Serum Magnesium Levels in Patients with Obstructive Sleep Apnoea: A Systematic Review and Meta-Analysis
Abstract
1. Introduction
2. Materials and Methods
2.1. Selection Criteria
2.2. Data Extraction and Meta-Analysis
2.3. Assessment of Methodological Quality
3. Results
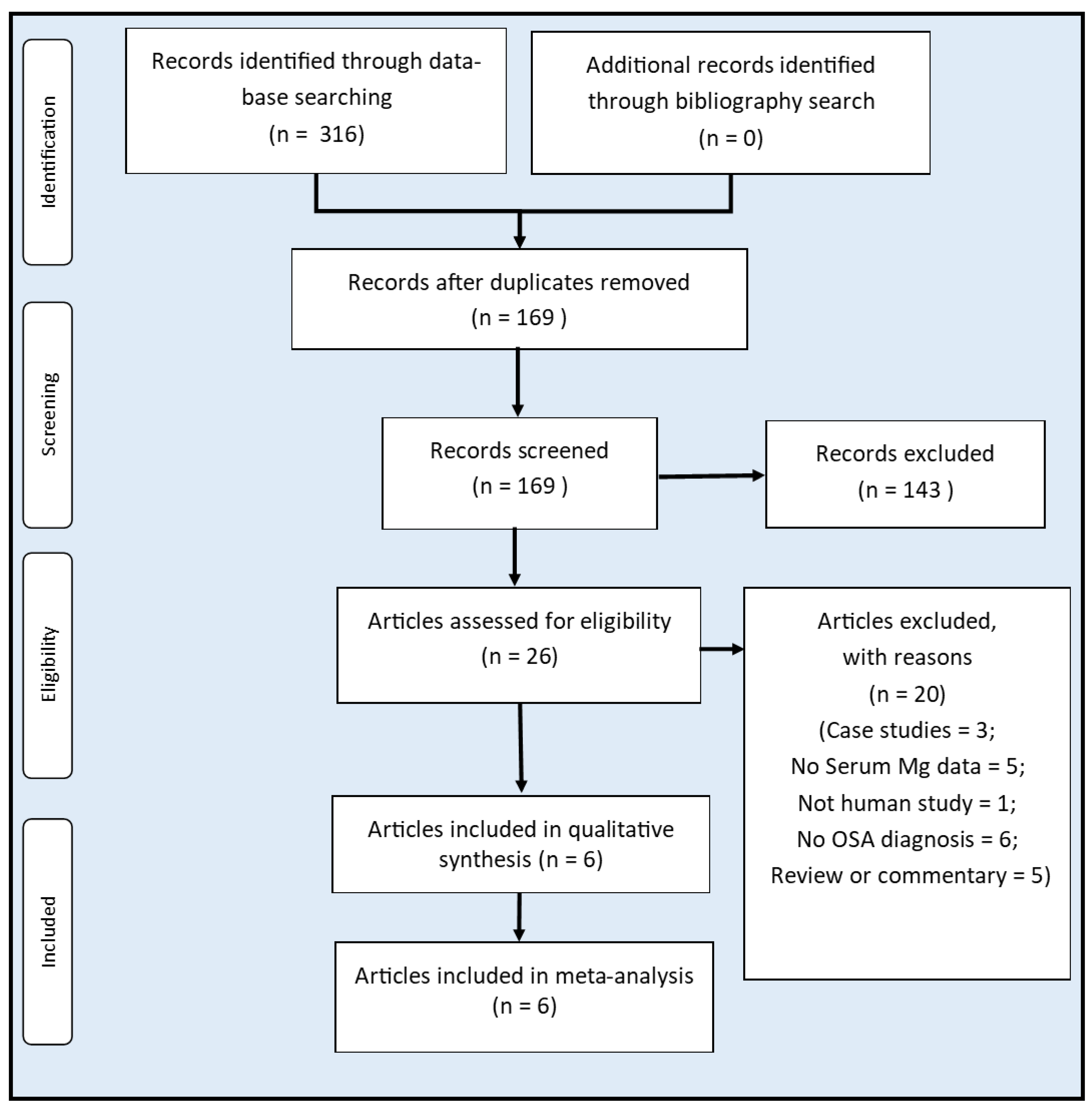
3.1. Search Results
3.2. Serum Mg and Risk of Mg Deficiency
3.2.1. Comparing to Healthy Controls
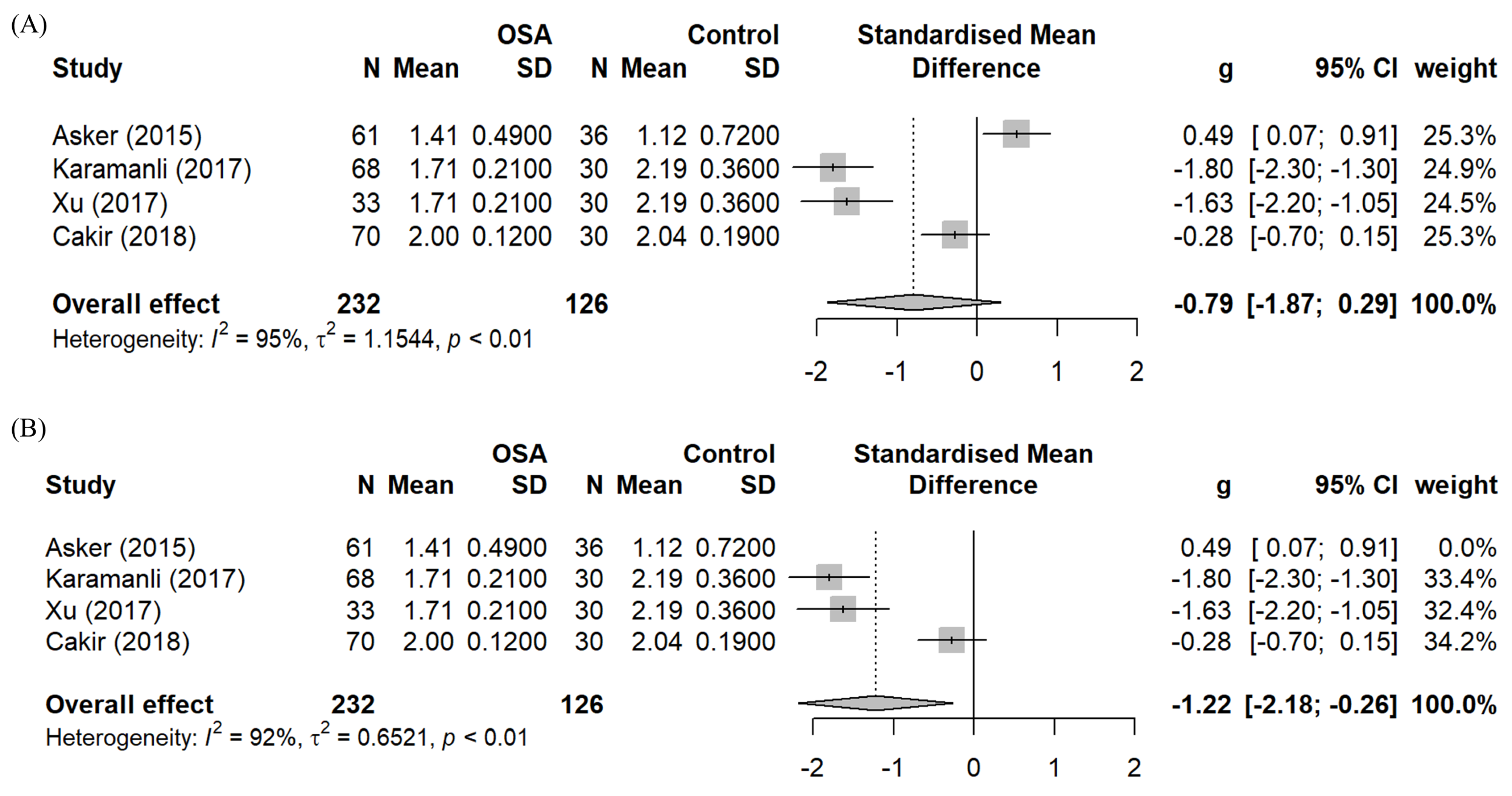
3.2.2. Meta-Analysis—Effect Size
3.2.3. Meta-Analysis—Pooled Mean
3.3. Serum Mg and OSA Severity
3.3.1. AHI
3.3.2. Before and after Treatment
3.4. Serum Mg and Biomarkers
3.4.1. Lipid Profile
3.4.2. Glucose Metabolism
3.4.3. Trace Minerals and Heavy Metals
3.4.4. C-Reactive Protein, Ischemia-Modified Albumin, and Carotid Intima-Media Thickness
