Lupus Nephritis and Dysbiosis
Abstract
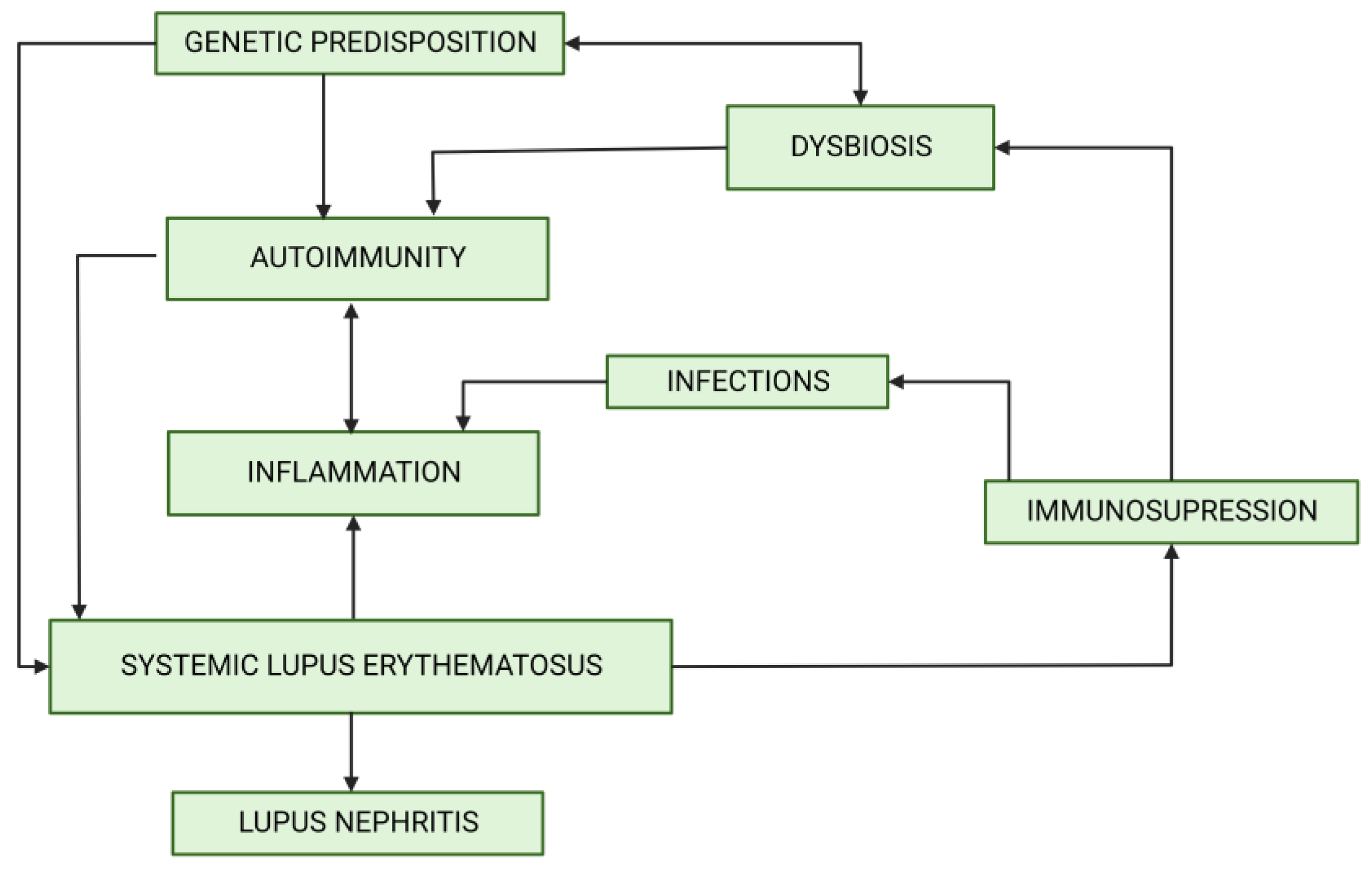
1. Introduction
2. Microbiota
3. Microbiota, Dysbiosis and LN
3.1. Intestinal Permeability
3.2. Molecular Mimicry
4. Oral Microbiota
5. Genital Microbiota
6. Skin Microbiota
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Chi, M.; Ma, K.; Wang, J.; Ding, Z.; Li, Y.; Zhu, S.; Liang, X.; Zhang, Q.; Song, L.; Liu, C. The Immunomodulatory Effect of the Gut Microbiota in Kidney Disease. J. Immunol. Res. 2021, 2021, 5516035. [Google Scholar] [CrossRef] [PubMed]
- Gomaa, E.Z. Human gut microbiota/microbiome in health and diseases: A review. Antonie Leeuwenhoek 2020, 113, 2019–2040. [Google Scholar] [CrossRef]
- Brady, M.P.; Korte, E.A.; Caster, D.J.; Powell, D.W. TNIP1/ABIN1 and lupus nephritis: Review. Lupus Sci. Med. 2020, 7, e000437. [Google Scholar] [CrossRef] [PubMed]
- Tanha, N.; Troelsen, L.; Hermansen, M.-L.F.; Kjær, L.; Faurschou, M.; Garred, P.; Jacobsen, S. MBL2 gene variants coding for mannose-binding lectin deficiency are associated with increased risk of nephritis in Danish patients with systemic lupus erythematosus. Lupus 2014, 23, 1105–1111. [Google Scholar] [CrossRef] [PubMed]
- Lee, G.R. The balance of Th17 versus treg cells in autoimmunity. Int. J. Mol. Sci. 2018, 19, 730. [Google Scholar] [CrossRef] [PubMed]
