Towards a Procedure-Optimised Steerable Catheter for Deep-Seated Neurosurgery
Abstract
1. Introduction
2. Materials and Methods
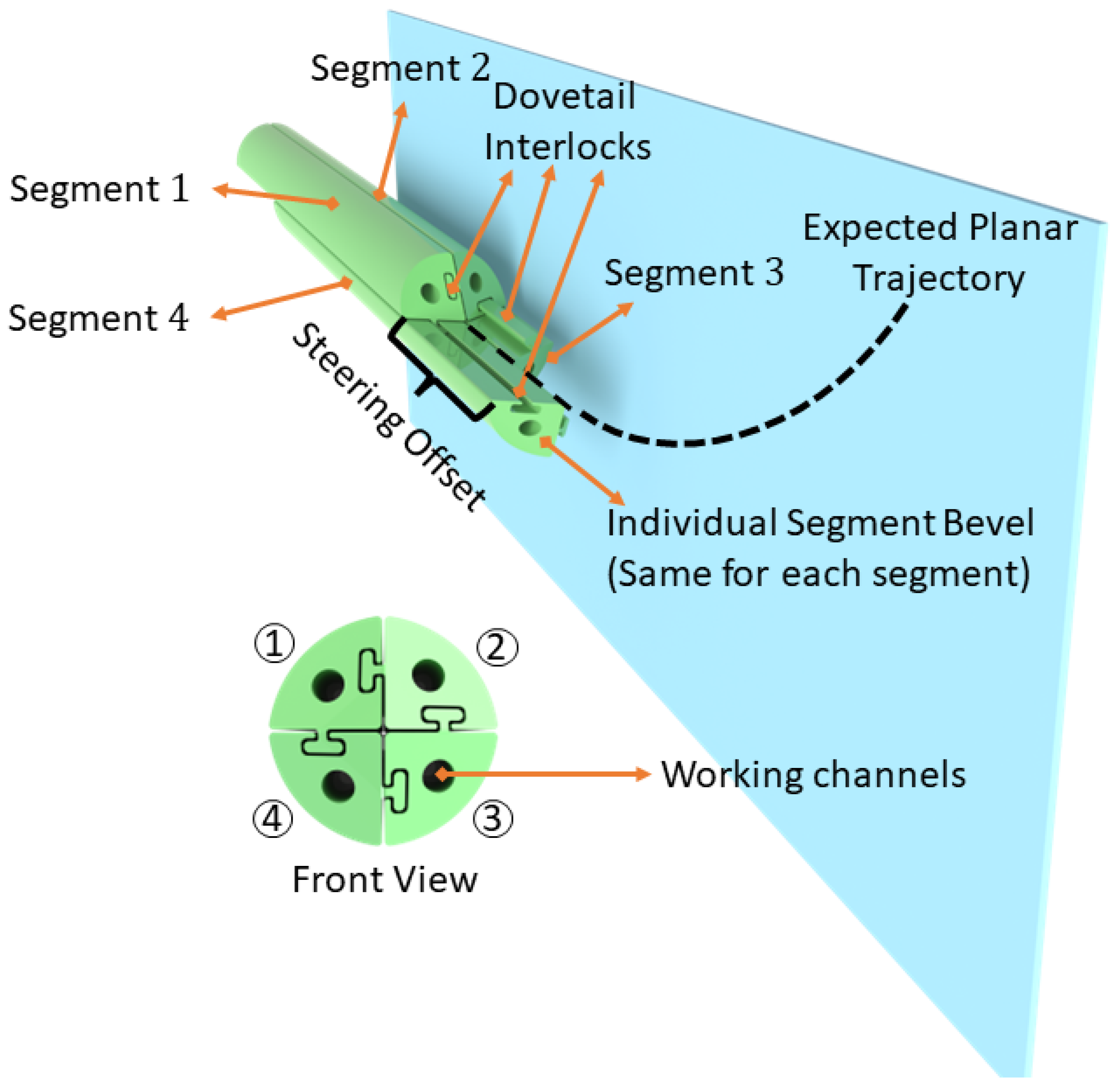
2.1. Catheter Design and Manufacturing
2.1.1. Extrusion-Manufactured (EM) PBN Catheter
2.1.2. Thermally Drawn Catheter (TD)
2.2. Characterization Methods
2.2.1. Mechanical Feature Testing
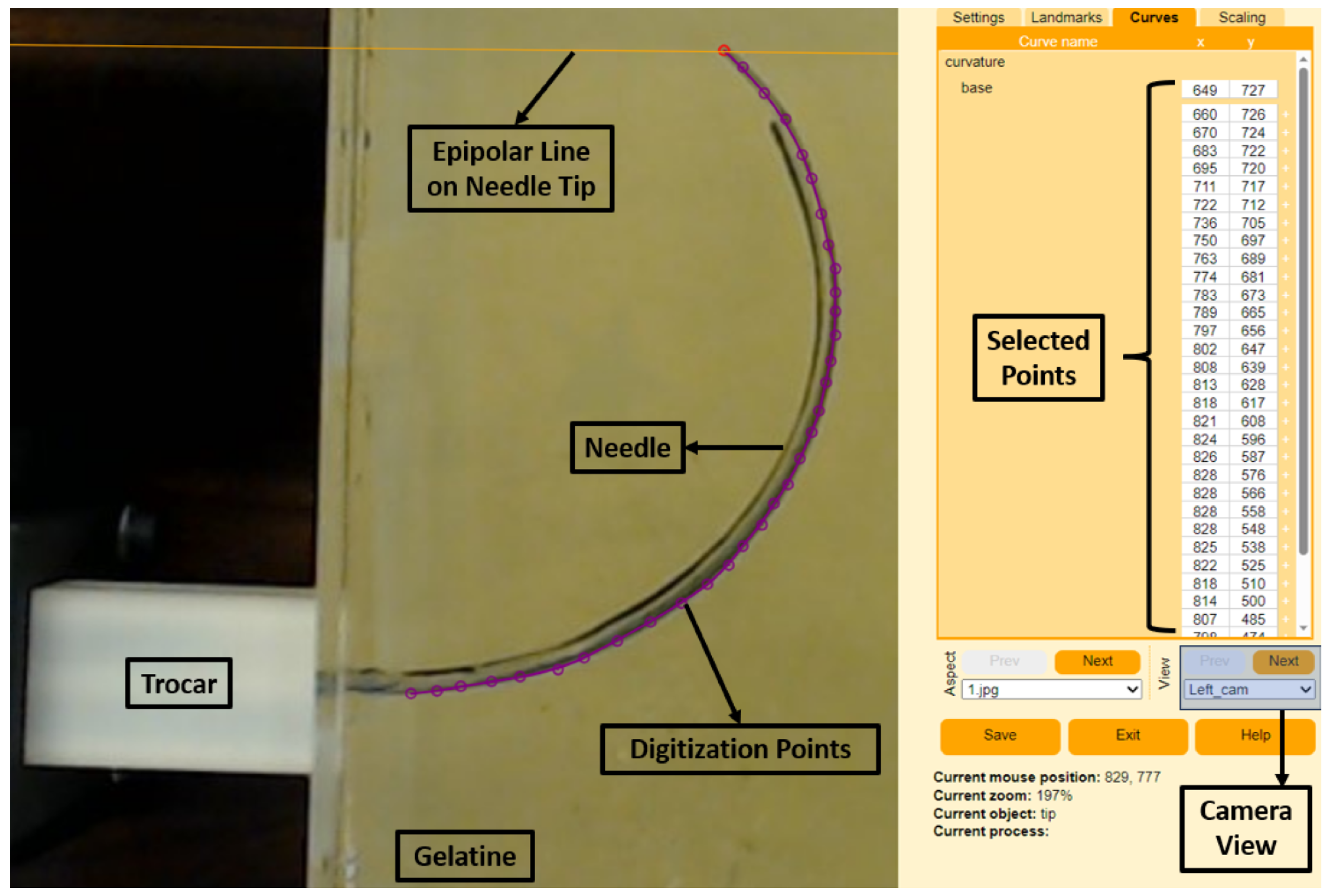
2.2.2. Curvature Estimation Method
3. Experimental Validation
4. Results
4.1. Mechanical Features
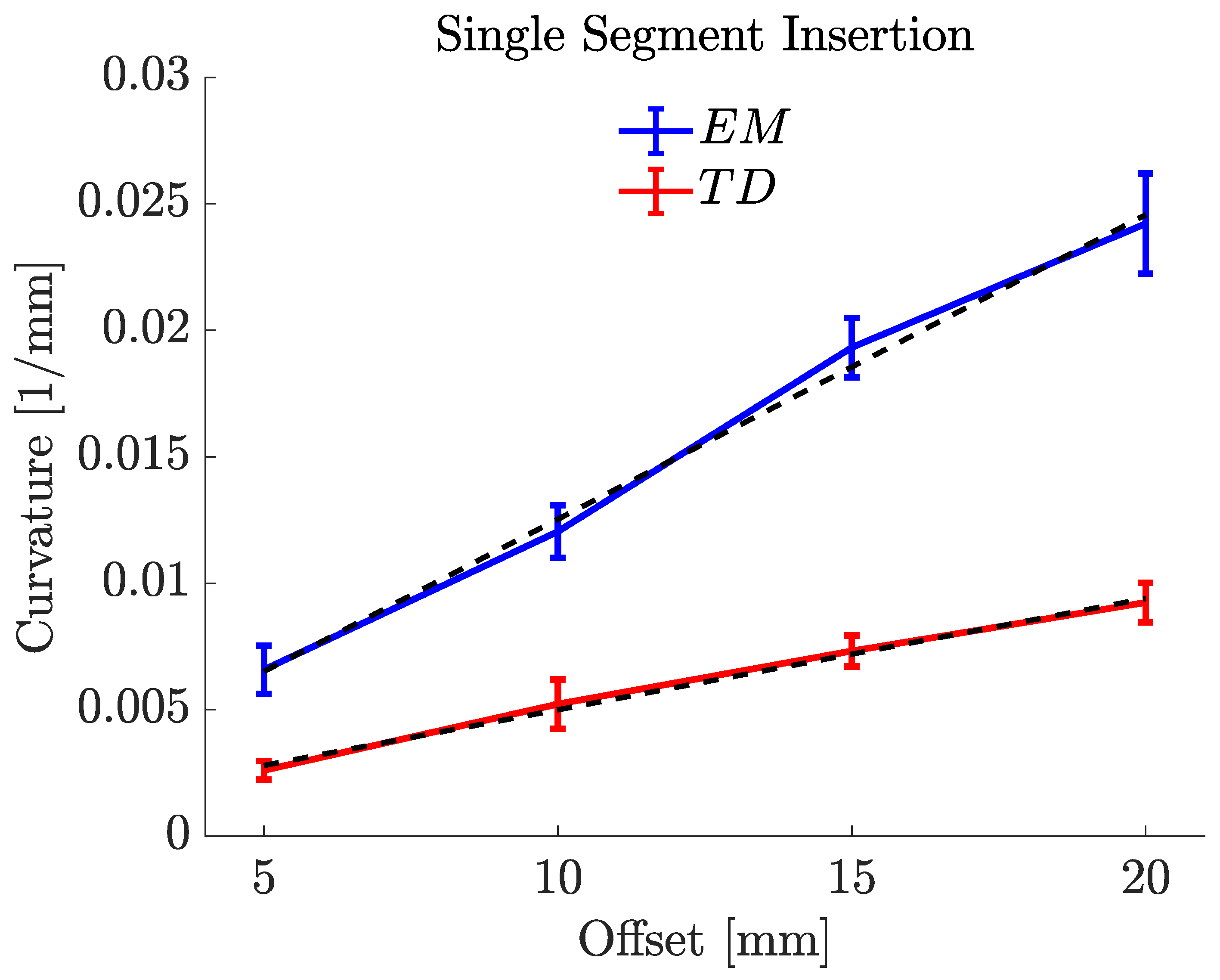
4.2. Offset vs. Curvature
5. Discussion
- Improved sliding behaviour: Enhances the sliding behaviour of the catheter segments, reducing the risk of tissue damage during insertion and removal;
- Stronger segment interlocking: Creates stronger interlocking segments, minimizing the likelihood of segment separation;
- Smaller catheter size: Enables the production of small-size catheters, beneficial for MIS procedures and patient comfort;
- Design flexibility: Offers adaptability in catheter designs to meet various surgical needs;
- Reduced post-production processes: Eliminates the need for additional post-production processes to improve sliding behaviour and ensure biocompatibility;
- Cost-effectiveness: Reduces manufacturing costs by eliminating complex tooling and moulds used in conventional techniques.
- Lower steering performance: The material’s stiffness in thermal drawing can limit the catheter’s manoeuvrability when navigating complex anatomical structures;
- Limited material selection: The method is primarily suitable for amorphous thermoplastics, limiting the choice of materials for catheter manufacturing;
- Challenges in material characterization: Extensive testing and characterization are necessary to evaluate material properties for thermal drawing;
- Process optimization: Achieving consistent and reliable size and catheter features requires meticulous parameter adjustments (though this is true for other manufacturing methods too);
- The complexity of preform fabrication: achieving preforms with desired properties through 3D printing and catheter design expertise can be challenging.