3.5. Quality Assessment
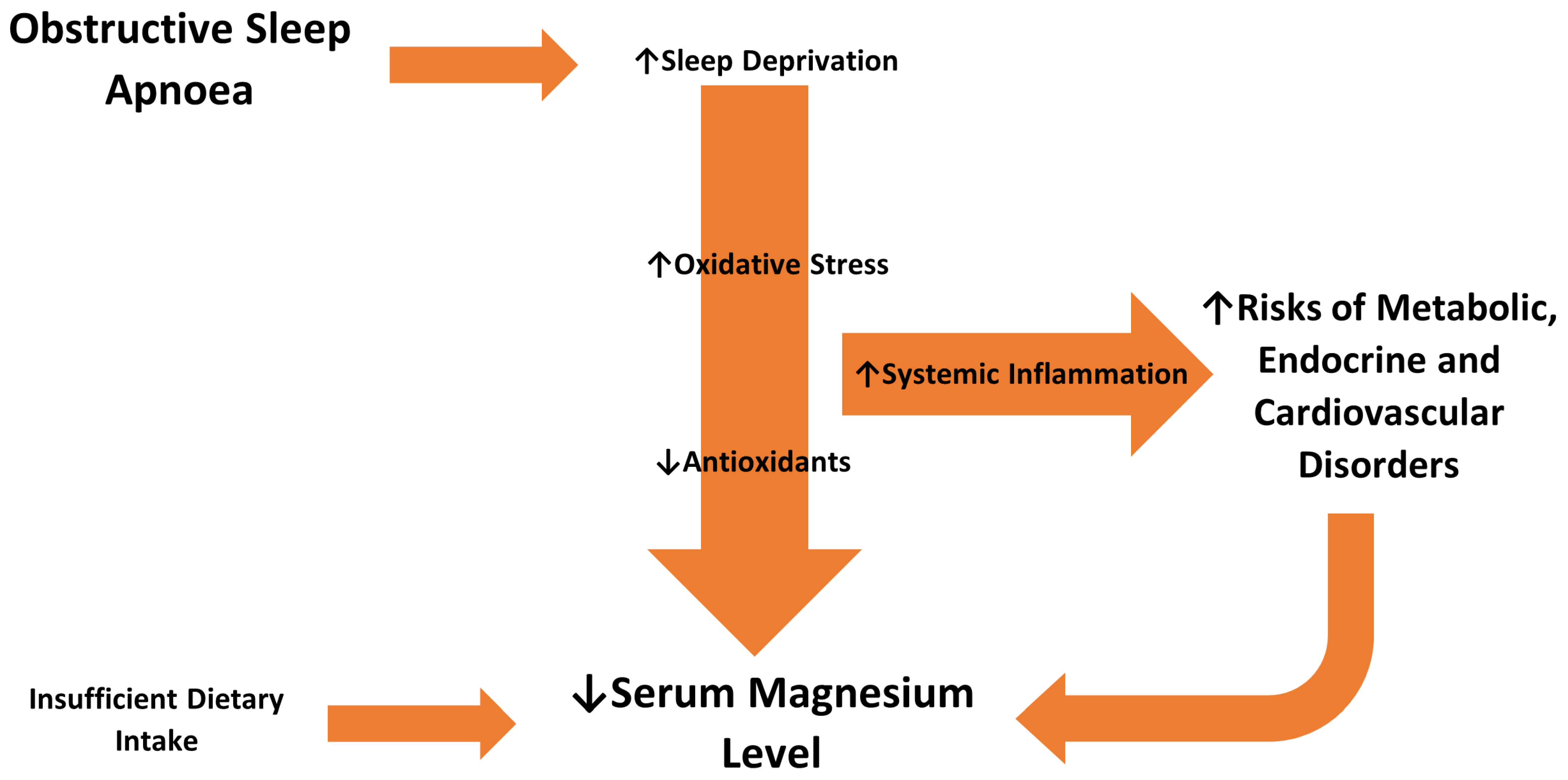
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| AHI | Apnoea-hypopnoea index |
| AUROC | Area under the receiver operating characteristics |
| BMI | Body mass index |
| Ca | Calcium |
| CPAP | Continuous positive airway pressure |
| CRP | C-reactive protein |
| CI | Confidence interval |
| CIMT | Carotid intima-media thickness |
| ESR | Erythrocyte sedimentation rate |
| HDL | High-density lipoproteins |
| hsCRP | high sensitivity CRP |
| IMA | Ischemia-modified albumin |
| LDL | Low-density lipoproteins |
| Mg | Magnesium |
| N | Sample size |
| OSA | Obstructive sleep apnoea |
| PSG | Polysomnography |
| PWV | Pulse wave velocity |
