, 1470–1478. [Google Scholar] [CrossRef] [PubMed]
- Cossa, A.; Castagnola, G.; Romeo, G.; Bellucci, M.; Nigri, G.; Bellotti, C. Utility of intraoperative neuromonitoring in detecting recurrent nerve’s anatomical anomalies during thyroidectomy. Endocrine 2020, 70, 194–197. [Google Scholar] [CrossRef] [PubMed]
- Barczyński, M.; Konturek, A.; Stopa, M.; Hubalewska-Dydejczyk, A.; Richter, P.; Nowak, W. Clinical value of intraoperative neuromonitoring of the recurrent laryngeal nerves in improving outcomes of surgery for well-differentiated thyroid cancer. Pol. Przegl. Chir. 2011, 83, 196–203. [Google Scholar] [CrossRef][Green Version]
- Serpell, J.W.; Yeung, M.J.; Grodski, S. The motor fibers of the recurrent laryngeal nerve are located in the anterior extralaryngeal branch. Ann. Surg. 2009, 249, 648–652. [Google Scholar] [CrossRef]
- Wojtczak, B.; Kaliszewski, K.; Sutkowski, K.; Głód, M.; Barczyński, M. The learning curve for intraoperative neuromonitoring of the recurrent laryngeal nerve in thyroid surgery. Langenbecks Arch. Surg. 2017, 402, 701–708. [Google Scholar] [CrossRef]
- Schneider, R.; Machens, A.; Lorenz, K.; Dralle, H. Intraoperative nerve monitoring in thyroid surgery-shifting current paradigms. Gland. Surg. 2020, 9, S120–S128. [Google Scholar] [CrossRef] [PubMed]
- Deniwar, A.; Bhatia, P.; Kandil, E. Electrophysiological neuromonitoring of the laryngeal nerves in thyroid and parathyroid surgery: A review. World J. Exp. Med. 2015, 5, 120–123. [Google Scholar] [CrossRef] [PubMed]
- Dralle, H.; Sekulla, C.; Haerting, J.; Timmermann, F.; Neumann, H.J.; Kruse, E.; Grond, S.; Muhlig, H.P.; Richter, C.; Voss, J.; et al. Risk factors of paralysis and functional outcome after recurrent laryngeal nerve monitoring in thyroid surgery. Surgery 2004, 136, 1310–1322. [Google Scholar] [CrossRef] [PubMed]
- Barczyński, M.; Konturek, A.; Cichoń, S. Randomizedclinicaltrialofvisualizationwersusneuromonitoringofrecurrentlaryngeal nerves during thyroidectomy. Br. J. Surg. 2009, 96, 240–246. [Google Scholar] [CrossRef] [PubMed]
- Barczyński, M.; Konturek, A.; Pragacz, K.; Papier, A.; Stopa, M.; Nowak, W. Intraoperative nerve monitoring can reduce prevalence of recurrent laryngeal nerve injury in thyroid reoperations: Results of a ret-rospective cohort study. World J. Surg. 2014, 38, 599–606. [Google Scholar] [CrossRef]
- Pisanu, A.; Porceddu, G.; Podda, M.; Cois, A.; Uccheddu, A. Systematic review with meta-analysis of studies comparing intraoperative neuromonitoring of recurrent laryngeal nerves versus visualization alone during thyroidectomy. J. Surg. Res. 2014, 188, 152–161. [Google Scholar]
- Henry, B.M.; Graves, M.J.; Vikse, J.; Sanna, B.; Pękala, P.A.; Walocha, J.A.; Barczyński, M.; Tomaszewski, K.A. The current state of inter- mittent intraoperative neural monitoring for prevention recurrent laryngeal nerve injury during thyroidectomy: A PRISMA-compliant systematic review of overlapping meta analyses. Langenbeck’s Arch. Surg. 2017, 402, 663–673. [Google Scholar]
- Yang, S.; Zhou, L.; Lu, Z.; Ma, B.; Ji, Q.; Wang, Y. Systematic review with meta-analysis of intraoperative neuromonitoring during thyroidectomy. Int. J. Surg. 2017, 39, 104–113. [Google Scholar] [CrossRef]
- Wong, K.P.; Mak, K.L.; Wong, C.K.; Lang, B.H. Systematic review and meta-analysis on intra-operative neuro-monitoring in high-risk thyroidectomy. Int. J. Surg. 2017, 38, 21–30. [Google Scholar] [CrossRef]
- Wojtczak, B.; Marciniak, D.; Kaliszewski, K.; Sutkowski, K.; Głód, M.; Rudnicki, J.; Bolanowski, M.; Barczyński, M. Proving the Superiority of Intraoperative Recurrent Laryngeal Nerve Monitoring over Visualization Alone during Thyroidectomy. Biomedicines 2023, 11, 880. [Google Scholar] [CrossRef]
