Opiate Antagonists for Chronic Pain: A Review on the Benefits of Low-Dose Naltrexone in Arthritis versus Non-Arthritic Diseases
Abstract
1. Introduction and Global Impact of Pain in Medicine
2. Applications of Naltrexone in Opioid Use Disorder and Alcohol Use Disorder
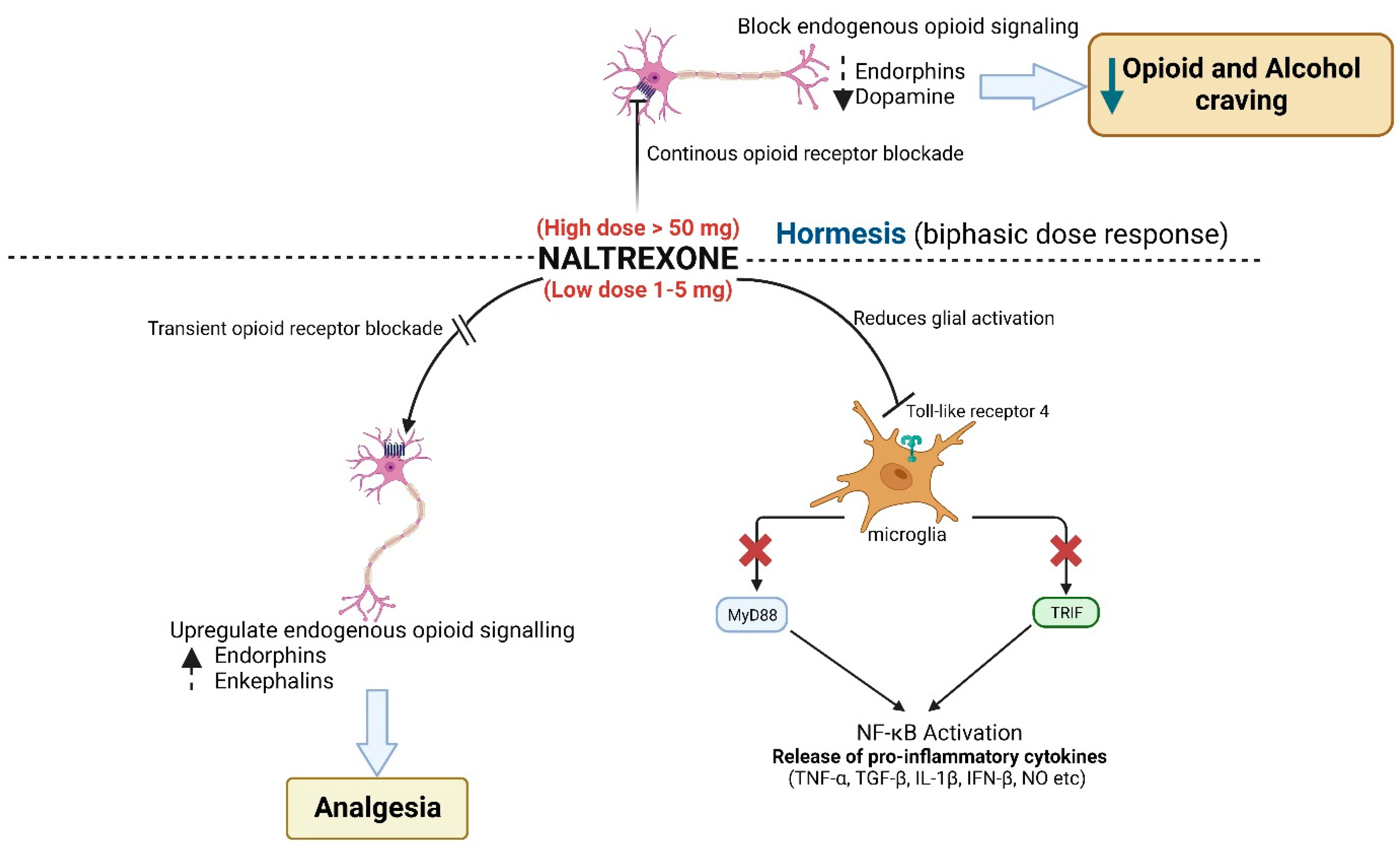
3. Pharmacotherapeutic Effects of Naltrexone Are Biphasic
4. Pharmaco-Kinetics of Naltrexone
5. Physiological Mechanisms of Action of Low-Dose Naltrexone
6. Therapeutic Potential of LDN in the Treatment of Arthritic Diseases
7. Therapeutic Potential of LDN in the Treatment of Non-Arthritic Diseases
8. The Relationship between LDN and Depression in Chronic Conditions
9. Strengths and Limitations
10. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| bid | bis in die (twice a day) |
| FDA | Food and Drug Administration |
| GPCR | G protein-coupled receptor |
| IFNB1 | interferon- β |
| IL1B | interleukin-1β |
| LDN | low-dose naltrexone |
| MS | multiple sclerosis |
| NSAID | non-steroidal anti-inflammatory drug |
| OA | osteoarthritis |
| OPRD1 | Opioid Receptor Delta 1 |
| OPRK1 | Opioid Receptor Kappa 1 |
| OPRM1 | Opioid Receptor Mu 1 (mu-opioid receptor, MOR) |
| qid | quater in die (four times a day) |
| TAC1 | Tachykinin Precursor 1 (Substance P) |
| TLR4 | Toll-like receptor 4 |
| TNF | tumor necrosis factor α |
| Ultra-LDN | ultra-low-dose naltrexone |
References
- Institute of Medicine. Relieving Pain in America: A Blueprint for Transforming Prevention, Care, Education, and Research; The National Academies Press: Washington, DC, USA, 2012; Volume 26. [Google Scholar] [CrossRef]
- Yong, R.J.; Mullins, P.M.; Bhattacharyya, N. Prevalence of chronic pain among adults in the United States. Pain 2022, 163, 332. [Google Scholar] [CrossRef] [PubMed]
- Cohen, S.P.; Vase, L.; Hooten, W.M. Chronic pain: An update on burden, best practices, and new advances. Lancet 2021, 397, 2082–2097. [Google Scholar] [CrossRef] [PubMed]
- Gleber, R.; Vilke, G.M.; Castillo, E.M.; Brennan, J.; Oyama, L.; Coyne, C.J. Trends in emergency physician opioid prescribing practices during the United States opioid crisis. Am. J. Emerg. Med. 2020, 38, 735–740. [Google Scholar] [CrossRef]
- Rosa, J.; Burke, J.R. Changes in opioid therapy use by an interprofessional primary care team: A descriptive study of opioid prescription data. J. Manip. Physiol. Ther. 2021, 44, 186–195. [Google Scholar] [CrossRef] [PubMed]
- Essack, Y.; Stanfliet, J. Opioid abuse. Prof. Nurs. Today 2016, 20, 20–21. [Google Scholar]
- Bennett, C.D. New Jersey’s opiod addiction health crisis. Md Advis. A J. New Jersey Med. Community 2017, 10, 5–6. [Google Scholar]
- Paul, S.M.; Allread, V. Opiod misuse, abuse and addiction part 2: Opiod prescriber responsibilities and resources. Md Advis. A J. New Jersey Med. Community 2017, 10, 4–16. [Google Scholar]
