Biological Role of Folic Acid in Pregnancy and Possible Therapeutic Application for the Prevention of Preeclampsia
Abstract
1. Introduction
2. Materials and Methods
3. Results and Discussion
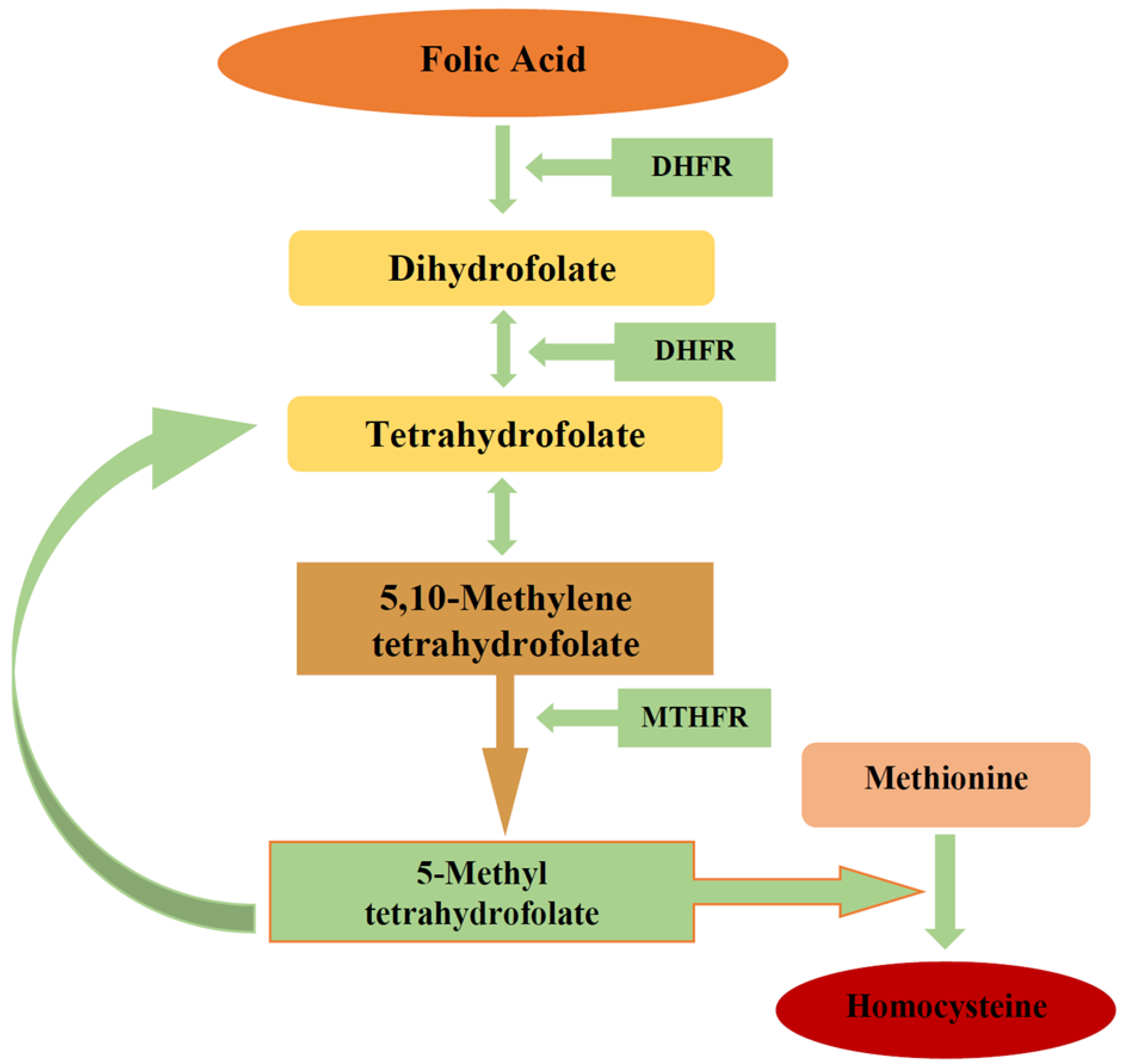
3.1. Folic Acid
3.1.1. Folic Acid Metabolism
3.1.2. Folic Acid Function
3.1.3. MTHFR Gene Physiology and Polymorphism
3.2. The Role of Folic Acid Supplementation in Pregnancy
Folic Acid in the Fetus and Placenta
3.3. Preeclampsia
3.3.1. Epidemiology of Preeclampsia
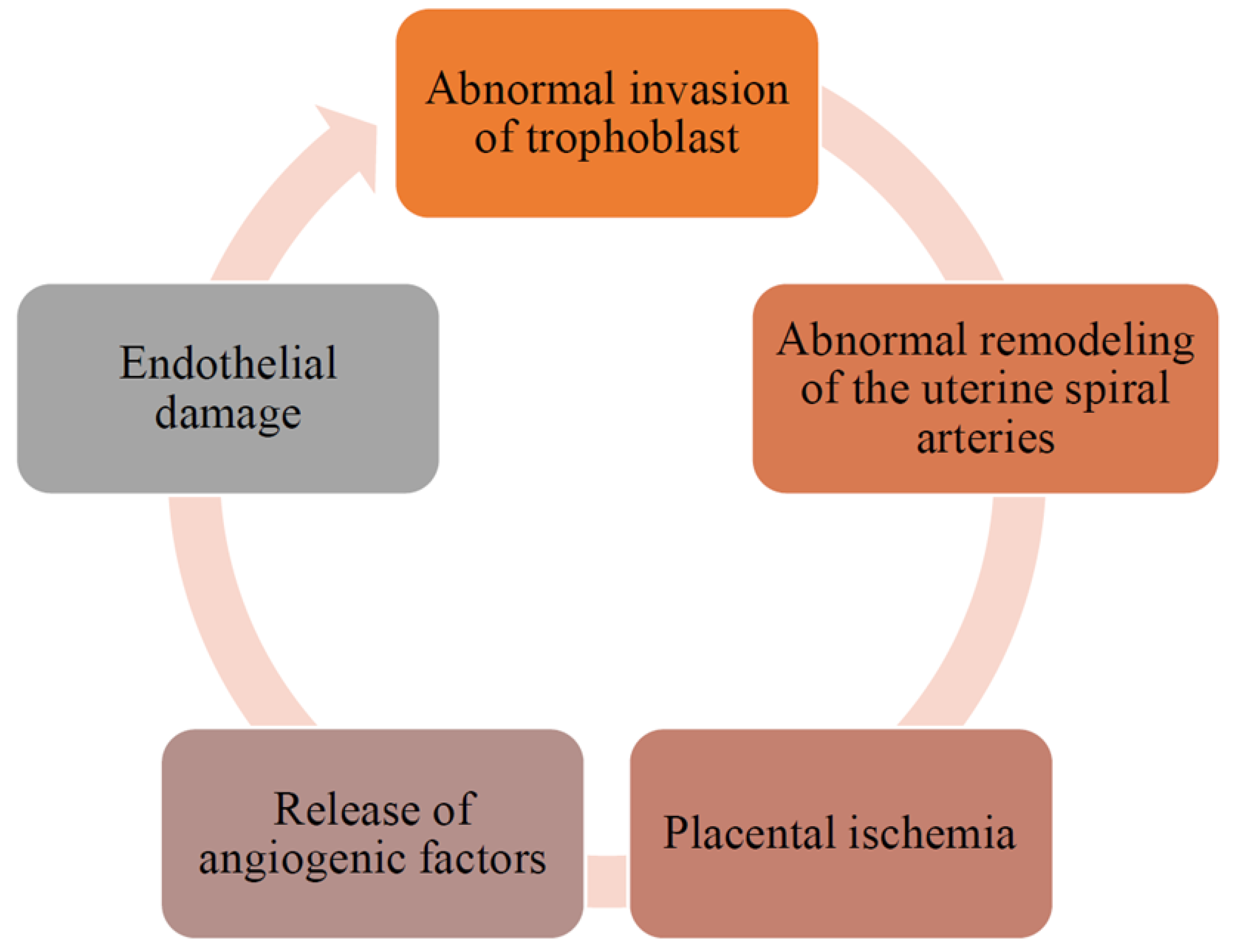
3.3.2. Pathogenesis of Preeclampsia
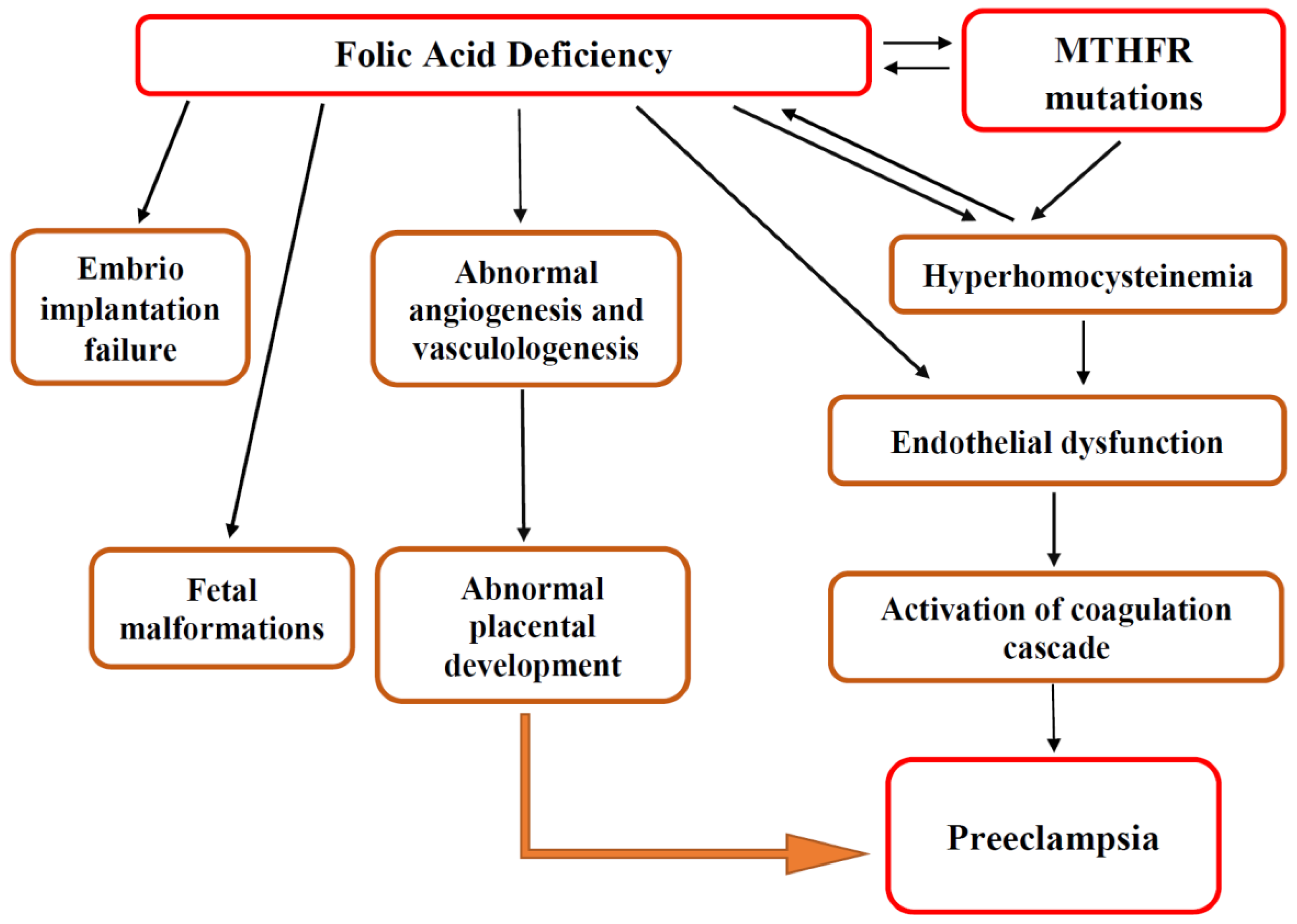
3.3.3. The Role of MTHFR Polymorphism and Hyperhomocysteinemia in Preeclampsia
3.4. Folic Acid Supplementation for Preeclampsia Prevention
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ives, C.W.; Sinkey, R.; Rajapreyar, I.; Tita, A.T.N.; Oparil, S. Preeclampsia—Pathophysiology and Clinical Presentations. J. Am. Coll. Cardiol. 2020, 76, 1690–1702. [Google Scholar] [CrossRef]
- Phipps, E.; Prasanna, D.; Brima, W.; Jim, B. Preeclampsia: Updates in Pathogenesis, Definitions, and Guidelines. Clin. J. Am. Soc. Nephrol. 2016, 11, 1102–1113. [Google Scholar] [CrossRef]
- ACOG. Gestational Hypertension and Preeclampsia. Obstet. Gynecol. 2020, 135, 1492–1495. [Google Scholar] [CrossRef]
- Ferrazzi, E.; Tiso, G.; Di Martino, D. Folic acid versus 5- methyl tetrahydrofolate supplementation in pregnancy. Eur. J. Obstet. Gynecol. Reprod. Biol. 2020, 253, 312–319. [Google Scholar] [CrossRef] [PubMed]
- El-Sayed, A.A.F. Preeclampsia: A Review of the Pathogenesis and Possible Management Strategies Based on Its Pathophysiological Derangements. Taiwan J. Obstet. Gynecol. 2017, 56, 593–598. [Google Scholar] [CrossRef]
- Rana, S.; Lemoine, E.; Granger, J.P.; Karumanchi, S.A. Preeclampsia: Pathophysiology, challenges, and perspectives. Circ. Res. 2019, 124, 1094–1112. [Google Scholar] [CrossRef]
- Hua, X.; Zhang, J.; Guo, Y.; Shen, M.; Gaudet, L.; Janoudi, G.; Walker, M.; Wen, S.W. Effect of folic acid supplementation during pregnancy on gestational hypertension/preeclampsia: A systematic review and meta-analysis. Hypertens. Pregnancy 2016, 35, 447–460. [Google Scholar] [CrossRef]
- Liu, C.; Liu, C.; Wang, Q.; Zhang, Z. Supplementation of folic acid in pregnancy and the risk of preeclampsia and gestational hypertension: A meta-analysis. Arch. Gynecol. Obstet. 2018, 298, 697–704. [Google Scholar] [CrossRef]
- Hauspurg, A.; Countouris, M.E.; Catov, J.M. Hypertensive Disorders of Pregnancy and Future Maternal Health: How Can the Evidence Guide Postpartum Management? Curr. Hypertens. Rep. 2019, 21, 96. [Google Scholar] [CrossRef]
- Sanchez, S.E.; Zhang, C.; Malinow, M.R.; Ware-Jauregui, S.; Larrabure, G.; Williams, M.A. Plasma Folate, Vitamin B12, and Homocyst(e)ine Concentrations in Preeclamptic and Normotensive Peruvian Women. Am. J. Epidemiol. 2001, 153, 474–480. [Google Scholar] [CrossRef]
- Vollset, S.E.; Refsum, H.; Irgens, L.M.; Emblem, B.M.; Tverdal, A.; Gjessing, H.K.; Monsen, A.L.B.; Ueland, P.M. Plasma total homocysteine, pregnancy complications, and adverse pregnancy outcomes: The Hordaland Homocysteine Study. Am. J. Clin. Nutr. 2000, 71, 962–968. [Google Scholar] [CrossRef] [PubMed]
- Manizheh, S.M.; Mandana, S.; Hassan, A.; Amir, G.H.; Mahlisha, K.S.; Morteza, G. Comparison study on the effect of prenatal administration of high dose and low dose folic acid. Saudi Med. J. 2009, 30, 88–97. [Google Scholar] [PubMed]
- Shahbazian, N.; Jafari, R.M.; Haghnia, S. The evaluation of serum homocysteine, folic acid, and vitamin B12 in patients complicated with preeclampsia. Electron. Physician 2016, 8, 3057–3061. [Google Scholar] [CrossRef] [PubMed]
