The Association between Delayed Gut Microbiota Maturity in Pre-Term Infants and the Feeding Intolerance—A Pilot Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Ethical Approval
2.2. Subjects
2.3. Fecal Collection
2.4. Feeding Tolerance Assessment
2.5. Gut Microbiota Analysis
2.6. Data Analysis
3. Results
3.1. Demographic Characteristics
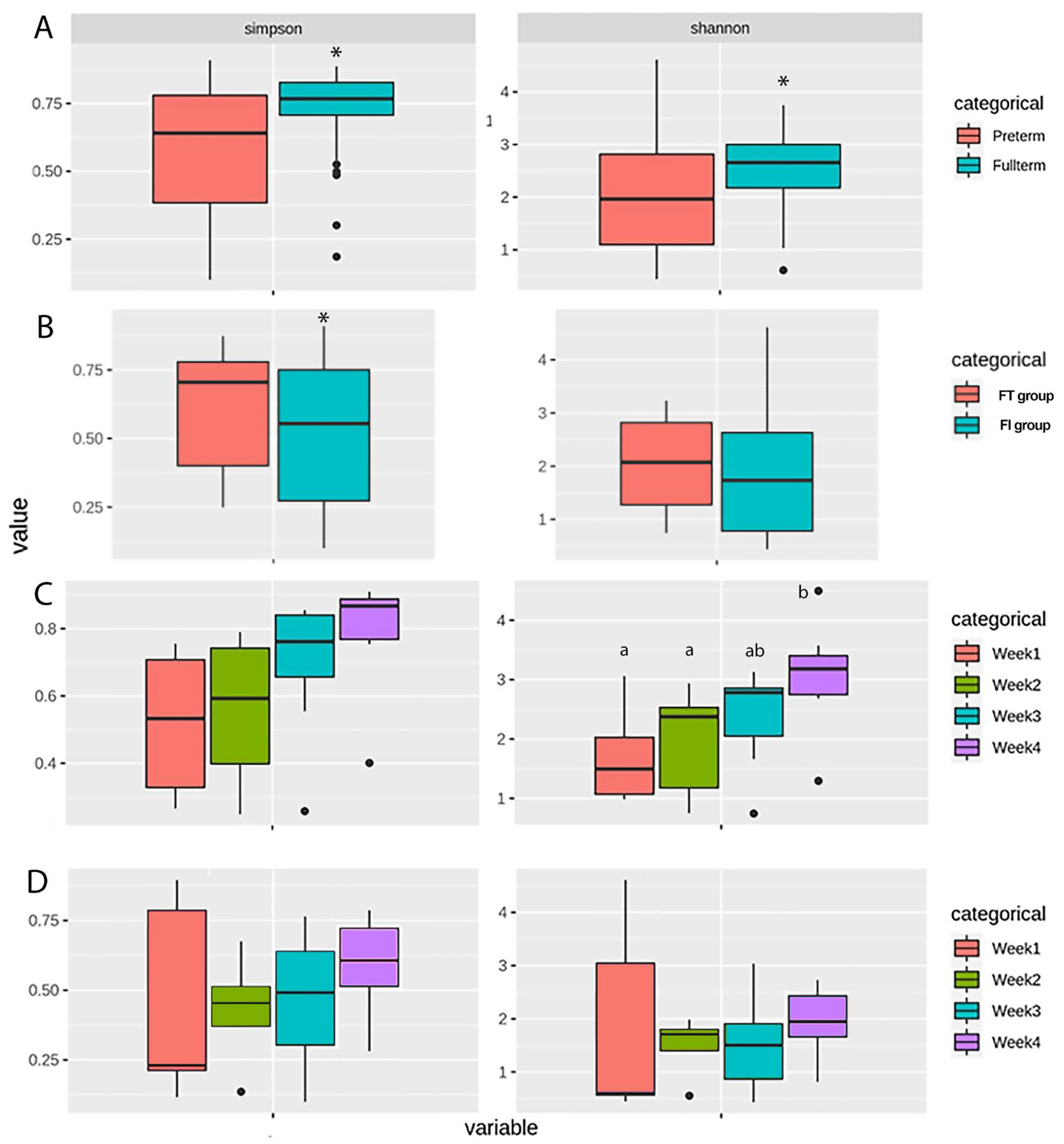
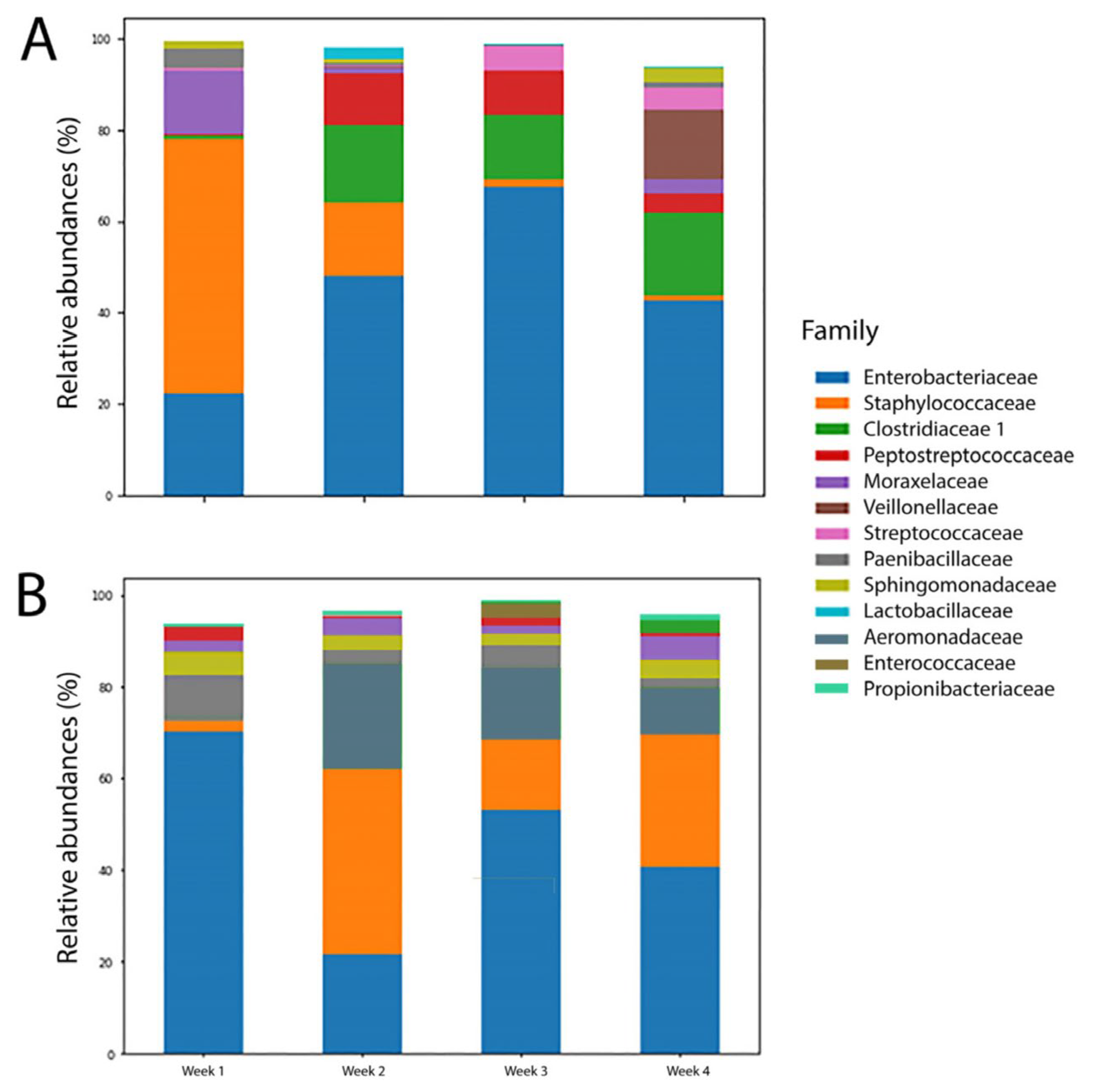
3.2. Gut Microbiome Profiles of the Full-Term and Pre-term Groups
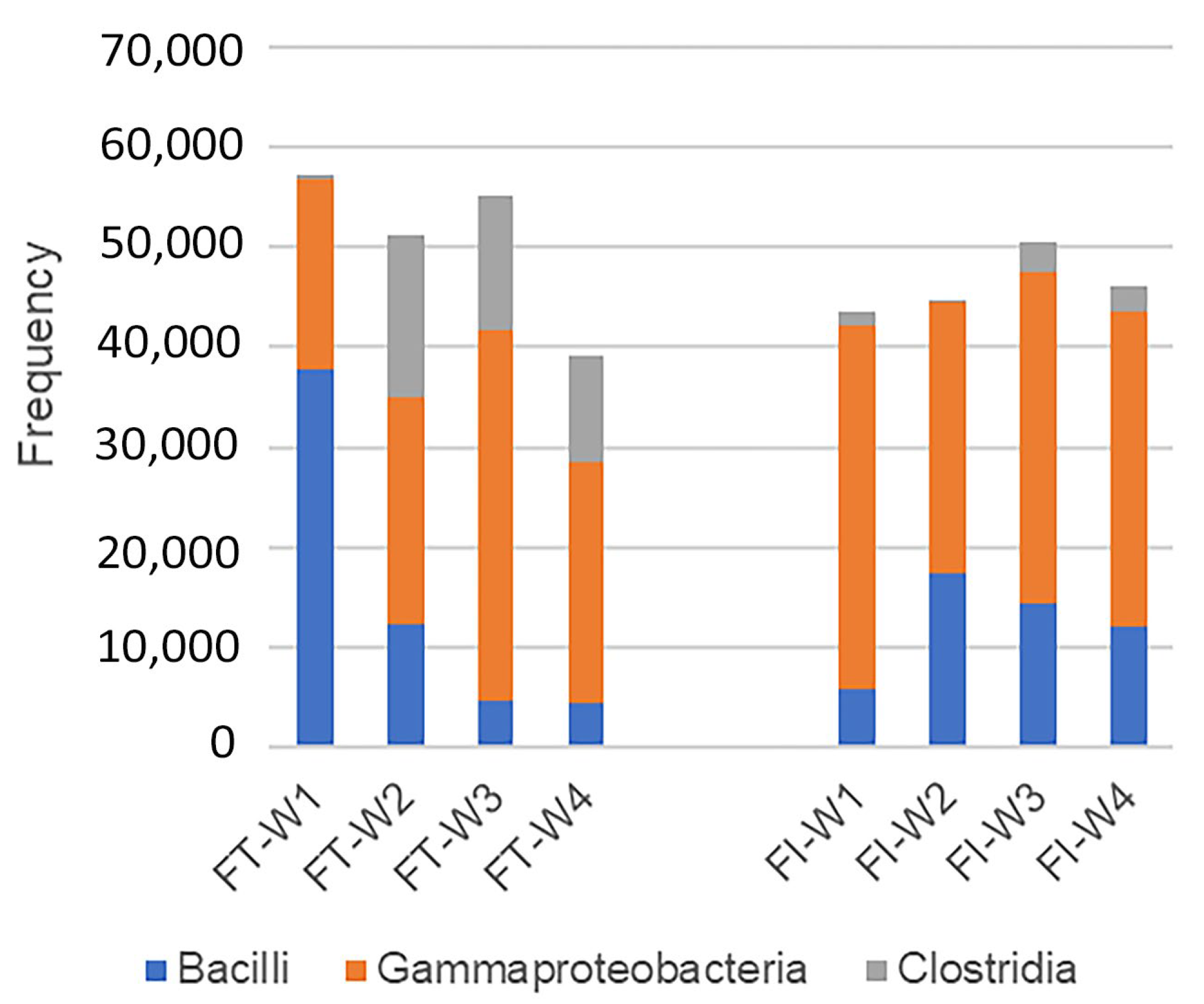
3.3. Overall Microbiome Profiles of the FT and FI Groups and the Changes over Four Weeks
3.4. Biomarkers in Full-Term and Pre-term Infants: FT and FI Groups
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Harmsen, H.J.; Wildeboer–Veloo, A.C.; Raangs, G.C.; Wagendorp, A.A.; Klijn, N.; Bindels, J.G.; Welling, G.W. Analysis of intestinal flora development in breast-fed and formula-fed infants by using molecular identification and detection methods. J. Pediatr. Gastroenterol. Nutr. 2000, 30, 61–67. [Google Scholar] [CrossRef] [PubMed]
- Matamoros, S.; Gras-Leguen, C.; Le Vacon, F.; Potel, G.; de La Cochetiere, M.-F. Development of intestinal microbiota in infants and its impact on health. Trends Microbiol. 2013, 21, 167–173. [Google Scholar] [CrossRef] [PubMed]
- Sakata, H.; Yoshioka, H.; Fujita, K. Development of the intestinal flora in very low birth weight infants compared to normal full-term newborns. Eur. J. Pediatr. 1985, 144, 186–190. [Google Scholar] [CrossRef] [PubMed]
- Rinninella, E.; Raoul, P.; Cintoni, M.; Franceschi, F.; Miggiano, G.A.D.; Gasbarrini, A.; Mele, M.C. What is the healthy gut microbiota composition? A changing ecosystem across age, environment, diet, and diseases. Microorganisms 2019, 7, 14. [Google Scholar] [CrossRef] [PubMed]