- Gilron, I.; Carr, D.B.; Desjardins, P.J.; Kehlet, H. Current methods and challenges for acute pain clinical trials. PAIN Rep. 2018, 4, e647. [Google Scholar] [CrossRef]
- Bruun, K.D.; Blichfeldt-Eckhardt, M.R.; Vægter, H.B.; Lauridsen, J.T.; Amris, K.; Toft, P. Low-dose naltrexone for the treatment of fibromyalgia: Investigation of dose-response relationships. Pain Med. 2020, 21, 2253–2261. [Google Scholar] [CrossRef]
- Shen, K.-F.; Crain, S.M. Ultra-low doses of naltrexone or etorphine increase morphine’s antinociceptive potency and attenuate tolerance/dependence in mice. Brain Res. 1997, 757, 176–190. [Google Scholar] [CrossRef]
- Shen, K.-F.; Crain, S.M.; Moate, P.; Boston, R.; de Kater, A.W.; Schoenhard, G.L. PTI-801, a novel formulation of oxycodone, shows absence of tolerance, physical dependence and naloxone-precipitated withdrawal effects in mice. J. Pain 2002, 3, 49. [Google Scholar]
- Shen, K.-F.; Crain, S.M.; Moate, P.; Boston, R.; de Kater, A.W.; Schoenhard, G.L. PTI-555, reverses and prevents morphine-induced tolerance and naloxone precipitated withdrawal in mice chronically treated with morphine. J. Pain 2002, 3, 50. [Google Scholar]
- Soin, A.; Soin, Y.; Dann, T.; Buenaventura, R.; Ferguson, K.; Atluri, S.; Sachdeva, H.; Sudarshan, G.; Akbik, H.; Italiano, J. Low-dose naltrexone use for patients with chronic regional pain syndrome: A systematic literature review. Pain Physician 2021, 24, 406. [Google Scholar]
- Larney, S.; Gowing, L.; Mattick, R.P.; Farrell, M.; Hall, W.; Degenhardt, L. A systematic review and meta-analysis of naltrexone implants for the treatment of opioid dependence. Drug Alcohol Rev. 2014, 33, 115–128. [Google Scholar] [CrossRef] [PubMed]
- Krupitsky, E.; Zvartau, E.; Blokhina, E.; Verbitskaya, E.; Wahlgren, V.; Tsoy-Podosenin, M.; Bushara, N.; Burakov, A.; Masalov, D.; Romanova, T.; et al. Anhedonia, depression, anxiety, and craving in opiate dependent patients stabilized on oral naltrexone or an extended release naltrexone implant. Am. J. Drug Alcohol Abus. 2016, 42, 614–620. [Google Scholar] [CrossRef]
- Edinoff, A.N.; Nix, C.A.; Orellana, C.V.; StPierre, S.M.; Crane, E.A.; Bulloch, B.T.; Cornett, E.M.; Kozinn, R.L.; Kaye, A.M.; Murnane, K.S.; et al. Naltrexone Implant for Opioid Use Disorder. Neurol. Int. 2021, 14, 49–61. [Google Scholar] [CrossRef] [PubMed]
- Cant, R.; Dalgleish, A.G.; Allen, R.L. Naltrexone inhibits IL-6 and TNFalpha production in human immune cell subsets following stimulation with ligands for intracellular toll-like receptors. Front. Immunol. 2017, 8, 809. [Google Scholar] [CrossRef]