- Dai, C.; Fei, Y.; Li, J.; Shi, Y.; Yang, X. A Novel Review of Homocysteine and Pregnancy Complications. BioMed Res. Int. 2021, 2021, 6652231. [Google Scholar] [CrossRef] [PubMed]
- Liew, S.-C.; Gupta, E.D. Methylenetetrahydrofolate reductase (MTHFR) C677T polymorphism: Epidemiology, metabolism and the associated diseases. Eur. J. Med. Genet. 2015, 58, 1–10. [Google Scholar] [CrossRef]
- Akilzhanova, A.; Takamura, N.; Kusano, Y.; Karazhanova, L.; Yamashita, S.; Saito, H.; Aoyagi, K. Association between C677T/MTHFR genotype and homocysteine concentration in a Kazakh population. Asia Pac. J. Clin. Nutr. 2008, 17, 325–329. [Google Scholar]
- Hiraoka, M.; Kagawa, Y. Genetic polymorphisms and folate status. Congenit. Anom. 2017, 57, 142–149. [Google Scholar] [CrossRef]
- Razbekova, M.; Issanov, A.; Chan, M.-Y.; Chan, R.; Yerezhepov, D.; Kozhamkulov, U.; Akilzhanova, A.; Chan, C.-K. Genetic factors associated with obesity risks in a Kazakhstani population. BMJ Nutr. Prev. Health 2021, 4, 90–101. [Google Scholar] [CrossRef]
- Kindler, H.L. Systemic Treatments for Mesothelioma: Standard and Novel. Curr. Treat. Options Oncol. 2008, 9, 171–179. [Google Scholar] [CrossRef]
- Bulloch, R.E.; Lovell, A.L.; Jordan, V.M.B.; McCowan, L.M.E.; Thompson, J.M.D.; Wall, C.R. Maternal folic acid supplementation for the prevention of preeclampsia: A systematic review and meta-analysis. Paediatr. Périnat. Epidemiol. 2018, 32, 346–357. [Google Scholar] [CrossRef]
- Wen, S.W.; White, R.R.; Rybak, N.; Gaudet, L.M.; Robson, S.; Hague, W.; Simms-Stewart, D.; Carroli, G.; Smith, G.; Fraser, W.D.; et al. Effect of high dose folic acid supplementation in pregnancy on pre-eclampsia (FACT): Double blind, phase III, randomised controlled, international, multicentre trial. BMJ 2018, 362, k3478. [Google Scholar] [CrossRef]
- De Ocampo, M.P.G.; Araneta, M.R.G.; Macera, C.A.; Alcaraz, J.E.; Moore, T.R.; Chambers, C.D. Folic acid supplement use and the risk of gestational hypertension and preeclampsia. Women Birth 2018, 31, e77–e83. [Google Scholar] [CrossRef]
- Catov, J.M.; Bodnar, L.M.; Olsen, J.; Olsen, S.; A Nohr, E. Periconceptional multivitamin use and risk of preterm or small-for-gestational-age births in the Danish National Birth Cohort. Am. J. Clin. Nutr. 2011, 94, 906–912. [Google Scholar] [CrossRef]
- Hassan, A.G.M.; Shehata, N.A. Comparison between the roles of low-dose aspirin and folic acid in preventing preeclampsia among high-risk women screened by uterine artery Doppler at 22–24 weeks of gestation. Evid. Based Women’s Health J. 2015, 5, 111–117. [Google Scholar] [CrossRef]
- Davis, R.E. Clinical Chemistry of Folic Acid. Adv. Clin. Chem. 1986, 25, 233–294. [Google Scholar] [CrossRef] [PubMed]
- Talaulikar, V.S.; Arulkumaran, S. Folic Acid in Obstetric Practice: A Review. Obstet. Gynecol. Surv. 2011, 66, 240–247. [Google Scholar] [CrossRef] [PubMed]
- Wright, A.J.A.; Dainty, J.R.; Finglas, P. Folic acid metabolism in human subjects revisited: Potential implications for proposed mandatory folic acid fortification in the UK. Br. J. Nutr. 2007, 98, 667–675. [Google Scholar] [CrossRef]
- Rosenberg, I.H. Absorption and malabsorption of folates. Clin. Haematol. 1976, 5, 589–618. [Google Scholar] [CrossRef]
- Ebara, S. Nutritional role of folate. Congenit. Anom. 2017, 57, 138–141. [Google Scholar] [CrossRef]
- Menezo, Y.; Elder, K.; Clement, A.; Clement, P. Folic Acid, Folinic Acid, 5 Methyl TetraHydroFolate Supplementation for Mutations that Affect Epigenesis through the Folate and One-Carbon Cycles. Biomolecules 2022, 12, 197. [Google Scholar] [CrossRef]
