Synthesis and Characterization of PCL-Idebenone Nanoparticles for Potential Nose-to-Brain Delivery
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Methods
2.2.1. Preparation of NPs
2.2.2. Particle Size Analysis, Size Distribution, and Zeta Potential
2.2.3. Scanning Electron Microscopy (SEM)
2.2.4. Transmission Electron Microscopy (TEM)
2.2.5. Fourier-Transform Infrared (FTIR) Spectroscopy
2.2.6. Estimation of IDB Loading (DL) and Entrapment Efficiency (EE)
2.2.7. In Vitro Drug Release Studies and Release Kinetics
3. Results and Discussion
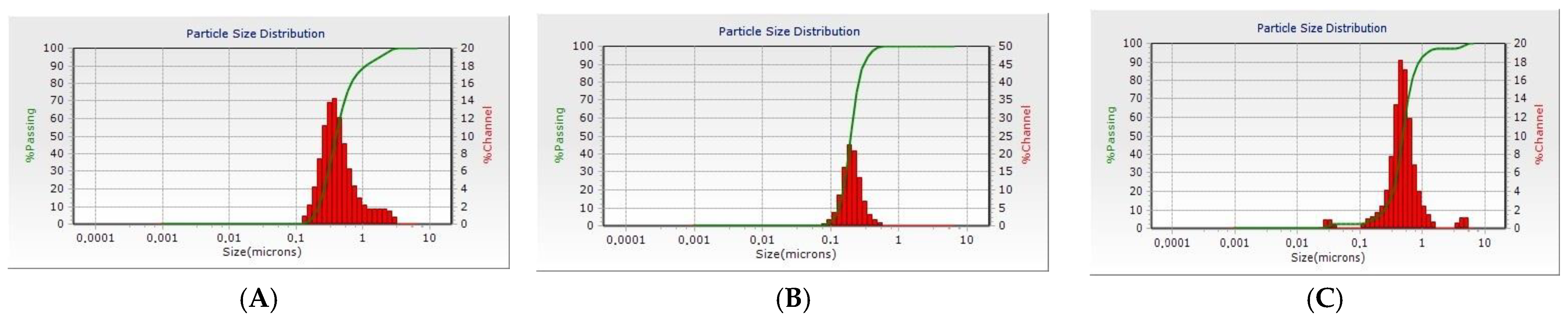
3.1. Synthesis and Characterization of Blank PCL NPs
3.2. Synthesis and Characterization of IDB-Loaded PCL NPs
3.2.1. Drug Loading and Entrapment Efficiency
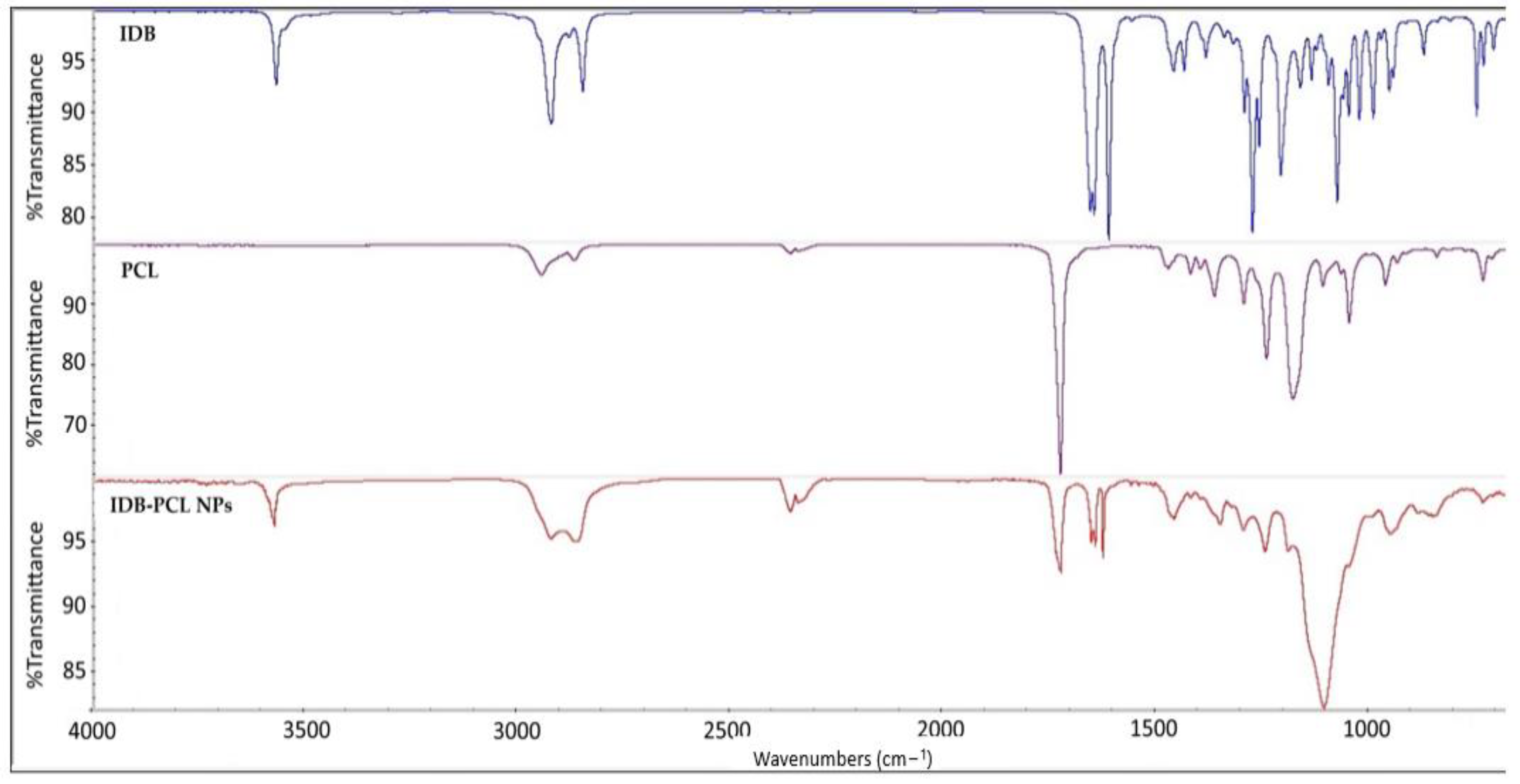
3.2.2. FTIR Analysis
3.2.3. Scanning Electron Microscopy and Transmission Electron Microscopy
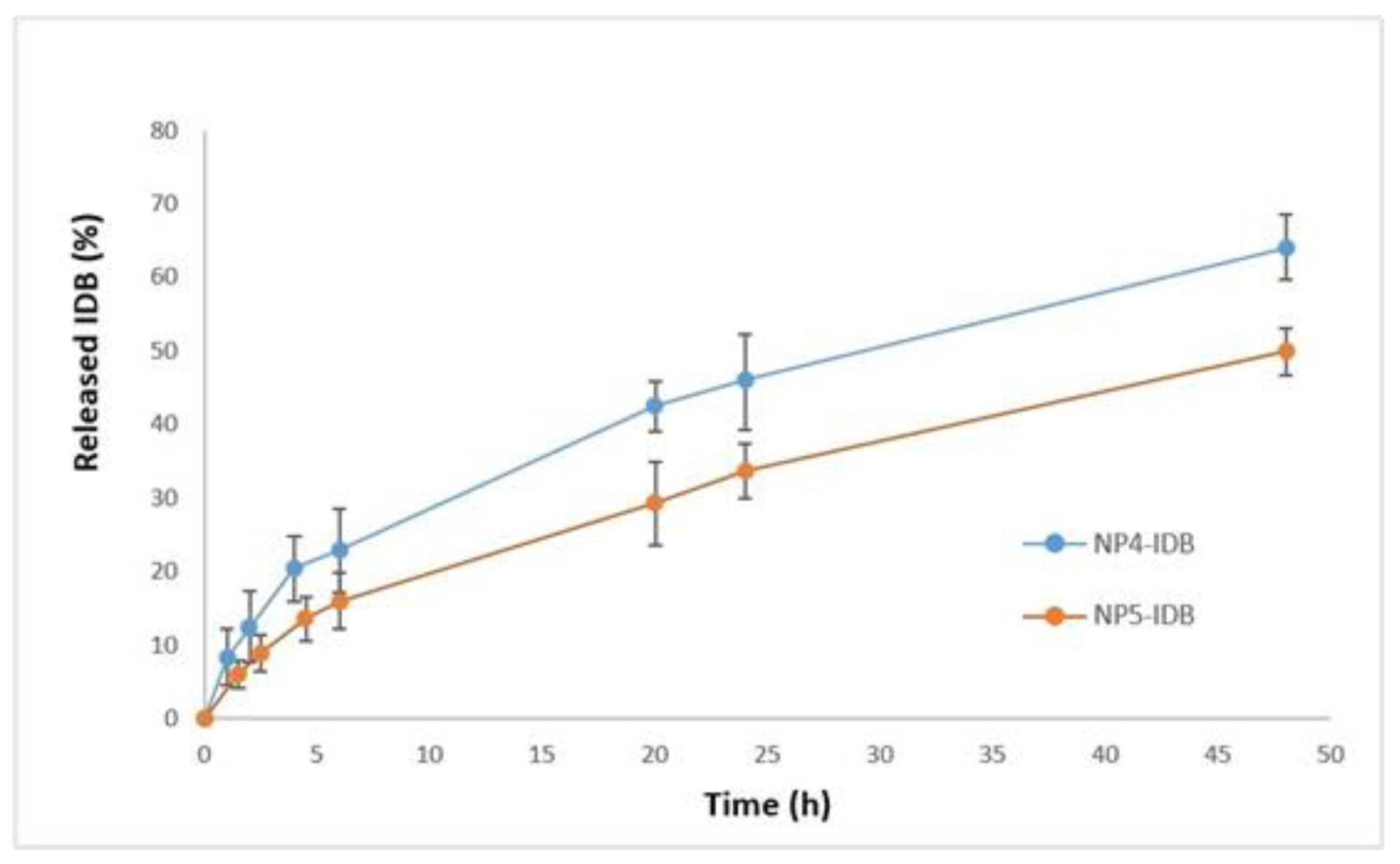
3.2.4. In Vitro Drug Release
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Albers, D.; Beal, M. Mitochondrial dysfunction and oxidative stress in aging and neurodegenerative disease. J. Neural Transm. Suppl. 2000, 59, 133–154. [Google Scholar] [CrossRef]
- Fischer, R.; Maier, M. Interrelation of oxidative stress and inflammation in neurodegenerative disease: Role of TNF. Oxidative Med. Cell. Longev. 2015, 2015, 610813. [Google Scholar] [CrossRef]
- Liu, Z.; Zhou, T.; Ziegler, A.C.; Dimitrion, P.; Zuo, L. Oxidative Stress in Neurodegenerative Diseases: From Molecular Mechanisms to Clinical Applications. Oxidative Med. Cell. Longev. 2017, 2017, 2525967. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Song, Y.; Chen, Z.; Leng, S.X. Connection between Systemic Inflammation and Neuroinflammation Underlies Neuroprotective Mechanism of Several Phytochemicals in Neurodegenerative Diseases. Oxidative Med. Cell. Longev. 2018, 2018, 1972714. [Google Scholar] [CrossRef]
