Behavioral and Cognitive Comorbidities in Genetic Rat Models of Absence Epilepsy (Focusing on GAERS and WAG/Rij Rats)
Abstract
1. Introduction
2. Genetic Rat Models
3. Investigation of Behavioral and Cognitive Functions in Rats
4. Cognitive Comorbidities in WAG/Rij Rats
- Cognitive impairment in WAG/Rij rats was secondary to absence epilepsy and to depressive-like behavior;
- Absence epilepsy, depressive-like behavior, and cognitive deficit may arise independently and separately in a lifetime from the same underlying network disease;
- Cognitive impairment in WAG/Rij rats was age-dependent and was linked to the age-dependent increase in spike-wave discharges (i.e., the electroencephalographic sign of absence epilepsy).
| Subjects | Tests | Test Results | References |
|---|---|---|---|
| Male WAG/Rij rats; Untreated and ethosximide-treated (300 mg/kg/day; 17 days); 6 and 12 months old | The Forced Swimming test The novel objects recognition test Social recognition test Morris water maze Passive Avoidance | Anxiety, learning and behavior (cognitive skills)
| Leo et al., 2019 [77] |
| Drug-naive adult male WAG/Rij rats and Wistar control rats, data compilation | The Open Field test The Forced Swimming test Sucrose Preference test for anhedonia (20% solution, two-bottles choice) The Light–Dark choice test The social interaction test in the open field The elevated plus-maze. | Anxiety and behavior
| Sarkisova and van Luijtelaar, 2011 [76] Sarkisova et al., 2003 [97] |
| Drug-naive male WAG/Rij rats and Brown Norway control rats; 13 months old | Test for spatial memory in 16-holes field (holeboard) | Spatial memory (working memory and reference memory
| van der Staay, 1999 [101] |
| Drug-naïve WAG/Rij rats, outbred and Wistar rats; adult and 2 months old | The Elevated Plus Maze | Anxiety and behavior
| Kliueva et al., 1999 [99] |
| Drug-naive female WAG/Rij rats and Wistar control rats; approx. 4 months old | The Open Field test of mothers and their pups on the postnatal days 4–6 | Maternal behavior
| Dobryakova et al., 2008 [102] |
| Drug-naive male WAG/Rij rats and Wistar control rats; 2 months old | The Open Field test The Forced Swimming test The Light–dark choice test Two-days passive avoidance learning test Two-days Active Avoidance learning test | Behavior and learning on the preclinical stage.
| Fedosova et al., 2015 [98] |
| Drug-naive male WAG/Rij rats with epileptic and non-epileptic phenotypes; 6 months old | The Active Avoidance test | The Active Avoidance test on the clinical stage (fear-motivated associative learning).
| Sitnikova & Smirnov, 2020 [83] |
| Drug-naive male and female WAG/Rij rats and control non-epileptic NEW rat substrain; 8.53 ± 1.15 months old | The Active Avoidance test | The Active Avoidance test on the clinical stage (fear-motivated associative learning). Both strains were prone to absence epilepsy, with WAG/Rij rats exhibiting seizures and a minor NEW substrain being non-epileptic.
| Alexandrov et al., 2023 [82] |
5. Behavioral and Cognitive Comorbidities in the GAERS
- Reduced consumption of 20% sucrose solution;
- Spending less time in the open arms of the Elevated Plus Maze;
- Reduced exploratory activity in the Open Field test;
- Spending less time in the inner area of the Open Field test.
- All three strains showed similar levels of locomotor activity as measured in their home cages during the lights-on period;
- The NECs and the GAERS were slightly less active in their home cages than Wistar rats during the light-off period;
- In the beam-walking test, the GAERS and the NECs showed good sensorimotor abilities. Among the three strains, Wistar rats showed the poorest sensorimotor abilities, likely because the body weight in Wistar rats exceeded that in the GAERS and the NECs;
- The GAERS showed a higher anxiety than NECs in the Open Field test (lower activity scores in both central and peripheral areas, and a lower number of rearings) and in the Plus Maze test (a lower number of entries in open arms). However, the results of the GAERS did not differ from those of the Wistar rats;
- When exposed to higher novelty in the Open Field, the GAERS showed a reduced exploration, as compared to the NECs and Wistar rats.
- During prepuberty, both sexes spent less time in open arms and had fewer total open and closed arm entries in the Elevated Plus Maze;
- During prepuberty and young adulthood, both sexes travelled less distance in both the inner and outer areas of the Open Field;
- During young adulthood, females spent less time in open arms in the Elevated Plus Maze, with no difference between the males;
- During prepuberty and young adulthood, both sexes exhibited higher startle responses;
- During prepuberty and young adulthood, males showed increased freezing relative in the low-intensity fear conditioning;
- Exaggerated cued and contextual Pavlovian fear conditioning and impaired fear extinction;
- An impairment of latent inhibition in a paradigm using Pavlovian fear conditioning.
- Deficits in working, spatial reference, and recognition memory as compared to both NEC and Wistar rats;
- Did not show an exaggerated anxiety-like phenotype, but rather a lower anxiety-like behavior in two out of three anxiety tests;
- Preferentially used egocentric strategies to perform spatial memory tasks.
