Small RNA-Seq Transcriptome Profiling of Mesothelial and Mesothelioma Cell Lines Revealed microRNA Dysregulation after Exposure to Asbestos-like Fibers
Abstract
1. Introduction
2. Materials and Methods
2.1. Cell Cultures
2.2. In Vitro Treatments
2.3. RNA Isolation
2.4. Small RNA-Seq
2.5. Small RNA-Seq Analysis
3. Results
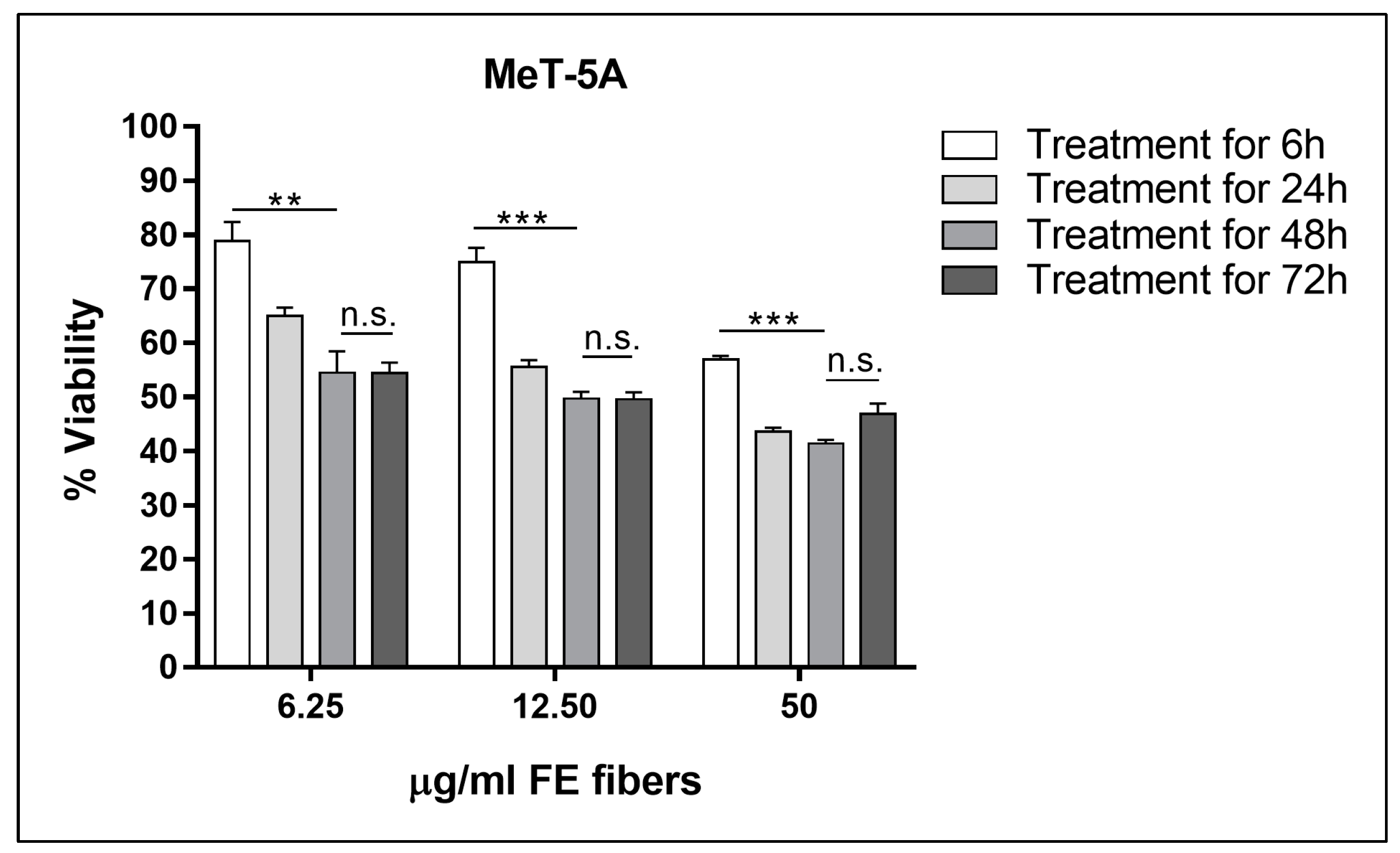
3.1. Choice of Exposure Time
3.2. Differential Expression Analysis
3.3. Pathways Analysis
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Bibby, A.C.; Tsim, S.; Kanellakis, N.; Ball, H.; Talbot, D.C.; Blyth, K.G.; Maskell, N.A.; Psallidas, I. Malignant Pleural Mesothelioma: An Update on Investigation, Diagnosis and Treatment. Eur. Respir. Rev. 2016, 25, 472–486. [Google Scholar] [CrossRef]
- Koskinen, K.; Rinne, J.-P.; Zitting, A.; Tossavainen, A.; Kivekäs, J.; Reijula, K.; Roto, P.; Huuskonen, M.S. Screening for Asbestos-Induced Diseases in Finland. Am. J. Ind. Med. 1996, 30, 241–251. [Google Scholar] [CrossRef]
- Pan, X.-L.; Day, H.W.; Wang, W.; Beckett, L.A.; Schenker, M.B. Residential Proximity to Naturally Occurring Asbestos and Mesothelioma Risk in California. Am. J. Respir. Crit. Care Med. 2005, 172, 1019–1025. [Google Scholar] [CrossRef] [PubMed]
- Luo, S.; Liu, X.; Mu, S.; Tsai, S.P.; Wen, C.P. Asbestos Related Diseases from Environmental Exposure to Crocidolite in Da-Yao, China. I. Review of Exposure and Epidemiological Data. Occup. Environ. Med. 2003, 60, 35–42. [Google Scholar] [CrossRef] [PubMed]
- Rey, F.; Boutin, C.; Steinbauer, J.; Viallat, J.R.; Alessandroni, P.; Jutisz, P.; Di Giambattista, D.; Billon-Galland, M.A.; Hereng, P.; Dumortier, P. Environmental Pleural Plaques in an Asbestos Exposed Population of Northeast Corsica. Eur. Respir. J. 1993, 6, 978–982. [Google Scholar] [CrossRef]
- Baumann, F.; Maurizot, P.; Mangeas, M.; Ambrosi, J.-P.; Douwes, J.; Robineau, B. Pleural Mesothelioma in New Caledonia: Associations with Environmental Risk Factors. Environ. Health Perspect. 2011, 119, 695–700. [Google Scholar] [CrossRef]
- McConnochie, K.; Simonato, L.; Mavrides, P.; Christofides, P.; Pooley, F.D.; Wagner, J.C. Mesothelioma in Cyprus: The Role of Tremolite. Thorax 1987, 42, 342–347. [Google Scholar] [CrossRef] [PubMed]
- Constantopoulos, S.H. Environmental Mesothelioma Associated with Tremolite Asbestos: Lessons from the Experiences of Turkey, Greece, Corsica, New Caledonia and Cyprus. Regul. Toxicol. Pharmacol. 2008, 52, S110–S115. [Google Scholar] [CrossRef] [PubMed]
- Filetti, V.; Vitale, E.; Broggi, G.; Hagnäs, M.P.; Candido, S.; Spina, A.; Lombardo, C. Update of in Vitro, in Vivo and Ex Vivo Fluoro-Edenite Effects on Malignant Mesothelioma: A Systematic Review (Review). Biomed. Rep. 2020, 13, 60. [Google Scholar] [CrossRef] [PubMed]
- Filetti, V.; Falzone, L.; Rapisarda, V.; Caltabiano, R.; Eleonora Graziano, A.C.; Ledda, C.; Loreto, C. Modulation of microRNA Expression Levels after Naturally Occurring Asbestiform Fibers Exposure as a Diagnostic Biomarker of Mesothelial Neoplastic Transformation. Ecotoxicol. Environ. Saf. 2020, 198, 110640. [Google Scholar] [CrossRef] [PubMed]
- Filetti, V.; Loreto, C.; Falzone, L.; Lombardo, C.; Cannizzaro, E.; Castorina, S.; Ledda, C.; Rapisarda, V. Diagnostic and Prognostic Value of Three microRNAs in Environmental Asbestiform Fibers-Associated Malignant Mesothelioma. J. Pers. Med. 2021, 11, 1205. [Google Scholar] [CrossRef] [PubMed]
- Biggeri, A.; Pasetto, R.; Belli, S.; Bruno, C.; Di Maria, G.; Mastrantonio, M.; Trinca, S.; Uccelli, R.; Comba, P. Mortality from Chronic Obstructive Pulmonary Disease and Pleural Mesothelioma in an Area Contaminated by Natural Fiber (fluoro-Edenite). Scand. J. Work Environ. Health 2004, 30, 249–252. [Google Scholar] [CrossRef]
- Grosse, Y.; Loomis, D.; Guyton, K.Z.; Lauby-Secretan, B.; El Ghissassi, F.; Bouvard, V.; Benbrahim-Tallaa, L.; Guha, N.; Scoccianti, C.; Mattock, H.; et al. Carcinogenicity of Fluoro-Edenite, Silicon Carbide Fibres and Whiskers, and Carbon Nanotubes. Lancet Oncol. 2014, 15, 1427–1428. [Google Scholar] [CrossRef]
- Loreto, C.; Caltabiano, R.; Graziano, A.C.E.; Castorina, S.; Lombardo, C.; Filetti, V.; Vitale, E.; Rapisarda, G.; Cardile, V.; Ledda, C.; et al. Defense and Protection Mechanisms in Lung Exposed to Asbestiform Fiber: The Role of Macrophage Migration Inhibitory Factor and Heme Oxygenase-1. Eur. J. Histochem. 2020, 64, 3073. [Google Scholar] [CrossRef] [PubMed]
- Loreto, C.; Ledda, C.; Tumino, R.; Lombardo, C.; Vitale, E.; Filetti, V.; Caltabiano, R.; Rapisarda, V. Activation of Caspase-3 in Malignant Mesothelioma Induced by Asbestiform Fiber: An in Vivo Study. J. Biol. Regul. Homeost. Agents 2020, 34, 1163–1166. [Google Scholar]
- Loreto, C.; Lombardo, C.; Caltabiano, R.; Ledda, C.; Hagnas, M.; Filetti, V.; Rapisarda, V. An in vivo immunohistochemical study on macroh2a.1 in lung and lymph-node tissues exposed to an asbestiform fiber. Curr. Mol. Med. 2020, 20, 653–660. [Google Scholar] [CrossRef]
- Broggi, G.; Angelico, G.; Filetti, V.; Ledda, C.; Lombardo, C.; Vitale, E.; Rapisarda, V.; Loreto, C.; Caltabiano, R. Immunohistochemical expression of serine and arginine-rich splicing factor 1 (Srsf1) in fluoro-edenite-induced malignant mesothelioma: A preliminary study. Int. J. Environ. Res. Public Health 2021, 18, 6249. [Google Scholar] [CrossRef] [PubMed]
- Romano, G.; Veneziano, D.; Acunzo, M.; Croce, C.M. Small Non-Coding RNA and Cancer. Carcinogenesis 2017, 38, 485–491. [Google Scholar] [CrossRef]