| RYGB | Roux-en-Y gastric bypass |
| T2DM | Type 2 diabetes mellitus |
| SD | Standard deviation |
References
- Osman, A.M.; Carter, S.G.; Carberry, J.C.; Eckert, D.J. Obstructive sleep apnea: Current perspectives. Nat. Sci. Sleep 2018, 10, 21–34. [Google Scholar] [CrossRef] [PubMed]
- Spicuzza, L.; Caruso, D.; Maria, G.D. Obstructive sleep apnoea syndrome and its management. Ther. Adv. Chronic Dis. 2015, 6, 273–285. [Google Scholar] [CrossRef]
- Stansbury, R.C.; Strollo, P.J. Clinical manifestations of sleep apnea. J. Thorac. Dis. 2015, 7, E298–E310. [Google Scholar] [CrossRef] [PubMed]
- Schwalfenberg, G.K.; Genuis, S.J. The importance of magnesium in clinical healthcare. Scientifica 2017, 2017, 4179326. [Google Scholar] [CrossRef] [PubMed]
- Romani, A.M.P. Magnesium in health and disease. In Interrelations between Essential Metal Ions and Human Diseases. Metal Ions in Life Sciences; Sigel, A., Sigel, H., Sigel, R., Eds.; Springer: Dordrecht, The Netherlands, 2013; Volume 13, pp. 49–79. [Google Scholar] [CrossRef]
- Jahnen-Dechent, W.; Ketteler, M. Magnesium basics. Clin. Kidney J. 2012, 5, i3–i14. [Google Scholar] [CrossRef]
- King, D.E.; Mainous, A.G.; Geesey, M.E.; Egan, B.M.; Rehman, S. Magnesium supplement intake and C-reactive protein levels in adults. Nutr. Res. 2006, 26, 193–196. [Google Scholar] [CrossRef]
- Australian Bureau of Statistics. Australian Health Survey: Usual Nutrient Intakes; 2011-12 (Cat. No 4364.0.55.008); Technical Report; Australian Bureau of Statistics: Canberra, Australia, 2015.
