Matrix Metalloproteinases in Oral Health—Special Attention on MMP-8
Abstract
1. Introduction
2. MMP-8
3. MMPs and MMP-8 in Periodontal Diseases
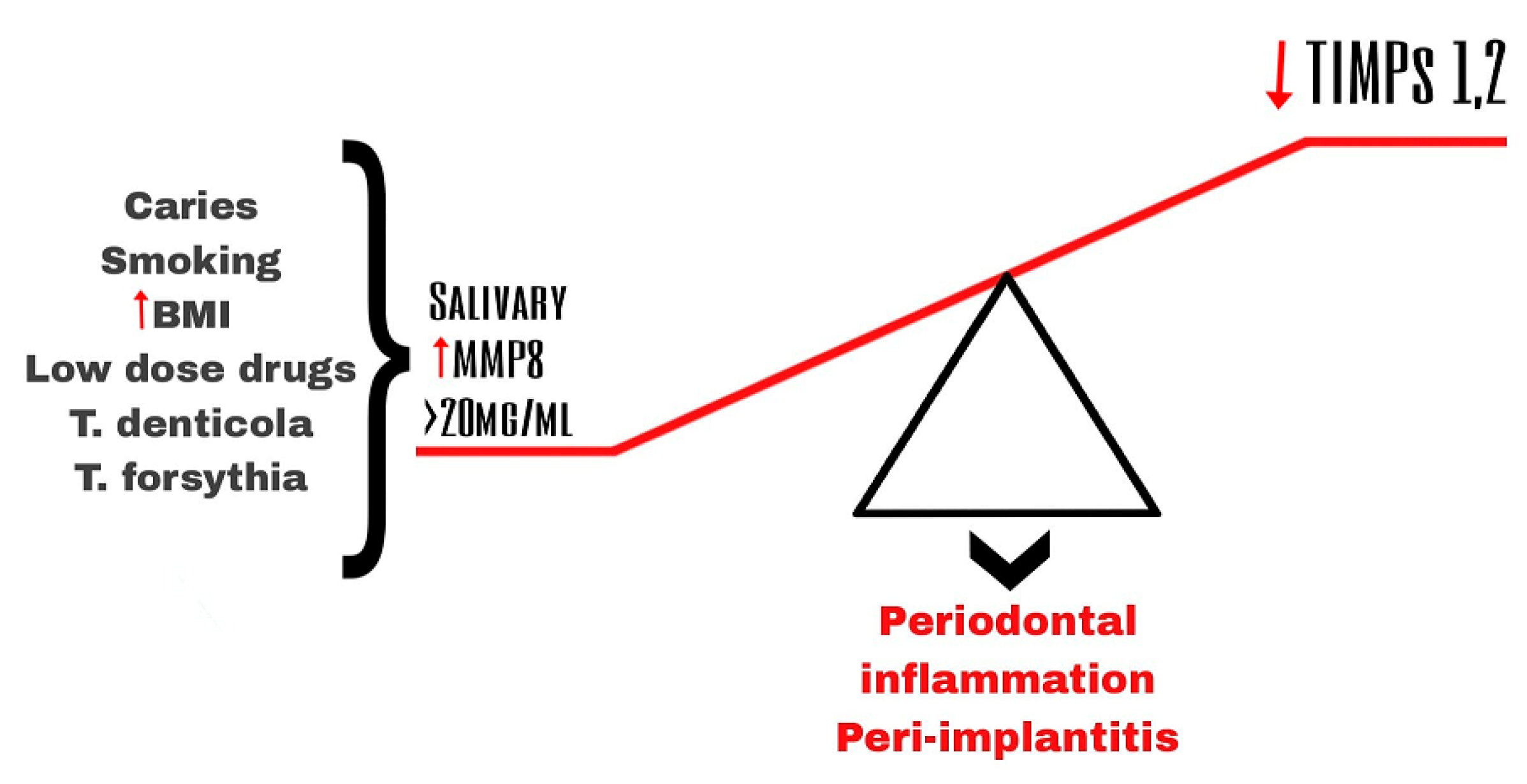
4. MMPs and MMP-8 in Peri-Implantitis
5. MMPs and MMP-8 in Carious Lesions
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Schubert-Unkmeir, A.; Konrad, C.; Slanina, H.; Czapek, F.; Hebling, S.; Frosch, M. Neisseria meningitidis induces brain microvascular endothelial cell detachment from the matrix and cleavage of occludin: A role for MMP-8. PLoS Pathog. 2010, 6, e1000874. [Google Scholar] [CrossRef] [PubMed]
- Checchi, V.; Maravic, T.; Bellini, P.; Generali, L.; Consolo, U.; Breschi, L.; Mazzoni, A. The Role of Matrix Metalloproteinases in Periodontal Disease. Int. J. Environ. Res. Public. Health 2020, 17, 4923. [Google Scholar] [CrossRef] [PubMed]
- Gul, S.S.; Abdulkareem, A.A.; Sha, A.M.; Rawlinson, A. Diagnostic Accuracy of Oral Fluids Biomarker Profile to Determine the Current and Future Status of Periodontal and Peri-Implant Diseases. Diagnostics 2020, 10, 838. [Google Scholar] [CrossRef] [PubMed]
- Luchian, I.; Goriuc, A.; Sandu, D.; Covasa, M. The Role of Matrix Metalloproteinases (MMP-8, MMP-9, MMP-13) in Periodontal and Peri-Implant Pathological Processes. Int. J. Mol. Sci. 2022, 23, 1806. [Google Scholar] [CrossRef] [PubMed]
- Franco, C.; Patricia, H.R.; Timo, S.; Claudia, B.; Marcela, H. Matrix Metalloproteinases as Regulators of Periodontal Inflammation. Int. J. Mol. Sci. 2017, 18, 440. [Google Scholar] [CrossRef]
- Wang, X.; Rojas-Quintero, J.; Wilder, J.; Tesfaigzi, Y.; Zhang, D.; Owen, C.A. Tissue Inhibitor of Metalloproteinase-1 Promotes Polymorphonuclear Neutrophil (PMN) Pericellular Proteolysis by Anchoring Matrix Metalloproteinase-8 and -9 to PMN Surfaces. J. Immunol. 2019, 202, 3267–3281. [Google Scholar] [CrossRef]
- Goldberg, M. Dental Tissues Remodeling by Matrix Metalloproteinases (MMPS) and Tissues Inhibitors of MMPS (TIMPS). SVOA Dent. 2022, 3, 203–212. [Google Scholar]
- Makela, M.; Salo, T.; Uitto, V.J.; Larjava, H. Matrix metalloproteinases (MMP-2 and MMP-9) of the oral cavity: Cellular origin and relationship to periodontal status. J. Dent. Res. 1994, 73, 1397–1406. [Google Scholar] [CrossRef]
- Tervahartiala, T.; Pirila, E.; Ceponis, A.; Maisi, P.; Salo, T.; Tuter, G.; Kallio, P.; Tornwall, J.; Srinivas, R.; Konttinen, Y.T.; et al. The in vivo expression of the collagenolytic matrix metalloproteinases (MMP-2, -8, -13, and -14) and matrilysin (MMP-7) in adult and localized juvenile periodontitis. J. Dent. Res. 2000, 79, 1969–1977. [Google Scholar] [CrossRef]