- Quinn, L.; Finn, S.P.; Cuffe, S.; Gray, S.G. Non-Coding RNA Repertoires in Malignant Pleural Mesothelioma. Lung Cancer 2015, 90, 417–426. [Google Scholar] [CrossRef]
- La Ferlita, A.; Battaglia, R.; Andronico, F.; Caruso, S.; Cianci, A.; Purrello, M.; Di Pietro, C. Non-Coding RNAs in Endometrial Physiopathology. Int. J. Mol. Sci. 2018, 19, 2120. [Google Scholar] [CrossRef]
- Peng, Y.; Croce, C.M. The Role of MicroRNAs in Human Cancer. Signal Transduct. Target. Ther. 2016, 1, 15004. [Google Scholar] [CrossRef]
- Wang, H.; Peng, R.; Wang, J.; Qin, Z.; Xue, L. Circulating microRNAs as Potential Cancer Biomarkers: The Advantage and Disadvantage. Clin. Epigenet. 2018, 10, 59. [Google Scholar] [CrossRef]
- Filetti, V.; La Ferlita, A.; Di Maria, A.; Cardile, V.; Graziano, A.C.E.; Rapisarda, V.; Ledda, C.; Pulvirenti, A.; Loreto, C. Dysregulation of microRNAs and tRNA-derived ncRNAs in mesothelial and mesothelioma cell lines after asbestiform fiber exposure. Sci. Rep. 2022, 12, 9181. [Google Scholar] [CrossRef] [PubMed]
- Andrews, S.; Krueger, F.; Segonds-Pichon, A.; Biggins, L.; Krueger, C.; Wingett, S. FastQC: A Quality Control Tool for High Throughput Sequence Data. 2010. Available online: https://www.bioinformatics.babraham.ac.uk/projects/fastqc/ (accessed on 12 October 2022).
- Martin, M. Cutadapt Removes Adapter Sequences from High-Throughput Sequencing Reads. EMBnet. J. 2011, 17, 10–12. [Google Scholar] [CrossRef]
- Kim, D.; Paggi, J.M.; Park, C.; Bennett, C.; Salzberg, S.L. Graph-Based Genome Alignment and Genotyping with HISAT2 and HISAT-Genotype. Nat. Biotechnol. 2019, 37, 907–915. [Google Scholar] [CrossRef] [PubMed]
- Kozomara, A.; Griffiths-Jones, S. miRBase: Annotating High Confidence microRNAs Using Deep Sequencing Data. Nucleic Acids Res. 2014, 42, D68–D73. [Google Scholar] [CrossRef]
- Robinson, M.D.; McCarthy, D.J.; Smyth, G.K. edgeR: A Bioconductor Package for Differential Expression Analysis of Digital Gene Expression Data. Bioinformatics 2010, 26, 139–140. [Google Scholar] [CrossRef]
- Ritchie, M.E.; Phipson, B.; Wu, D.; Hu, Y.; Law, C.W.; Shi, W.; Smyth, G.K. Limma Powers Differential Expression Analyses for RNA-Sequencing and Microarray Studies. Nucleic Acids Res. 2015, 43, e47. [Google Scholar] [CrossRef]
- Alaimo, S.; Giugno, R.; Acunzo, M.; Veneziano, D.; Ferro, A.; Pulvirenti, A. Post-Transcriptional Knowledge in Pathway Analysis Increases the Accuracy of Phenotypes Classification. Oncotarget 2016, 7, 54572–54582. [Google Scholar] [CrossRef] [PubMed]
- La Ferlita, A.; Alaimo, S.; Di Bella, S.; Martorana, E.; Laliotis, G.I.; Bertoni, F.; Cascione, L.; Tsichlis, P.N.; Ferro, A.; Bosotti, R.; et al. RNAdetector: A Free User-Friendly Stand-Alone and Cloud-Based System for RNA-Seq Data Analysis. BMC Bioinform. 2021, 22, 298. [Google Scholar] [CrossRef] [PubMed]
- Musumeci, G.; Loreto, C.; Giunta, S.; Rapisarda, V.; Szychlinska, M.A.; Imbesi, R.; Castorina, A.; Annese, T.; Castorina, S.; Castrogiovanni, P.; et al. Angiogenesis Correlates with Macrophage and Mast Cell Infiltration in Lung Tissue of Animals Exposed to Fluoro-Edenite Fibers. Exp. Cell Res. 2016, 346, 91–98. [Google Scholar] [CrossRef] [PubMed]
- Sayan, M.; Mossman, B.T. The NLRP3 Inflammasome in Pathogenic Particle and Fibre-Associated Lung Inflammation and Diseases. Part. Fibre Toxicol. 2016, 13, 51. [Google Scholar] [CrossRef] [PubMed]
- Dostert, C.; Petrilli, V.; Van Bruggen, R.; Steele, C.; Mossman, B.T.; Tschopp, J. Innate Immune Activation Through Nalp3 Inflammasome Sensing of Asbestos and Silica. Science 2008, 320, 674–677. [Google Scholar] [CrossRef] [PubMed]
- Vermeire, K.; Heremans, H.; Vandeputte, M.; Huang, S.; Billiau, A.; Matthys, P. Accelerated Collagen-Induced Arthritis in IFN-Gamma Receptor-Deficient Mice. J. Immunol. 1997, 158, 5507–5513. [Google Scholar] [CrossRef]
- Loreto, C.; Rapisarda, V.; Carnazza, M.L.; Musumeci, G.; Valentino, M.; Fenga, C.; Martinez, G. Fluoro-Edenite Fibres Induce Lung Cell Apoptosis: An in Vivo Study. Histol. Histopathol. 2008, 23, 319–326. [Google Scholar]
- Lin, W.-C.; Lin, C.-F.; Chen, C.-L.; Chen, C.-W.; Lin, Y.-S. Inhibition of Neutrophil Apoptosis via Sphingolipid Signaling in Acute Lung Injury. J. Pharmacol. Exp. Ther. 2011, 339, 45–53. [Google Scholar] [CrossRef]
- Zhao, B.; Tumaneng, K.; Guan, K.-L. The Hippo Pathway in Organ Size Control, Tissue Regeneration and Stem Cell Self-Renewal. Nat. Cell Biol. 2011, 13, 877–883. [Google Scholar] [CrossRef]
- Matsuzaki, H.; Lee, S.; Maeda, M.; Kumagai-Takei, N.; Nishimura, Y.; Otsuki, T. FoxO1 Regulates Apoptosis Induced by Asbestos in the MT-2 Human T-Cell Line. J. Immunotoxicol. 2016, 13, 620–627. [Google Scholar] [CrossRef]
- Lee, Y.A.; Noon, L.A.; Akat, K.M.; Ybanez, M.D.; Lee, T.-F.; Berres, M.-L.; Fujiwara, N.; Goossens, N.; Chou, H.-I.; Parvin-Nejad, F.P.; et al. Autophagy Is a Gatekeeper of Hepatic Differentiation and Carcinogenesis by Controlling the Degradation of Yap. Nat. Commun. 2018, 9, 4962. [Google Scholar] [CrossRef]
- Zhang, W.-Q.; Dai, Y.-Y.; Hsu, P.-C.; Wang, H.; Cheng, L.; Yang, Y.-L.; Wang, Y.-C.; Xu, Z.-D.; Liu, S.; Chan, G.; et al. Targeting YAP in Malignant Pleural Mesothelioma. J. Cell. Mol. Med. 2017, 21, 2663–2676. [Google Scholar] [CrossRef]