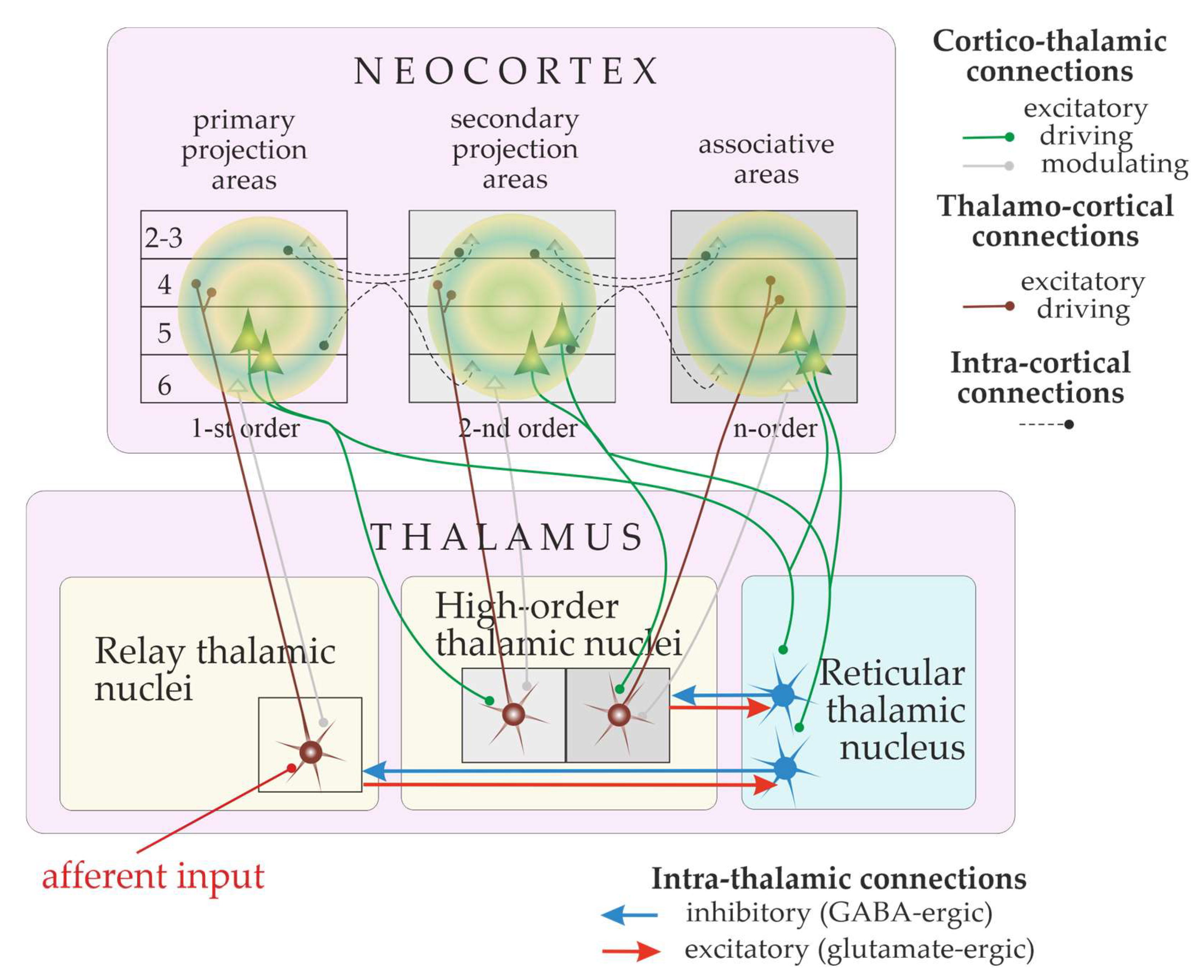
6. The Thalamocortical Network and “the Cognitive Thalamus”
7. Some Translational Issues
8. Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| EEG | electroencephalogram |
| GAERS | the Genetic Absence Epilepsy Rats from Strasbourg, genetic model of absence epilepsy |
| NEW | non-epileptic WAG/Rij |
| NEC | non-epileptic control (for GAERS) |
| SWDs | Spike-wave discharges, EEG hallmarks of absence epilepsy |
| WAG/Rij | Wistar Albino Glaxo Rats from Rijswijk, genetic model of absence epilepsy |
References
- Fisher, R.S.; van Emde Boas, W.; Blume, W.; Elger, C.; Genton, P.; Lee, P.; Engel, J. Response: Definitions Proposed by the International League Against Epilepsy (ILAE) and the International Bureau for Epilepsy (IBE). Epilepsia 2005, 46, 1701–1702. [Google Scholar] [CrossRef]
- Fisher, R.S.; Cross, J.H.; D’Souza, C.; French, J.A.; Haut, S.R.; Higurashi, N.; Hirsch, E.; Jansen, F.E.; Lagae, L.; Moshé, S.L.; et al. Instruction Manual for the ILAE 2017 Operational Classification of Seizure Types. Epilepsia 2017, 58, 531–542. [Google Scholar] [CrossRef] [PubMed]
- Fisher, R.S.; Cross, J.H.; French, J.A.; Higurashi, N.; Hirsch, E.; Jansen, F.E.; Lagae, L.; Moshé, S.L.; Peltola, J.; Roulet Perez, E.; et al. Operational Classification of Seizure Types by the International League Against Epilepsy: Position Paper of the ILAE Commission for Classification and Terminology. Epilepsia 2017, 58, 522–530. [Google Scholar] [CrossRef] [PubMed]
- Hirsch, E.; French, J.; Scheffer, I.E.; Bogacz, A.; Alsaadi, T.; Sperling, M.R.; Abdulla, F.; Zuberi, S.M.; Trinka, E.; Specchio, N.; et al. ILAE Definition of the Idiopathic Generalized Epilepsy Syndromes: Position Statement by the ILAE Task Force on Nosology and Definitions. Epilepsia 2022, 63, 1475–1499. [Google Scholar] [CrossRef] [PubMed]
- Matricardi, S.; Verrotti, A.; Chiarelli, F.; Cerminara, C.; Curatolo, P. Current Advances in Childhood Absence Epilepsy. Pediatr. Neurol. 2014, 50, 205–212. [Google Scholar] [CrossRef] [PubMed]
- Verrotti, A.; Matricardi, S.; Rinaldi, V.E.; Prezioso, G.; Coppola, G. Neuropsychological Impairment in Childhood Absence Epilepsy: Review of the Literature. J. Neurol. Sci. 2015, 359, 59–66. [Google Scholar] [CrossRef] [PubMed]
- Panayiotopoulos, C.P.; Chroni, E.; Daskalopoulos, C.; Baker, A.; Rowlinson, S.; Walsh, P. Typical Absence Seizures in Adults: Clinical, EEG, Video-EEG Findings and Diagnostic/Syndromic Considerations. J. Neurol. Neurosurg. Psychiatry 1992, 55, 1002–1008. [Google Scholar] [CrossRef] [PubMed]
- Guo, J.N.; Kim, R.; Chen, Y.; Negishi, M.; Jhun, S.; Weiss, S.; Ryu, J.H.; Bai, X.; Xiao, W.; Feeney, E.; et al. Impaired Consciousness in Patients with Absence Seizures Investigated by Functional MRI, EEG, and Behavioural Measures: A Cross-Sectional Study. Lancet Neurol. 2016, 15, 1336–1345. [Google Scholar] [CrossRef]
- De Curtis, M.; Avanzini, G. Thalamic Regulation of Epileptic Spike and Wave Discharges. Funct. Neurol. 1994, 9, 307–326. [Google Scholar] [CrossRef]
- Steriade, M. Neuronal Substrates of Sleep and Epilepsy; Cambridge University Press: Cambridge, UK, 2003; ISBN 978-0521817073. [Google Scholar]
- Avoli, M. A Brief History on the Oscillating Roles of Thalamus and Cortex in Absence Seizures. Epilepsia 2012, 53, 779–789. [Google Scholar] [CrossRef]
- Van Luijtelaar, G.; Sitnikova, E. Global and Focal Aspects of Absence Epilepsy: The Contribution of Genetic Models. Neurosci. Biobehav. Rev. 2006, 30, 983–1003. [Google Scholar] [CrossRef]
- Sitnikova, E. Thalamo-Cortical Mechanisms of Sleep Spindles and Spike–Wave Discharges in Rat Model of Absence Epilepsy (a Review). Epilepsy Res. 2010, 89, 17–26. [Google Scholar] [CrossRef] [PubMed]
- Huguenard, J. Current Controversy: Spikes, Bursts, and Synchrony in Generalized Absence Epilepsy: Unresolved Questions Regarding Thalamocortical Synchrony in Absence Epilepsy. Epilepsy Curr. 2019, 19, 105–111. [Google Scholar] [CrossRef]