- Wang, H.; Parry, S.; Macones, G.; Sammel, M.D.; Ferrand, P.E.; Kuivaniemi, H.; Tromp, G.; Halder, I.; Shriver, M.D.; Romero, R.; et al. Functionally significant SNP MMP8 promoter haplotypes and preterm premature rupture of membranes (PPROM). Hum. Mol. Genet. 2004, 13, 2659–2669. [Google Scholar] [CrossRef]
- Quintero, P.A.; Knolle, M.D.; Cala, L.F.; Zhuang, Y.; Owen, C.A. Matrix metalloproteinase-8 inactivates macrophage inflammatory protein-1 alpha to reduce acute lung inflammation and injury in mice. J. Immunol. 2010, 184, 1575–1588. [Google Scholar] [CrossRef] [PubMed]
- Pollanen, M.T.; Salonen, J.I.; Uitto, V.J. Structure and function of the tooth-epithelial interface in health and disease. Periodontology 2000, 31, 12–31. [Google Scholar] [CrossRef] [PubMed]
- Luo, K.W.; Wei, C.; Lung, W.Y.; Wei, X.Y.; Cheng, B.H.; Cai, Z.M.; Huang, W.R. EGCG inhibited bladder cancer SW780 cell proliferation and migration both in vitro and in vivo via down-regulation of NF-κB and MMP-9. J. Nutr. Biochem. 2017, 41, 56–64. [Google Scholar] [CrossRef]
- Charles, K.; Honibald, E.N.; Reddy, N.R.; Palani, A.; Ramamurthy, R.D.; Sankaralingam, T. Role of matrix metalloproteinases (MMPS) in periodontitis and its management. J. Indian Acad. Dent. Spec. Res. 2014, 1, 65–69. [Google Scholar]
- Ye, S. Polymorphism in matrix metalloproteinase gene promoters: Implication in regulation of gene expression and susceptibility of various diseases. Matrix Biol. 2000, 19, 623–629. [Google Scholar] [CrossRef]
- Djuric, T.; Zivkovic, M. Overview of MMP biology and gene associations in human diseases. In The Role of Matrix Metalloproteinase in Human Body Pathologies; IntechOpen: London, UK, 2017. [Google Scholar]
- Li, W.; Zhu, Y.; Singh, P.; Ajmera, D.H.; Song, J.; Ji, P. Association of Common Variants in MMPs with Periodontitis Risk. Dis. Markers 2016, 2016, 1545974. [Google Scholar] [CrossRef]
- Emingil, G.; Han, B.; Gurkan, A.; Berdeli, A.; Tervahartiala, T.; Salo, T.; Pussinen, P.J.; Kose, T.; Atilla, G.; Sorsa, T. Matrix metalloproteinase (MMP)-8 and tissue inhibitor of MMP-1 (TIMP-1) gene polymorphisms in generalized aggressive periodontitis: Gingival crevicular fluid MMP-8 and TIMP-1 levels and outcome of periodontal therapy. J. Periodontol. 2014, 85, 1070–1080. [Google Scholar] [CrossRef]
- Maghajothi, S.; Subramanian, L.; Mani, P.; Singh, M.; Iyer, D.R.; Sharma, S.; Khullar, M.; Victor, S.M.; Asthana, S.; Mullasari, A.S.; et al. A common Matrix metalloproteinase 8 promoter haplotype enhances the risk for hypertension via diminished interactions with nuclear factor kappa B. J. Hypertens. 2022, 40, 2147–2160. [Google Scholar] [CrossRef]