- Dallas, D.C.; Underwood, M.A.; Zivkovic, A.M.; German, J.B. Digestion of protein in premature and term infants. J. Nutr. Disord. Ther. 2012, 2, 112. [Google Scholar] [CrossRef]
- Demers-Mathieu, V.; Qu, Y.; Underwood, M.A.; Dallas, D.C. The preterm infant stomach actively degrades milk proteins with increasing breakdown across digestion time. Acta Paediatr. 2018, 107, 967–974. [Google Scholar] [CrossRef] [PubMed]
- Hamosh, M.; Scanlon, J.W.; Ganot, D.; Likel, M.; Scanlon, K.B.; Hamosh, P. Fat digestion in the newborn: Characterization of lipase in gastric aspirates of premature and term infants. J. Clin. Investig. 1981, 67, 838–846. [Google Scholar] [CrossRef] [PubMed]
- Heintz-Buschart, A.; Wilmes, P. Human Gut Microbiome: Function Matters. Trends Microbiol. 2018, 26, 563–574. [Google Scholar] [CrossRef]
- Fanaro, S. Feeding intolerance in the preterm infant. Early Hum. Dev. 2013, 89 (Suppl. S2), S13–S20. [Google Scholar] [CrossRef]
- Ortigoza, E.B. Feeding intolerance. Early Hum. Dev. 2022, 171, 105601. [Google Scholar] [CrossRef]
- Bolger, A.M.; Lohse, M.; Usadel, B. Trimmomatic: A flexible trimmer for Illumina sequence data. Bioinformatics 2014, 30, 2114–2120. [Google Scholar] [CrossRef] [PubMed]
- Martin, M. Cutadapt removes adapter sequences from high-throughput sequencing reads. EMBnet. J. 2011, 17, 10–12. [Google Scholar] [CrossRef]
- Callahan, B.J.; McMurdie, P.J.; Rosen, M.J.; Han, A.W.; Johnson, A.J.A.; Holmes, S.P. DADA2: High-resolution sample inference from Illumina amplicon data. Nat. Methods 2016, 13, 581–583. [Google Scholar] [CrossRef] [PubMed]