- Roeckel, L.-A.; Utard, V.; Reiss, D.; Mouheiche, J.; Maurin, H.; Robé, A.; Audouard, E.; Wood, J.N.; Goumon, Y.; Simonin, F.; et al. Morphine-induced hyperalgesia involves mu opioid receptors and the metabolite morphine-3-glucuronide. Sci. Rep. 2017, 7, 10406. [Google Scholar] [CrossRef]
- Younger, J.; Parkitny, L.; McLain, D. The use of low-dose naltrexone (LDN) as a novel anti-inflammatory treatment for chronic pain. Clin. Rheumatol. 2014, 33, 451–459. [Google Scholar] [CrossRef]
- Kim, Y.H.; West, K. Treating chronic pain with low dose naltrexone and ultralow dose naltrexone: A review paper. J. Pain Manag. Ther. 2019, 3, 1. [Google Scholar]
- Afshari, R.; Khadem-Rezaiyan, M.; Moghadam, H.K.; Talebi, M. Very low dose naltrexone in opioid detoxification: A double-blind, randomized clinical trial of efficacy and safety. Toxicol. Res. 2020, 36, 21–27. [Google Scholar] [CrossRef]
- Greig, S.L.; Keating, G.M. Naltrexone ER/Bupropion ER: A Review in Obesity Management. Drugs 2015, 75, 1269–1280. [Google Scholar] [CrossRef]
- Sudakin, D. Naltrexone: Not just for opioids anymore. J. Med. Toxicol. 2015, 12, 71–75. [Google Scholar] [CrossRef] [PubMed]
- Krieter, P.; Gyaw, S.; Chiang, C.N.; Crystal, R.; Skolnick, P. Enhanced intranasal absorption of naltrexone by dodecyl maltopyranoside: Implications for the treatment of opioid overdose. J. Clin. Pharmacol. 2019, 59, 947–957. [Google Scholar] [CrossRef]
- Kim, P.S.; Fishman, M.A. Low-dose naltrexone for chronic pain: Update and systemic review. Curr. Pain Headache Rep. 2020, 24, 64. [Google Scholar] [CrossRef]
- Colomer-Carbonell, A.; Sanabria-Mazo, J.P.; Hernández-Negrín, H.; Borràs, X.; Suso-Ribera, C.; García-Palacios, A.; Muchart, J.; Munuera, J.; D’Amico, F.; Maes, M.; et al. Study protocol for a randomised, double-blinded, placebo-controlled phase III trial examining the add-on efficacy, cost–utility and neurobiological effects of low-dose naltrexone (LDN) in patients with fibromyalgia (INNOVA study). BMJ Open 2022, 12, e055351. [Google Scholar] [CrossRef] [PubMed]
- Jackson, D.; Singh, S.; Zhang-James, Y.; Faraone, S.; Johnson, B. The effects of low dose naltrexone on opioid induced hyperalgesia and fibromyalgia. Front. Psychiatry 2021, 12, 136. [Google Scholar] [CrossRef]
- Rivera, V.; DeCicco, J.; Espiritu, S. Proceedings# 47: Low dose naltrexone: A viable alternative for long term chronic pain? Brain Stimul. Basic Transl. Clin. Res. Neuromodulation 2019, 12, e118–e120. [Google Scholar] [CrossRef]
- Andreesen, J.; Svoboda, A.; Fiacp, P.D. Low dose naltrexone in dermatology. J. Drugs Dermatol. 2019, 18, 235–238. [Google Scholar]
- Wahid, H.H.; Chin, P.Y.; Sharkey, D.J.; Diener, K.R.; Hutchinson, M.R.; Rice, K.C.; Moldenhauer, L.M.; Robertson, S.A. Toll-like receptor-4 antagonist (+)-naltrexone protects against carbamyl-platelet activating factor (cPAF)-induced preterm labor in mice. Am. J. Pathol. 2020, 190, 1030–1045. [Google Scholar] [CrossRef]