- Zhao, Y.; Zhang, Q.; Zhang, X.; Zhang, Y.; Lu, Y.; Ma, X.; Li, W.; Niu, X.; Zhang, G.; Chang, M.; et al. The roles of MMP8/MMP10 polymorphisms in ischemic stroke susceptibility. Brain Behav. 2022, 12, e2797. [Google Scholar] [CrossRef]
- Lee, M.R.; Chen, Y.L.; Wu, C.W.; Chen, L.C.; Chang, L.Y.; Chen, J.Y.; Huang, Y.T.; Wang, J.Y.; Shih, J.Y.; Yu, C.J. Toll-like receptor and matrix metalloproteinase single-nucleotide polymorphisms, haplotypes, and polygenic risk score differentiated between tuberculosis disease and infection. Int. J. Infect. Dis. 2022, 125, 61–66. [Google Scholar] [CrossRef]
- Juurikka, K.; Butler, G.S.; Salo, T.; Nyberg, P.; Astrom, P. The Role of MMP8 in Cancer: A Systematic Review. Int. J. Mol. Sci. 2019, 20, 4506. [Google Scholar] [CrossRef] [PubMed]
- Decock, J.; Long, J.R.; Laxton, R.C.; Shu, X.O.; Hodgkinson, C.; Hendrickx, W.; Pearce, E.G.; Gao, Y.T.; Pereira, A.C.; Paridaens, R.; et al. Association of matrix metalloproteinase-8 gene variation with breast cancer prognosis. Cancer Res. 2007, 67, 10214–10221. [Google Scholar] [CrossRef] [PubMed]
- Tai, J.; Sun, D.; Wang, X.; Kang, Z. Matrix metalloproteinase-8 rs11225395 polymorphism correlates with colorectal cancer risk and survival in a Chinese Han population: A case-control study. Aging 2020, 12, 19618–19627. [Google Scholar] [CrossRef] [PubMed]
- Rella, J.M.; Jilma, B.; Fabry, A.; Kaynar, A.M.; Mayr, F.B. MMP-8 genotypes influence the inflammatory response in human endotoxemia. Inflammation 2014, 37, 451–456. [Google Scholar] [CrossRef] [PubMed]
- Shimoda, T.; Obase, Y.; Kishikawa, R.; Iwanaga, T. Association of matrix metalloproteinase 8 genetic polymorphisms with bronchial asthma in a Japanese population. Allergy Rhinol. 2013, 4, e132–e139. [Google Scholar] [CrossRef] [PubMed]
- Milaras, C.; Lepetsos, P.; Dafou, D.; Potoupnis, M.; Tsiridis, E. Association of Matrix Metalloproteinase (MMP) Gene Polymorphisms With Knee Osteoarthritis: A Review of the Literature. Cureus 2021, 13, e18607. [Google Scholar] [CrossRef] [PubMed]
- Heikkinen, A.M.; Kettunen, K.; Kovanen, L.; Haukka, J.; Elg, J.; Husu, H.; Tervahartiala, T.; Pussinen, P.; Meurman, J.; Sorsa, T. Inflammatory mediator polymorphisms associate with initial periodontitis in adolescents. Clin. Exp. Dent. Res. 2016, 2, 208–215. [Google Scholar] [CrossRef]
- Weng, H.; Yan, Y.; Jin, Y.H.; Meng, X.Y.; Mo, Y.Y.; Zeng, X.T. Matrix metalloproteinase gene polymorphisms and periodontitis susceptibility: A meta-analysis involving 6162 individuals. Sci. Rep. 2016, 6, 24812. [Google Scholar] [CrossRef]