- DiNicolantonio, J.J.; O’Keefe, J.H.; Wilson, W. Subclinical magnesium deficiency: A principal driver of cardiovascular disease and a public health crisis. Open Heart 2018, 5, e000668. [Google Scholar] [CrossRef]
- Costello, R.B.; Nielsen, F. Interpreting magnesium status to enhance clinical care: Key indicators. Curr. Opin. Clin. Nutr. Metab. Care 2017, 20, 504–511. [Google Scholar] [CrossRef]
- Costello, R.B.; Elin, R.J.; Rosanoff, A.; Wallace, T.C.; Guerrero-Romero, F.; Hruby, A.; Lutsey, P.L.; Nielsen, F.H.; Rodriguez-Moran, M.; Song, Y.; et al. Perspective: The case for an evidence-based reference interval for serum magnesium: The time has come. Adv. Nutr. 2016, 7, 977–993. [Google Scholar] [CrossRef]
- Tangvoraphonkchai, K.; Davenport, A. Magnesium and cardiovascular disease. Adv. Chronic Kidney Dis. 2018, 25, 251–260. [Google Scholar] [CrossRef]
- Ju, S.Y.; Choi, W.S.; Ock, S.M.; Kim, C.M.; Kim, D.H. Dietary magnesium intake and metabolic syndrome in the adult population: Dose-response meta-analysis and meta-regression. Nutrients 2014, 6, 6005–6019. [Google Scholar] [CrossRef] [PubMed]
- Barbagallo, M.; Dominguez, L.J. Magnesium and type 2 diabetes. World J. Diabetes 2015, 6, 1152–1157. [Google Scholar] [CrossRef] [PubMed]
- Hassan, S.A.U.; Ahmed, I.; Nasrullah, A.; Haq, S.; Ghazanfar, H.; Sheikh, A.B.; Zafar, R.; Askar, G.; Hamid, Z.; Khushdil, A.; et al. Comparison of serum magnesium levels in overweight and obese children and normal weight children. Cureus 2017, 9, e1607. [Google Scholar] [CrossRef] [PubMed]
- Hein, M.; Lanquart, J.P.; Loas, G.; Hubain, P.; Linkowski, P. Prevalence and risk factors of moderate to severe obstructive sleep apnea syndrome in insomnia sufferers: A study on 1311 subjects. Respir. Res. 2017, 18, 135. [Google Scholar] [CrossRef]
- Yacoub, M.; Youssef, I.; Salifu, M.O.; McFarlane, S.I. Cardiovascular Disease Risk in Obstructive Sleep apnea: An Update. J. Sleep Disord. Ther. 2017, 7, 283. [Google Scholar] [CrossRef]
- Alawi, A.M.A.; Majoni, S.W.; Falhammar, H. Magnesium and human health: Perspectives and research directions. Int. J. Endocrinol. 2018, 2018, 9041694. [Google Scholar] [CrossRef]
- Drahota, A.; Beller, E. RevMan Calculator. Cochrane Training. Available online: https://training.cochrane.org/resource/revman-calculator (accessed on 8 December 2020).
- National Institutes of Health. Quality Assessment Tool for Observational Cohort and Cross-Sectional Studies. Available online: https://www.nhlbi.nih.gov/health-topics/study-quality-assessment-tools (accessed on 8 December 2020).
- Asker, S.; Asker, M.; Yeltekin, A.C.; Aslan, M.; Demir, H. Serum levels of trace minerals and heavy metals in severe obstructive sleep apnea patients: Correlates and clinical implications. Sleep Breath. 2015, 19, 547–552. [Google Scholar] [CrossRef]
- Jiao, X.; Zou, J.; Zhang, P.; Yu, H.; Di, J.; Han, X.; Yin, S.; Yi, H. Roux-en-Y Gastric Bypass Surgery on Obstructive Sleep Apnea-Hypopnea Syndrome: Factors Associated with Postoperative Efficacy. Obes. Surg. 2016, 26, 2924–2930. [Google Scholar] [CrossRef]
- Karamanli, H.; Kizilirmak, D.; Akgedik, R.; Bilgi, M. Serum levels of magnesium and their relationship with CRP in patients with OSA. Sleep Breath. 2017, 21, 549–556. [Google Scholar] [CrossRef]
- Xu, Q.; Du, J.; Ling, X.; Lu, Y. Evaluation of MIh scoring system in diagnosis of obstructive sleep apnea syndrome. Med Sci. Monit. Int. Med. J. Exp. Clin. Res. 2017, 23, 4715–4722. [Google Scholar] [CrossRef]