- Monnet, E.; Choy, E.H.; McInnes, I.; Kobakhidze, T.; De Graaf, K.; Jacqmin, P.; Lapeyre, G.; De Min, C. Efficacy and safety of NI-0101, an anti-toll-like receptor 4 monoclonal antibody, in patients with rheumatoid arthritis after inadequate response to methotrexate: A phase II study. Ann. Rheum. Dis. 2020, 79, 316–323. [Google Scholar] [CrossRef] [PubMed]
- Cattaneo, I.; Kayser, V.; Guilbaud, G. Differential effects of specific δ and κ opioid receptor antagonists on the bidirectional dose-dependent effect of systemic naloxone in arthritic rats, an experimental model of persistent pain. Brain Res. 1993, 623, 201–207. [Google Scholar] [CrossRef] [PubMed]
- Desmeules, J.; Kayser, V.; Gacel, G.; Guilbaud, G.; Roques, B. The highly selective δ agonist BUBU induces an analgesic effect in normal and arthritic rat and this action is not affected by repeated administration of low doses of morphine. Brain Res. 1993, 611, 243–248. [Google Scholar] [CrossRef]
- Mayo Foundation for Medical Education and Research. (18 May 2021). Rheumatoid arthritis. MayoClinic. Available online: https://www.mayoclinic.org/diseases-conditions/rheumatoid-arthritis/symptoms-causes/syc-20353648 (accessed on 10 December 2022).
- WebMD. (n.d.). Types of Rheumatoid arthritis: Seropositive RA vs. Seronegative Ra. WebMD. Available online: https://www.webmd.com/rheumatoid-arthritis/rheumatoid-arthritis-types#:~:text=Having%20seropositive%20RA%20means%20your,if%20you%20have%20anti%2DCCPs (accessed on 10 December 2022).
- Raknes, G.; Småbrekke, L. Low dose naltrexone: Effects on medication in rheumatoid and seropositive arthritis. A nationwide register-based controlled quasi-experimental before-after study. PLoS ONE 2019, 14, e0212460. [Google Scholar] [CrossRef]
- Mannelli, P.; Patkar, A.; Peindl, K.; Gottheil, E.; Wu, L.-T.; Gorelick, D. Early outcomes following low dose naltrexone enhancement of opioid detoxification. Am. J. Addict. 2009, 18, 109–116. [Google Scholar] [CrossRef]
- Webster, L.R.; Butera, P.G.; Moran, L.V.; Wu, N.; Burns, L.H.; Friedmann, N. Oxytrex minimizes physical dependence while providing effective analgesia: A randomized controlled trial in low back pain. J. Pain 2006, 7, 937–946. [Google Scholar] [CrossRef]
- Partridge, S.; Quadt, L.; Bolton, M.; Eccles, J.; Thompson, C.; Colasanti, A.; Bremner, S.; Jones, C.I.; Bruun, K.D.; Van Marwijk, H. A systematic literature review on the clinical efficacy of low dose naltrexone and its effect on putative pathophysiological mechanisms among patients diagnosed with fibromyalgia. Heliyon 2023, 9, e15638. [Google Scholar] [CrossRef]
- Naltrexone lowers hospital admissions in alcohol use disorder. Practitioner 2021, 1852, 9.