- Salzman, N.H.; Underwood, M.A.; Bevins, C.L. Paneth cells, defensins, and the commensal microbiota: A hypothesis on intimate interplay at the intestinal mucosa. Semin. Immunol. 2007, 19, 70–83. [Google Scholar] [CrossRef]
- Klein, E.Y.; Milkowska-Shibata, M.; Tseng, K.K.; Sharland, M.; Gandra, S.; Pulcini, C.; Laxminarayan, R. Assessment of WHO antibiotic consumption and access targets in 76 countries, 2000–2015: An analysis of pharmaceutical sales data. Lancet Infect. Dis. 2021, 21, 107–115. [Google Scholar] [CrossRef] [PubMed]
- Available online: https://www.cdc.gov/vitalsigns/painkilleroverdoses/infographic.html (accessed on 14 February 2023).
- Schjerning Olsen, A.M.; Lindhardsen, J.; Gislason, G.H.; McGettigan, P.; Hlatky, M.A.; Fosbøl, E.; Køber, L.; Torp-Pedersen, C.; Lamberts, M. Impact of proton pump inhibitor treatment on gastrointestinal bleeding associated with non-steroidal anti-inflammatory drug use among post-myocardial infarction patients taking antithrombotics: Nationwide study. BMJ 2015, 351, h5096. [Google Scholar] [CrossRef]
- Duarte-Garcia, A.; Hocaoglu, M.; Osei-Onomah, S.; Dabit, J.; Giblon, R.; Crowson, C. Time Trends in the Incidence of Systemic Lupus Erythematosus: A 40-Year Study. Arthritis Rheumatol. 2020, 72 (Suppl. 10). Available online: https://acrabstracts.org/abstrct/time-trends-in-the-incidence-of-systemic-lupus-erythematosus-a-40-year-study/ (accessed on 14 February 2023).