- Kim, J.B.; Suh, S.; Seo, W.-K.; Oh, K.; Koh, S.-B.; Kim, J.H. Altered Thalamocortical Functional Connectivity in Idiopathic Generalized Epilepsy. Epilepsia 2014, 55, 592–600. [Google Scholar] [CrossRef]
- Sherman, S.M.; Guillery, R.W. Exploring the Thalamus and Its Role in Cortical Function, 2nd ed.; MIT Press: Cambridge, MA, USA, 2006; ISBN 0-262-19532-1. [Google Scholar]
- McCormick, D.A.; Bal, T. Sleep and Arousal: Thalamocortical Mechanisms. Annu. Rev. Neurosci. 1997, 20, 185–215. [Google Scholar] [CrossRef]
- Buzsáki, G. Rhythms of the Brain; Oxford University Press: Oxford, UK, 2009; ISBN 9780199863716. [Google Scholar]
- Wolff, M.; Vann, S.D. The Cognitive Thalamus as a Gateway to Mental Representations. J. Neurosci. 2019, 39, 3–14. [Google Scholar] [CrossRef] [PubMed]
- Constantinople, C.M.; Bruno, R.M. Deep Cortical Layers Are Activated Directly by Thalamus. Science 2013, 340, 1591–1594. [Google Scholar] [CrossRef] [PubMed]
- Halász, P. Sleep and Epilepsy. In Handbook of Clinical Neurology; Stefan, H., Theodore, W.H., Eds.; Elsevier: Amsterdam, The Netherlands, 2012; Volume 107, pp. 305–322. ISBN 0072-9752. [Google Scholar]
- Buzsáki, G. The Thalamic Clock: Emergent Network Properties. Neuroscience 1991, 41, 351–364. [Google Scholar] [CrossRef]
- McCormick, D.A.; Bal, T. Sensory Gating Mechanisms of the Thalamus. Curr. Opin. Neurobiol. 1994, 4, 550–556. [Google Scholar] [CrossRef]
- Steriade, M. Grouping of Brain Rhythms in Corticothalamic Systems. Neuroscience 2006, 137, 1087–1106. [Google Scholar] [CrossRef]
- Polack, P.O.; Charpier, S. Intracellular Activity of Cortical and Thalamic Neurones during High-Voltage Rhythmic Spike Discharge in Long-Evans Rats. J. Physiol. 2006, 571, 461–476. [Google Scholar] [CrossRef] [PubMed]
- Halász, P. How Sleep Activates Epileptic Networks? Epilepsy Res. Treat. 2013, 2013, 425697. [Google Scholar] [CrossRef] [PubMed]
- Llinas, R.R.; Ribary, U.; Jeanmonod, D.; Kronberg, E.; Mitra, P.P. Thalamocortical Dysrhythmia: A Neurological and Neuropsychiatric Syndrome Characterized by Magnetoencephalography. Proc. Natl. Acad. Sci. USA 1999, 96, 15222–15227. [Google Scholar] [CrossRef] [PubMed]
- Zobeiri, M.; van Luijtelaar, G.; Budde, T.; Sysoev, I.V. The Brain Network in a Model of Thalamocortical Dysrhythmia. Brain Connect. 2019, 9, 273–284. [Google Scholar] [CrossRef] [PubMed]
- Scheffer, I.E.; Berkovic, S.; Capovilla, G.; Connolly, M.B.; French, J.; Guilhoto, L.; Hirsch, E.; Jain, S.; Mathern, G.W.; Moshé, S.L.; et al. ILAE Classification of the Epilepsies: Position Paper of the ILAE Commission for Classification and Terminology. Epilepsia 2017, 58, 512–521. [Google Scholar] [CrossRef] [PubMed]
- Van Luijtelaar, E.L.J.M.; Coenen, A.M.L. Two Types of Electrocortical Paroxysms in an Inbred Strain of Rats. Neurosci. Lett. 1986, 70, 393–397. [Google Scholar] [CrossRef] [PubMed]
- Marescaux, C.; Vergnes, M. Genetic Absence Epilepsy in Rats from Strasbourg (GAERS). Ital. J. Neurol. Sci. 1995, 16, 113–118. [Google Scholar] [CrossRef]
- Coenen, A.M.L.; Van Luijtelaar, E.L.J.M. Genetic Animal Models for Absence Epilepsy: A Review of the WAG/Rij Strain of Rats. Behav. Genet. 2003, 33, 635–655. [Google Scholar] [CrossRef]
- Bhise, V.V.; Burack, G.D.; Mandelbaum, D.E. Baseline Cognition, Behavior, and Motor Skills in Children with New-Onset, Idiopathic Epilepsy. Dev. Med. Child Neurol. 2010, 52, 22–26. [Google Scholar] [CrossRef]
- Caplan, R.; Siddarth, P.; Stahl, L.; Lanphier, E.; Vona, P.; Gurbani, S.; Koh, S.; Sankar, R.; Shields, W.D. Childhood Absence Epilepsy: Behavioral, Cognitive, and Linguistic Comorbidities. Epilepsia 2008, 49, 1838–1846. [Google Scholar] [CrossRef]
- D’Agati, E.; Cerminara, C.; Casarelli, L.; Pitzianti, M.; Curatolo, P. Attention and Executive Functions Profile in Childhood Absence Epilepsy. Brain Dev. 2012, 34, 812–817. [Google Scholar] [CrossRef] [PubMed]