- Patten, D.K.; Schultz, B.G.; Berlau, D.J. The safety and efficacy of low-dose naltrexone in the management of chronic pain and inflammation in multiple sclerosis, fibromyalgia, crohn’s disease, and other chronic pain disorders. Pharmacother. J. Hum. Pharmacol. Drug Ther. 2018, 38, 382–389. [Google Scholar] [CrossRef]
- National Multiple Sclerosis Society. What Is MS? Available online: www.nationalmssociety.org/about-multiple-sclerosis/index.aspx (accessed on 5 August 2016).
- Leddy, S.; Dobson, R. Multiple sclerosis. Medicine 2020, 48, 588–594. [Google Scholar] [CrossRef]
- Katsavos, S.; Anagnostouli, M. Biomarkers in Multiple Sclerosis: An Up-to-Date Overview. Mult. Scler. Int. 2013, 2013, 20. [Google Scholar] [CrossRef]
- Turel, A.P.; Oh, K.H.; Zagon, I.S.; McLaughlin, P.J. Low dose naltrexone for treatment of multiple sclerosis: A retrospective chart review of safety and tolerability (Letter). J. Clin. Psychopharmacol. 2015, 35, 609–611. [Google Scholar] [CrossRef] [PubMed]
- Codino, H.; Hardin, A. Low Dose Naltrexone in Conjunction with the Wahls Protocol to Reduce the Frequency of Chronic Migraines in a Patient with Multiple Sclerosis: A Case Study. Integr. Med. 2021, 20, 30–34. [Google Scholar]
- Rosenblum, M.D.; Remedios, K.A.; Abbas, A.K. Mechanisms of human autoimmunity. J. Clin. Investig. 2015, 125, 2228–2233. [Google Scholar] [CrossRef] [PubMed]
- Ludwig, M.D.; Zagon, I.S.; McLaughlin, P.J. Featured Article: Serum [Met5]-enkephalin levels are reduced in multiple sclerosis and restored by low-dose naltrexone. Exp. Biol. Med. 2017, 242, 1524–1533. [Google Scholar] [CrossRef]
- Smith, J.P.; Stock, H.; Bingaman, S.; Mauger, D.; Rogosnitzky, M.; Zagon, I.S. Low-dose naltrexone therapy improves active Crohn’s disease. Am. J. Gastroenterol. 2007, 102, 820–828. [Google Scholar] [CrossRef]
- Lie, M.R.K.L.; Van Der Giessen, J.; Fuhler, G.M.; De Lima, A.; Peppelenbosch, M.P.; Van Der Ent, C.; Van Der Woude, C.J. Low dose Naltrexone for induction of remission in inflammatory bowel disease patients. J. Transl. Med. 2018, 16, 55. [Google Scholar] [CrossRef]
- Segal, D.; Macdonald, J.K.; Chande, N. Low dose naltrexone for induction of remission in Crohn’s disease. Cochrane Database Syst. Rev. 2014, 2, 010410. [Google Scholar] [CrossRef]
- Sharma, A.; Kudesia, P.; Shi, Q.; Gandhi, R. Anxiety and depression in patients with osteoarthritis: Impact and management challenges. Open Access Rheumatol. Res. Rev. 2016, 8, 103–113. [Google Scholar] [CrossRef]
- Doan, L.; Manders, T.; Wang, J. Neuroplasticity Underlying the Comorbidity of Pain and Depression. Neural Plast. 2015, 2015, 1–16. [Google Scholar] [CrossRef]
- Vallerand, I.A.; Patten, S.B.; Barnabe, C. Depression and the risk of rheumatoid arthritis. Curr. Opin. Rheumatol. 2019, 31, 279–284. [Google Scholar] [CrossRef] [PubMed]
- Mischoulon, D.; Hylek, L.; Yeung, A.S.; Clain, A.J.; Baer, L.; Cusin, C.; Ionescu, D.F.; Alpert, J.E.; Soskin, D.P.; Fava, M. Randomized, proof-of-concept trial of low dose naltrexone for patients with breakthrough symptoms of major depressive disorder on antidepressants. J. Affect. Disord. 2017, 208, 6–14. [Google Scholar] [CrossRef] [PubMed]
- Siembida, J.; Johnson, B. Depression in fibromyalgia patients may require low-dose naltrexone to respond: A case report. Cureus 2022, 14, 22677. [Google Scholar] [CrossRef] [PubMed]
- McLaughlin, P.J.; Odom, L.B.; Arnett, P.A.; Orehek, S.; Thomas, G.A.; Zagon, I.S. Low-dose naltrexone reduced anxiety in persons with multiple sclerosis during the COVID-19 pandemic. Int. Immunopharmacol. 2022, 113 Pt B, 109438. [Google Scholar] [CrossRef]