- Hall, A.B.; Tolonen, A.; Xavier, R.J. Human genetic variation and the gut microbiome in disease. Nat. Rev. Genet. 2017, 18, 690–699. [Google Scholar] [CrossRef]
- Hevia, A.; Milani, C.; López, P.; Cuervo, A.; Arboleya, S.; Duranti, S.; Turroni, F.; González, S.; Suárez, A.; Gueimonde, M.; et al. Intestinal Dysbiosis Associated with Systemic Lupus Erythematosus. mBio 2014, 5, e01548-14. [Google Scholar] [CrossRef] [PubMed]
- Neuman, H.; Koren, O. The gut microbiota: A possible factor influencing systemic lupus erythematosus. Curr. Opin. Rheumatol. 2017, 29, 374–377. [Google Scholar] [CrossRef]
- Ruiz, L.; López, P.; Suárez, A.; Sánchez, B.; Margolles, A. The role of gut microbiota in lupus: What we know in 2018? Expert Rev. Clin. Immunol. 2018, 14, 787–792. [Google Scholar] [CrossRef]
- Li, Y.; Wang, H.-F.; Li, X.; Li, H.-X.; Zhang, Q.; Zhou, H.-W.; He, Y.; Li, P.; Fu, C.; Zhang, X.-H.; et al. Disordered intestinal microbes are associated with the activity of Systemic Lupus Erythematosus. Clin. Sci. 2019, 133, 821–838. [Google Scholar] [CrossRef]
- van der Meulen, T.A.; Harmsen, H.J.M.; Vila, A.V.; Kurilshikov, A.; Liefers, S.C.; Zhernakova, A.; Fu, J.; Wijmenga, C.; Weersma, R.K.; de Leeuw, K.; et al. Shared gut, but distinct oral microbiota composition in primary Sjögren’s syndrome and systemic lupus erythematosus. J. Autoimmun. 2019, 97, 77–87. [Google Scholar] [CrossRef]
- Rojo, D.; Méndez-García, C.; Raczkowska, B.A.; Bargiela, R.; Moya, A.; Ferrer, M.; Barbas, C. Exploring the human microbiome from multiple perspectives: Factors altering its composition and function. FEMS Microbiol. Rev. 2017, 41, 453–478. [Google Scholar] [CrossRef] [PubMed]
- Knip, M.; Siljander, H. The role of the intestinal microbiota in type 1 diabetes mellitus. Nat. Rev. Endocrinol. 2016, 12, 154–167. [Google Scholar] [CrossRef]
- Van de Wiele, T.; Van Praet, J.T.; Marzorati, M.; Drennan, M.B.; Elewaut, D. How the microbiota shapes rheumatic diseases. Nat. Rev. Rheumatol. 2016, 12, 398–411. [Google Scholar] [CrossRef]
- Integrative HMP (iHMP) Research Network Consortium. The Integrative Human Microbiome Project. Nature 2019, 569, 641–648. [Google Scholar] [CrossRef] [PubMed]
- Blekhman, R.; Goodrich, J.K.; Huang, K.; Sun, Q.; Bukowski, R.; Bell, J.T.; Spector, T.D.; Keinan, A.; Ley, R.E.; Gevers, D.; et al. Host genetic variation impacts microbiome composition across human body sites. Genome Biol. 2015, 16, 191. [Google Scholar] [CrossRef]
- Wang, J.; Thingholm, L.B.; Skiecevičienė, J.; Rausch, P.; Kummen, M.; Hov, J.R.; Degenhardt, F.; Heinsen, F.A.; Rühlemann, M.C.; Szymczak, S.; et al. Genome-wide association analysis identifies variation in vitamin D receptor and other host factors influencing the gut microbiota. Nat. Genet. 2016, 48, 1396–1406. [Google Scholar] [CrossRef] [PubMed]
- Vorobyev, A.; Gupta, Y.; Sezin, T.; Koga, H.; Bartsch, Y.C.; Belheouane, M.; Künzel, S.; Sina, C.; Schilf, P.; Körber-Ahrens, H.; et al. Gene-diet interactions associated with complex trait variation in an advanced intercross outbred mouse line. Nat. Commun. 2019, 10, 4097. [Google Scholar] [CrossRef] [PubMed]