- Rapisarda, V.; Broggi, G.; Caltabiano, R.; Lombardo, C.; Castorina, S.; Trovato, A.; Ledda, C.; Filetti, V.; Loreto, C. ATG7 immunohistochemical expression in Malignant Pleural Mesothelioma. A preliminary report. Histol. Histopathol. 2021, 36, 1301–1308. [Google Scholar] [PubMed]
- Coussens, L.M.; Werb, Z. Inflammation and Cancer. Nature 2002, 420, 860–867. [Google Scholar] [CrossRef]
- Ledda, C.; Loreto, C.; Matera, S.; Massimino, N.; Cannizzaro, E.; Musumeci, A.; Migliore, M.; Fenga, C.; Pomara, C.; Rapisarda, V. Early Effects of Fluoro-Edenite: Correlation between IL-18 Serum Levels and Pleural and Parenchymal Abnormalities. Future Oncol. 2016, 12, 59–62. [Google Scholar] [CrossRef] [PubMed]
- Kumar-Singh, S.; Weyler, J.; Martin, M.J.; Vermeulen, P.B.; Van Marck, E. Angiogenic Cytokines in Mesothelioma: A Study of VEGF, FGF-1 and -2, and TGF Beta Expression. J. Pathol. 1999, 189, 72–78. [Google Scholar] [CrossRef]
- Aoe, K.; Hiraki, A.; Tanaka, T.; Gemba, K.-I.; Taguchi, K.; Murakami, T.; Sueoka, N.; Kamei, T.; Ueoka, H.; Sugi, K.; et al. Expression of Vascular Endothelial Growth Factor in Malignant Mesothelioma. Anticancer Res. 2006, 26, 4833–4836. [Google Scholar]
- Yano, S.; Shinohara, H.; Herbst, R.S.; Kuniyasu, H.; Bucana, C.D.; Ellis, L.M.; Fidler, I.J. Production of Experimental Malignant Pleural Effusions Is Dependent on Invasion of the Pleura and Expression of Vascular Endothelial Growth Factor/vascular Permeability Factor by Human Lung Cancer Cells. Am. J. Pathol. 2000, 157, 1893–1903. [Google Scholar] [CrossRef] [PubMed]
- Pugnaloni, A.; Lucarini, G.; GiantomassI, F.; Lombardo, L.; Capella, S.; Belluso, E.; Zizzi, A.; Panico, A.M.; Biagini, G.; Cardile, V. In Vitro Study of Biofunctional Indicators after Exposure to Asbestos-like Fluoro-Edenite Fibres. Cell. Mol. Biol. 2007, 53, OL965–OL980. [Google Scholar]
- Graziano, A.C.E.; Ledda, C.; Loreto, C.; Cardile, V. Adaption of Lung Fibroblasts to Fluoro-Edenite Fibers: Evaluation of Molecular and Physiological Dynamics. Cell. Physiol. Biochem. 2021, 55, 327–343. [Google Scholar]
- Jackaman, C.; Lansley, S.; Allan, J.E.; Robinson, B.W.S.; Nelson, D.J. IL-2/CD40-Driven NK Cells Install and Maintain Potency in the Anti-Mesothelioma Effector/memory Phase. Int. Immunol. 2012, 24, 357–368. [Google Scholar] [CrossRef]
- Patel, M.R.; Jacobson, B.A.; De, A.; Frizelle, S.P.; Janne, P.; Thumma, S.C.; Whitson, B.A.; Farassati, F.; Kratzke, R.A. Ras Pathway Activation in Malignant Mesothelioma. J. Thorac. Oncol. 2007, 2, 789–795. [Google Scholar] [CrossRef]
- Papp, T.; Schipper, H.; Pemsel, H.; Bastrop, R.; Muller, K.M.; Wiethege, T.; Weiss, D.G.; Dopp, E.; Schiffmann, D.; Rahman, Q. Mutational Analysis of N-Ras, p53, p16INK4a, p14ARF and CDK4 Genes in Primary Human Malignant Mesotheliomas. Int. J. Oncol. 2001, 18, 425–433. [Google Scholar] [CrossRef] [PubMed]
- Ni, Z.; Liu, Y.; Keshava, N.; Zhou, G.; Whong, W.; Ong, T. Analysis of K-Ras and p53 Mutations in Mesotheliomas from Humans and Rats Exposed to Asbestos. Mutat. Res. 2000, 468, 87–92. [Google Scholar] [CrossRef] [PubMed]
- Thirkettle, I.; Harvey, P.; Hasleton, P.S.; Ball, R.Y.; Warn, R.M. Immunoreactivity for Cadherins, HGF/SF, Met, and erbB-2 in Pleural Malignant Mesotheliomas. Histopathology 2000, 36, 522–528. [Google Scholar] [CrossRef] [PubMed]
- Giuliano, M.; Catalano, A.; Strizzi, L.; Vianale, G.; Capogrossi, M.; Procopio, A. Adenovirus-Mediated Wild-Type p53 Overexpression Reverts Tumourigenicity of Human Mesothelioma Cells. Int. J. Mol. Med. 2000, 5, 591–596. [Google Scholar] [CrossRef]
- Tolnay, E.; Kuhnen, C.; Wiethege, T.; König, J.-E.; Voss, B.; Müller, K.-M. Hepatocyte Growth Factor/scatter Factor and Its Receptor c-Met Are Overexpressed and Associated with an Increased Microvessel Density in Malignant Pleural Mesothelioma. J. Cancer Res. Clin. Oncol. 1998, 124, 291–296. [Google Scholar] [CrossRef]
- Altomare, D.A.; Testa, J.R. Perturbations of the AKT Signaling Pathway in Human Cancer. Oncogene 2005, 24, 7455–7464. [Google Scholar] [CrossRef]
- Hennessy, B.T.; Smith, D.L.; Ram, P.T.; Lu, Y.; Mills, G.B. Exploiting the PI3K/AKT Pathway for Cancer Drug Discovery. Nat. Rev. Drug Discov. 2005, 4, 988–1004. [Google Scholar] [CrossRef]
- Cedrés, S.; Montero, M.A.; Martinez, P.; Martinez, A.; Rodríguez-Freixinós, V.; Torrejon, D.; Gabaldon, A.; Salcedo, M.; Ramon y Cajal, S.; Felip, E. Exploratory Analysis of Activation of PTEN–PI3K Pathway and Downstream Proteins in Malignant Pleural Mesothelioma (MPM). Lung Cancer 2012, 77, 192–198. [Google Scholar] [CrossRef]
- Tomasetti, M.; Amati, M.; Neuzil, J.; Santarelli, L. Circulating Epigenetic Biomarkers in Lung Malignancies: From Early Diagnosis to Therapy. Lung Cancer 2017, 107, 65–72. [Google Scholar] [CrossRef] [PubMed]




| JU77NTvsMeT5ANT | JU10vsMeT10 | JU50vsMeT50 | |||
|---|---|---|---|---|---|
| Up-Regulated miRNAs | Down-Regulated miRNAs | Up-Regulated miRNAs | Down-Regulated miRNAs | Up-Regulated miRNAs | Down-Regulated miRNAs |
| hsa-miR-144-5p | hsa-miR-196b-3p | hsa-miR-139-5p | hsa-miR-4466 | hsa-miR-139-5p | hsa-miR-23a-5p |
| hsa-miR-139-5p | hsa-miR-550a-3p | hsa-miR-328-3p | hsa-miR-550a-3p | hsa-miR-328-3p | hsa-miR-615-3p |
| hsa-miR-328-3p | hsa-miR-615-3p | hsa-miR-549a | hsa-miR-615-3p | hsa-miR-549a | hsa-miR-3912-5p |
| hsa-miR-3661 | hsa-miR-3912-5p | hsa-miR-3167 | hsa-miR-3912-5p | hsa-miR-3167 | hsa-miR-2116-5p |
| hsa-miR-549a | hsa-miR-2116-5p | hsa-miR-6844 | hsa-miR-2116-5p | hsa-miR-6844 | hsa-miR-3618 |