- Ludwig, W.; Strunk, O.; Westram, R.; Richter, L.; Meier, H.; Yadhukumar, A.; Buchner, A.; Lai, T.; Steppi, S.; Jobb, G. ARB: A software environment for sequence data. Nucleic Acids Res. 2004, 32, 1363–1371. [Google Scholar] [CrossRef] [PubMed]
- Caporaso, J.G.; Kuczynski, J.; Stombaugh, J.; Bittinger, K.; Bushman, F.D.; Costello, E.K.; Fierer, N.; Peña, A.G.; Goodrich, J.K.; Gordon, J.I. QIIME allows analysis of high-throughput community sequencing data. Nat. Methods 2010, 7, 335–336. [Google Scholar] [CrossRef]
- Ondov, B.D.; Bergman, N.H.; Phillippy, A.M. Interactive metagenomic visualization in a Web browser. BMC Bioinform. 2011, 12, 385. [Google Scholar] [CrossRef]
- Tong, S.T. The use of non-metric multidimensional scaling as an ordination technique in resource survey and evaluation: A case study from southeast Spain. Appl. Geogr. 1992, 12, 243–260. [Google Scholar] [CrossRef]
- Segata, N.; Izard, J.; Waldron, L.; Gevers, D.; Miropolsky, L.; Garrett, W.S.; Huttenhower, C. Metagenomic biomarker discovery and explanation. Genome Biol. 2011, 12, R60. [Google Scholar] [CrossRef]
- Clarke, K.R. Non-parametric multivariate analyses of changes in community structure. Austral. J. Ecol. 1993, 18, 117–143. [Google Scholar] [CrossRef]
- Grier, A.; Qiu, X.; Bandyopadhyay, S.; Holden-Wiltse, J.; Kessler, H.A.; Gill, A.L.; Hamilton, B.; Huyck, H.; Misra, S.; Mariani, T.J. Impact of prematurity and nutrition on the developing gut microbiome and preterm infant growth. Microbiome 2017, 5, 158. [Google Scholar] [CrossRef]
- Henderickx, J.G.; Zwittink, R.D.; Van Lingen, R.A.; Knol, J.; Belzer, C. The preterm gut microbiota: An inconspicuous challenge in nutritional neonatal care. Front. Cell. Infect. Microbiol. 2019, 9, 85. [Google Scholar] [CrossRef]