- Xu, F.; Fu, Y.; Sun, T.-Y.; Jiang, Z.; Miao, Z.; Shuai, M.; Gou, W.; Ling, C.-W.; Yang, J.; Wang, J.; et al. The interplay between host genetics and the gut microbiome reveals common and distinct microbiome features for complex human diseases. Microbiome 2020, 8, 145. [Google Scholar] [CrossRef]
- Honda, K.; Littman, D.R. The microbiota in adaptive immune homeostasis and disease. Nature 2016, 535, 75–84. [Google Scholar] [CrossRef]
- Thaiss, C.A.; Itav, S.; Rothschild, D.; Meijer, M.T.; Levy, M.; Moresi, C.; Dohnalová, L.; Braverman, S.; Rozin, S.; Malitsky, S.; et al. Persistent microbiome alterations modulate the rate of post-dieting weight regain. Nature 2016, 540, 544–551. [Google Scholar] [CrossRef] [PubMed]
- Clemente, J.C.; Manasson, J.; Scher, J.U. The role of the gut microbiome in systemic inflammatory disease. BMJ 2018, 360, j5145. [Google Scholar] [CrossRef] [PubMed]
- Ruff, W.E.; Greiling, T.M.; Kriegel, M.A. Host-microbiota interactions in immune-mediated diseases. Nat. Rev. Microbiol. 2020, 18, 521–538. [Google Scholar] [CrossRef] [PubMed]
- Manfredo Vieira, S.; Hiltensperger, M.; Kumar, V.; Zegarra-Ruiz, D.; Dehner, C.; Khan, N.; Costa, F.R.C.; Tiniakou, E.; Greiling, T.; Ruff, W.; et al. Translocation of a gut pathobiont drives autoimmunity in mice and humans. Science 2018, 359, 1156–1161. [Google Scholar] [CrossRef]
- Greiling, T.M.; Dehner, C.; Chen, X.; Hughes, K.; Iñiguez, A.J.; Boccitto, M.; Ruiz, D.Z.; Renfroe, S.C.; Vieira, S.M.; Ruff, W.E.; et al. Commensal orthologs of the human autoantigen Ro60 as triggers of autoimmunity in lupus. Sci. Transl. Med. 2018, 10, eaan2306. [Google Scholar] [CrossRef]
- Valiente, G.R.; Munir, A.; Hart, M.L.; Blough, P.; Wada, T.T.; Dalan, E.E.; Willis, W.L.; Wu, L.-C.; Freud, A.G.; Jarjour, W.N. Gut dysbiosis is associated with acceleration of lupus nephritis. Sci. Rep. 2022, 12, 152. [Google Scholar] [CrossRef] [PubMed]
- Mu, Q.; Zhang, H.; Liao, X.; Lin, K.; Liu, H.; Edwards, M.R.; Ahmed, S.A.; Yuan, R.; Li, L.; Cecere, T.E.; et al. Control of lupus nephritis by changes of gut microbiota. Microbiome 2017, 5, 73. [Google Scholar] [CrossRef]
- Azzouz, D.; Omarbekova, A.; Heguy, A.; Schwudke, D.; Gisch, N.; Rovin, B.H.; Caricchio, R.; Buyon, J.P.; Alekseyenko, A.V.; Silverman, G.J. Lupus nephritis is linked to disease-activity associated expansions and immunity to a gut commensal. Ann. Rheum. Dis. 2019, 78, 947–956. [Google Scholar] [CrossRef]
- Luo, X.M.; Edwards, M.R.; Mu, Q.; Yu, Y.; Vieson, M.D.; Reilly, C.; Ahmed, S.A.; Bankole, A.A. Gut Microbiota in Human Systemic Lupus Erythematosus and a Mouse Model of Lupus. Appl. Environ. Microbiol. 2018, 84, e02288-17. [Google Scholar] [CrossRef] [PubMed]
- Watanabe-Fukunaga, R.; Brannan, C.I.; Copeland, N.G.; Jenkins, N.A.; Nagata, S. Lymphoproliferation disorder in mice explained by defects in Fas antigen that mediates apoptosis. Nature 1992, 356, 314–317. [Google Scholar] [CrossRef] [PubMed]
- Richard, M.L.; Gilkeson, G. Mouse models of lupus: What they tell us and what they don’t. Lupus Sci. Med. 2018, 5, e000199. [Google Scholar] [CrossRef]