| hsa-miR-3140-5p | hsa-miR-3613-3p | hsa-miR-18b-5p | hsa-miR-3613-3p | hsa-miR-18b-5p | hsa-miR-1262 |
| hsa-miR-3167 | hsa-miR-545-3p | hsa-let-7a-2-3p | hsa-miR-627-3p | hsa-let-7a-2-3p | hsa-miR-143-3p |
| hsa-miR-6844 | hsa-miR-627-3p | hsa-let-7c-3p | hsa-miR-664a-5p | hsa-let-7c-3p | hsa-miR-16-1-3p |
| hsa-miR-18b-5p | hsa-miR-664a-5p | hsa-miR-100-3p | hsa-miR-3618 | hsa-miR-100-3p | hsa-miR-215-5p |
| hsa-miR-4466 | hsa-miR-935 | hsa-miR-125b-2-3p | hsa-miR-1262 | hsa-miR-125b-2-3p | hsa-miR-25-5p |
| hsa-let-7a-2-3p | hsa-miR-3618 | hsa-miR-222-5p | hsa-miR-143-3p | hsa-miR-222-5p | hsa-miR-3176 |
| hsa-let-7c-3p | hsa-miR-1262 | hsa-miR-29a-5p | hsa-miR-16-1-3p | hsa-miR-29a-5p | hsa-miR-4326 |
| hsa-miR-100-3p | hsa-miR-143-3p | hsa-miR-29b-1-5p | hsa-miR-215-5p | hsa-miR-29b-1-5p | hsa-miR-484 |
| hsa-miR-125b-2-3p | hsa-miR-16-1-3p | hsa-miR-363-3p | hsa-miR-25-5p | hsa-miR-363-3p | hsa-miR-7-5p_1 |
| hsa-miR-128-1-5p | hsa-miR-215-5p | hsa-miR-449c-5p | hsa-miR-3176 | hsa-miR-449c-5p | hsa-let-7f-2-3p |
| hsa-miR-152-5p | hsa-miR-25-5p | hsa-miR-501-5p | hsa-miR-4326 | hsa-miR-501-5p | hsa-miR-16-2-3p |
| hsa-miR-19a-5p | hsa-miR-3176 | hsa-miR-503-3p | hsa-miR-484 | hsa-miR-503-3p | hsa-miR-2278 |
| hsa-miR-200a-3p | hsa-miR-4326 | hsa-miR-410-3p | hsa-miR-7-5p_1 | hsa-miR-410-3p | hsa-miR-30d-3p |
| hsa-miR-200b-3p | hsa-miR-484 | hsa-miR-125b-1-3p | hsa-let-7f-2-3p | hsa-miR-125b-1-3p | hsa-miR-3129-5p |
| hsa-miR-222-5p | hsa-miR-7-5p_1 | hsa-miR-1272 | hsa-miR-16-2-3p | hsa-miR-1272 | hsa-miR-340-3p |
| hsa-miR-29a-5p | hsa-let-7f-2-3p | hsa-miR-188-5p | hsa-miR-2278 | hsa-miR-433-3p | hsa-miR-34c-5p |
| hsa-miR-29b-1-5p | hsa-miR-16-2-3p | hsa-miR-23a-5p | hsa-miR-30d-3p | hsa-miR-548u | hsa-miR-454-5p |
| hsa-miR-362-5p | hsa-miR-2278 | hsa-miR-3934-5p | hsa-miR-3129-5p | hsa-miR-99a-5p | hsa-miR-561-3p |
| hsa-miR-363-3p | hsa-miR-30d-3p | hsa-miR-433-3p | hsa-miR-340-3p | hsa-miR-7706 | hsa-miR-570-3p |
| hsa-miR-429 | hsa-miR-3129-5p | hsa-miR-495-5p | hsa-miR-34c-5p | hsa-miR-135b-5p | hsa-miR-579-3p |
| hsa-miR-449c-5p | hsa-miR-340-3p | hsa-miR-548u | hsa-miR-454-5p | hsa-miR-154-3p | hsa-miR-624-3p |
| hsa-miR-4517 | hsa-miR-34c-5p | hsa-miR-99a-5p | hsa-miR-561-3p | hsa-miR-221-5p | hsa-miR-624-5p |
| hsa-miR-4521 | hsa-miR-454-5p | hsa-miR-7706 | hsa-miR-570-3p | hsa-miR-660-3p | hsa-miR-627-5p |
| hsa-miR-501-5p | hsa-miR-561-3p | hsa-miR-135b-5p | hsa-miR-579-3p | hsa-miR-496 | hsa-miR-7-1-3p |
| hsa-miR-503-3p | hsa-miR-570-3p | hsa-miR-449a | hsa-miR-624-3p | hsa-miR-636 | hsa-miR-3653 |
| hsa-miR-573 | hsa-miR-579-3p | hsa-miR-154-3p | hsa-miR-624-5p | hsa-miR-6720-3p | hsa-miR-1278 |
| hsa-miR-6513-3p | hsa-miR-624-3p | hsa-miR-221-5p | hsa-miR-627-5p | hsa-miR-5586-5p | hsa-miR-195-3p |
| hsa-miR-410-3p | hsa-miR-624-5p | hsa-miR-660-3p | hsa-miR-7-1-3p | hsa-let-7c-5p | hsa-miR-23b-5p |
| hsa-miR-125b-1-3p | hsa-miR-627-5p | hsa-miR-496 | hsa-miR-3653 | hsa-miR-146a-5p | hsa-miR-33b-5p |
| hsa-miR-1272 | hsa-miR-7-1-3p | hsa-miR-636 | hsa-miR-1278 | hsa-miR-708-5p | hsa-miR-548j-5p |
| hsa-miR-128-3p_1 | hsa-miR-3653 | hsa-miR-6720-3p | hsa-miR-195-3p | hsa-miR-431-3p | hsa-miR-7976 |
| hsa-miR-188-5p | hsa-miR-4645-3p | hsa-miR-5586-5p | hsa-miR-23b-5p | hsa-miR-1226-3p | hsa-miR-664a-3p |
| hsa-miR-23a-5p | hsa-miR-1278 | hsa-let-7c-5p | hsa-miR-33b-5p | hsa-miR-218-1-3p | hsa-let-7f-1-3p |
| hsa-miR-3934-5p | hsa-miR-195-3p | hsa-miR-146a-5p | hsa-miR-548j-5p | hsa-let-7a-3p | hsa-miR-106b-3p |
| hsa-miR-433-3p | hsa-miR-23b-5p | hsa-miR-708-5p | hsa-miR-7976 | hsa-let-7a-5p | hsa-miR-132-3p |
| hsa-miR-487b-5p | hsa-miR-33b-3p | hsa-miR-431-3p | hsa-miR-1248 | hsa-let-7a-5p_1 | hsa-miR-181b-3p |
| hsa-miR-495-5p | hsa-miR-33b-5p | hsa-miR-629-3p | hsa-miR-3651 | hsa-miR-100-5p | hsa-miR-3173-5p |
| hsa-miR-548u | hsa-miR-380-3p | hsa-miR-1226-3p | hsa-miR-664a-3p | hsa-miR-106a-5p | hsa-miR-345-5p |
| hsa-miR-6747-3p | hsa-miR-380-5p | hsa-miR-218-1-3p | hsa-let-7f-1-3p | hsa-miR-10a-3p | hsa-miR-3611 |
| hsa-miR-99a-5p | hsa-miR-4746-5p | hsa-let-7a-3p | hsa-miR-106b-3p | hsa-miR-10a-5p | hsa-miR-92a-3p |
| hsa-miR-7706 | hsa-miR-548j-5p | hsa-let-7a-5p | hsa-miR-132-3p | hsa-miR-10b-5p | hsa-miR-93-3p |
| hsa-miR-135b-5p | hsa-miR-7976 | hsa-let-7a-5p_1 | hsa-miR-181b-3p | hsa-miR-125b-5p | hsa-miR-1291 |
| hsa-miR-92a-1-5p | hsa-miR-1248 | hsa-miR-100-5p | hsa-miR-3173-5p | hsa-miR-125b-5p_1 | hsa-miR-3614-3p |
| hsa-miR-6514-5p | hsa-miR-3651 | hsa-miR-106a-5p | hsa-miR-345-5p | hsa-miR-137 | hsa-miR-615-5p |
| hsa-miR-449a | hsa-miR-664a-3p | hsa-miR-10a-3p | hsa-miR-3611 | hsa-miR-138-5p_1 | hsa-let-7a-5p_2 |
| hsa-miR-154-3p | hsa-let-7f-1-3p | hsa-miR-10a-5p | hsa-miR-92a-3p | hsa-miR-148b-3p | hsa-let-7d-3p |
| hsa-miR-17-5p | hsa-miR-106b-3p | hsa-miR-10b-5p | hsa-miR-93-3p | hsa-miR-155-5p | hsa-let-7d-5p |
| hsa-miR-190b | hsa-miR-132-3p | hsa-miR-125b-5p | hsa-miR-1291 | hsa-miR-193a-3p | hsa-let-7f-5p |
| hsa-miR-221-5p | hsa-miR-181b-3p | hsa-miR-125b-5p_1 | hsa-miR-3614-3p | hsa-miR-193a-5p | hsa-let-7f-5p_1 |
| hsa-miR-424-3p | hsa-miR-3173-5p | hsa-miR-137 | hsa-miR-615-5p | hsa-miR-20b-5p | hsa-let-7i-3p |
| hsa-miR-487a-3p | hsa-miR-345-5p | hsa-miR-138-5p_1 | hsa-let-7a-5p_2 | hsa-miR-218-5p | hsa-let-7i-5p |
| hsa-miR-502-5p | hsa-miR-3611 | hsa-miR-148b-3p | hsa-let-7d-3p | hsa-miR-218-5p_1 | hsa-miR-101-3p |
| hsa-miR-660-3p | hsa-miR-92a-3p | hsa-miR-155-5p | hsa-let-7d-5p | hsa-miR-221-3p | hsa-miR-106b-5p |