- Majumder, P.; Ghosh, S.; Dey, S.K. Matrix metalloproteinase gene polymorphisms in chronic periodontitis: A case-control study in the Indian population. J. Genet. 2019, 98, 32. [Google Scholar] [CrossRef]
- Putri, H.; Sulijaya, B.; Hartomo, B.T.; Suhartono, A.W.; Auerkari, E.I. + 17 C/G polymorphism in matrix metalloproteinase (MMP)-8 gene and its association with periodontitis. J. Stomatol. 2020, 73, 154–158. [Google Scholar] [CrossRef]
- Costa-Junior, F.R.; Alvim-Pereira, C.C.; Alvim-Pereira, F.; Trevilatto, P.C.; de Souza, A.P.; Santos, M.C. Influence of MMP-8 promoter polymorphism in early osseointegrated implant failure. Clin. Oral. Investig. 2013, 17, 311–316. [Google Scholar] [CrossRef] [PubMed]
- Chapple, I.L.C.; Mealey, B.L.; Van Dyke, T.E.; Bartold, P.M.; Dommisch, H.; Eickholz, P.; Geisinger, M.L.; Genco, R.J.; Glogauer, M.; Goldstein, M.; et al. Periodontal health and gingival diseases and conditions on an intact and a reduced periodontium: Consensus report of workgroup 1 of the 2017 World Workshop on the Classification of Periodontal and Peri-Implant Diseases and Conditions. J. Periodontol. 2018, 89, S74–S84. [Google Scholar] [CrossRef] [PubMed]
- de Morais, E.F.; Pinheiro, J.C.; Leite, R.B.; Santos, P.P.A.; Barboza, C.A.G.; Freitas, R.A. Matrix metalloproteinase-8 levels in periodontal disease patients: A systematic review. J. Periodontal Res. 2018, 53, 156–163. [Google Scholar] [CrossRef] [PubMed]
- Letra, A.; Silva, R.M.; Rylands, R.J.; Silveira, E.M.; de Souza, A.P.; Wendell, S.K.; Garlet, G.P.; Vieira, A.R. MMP3 and TIMP1 variants contribute to chronic periodontitis and may be implicated in disease progression. J. Clin. Periodontol. 2012, 39, 707–716. [Google Scholar] [CrossRef]
- Yakob, M.; Kari, K.; Tervahartiala, T.; Sorsa, T.; Soder, P.; Meurman, J.H.; Soder, B. Associations of periodontal microorganisms with salivary proteins and MMP-8 in gingival crevicular fluid. J. Clin. Periodontol. 2012, 39, 256–263. [Google Scholar] [CrossRef] [PubMed]
- Yakob, M.; Meurman, J.H.; Sorsa, T.; Söder, B. Treponema denticola associates with increased levels of MMP-8 and MMP-9 in gingival crevicular fluid. Oral. Dis. 2013, 19, 694–701. [Google Scholar] [CrossRef]
- Sapna, G.; Gokul, S.; Bagri-Manjrekar, K. Matrix metalloproteinases and periodontal diseases. Oral. Dis. 2014, 20, 538–550. [Google Scholar] [CrossRef]
- Emingil, G.; Atilla, G.; Sorsa, T.; Luoto, H.; Kirilmaz, L.; Baylas, H. The effect of adjunctive low-dose doxycycline therapy on clinical parameters and gingival crevicular fluid matrix metalloproteinase-8 levels in chronic periodontitis. J. Periodontol. 2004, 75, 106–115. [Google Scholar] [CrossRef]