- Bastian, H. Lucy Wills (1888–1964): The life and research of an adventurous independent woman. J. R. Coll. Physicians Edinb. 2008, 38, 89–91. [Google Scholar] [PubMed]
- Angier, R.B.; Boothe, J.H.; Hutchings, B.L.; Mowat, J.H.; Semb, J.; Stokstad, E.L.; Subbarow, Y.; Waller, C.W.; Cosulich, D.B.; Fahrenbach, M.J.; et al. Synthesis of a compound identical with the L. casei factor isolated from liver. Science 1945, 102, 227–228. [Google Scholar] [CrossRef] [PubMed]
- Hasan, T.; Arora, R.; Bansal, A.K.; Bhattacharya, R.; Sharma, G.S.; Singh, L.R. Disturbed homocysteine metabolism is associated with cancer. Exp. Mol. Med. 2019, 51, 1–13. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Recommendations on Antenatal Care for a Positive Pregnancy Experience. Available online: https://apps.who.int/iris/bitstream/handle/10665/250796/97892415?sequence (accessed on 18 November 2022).
- Oh, C.; Keats, E.C.; Bhutta, Z.A. Vitamin and Mineral Supplementation During Pregnancy on Maternal, Birth, Child Health and Development Outcomes in Low- and Middle-Income Countries: A Systematic Review and Meta-Analysis. Nutrients 2020, 12, 491. [Google Scholar] [CrossRef]
- van Gool, J.D.; Hirche, H.; Lax, H.; De Schaepdrijver, L. Folic acid and primary prevention of neural tube defects: A review. Reprod. Toxicol. 2018, 80, 73–84. [Google Scholar] [CrossRef]
- Lan, X.; Field, M.S.; Stover, P.J. Cell cycle regulation of folate-mediated one-carbon metabolism. Wiley Interdiscip. Rev. Syst. Biol. Med. 2018, 10, e1426. [Google Scholar] [CrossRef]
- Williams, P.J.; Bulmer, J.N.; Innes, B.A.; Broughton Pipkin, F. Possible Roles for Folic Acid in the Regulation of Trophoblast Invasion and Placental Development in Normal Early Human Pregnancy. Biol. Reprod. 2011, 84, 1148–1153. [Google Scholar] [CrossRef]
- Molloy, A.M. Genetic aspects of folate metabolism. In Water Soluble Vitamins. Clinical Research and Future Application; Stanger, O., Ed.; Springer: Dordrecht, The Netherlands, 2011; pp. 105–130. [Google Scholar]
- Rahat, B.; Hamid, A.; Bagga, R.; Kaur, J. Folic Acid Levels During Pregnancy Regulate Trophoblast Invasive Behavior and the Possible Development of Preeclampsia. Front. Nutr. 2022, 9, 847136. [Google Scholar] [CrossRef]
- Goyette, P.; Sumner, J.S.; Milos, R.; Duncan, A.M.V.; Rosenblatt, D.S.; Matthews, R.G.; Rozen, R. Human methylenetetrahydrofolate reductase: Isolation of cDNA, mapping, and mutation identification. Nat. Genet. 1994, 7, 195–200. [Google Scholar] [CrossRef]
- Al Mutairi, F. Hyperhomocysteinemia: Clinical Insights. J. Central Nerv. Syst. Dis. 2020, 12, 1179573520962230. [Google Scholar] [CrossRef]
- Kang, S.; Zhou, J.; Wong, P.; Kowalisyn, J.; Strokosch, G. Intermediate homocysteinaemia: A thermolabile variant of meth-ylenetrahydrofolate reductase. Am. J. Hum. Genet. 1988, 43, 414–421. [Google Scholar]
- Kumar, A.; Palfrey, H.A.; Pathak, R.; Kadowitz, P.J.; Gettys, T.W.; Murthy, S.N. The metabolism and significance of homocysteine in nutrition and health. Nutr. Metab. 2017, 14, 78. [Google Scholar] [CrossRef] [PubMed]
- Xie, R.; Jia, D.; Gao, C.; Zhou, J.; Sui, H.; Wei, X.; Zhang, T.; Han, Y.; Shi, J.; Bai, Y. Homocysteine induces procoagulant activity of red blood cells via phosphatidylserine exposure and microparticles generation. Amino Acids 2014, 46, 1997–2004. [Google Scholar] [CrossRef] [PubMed]
- Dean, L. Methylenetetrahydrofolate reductase deficiency. In Medical Genetics Summaries; Pratt, V.M., Scott, S.A., Pirmohamed, M., Esquivel, B., Kattman, B.L., Malheiro, A.J., Eds.; National Center for Biotechnology Information (US): Bethesda, MD, USA, 2012. [Google Scholar]