| hsa-miR-487a-5p | hsa-miR-93-3p | hsa-miR-193a-3p | hsa-let-7f-5p | hsa-miR-222-3p | hsa-miR-107 |
| hsa-miR-496 | hsa-miR-1291 | hsa-miR-193a-5p | hsa-let-7f-5p_1 | hsa-miR-27a-5p | hsa-miR-1179 |
| hsa-miR-636 | hsa-miR-6803-3p | hsa-miR-20b-5p | hsa-let-7i-3p | hsa-miR-29a-3p | hsa-miR-1180-3p |
| hsa-miR-6720-3p | hsa-miR-2110 | hsa-miR-218-5p | hsa-let-7i-5p | hsa-miR-29b-3p_1 | hsa-miR-125a-5p |
| hsa-miR-5586-5p | hsa-miR-3614-3p | hsa-miR-218-5p_1 | hsa-miR-101-3p | hsa-miR-30c-5p_1 | hsa-miR-126-3p |
| hsa-let-7c-5p | hsa-miR-381-5p | hsa-miR-221-3p | hsa-miR-106b-5p | hsa-miR-3188 | hsa-miR-126-5p |
| hsa-miR-146a-5p | hsa-miR-615-5p | hsa-miR-222-3p | hsa-miR-107 | hsa-miR-326 | hsa-miR-1271-5p |
| hsa-miR-708-5p | hsa-let-7a-5p_2 | hsa-miR-27a-5p | hsa-miR-1179 | hsa-miR-335-3p | hsa-miR-128-3p |
| hsa-miR-1228-3p | hsa-let-7b-5p | hsa-miR-29a-3p | hsa-miR-1180-3p | hsa-miR-34a-3p | hsa-miR-1296-5p |
| hsa-miR-370-5p | hsa-let-7d-3p | hsa-miR-29b-3p_1 | hsa-miR-125a-5p | hsa-miR-34a-5p | hsa-miR-130b-3p |
| hsa-miR-431-3p | hsa-let-7d-5p | hsa-miR-30c-5p_1 | hsa-miR-126-3p | hsa-miR-412-5p | hsa-miR-130b-5p |
| hsa-miR-501-3p | hsa-let-7f-5p | hsa-miR-3188 | hsa-miR-126-5p | hsa-miR-431-5p | hsa-miR-142-3p |
| hsa-miR-629-3p | hsa-let-7f-5p_1 | hsa-miR-326 | hsa-miR-1271-5p | hsa-miR-432-5p | hsa-miR-146b-5p |
| hsa-miR-1226-3p | hsa-let-7i-3p | hsa-miR-335-3p | hsa-miR-128-3p | hsa-miR-455-3p | hsa-miR-148a-3p |
| hsa-miR-218-1-3p | hsa-let-7i-5p | hsa-miR-34a-3p | hsa-miR-1296-5p | hsa-miR-455-5p | hsa-miR-148a-5p |
| hsa-let-7a-3p | hsa-miR-101-3p | hsa-miR-34a-5p | hsa-miR-1301-3p | hsa-miR-495-3p | hsa-miR-149-5p |
| hsa-let-7a-5p | hsa-miR-106b-5p | hsa-miR-376b-3p | hsa-miR-130b-3p | hsa-miR-5001-3p | hsa-miR-151a-5p |
| hsa-let-7a-5p_1 | hsa-miR-107 | hsa-miR-412-5p | hsa-miR-130b-5p | hsa-miR-543 | hsa-miR-15a-5p |
| hsa-miR-100-5p | hsa-miR-1179 | hsa-miR-431-5p | hsa-miR-142-3p | hsa-miR-574-3p | hsa-miR-15b-3p |
| hsa-miR-106a-5p | hsa-miR-1180-3p | hsa-miR-432-5p | hsa-miR-146b-5p | hsa-miR-584-5p | hsa-miR-15b-5p |
| hsa-miR-10a-3p | hsa-miR-125a-5p | hsa-miR-455-3p | hsa-miR-148a-3p | hsa-miR-625-5p | hsa-miR-16-5p |
| hsa-miR-10a-5p | hsa-miR-126-3p | hsa-miR-455-5p | hsa-miR-148a-5p | hsa-miR-628-3p | hsa-miR-16-5p_1 |
| hsa-miR-10b-5p | hsa-miR-126-5p | hsa-miR-495-3p | hsa-miR-149-5p | hsa-miR-628-5p | hsa-miR-181a-3p |
| hsa-miR-1185-1-3p | hsa-miR-1271-5p | hsa-miR-5001-3p | hsa-miR-151a-5p | hsa-miR-660-5p | hsa-miR-181a-5p |
| hsa-miR-1185-2-3p | hsa-miR-128-3p | hsa-miR-543 | hsa-miR-15a-5p | hsa-miR-6720-5p | hsa-miR-181a-5p_1 |
| hsa-miR-125b-5p | hsa-miR-1296-5p | hsa-miR-574-3p | hsa-miR-15b-3p | hsa-miR-99a-3p | hsa-miR-181b-5p_1 |
| hsa-miR-125b-5p_1 | hsa-miR-1301-3p | hsa-miR-584-5p | hsa-miR-15b-5p | hsa-miR-4797-3p | hsa-miR-182-5p |
| hsa-miR-137 | hsa-miR-130b-3p | hsa-miR-625-5p | hsa-miR-16-5p | hsa-miR-6783-5p | hsa-miR-185-3p |
| hsa-miR-138-5p_1 | hsa-miR-130b-5p | hsa-miR-628-3p | hsa-miR-16-5p_1 | hsa-miR-101-5p | hsa-miR-185-5p |
| hsa-miR-148b-3p | hsa-miR-134-5p | hsa-miR-628-5p | hsa-miR-181a-3p | hsa-miR-548aw | hsa-miR-191-5p |
| hsa-miR-148b-5p | hsa-miR-142-3p | hsa-miR-660-5p | hsa-miR-181a-5p | hsa-miR-335-5p | hsa-miR-192-5p |
| hsa-miR-155-5p | hsa-miR-146b-5p | hsa-miR-6720-5p | hsa-miR-181a-5p_1 | hsa-miR-4742-3p | hsa-miR-193b-3p |
| hsa-miR-193a-3p | hsa-miR-148a-3p | hsa-miR-99a-3p | hsa-miR-181b-5p_1 | hsa-miR-3138 | hsa-miR-195-5p |
| hsa-miR-193a-5p | hsa-miR-148a-5p | hsa-miR-4797-3p | hsa-miR-182-5p | hsa-miR-877-5p | hsa-miR-196b-5p |
| hsa-miR-20b-5p | hsa-miR-149-5p | hsa-miR-6783-5p | hsa-miR-185-3p | hsa-miR-144-5p | hsa-miR-19a-3p |
| hsa-miR-218-5p | hsa-miR-151a-5p | hsa-miR-101-5p | hsa-miR-185-5p | hsa-miR-3661 | hsa-miR-210-3p |
| hsa-miR-218-5p_1 | hsa-miR-15a-5p | hsa-miR-199b-5p | hsa-miR-191-5p | hsa-miR-429 | hsa-miR-2116-3p |
| hsa-miR-221-3p | hsa-miR-15b-3p | hsa-miR-548aw | hsa-miR-192-5p | hsa-miR-4521 | hsa-miR-21-3p |
| hsa-miR-222-3p | hsa-miR-15b-5p | hsa-miR-6716-3p | hsa-miR-193b-3p | hsa-miR-573 | hsa-miR-21-5p |
| hsa-miR-2277-5p | hsa-miR-16-5p | hsa-miR-449b-5p | hsa-miR-195-5p | hsa-miR-6513-3p | hsa-miR-219a-5p |
| hsa-miR-27a-3p | hsa-miR-16-5p_1 | hsa-miR-130a-5p | hsa-miR-196b-5p | hsa-miR-92a-1-5p | hsa-miR-224-5p |
| hsa-miR-27a-5p | hsa-miR-181a-3p | hsa-miR-335-5p | hsa-miR-19a-3p | hsa-miR-6514-5p | hsa-miR-22-5p |
| hsa-miR-29a-3p | hsa-miR-181a-5p | hsa-miR-551a | hsa-miR-210-3p | hsa-miR-190b | hsa-miR-2355-3p |
| hsa-miR-29b-2-5p | hsa-miR-181a-5p_1 | hsa-miR-2116-3p | hsa-miR-502-5p | hsa-miR-2355-5p | |
| hsa-miR-29b-3p | hsa-miR-181b-5p_1 | hsa-miR-21-3p | hsa-miR-487a-5p | hsa-miR-23b-3p | |
| hsa-miR-29b-3p_1 | hsa-miR-182-5p | hsa-miR-21-5p | hsa-miR-1228-3p | hsa-miR-25-3p | |
| hsa-miR-30c-2-3p | hsa-miR-185-3p | hsa-miR-219a-5p | hsa-miR-501-3p | hsa-miR-27b-3p | |
| hsa-miR-30c-5p_1 | hsa-miR-185-5p | hsa-miR-224-5p | hsa-miR-2277-5p | hsa-miR-27b-5p | |
| hsa-miR-31-5p | hsa-miR-191-5p | hsa-miR-22-5p | hsa-miR-29b-2-5p | hsa-miR-28-3p | |
| hsa-miR-3188 | hsa-miR-192-5p | hsa-miR-2355-3p | hsa-miR-30c-2-3p | hsa-miR-28-5p | |
| hsa-miR-326 | hsa-miR-193b-3p | hsa-miR-2355-5p | hsa-miR-409-5p | hsa-miR-299-3p | |
| hsa-miR-335-3p | hsa-miR-195-5p | hsa-miR-23b-3p | hsa-miR-485-3p | hsa-miR-299-5p | |
| hsa-miR-339-5p | hsa-miR-196a-5p_1 | hsa-miR-25-3p | hsa-miR-493-3p | hsa-miR-301b | |