- Feng, Y.; Wang, X. Antioxidant Therapies for Alzheimer’s Disease. Oxidative Med. Cell. Longev. 2012, 2012, 472932. [Google Scholar] [CrossRef] [PubMed]
- Collins, A.; Saleh, T.; Kalisch, B. Naturally Occurring Antioxidant Therapy in Alzheimer’s Disease. Antioxidants 2022, 11, 213. [Google Scholar] [CrossRef]
- Blesa, J.; Trigo-Damas, I.; Quiroga-Varela, A.; Jackson-Lewis, V.R. Oxidative Stress and Parkinson’s Disease. Front. Neuroanat. 2015, 9, 91. [Google Scholar] [CrossRef] [PubMed]
- Persson, T.; Popescu, B.; Cedazo-Minguez, A. Oxidative Stress in Alzheimer’s Disease: Why Did Antioxidant Therapy Fail? Oxidative Med. Cell. Longev. 2014, 2014, 427318. [Google Scholar] [CrossRef]
- Haefeli, R.; Erb, M.; Gemperli, A.; Robay, D.; Fruh, I.C.; Anklin, C.; Dallmann, R.; Gueven, N. NQO1-dependent redox cycling of idebenone: Effects on cellular redox potential and energy levels. PLoS ONE 2011, 6, e17963. [Google Scholar] [CrossRef]
- Gueven, N.; Ravishankar, P.; Eri, R.; Rybalka, E. Idebenone: When an antioxidant is not an antioxidant. Redox Biol. 2021, 38, 101812. [Google Scholar] [CrossRef]
- Yan, A.; Liu, Z.; Song, L.; Wang, X.; Zhang, Y.; Wu, N.; Lin, J.; Liu, Y.; Liu, Z. Idebenone Alleviates Neuroinflammation and Modulates Microglial Polarization in LPS-Stimulated BV2 Cells and MPTP-Induced Parkinson’s Disease Mice. Front. Cell Neurosci. 2019, 12, 29. [Google Scholar] [CrossRef] [PubMed]
- Peng, J.; Wang, H.; Gong, Z.; Li, X.; He, L.; Shen, Q.; Pan, J.; Peng, Y. Idebenone attenuates cerebral inflammatory injury in ischemia and reperfusion via dampening NLRP3 inflammasome activity. Mol. Immunol. 2020, 123, 74–87. [Google Scholar] [CrossRef] [PubMed]
- Fadda, L.; Hagar, H.; Mohamed, A.; Ali, H. Quercetin and Idebenone Ameliorate Oxidative Stress, Inflammation, DNA damage, and Apoptosis Induced by Titanium Dioxide Nanoparticles in Rat Liver. Dose Response 2018, 16, 4. [Google Scholar] [CrossRef]
