The Mito-Hormetic Mechanisms of Ozone in the Clearance of SARS-CoV2 and in the COVID-19 Therapy
Abstract
1. Introduction
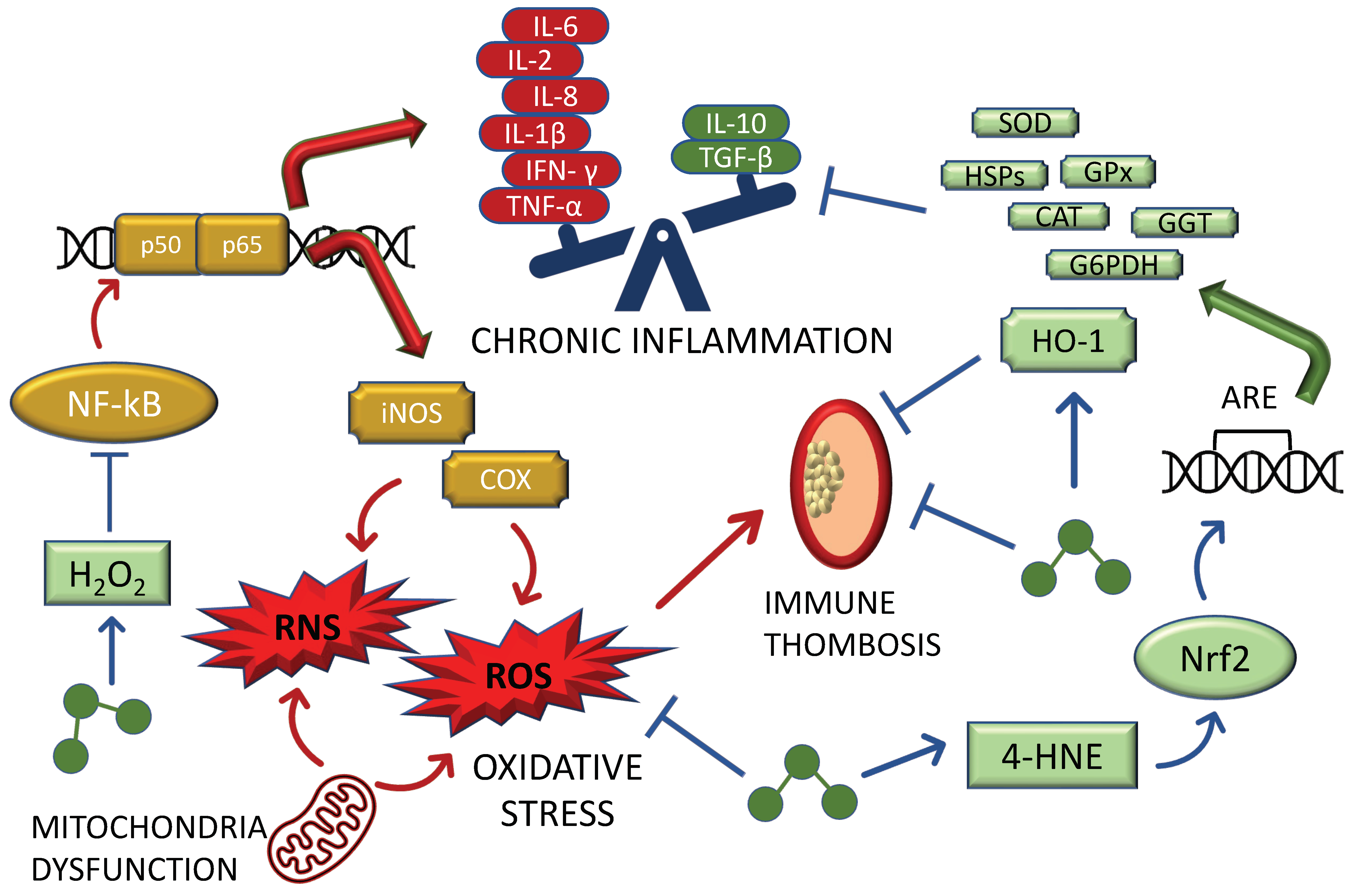
2. The Role of Ozone in the Mito-Hormetic Machinery
2.1. Insights on the Anti-Oxidant/Pro-Inflammatory Relationship Caused by Ozone
2.2. Insights into the Mito-Hormetic Ability of Ozone and Its Major Biochemical Mediators in Counteracting SARS-CoV2 Infection and COVID-19 Development: Endothelia and NO
2.3. Insights into the Mito-Hormetic Ability of Ozone and Its Major Biochemical Mediators in Counteracting SARS-CoV2 Infection and COVID-19 Development: The HO-1 and the Hypoxic Pathway
2.4. Ozone in COVID-19: The Key Mechanism of Ozone-Induced Hormetic Treatment against SARS-CoV2: Inflammasome, AhRs, and Nitric Oxide
2.5. Ozone in Post-COVID-19 (PASC)
3. Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Ivanov, S.V.; Panchenko, V.Y. Infrared and microwave spectroscopy of ozone: Historical aspects. Phys. -Uspekhi 1994, 37, 677–695. [Google Scholar] [CrossRef]
- Bocci, V. Is it true that ozone is always toxic? The end of a dogma. Toxicol. Appl. Pharmacol. 2006, 216, 493–504. [Google Scholar] [CrossRef] [PubMed]
- Bocci, V.; Borrelli, E.; Travagli, V.; Zanardi, I. The ozone paradox: Ozone is a strong oxidant as well as a medical drug. Med. Res. Rev. 2009, 29, 646–682. [Google Scholar] [CrossRef]
- Scassellati, C.; Galoforo, A.C.; Bonvicini, C.; Esposito, C.; Ricevuti, G. Ozone: A natural bioactive molecule with antioxidant property as potential new strategy in aging and in neurodegenerative disorders. Ageing Res. Rev. 2020, 63, 101138. [Google Scholar] [CrossRef] [PubMed]
- Galiè, M.; Covi, V.; Tabaracci, G.; Malatesta, M. The Role of Nrf2 in the Antioxidant Cellular Response to Medical Ozone Exposure. Int. J. Mol. Sci. 2019, 20, 4009. [Google Scholar] [CrossRef]
- Viebahn-Haensler, R.; Fernández, O.L. Ozone in Medicine. The Low-Dose Ozone Concept and Its Basic Biochemical Mechanisms of Action in Chronic Inflammatory Diseases. Int. J. Mol. Sci. 2021, 22, 7890. [Google Scholar] [CrossRef] [PubMed]
- Ezraty, B.; Gennaris, A.; Barras, F.; Collet, J.-F. Oxidative stress, protein damage and repair in bacteria. Nat. Rev. Microbiol. 2017, 15, 385–396. [Google Scholar] [CrossRef] [PubMed]
- Juan, C.; de la Lastra, J.P.; Plou, F.; Pérez-Lebeña, E. The Chemistry of Reactive Oxygen Species (ROS) Revisited: Outlining Their Role in Biological Macromolecules (DNA, Lipids and Proteins) and Induced Pathologies. Int. J. Mol. Sci. 2021, 22, 4642. [Google Scholar] [CrossRef]