| hsa-miR-34a-3p | hsa-miR-196b-5p | hsa-miR-26a-5p_1 | hsa-miR-500a-3p | hsa-miR-3074-3p | |
| hsa-miR-34a-5p | hsa-miR-19a-3p | hsa-miR-27b-3p | hsa-miR-502-3p | hsa-miR-30b-5p | |
| hsa-miR-3605-3p | hsa-miR-210-3p | hsa-miR-27b-5p | hsa-miR-505-5p | hsa-miR-30d-5p | |
| hsa-miR-376a-2-5p | hsa-miR-2116-3p | hsa-miR-28-3p | hsa-miR-532-3p | hsa-miR-3129-3p | |
| hsa-miR-376a-5p | hsa-miR-212-3p | hsa-miR-28-5p | hsa-miR-532-5p | hsa-miR-3200-3p | |
| hsa-miR-376b-3p | hsa-miR-21-3p | hsa-miR-299-3p | hsa-miR-92a-3p_1 | hsa-miR-32-5p | |
| hsa-miR-377-3p | hsa-miR-21-5p | hsa-miR-299-5p | hsa-miR-933 | hsa-miR-33a-3p | |
| hsa-miR-409-5p | hsa-miR-219a-5p | hsa-miR-301b | hsa-miR-370-3p | hsa-miR-33a-5p | |
| hsa-miR-412-5p | hsa-miR-224-5p | hsa-miR-3074-3p | hsa-miR-181c-3p | hsa-miR-340-5p | |
| hsa-miR-431-5p | hsa-miR-22-5p | hsa-miR-30b-5p | hsa-miR-183-3p | hsa-miR-342-3p | |
| hsa-miR-432-5p | hsa-miR-2355-3p | hsa-miR-30d-5p | hsa-miR-337-3p | hsa-miR-342-5p | |
| hsa-miR-455-3p | hsa-miR-2355-5p | hsa-miR-3129-3p | hsa-miR-4677-5p | hsa-miR-34b-5p | |
| hsa-miR-455-5p | hsa-miR-23b-3p | hsa-miR-3136-5p | hsa-miR-625-3p | hsa-miR-3613-5p | |
| hsa-miR-4775 | hsa-miR-25-3p | hsa-miR-3200-3p | hsa-miR-361-3p | ||
| hsa-miR-485-3p | hsa-miR-26a-5p_1 | hsa-miR-324-5p | hsa-miR-361-5p | ||
| hsa-miR-485-5p | hsa-miR-27b-3p | hsa-miR-32-5p | hsa-miR-374a-3p | ||
| hsa-miR-493-3p | hsa-miR-27b-5p | hsa-miR-33a-3p | hsa-miR-374a-5p | ||
| hsa-miR-495-3p | hsa-miR-28-3p | hsa-miR-33a-5p | hsa-miR-376b-5p | ||
| hsa-miR-5001-3p | hsa-miR-28-5p | hsa-miR-340-5p | hsa-miR-3912-3p | ||
| hsa-miR-500a-3p | hsa-miR-299-3p | hsa-miR-342-3p | hsa-miR-450a-1-3p | ||
| hsa-miR-502-3p | hsa-miR-299-5p | hsa-miR-342-5p | hsa-miR-450a-2-3p | ||
| hsa-miR-505-5p | hsa-miR-301b | hsa-miR-34b-5p | hsa-miR-450b-5p | ||
| hsa-miR-532-3p | hsa-miR-3074-3p | hsa-miR-3613-5p | hsa-miR-452-3p | ||
| hsa-miR-532-5p | hsa-miR-30b-5p | hsa-miR-361-3p | hsa-miR-452-5p | ||
| hsa-miR-543 | hsa-miR-30d-5p | hsa-miR-361-5p | hsa-miR-454-3p | ||
| hsa-miR-574-3p | hsa-miR-3129-3p | hsa-miR-374a-3p | hsa-miR-4766-3p | ||
| hsa-miR-584-5p | hsa-miR-3136-5p | hsa-miR-374a-5p | hsa-miR-491-5p | ||
| hsa-miR-625-5p | hsa-miR-3200-3p | hsa-miR-376b-5p | hsa-miR-494-3p | ||
| hsa-miR-628-3p | hsa-miR-324-5p | hsa-miR-3912-3p | hsa-miR-497-5p | ||
| hsa-miR-628-5p | hsa-miR-32-5p | hsa-miR-450a-1-3p | hsa-miR-5008-5p | ||
| hsa-miR-660-5p | hsa-miR-33a-3p | hsa-miR-450a-2-3p | hsa-miR-542-3p | ||
| hsa-miR-6720-5p | hsa-miR-33a-5p | hsa-miR-450b-5p | hsa-miR-542-5p | ||
| hsa-miR-92a-3p_1 | hsa-miR-340-5p | hsa-miR-452-3p | hsa-miR-556-3p | ||
| hsa-miR-99a-3p | hsa-miR-342-3p | hsa-miR-452-5p | hsa-miR-561-5p | ||
| hsa-miR-342-5p | hsa-miR-454-3p | hsa-miR-5699-3p | |||
| hsa-miR-34b-5p | hsa-miR-4766-3p | hsa-miR-5699-5p | |||
| hsa-miR-3613-5p | hsa-miR-491-5p | hsa-miR-585-3p | |||
| hsa-miR-361-3p | hsa-miR-494-3p | hsa-miR-590-3p | |||
| hsa-miR-361-5p | hsa-miR-497-5p | hsa-miR-590-5p | |||
| hsa-miR-374a-3p | hsa-miR-5008-5p | hsa-miR-598-3p | |||
| hsa-miR-374a-5p | hsa-miR-542-3p | hsa-miR-671-5p | |||
| hsa-miR-376b-5p | hsa-miR-542-5p | hsa-miR-7705 | |||
| hsa-miR-3912-3p | hsa-miR-556-3p | hsa-miR-92b-3p | |||
| hsa-miR-450a-1-3p | hsa-miR-561-5p | hsa-miR-92b-5p | |||
| hsa-miR-450a-2-3p | hsa-miR-5699-3p | hsa-miR-93-5p | |||
| hsa-miR-450b-5p | hsa-miR-5699-5p | hsa-miR-942-5p | |||
| hsa-miR-452-3p | hsa-miR-585-3p | hsa-miR-95-3p | |||
| hsa-miR-452-5p | hsa-miR-590-3p | hsa-miR-96-5p | |||
| hsa-miR-454-3p | hsa-miR-590-5p | hsa-miR-99b-3p | |||
| hsa-miR-4766-3p | hsa-miR-598-3p | hsa-miR-579-5p | |||
| hsa-miR-491-5p | hsa-miR-641 | hsa-miR-3657 | |||
| hsa-miR-494-3p | hsa-miR-671-5p | hsa-let-7g-3p | |||
| hsa-miR-497-5p | hsa-miR-7705 | hsa-miR-1305 | |||
| hsa-miR-5008-5p | hsa-miR-92b-3p | hsa-miR-132-5p | |||
| hsa-miR-542-3p | hsa-miR-92b-5p | hsa-miR-15a-3p | |||
| hsa-miR-542-5p | hsa-miR-93-5p | hsa-miR-32-3p | |||
| hsa-miR-556-3p | hsa-miR-942-5p | hsa-miR-548k | |||
| hsa-miR-561-5p | hsa-miR-95-3p | hsa-miR-7974 | |||
| hsa-miR-5699-3p | hsa-miR-96-5p | hsa-miR-19b-3p_1 | |||
| hsa-miR-5699-5p | hsa-miR-99b-3p | hsa-miR-30b-3p | |||
| hsa-miR-585-3p | hsa-miR-186-3p | hsa-miR-580-3p | |||
| hsa-miR-590-3p | hsa-miR-579-5p | hsa-miR-671-3p | |||
| hsa-miR-590-5p | hsa-miR-3657 | hsa-miR-1197 | |||
| hsa-miR-598-3p | hsa-let-7g-3p | hsa-miR-140-3p | |||
| hsa-miR-641 | hsa-miR-1305 | hsa-miR-192-3p | |||
| hsa-miR-651-5p | hsa-miR-132-5p | hsa-miR-219a-1-3p | |||
| hsa-miR-655-3p | hsa-miR-15a-3p | hsa-miR-22-3p | |||
| hsa-miR-671-5p | hsa-miR-32-3p | hsa-miR-296-5p | |||
| hsa-miR-7705 | hsa-miR-548k | hsa-miR-323a-3p | |||
| hsa-miR-92b-3p | hsa-miR-7974 | hsa-miR-378a-5p | |||
| hsa-miR-92b-5p | hsa-miR-19b-3p_1 | hsa-miR-381-3p | |||
| hsa-miR-93-5p | hsa-miR-30b-3p | hsa-miR-5582-3p | |||
| hsa-miR-942-5p | hsa-miR-580-3p | hsa-miR-643 | |||
| hsa-miR-95-3p | hsa-miR-671-3p | hsa-miR-19a-5p | |||
| hsa-miR-96-5p | hsa-miR-4742-3p | hsa-miR-196b-3p | |||
| hsa-miR-99b-3p | hsa-miR-1296-3p | hsa-miR-4645-3p | |||
| hsa-miR-99b-5p | hsa-miR-26a-1-3p | hsa-miR-33b-3p | |||
| hsa-miR-3187-3p | hsa-miR-380-3p | ||||
| hsa-miR-3684 | hsa-miR-4746-5p | ||||
| hsa-miR-548e-3p | hsa-miR-3140-3p | ||||
| hsa-miR-744-3p | hsa-miR-19b-1-5p | ||||
| hsa-miR-200c-3p | hsa-miR-362-3p | ||||
| hsa-miR-3138 | hsa-miR-320b_1 | ||||