- Ratcliffe, C.; Wandschneider, B.; Baxendale, S.; Thompson, P.; Koepp, M.J.; Caciagli, L. Cognitive Function in Genetic Generalized Epilepsies: Insights from Neuropsychology and Neuroimaging. Front. Neurol. 2020, 11, 144. [Google Scholar] [CrossRef]
- Blumenfeld, H. Cellular and Network Mechanisms of Spike-Wave Seizures. Epilepsia 2005, 46, 21–33. [Google Scholar] [CrossRef] [PubMed]
- van Luijtelaar, G.; van Oijen, G. Establishing Drug Effects on Electrocorticographic Activity in a Genetic Absence Epilepsy Model: Advances and Pitfalls. Front. Pharmacol. 2020, 11, 395. [Google Scholar] [CrossRef] [PubMed]
- Depaulis, A.; van Luijtelaar, G. Genetic Models of Absence Epilepsy in the Rat. In Models of Seizures and Epilepsy; Elsevier: Amsterdam, The Netherlands, 2006; pp. 233–248. ISBN 9780120885541. [Google Scholar]
- Sitnikova, E.; van Luijtelaar, G. Electroencephalographic Characterization of Spike-Wave Discharges in Cortex and Thalamus in WAG/Rij Rats. Epilepsia 2007, 48, 2296–2311. [Google Scholar] [CrossRef] [PubMed]
- Van Luijtelaar, E.L.J.M.; Van der Werf, S.J.; Vossen, J.M.H.; Coenen, A.M.L. Arousal, Performance and Absence Seizures in Rats. Electroencephalogr. Clin. Neurophysiol. 1991, 79, 430–434. [Google Scholar] [CrossRef] [PubMed]
- Drinkenburg, W.H.I.M.; Coenen, A.M.L.; Vossen, J.M.H.; Van Luijtelaar, E.L.J.M. Spike-Wave Discharges and Sleep-Wake States in Rats with Absence Epilepsy. Epilepsy Res. 1991, 9, 218–224. [Google Scholar] [CrossRef]
- Drinkenburg, W.H.I.M.; Schuurmans, M.L.E.J.; Coenen, A.M.L.; Vossen, J.M.H.; Van Luijtelaar, E.L.J.M. Ictal Stimulus Processing during Spike-Wave Discharges in Genetic Epileptic Rats. Behav. Brain Res. 2003, 143, 141–146. [Google Scholar] [CrossRef]
- Glauser, T.A.; Cnaan, A.; Shinnar, S.; Hirtz, D.G.; Dlugos, D.; Masur, D.; Clark, P.O.; Adamson, P.C. Ethosuximide, Valproic Acid, and Lamotrigine in Childhood Absence Epilepsy: Initial Monotherapy Outcomes at 12 Months. Epilepsia 2013, 54, 141–155. [Google Scholar] [CrossRef]
- Masur, D.; Shinnar, S.; Cnaan, A.; Shinnar, R.C.; Clark, P.; Wang, J.; Weiss, E.F.; Hirtz, D.G.; Glauser, T.A. Pretreatment Cognitive Deficits and Treatment Effects on Attention in Childhood Absence Epilepsy. Neurology 2013, 81, 1572–1580. [Google Scholar] [CrossRef]
- Fonseca Wald, E.L.A.; Hendriksen, J.G.M.; Drenthen, G.S.; Kuijk, S.M.J.V.; Aldenkamp, A.P.; Vles, J.S.H.; Vermeulen, R.J.; Debeij-van Hall, M.H.J.A.; Klinkenberg, S. Towards a Better Understanding of Cognitive Deficits in Absence Epilepsy: A Systematic Review and Meta-Analysis. Neuropsychol. Rev. 2019, 29, 421–449. [Google Scholar] [CrossRef] [PubMed]
- Danober, L.; Deransart, C.; Depaulis, A.; Vergnes, M.; Marescaux, C. Pathophysiological Mechanisms of Genetic Absence Epilepsy in The Rat. Prog. Neurobiol. Vol. 1998, 55, 27–57. [Google Scholar] [CrossRef] [PubMed]
- Van der Staay, F.J.; Arndt, S.S.; Nordquist, R.E. Evaluation of Animal Models of Neurobehavioral Disorders. Behav. Brain Funct. 2009, 5, 11. [Google Scholar] [CrossRef]
- Keeler, J.F.; Robbins, T.W. Translating Cognition from Animals to Humans. Biochem. Pharmacol. 2011, 81, 1356–1366. [Google Scholar] [CrossRef] [PubMed]
- Dawson, T.M.; Ko, H.S.; Dawson, V.L. Genetic Animal Models of Parkinson’s Disease. Neuron 2010, 66, 646–661. [Google Scholar] [CrossRef]
- Caldarone, B.J.; Zachariou, V.; King, S.L. Rodent Models of Treatment-Resistant Depression. Eur. J. Pharmacol. 2015, 753, 51–65. [Google Scholar] [CrossRef]
- Gainetdinov, R.R.; Mohn, A.R.; Caron, M.G. Genetic Animal Models: Focus on Schizophrenia. Trends Neurosci. 2001, 24, 527–533. [Google Scholar] [CrossRef]
- Löscher, W.; Stafstrom, C.E. Epilepsy and Its Neurobehavioral Comorbidities: Insights Gained from Animal Models. Epilepsia 2023, 64, 54–91. [Google Scholar] [CrossRef]
- Nam, H.; Clinton, S.M.; Jackson, N.L.; Kerman, I.A. Learned Helplessness and Social Avoidance in the Wistar-Kyoto Rat. Front. Behav. Neurosci. 2014, 8, 109. [Google Scholar] [CrossRef]