- Schneider, R.; Machens, A.; Sekulla, C.; Lorenz, K.; Elwerr, M.; Dralle, H. Superiority of continuous over intermittent intraoperative nerve monitoring in preventing vocal cord palsy. Br. J. Surg. 2021, 108, 566–573. [Google Scholar] [CrossRef]
- Masuoka, H.; Miyauchi, A.; Higashiyama, T.; Yabuta, T.; Fukushima, M.; Ito, Y.; Kihara, M.; Kobayashi, K.; Yamada, O.; Nakayama, A.; et al. Prospective randomized study on injury of the external branch of the superior laryngeal nerve during thyroidectomy comparing intraoperative nerve monitoring and a conventional technique. Head Neck. 2015, 10, 1456–1460. [Google Scholar] [CrossRef] [PubMed]
- Aygün, N.; Uludağ, M.; İşgör, A. Contribution of intraoperative neuromonitoring to the identification of the external branch of superior laryngeal nerve. Turk. J. Surg. 2017, 33, 169–174. [Google Scholar] [CrossRef]
- Yuan, Q.; Zheng, L.; Hou, J.; Zhou, R.; Xu, G.; Li, C.; Wu, G. Visual identification and neuromonitoring vs. no sighting the external branch of the superior laryngeal nerve in thyroid surgery: A randomized clinical trial. Updates Surg. 2022, 74, 727–734. [Google Scholar] [CrossRef]
- Al-Qurayshi, Z.; Kandil, E.; Randolph, G.W. Cost-effectiveness of intraoperative nerve monitoring in avoidance of bilateral recurrent laryngeal nerve injury in patients undergoing total thyroidectomy. Br. J. Surg. 2017, 104, 1523–1531. [Google Scholar] [PubMed]
- Wojtczak, B.; Sutkowski, K.; Kaliszewski, K.; Głód, M.; Barczyński, M. Experience with intraoperative neuromonitoring of the recurrent laryngeal nerve improves surgical skills and outcomes of non-monitored thyroidectomy. Langenbeck’s Arch. Surg. 2017, 402, 709–717. [Google Scholar] [CrossRef] [PubMed]
- Lorenz, K.; Raffaeli, M.; Barczyński, M.; Lorente-Poch, L.; Sancho, J. Volume, outcomes and quality standards in thyroid surgery: An evidence-based analysis—European Society of Endocrine Surgeons (ESES) positional statement. Langenbeck’s Arch. Surg. 2020, 405, 401–425. [Google Scholar]
- Wu, C.W.; Randolph, G.W.; Barczyński, M.; Schneider, R.; Chiang, F.Y.; Huang, T.Y.; Karcioglu, A.S.; Konturek, A.; Frattini, F.; Weber, F.; et al. Training Courses in Laryngeal Nerve Monitoring in Thyroid and Parathyroid Surgery- The INMSG Consensus Statement. Front. Endocrinol. 2021, 12, 705346. [Google Scholar] [CrossRef]
- Zhang, D.; Pino, A.; Caruso, E.; Dionigi, G.; Sun, H. Neural monitoring in thyroid surgery is here to stay. Gland. Surg. 2020, 9, S43–S46. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.W.; Huang, T.Y.; Randolph, G.W.; Barczyński, M.; Schneider, R.; Chiang, F.Y.; Silver Karcioglu, A.; Wojtczak, B.; Frattini, F.; Gualniera, P.; et al. Informed Consent for Intraoperative Neural Monitoring in Thyroid and Parathyroid Surgery—Consensus Statement of the International Neural Monitoring Study Group. Front. Endocrinol. 2021, 12, 795281. [Google Scholar] [CrossRef]
- Musholt, T.J.; Clerici, T.; Dralle, H.; Frilling, A.; Goretzki, P.E.; Hermann, M.M.; Kußmann, J.; Lorenz, K.; Nies, C.; Schabram, J.; et al. German Association of Endocrine Surgeons practice guidelines for the surgical treatment of benign thyroid disease. Langenbeck’s Arch. Surg. 2011, 396, 639–649. [Google Scholar] [CrossRef]
- Bryan, R.; Haugen, E.; Alexander, E.; Bible, K.C.; Doherty, G.M.; Mandel, S.J.; Nikiforov, E.Y.; Pacini, F.; Randolph, G.W.; Sawka, J.; et al. American Thyroid Association Management Guidelines for Adult Patients with Thyroid Nodules and Differentiated Thyroid Cancer The American Thyroid Association Guidelines Task Force on Thyroid Nodules and Differentiated Thyroid Cancer. Thyroid 2016, 26, 1–133. [Google Scholar] [CrossRef]
- Patel, K.N.; Yip, L.; Lubitz, C.C.; Grubbs, E.G.; Miller, B.S.; Shen, W.; Angelos, P.; Chen, H.; Doherty, G.M.; Fahey, T.J.I.; et al. The American Association of Endocrine Surgeons Guidelines for the Definitive Surgical Management of Thyroid Disease in Adults. Ann Surg. 2020, 3, 21–93. [Google Scholar] [CrossRef]