| hsa-miR-330-3p | hsa-miR-331-5p | ||||
| hsa-miR-877-5p | hsa-miR-548n | ||||
| hsa-let-7g-5p | hsa-miR-651-3p | ||||
| hsa-miR-1197 | hsa-miR-758-5p | ||||
| hsa-miR-125a-3p | hsa-miR-193b-5p | ||||
| hsa-miR-1292-5p | hsa-miR-323b-3p | ||||
| hsa-miR-1307-5p | hsa-miR-548e-5p | ||||
| hsa-miR-140-3p | |||||
| hsa-miR-191-3p | |||||
| hsa-miR-192-3p | |||||
| hsa-miR-194-3p | |||||
| hsa-miR-197-3p | |||||
| hsa-miR-219a-1-3p | |||||
| hsa-miR-22-3p | |||||
| hsa-miR-26a-5p | |||||
| hsa-miR-296-5p | |||||
| hsa-miR-323a-3p | |||||
| hsa-miR-324-3p | |||||
| hsa-miR-330-5p | |||||
| hsa-miR-3615 | |||||
| hsa-miR-3679-5p | |||||
| hsa-miR-378a-5p | |||||
| hsa-miR-381-3p | |||||
| hsa-miR-3940-3p | |||||
| hsa-miR-5582-3p | |||||
| hsa-miR-643 | |||||
| hsa-miR-656-3p | |||||
| hsa-miR-744-5p | |||||
| Up-Regulated miRNAs in Common | ||
| JU77NTvsMeT5ANT and JU10vsMeT10 | JU77NTvsMeT5ANT and JU50vsMeT50 | JU10vsMeT10 and JU50vsMeT50 |
| hsa-miR-376b-3p | hsa-miR-4521 | hsa-miR-335-5p |
| hsa-miR-188-5p | hsa-miR-92a-3p_1 | hsa-miR-5586-5p |
| hsa-miR-410-3p | hsa-miR-532-5p | |
| hsa-miR-29b-3p_1 | hsa-miR-429 | |
| hsa-miR-449a | hsa-miR-485-3p | |
| hsa-miR-548u | hsa-miR-30c-2-3p | |
| hsa-miR-493-3p | ||
| hsa-miR-29b-2-5p | ||
| hsa-miR-190b | ||
| hsa-miR-502-3p | ||
| hsa-miR-532-3p | ||
| hsa-miR-500a-3p | ||
| hsa-miR-409-5p | ||
| hsa-miR-431-3p | ||
| hsa-miR-660-3p | ||
| hsa-miR-505-5p | ||
| hsa-miR-502-5p | ||
| hsa-miR-2277-5p | ||
| Down-Regulated miRNAs in Common | ||
| JU77NTvsMeT5ANT and JU10vsMeT10 | JU77NTvsMeT5ANT and JU50vsMeT50 | JU10vsMeT10 and JU50vsMeT50 |
| hsa-miR-1248 | hsa-miR-4645-3p | hsa-miR-22-3p |
| hsa-miR-26a-5p_1 | hsa-miR-33a-3p | hsa-miR-140-3p |
| hsa-miR-324-5p | hsa-miR-2355-5p | hsa-miR-381-3p |
| hsa-miR-1301-3p | hsa-miR-132-5p | |
| hsa-miR-664a-3p | hsa-miR-3176 | |
| hsa-miR-641 | hsa-miR-323a-3p | |
| hsa-miR-3651 | hsa-miR-296-5p | |
| hsa-miR-627-3p | hsa-let-7g-3p | |
| hsa-miR-19b-3p_1 | ||
| hsa-miR-32-3p | ||
| hsa-miR-378a-5p | ||
| hsa-miR-15a-3p | ||
| hsa-miR-548k | ||
| hsa-miR-7974 | ||
| hsa-miR-671-3p | ||
| hsa-miR-561-3p | ||
| hsa-miR-7-5p_1 | ||
| MeT10vsMeT5ANT | MeT50vsMeT5ANT | JU10vsJU77NT | JU50vsJU77NT | ||||
|---|---|---|---|---|---|---|---|
| Up-Regulated miRNAs | Down-Regulated miRNAs | Up-Regulated miRNAs | Down-Regulated miRNAs | Up-Regulated miRNAs | Down-Regulated miRNAs | Up-Regulated miRNAs | Down-Regulated miRNAs |
| hsa-miR-1228-3p | hsa-miR-1226-3p | hsa-miR-144-5p | hsa-miR-1248 | hsa-miR-3653 | hsa-miR-3140-5p | hsa-miR-4797-3p | hsa-miR-3140-3p |
| hsa-miR-144-5p | hsa-miR-1248 | hsa-miR-3140-5p | hsa-miR-1291 | hsa-miR-4645-3p | hsa-miR-3167 | hsa-miR-6783-5p | hsa-miR-3140-5p |
| hsa-miR-18b-5p | hsa-miR-1291 | hsa-miR-3167 | hsa-miR-3651 | hsa-miR-539-5p | hsa-miR-1248 | hsa-miR-101-5p | hsa-miR-3167 |
| hsa-miR-193b-5p | hsa-miR-139-5p | hsa-miR-487a-5p | hsa-miR-3912-5p | hsa-miR-3157-5p | hsa-miR-3651 | hsa-miR-196b-3p | hsa-miR-186-3p |
| hsa-miR-3140-5p | hsa-miR-218-1-3p | hsa-miR-496 | hsa-miR-548l | hsa-miR-4797-3p | hsa-miR-135b-5p | hsa-miR-199b-5p | hsa-miR-3618 |
| hsa-miR-3157-5p | hsa-miR-3614-3p | hsa-miR-636 | hsa-miR-5586-5p | hsa-miR-6783-5p | hsa-miR-186-3p | hsa-miR-548aw | hsa-miR-579-5p |
| hsa-miR-3167 | hsa-miR-3651 | hsa-miR-6720-3p | hsa-miR-652-3p | hsa-miR-7706 | hsa-miR-3618 | hsa-miR-550a-3p | hsa-miR-6844 |
| hsa-miR-330-3p | hsa-miR-3657 | hsa-miR-758-5p | hsa-miR-6803-3p | hsa-miR-101-5p | hsa-miR-4742-3p | hsa-miR-615-3p | hsa-miR-18b-5p |
| hsa-miR-370-5p | hsa-miR-381-5p | hsa-let-7c-5p | hsa-miR-1278 | hsa-miR-579-5p | hsa-miR-144-5p | hsa-miR-4466 | |
| hsa-miR-431-3p | hsa-miR-3912-5p | hsa-miR-135b-5p | hsa-miR-195-3p | hsa-miR-664a-3p | hsa-miR-3912-5p | hsa-miR-3657 | |
| hsa-miR-4466 | hsa-miR-410-3p | hsa-miR-146a-5p | hsa-miR-196b-3p | hsa-miR-6844 | hsa-miR-933 | hsa-miR-6716-3p | |
| hsa-miR-4797-3p | hsa-miR-449a | hsa-miR-186-3p | hsa-miR-199b-5p | hsa-miR-92a-1-5p | hsa-miR-139-5p | hsa-let-7a-2-3p | |
| hsa-miR-487a-5p | hsa-miR-548l | hsa-miR-200c-3p | hsa-miR-23b-5p | hsa-miR-18b-5p | hsa-miR-2116-5p | hsa-let-7c-3p | |
| hsa-miR-496 | hsa-miR-5586-5p | hsa-miR-2110 | hsa-miR-3140-3p | hsa-miR-4466 | hsa-miR-328-3p | hsa-let-7g-3p | |
| hsa-miR-501-3p | hsa-miR-615-5p | hsa-miR-3138 | hsa-miR-33b-3p | hsa-miR-6514-5p | hsa-miR-3613-3p | hsa-miR-100-3p | |
| hsa-miR-629-3p | hsa-miR-652-3p | hsa-miR-3143 | hsa-miR-33b-5p | hsa-miR-3657 | hsa-miR-3661 | hsa-miR-125b-2-3p | |
| hsa-miR-636 | hsa-miR-6716-3p | hsa-miR-3618 | hsa-miR-380-3p | hsa-miR-449a | hsa-miR-370-3p | hsa-miR-1262 | |
| hsa-miR-6514-5p | hsa-miR-6803-3p | hsa-miR-3653 | hsa-miR-380-5p | hsa-miR-6716-3p | hsa-miR-545-3p | hsa-miR-128-1-5p | |
| hsa-miR-6783-5p | hsa-miR-3934-3p | hsa-miR-4746-5p | hsa-let-7a-2-3p | hsa-miR-549a | hsa-miR-1305 | ||
| hsa-miR-7706 | hsa-miR-4645-3p | hsa-miR-548aw | hsa-let-7c-3p | hsa-miR-627-3p | hsa-miR-132-5p | ||
| hsa-miR-877-5p | hsa-miR-4742-3p | hsa-miR-548j-5p | hsa-let-7f-1-3p | hsa-miR-664a-5p | hsa-miR-143-3p | ||
| hsa-miR-933 | hsa-miR-539-5p | hsa-miR-550a-3p | hsa-let-7g-3p | hsa-miR-935 | hsa-miR-152-5p | ||
| hsa-miR-579-5p | hsa-miR-615-3p | hsa-miR-100-3p | hsa-miR-15a-3p | ||||
| hsa-miR-664a-3p | hsa-miR-7976 | hsa-miR-106b-3p | hsa-miR-16-1-3p | ||||
| hsa-miR-6844 | hsa-miR-125b-2-3p | hsa-miR-18a-3p | |||||
| hsa-miR-708-5p | hsa-miR-1262 | hsa-miR-18a-5p | |||||