- Qin, J.-M.; Wang, X.-M.; Chen, B.; Yang, L.; Li, F.; He, L.; Liao, P.-H. Study on the ingestion of folate and polymorphism of MTHFR C677T with esophageal cancer in Xinjiang Kazakh. Zhonghua Liu Xing Bing Xue Za Zhi = Zhonghua Liuxingbingxue Zazhi 2008, 29, 30–33. [Google Scholar]
- Yang, Y.-B.; Shang, Y.-H.; Tan, Y.-L.; Kang, X.-J.; Meng, M.; Zhao, Z.-X. Methylenetetrahydrofolate Reductase Polymorphisms and Susceptibility to Esophageal Cancer in Chinese Populations: A Meta-Analysis. Asian Pac. J. Cancer Prev. 2014, 15, 1345–1349. [Google Scholar] [CrossRef]
- Dong, Y.; Yi, X.; Yujie, Z.; Huixia, Z.; Yan, C. Relationship between the Methylation of Folic Acid Metabolism-Related Genes and the Incidence and Prognosis of Esophageal Cancer among Ethnic Kazakhs. J. Cancer 2018, 9, 2865–2875. [Google Scholar] [CrossRef]
- Guo, S.; Pang, H.; Guo, H.; Zhang, M.; He, J.; Yan, Y.; Niu, Q.; Muratbek; Rui, D.; Li, S.; et al. Ethnic Differences in the Prevalence of High Homocysteine Levels Among Low-Income Rural Kazakh and Uyghur Adults in Far Western China and Its Implications for Preventive Public Health. Int. J. Environ. Res. Public Health 2015, 12, 5373–5385. [Google Scholar] [CrossRef] [PubMed]
- Adjalla, C.E.; Amouzou, E.K.; Sanni, A.; Abdelmouttaleb, I.; Chabi, N.W.; Namour, F.; Soussou, B.; Guéant, J.-L. Low Frequency of Mutated Methylenetetrahydrofolate Reductase 677 C→T and 1298 A→C Genetics Single Nucleotide Polymorphisms (SNPs) in Sub-Saharan Populations. Clin. Chem. Lab. Med. (CCLM) 2003, 41, 1028–1032. [Google Scholar] [CrossRef]
- Osunkalu, V.; Taiwo, I.; Makwe, C.; Quao, R. Methylene tetrahydrofolate reductase and methionine synthase gene polymorphisms as genetic determinants of pre-eclampsia. Pregnancy Hypertens. 2020, 20, 7–13. [Google Scholar] [CrossRef] [PubMed]
- Ganguly, P.; Alam, S.F. Role of homocysteine in the development of cardiovascular disease. Nutr. J. 2015, 14, 6. [Google Scholar] [CrossRef]
- Gaiday, A.N.; Tussupkaliyev, A.B.T.; Bermagambetova, S.K.; Zhumagulova, S.S.; Sarsembayeva, L.K.; Dossimbetova, M.B.; Daribay, Z.Z. Effect of homocysteine on pregnancy: A systematic review. Chem. Biol. Interact. 2018, 293, 70–76. [Google Scholar] [CrossRef]
- Saussele, T. Supplementation with folic acid before and during pregnancy. Med. Mon. Fur Pharm. 2008, 31, 469–472. [Google Scholar]
- Berger, M.M.; Shenkin, A.; Schweinlin, A.; Amrein, K.; Augsburger, M.; Biesalski, H.-K.; Bischoff, S.C.; Casaer, M.P.; Gundogan, K.; Lepp, H.-L.; et al. ESPEN micronutrient guideline. Clin. Nutr. 2022, 41, 1357–1424. [Google Scholar] [CrossRef]
- Wilson, R.D.; O’Connor, D.L. Guideline No. 427: Folic Acid and Multivitamin Supplementation for Prevention of Folic Acid–Sensitive Congenital Anomalies. J. Obstet. Gynaecol. Can. 2022, 44, 707–719.e1. [Google Scholar] [CrossRef]
- Jankovic-Karasoulos, T.; Furness, D.L.; Leemaqz, S.Y.; Dekker, G.A.; Grzeskowiak, L.E.; Grieger, J.A.; Andraweera, P.H.; McCullough, D.; McAninch, D.; McCowan, L.M.; et al. Maternal folate, one-carbon metabolism and pregnancy outcomes. Matern. Child Nutr. 2020, 17, e13064. [Google Scholar] [CrossRef]
- Tian, T.; Yang, K.-Q.; Cui, J.-G.; Zhou, L.-L.; Zhou, X.-L. Folic Acid Supplementation for Stroke Prevention in Patients with Cardiovascular Disease. Am. J. Med. Sci. 2017, 354, 379–387. [Google Scholar] [CrossRef]
- George, L.; Mills, J.L.; Johansson, A.L.; Nordmark, A.; Olander, B.; Granath, F.; Cnattingius, S. Plasma folate levels and risk of spontaneous abortion. JAMA 2002, 288, 1867–1873. [Google Scholar] [CrossRef]
- Wen, S.W.; Zhou, J.; Yang, Q.; Fraser, W.; Olatunbosun, O.; Walker, M. Maternal exposure to folic acid antagonists and placenta-mediated adverse pregnancy outcomes. CMAJ 2008, 179, 1263–1268. [Google Scholar] [CrossRef]