- Baldassarre, M.E.; Di Mauro, A.; Capozza, M.; Rizzo, V.; Schettini, F.; Panza, R.; Laforgia, N. Dysbiosis and Prematurity: Is There a Role for Probiotics? Nutrients 2019, 11, 1273. [Google Scholar] [CrossRef] [PubMed]
- Hong, L.; Huang, Y.; Han, J.; Li, S.; Zhang, L.; Jiang, S.; Zhou, Q.; Cao, X.; Yu, W.; Yang, Y.; et al. Dynamics and Crosstalk between Gut Microbiota, Metabolome, and Fecal Calprotectin in Very Preterm Infants: Insights into Feeding Intolerance. Nutrients 2023, 15, 4849. [Google Scholar] [CrossRef] [PubMed]
- La Rosa, P.S.; Warner, B.B.; Zhou, Y.; Weinstock, G.M.; Sodergren, E.; Hall-Moore, C.M.; Stevens, H.J.; Bennett, W.E., Jr.; Shaikh, N.; Linneman, L.A. Patterned progression of bacterial populations in the premature infant gut. Proc. Natl. Acad. Sci. USA 2014, 111, 12522–12527. [Google Scholar] [CrossRef] [PubMed]
- Xiang, Q.; Yan, X.; Shi, W.; Li, H.; Zhou, K. Early gut microbiota intervention in premature infants: Application perspectives. J. Adv. Res. 2022, 51, 59–72. [Google Scholar] [CrossRef] [PubMed]
- Shaw, A.G.; Sim, K.; Randell, P.; Cox, M.J.; McClure, Z.E.; Li, M.S.; Donaldson, H.; Langford, P.R.; Cookson, W.O.C.M.; Moffatt, M.F.; et al. Late-onset bloodstream infection and perturbed maturation of the gastrointestinal microbiota in premature infants. PLoS ONE 2015, 10, e0132923. [Google Scholar] [CrossRef] [PubMed]
- Korpela, K.; Blakstad, E.W.; Moltu, S.J.; Strømmenm, K.; Nakstad, B.; Rønnestad, A.E.; Brækkem, K.; Iversen, P.O.; Drevon, C.A.; De Vos, W. Intestinal microbiota development and gestational age in preterm neonates. Sci. Rep. 2018, 8, 2453. [Google Scholar] [CrossRef]