- Paré, W.P. Open Field, Learned Helplessness, Conditioned Defensive Burying, and Forced-Swim Tests in WKY Rats. Physiol. Behav. 1994, 55, 433–439. [Google Scholar] [CrossRef]
- Malkesman, O.; Weller, A. Two Different Putative Genetic Animal Models of Childhood Depression—A Review. Prog Neurobiol. 2009, 88, 153–169. [Google Scholar] [CrossRef]
- Overstreet, D.H. Modeling Depression in Animal Models. Methods Mol. Biol. 2012, 829, 125–144. [Google Scholar] [CrossRef] [PubMed]
- Overstreet, D.H. The Flinders Sensitive Line Rats: A Genetic Animal Model of Depression. Neurosci. Biobehav. Rev. 1993, 17, 51–68. [Google Scholar] [CrossRef] [PubMed]
- Overstreet, D.H.; Friedman, E.; Mathé, A.A.; Yadid, G. The Flinders Sensitive Line Rat: A Selectively Bred Putative Animal Model of Depression. Neurosci. Biobehav. Rev. 2005, 29, 739–759. [Google Scholar] [CrossRef]
- Del Río, C.; Oliveras, I.; Cañete, T.; Blázquez, G.; Tobeña, A.; Fernández-Teruel, A. Genetic Rat Models of Schizophrenia-Relevant Symptoms. World J. Neurosci. 2014, 04, 261–278. [Google Scholar] [CrossRef][Green Version]
- Depaulis, A.; Charpier, S. Pathophysiology of Absence Epilepsy: Insights from Genetic Models. Neurosci. Lett. 2018, 667, 53–65. [Google Scholar] [CrossRef] [PubMed]
- Belzung, C.; Lemoine, M. Criteria of Validity for Animal Models of Psychiatric Disorders: Focus on Anxiety Disorders and Depression. Biol. Mood Anxiety Disord. 2011, 1, 9. [Google Scholar] [CrossRef]
- Willner, P. The Validity of Animal Models of Depression. Psychopharmacology 1984, 83, 1–16. [Google Scholar] [CrossRef]
- Pollak, D.D.; Monje, F.J.; Zuckerman, L.; Denny, C.A.; Drew, M.R.; Kandel, E.R. An Animal Model of a Behavioral Intervention for Depression. Neuron 2008, 60, 149–161. [Google Scholar] [CrossRef]
- van der Staay, F.J. Animal Models of Behavioral Dysfunctions: Basic Concepts and Classifications, and an Evaluation Strategy. Brain Res. Rev. 2006, 52, 131–159. [Google Scholar] [CrossRef]
- Coenen, A.M.L.; Van Luijtelaar, E.L.J.M. The WAG/Rij Rat Model for Absence Epilepsy: Age and Sex Factors. Epilepsy Res. 1987, 1, 297–301. [Google Scholar] [CrossRef] [PubMed]
- Russo, E.; Citraro, R.; Constanti, A.; Leo, A.; Lüttjohann, A.; van Luijtelaar, G.; De Sarro, G. Upholding WAG/Rij Rats as a Model of Absence Epileptogenesis: Hidden Mechanisms and a New Theory on Seizure Development. Neurosci. Biobehav. Rev. 2016, 71, 388–408. [Google Scholar] [CrossRef] [PubMed]
- Leo, A.; De Caro, C.; Nesci, V.; Tallarico, M.; Mangano, G.; Palma, E.; Iannone, M.; De Sarro, G.; Citraro, R.; Russo, E. WAG/Rij Rat Model: A Resource for the Pharmacology of Epileptogenesis and Related Neurological/Psychiatric Comorbidities. Neurosci. Res. Notes 2019, 1, 18–34. [Google Scholar] [CrossRef]
- Sitnikova, E.; Grubov, V.; Hramov, A.E. Slow—Wave Activity Preceding the Onset of 10–15-Hz Sleep Spindles and 5–9-Hz Oscillations in Electroencephalograms in Rats with and without Absence Seizures. J. Sleep Res. 2019, 29, e12927. [Google Scholar] [CrossRef]
- Perescis, M.F.J.; van Luijtelaar, G.; van Rijn, C.M. Neonatal Exposure to AY-9944 Increases Typical Spike and Wave Discharges in WAG/Rij and Wistar Rats. Epilepsy Res. 2019, 157, 106184. [Google Scholar] [CrossRef] [PubMed]
- Vergnes, M.; Marescaux, C.; Micheletti, G.; Reis, J.; Depaulis, A.; Rumbach, L.; Warter, J.M. Spontaneous Paroxysmal Electroclinical Patterns in Rat: A Model of Generalized Non-Convulsive Epilepsy. Neurosci. Lett. 1982, 33, 97–101. [Google Scholar] [CrossRef] [PubMed]
- Marques-Carneiro, J.E.; Faure, J.-B.; Cosquer, B.; Koning, E.; Ferrandon, A.; de Vasconcelos, A.P.; Cassel, J.-C.; Nehlig, A. Anxiety and Locomotion in Genetic Absence Epilepsy Rats from Strasbourg (GAERS): Inclusion of Wistar Rats as a Second Control. Epilepsia 2014, 55, 1460–1468. [Google Scholar] [CrossRef]
- André, E.S.; Bruno-Neto, R.; Marino-Neto, J.; do Valle, A.C.; Timo-Iaria, C. Spontaneous Absence-like Activity in Wistar Rats: Behavioral and Electrographic Characteristics and the Effects of Antiepileptic Drugs. Acta Sci. Biol. Sci. 2014, 36, 231. [Google Scholar] [CrossRef]