- Ding, Y.-X.; Cui, H. Effects of Folic Acid on DNMT1, GAP43, and VEGFR1 in Intrauterine Growth Restriction Filial Rats. Reprod. Sci. 2017, 25, 366–371. [Google Scholar] [CrossRef]
- Alvestad, S.; Husebye, E.S.N.; Christensen, J.; Dreier, J.W.; Sun, Y.; Igland, J.; Leinonen, M.K.; Gissler, M.; Gilhus, N.E.; Tomson, T.; et al. Folic Acid and Risk of Preterm Birth, Preeclampsia, and Fetal Growth Restriction Among Women with Epilepsy. Neurology 2022, 99, e605–e615. [Google Scholar] [CrossRef]
- Hovdenak, N.; Haram, K. Influence of mineral and vitamin supplements on pregnancy outcome. Eur. J. Obstet. Gynecol. Reprod. Biol. 2012, 164, 127–132. [Google Scholar] [CrossRef]
- Saccone, G.; Berghella, V. Folic acid supplementation in pregnancy to prevent preterm birth: A systematic review and meta-analysis of randomized controlled trials. Eur. J. Obstet. Gynecol. Reprod. Biol. 2016, 199, 76–81. [Google Scholar] [CrossRef]
- Sharif, M.E.; Mohamedain, A.; Ahmed, A.A.; Nasr, A.M.; Adam, I. Folic acid level and preterm birth among Sudanese women. Matern. Health Neonatol. Perinatol. 2017, 3, 25. [Google Scholar] [CrossRef]
- Li, B.; Zhang, X.; Peng, X.; Zhang, S.; Wang, X.; Zhu, C. Folic Acid and Risk of Preterm Birth: A Meta-Analysis. Front. Neurosci. 2019, 13, 1284. [Google Scholar] [CrossRef]
- Chen, X.; Zhang, Y.; Chen, H.; Jiang, Y.; Wang, Y.; Wang, D.; Li, M.; Dou, Y.; Sun, X.; Huang, G.; et al. Association of Maternal Folate and Vitamin B12 in Early Pregnancy with Gestational Diabetes Mellitus: A Prospective Cohort Study. Diabetes Care 2020, 44, 217–223. [Google Scholar] [CrossRef]
- Obeid, R.; Kasoha, M.; Kirsch, S.H.; Munz, W.; Herrmann, W. Concentrations of unmetabolized folic acid and primary folate forms in pregnant women at delivery and in umbilical cord blood. Am. J. Clin. Nutr. 2010, 92, 1416–1422. [Google Scholar] [CrossRef]
- Ahmed, T.; Fellus, I.; Gaudet, J.; MacFarlane, A.J.; Fontaine-Bisson, B.; Bainbridge, S.A. Effect of folic acid on human trophoblast health and function in vitro. Placenta 2016, 37, 7–15. [Google Scholar] [CrossRef]
- Luan, Y.; Leclerc, D.; Cosín-Tomás, M.; Malysheva, O.V.; Wasek, B.; Bottiglieri, T.; Caudill, M.A.; Rozen, R. Moderate Folic Acid Supplementation in Pregnant Mice Results in Altered Methyl Metabolism and in Sex-Specific Placental Transcription Changes. Mol. Nutr. Food Res. 2021, 65, e2100197. [Google Scholar] [CrossRef]
- Johnson, J.D.; Louis, J.M. Does race or ethnicity play a role in the origin, pathophysiology, and outcomes of preeclampsia? An expert review of the literature. Am. J. Obstet. Gynecol. 2020, 226, S876–S885. [Google Scholar] [CrossRef]
- Yong, H.E.J.; Murthi, P.; Brennecke, S.P.; Moses, E.K. Genetic approaches in preeclampsia. In Preeclampsia. Methods and Protocols. Murthi, P., Vaillancourt, C., Eds.; Springer: New York, NY, USA, 2017; pp. 53–72. [Google Scholar]
- Lokki, A.I.; Heikkinen-Eloranta, J.K.; Laivuori, H. The Immunogenetic Conundrum of Preeclampsia. Front. Immunol. 2018, 9, 2630. [Google Scholar] [CrossRef]
- Dunk, C.E.; Bucher, M.; Zhang, J.; Hayder, H.; Geraghty, D.E.; Lye, S.J.; Myatt, L.; Hackmon, R. Human Leukocyte Antigen HLA-C, HLA-G, HLA-F and HLA-E placental profiles are altered in Early Severe Preeclampsia and Preterm Birth with Chorioamnionitis. Am. J. Obstet. Gynecol. 2022, 227, 641.e1–641.e13. [Google Scholar] [CrossRef] [PubMed]
- Loisel, D.A.; Billstrand, C.; Murray, K.; Patterson, K.; Chaiworapongsa, T.; Romero, R.; Ober, C. The maternal HLA-G 1597 C null mutation is associated with increased risk of pre-eclampsia and reduced HLA-G expression during pregnancy in African-American women. Mol. Hum. Reprod. 2012, 19, 144–152. [Google Scholar] [CrossRef]