- D’Autréaux, B.; Toledano, M.B. ROS as signalling molecules: Mechanisms that generate specificity in ROS homeostasis. Nat. Rev. Mol. Cell Biol. 2007, 8, 813–824. [Google Scholar] [CrossRef]
- Mittler, R.; Vanderauwera, S.; Suzuki, N.; Miller, G.; Tognetti, V.B.; Vandepoele, K.; Gollery, M.; Shulaev, V.; Van Breusegem, F. ROS signaling: The new wave? Trends Plant Sci. 2011, 16, 300–309. [Google Scholar] [CrossRef]
- Zhang, J.; Wang, X.; Vikash, V.; Ye, Q.; Wu, D.; Liu, Y.; Dong, W. ROS and ROS-mediated cellular signaling. Oxid. Med. Cell. Longev. 2016, 2016, 4350965. [Google Scholar] [CrossRef] [PubMed]
- Phull, A.-R.; Nasir, B.; Haq, I.U.; Kim, S.J. Oxidative stress, consequences and ROS mediated cellular signaling in rheumatoid arthritis. Chem. Interact. 2018, 281, 121–136. [Google Scholar] [CrossRef] [PubMed]
- Rigotti, G.; Chirumbolo, S. Biological Morphogenetic Surgery: A Minimally Invasive Procedure to Address Different Biological Mechanisms. Aesthetic Surg. J. 2018, 39, 745–755. [Google Scholar] [CrossRef] [PubMed]
- Chirumbolo, S.; Bjørklund, G. PERM Hypothesis: The Fundamental Machinery Able to Elucidate the Role of Xenobiotics and Hormesis in Cell Survival and Homeostasis. Int. J. Mol. Sci. 2017, 18, 165. [Google Scholar] [CrossRef]
- Degechisa, S.T.; Dabi, Y.T.; Gizaw, S.T. The mitochondrial associated endoplasmic reticulum membranes: A platform for the pathogenesis of inflammation-mediated metabolic diseases. Immun. Inflamm. Dis. 2022, 10, e647. [Google Scholar] [CrossRef]
- Canella, R.; Martini, M.; Borriello, R.; Cavicchio, C.; Muresan, X.M.; Benedusi, M.; Cervellati, F.; Valacchi, G. Modulation of Chloride Currents in Human Lung Epithelial Cells Exposed to Exogenous Oxidative Stress. J. Cell. Physiol. 2017, 232, 1817–1825. [Google Scholar] [CrossRef]
- Canella, R.; Benedusi, M.; Martini, M.; Cervellati, F.; Cavicchio, C.; Valacchi, G. Role of Nrf2 in preventing oxidative stress induced chloride current alteration in human lung cells. J. Cell. Physiol. 2017, 233, 6018–6027. [Google Scholar] [CrossRef]
- Swanton, T.; Beswick, J.A.; Hammadi, H.; Morris, L.; Williams, D.; de Cesco, S.; El-Sharkawy, L.; Yu, S.; Green, J.; Davis, J.B.; et al. Selective inhibition of the K+ efflux sensitive NLRP3 pathway by Cl− channel modulation. Chem. Sci. 2020, 11, 11720–11728. [Google Scholar] [CrossRef]
- Kaivola, J.; Nyman, T.A.; Matikainen, S. Inflammasomes and SARS-CoV-2 Infection. Viruses 2021, 13, 2513. [Google Scholar] [CrossRef]