- Konopka, L.; Pietrzak, A.; Brzezinska-Blaszczyk, E. Effect of scaling and root planing on interleukin-1β, interleukin-8 and MMP-8 levels in gingival crevicular fluid from chronic periodontitis patients. J. Periodontal. Res. 2012, 47, 681–688. [Google Scholar] [CrossRef]
- Honibald, E.N.; Mathew, S.; Padmanaban, J.; Sundaram, E.; Ramamoorthy, R.D. Perioceutics: Matrix metalloproteinase inhibitors as an adjunctive therapy for inflammatory periodontal disease. J. Pharm. Bioallied Sci. 2012, 4, 0975–7406. [Google Scholar] [CrossRef]
- Sorsa, T.; Gursoy, U.K.; Nwhator, S.; Hernandez, M.; Tervahartiala, T.; Leppilahti, J.; Gursoy, M.; Könönen, E.; Emingil, G.; Pussinen, P.J.; et al. Analysis of matrix metalloproteinases, especially MMP-8, in gingival creviclular fluid, mouthrinse and saliva for monitoring periodontal diseases. Periodontology 2000, 70, 142–163. [Google Scholar] [CrossRef] [PubMed]
- da Silva, M.K.; de Carvalho, A.C.G.; Alves, E.H.P.; da Silva, F.R.P.; Pessoa, L.D.S.; Vasconcelos, D.F.P. Genetic Factors and the Risk of Periodontitis Development: Findings from a Systematic Review Composed of 13 Studies of Meta-Analysis with 71,531 Participants. Int. J. Dent. 2017, 2017, 1914073. [Google Scholar] [CrossRef] [PubMed]
- Chou, Y.H.; Ho, Y.P.; Lin, Y.C.; Hu, K.F.; Yang, Y.H.; Ho, K.Y.; Wu, Y.M.; Hsi, E.; Tsai, C.C. MMP-8 -799 C>T genetic polymorphism is associated with the susceptibility to chronic and aggressive periodontitis in Taiwanese. J. Clin. Periodontol. 2011, 38, 1078–1084. [Google Scholar] [CrossRef] [PubMed]
- Izakovicova Holla, L.; Hrdlickova, B.; Vokurka, J.; Fassmann, A. Matrix metalloproteinase 8 (MMP8) gene polymorphisms in chronic periodontitis. Arch. Oral. Biol. 2012, 57, 188–196. [Google Scholar] [CrossRef]
- Goncalves, P.F.; Huang, H.; McAninley, S.; Alfant, B.; Harrison, P.; Aukhil, I.; Walker, C.; Shaddox, L.M. Periodontal treatment reduces matrix metalloproteinase levels in localized aggressive periodontitis. J. Periodontol. 2013, 84, 1801–1808. [Google Scholar] [CrossRef]
- Cifcibasi, E.; Kantarci, A.; Badur, S.; Issever, H.; Cintan, S. Impact of metronidazole and amoxicillin combination on matrix metalloproteinases-1 and tissue inhibitors of matrix metalloproteinases balance in generalized aggressive periodontitis. Eur. J. Dent. 2015, 9, 53–59. [Google Scholar] [CrossRef]
- Luizon, M.R.; de Almeida Belo, V. Matrix metalloproteinase (MMP)-2 and MMP-9 polymorphisms and haplotypes as disease biomarkers. Biomarkers 2012, 17, 286–288. [Google Scholar] [CrossRef]
- Renvert, S.; Persson, G.R.; Pirih, F.Q.; Camargo, P.M. Peri-implant health, peri-implant mucositis, and peri-implantitis: Case definitions and diagnostic considerations. J. Periodontol. 2018, 2018, S304–S312. [Google Scholar] [CrossRef]