- Taylor, J.A.; Reuter, J.D.; Kubiak, R.A.; Mufford, T.T.; Booth, C.J.; Dudek, F.E.; Barth, D.S. Spontaneous Recurrent Absence Seizure-like Events in Wild-Caught Rats. J. Neurosci. 2019, 39, 4829–4841. [Google Scholar] [CrossRef]
- McCafferty, C.; Gruenbaum, B.F.; Tung, R.; Li, J.-J.; Zheng, X.; Salvino, P.; Vincent, P.; Kratochvil, Z.; Ryu, J.H.; Khalaf, A.; et al. Decreased but Diverse Activity of Cortical and Thalamic Neurons in Consciousness-Impairing Rodent Absence Seizures. Nat. Commun. 2023, 14, 117. [Google Scholar] [CrossRef]
- Sarkisova, K.; van Luijtelaar, G. The WAG/Rij Strain: A Genetic Animal Model of Absence Epilepsy with Comorbidity of Depressiony. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 2011, 35, 854–876. [Google Scholar] [CrossRef] [PubMed]
- Leo, A.; Citraro, R.; Tallarico, M.; Iannone, M.; Fedosova, E.; Nesci, V.; De Sarro, G.; Sarkisova, K.; Russo, E. Cognitive Impairment in the WAG/Rij Rat Absence Model Is Secondary to Absence Seizures and Depressive-like Behavior. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 2019, 94, 109652. [Google Scholar] [CrossRef] [PubMed]
- Midzyanovskaya, I.S.; Shatskova, A.B.; Sarkisova, K.Y.; van Luijtelaar, G.; Tuomisto, L.; Kuznetsova, G.D. Convulsive and Nonconvulsive Epilepsy in Rats: Effects on Behavioral Response to Novelty Stress. Epilepsy Behav. 2005, 6, 543–551. [Google Scholar] [CrossRef] [PubMed]
- Karson, A.; Utkan, T.; Balcı, F.; Arıcıoğlu, F.; Ateş, N. Age-Dependent Decline in Learning and Memory Performances of WAG/Rij Rat Model of Absence Epilepsy. Behav. Brain Funct. 2012, 8, 51. [Google Scholar] [CrossRef] [PubMed]
- Gruenbaum, B.F.; Sandhu, M.R.S.; Bertasi, R.A.O.; Bertasi, T.G.O.; Schonwald, A.; Kurup, A.; Gruenbaum, S.E.; Freedman, I.G.; Funaro, M.C.; Blumenfeld, H.; et al. Absence Seizures and Their Relationship to Depression and Anxiety: Evidence for Bidirectionality. Epilepsia 2021, 62, 1041–1056. [Google Scholar] [CrossRef] [PubMed]
- Jafarian, M.; Karimzadeh, F.; Alipour, F.; Attari, F.; Lotfinia, A.A.; Speckmann, E.-J.; Zarrindast, M.-R.; Gorji, A. Cognitive Impairments and Neuronal Injury in Different Brain Regions of a Genetic Rat Model of Absence Epilepsy. Neuroscience 2015, 298, 161–170. [Google Scholar] [CrossRef] [PubMed]
- Alexandrov, P.; Pupikina, M.; Adaeva, Z.; Sitnikova, E. The Difference between Male and Female Rats in Terms of Freezing and Aversive Ultrasonic Vocalization in an Active Avoidance Test. Physiologia 2023, 3, 406–420. [Google Scholar] [CrossRef]
- Sitnikova, E.; Smirnov, K. Active Avoidance Learning in WAG/Rij Rats with Genetic Predisposition to Absence Epilepsy. Brain Res. Bull. 2020, 165, 198–208. [Google Scholar] [CrossRef]
- Pollak, D.D.; Rey, C.E.; Monje, F.J. Rodent Models in Depression Research: Classical Strategies and New Directions. Ann. Med. 2010, 42, 252–264. [Google Scholar] [CrossRef]
- Jones, N.C.; Salzberg, M.R.; Kumar, G.; Couper, A.; Morris, M.J.; O’Brien, T.J. Elevated Anxiety and Depressive-like Behavior in a Rat Model of Genetic Generalized Epilepsy Suggesting Common Causation. Exp. Neurol. 2008, 209, 254–260. [Google Scholar] [CrossRef]
- Berrio, J.P.; Hestehave, S.; Kalliokoski, O.; Paola, J.; Sanchez, B. Reliability of Sucrose Preference Testing Following Short or No Food and Water Deprivation—A Systematic Review and Meta–Analysis of Rat Models of Chronic Unpredictable Stress. bioRxiv 2023. [Google Scholar] [CrossRef]
- Porsolt, R.D.; Anton, G.; Blavet, N.; Jalfre, M. Behavioural Despair in Rats: A New Model Sensitive to Antidepressant Treatments. Eur. J. Pharmacol. 1978, 47, 379–391. [Google Scholar] [CrossRef]
- Steru, L.; Chermat, R.; Thierry, B.; Simon, P. The Tail Suspension Test: A New Method for Screening Antidepressants in Mice. Psychopharmacology 1985, 85, 367–370. [Google Scholar] [CrossRef] [PubMed]