- Shang, C.; Liu, Z.; Zhu, Y.; Lu, J.; Ge, C.; Zhang, C.; Li, N.; Jin, N.; Li, Y.; Tian, M.; et al. SARS-CoV-2 Causes Mitochondrial Dysfunction and Mitophagy Impairment. Front. Microbiol. 2022, 12, 780768. [Google Scholar] [CrossRef]
- Hennig, P.; Garstkiewicz, M.; Grossi, S.; Di Filippo, M.; French, L.E.; Beer, H.-D. The Crosstalk between Nrf2 and Inflammasomes. Int. J. Mol. Sci. 2018, 19, 562. [Google Scholar] [CrossRef] [PubMed]
- Chirumbolo, S.; Bjørklund, G. The bimodal SARS-CoV-2 outbreak in Italy as an effect of environmental and allergic causes. J. Allergy Clin. Immunol. 2020, 146, 331–332. [Google Scholar] [CrossRef] [PubMed]
- Kettle, A.J.; Winterbourn, C.C. Do neutrophils produce ozone? An appraisal of current evidence. BioFactors 2005, 24, 41–45. [Google Scholar] [CrossRef] [PubMed]
- Babior, B.M.; Takeuchi, C.; Ruedi, J.; Gutierrez, A.; Wentworth, P. Investigating antibody-catalyzed ozone generation by human neutrophils. Proc. Natl. Acad. Sci. USA 2003, 100, 3031–3034. [Google Scholar] [CrossRef]
- Bárcena, C.; Mayoral, P.; Quirós, P.M. Mitohormesis, an Antiaging Paradigm. Int. Rev. Cell Mol. Biol. 2018, 340, 35–77. [Google Scholar] [CrossRef]
- Ristow, M.; Schmeisser, K. Mitohormesis: Promoting Health and Lifespan by Increased Levels of Reactive Oxygen Species (ROS). Dose-Response 2014, 12, 288–341. [Google Scholar] [CrossRef]
- Olagnier, D.; Farahani, E.; Thyrsted, J.; Blay-Cadanet, J.; Herengt, A.; Idorn, M.; Hait, A.; Hernaez, B.; Knudsen, A.; Iversen, M.B.; et al. Author Correction: SARS-CoV2-mediated suppression of NRF2-signaling reveals potent antiviral and anti-inflammatory activity of 4-octyl-itaconate and dimethyl fumarate. Nat. Commun. 2020, 11, 5419. [Google Scholar] [CrossRef]
- Ma, Q. Role of Nrf2 in Oxidative Stress and Toxicity. Annu. Rev. Pharmacol. Toxicol. 2013, 53, 401–426. [Google Scholar] [CrossRef]
- Itoh, K.; Chiba, T.; Takahashi, S.; Ishii, T.; Igarashi, K.; Katoh, Y.; Oyake, T.; Hayashi, N.; Satoh, K.; Hatayama, I.; et al. An Nrf2/Small Maf Heterodimer Mediates the Induction of Phase II Detoxifying Enzyme Genes through Antioxidant Response Elements. Biochem. Biophys. Res. Commun. 1997, 236, 313–322. [Google Scholar] [CrossRef]
- Venugopal, R.; Jaiswal, A.K. Nrf1 and Nrf2 positively and c-Fos and Fra1 negatively regulate the human antioxidant response element-mediated expression of NAD(P)H:quinone oxidoreductase 1 gene. Proc. Natl. Acad. Sci. USA 1996, 93, 14960–14965. [Google Scholar] [CrossRef]