- Çakır, I.; Uluhan, M. Evaluation of metabolic risk markers: Calcium/magnesium ratio, lipoproteins and insulin resistance in patients with obstructive sleep apnea syndrome. Erciyes Med. J. 2018, 40, 131–135. [Google Scholar] [CrossRef]
- Zota, I.M.; Sascau, R.; Statescu, C.; Roca, M.; Boisteanu, D.; Constantin, M.M.L.; Mastaleru, A.; Vasilcu, T.; Gavril, R.; Mitu, F. Arterial stiffness in moderate-severe obstructive sleep apnea. Rom. J. Cardiol. Vol. 2019, 29, 27–32. [Google Scholar]
- Rodrigues, A.K.; Melo, A.E.; Domingueti, C.P. Association between reduced serum levels of magnesium and the presence of poor glycemic control and complications in type 1 diabetes mellitus: A systematic review and meta-analysis. Diabetes Metab. Syndr. 2020, 14, 127–134. [Google Scholar] [CrossRef]
- Severino, P.; Netti, L.; Mariani, M.V.; Maraone, A.; D’Amato, A.; Scarpati, R.; Infusino, F.; Pucci, M.; Lavalle, C.; Maestrini, V.; et al. Prevention of cardiovascular disease: Screening for magnesium deficiency. Cardiol. Res. Pract. 2019, 2019, 4874921. [Google Scholar] [CrossRef] [PubMed]
- Ismail, A.A.; Ismail, N.A. Magnesium: A mineral essential for health yet generally underestimated or even ignored. J. Nutr. Food Sci. 2016, 6, 4–11. [Google Scholar] [CrossRef]
- Akanmu, M.A.; Meludu, S.C.; Honda, K. Effects of 6-hour total sleep deprivation on plasma magnesium and calcium levels in rats. Sleep Biol. Rhythm. 2003, 1, 97–100. [Google Scholar] [CrossRef]
- Tanabe, K.; Yamamoto, A.; Suzuki, N.; Osada, N.; Yokoyama, Y.; Samejima, H.; Seki, A.; Oya, M.; Murabayashi, T.; Nakayama, M.; et al. Efficacy of oral magnesium administration on decreased exercise tolerance in a state of chronic sleep deprivation. Jpn. Circ. J. 1998, 62, 341–346. [Google Scholar] [CrossRef][Green Version]
- Orrù, G.; Storari, M.; Scano, A.; Piras, V.; Taibi, R.; Viscuso, D. Obstructive Sleep Apnea, oxidative stress, inflammation and endothelial dysfunction-An overview of predictive laboratory biomarkers. Eur. Rev. Med. Pharmacol. Sci. 2020, 24, 6939–6948. [Google Scholar] [CrossRef]
- Nielsen, F.H.; Johnson, L.K.; Zeng, H. Magnesium supplementation improves indicators of low magnesium status and inflammatory stress in adults older than 51 years with poor quality sleep. Magnes. Res. 2010, 23, 158–168. [Google Scholar] [CrossRef]
- Kaliaperumal, R.; Venkatachalam, R.; Nagarajan, P.; Sabapathy, S.K. Association of serum magnesium with oxidative stress in the pathogenesis of diabetic cataract. Biol. Trace Elem. Res. 2021, 199, 2869–2873. [Google Scholar] [CrossRef]
- Volná, J.; Kemlink, D.; Kalousová, M.; Vávrová, J.; Majerová, V.; Mestek, O.; Svarcová, J.; Sonka, K.; Zima, T. Biochemical oxidative stress-related markers in patients with obstructive sleep apnea. Med Sci. Monit. Int. Med. J. Exp. Clin. Res. 2011, 17, CR491–CR497. [Google Scholar] [CrossRef] [PubMed]
- Tapiero, H.; Townsend, D.M.; Tew, K.D. Trace elements in human physiology and pathology. Copper. Biomed. Pharmacother. 2003, 57, 386–398. [Google Scholar] [CrossRef]
- Kocyigit, A.; Armutcu, F.; Gurel, A.; Ermis, B. Alterations in plasma essential trace elements selenium, manganese, zinc, copper, and iron concentrations and the possible role of these elements on oxidative status in patients with childhood asthma. Biol. Trace Elem. Res. 2004, 97, 31–41. [Google Scholar] [CrossRef]