| Naltrexone Dosage | Clinical Uses |
|---|---|
| 50–300 mg (regular dose) | Opiate, Alcohol addiction |
| 1–5 mg (LDN) | Fibromyalgia, arthritis, Crohn’s disease, multiple sclerosis |
| 0.001–1 mg very low-dose naltrexone (VLDN) | Reduced side effects and withdrawal symptoms of opiates |
| <0.001 mg (ULDN) | Similar to VLDN, OA, low back pain, postoperative pain control |
| Disease | Characteristics of the Study | LDN Benefits |
|---|---|---|
| RA and seropositive arthritis | Quasi-experimental study with controlled before and after comparisons | Reduced dispensing of medications (antirheumatic agents and NSAIDS) |
| OA | A clinical study with Oxytrex (opiate+LDN) with a naltrexone dose of 0.004 mg | Reduction in pain and less dependence compared to opioids |
| Number of Participants | Length of Study, and Dosage | Outcomes of Study | Reference |
|---|---|---|---|
| 17 participants | 12 weeks, 4.5 mg naltrexone/day | Crohn’s disease activity index (CDAI) scores decreased significantly; 67% achieved remission (p < 0.001). | Smith [50] |
| 47 participants | 12 weeks, 4.5 mg naltrexone/day | LDN led to clinical improvement in 74.5% and remission in 25.5% of patients. Naltrexone improved epithelial barrier function by improving wound healing. | Lie [51] |
| Two studies were evaluated:34 adult patients,12 pediatric patients | 12 weeks, 4.5 mg naltrexone/day 0.1 mg/kg up to 4.5 mg | LDN was safe and had minimal adverse effects. | Segal [52] |
| Disease | Characteristics of the Study | LDN Benefits |
|---|---|---|
| Fibromyalgia | LDN doses of 3.88 mg or 5.40 mg were used. | Reduces pain intensity and quality |
| Multiple sclerosis | LDN dose of 4.5 mg was used in one study. In another study, inhibitory peptides were indirectly measured as a way of assessing efficacy. | Reduces suppression of lymphocytes and improves quality of life |
| Crohn’s disease | LDN dosage of 4.5 mg naltrexone/day. | Wound healing, minimal adverse effects, improved symptoms |
| Depression and anxiety | Small-scale studies based on subjective assessments for patients with treatment regimens supplemented with 1mg LDN (bid). | Decreased levels of depression and anxiety |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dara, P.; Farooqui, Z.; Mwale, F.; Choe, C.; van Wijnen, A.J.; Im, H.-J. Opiate Antagonists for Chronic Pain: A Review on the Benefits of Low-Dose Naltrexone in Arthritis versus Non-Arthritic Diseases. Biomedicines 2023, 11, 1620. https://doi.org/10.3390/biomedicines11061620
Dara P, Farooqui Z, Mwale F, Choe C, van Wijnen AJ, Im H-J. Opiate Antagonists for Chronic Pain: A Review on the Benefits of Low-Dose Naltrexone in Arthritis versus Non-Arthritic Diseases. Biomedicines. 2023; 11(6):1620. https://doi.org/10.3390/biomedicines11061620
Chicago/Turabian StyleDara, Praneet, Zeba Farooqui, Fackson Mwale, Chungyoul Choe, Andre J. van Wijnen, and Hee-Jeong Im. 2023. "Opiate Antagonists for Chronic Pain: A Review on the Benefits of Low-Dose Naltrexone in Arthritis versus Non-Arthritic Diseases" Biomedicines 11, no. 6: 1620. https://doi.org/10.3390/biomedicines11061620
APA StyleDara, P., Farooqui, Z., Mwale, F., Choe, C., van Wijnen, A. J., & Im, H.-J. (2023). Opiate Antagonists for Chronic Pain: A Review on the Benefits of Low-Dose Naltrexone in Arthritis versus Non-Arthritic Diseases. Biomedicines, 11(6), 1620. https://doi.org/10.3390/biomedicines11061620