- Eche, S.; Mackraj, I.; Moodley, J. Circulating fetal and total cell-free DNA, and sHLA-G in black South African women with gestational hypertension and pre-eclampsia. Hypertens. Pregnancy 2017, 36, 295–301. [Google Scholar] [CrossRef]
- Phoswa, W.N.; Ramsuran, V.; Naicker, T.; Singh, R.; Moodley, J. HLA-G Polymorphisms Associated with HIV Infection and Preeclampsia in South Africans of African Ancestry. BioMed Res. Int. 2020, 2020, 1697657. [Google Scholar] [CrossRef] [PubMed]
- McGinnis, R.; The FINNPEC Consortium; Steinthorsdottir, V.; O Williams, N.; Thorleifsson, G.; Shooter, S.; Hjartardottir, S.; Bumpstead, S.; Stefansdottir, L.; Hildyard, L.; et al. Variants in the fetal genome near FLT1 are associated with risk of preeclampsia. Nat. Genet. 2017, 49, 1255–1260. [Google Scholar] [CrossRef] [PubMed]
- Gray, K.; Saxena, R.; Karumanchi, S.A. Genetic predisposition to preeclampsia is conferred by fetal DNA variants near FLT1, a gene involved in the regulation of angiogenesis. Am. J. Obstet. Gynecol. 2017, 218, 211–218. [Google Scholar] [CrossRef]
- Kikas, T.; Inno, R.; Ratnik, K.; Rull, K.; Laan, M. C-allele of rs4769613 Near FLT1 Represents a High-Confidence Placental Risk Factor for Preeclampsia. Hypertension 2020, 76, 884–891. [Google Scholar] [CrossRef]
- Say, L.; Chou, D.; Gemmill, A.; Tunçalp, Ö.; Moller, A.-B.; Daniels, J.; Gülmezoglu, A.M.; Temmerman, M.; Alkema, L. Global causes of maternal death: A WHO systematic analysis. Lancet Glob. Health 2014, 2, E323–E333. [Google Scholar] [CrossRef]
- Geller, S.E.; Koch, A.R.; Garland, C.E.; MacDonald, E.J.; Storey, F.; Lawton, B. A global view of severe maternal morbidity: Moving beyond maternal mortality. Reprod. Health 2018, 15, 31–43. [Google Scholar] [CrossRef]
- McLaren, Z.M.; Sharp, A.; Hessburg, J.P.; Sarvestani, A.S.; Parker, E.; Akazili, J.; Johnson, T.R.; Sienko, K.H. Cost effectiveness of medical devices to diagnose pre-eclampsia in low-resource settings. Dev. Eng. 2017, 2, 99–106. [Google Scholar] [CrossRef]
- Wallis, A.B.; Saftlas, A.F.; Hsia, J.; Atrash, H.K. Secular Trends in the Rates of Preeclampsia, Eclampsia, and Gestational Hypertension, United States, 1987–2004. Am. J. Hypertens. 2008, 21, 521–526. [Google Scholar] [CrossRef] [PubMed]
- Roberts, J.; Cooper, D. Pathogenesis and genetics of pre-eclampsia. Lancet 2001, 357, 53–56. [Google Scholar] [CrossRef] [PubMed]
- Mishra, J.; Talwar, S.; Kaur, L.; Chandiok, K.; Yadav, S.; Puri, M.; Sachdeva, M.; Lakshmy, R.; Saraswathy, K. Differential global and MTHFR gene specific methylation patterns in preeclampsia and recurrent miscarriages: A case-control study from North India. Gene 2019, 704, 68–73. [Google Scholar] [CrossRef] [PubMed]
- Moussa, H.N.; Nasab, S.H.; A Haidar, Z.; Blackwell, S.C.; Sibai, B.M. Folic acid supplementation: What is new? Fetal, obstetric, long-term benefits and risks. Future Sci. OA 2016, 2, FSO116. [Google Scholar] [CrossRef]
- Cotter, A.M.; Molloy, A.; Scott, J.M.; Daly, S.F. Elevated plasma homocysteine in early pregnancy: A risk factor for the development of severe preeclampsia. Am. J. Obstet. Gynecol. 2001, 185, 781–785. [Google Scholar] [CrossRef]
- Mujawar, S.A.; Patil, V.W.; Daver, R.G. Study of Serum Homocysteine, Folic Acid and Vitamin B12 in Patients with Preeclampsia. Indian J. Clin. Biochem. 2011, 26, 257–260. [Google Scholar] [CrossRef]
- Kharb, S.; Aggarwal, D.; Bala, J.; Nanda, S. Evaluation of Homocysteine, Vitamin B12 and Folic Acid Levels During all the Trimesters in Pregnant and Preeclamptic Womens. Curr. Hypertens. Rev. 2017, 12, 234–238. [Google Scholar] [CrossRef]