- O’Connor, J.M. Trace elements and DNA damage. Biochem. Soc. Trans. 2001, 29, 354–357. [Google Scholar] [CrossRef] [PubMed]
- Krachler, M.; Rossipal, E.; Micetic-Turk, D. Concentrations of trace elements in sera of newborns, young infants, and adults. Biol. Trace Elem. Res. 1999, 68, 121. [Google Scholar] [CrossRef]
- Wyparło-Wszelaki, M.; Wąsik, M.; Machoń-Grecka, A.; Kasperczyk, A.; Bellanti, F.; Kasperczyk, S.; Dobrakowski, M. Blood magnesium level and selected oxidative stress indices in lead-exposed workers. Biol. Trace Elem. Res. 2021, 199, 465–472. [Google Scholar] [CrossRef]
- Bronkowska, M.; Gołecki, M.; Słomian, J.; Mikołajczak, J.; Kosacka, M.; Porȩbska, I.; Jankowska, R.; Biernat, J. Evaluation of vitamin and mineral intake in the daily food rations of overweight and obese patients diagnosed with obstructive sleep apnea. Adv. Clin. Exp. Med. 2010, 19, 721–722. [Google Scholar]
- Cao, Y.; Zhen, S.; Taylor, A.W.; Appleton, S.; Atlantis, E.; Shi, Z. Magnesium intake and sleep disorder symptoms: Findings from the Jiangsu nutrition study of Chinese adults at five-year follow-up. Nutrients 2018, 10, 1354. [Google Scholar] [CrossRef]
- Page, M.J.; McKenzie, J.E.; Bossuyt, A.W.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. BMJ 2021, 372, n71. [Google Scholar] [CrossRef]


| Study and Design | Participants | Objective | Findings: Mean Serum Mg (mg/dL) and Metabolic Parameters | Ref. |
|---|---|---|---|---|
| Asker (2015) Turkey Cross-sectional case-control study | OSA: AHI ≥ 30, n = 61. Male/Female: 42/19. Mean age: 48.2 ± 9.1. Control: healthy volunteers, n = 36. Male/Female: 11/25. Mean age: 46.9 ± 10.2. | Evaluation of serum levels of trace minerals and heavy metals in severe OSA patients before any therapeutic intervention. | OSA: 1.413 ± 0.488; Control: 1.12 ± 0.722; OSA > Control (p < 0.001). The OSA group had significantly higher cholesterol, triglycerides, trace minerals, heavy minerals, and CIMT compared to the control (p < 0.005 for all). CIMT and triglycerides were positively correlated with Mg. HDL was negatively correlated with Mg. | [21] |
| Jiao (2016) China Case-control follow up observational study | OSA patients (AHI ≥ 5) with obesity and T2DM receiving RYGB surgery, n = 39. Male/Female: 15/24. Mean age: 44.20 ± 8.95 (Males); 50.50 ± 12.29 (Females). | Assessing the efficacy of RYGB surgery on patients with OSA using PSG and biochemical tests (including Mg levels), pre- and post-surgery. Study duration: 6–12 months post-intervention. | Preoperative: 2.04 ± 0.17; Postoperative: 2.19 ± 0.17; Post > Pre (p < 0.001). RYGB surgery significantly lowered the AHI and BMI in OSA patients. Postoperative blood Mg levels were significantly increased in OSA patients when compared to the time of diagnosis (p < 0.05). Mg was not correlated with the improvement in AHI. | [22] |
| Karamanli (2017) Turkey Retrospective cross-sectional study | OSA: AHI ≥ 5, n = 68. Male/Female: 46/22. Control: healthy volunteers, n = 30. Male/Female: 14/16. | To evaluate the relationship between serum levels of Mg and the inflammatory response (CRP) in patients with newly diagnosed OSA. | OSA: 1.71 ± 0.21; Control: 2.19 ± 0.36; OSA < Control (p < 0.0001). Mg levels were lower in OSA patients than those in controls. Those with severe OSA also had significantly lower Mg (p = 0.03) than those with mild OSA. OSA group had a significantly higher CRP. A significant negative correlation was observed between Mg and CRP levels (p < 0.0001). | [23] |