- Jiang, W.; Geng, H.; Lv, X.; Ma, J.; Liu, F.; Lin, P.; Yan, C. Idebenone Protects against Atherosclerosis in Apolipoprotein E-Deficient Mice Via Activation of the SIRT3-SOD2-mtROS Pathway. Cardiovasc. Drugs Ther. 2021, 6, 1129–1145. [Google Scholar] [CrossRef] [PubMed]
- Lauro, F.; Ilari, S.; Giancotti, L.; Ventura, C.; Morabito, C.; Gliozzi, M.; Malafoglia, V.; Palma, E.; Paolino, D.; Mollace, V.; et al. Pharmacological effect of a new idebenone formulation in a model of carrageenan-induced inflammatory pain. Pharmacol. Res. 2016, 111, 767–773. [Google Scholar] [CrossRef]
- Thal, L.; Grundman, M.; Berg, J.; Ernstrom, K.; Margolin, R.; Pfeiffer, E.; Weiner, M.; Zamrini, E.; Thomas, R. Idebenone treatment fails to slow cognitive decline in Alzheimer’s disease. Neurology 2003, 61, 1498–1502. [Google Scholar] [CrossRef]
- Schlatter, J.; Bourguignon, E.; Majoul, E.; Kabiche, S.; Balde, I.; Cisternino, S.; Fontan, J. Stability study of oral pediatric idebenone suspensions. Pharm. Dev. Technol. 2017, 22, 296–299. [Google Scholar] [CrossRef]
- Qian, X.; Wang, G.; Li, J.; Zhang, Z.; Zhang, M.; Yang, Q.; Zhang, Z.; Li, Y. Improving oral bioavailability of water-insoluble idebenone with bioadhesive liposomes. J. Drug Deliv. Sci. Thecnol. 2022, 75, 103640. [Google Scholar] [CrossRef]
- Venuti, V.; Crupi, V.; Fazio, B.; Majolino, D.; Acri, G.; Testagrossa, B.; Stancanelli, R.; De Gaetano, F.; Gagliardi, A.; Paolino, D.; et al. Physicochemical Characterization and Antioxidant Activity Evaluation of Idebenone/Hydroxypropyl-β-Cyclodextrin Inclusion Complex. Biomolecules 2019, 9, 531. [Google Scholar] [CrossRef]
- Huang, Y.; Ma, M.; Zhu, X.; Li, M.; Guo, M.; Liu, P.; He, Z.; Fu, Q. Effectiveness of idebenone nanorod formulations in the treatment of Alzheimer’s disease. J. Control. Release 2021, 336, 169–180. [Google Scholar] [CrossRef]