- Larini, A.; Bianchi, L.; Bocci, V. Effect of 4-hydroxynonenal on antioxidant capacity and apoptosis induction in Jurkat T cells. Free Radic. Res. 2004, 38, 509–516. [Google Scholar] [CrossRef] [PubMed]
- Singhal, S.S.; Singh, S.P.; Singhal, P.; Horne, D.; Singhal, J.; Awasthi, S. Antioxidant role of glutathione S-transferases: 4-Hydroxynonenal, a key molecule in stress-mediated signaling. Toxicol. Appl. Pharmacol. 2015, 289, 361–370. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, T.; Sherratt, P.J.; Pickett, C.B. Regulatory Mechanisms Controlling Gene Expression Mediated by the Antioxidant Response Element. Annu. Rev. Pharmacol. Toxicol. 2003, 43, 233–260. [Google Scholar] [CrossRef] [PubMed]
- Ma, Q. Xenobiotic-Activated Receptors: From Transcription to Drug Metabolism to Disease. Chem. Res. Toxicol. 2008, 21, 1651–1671. [Google Scholar] [CrossRef]
- Chirumbolo, S.; Valdenassi, L.; Simonetti, V.; Bertossi, D.; Ricevuti, G.; Franzini, M.; Pandolfi, S. Insights on the mechanisms of action of ozone in the medical therapy against COVID-19. Int. Immunopharmacol. 2021, 96, 107777. [Google Scholar] [CrossRef]
- Kovac, S.; Angelova, P.R.; Holmström, K.M.; Zhang, Y.; Dinkova-Kostova, A.T.; Abramov, A.Y. Nrf2 regulates ROS production by mitochondria and NADPH oxidase. Biochim. Biophys. Acta 2015, 1850, 794–801. [Google Scholar] [CrossRef]
- Kasai, S.; Shimizu, S.; Tatara, Y.; Mimura, J.; Itoh, K. Regulation of Nrf2 by Mitochondrial Reactive Oxygen Species in Physiology and Pathology. Biomolecules 2020, 10, 320. [Google Scholar] [CrossRef]
- Ryan, D.G.; Knatko, E.V.; Casey, A.M.; Hukelmann, J.L.; Naidu, S.D.; Brenes, A.J.; Ekkunagul, T.; Baker, C.; Higgins, M.; Tronci, L.; et al. Nrf2 activation reprograms macrophage intermediary metabolism and suppresses the type I interferon response. iScience 2022, 25, 103827. [Google Scholar] [CrossRef]
- Cattel, F.; Giordano, S.; Bertiond, C.; Lupia, T.; Corcione, S.; Scaldaferri, M.; Angelone, L.; De Rosa, F.G. Ozone therapy in COVID-19: A narrative review. Virus Res. 2020, 291, 198207. [Google Scholar] [CrossRef]
- Izadi, M.; Cegolon, L.; Javanbakht, M.; Sarafzadeh, A.; Abolghasemi, H.; Alishiri, G.; Zhao, S.; Einollahi, B.; Kashaki, M.; Jonaidi-Jafari, N.; et al. Ozone therapy for the treatment of COVID-19 pneumonia: A scoping review. Int. Immunopharmacol. 2020, 92, 107307. [Google Scholar] [CrossRef]