- Benevides, G.N.; Hein, N.; Lo, D.S.; Ferronato, A.E.; Ragazzi, S.L.B.; Yoshioka, C.R.M.; Hirose, M.; Cardoso, D.M.; Dos Santos, S.R.; Gilio, A.E. Otomastoiditis caused by Sphingomonas paucimobilis: Case report and literature review. Autops. Case Rep. 2014, 4, 13. [Google Scholar] [CrossRef][Green Version]
- Stewart, C.J.; Nelson, A.; Scribbins, D.; Marrs, E.C.L.; Lanyon, C.; Perry, J.D.; Embleton, N.D.; Cummings, S.P.; Berrington, J.E. Bacterial and fungal viability in the preterm gut: NEC and sepsis. Arch. Dis. Child. Fetal Neonatal Ed. 2013, 98, F298–F303. [Google Scholar] [CrossRef]
- Lindberg, T.P.; Caimano, M.J.; Hagadorn, J.I.; Bennett, E.M.; Maas, K.; Brownell, E.A.; Matson, A.P. Preterm infant gut microbial patterns related to the development of necrotizing enterocolitis. J. Matern. Fetal Neonatal Med. 2020, 33, 349–358. [Google Scholar] [CrossRef]
- Lee, J.K.; Tan, L.T.H.; Ramadas, A.; Ab Mutalib, N.S.; Lee, L.H. Exploring the Role of Gut Bacteria in Health and Disease in Preterm Neonates. Int. J. Environ. Res. Public. Health 2020, 17, 6963. [Google Scholar] [CrossRef]
- Patel, R.M.; Ferguson, J.; McElroy, S.J.; Khashu, M.; Caplan, M.S. Defining necrotizing enterocolitis: Current difficulties and future opportunities. Pediatr. Res. 2020, 88, 10–15. [Google Scholar] [CrossRef]
- Battersby, C.; Santhalingam, T.; Costeloe, K.; Modi, N. Incidence of neonatal necrotising enterocolitis in high-income countries: A systematic review. Arch. Dis. Child. Fetal Neonatal Ed. 2018, 103, F182–F189. [Google Scholar] [CrossRef]
- Brehin, C.; Dubois, D.; Dicky, O.; Breinig, S.; Oswald, E.; Serino, M. Evolution of Gut Microbiome and Metabolome in Suspected Necrotizing Enterocolitis: A Case-Control Study. J. Clin. Med. 2020, 9, 2278. [Google Scholar] [CrossRef]