- Guven, M.A.; Coskun, A.; Ertas, I.E.; Aral, M.; Zencırcı, B.; Oksuz, H. Association of Maternal Serum CRP, IL-6, TNF-α, Homocysteine, Folic Acid and Vitamin B12 Levels with the Severity of Preeclampsia and Fetal Birth Weight. Hypertens. Pregnancy 2009, 28, 190–200. [Google Scholar] [CrossRef]
- Kharb, S.; Nanda, S. Patterns of Biomarkers in Cord Blood During Pregnancy and Preeclampsia. Curr. Hypertens. Rev. 2017, 13, 57–64. [Google Scholar] [CrossRef]
- Acilmis, Y.G.; Dikensoy, E.; Kutlar, A.I.; Balat, O.; Cebesoy, F.B.; Ozturk, E.; Cicek, H.; Pence, S. Homocysteine, folic acid and vitamin B12 levels in maternal and umbilical cord plasma and homocysteine levels in placenta in pregnant women with pre-eclampsia. J. Obstet. Gynaecol. Res. 2010, 37, 45–50. [Google Scholar] [CrossRef]
- McNulty, B.; McNulty, H.; Marshall, B.; Ward, M.; Molloy, A.M.; Scott, J.M.; Dornan, J.; Pentieva, K. Impact of continuing folic acid after the first trimester of pregnancy: Findings of a randomized trial of Folic Acid Supplementation in the Second and Third Trimesters. Am. J. Clin. Nutr. 2013, 98, 92–98. [Google Scholar] [CrossRef]
- Wen, S.W.; Chen, X.-K.; Rodger, M.; White, R.R.; Yang, Q.; Smith, G.; Sigal, R.J.; Perkins, S.L.; Walker, M.C. Folic acid supplementation in early second trimester and the risk of preeclampsia. Am. J. Obstet. Gynecol. 2008, 198, 45.e1–45.e7. [Google Scholar] [CrossRef]
- Wen, S.W.; Guo, Y.; Rodger, M.; White, R.R.; Yang, Q.; Smith, G.N.; Perkins, S.L.; Walker, M.C. Folic Acid Supplementation in Pregnancy and the Risk of Pre-Eclampsia—A Cohort Study. PLoS ONE 2016, 11, e0149818. [Google Scholar] [CrossRef]
- Wang, Y.; Zhao, N.; Qiu, J.; He, X.; Zhou, M.; Cui, H.; Lv, L.; Lin, X.; Zhang, C.; Zhang, H.; et al. Folic acid supplementation and dietary folate intake, and risk of preeclampsia. Eur. J. Clin. Nutr. 2015, 69, 1145–1150. [Google Scholar] [CrossRef]
- Vanderlelie, J.; Scott, R.; Shibl, R.; Lewkowicz, J.; Perkins, A.; Scuffham, P. First trimester multivitamin/mineral use is associated with reduced risk of pre-eclampsia among overweight and obese women. Matern. Child Nutr. 2014, 12, 339–348. [Google Scholar] [CrossRef]
- Han, T.B.; Guo, L.L.; Yang, F.F.; Zhao, F.; Du, W.Q.; Wang, Y.; Shen, J.X.; Feng, Y.L.; Yang, H.L.; Zhang, Y.W.; et al. Folic acid supplementation before and during pregnancy and the risk of preeclampsia. Zhonghua Liu Xing Bing Xue Za Zhi 2020, 41, 1894–1899. [Google Scholar]
- Kim, M.W.; Ahn, K.H.; Ryu, K.-J.; Hong, S.-C.; Lee, J.S.; Nava-Ocampo, A.A.; Oh, M.-J.; Kim, H.-J. Preventive Effects of Folic Acid Supplementation on Adverse Maternal and Fetal Outcomes. PLoS ONE 2014, 9, e97273. [Google Scholar] [CrossRef]
- Martinussen, M.P.; Bracken, M.B.; Triche, E.W.; Jacobsen, G.W.; Risnes, K.R. Folic acid supplementation in early pregnancy and the risk of preeclampsia, small for gestational age offspring and preterm delivery. Eur. J. Obstet. Gynecol. Reprod. Biol. 2015, 195, 94–99. [Google Scholar] [CrossRef]
- Zheng, L.; Huang, J.; Kong, H.; Wang, F.; Su, Y.; Xin, H. The effect of folic acid throughout pregnancy among pregnant women at high risk of pre-eclampsia: A randomized clinical trial. Pregnancy Hypertens. 2020, 19, 253–258. [Google Scholar] [CrossRef]
- Li, Z.; Ye, R.; Zhang, L.; Li, H.; Liu, J.; Ren, A. Folic Acid Supplementation During Early Pregnancy and the Risk of Gestational Hypertension and Preeclampsia. Hypertension 2013, 61, 873–879. [Google Scholar] [CrossRef]
- Ayala-Ramírez, P.; Serrano, N.; Barrera, V.; Bejarano, J.P.; Silva, J.L.; Martínez, R.; Gil, F.; Olaya-C, M.; García-Robles, R. Risk factors and fetal outcomes for preeclampsia in a Colombian cohort. Heliyon 2020, 6, e05079. [Google Scholar] [CrossRef] [PubMed]