6. Conclusions and Future Work
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| MIS | minimally invasive surgery |
| CED | convection-enhanced delivery |
| TD | thermally drawn |
| EM | extrusion-manufactured |
| PBN | programmable bevel-tip needle |
| LiTT | laser interstitial thermal therapy |
| PC | poly carbonate |
References
- van der Putten, E.P.W.; Goossens, R.H.M.; Jakimowicz, J.J.; Dankelman, J. Haptics in minimally invasive surgery a review. Minim. Invasive Ther. Allied Technol. 2008, 17, 3–16. [Google Scholar] [CrossRef] [PubMed]
- Ohuchida, K. Robotic Surgery in Gastrointestinal Surgery. Cyborg Bionic Syst. 2020, 2020, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Gu, X.; Ren, H. A Survey of Transoral Robotic Mechanisms: Distal Dexterity, Variable Stiffness, and Triangulation. Cyborg Bionic Syst. 2023, 4, 0007. [Google Scholar] [CrossRef]
- Nakadate, R.; Iwasa, T.; Onogi, S.; Arata, J.; Oguri, S.; Okamoto, Y.; Akahoshi, T.; Eto, M.; Hashizume, M. Surgical Robot for Intraluminal Access: An Ex Vivo Feasibility Study. Cyborg Bionic Syst. 2020, 2020, 1–9. [Google Scholar] [CrossRef]
- Secoli, R.; Matheson, E.; Pinzi, M.; Galvan, S.; Donder, A.; Watts, T.; Riva, M.; Zani, D.D.; Bello, L.; Rodriguez y Baena, F. Modular robotic platform for precision neurosurgery with a bio-inspired needle: System overview and first in-vivo deployment. PLoS ONE 2022, 17, e0275686. [Google Scholar] [CrossRef] [PubMed]
- Kulkarni, P.; Sikander, S.; Biswas, P.; Frawley, S.; Song, S.E. Review of robotic needle guide systems for percutaneous intervention. Ann. Biomed. Eng. 2019, 47, 2489–2513. [Google Scholar] [CrossRef]
- Patel, N.K.; Plaha, P.; Gill, S.S. Magnetic resonance imaging-directed method for functional neurosurgery using implantable guide tubes. Oper. Neurosurg. 2007, 61, ONS358–ONS366. [Google Scholar] [CrossRef]
- van de Berg, N.J.; van Gerwen, D.J.; Dankelman, J.; van den Dobbelsteen, J.J. Design Choices in Needle Steering-A Review. IEEE/ASME Trans. Mechatron. 2015, 20, 2172–2183. [Google Scholar] [CrossRef]
- Misra, S.; Reed, K.B.; Schafer, B.W.; Ramesh, K.; Okamura, A.M. Mechanics of flexible needles robotically steered through soft tissue. Int. J. Robot. Res. 2010, 29, 1640–1660. [Google Scholar] [CrossRef]
- Dupont, P.E.; Nelson, B.J.; Goldfarb, M.; Hannaford, B.; Menciassi, A.; O’Malley, M.K.; Simaan, N.; Valdastri, P.; Yang, G.Z. A decade retrospective of medical robotics research from 2010 to 2020. Sci. Robot. 2021, 6, eabi8017. [Google Scholar] [CrossRef]
- Mignon, P.; Poignet, P.; Troccaz, J. Automatic robotic steering of flexible needles from 3D ultrasound images in phantoms and ex vivo biological tissue. Ann. Biomed. Eng. 2018, 46, 1385–1396. [Google Scholar] [CrossRef] [PubMed]