- Heitz-Mayfield, L.J.A.; Salvi, G.E. Peri-implant mucositis. J. Periodontol. 2018, 2018, S257–S266. [Google Scholar] [CrossRef]
- Prathapachandran, J.; Suresh, N. Management of peri-implantitis. Dent. Res. J. 2012, 9, 516–521. [Google Scholar] [CrossRef]
- Tonetti, M.S.; Greenwell, H.; Kornman, K.S. Staging and grading of periodontitis: Framework and proposal of a new classification and case definition. J. Periodontol. 2018, 89, S159–S172. [Google Scholar] [CrossRef] [PubMed]
- Kinney, J.S.; Morelli, T.; Oh, M.; Braun, T.M.; Ramseier, C.A.; Sugai, J.V.; Giannobile, W.V. Crevicular fluid biomarkers and periodontal disease progression. J. Clin. Periodontol. 2014, 41, 113–120. [Google Scholar] [CrossRef] [PubMed]
- Taylor, J.J.; Preshaw, P.M. Gingival crevicular fluid and saliva. Periodontology 2000, 70, 7–10. [Google Scholar] [CrossRef]
- Basegmez, C.; Yalcin, S.; Yalcin, F.; Ersanli, S.; Mijiritsky, E. Evaluation of periimplant crevicular fluid prostaglandin E2 and matrix metalloproteinase-8 levels from health to periimplant disease status: A prospective study. Implant. Dent. 2012, 21, 306–310. [Google Scholar] [CrossRef] [PubMed]
- Thierbach, R.; Maier, K.; Sorsa, T.; Mentyla, P. Peri-Implant Sulcus Fluid (PISF) Matrix Metalloproteinase (MMP)-8 Levels in Peri-Implantitis. J. Clin. Diagn. Res. 2016, 10, ZC34–ZC38. [Google Scholar] [CrossRef] [PubMed]
- Dursun, E.; Tozum, T.F. Peri-Implant Crevicular Fluid Analysis, Enzymes and Biomarkers: A Systemetic Review. J. Oral. Maxillofac. Res. 2016, 7, e9. [Google Scholar] [CrossRef]
- Ghassib, I.; Chen, Z.; Zhu, J.; Wang, H.L. Use of IL-1b, IL-6, TNF-a, and MMP-8 biomarkers to distinguish peri-implant diseases: A systematic review and meta-analysis. Clin. Implant. Dent. Relat. Res. 2019, 21, 190–207. [Google Scholar] [CrossRef]
- Sorsa, T.; Tervahartiala, T.; Leppilahti, J.; Hernandez, M.; Gamonal, J.; Tuomainen, A.M.; Lauhio, A.; Pussinen, P.J.; Mäntylä, P. Collagenase-2 (MMP-8) as a point-of-care biomarker in periodontitis and cardiovascular diseases. Therapeutic response to non-antimicrobial properties of tetracyclines. Pharmacol. Res. 2011, 63, 108–113. [Google Scholar] [CrossRef]
- Alassiri, S.; Parnanen, P.; Rathnayake, N.; Johannsen, G.; Heikkinen, A.M.; Lazzara, R.; van der Schoor, P.; van der Schoor, J.G.; Tervahartiala, T.; Gieselmann, D.; et al. The Ability of Quantitative, Specific, and Sensitive Point-of-Care/Chair-Side Oral Fluid Immunotests for aMMP-8 to Detect Periodontal and Peri-Implant Diseases. Dis. Markers 2018, 2018, 1306396. [Google Scholar] [CrossRef]