- Villani, E.; Ranaldi, G.; Franza, L. Rationale for ozone-therapy as an adjuvant therapy in COVID-19: A narrative review. Med. Gas. Res. 2020, 10, 134–138. [Google Scholar] [CrossRef]
- Shah, M.; Captain, J.; Vaidya, V.; Kulkarni, A.; Valsangkar, K.; Nair, P.M.; Ganu, G. Safety and efficacy of ozone therapy in mild to moderate COVID-19 patients: A phase 1/11 randomized control trial (SEOT study). Int. Immunopharmacol. 2020, 91, 107301. [Google Scholar] [CrossRef] [PubMed]
- Hernández, A.; Viñals, M.; Pablos, A.; Vilás, F.; Papadakos, P.J.; Wijeysundera, D.N.; Bergese, S.D.; Vives, M. Ozone therapy for patients with COVID-19 pneumonia: Preliminary report of a prospective case-control study. Int. Immunopharmacol. 2020, 90, 107261. [Google Scholar] [CrossRef] [PubMed]
- Çolak, Ş.; Yavuz, B.G.; Yavuz, M.; Özçelik, B.; Öner, M.; Özgültekin, A.; Şenbayrak, S. Effectiveness of ozone therapy in addition to conventional treatment on mortality in patients with COVID-19. Int. J. Clin. Pract. 2021, 75, e14321. [Google Scholar] [CrossRef] [PubMed]
- Franzini, M.; Valdenassi, L.; Ricevuti, G.; Chirumbolo, S.; Depfenhart, M.; Bertossi, D.; Tirelli, U. Oxygen-ozone (O2-O3) immunoceutical therapy for patients with COVID-19. Preliminary evidence reported. Int. Immunopharmacol. 2020, 88, 106879. [Google Scholar] [CrossRef] [PubMed]
- Cenci, A.; Macchia, I.; La Sorsa, V.; Sbarigia, C.; Di Donna, V.; Pietraforte, D. Mechanisms of Action of Ozone Therapy in Emerging Viral Diseases: Immunomodulatory Effects and Therapeutic Advantages with Reference to SARS-CoV-2. Front. Microbiol. 2022, 13, 871645. [Google Scholar] [CrossRef]
- Hernández, A.; Viñals, M.; Isidoro, T.; Vilás, F. Potential Role of Oxygen–Ozone Therapy in Treatment of COVID-19 Pneumonia. Am. J. Case Rep. 2020, 21, e925849-1–e925849-6. [Google Scholar] [CrossRef]
- Zheng, Z.; Dong, M.; Hu, K. A preliminary evaluation on the efficacy of ozone therapy in the treatment of COVID-19. J. Med. Virol. 2020, 92, 2348–2350. [Google Scholar] [CrossRef]
- Araimo, F.; Imperiale, C.; Tordiglione, P.; Ceccarelli, G.; Borrazzo, C.; Alessandri, F.; Santinelli, L.; Innocenti, G.P.; Pinacchio, C.; Mauro, V.; et al. Ozone as adjuvant support in the treatment of COVID-19: A preliminary report of probiozovid trial. J. Med. Virol. 2020, 93, 2210–2220. [Google Scholar] [CrossRef]
- Fernández-Cuadros, M.E.; Albaladejo-Florín, M.J.; Álava-Rabasa, S.; Gallego-Galiana, J.; Pérez-Cruz, G.F.; Usandizaga-Elio, I.; Pacios, E.; Torres-García, D.E.; Peña-Lora, D.; Casique-Bocanegra, L.; et al. Compassionate Use of Rectal Ozone (O3) in Severe COVID-19 Pneumonia: A Case-Control Study. SN Compr. Clin. Med. 2021, 3, 1185–1199. [Google Scholar] [CrossRef]