- Gänger, S.; Schindowski, K. Tailoring Formulations for Intranasal Nose-to-Brain Delivery: A Review on Architecture, Physico-Chemical Characteristics and Mucociliary Clearance of the Nasal Olfactory Mucosa. Pharmaceutics 2018, 10, 116. [Google Scholar] [CrossRef] [PubMed]
- Schiöth, H.; Craft, S.; Brooks, S.; Frey, W.; Benedict, C. Brain insulin signaling and Alzheimer’s disease: Current evidence and future directions. Mol. Neurobiol. 2012, 46, 4–10. [Google Scholar] [CrossRef] [PubMed]
- Mistry, A.; Stolnik, S.; Illum, L. Nanoparticles for direct nose-to-brain delivery of drugs. Int. J. Pharm. 2009, 379, 146–157. [Google Scholar] [CrossRef] [PubMed]
- Chen, F.; Zhang, Z.; Yuan, F.; Qin, X.; Wang, M.; Huang, Y. In vitro and in vivo study of N-trimethyl chitosan nanoparticles for oral protein delivery. Int. J. Pharm. 2008, 349, 226–233. [Google Scholar] [CrossRef]
- Li, L.; LaBarbera, D. 3D High-Content Screening of Organoids for Drug Discovery. In Comprehensive Medicinal Chemistry III, 3rd ed.; Chackalamannil, S., Rotella, D., Ward, S.E., Eds.; Elsevier: Amsterdam, The Netherlands, 2017; pp. 388–415. [Google Scholar] [CrossRef]
- Nava-Arzaluz, M.; Piñón-Segundo, E.; Ganem-Rondero, A.; Lechuga-Ballesteros, D. Single emulsion-solvent evaporation technique and modifications for the preparation of pharmaceutical polymeric nanoparticles. Recent Pat. Drug Deliv. Formul. 2012, 6, 209–223. [Google Scholar] [CrossRef]
- Hakansson, A.; Tragardh, C.; Bergenstahl, B. Studying the effects of adsorption, recoalescence and fragmentation in a high pressure homogenizer using a dynamic simulation model. Food Hydrocoll. 2009, 23, 1177–1183. [Google Scholar] [CrossRef]
- McClements, D.; Jafari, S. Improving emulsion formation, stability and performance using mixed emulsifiers: A review. Adv. Colloid Interface Sci. 2018, 251, 55–79. [Google Scholar] [CrossRef]
- Cirin, D.; Krstonošic, V. Influence of Poloxamer 407 on Surface Properties of Aqueous Solutions of Polysorbate Surfactants. J. Surfactants Deterg. 2020, 23, 595–602. [Google Scholar] [CrossRef]