- Hedenbjork-Lager, A.; Bjorndal, L.; Gustafsson, A.; Sorsa, T.; Tjaderhane, L.; Akerman, S.; Ericson, D. Caries correlates strongly to salivary levels of matrix metalloproteinase-8. Caries Res. 2015, 49, 1–8. [Google Scholar] [CrossRef]
- Preshaw, P.M. Host modulation therapy with anti-inflammatory agents. Periodontology 2000, 76, 131–149. [Google Scholar] [CrossRef] [PubMed]
- Golub, L.M.; Elburki, M.S.; Walker, C.; Ryan, M.; Sorsa, T.; Tenenbaum, H.; Goldberg, M.; Wolff, M.; Gu, Y. Non-antibacterial tetracycline formulations: Host-modulators in the treatment of periodontitis and relevant systemic diseases. Int. Dent. J. 2016, 66, 127–135. [Google Scholar] [CrossRef] [PubMed]
- Leite, M.F.; Santos, M.C.; de Souza, A.P.; Line, S.R. Osseointegrated implant failure associated with MMP-1 promotor polymorphisms (-1607 and -519). Int. J. Oral. Maxillofac. Implants 2008, 23, 653–658. [Google Scholar] [PubMed]
- de Araujo Munhoz, F.B.; Branco, F.P.; Souza, R.L.R.; Dos Santos, M. Matrix metalloproteinases gene polymorphism haplotype is a risk factor to implant loss: A case-control study. Clin. Implant. Dent. Relat. Res. 2018, 20, 1003–1008. [Google Scholar] [CrossRef] [PubMed]
- Zhang, F.; Liu, E.; Radaic, A.; Yu, X.; Yang, S.; Yu, C.; Xiao, S.; Ye, C. Diagnostic potential and future directions of matrix metalloproteinases as biomarkers in gingival crevicular fluid of oral and systemic diseases. Int. J. Biol. Macromol. 2021, 188, 180–196. [Google Scholar] [CrossRef]
- Ashwini, A.; Dineshkumar, T.; Rameshkumar, A.; Swarnalakshmi, R.; Shahnaz, A.; Nagarathinam, A.E.; Rajkumar, K. Dentin degradonomics—The potential role of salivary MMP-8 in dentin caries. J. Clin. Exp. Dent. 2010, 12, e108–e115. [Google Scholar] [CrossRef]
- Jain, A.; Bahuguna, R. Role of matrix metalloproteinases in dental caries, pulp and periapical inflammation: An overview. J. Oral. Biol. Craniofac Res. 2015, 5, 212–218. [Google Scholar] [CrossRef]
- Mazzoni, A.; Tjäderhane, L.; Checchi, V.; Di Lenarda, R.; Salo, T.; Tay, F.R.; Pashley, D.H.; Breschi, L. Role of dentin MMPs in caries progression and bond stability. J. Dent. Res. 2015, 94, 241–251. [Google Scholar] [CrossRef]
- Jacobsen, J.A.; Fullagar, J.L.; Miller, M.T.; Cohen, S.M. Identifying chelators for metalloprotein inhibitors using a fragment-based approach. J. Med. Chem. 2011, 54, 591–602. [Google Scholar] [CrossRef]
- Tannure, P.N.; Kuchler, E.C.; Falagan-Lotsch, P.; Amorim, L.M.; Raggio Luiz, R.; Costa, M.C.; Vieira, A.R.; Granjeiro, J.M. MMP13 polymorphism decreases risk for dental caries. Caries Res. 2012, 46, 401–407. [Google Scholar] [CrossRef]