- Hendawy, H.A.; Mosallam, W.; Abuelnaga, M.E.; Sabry, A.M. Old Treatment for a New Disease: Can Rectal Ozone Insufflation Be Used for COVID-19 Management? A Case Report. SN Compr. Clin. Med. 2021, 3, 1424–1427. [Google Scholar] [CrossRef] [PubMed]
- Sharma, A.; Shah, M.; Sane, H.; Gokulchandran, N.; Paranjape, A.; Khubchandani, P.; Captain, J.; Shirke, S.; Kul-karni, P. Intravenous ozonized saline therapy as prophylaxis for healthcare workers (HCWs) in a dedicated COVID-19 hospital in India –A retrospective study. Eur. Rev. Med. Pharm. Sci. 2021, 25, 3632–3639. [Google Scholar] [CrossRef]
- Sozio, E.; De Monte, A.; Sermann, G.; Bassi, F.; Sacchet, D.; Sbrana, F.; Ripoli, A.; Curcio, F.; Fabris, M.; Marengo, S.; et al. CORonavirus-19 mild to moderate pneumonia Management with blood Ozonization in patients with Respiratory failure (CORMOR) multicentric prospective randomized clinical trial. Int. Immunopharmacol. 2021, 98, 107874. [Google Scholar] [CrossRef] [PubMed]
- Sharma, A.; Shah, M.; Lakshmi, S.; Sane, H.; Captain, J.; Gokulchandran, N.; Khubchandani, P.; Pradeep, M.; Gote, P.; Tuppekar, B.; et al. A pilot study for treatment of COVID-19 patients in moderate stage using intravenous administration of ozonized saline as an adjuvant treatment-registered clinical trial. Int. Immunopharmacol. 2021, 96, 107743. [Google Scholar] [CrossRef]
- Tascini, C.; Sermann, G.; Pagotto, A.; Sozio, E.; De Carlo, C.; Giacinta, A.; Sbrana, F.; Ripoli, A.; Castaldo, N.; Merelli, M.; et al. Blood ozonization in patients with mild to moderate COVID-19 pneumonia: A single centre experience. Intern. Emerg. Med. 2020, 16, 669–675. [Google Scholar] [CrossRef]
- Dengiz, E.; Özcan, Ç.; Güven, Y.İ.; Uçar, S.; Ener, B.K.; Sözen, S.; Yağcı, B.; Güzel, İ.A.; Yiğit, B.; Andaç, A.; et al. Ozone Gas Applied through Nebulization as Adjuvant Treatment for Lung Respiratory Diseases Due to COVID-19 Infections: A Prospective Randomized Trial. Med. Gas. Res. 2022, 12, 55–59. [Google Scholar] [CrossRef]
- Tirelli, U.; Franzini, M.; Valdenassi, L.; Pisconti, S.; Taibi, R.; Torrisi, C.; Pandolfi, S.; Chirumbolo, S. Fatigue in Post-Acute Sequelae of SARS-CoV2 (PASC) Treated with Oxygen-Ozone Autohemotherapy-Preliminary Results on 100 Patients. Eur. Rev. Med. Pharm. Sci. 2021, 25, 5871–5875. [Google Scholar] [CrossRef]