- Yang, W.; Cong, Y. Gut microbiota-derived metabolites in the regulation of host immune responses and immune-related inflammatory diseases. Cell. Mol. Immunol. 2021, 18, 866–877. [Google Scholar] [CrossRef] [PubMed]
- López, P.; Sánchez, B.; Margolles, A.; Suárez, A. Intestinal dysbiosis in systemic lupus erythematosus: Cause or consequence? Curr. Opin. Rheumatol. 2016, 28, 515–522. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Qing, P.; Yang, H.; Wu, Y.; Liu, Y.; Luo, Y. Gut Microbiome and Metabolites in Systemic Lupus Erythematosus: Link, Mechanisms and Intervention. Front. Immunol. 2021, 12, 686501. [Google Scholar] [CrossRef] [PubMed]
- Pan, Q.; Guo, F.; Huang, Y.; Li, A.; Chen, S.; Chen, J.; Liu, H.-F.; Pan, Q. Gut Microbiota Dysbiosis in Systemic Lupus Erythematosus: Novel Insights into Mechanisms and Promising Therapeutic Strategies. Front. Immunol. 2021, 12, 799788. [Google Scholar] [CrossRef]
- Zhang, W.; Reichlin, M. A Possible Link between Infection with Burkholderia Bacteria and Systemic Lupus Erythematosus Based on Epitope Mimicry. Clin. Dev. Immunol. 2008, 2008, 683489. [Google Scholar] [CrossRef] [PubMed]
- Soliman, S.; Mohan, C. Lupus nephritis biomarkers. Clin. Immunol. 2017, 185, 10–20. [Google Scholar] [CrossRef] [PubMed]
- Gladman, D.D.; Ibañez, D.; Urowitz, M.B. Systemic lupus erythematosus disease activity index 2000. J. Rheumatol. 2002, 29, 288–291. [Google Scholar]
- Shoenfeld, Y.; Vilner, Y.; Coates, A.R.; Rauch, J.; Lavie, G.; Shaul, D.; Pinkhas, J. Monoclonal anti-tuberculosis antibodies react with DNA, and monoclonal anti-DNA autoantibodies react with Mycobacterium tuberculosis. Clin. Exp. Immunol. 1986, 66, 255–261. [Google Scholar]
- Garabatos, N.; Santamaria, P. Gut Microbial Antigenic Mimicry in Autoimmunity. Front. Immunol. 2022, 13, 873607. [Google Scholar] [CrossRef] [PubMed]
- Szymula, A.; Rosenthal, J.; Szczerba, B.M.; Bagavant, H.; Fu, S.M.; Deshmukh, U.S. T cell epitope mimicry between Sjögren’s syndrome Antigen A (SSA)/Ro60 and oral, gut, skin and vaginal bacteria. Clin. Immunol. 2014, 152, 1–9. [Google Scholar] [CrossRef] [PubMed]
- López, P.; de Paz, B.; Rodríguez-Carrio, J.; Hevia, A.; Sánchez, B.; Margolles, A.; Suárez, A. Th17 responses and natural IgM antibodies are related to gut microbiota composition in systemic lupus erythematosus patients. Sci. Rep. 2016, 6, 24072. [Google Scholar] [CrossRef]
- Guo, M.; Wang, H.; Xu, S.; Zhuang, Y.; An, J.; Su, C.; Xia, Y.; Chen, J.; Xu, Z.Z.; Liu, Q.; et al. Alteration in gut microbiota is associated with dysregulation of cytokines and glucocorticoid therapy in systemic lupus erythematosus. Gut Microbes 2020, 11, 1758–1773. [Google Scholar] [CrossRef] [PubMed]
- Wauters, L.; Tito, R.Y.; Ceulemans, M.; Lambaerts, M.; Accarie, A.; Rymenans, L.; Verspecht, C.; Toth, J.; Mols, R.; Augustijns, P.; et al. Duodenal Dysbiosis and Relation to the Efficacy of Proton Pump Inhibitors in Functional Dyspepsia. Int. J. Mol. Sci. 2021, 22, 13609. [Google Scholar] [CrossRef]
- Crump, K.; Sahingur, S. Microbial Nucleic Acid Sensing in Oral and Systemic Diseases. J. Dent. Res. 2016, 95, 17–25. [Google Scholar] [CrossRef]