- Vasconcelos, K.R.; Arid, J.; Evangelista, S.; Oliveira, S.; Dutra, A.L.; Silva, L.A.B.; Segato, R.A.B.; Vieira, A.R.; Nelson-Filho, P.; Kuchler, E.C. MMP13 Contributes to Dental Caries Associated with Developmental Defects of Enamel. Caries Res. 2019, 53, 441–446. [Google Scholar] [CrossRef] [PubMed]

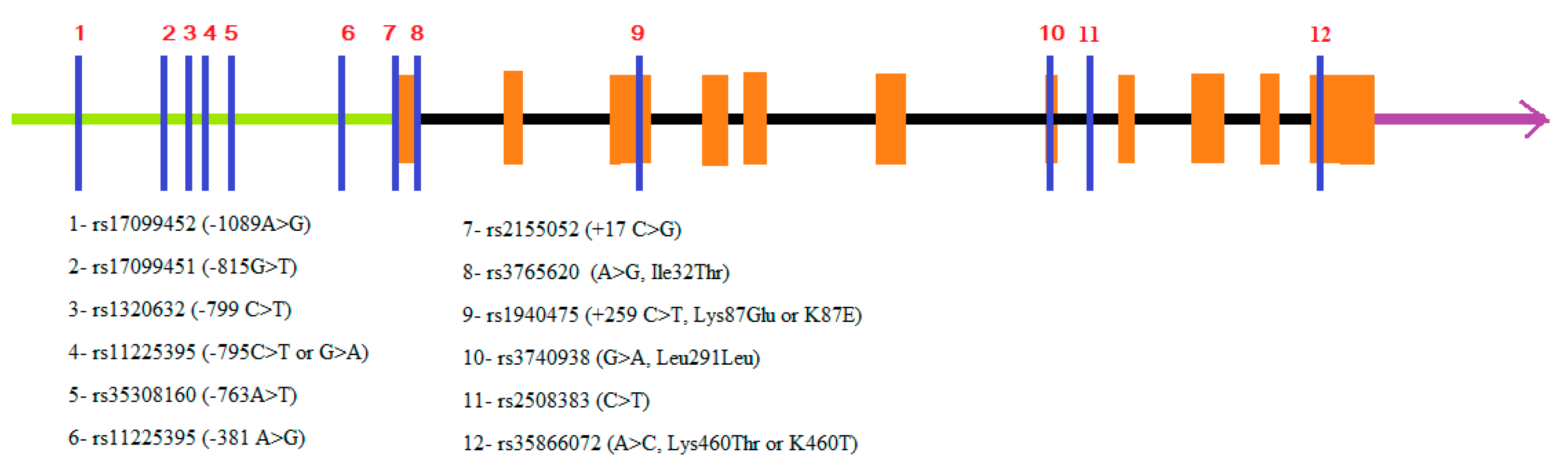
| Polymorphism in MMP8 | Population | Disease | Patients/Controls | Observation |
|---|---|---|---|---|
| −799 (C > T) | Taiwan | AgP + CP | 96 + 361/106 | Increased risk for AgP (p = 0.04) and CP (p = 0.007) in carriers of T allele [44]. |
| −799 (C > T) | No differences in allele (p = 0.06) and genotype (p = 0.280 distributions. No associations with particular periodontal pathogen [45] | |||
| Czech | CP | 341/278 | ||
| +17 (C > G) | Czech | CP | 341/278 | No differences in allele (p = 0.38) and genotype (p = 0.09) distributions. No associations with particular periodontal pathogen [45] |
| −799C>T/+17C > G haplotypes | Czech | CP | 341/278 | -799T/+17C haplotype is associated with 1.273-fold risk of CP (p = 0.038) [45] |
| −799 (C > T) | Turkish | GAgP | 100/267 | T allele (p < 0.0001) and T allele genotypes (CT + TT, p < 0.0001) were more common in GAgP determining 2.878-; 6.76-fold higher risk of GAgP compared to the wild C allele and CC genotype [18]. |
| +17 (C > G) | Turkish | GAgP | 100/267 | No differences in allele (p = 0.290) and genotype (p = 0.581) distributions [18] |
| −381 (A > G) | Turkish | GAgP | 100/267 | G allele (p = 0.027) and G allele genotypes (AG + GG, p = 0.015) were less common in GAgP determining 1.5- and 2.27-fold lower risk of GAgP (OR = 0.664 and OR = 0.44) compared to the wild A allele and AA genotype [18] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Atanasova, T.; Stankova, T.; Bivolarska, A.; Vlaykova, T. Matrix Metalloproteinases in Oral Health—Special Attention on MMP-8. Biomedicines 2023, 11, 1514. https://doi.org/10.3390/biomedicines11061514
Atanasova T, Stankova T, Bivolarska A, Vlaykova T. Matrix Metalloproteinases in Oral Health—Special Attention on MMP-8. Biomedicines. 2023; 11(6):1514. https://doi.org/10.3390/biomedicines11061514
Chicago/Turabian StyleAtanasova, Tsvetelina, Teodora Stankova, Anelia Bivolarska, and Tatyana Vlaykova. 2023. "Matrix Metalloproteinases in Oral Health—Special Attention on MMP-8" Biomedicines 11, no. 6: 1514. https://doi.org/10.3390/biomedicines11061514
APA StyleAtanasova, T., Stankova, T., Bivolarska, A., & Vlaykova, T. (2023). Matrix Metalloproteinases in Oral Health—Special Attention on MMP-8. Biomedicines, 11(6), 1514. https://doi.org/10.3390/biomedicines11061514