- Pammi, M.; Cope, J.; Tarr, P.I.; Warner, B.B.; Morrow, A.L.; Mai, V.; Gregory, K.E.; Kroll, J.S.; McMurtry, V.; Ferris, M.J.; et al. Intestinal dysbiosis in preterm infants preceding necrotizing enterocolitis: A systematic review and meta-analysis. Microbiome 2017, 5, 31–45. [Google Scholar] [CrossRef] [PubMed]
- El Manouni el Hassani, S.; Niemarkt, H.J.; Berkhout, D.J.C.; Peeters, C.F.W.; Hulzebos, C.V.; van Kaam, A.H.; Kramer, B.W.; van Lingen, R.A.; Jenken, F.; de Boode, W.P.; et al. Profound pathogen-specific alterations in intestinal microbiota composition precede late-onset sepsis in preterm infants: A longitudinal, multicenter, case-control study. Clin. Infect. Dis. 2021, 73, e224–e232. [Google Scholar] [CrossRef] [PubMed]
- Hiltunen, H.; Hanani, H.; Luoto, R.; Turjeman, S.; Ziv, O.; Isolauri, E.; Salminen, S.; Koren, O.; Rautava, S. Preterm infant meconium microbiota transplant induces growth failure, inflammatory activation, and metabolic disturbances in germ-free mice. Cell Rep. Med. 2021, 2, 100447. [Google Scholar] [CrossRef] [PubMed]
- Rautava, S. Feeding the preterm infant gut microbiota. Cell Host Microbe 2022, 30, 1199–1200. [Google Scholar] [CrossRef] [PubMed]
- Yao, Y.; Cai, X.; Ye, Y.; Wang, F.; Chen, F.; Zheng, C. The Role of Microbiota in Infant Health: From Early Life to Adulthood. Front. Immunol. 2021, 12, 708472. [Google Scholar] [CrossRef] [PubMed]
- Moore, R.E.; Townsend, S.D. Temporal development of the infant gut microbiome. Open Biol. 2019, 9, 190128. [Google Scholar] [CrossRef] [PubMed]




| Grouping | R Statistic Value 1 | p Value |
|---|---|---|
| Pre-term vs. full-term | 0.2955 | 0.0001 |
| FT vs. FI | 0.1733 | 0.0009 |
| FT Group | ||
| Week 1 vs. 2 | 0.3188 | 0.0160 |