- Corrêa, J.D.; Calderaro, D.C.; Ferreira, G.A.; Mendonça, S.M.S.; Fernandes, G.R.; Xiao, E.; Teixeira, A.L.; Leys, E.J.; Graves, D.T.; Silva, T.A. Subgingival microbiota dysbiosis in systemic lupus erythematosus: Association with periodontal status. Microbiome 2017, 5, 34. [Google Scholar] [CrossRef]
- Nikitakis, N.G.; Papaioannou, W.; Sakkas, L.I.; Kousvelari, E. The autoimmunity-oral microbiome connection. Oral Dis. 2017, 23, 828–839. [Google Scholar] [CrossRef]
- Pessoa, L.; Aleti, G.; Choudhury, S.; Nguyen, D.; Yaskell, T.; Zhang, Y.; Li, W.; Nelson, K.E.; Neto, L.L.S.; Sant’Ana, A.C.P.; et al. Host microbial Interactions in Systemic Lupus Erythematosus and Periodontitis. Front. Immunol. 2019, 10, 2602. [Google Scholar] [CrossRef]
- Chen, B.D.; Jia, X.M.; Xu, J.Y.; Zhao, L.D.; Ji, J.Y.; Wu, B.X.; Ma, Y.; Li, H.; Zuo, X.X.; Pan, W.Y.; et al. An Autoimmunogenic and Proinflammatory Profile Defined by the Gut Microbiota of Patients With Untreated Systemic Lupus Erythematosus. Arthritis Rheumatol. 2021, 73, 232–243. [Google Scholar] [CrossRef]
- Méndez-Martínez, S.; García-Carrasco, M.; Cedillo-Ramírez, M.L.; Mendoza-Pinto, C.; Etchegaray-Morales, I.; Gil-Juárez, C.; Montiel-Jarquín, J.; Taboada-Cole, A.; Jiménez-Herrera, E.A.; Muñóz-Guarneros, M.; et al. Genital Mycoplasma infection among Mexican women with systemic lupus erythematosus. Int. J. Gynecol. Obstet. 2017, 138, 17–22. [Google Scholar] [CrossRef] [PubMed]
- Ekiel, A.; Romanik, M.; Aptekorz, M.; Semik-Grabarczyk, E.; Cieślik, P.; Smolec, D.; Jóźwiak, J.; Holecki, M.; Gayane, M. Prevalence of urogenital mycoplasmas in women with systemic lupus erythematosus (SLE): Preliminary study. Eur. J. Clin. Microbiol. Infect. Dis. 2020, 39, 717–721. [Google Scholar]
- Rotjanapan, P.; Jaroensukrungruang, A.; Pisitkun, P.; Ngamjanyaporn, P.; Manonai, J.; Sawaswong, V.; Chanchaem, P.; Payungporn, S. Vaginal microbiota affects urinary tract infection risk in women with systemic lupus erythematosus: A pilot cross-sectional study from Thailand. Lupus Sci. Med. 2021, 8, e000551. [Google Scholar] [CrossRef]
- Ribero, S.; Sciascia, S.; Borradori, L.; Lipsker, D. The Cutaneous Spectrum of Lupus Erythematosus. Clin. Rev. Allergy Immunol. 2017, 53, 291–305. [Google Scholar] [CrossRef]
- Zhou, H.Y.; Cao, N.W.; Guo, B.; Chen, W.J.; Tao, J.H.; Chu, X.J.; Meng, X.; Zhang, T.X.; Li, B.Z. Systemic lupus erythematosus patients have a distinct structural and functional skin microbiota compared with controls. Lupus 2021, 30, 1553–1564. [Google Scholar] [CrossRef] [PubMed]


Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Monticolo, M.; Mucha, K.; Foroncewicz, B. Lupus Nephritis and Dysbiosis. Biomedicines 2023, 11, 1165. https://doi.org/10.3390/biomedicines11041165
Monticolo M, Mucha K, Foroncewicz B. Lupus Nephritis and Dysbiosis. Biomedicines. 2023; 11(4):1165. https://doi.org/10.3390/biomedicines11041165
Chicago/Turabian StyleMonticolo, Marta, Krzysztof Mucha, and Bartosz Foroncewicz. 2023. "Lupus Nephritis and Dysbiosis" Biomedicines 11, no. 4: 1165. https://doi.org/10.3390/biomedicines11041165
APA StyleMonticolo, M., Mucha, K., & Foroncewicz, B. (2023). Lupus Nephritis and Dysbiosis. Biomedicines, 11(4), 1165. https://doi.org/10.3390/biomedicines11041165