- Babaiasl, M.; Yang, F.; Swensen, J.P. Robotic needle steering: State-of-the-art and research challenges. Intell. Serv. Robot. 2022, 15, 679–711. [Google Scholar] [CrossRef]
- Lu, M.; Zhang, Y.; Lim, C.M.; Ren, H. Flexible Needle Steering with Tethered and Untethered Actuation: Current States, Targeting Errors, Challenges and Opportunities. Ann. Biomed. Eng. 2023, 51, 905–924. [Google Scholar] [CrossRef] [PubMed]
- Robert, J.; Webster, I.; Jones, B.A. Design and Kinematic Modeling of Constant Curvature Continuum Robots: A Review. Int. J. Robot. Res. 2010, 29, 1661–1683. [Google Scholar] [CrossRef]
- Glozman, D.; Shoham, M. Image-Guided Robotic Flexible Needle Steering. IEEE Trans. Robot. 2007, 23, 459–467. [Google Scholar] [CrossRef]
- Majewicz, A.; Marra, S.P.; van Vledder, M.G.; Lin, M.; Choti, M.A.; Song, D.Y.; Okamura, A.M. Behavior of Tip-Steerable Needles in Ex Vivo and In Vivo Tissue. IEEE Trans. Biomed. Eng. 2012, 59, 2705–2715. [Google Scholar] [CrossRef]
- DiMaio, S.; Salcudean, S. Interactive simulation of needle insertion models. IEEE Trans. Biomed. Eng. 2005, 52, 1167–1179. [Google Scholar] [CrossRef]
- Roesthuis, R.J.; van de Berg, N.J.; van den Dobbelsteen, J.J.; Misra, S. Modeling and steering of a novel actuated-tip needle through a soft-tissue simulant using Fiber Bragg Grating sensors. In Proceedings of the 2015 IEEE International Conference on Robotics and Automation (ICRA), Seattle, WA, USA, 26–30 May 2015; pp. 2283–2289. [Google Scholar] [CrossRef]
- Ayvali, E.; Liang, C.P.; Ho, M.; Chen, Y.; Desai, J.P. Towards a discretely actuated steerable cannula for diagnostic and therapeutic procedures. Int. J. Robot. Res. 2012, 31, 588–603. [Google Scholar] [CrossRef]
- Ilami, M.; Ahmed, R.J.; Petras, A.; Beigzadeh, B.; Marvi, H. Magnetic needle steering in soft phantom tissue. Sci. Rep. 2020, 10, 2500. [Google Scholar] [CrossRef]
- Frasson, L.; Ferroni, F.; Ko, S.Y.; Dogangil, G.; Rodriguez y Baena, F. Experimental evaluation of a novel steerable probe with a programmable bevel tip inspired by nature. J. Robot. Surg. 2012, 6, 189–197. [Google Scholar] [CrossRef]
- Ko, S.Y.; Frasson, L.; Rodriguez y Baena, F. Closed-Loop Planar Motion Control of a Steerable Probe With a “Programmable Bevel” Inspired by Nature. IEEE Trans. Robot. 2011, 27, 970–983. [Google Scholar] [CrossRef]
- Matheson, E.; Watts, T.; Secoli, R.; Baena, F.R.y. Cyclic Motion Control for Programmable Bevel-Tip Needle 3D Steering: A Simulation Study. In Proceedings of the 2018 IEEE International Conference on Robotics and Biomimetics (ROBIO), Kuala Lumpur, Malaysia, 12–15 December 2018; pp. 444–449. [Google Scholar] [CrossRef]