- Stack, B.C., Jr.; Tolley, N.S.; Bartel, T.B.; Bilezikian, J.P.; Bodenner, D.; Camacho, P.; Cox, J.P.D.T.; Dralle, H.; Jackson, J.E.; Morris, J.C.; et al. AHNS Series: Do you know your guidelines? Optimizing outcomes in reoperative parathyroid surgery: Definitive multidisciplinary joint consensus guidelines of the American Head and Neck Society and the British Association of Endocrine and Thyroid Surgeons. Head Neck 2018, 40, 1617–1629. [Google Scholar] [CrossRef]
- Del Rio, P.; Polistena, A.; Chiofalo, M.G.; De Pasquale, L.; Dionigi, G.; Docimo, G.; Graceffa, G.; Iacobone, M.; Medas, F.; Pezzolla, A.; et al. Management of surgical diseases of thyroid gland indications of the United Italian Society of Endocrine Surgery (SIUEC). Updates Surg. 2023, 75, 1393–1417. [Google Scholar] [CrossRef] [PubMed]
- Krajewska, J.; Chmielik, E.; Dedecjus, M.; Jarząb, B.; Hubalewska-Dydejczyk, A.; Karbownik-Lewińska, M.; Kos-Kudła, B.; Lewiński, A.; Ruchała, M. Diagnosis and treatment of thyroid cancer in adult patients—Recommendations of Polish Scientific Societies and the National Oncological Strategy. Update of the 2022 Update [Diagnostyka i leczenie raka tarczycy u chorych dorosłych—Rekomendacje Polskich Towarzystw Naukowych oraz Narodowej Strategii Onkologicznej. Aktualizacja na rok 2022—Uzupełnienie]. Endokrynol. Pol. 2022, 73, 799–802. [Google Scholar] [CrossRef]
- Sun, H.; Tian, W.; Chinese Thyroid Association, College of Surgeons, Chinese Medical Doctor Association; Chinese Research Hospital Association Thyroid Disease Committee. Chinese guidelines on intraoperative neuromonitoring in thyroid and parathyroid surgery (2023 edition). Gland. Surg. 2023, 12, 1031–1049. [Google Scholar] [CrossRef]
- Available online: www.eurocrine.eu (accessed on 1 January 2022).
- Caruso, E.; Pino, A.; Dionigi, G.; Pontin, A. Safety of intraoperative neuromonitoring. Ann. Thyroid 2019, 4, 6. [Google Scholar] [CrossRef]
- Friedrich, C.; Ulmer, C.; Rieber, F.; Kern, E.; Kohler, A.; Schymik, K.; Thon, K.; Lamadé, W. Safety analysis of vagal nerve stimulation for continuous nerve monitoring during thyroid surgery. Laryngoscope 2012, 122, 1979–1987. [Google Scholar] [CrossRef] [PubMed]
- Schneider, R.; Randolph, G.W.; Barczynski, M.; Dionigi, G.; Wu, C.W.; Chiang, F.Y.; Machens, A.; Kamani, D.; Dralle, H. Continuous intraoperative neural monitoring of the recurrent nerves in thyroid surgery: A quantum leap in technology. Gland. Surg. 2016, 5, 607–616. [Google Scholar] [CrossRef]
- Schneider, R.; Machens, A.; Bucher, M.; Raspé, C.; Heinroth, K.; Dralle, H. Continuous intraoperative monitoring of vagus and recurrent laryngeal nerve function in patients with advanced atrioventricular block. Langenbeck’s Arch. Surg. 2016, 401, 551–556. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wojtczak, B.; Sutkowska-Stępień, K.; Głód, M.; Kaliszewski, K.; Sutkowski, K.; Barczyński, M. Current Knowledge on the Use of Neuromonitoring in Thyroid Surgery. Biomedicines 2024, 12, 675. https://doi.org/10.3390/biomedicines12030675
Wojtczak B, Sutkowska-Stępień K, Głód M, Kaliszewski K, Sutkowski K, Barczyński M. Current Knowledge on the Use of Neuromonitoring in Thyroid Surgery. Biomedicines. 2024; 12(3):675. https://doi.org/10.3390/biomedicines12030675
Chicago/Turabian StyleWojtczak, Beata, Karolina Sutkowska-Stępień, Mateusz Głód, Krzysztof Kaliszewski, Krzysztof Sutkowski, and Marcin Barczyński. 2024. "Current Knowledge on the Use of Neuromonitoring in Thyroid Surgery" Biomedicines 12, no. 3: 675. https://doi.org/10.3390/biomedicines12030675
APA StyleWojtczak, B., Sutkowska-Stępień, K., Głód, M., Kaliszewski, K., Sutkowski, K., & Barczyński, M. (2024). Current Knowledge on the Use of Neuromonitoring in Thyroid Surgery. Biomedicines, 12(3), 675. https://doi.org/10.3390/biomedicines12030675