- Mainardes, R.; Urban, M.; Cinto, P.; Gremiao, M. Liposomes and micro/nanoparticles as colloidal carriers for nasal drug delivery. Curr. Drug Deliv. 2006, 3, 275–285. [Google Scholar] [CrossRef]
- Naqvi, S.; Panghal, A.; Flora, S. Nanotechnology: A Promising Approach for Delivery of Neuroprotective Drugs. Front. Neurosci. 2020, 14, 494. [Google Scholar] [CrossRef]
- Al-Maaieh, A.; Flanagan, D. Salt and cosolvents effects on ionic drug loading into microspheres using an O/W method. J. Control. Release 2001, 70, 169–181. [Google Scholar] [CrossRef] [PubMed]




| Conditions | Results |
|---|---|
| Oil/water ratio | |
| 1:2 | Unstable emulsion |
| 1:5 | Stable emulsion, larger particles |
| 1:10 | Stable emulsion, optimal particle size |
| Stirring speed | |
| 16,000 rpm | Larger particles |
| 25,000 rpm | Optimal particle size |
| 35,000 rpm | High dispersity, very small particle size |
| PCL concentration | |
| 0.10% | Larger particles |
| 0.25% | Optimal particle size |
| 0.50% | Larger particles |
| Poloxamer 407 concentration | |
| 2.0% | Unstable emulsion |
| 4.0% | Stable emulsion |
| 6.0% | Stable emulsion |
| Polysorbate 20 concentration | |
| 0.5% | Stable emulsion |
| 1.0% | Stable emulsion |
| 1.5% | Stable emulsion |
| Composition (%) | Sample Code | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| NP1 | NP2 | NP3 | NP4 | NP5 | NP6 | NP7 | NP8 | NP9 | |
| PCL (14 kDa) | 0.25 | - | 0.125 | 0.25 | - | 0.125 | 0.25 | - | 0.125 |
| PCL (80 kDa) | - | 0.25 | 0.125 | - | 0.25 | 0.125 | - | 0.25 | 0.125 |
| Poloxamer 407 | 4.00 | 4.00 | 4.000 | - | - | - | 2.00 | 2.00 | 2.000 |
| Polysorbate 20 | - | - | - | 1.00 | 1.00 | 1.000 | 0.20 | 0.20 | 0.200 |
| Sample Code | Mean Diameter (nm) | PDI | ζ-Potential (mV) |
|---|---|---|---|
| NP1 | 474 ± 250 | 17.73 | −5.3 ± 0.2 |
| NP2 | 628 ± 380 | 22.30 | −7.9 ± 0.9 |
| NP3 | 536 ± 204 | 5.75 | −10.9 ± 0.3 |