- Pinzi, M.; Vakharia, V.N.; Hwang, B.Y.; Anderson, W.S.; Duncan, J.S.; Baena, F.R.y. Computer Assisted Planning for Curved Laser Interstitial Thermal Therapy. IEEE Trans. Biomed. Eng. 2021, 68, 2957–2964. [Google Scholar] [CrossRef] [PubMed]
- Chen, R.; Canales, A.; Anikeeva, P. Neural recording and modulation technologies. Nat. Rev. Mater. 2017, 2, 1–16. [Google Scholar] [CrossRef]
- van der Elst, L.; de Lima, C.F.; Kurtoglu, M.G.; Koraganji, V.N.; Zheng, M.; Gumennik, A. 3D Printing in Fiber-Device Technology. Adv. Fiber Mater. 2021, 3, 59–75. [Google Scholar] [CrossRef]
- Cook, K.; Canning, J.; Leon-Saval, S.; Reid, Z.; Hossain, M.A.; Comatti, J.E.; Luo, Y.; Peng, G.D. Air-structured optical fiber drawn from a 3D-printed preform. Opt. Lett. 2015, 40, 3966–3969. [Google Scholar] [CrossRef] [PubMed]
- Leber, A.; Dong, C.; Laperrousaz, S.; Banerjee, H.; Abdelaziz, M.E.M.K.; Bartolomei, N.; Schyrr, B.; Temelkuran, B.; Sorin, F. Highly Integrated Multi-Material Fibers for Soft Robotics. Adv. Sci. 2023, 10, 2204016. [Google Scholar] [CrossRef]
- Abdelaziz, M.E.M.K.; Zhao, J.; Rosa, B.G.; Lee, H.T.; Simon, D.; Vyas, K.; Li, B.; Koguna, H.; Li, Y.; Demircali, A.A.; et al. Fiberbots: Robotic fibers for high-precision minimally invasive surgery. bioRxiv 2023. [Google Scholar] [CrossRef]
- Hart, S.D.; Maskaly, G.R.; Temelkuran, B.; Prideaux, P.H.; Joannopoulos, J.D.; Fink, Y. External Reflection from Omnidirectional Dielectric Mirror Fibers. Science 2002, 296, 510–513. [Google Scholar] [CrossRef]
- Temelkuran, B.; Hart, S.D.; Benoit, G.; Joannopoulos, J.D.; Fink, Y. Wavelength-scalable hollow optical fibres with large photonic bandgaps for CO2 laser transmission. Nature 2002, 420, 650–653. [Google Scholar] [CrossRef]
- Sorin, F.; Abouraddy, A.F.; Orf, N.; Shapira, O.; Viens, J.; Arnold, J.; Joannopoulos, J.D.; Fink, Y. Multimaterial photodetecting fibers: A geometric and structural study. Adv. Mater. 2007, 19, 3872–3877. [Google Scholar] [CrossRef]
- Bayindir, M.; Abouraddy, A.; Arnold, J.; Joannopoulos, J.; Fink, Y. Thermal-Sensing Fiber Devices by Multimaterial Codrawing. Adv. Mater. 2006, 18, 845–849. [Google Scholar] [CrossRef]
- Gumennik, A.; Stolyarov, A.M.; Schell, B.R.; Hou, C.; Lestoquoy, G.; Sorin, F.; McDaniel, W.; Rose, A.; Joannopoulos, J.D.; Fink, Y. All-in-Fiber Chemical Sensing. Adv. Mater. 2012, 24, 6005–6009. [Google Scholar] [CrossRef]
- Yan, W.; Page, A.; Nguyen-Dang, T.; Qu, Y.; Sordo, F.; Wei, L.; Sorin, F. Advanced Multimaterial Electronic and Optoelectronic Fibers and Textiles. Adv. Mater. 2019, 31, 1802348. [Google Scholar] [CrossRef]