- Corsi, D.J.; Gaudet, L.M.; El-Chaar, D.; White, R.R.; Rybak, N.; Harvey, A.; Muldoon, K.; Wen, S.W.; Walker, M. Effect of high-dose folic acid supplementation on the prevention of preeclampsia in twin pregnancy. J. Matern. Neonatal Med. 2020, 35, 503–508. [Google Scholar] [CrossRef] [PubMed]
- Antoniades, C.; Shirodaria, C.; Warrick, N.; Cai, S.; De Bono, J.; Lee, J.; Leeson, P.; Neubauer, S.; Ratnatunga, C.; Pillai, R.; et al. 5-Methyltetrahydrofolate Rapidly Improves Endothelial Function and Decreases Superoxide Production in Human Vessels. Circulation 2006, 114, 1193–1201. [Google Scholar] [CrossRef] [PubMed]
- Antoniades, C.; Shirodaria, C.; Leeson, P.; Baarholm, O.A.; Van-Assche, T.; Cunnington, C.; Pillai, R.; Ratnatunga, C.; Tousoulis, D.; Stefanadis, C.; et al. MTHFR 677 C>T Polymorphism Reveals Functional Importance for 5-Methyltetrahydrofolate, Not Homocysteine, in Regulation of Vascular Redox State and Endothelial Function in Human Atherosclerosis. Circulation 2009, 119, 2507–2515. [Google Scholar] [CrossRef]
- Yuyun, M.F.; Ng, L.; Ng, G.A. Endothelial dysfunction, endothelial nitric oxide bioavailability, tetrahydrobiopterin, and 5-methyltetrahydrofolate in cardiovascular disease. Where are we with therapy? Microvasc. Res. 2018, 119, 7–12. [Google Scholar] [CrossRef]
| Authors | Population | Recommendations | Reference | |
|---|---|---|---|---|
| Dose | Time and Duration | |||
| Manizheh et al., 2009 | Iranian | Folic acid, 500 μg/day–5 mg/day | From early pregnancy until delivery | [12] |
| Kim et al., 2014 | South Korean | Folic acid, 400 μg/day–1.0 mg/day daily for at least | Two-three months before conception and throughout pregnancy | [101] |
| Martinussen et al., 2015 | USA | Folic acid, <200 μg/day, 200 < 600 μg/day, ≥600 μg/day | One month before pregnancy, first-trimester pregnancy | [102] |
| Wang et al., 2015 | Chinese | Dietary folic acid intake (from 151.6 μg/day to ≥274 μg/day) | Throughout the whole pregnancy | [98] |
| Vanderlelie et al., 2016 | Australian | Folic acid, 800 μg/day | The first trimester of pregnancy | [99] |
| Wen et al., 2016 | Canadian | Folic acid, ≥1.0 mg/day | First and early second trimester of pregnancy | [97] |
| Han et al., 2020 | Chinese | Folic acid, <400, 400, and >400 μg/day | Before and during pregnancy | [100] |
| Zheng et al., 2020 | Chinese | Folic acid, 4 mg/day | Three months before pregnancy for the entire pregnancy | [103] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kaldygulova, L.; Ukybassova, T.; Aimagambetova, G.; Gaiday, A.; Tussupkaliyev, A. Biological Role of Folic Acid in Pregnancy and Possible Therapeutic Application for the Prevention of Preeclampsia. Biomedicines 2023, 11, 272. https://doi.org/10.3390/biomedicines11020272
Kaldygulova L, Ukybassova T, Aimagambetova G, Gaiday A, Tussupkaliyev A. Biological Role of Folic Acid in Pregnancy and Possible Therapeutic Application for the Prevention of Preeclampsia. Biomedicines. 2023; 11(2):272. https://doi.org/10.3390/biomedicines11020272
Chicago/Turabian StyleKaldygulova, Lyazzat, Talshyn Ukybassova, Gulzhanat Aimagambetova, Andrey Gaiday, and Akylbek Tussupkaliyev. 2023. "Biological Role of Folic Acid in Pregnancy and Possible Therapeutic Application for the Prevention of Preeclampsia" Biomedicines 11, no. 2: 272. https://doi.org/10.3390/biomedicines11020272
APA StyleKaldygulova, L., Ukybassova, T., Aimagambetova, G., Gaiday, A., & Tussupkaliyev, A. (2023). Biological Role of Folic Acid in Pregnancy and Possible Therapeutic Application for the Prevention of Preeclampsia. Biomedicines, 11(2), 272. https://doi.org/10.3390/biomedicines11020272