- Chermat, R.; Thierry, B.; Mico, J.A.; Steru, L.; Simon, P. Adaptation of the Tail Suspension Test to the Rat. J. Pharmacol. 1986, 17, 348–350. [Google Scholar] [PubMed]
- Scheggi, S.; De Montis, M.G.; Gambarana, C. Making Sense of Rodent Models of Anhedonia. Int. J. Neuropsychopharmacol. 2018, 21, 1049–1065. [Google Scholar] [CrossRef] [PubMed]
- Markov, D.D. Sucrose Preference Test as a Measure of Anhedonic Behavior in a Chronic Unpredictable Mild Stress Model of Depression: Outstanding Issues. Brain Sci. 2022, 12, 1287. [Google Scholar] [CrossRef] [PubMed]
- Berrio, J.P.; Kalliokoski, O. Rethinking Data Treatment: The Sucrose Preference Threshold for Anhedonia in Stress-Induced Rat Models of Depression. J. Neurosci. Methods 2023, 395, 109910. [Google Scholar] [CrossRef] [PubMed]
- Arrant, A.E.; Schramm-Sapyta, N.L.; Kuhn, C.M. Use of the Light/Dark Test for Anxiety in Adult and Adolescent Male Rats. Behav. Brain Res. 2013, 256, 119–127. [Google Scholar] [CrossRef]
- Kraeuter, A.-K.; Guest, P.C.; Sarnyai, Z. The Open Field Test for Measuring Locomotor Activity and Anxiety-Like Behavior. In Pre-Clinical Models. Methods in Molecular Biology; Guest, P., Ed.; Humana Press: New York, NY, USA, 2019; Volume 1897, pp. 99–103. ISBN 9784431538592. [Google Scholar]
- Pellow, S.; Chopin, P.; File, S.E.; Briley, M. Validation of Open: Closed Arm Entries in an Elevated plus-Maze as a Measure of Anxiety in the Rat. J. Neurosci. Methods 1985, 14, 149–167. [Google Scholar] [CrossRef]
- Sarkisova, K.Y.; Kuznetsova, G.D.; Kulikov, M.A.; van Luijtelaar, G. Spike-Wave Discharges Are Necessary for the Expression of Behavioral Depression-like Symptoms. Epilepsia 2010, 51, 146–160. [Google Scholar] [CrossRef]
- Sarkisova, K.Y.; Midzianovskaia, I.S.; Kulikov, M.A. Depressive-like Behavioral Alterations and c-Fos Expression in the Dopaminergic Brain Regions in WAG/Rij Rats with Genetic Absence Epilepsy. Behav. Brain Res. 2003, 144, 211–226. [Google Scholar] [CrossRef] [PubMed]
- Fedosova, E.A.; Sarkisova, K.Y.; Kudrin, V.S.; Narkevich, V.B.; Bazyan, A.S. Behavioral and Neurochemical Characteristics of Two Months Old WAG/Rij Rats with Genetic Absence Epilepsy. Int. J. Clin. Exp. Neurol. 2015, 3, 32–44. [Google Scholar]
- Kliueva, I.A.; Chepurnova, N.E.; Berdiev, R.K.; Mart’ianov, A.A.; Mogimi, A.; Chepurnov, S.A. Anxiety and Behavior in WAG/Rij Strain Rats with Genetically Induced Absence Attacks. Zh Vyss. Nerv Deiat Im I P Pavlov. 1999, 49, 1018–1026. [Google Scholar]
- Sarkisova, K.Y.; Kulikov, M.A. Behavioral Characteristics of WAG/Rij Rats Susceptible and Non-Susceptible to Audiogenic Seizures. Behav. Brain Res. 2006, 166, 9–18. [Google Scholar] [CrossRef] [PubMed]
- Van der Staay, F.J. Spatial Working Memory and Reference Memory of Brown Norway and WAG Rats in a Holeboard Discrimination Task. Neurobiol. Learn. Mem. 1999, 71, 113–125. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Dobryakova, Y.V.; Dubynin, V.A.; van Luijtelaar, G. Maternal Behavior in a Genetic Animal Model of Absence Epilepsy. Acta Neurobiol. Exp. 2008, 68, 502–508. [Google Scholar] [CrossRef] [PubMed]
- Marks, W.N.; Cavanagh, M.E.; Greba, Q.; Cain, S.M.; Snutch, T.P.; Howland, J.G. The Genetic Absence Epilepsy Rats from Strasbourg Model of Absence Epilepsy Exhibits Alterations in Fear Conditioning and Latent Inhibition Consistent with Psychiatric Comorbidities in Humans. Eur. J. Neurosci. 2016, 43, 25–40. [Google Scholar] [CrossRef]
- Neuparth-Sottomayor, M.; Pina, C.C.; Morais, T.P.; Farinha-Ferreira, M.; Abreu, D.S.; Solano, F.; Mouro, F.; Good, M.; Sebastião, A.M.; Di Giovanni, G.; et al. Cognitive Comorbidities of Experimental Absence Seizures Are Independent of Anxiety. Neurobiol. Dis. 2023, 186, 106275. [Google Scholar] [CrossRef]
- Crunelli, V.; Lőrincz, M.L.; McCafferty, C.; Lambert, R.C.; Leresche, N.; Di Giovanni, G.; David, F. Clinical and Experimental Insight into Pathophysiology, Comorbidity and Therapy of Absence Seizures. Brain 2020, 143, 2341–2368. [Google Scholar] [CrossRef]