- Khudiyev, T.; Lee, J.T.; Cox, J.R.; Argentieri, E.; Loke, G.; Yuan, R.; Noel, G.H.; Tatara, R.; Yu, Y.; Logan, F.; et al. 100 m Long Thermally Drawn Supercapacitor Fibers with Applications to 3D Printing and Textiles. Adv. Mater. 2020, 32, 2004971. [Google Scholar] [CrossRef] [PubMed]
- Burrows, C.; Secoli, R.; Rodriguez y Baena, F. Experimental characterisation of a biologically inspired 3D steering needle. In Proceedings of the 2013 13th International Conference on Control, Automation and Systems (ICCAS 2013), Gwangju, Republic of Korea, 20–23 October 2013; pp. 1252–1257. [Google Scholar] [CrossRef]
- Watts, T.; Secoli, R.; Baena, F.R.y. A Mechanics-Based Model for 3-D Steering of Programmable Bevel-Tip Needles. IEEE Trans. Robot. 2019, 35, 371–386. [Google Scholar] [CrossRef]
- Virdyawan, V. Sensorisation of a Novel Biologically Inspired Flexible Needle. Ph.D. Thesis, Imperial College London, London, UK, 2018. [Google Scholar] [CrossRef]
- Olsen, A.M.; Westneat, M.W. StereoMorph: An R package for the collection of 3D landmarks and curves using a stereo camera set-up. Methods Ecol. Evol. 2015, 6, 351–356. [Google Scholar] [CrossRef]
- Ko, S.Y.; Davies, B.L.; Rodriguez y Baena, F. Two-dimensional needle steering with a “programmable bevel” inspired by nature: Modeling preliminaries. In Proceedings of the 2010 IEEE/RSJ International Conference on Intelligent Robots and Systems, Taipei, Taiwan, 18–22 October 2010; pp. 2319–2324. [Google Scholar] [CrossRef]
- Donder, A.; Baena, F.R.y. Kalman-Filter-Based, Dynamic 3-D Shape Reconstruction for Steerable Needles With Fiber Bragg Gratings in Multicore Fibers. IEEE Trans. Robot. 2022, 38, 2262–2275. [Google Scholar] [CrossRef]
- Budday, S.; Steinmann, P.; Kuhl, E. The role of mechanics during brain development. J. Mech. Phys. Solids 2014, 72, 75–92. [Google Scholar] [CrossRef]
- Cao, B.; Deng, R.; Zhu, S. Universal algorithm for water depth refraction correction in through-water stereo remote sensing. Int. J. Appl. Earth Obs. Geoinf. 2020, 91, 102108. [Google Scholar] [CrossRef]
- Murthy, S. Best Fit 3D Circle to a Set of Points, MATLAB Central File Exchange. 2022. Available online: https://uk.mathworks.com/matlabcentral/fileexchange/55304-best-fit-3d-circle-to-a-set-of-points (accessed on 1 November 2022).
- Leibinger, A.; Forte, A.E.; Tan, Z.; Oldfield, M.J.; Beyrau, F.; Dini, D.; Rodriguez y Baena, F. Soft tissue phantoms for realistic needle insertion: A comparative study. Ann. Biomed. Eng. 2016, 44, 2442–2452. [Google Scholar] [CrossRef]