| hsa-miR-92a-1-5p | hsa-miR-128-1-5p | hsa-miR-19a-5p | |||||
| hsa-miR-1296-3p | hsa-miR-19b-1-5p | ||||||
| hsa-miR-1305 | hsa-miR-200a-3p | ||||||
| hsa-miR-132-3p | hsa-miR-200b-3p | ||||||
| hsa-miR-132-5p | hsa-miR-20a-3p | ||||||
| hsa-miR-136-5p | hsa-miR-215-5p | ||||||
| hsa-miR-143-3p | hsa-miR-222-5p | ||||||
| hsa-miR-152-5p | hsa-miR-25-5p | ||||||
| hsa-miR-154-3p | hsa-miR-29a-5p | ||||||
| hsa-miR-15a-3p | hsa-miR-29b-1-5p | ||||||
| hsa-miR-16-1-3p | hsa-miR-3176 | ||||||
| hsa-miR-17-5p | hsa-miR-32-3p | ||||||
| hsa-miR-181b-3p | hsa-miR-362-3p | ||||||
| hsa-miR-18a-3p | hsa-miR-362-5p | ||||||
| hsa-miR-18a-5p | hsa-miR-363-3p | ||||||
| hsa-miR-190b | hsa-miR-3662 | ||||||
| hsa-miR-19a-5p | hsa-miR-429 | ||||||
| hsa-miR-19b-1-5p | hsa-miR-4326 | ||||||
| hsa-miR-200a-3p | hsa-miR-449c-5p | ||||||
| hsa-miR-200b-3p | hsa-miR-4517 | ||||||
| hsa-miR-20a-3p | hsa-miR-4521 | ||||||
| hsa-miR-215-5p | hsa-miR-484 | ||||||
| hsa-miR-221-5p | hsa-miR-501-5p | ||||||
| hsa-miR-222-5p | hsa-miR-503-3p | ||||||
| hsa-miR-25-5p | hsa-miR-503-5p | ||||||
| hsa-miR-26a-1-3p | hsa-miR-548k | ||||||
| hsa-miR-29a-5p | hsa-miR-573 | ||||||
| hsa-miR-29b-1-5p | hsa-miR-597-3p | ||||||
| hsa-miR-3173-5p | hsa-miR-597-5p | ||||||
| hsa-miR-3176 | hsa-miR-6513-3p | ||||||
| hsa-miR-3187-3p | hsa-miR-7-5p_1 | ||||||
| hsa-miR-32-3p | hsa-miR-7974 | ||||||
| hsa-miR-345-5p | hsa-miR-3934-3p | ||||||
| hsa-miR-3611 | hsa-miR-410-3p | ||||||
| hsa-miR-362-3p | hsa-let-7b-3p | ||||||
| hsa-miR-362-5p | hsa-let-7f-2-3p | ||||||
| hsa-miR-363-3p | hsa-miR-125b-1-3p | ||||||
| hsa-miR-3662 | hsa-miR-1272 | ||||||
| hsa-miR-3684 | hsa-miR-128-3p_1 | ||||||
| hsa-miR-424-3p | hsa-miR-16-2-3p | ||||||
| hsa-miR-429 | hsa-miR-17-3p | ||||||
| hsa-miR-4326 | hsa-miR-188-5p | ||||||
| hsa-miR-449c-5p | hsa-miR-190a-3p | ||||||
| hsa-miR-4517 | hsa-miR-19b-3p | ||||||
| hsa-miR-4521 | hsa-miR-19b-3p_1 | ||||||
| hsa-miR-484 | hsa-miR-2278 | ||||||
| hsa-miR-487a-3p | hsa-miR-23a-5p | ||||||
| hsa-miR-501-5p | hsa-miR-26b-3p | ||||||
| hsa-miR-502-5p | hsa-miR-30b-3p | ||||||
| hsa-miR-503-3p | hsa-miR-30d-3p | ||||||
| hsa-miR-503-5p | hsa-miR-3129-5p | ||||||
| hsa-miR-548e-3p | hsa-miR-320b_1 | ||||||
| hsa-miR-548k | hsa-miR-331-5p | ||||||
| hsa-miR-573 | hsa-miR-340-3p | ||||||
| hsa-miR-597-3p | hsa-miR-34c-5p | ||||||
| hsa-miR-597-5p | hsa-miR-3934-5p | ||||||
| hsa-miR-6513-3p | hsa-miR-411-3p | ||||||
| hsa-miR-660-3p | hsa-miR-433-3p | ||||||
| hsa-miR-744-3p | hsa-miR-449b-5p | ||||||
| hsa-miR-7-5p_1 | hsa-miR-454-5p | ||||||
| hsa-miR-7974 | hsa-miR-487b-5p | ||||||
| hsa-miR-92a-3p | hsa-miR-495-5p | ||||||
| hsa-miR-93-3p | hsa-miR-545-5p | ||||||
| hsa-miR-548n | |||||||
| hsa-miR-548u | |||||||
| hsa-miR-561-3p | |||||||
| hsa-miR-570-3p | |||||||
| hsa-miR-579-3p | |||||||
| hsa-miR-580-3p | |||||||
| hsa-miR-624-3p | |||||||
| hsa-miR-624-5p | |||||||
| hsa-miR-627-5p | |||||||
| hsa-miR-651-3p | |||||||
| hsa-miR-671-3p | |||||||
| hsa-miR-6747-3p | |||||||
| hsa-miR-7-1-3p | |||||||
| hsa-miR-99a-5p | |||||||
| Up-Regulated miRNAs in Common | |
| MeT10vsMeT5ANT and MeT50vsMeT5ANT | JU10vsJU77NT and JU50vsJU77NT |
| - | hsa-miR-615-3p |
| Down-Regulated miRNAs in Common | |
| MeT10vsMeT5ANT and MeT50vsMeT5ANT | JU10vsJU77NT and JU50vsJU77NT |
| - | hsa-let-7g-3p |
| hsa-miR-200b-3p | |
| hsa-let-7c-3p | |
| hsa-miR-7974 | |
| hsa-miR-100-3p | |
| hsa-miR-503-5p | |
| hsa-miR-222-5p | |
| hsa-miR-484 | |
| hsa-miR-363-3p | |
| hsa-miR-4521 | |
| hsa-miR-29a-5p | |
| hsa-miR-18a-5p | |
| hsa-miR-29b-1-5p | |
| hsa-miR-18a-3p | |
| hsa-miR-503-3p | |
| hsa-miR-362-5p | |
| hsa-miR-20a-3p | |
| hsa-let-7a-2-3p | |
| hsa-miR-200a-3p | |
| hsa-miR-132-5p | |
| hsa-miR-32-3p | |
| hsa-miR-125b-2-3p | |
| hsa-miR-16-1-3p | |
| hsa-miR-19b-1-5p | |
| hsa-miR-429 | |
| hsa-miR-548k | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Filetti, V.; Lombardo, C.; Loreto, C.; Dounias, G.; Bracci, M.; Matera, S.; Rapisarda, L.; Rapisarda, V.; Ledda, C.; Vitale, E. Small RNA-Seq Transcriptome Profiling of Mesothelial and Mesothelioma Cell Lines Revealed microRNA Dysregulation after Exposure to Asbestos-like Fibers. Biomedicines 2023, 11, 538. https://doi.org/10.3390/biomedicines11020538
Filetti V, Lombardo C, Loreto C, Dounias G, Bracci M, Matera S, Rapisarda L, Rapisarda V, Ledda C, Vitale E. Small RNA-Seq Transcriptome Profiling of Mesothelial and Mesothelioma Cell Lines Revealed microRNA Dysregulation after Exposure to Asbestos-like Fibers. Biomedicines. 2023; 11(2):538. https://doi.org/10.3390/biomedicines11020538
Chicago/Turabian StyleFiletti, Veronica, Claudia Lombardo, Carla Loreto, George Dounias, Massimo Bracci, Serena Matera, Lucia Rapisarda, Venerando Rapisarda, Caterina Ledda, and Ermanno Vitale. 2023. "Small RNA-Seq Transcriptome Profiling of Mesothelial and Mesothelioma Cell Lines Revealed microRNA Dysregulation after Exposure to Asbestos-like Fibers" Biomedicines 11, no. 2: 538. https://doi.org/10.3390/biomedicines11020538
APA StyleFiletti, V., Lombardo, C., Loreto, C., Dounias, G., Bracci, M., Matera, S., Rapisarda, L., Rapisarda, V., Ledda, C., & Vitale, E. (2023). Small RNA-Seq Transcriptome Profiling of Mesothelial and Mesothelioma Cell Lines Revealed microRNA Dysregulation after Exposure to Asbestos-like Fibers. Biomedicines, 11(2), 538. https://doi.org/10.3390/biomedicines11020538