- Lüttjohann, A.; van Luijtelaar, G. Dynamics of Networks during Absence Seizure’s on- and Offset in Rodents and Man. Front. Physiol. 2015, 6, 16. [Google Scholar] [CrossRef]
- Hughes, S.W.; Crunelli, V. Thalamic Mechanisms of EEG Alpha Rhythms and Their Pathological Implications. Neuroscientist 2005, 11, 357–372. [Google Scholar] [CrossRef]
- Moustafa, A.A.; McMullan, R.D.; Rostron, B.; Hewedi, D.H.; Haladjian, H.H. The Thalamus as a Relay Station and Gatekeeper: Relevance to Brain Disorders. Rev. Neurosci. 2017, 28, 203–218. [Google Scholar] [CrossRef]
- Hwang, K.; Bertolero, M.A.; Liu, W.B.; D’Esposito, M. The Human Thalamus Is an Integrative Hub for Functional Brain Networks. J. Neurosci. 2017, 37, 5594–5607. [Google Scholar] [CrossRef] [PubMed]
- Jones, E.G. Thalamocortical Dysrhythmia and Chronic Pain. Pain 2010, 150, 4–5. [Google Scholar] [CrossRef] [PubMed]
- Llinás, R.; Ribary, U.; Jeanmonod, D.; Cancro, R.; Kronberg, E.; Schulman, J.; Zonenshayn, M.; Magnin, M.; Morel, A.; Siegmund, M. Thalamocortical Dysrhythmia I. Functional and Imaging Aspects. Thalamus Relat. Syst. 2001, 1, 237–244. [Google Scholar] [CrossRef]
- Schulman, J.J.; Cancro, R.; Lowe, S.; Lu, F.; Walton, K.D.; Llinás, R.R. Imaging of Thalamocortical Dysrhythmia in Neuropsychiatry. Front. Hum. Neurosci. 2011, 5, 69. [Google Scholar] [CrossRef] [PubMed]
- Smyk, M.K.; Coenen, A.M.L.; Lewandowski, M.H.; van Luijtelaar, G. Endogenous Rhythm of Absence Epilepsy: Relationship with General Motor Activity and Sleep-Wake States. Epilepsy Res. 2011, 93, 120–127. [Google Scholar] [CrossRef] [PubMed]
- Coenen, A.M.L.; Drinkenburg, W.H.I.M.; Peeters, B.W.M.M.; Vossen, J.M.H.; van Luijtelaar, E.L.J.M. Absence Epilepsy and the Level of Vigilance in Rats of the WAG/Rij Strain. Neurosci. Biobehav. Rev. 1991, 15, 259–263. [Google Scholar] [CrossRef] [PubMed]
- Vega, C.; Vestal, M.; DeSalvo, M.; Berman, R.; Chung, M.H.; Blumenfeld, H.; Spann, M.N. Differentiation of Attention-Related Problems in Childhood Absence Epilepsy. Epilepsy Behav. 2010, 19, 82–85. [Google Scholar] [CrossRef]
- Leo, A.; De Caro, C.; Nesci, V.; Palma, E.; Tallarico, M.; Iannone, M.; Constanti, A.; De Sarro, G.; Russo, E.; Citraro, R. Antiepileptogenic Effects of Ethosuximide and Levetiracetam in WAG/Rij Rats Are Only Temporary. Pharmacol. Rep. 2019, 71, 833–838. [Google Scholar] [CrossRef]
- Pearce, P.S.; Friedman, D.; LaFrancois, J.J.; Iyengar, S.S.; Fenton, A.A.; MacLusky, N.J.; Scharfman, H.E. Spike–Wave Discharges in Adult Sprague–Dawley Rats and Their Implications for Animal Models of Temporal Lobe Epilepsy. Epilepsy Behav. 2014, 32, 121–131. [Google Scholar] [CrossRef]
- Vergnes, M.; Marescaux, C.; Depaulis, A. Mapping of Spontaneous Spike and Wave Discharges in Wistar Rats with Genetic Generalized Non-Convulsive Epilepsy. Brain Res. 1990, 523, 87–91. [Google Scholar] [CrossRef]
- Shaw, F.Z. Is Spontaneous High-Voltage Rhythmic Spike Discharge in Long Evans Rats an Absence-Like Seizure Activity? J. Neurophysiol. 2004, 91, 63–77. [Google Scholar] [CrossRef]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sitnikova, E. Behavioral and Cognitive Comorbidities in Genetic Rat Models of Absence Epilepsy (Focusing on GAERS and WAG/Rij Rats). Biomedicines 2024, 12, 122. https://doi.org/10.3390/biomedicines12010122
Sitnikova E. Behavioral and Cognitive Comorbidities in Genetic Rat Models of Absence Epilepsy (Focusing on GAERS and WAG/Rij Rats). Biomedicines. 2024; 12(1):122. https://doi.org/10.3390/biomedicines12010122
Chicago/Turabian StyleSitnikova, Evgenia. 2024. "Behavioral and Cognitive Comorbidities in Genetic Rat Models of Absence Epilepsy (Focusing on GAERS and WAG/Rij Rats)" Biomedicines 12, no. 1: 122. https://doi.org/10.3390/biomedicines12010122
APA StyleSitnikova, E. (2024). Behavioral and Cognitive Comorbidities in Genetic Rat Models of Absence Epilepsy (Focusing on GAERS and WAG/Rij Rats). Biomedicines, 12(1), 122. https://doi.org/10.3390/biomedicines12010122