- Emerson, M.; Ferguson, J.M.; Ertop, T.E.; Rox, M.; Granna, J.; Lester, M.; Maldonado, F.; Gillaspie, E.A.; Alterovitz, R.; Webster, R.J.; et al. A recurrent neural network approach to roll estimation for needle steering. In Experimental Robotics. ISER 2020. Springer Proceedings in Advanced Robotics; Springer: Cham, Switzerland, 2021; pp. 334–342. [Google Scholar] [CrossRef]
- Morley, C.; Patel, R.V. Steering of Flexible Needles Using an LSTM Encoder with Model Predictive Control. In Proceedings of the 2022 2nd International Conference on Robotics, Automation and Artificial Intelligence (RAAI), Singapore, 9–11 December 2022; pp. 99–104. [Google Scholar] [CrossRef]






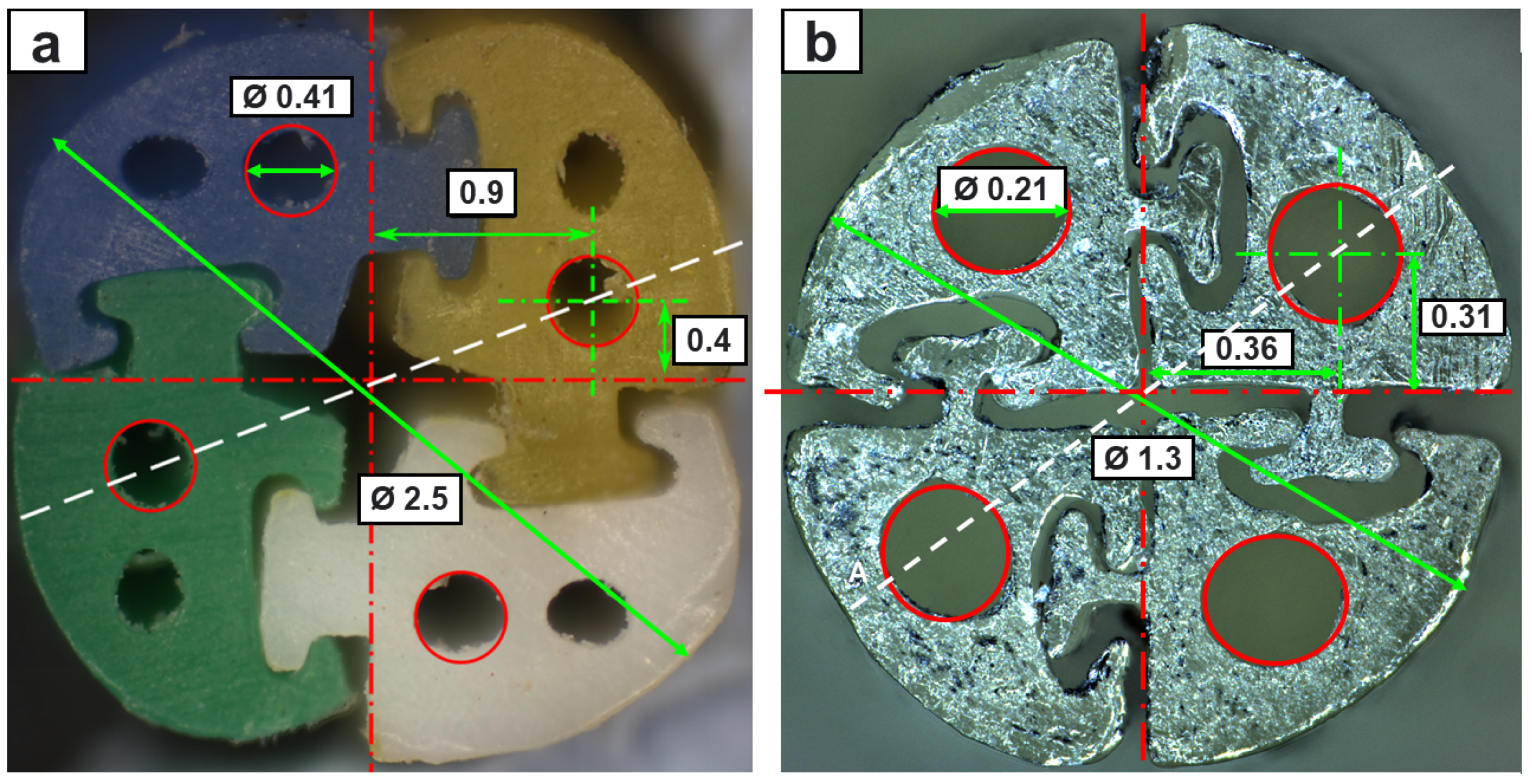

| Mean Flexural Stiffness (N/mm) | Mean Tensile Stress (MPa) | Mean Interlocking Breakout Force (N) | |
|---|---|---|---|
| 2.5 mm EM | 0.023 | 13.10 | 5.47 |
| 2.5 mm TD | 0.38 | 53.66 | 18.52 |
| 1.3 mm TD | 0.031 | 51.45 | 10.94 |
| Offsets (mm) | Mean (1/mm) | Mean R (mm) | Mean (degree) | |
|---|---|---|---|---|
| EM PBN | 5 | 0.0066 | 151.488 | 28.89 |
| 10 | 0.0120 | 83.306 | 42.68 | |
| 15 | 0.0193 | 52.454 | 54.58 | |
| 20 | 0.0242 | 41.307 | 69.20 | |
| TD PBN | 5 | 0.0026 | 385.516 | 8.93 |
| 10 | 0.0052 | 192.432 | 19.24 | |
| 15 | 0.0073 | 136.410 | 28.12 | |
| 20 | 0.0092 | 109.113 | 34.30 |
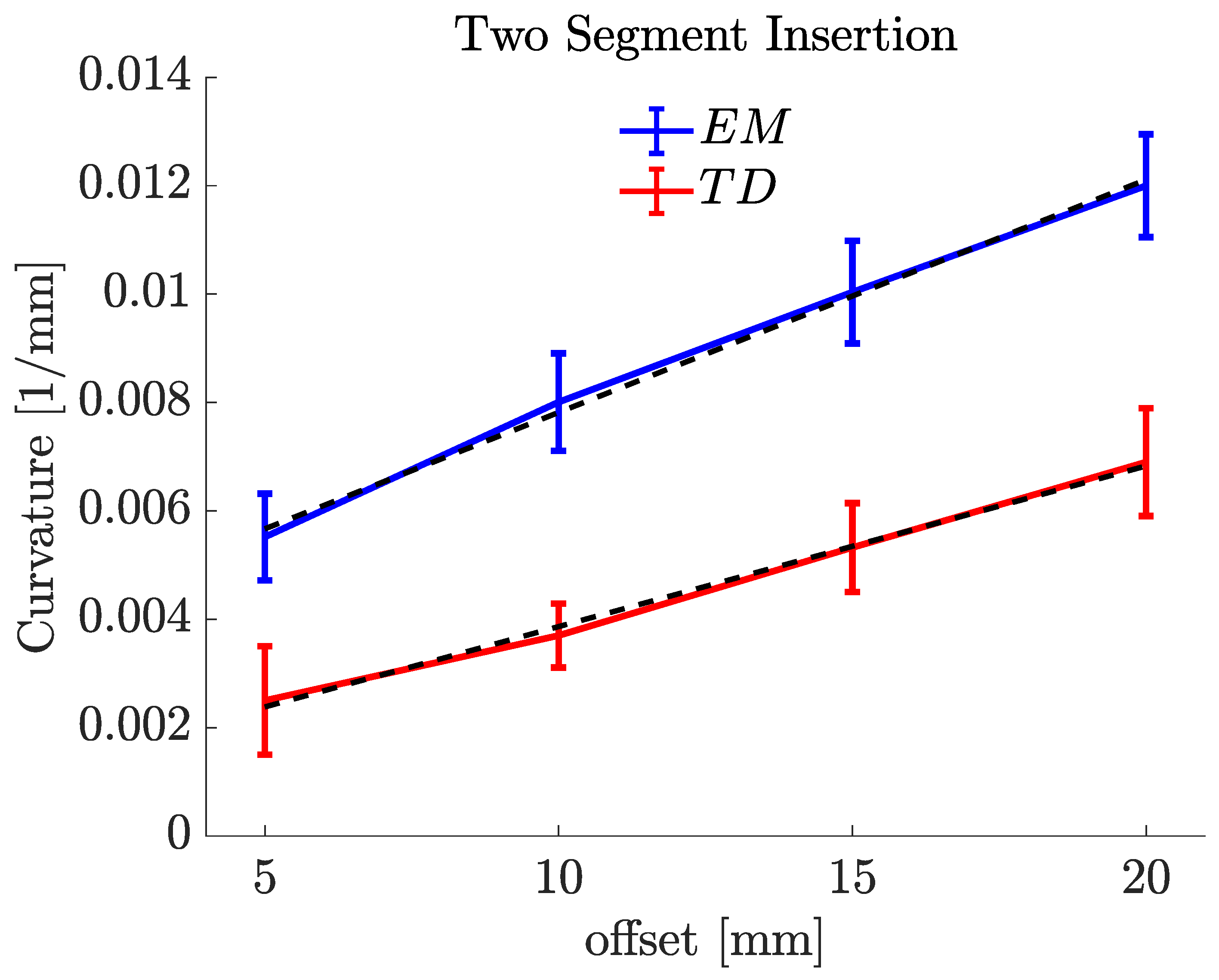
| Offsets (mm) | Mean (1/mm) | Mean R (mm) | |
|---|---|---|---|
| EM PBN | 5 | 0.0055 | 181.488 |
| 10 | 0.0080 | 125.036 | |
| 15 | 0.0102 | 98.034 | |
| 20 | 0.0121 | 82.644 | |
| TD PBN | 5 | 0.0025 | 400.056 |
| 10 | 0.0037 | 270.270 | |
| 15 | 0.0053 | 188.679 | |
| 20 | 0.0069 | 144.921 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Aktas, A.; Demircali, A.A.; Secoli, R.; Temelkuran, B.; Rodriguez y Baena, F. Towards a Procedure-Optimised Steerable Catheter for Deep-Seated Neurosurgery. Biomedicines 2023, 11, 2008. https://doi.org/10.3390/biomedicines11072008
Aktas A, Demircali AA, Secoli R, Temelkuran B, Rodriguez y Baena F. Towards a Procedure-Optimised Steerable Catheter for Deep-Seated Neurosurgery. Biomedicines. 2023; 11(7):2008. https://doi.org/10.3390/biomedicines11072008
Chicago/Turabian StyleAktas, Ayhan, Ali Anil Demircali, Riccardo Secoli, Burak Temelkuran, and Ferdinando Rodriguez y Baena. 2023. "Towards a Procedure-Optimised Steerable Catheter for Deep-Seated Neurosurgery" Biomedicines 11, no. 7: 2008. https://doi.org/10.3390/biomedicines11072008
APA StyleAktas, A., Demircali, A. A., Secoli, R., Temelkuran, B., & Rodriguez y Baena, F. (2023). Towards a Procedure-Optimised Steerable Catheter for Deep-Seated Neurosurgery. Biomedicines, 11(7), 2008. https://doi.org/10.3390/biomedicines11072008