| Week 1 vs. 3 | 0.5556 | 0.0013 |
| Week 1 vs. 4 | 0.5251 | 0.0019 |
| Week 2 vs. 3 | −0.1453 | 0.9499 |
| Week 2 vs. 4 | −0.0836 | 0.7585 |
| Week 3 vs. 4 | −0.0651 | 0.7016 |
| FI Group | ||
| Week 1 vs. 2 | 0.3125 | 0.0400 |
| Week 1 vs. 3 | −0.1440 | 0.9933 |
| Week 1 vs. 4 | −0.0720 | 0.7285 |
| Week 2 vs. 3 | −0.0992 | 0.6953 |
| Week 2 vs. 4 | −0.0714 | 0.6192 |
| Week 3 vs. 4 | −0.1448 | 0.8866 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hsu, Y.-C.; Lin, M.-C.; Ardanareswari, K.; Lowisia, W.; Lin, Y.-H.; Chen, Y.-J.; Hsu, C.-K.; Chung, Y.-C. The Association between Delayed Gut Microbiota Maturity in Pre-Term Infants and the Feeding Intolerance—A Pilot Study. Biomedicines 2024, 12, 539. https://doi.org/10.3390/biomedicines12030539
Hsu Y-C, Lin M-C, Ardanareswari K, Lowisia W, Lin Y-H, Chen Y-J, Hsu C-K, Chung Y-C. The Association between Delayed Gut Microbiota Maturity in Pre-Term Infants and the Feeding Intolerance—A Pilot Study. Biomedicines. 2024; 12(3):539. https://doi.org/10.3390/biomedicines12030539
Chicago/Turabian StyleHsu, Ya-Chi, Ming-Chih Lin, Katharina Ardanareswari, Webiana Lowisia, Yi-Hsuan Lin, Yi-Jhen Chen, Cheng-Kuang Hsu, and Yun-Chin Chung. 2024. "The Association between Delayed Gut Microbiota Maturity in Pre-Term Infants and the Feeding Intolerance—A Pilot Study" Biomedicines 12, no. 3: 539. https://doi.org/10.3390/biomedicines12030539
APA StyleHsu, Y.-C., Lin, M.-C., Ardanareswari, K., Lowisia, W., Lin, Y.-H., Chen, Y.-J., Hsu, C.-K., & Chung, Y.-C. (2024). The Association between Delayed Gut Microbiota Maturity in Pre-Term Infants and the Feeding Intolerance—A Pilot Study. Biomedicines, 12(3), 539. https://doi.org/10.3390/biomedicines12030539