| Xu (2017) China Case-control study with subgroup follow-up | OSA: AHI ≥ 5, n = 33. Male/Female: 23/10. Mean age: 51.6 ± 9.8. Control: healthy volunteers, n = 30. Male/Female: 21/9. Mean age: 52.1 ± 10.9. | Investigating Mg, hsCRP, and IMA as a non-invasive diagnosis method for OSA. Participants followed up after 3 months of CPAP treatment. | OSA: 1.71 ± 0.21; Control: 2.19 ± 0.36; OSA < Control (p = 0.021). Post-CPAP: 2.02 ± 0.25 (n = 22 only); Post > Baseline (p < 0.001)
OSA patients had significantly higher levels of hsCRP and IMA and significantly lower Mg compared to control (p < 0.05 for all). Mg, hsCRP, and IMA were used in a novel model to diagnose OSA with AUROC of 0.93 (0.83–0.98). OSA patients showed significant improvements in Mg, hsCRP, and IMA after CPAP treatment. | [24] |
| Cakir (2018) Turkey Clinical case-control study | OSA: AHI ≥ 5, n = 70 (55 severe, 11 moderate and 4 mild). Male only. Mean age: 47.57 ± 12.15. Control: non-apnoeic individuals (AHI < 5), n = 30. Male only. Mean age: 43.23 ± 10.5. | Comparing OSA patients’ metabolic markers (BMI, PSG, insulin sensitivity–resistance markers, lipid profiles) and mineral levels with those of control subjects. | OSA: 2.0 ± 0.12; Control: 2.04 ± 0.19.
There were no statistically significant differences in serum Mg, Ca, and Ca/Mg ratios between OSA patients and controls. Fasting glucose and insulin levels were significantly higher in the OSA group (p < 0.05 for all). Fasting glucose levels were correlated with Ca, Mg, and Ca/Mg ratios. Severe OSA patients had significantly higher Ca/Mg ratios than mild/moderate groups (p = 0.017). | [25] |
| Zota (2019) Romania Cross-sectional, single cohort study | OSA: AHI ≥ 5, n = 41 (23 severe and 18 moderate/mild). Mean age: 55.83 (moderate/mild), 57.34 (severe). | Assessed the relationship between OSA severity, arterial stiffness and clinic-biological parameters in moderate-severe OSA patients prior to the use of CPAP therapy. | Moderate: 2.09; Severe: 1.9; SD not reported. Between-group not significant with p = 0.1. No significant difference was found between severe and moderate groups in Mg levels. ESR was significantly higher in the severe group (p = 0.012). Inflammation markers (CRP and ESR) were correlated with OSA severity. Mg was negatively correlated with PWV, which measured arterial stiffness. | [26] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Al Wadee, Z.; Ooi, S.L.; Pak, S.C. Serum Magnesium Levels in Patients with Obstructive Sleep Apnoea: A Systematic Review and Meta-Analysis. Biomedicines 2022, 10, 2273. https://doi.org/10.3390/biomedicines10092273
Al Wadee Z, Ooi SL, Pak SC. Serum Magnesium Levels in Patients with Obstructive Sleep Apnoea: A Systematic Review and Meta-Analysis. Biomedicines. 2022; 10(9):2273. https://doi.org/10.3390/biomedicines10092273
Chicago/Turabian StyleAl Wadee, Zahraa, Soo Liang Ooi, and Sok Cheon Pak. 2022. "Serum Magnesium Levels in Patients with Obstructive Sleep Apnoea: A Systematic Review and Meta-Analysis" Biomedicines 10, no. 9: 2273. https://doi.org/10.3390/biomedicines10092273
APA StyleAl Wadee, Z., Ooi, S. L., & Pak, S. C. (2022). Serum Magnesium Levels in Patients with Obstructive Sleep Apnoea: A Systematic Review and Meta-Analysis. Biomedicines, 10(9), 2273. https://doi.org/10.3390/biomedicines10092273