| NP4 | 188 ± 61 | 0.84 | −14.5 ± 0.5 |
| NP5 | 201 ± 80 | 1.04 | −16.9 ± 0.2 |
| NP6 | 196 ± 70 | 0.91 | −17.9 ± 0.7 |
| NP7 | 336 ± 150 | 2.43 | −6.7 ± 0.8 |
| NP8 | 398 ± 250 | 3.72 | −8.5 ± 0.5 |
| NP9 | 390 ± 200 | 3.05 | −9.5 ± 0.2 |
| IDB (mg) | IDB (%) | PCL (mg) | PCL (%) | IDB:PCL |
|---|---|---|---|---|
| 10 | 0.2 | 12.5 | 0.25 | 1:1.25 |
| Sample Code | Mean Diameter (nm) | PDI | ζ-Potential (mV) | EE (%) | DL (%) |
|---|---|---|---|---|---|
| NP4-IDB | 195 ± 80 | 0.82 | −13.7 ± 0.3 | 53.22 ± 0.60 | 29.86 ± 0.20 |
| NP5-IDB | 212 ± 90 | 1.11 | −15.3 ± 0.9 | 36.98 ± 0.57 | 22.83 ± 0.22 |
| NP6-IDB | 205 ± 60 | 1.25 | −17.4 ± 0.7 | 34.15 ± 0.23 | 21.45 ± 0.31 |
| Sample Code | Zero Order | First Order | Higuchi | Korsmeyer–Peppas | Hixson–Crowell |
|---|---|---|---|---|---|
| NP4-IDB | 0.9490 | 0.9872 | 0.9985 | 0.9973 | 0.9774 |
| NP5-IDB | 0.9683 | 0.9912 | 0.9986 | 0.9992 | 0.9853 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Boyuklieva, R.; Hristozova, A.; Pilicheva, B. Synthesis and Characterization of PCL-Idebenone Nanoparticles for Potential Nose-to-Brain Delivery. Biomedicines 2023, 11, 1491. https://doi.org/10.3390/biomedicines11051491
Boyuklieva R, Hristozova A, Pilicheva B. Synthesis and Characterization of PCL-Idebenone Nanoparticles for Potential Nose-to-Brain Delivery. Biomedicines. 2023; 11(5):1491. https://doi.org/10.3390/biomedicines11051491
Chicago/Turabian StyleBoyuklieva, Radka, Asya Hristozova, and Bissera Pilicheva. 2023. "Synthesis and Characterization of PCL-Idebenone Nanoparticles for Potential Nose-to-Brain Delivery" Biomedicines 11, no. 5: 1491. https://doi.org/10.3390/biomedicines11051491
APA StyleBoyuklieva, R., Hristozova, A., & Pilicheva, B. (2023). Synthesis and Characterization of PCL-Idebenone Nanoparticles for Potential Nose-to-Brain Delivery. Biomedicines, 11(5), 1491. https://doi.org/10.3390/biomedicines11051491