- Liu, Q.; Zhang, D.; Hu, D.; Zhou, X.; Zhou, Y. The role of mitochondria in NLRP3 inflammasome activation. Mol. Immunol. 2018, 103, 115–124. [Google Scholar] [CrossRef]
- Patergnani, S.; Bouhamida, E.; Leo, S.; Pinton, P.; Rimessi, A. Mitochondrial Oxidative Stress and “Mito-Inflammation”: Actors in the Diseases. Biomedicines 2021, 9, 216. [Google Scholar] [CrossRef]
- Chen, Y.; Zhou, Z.; Min, W. Mitochondria, Oxidative Stress and Innate Immunity. Front. Physiol. 2018, 9, 1487. [Google Scholar] [CrossRef]
- Bonaventura, A.; Vecchié, A.; Dagna, L.; Martinod, K.; Dixon, D.L.; Van Tassell, B.W.; Dentali, F.; Montecucco, F.; Massberg, S.; Levi, M.; et al. Endothelial dysfunction and immunothrombosis as key pathogenic mechanisms in COVID-19. Nat. Rev. Immunol. 2021, 21, 319–329. [Google Scholar] [CrossRef]
- Infantes, E.C.; Bautista, J.T.; Beltrán-Povea, A.; Cahuana, G.M.; Soria, B.; Nabil, H.; Bedoya, F.; Tejedo, J.R. Regulation of mitochondrial function and endoplasmic reticulum stress by nitric oxide in pluripotent stem cells. World J. Stem Cells 2017, 9, 26–36. [Google Scholar] [CrossRef] [PubMed]
- Nisoli, E.; Falcone, S.; Tonello, C.; Cozzi, V.; Palomba, L.; Fiorani, M.; Pisconti, A.; Brunelli, S.; Cardile, A.; Francolini, M.; et al. Mitochondrial biogenesis by NO yields functionally active mitochondria in mammals. Proc. Natl. Acad. Sci. USA 2004, 101, 16507–16512. [Google Scholar] [CrossRef] [PubMed]
- Valerio, A.; Nisoli, E. Nitric oxide, interorganelle communication, and energy flow: A novel route to slow aging. Front. Cell Dev. Biol. 2015, 3, 6. [Google Scholar] [CrossRef] [PubMed]
- Åkerström, S.; Mousavi-Jazi, M.; Klingström, J.; Leijon, M.; Lundkvist, A.; Mirazimi, A. Nitric Oxide Inhibits the Replication Cycle of Severe Acute Respiratory Syndrome Coronavirus. J. Virol. 2005, 79, 1966–1969. [Google Scholar] [CrossRef]
- Martínez, M.C.; Morell, F.B.; Raya, A.; Romá, J.; Aldasoro, M.; Vila, J.; Lluch, S.; Romero, F.J. 4-Hydroxynonenal, a Lipid Peroxidation Product, Induces Relaxation of Human Cerebral Arteries. J. Cereb. Blood Flow Metab. 1994, 14, 693–696. [Google Scholar] [CrossRef]
- Romero, F.J.; Romero, M.J.; Morell, F.B.; Martínez, M.; Medina, P.; Lluch, S. 4-Hydroxynonenal-Induced Relaxation of Human Mesenteric Arteries. Free Radic. Biol. Med. 1997, 23, 521–523. [Google Scholar] [CrossRef]
- Maulucci, G.; Daniel, B.; Cohen, O.; Avrahami, Y.; Sasson, S. Hormetic and regulatory effects of lipid peroxidation mediators in pancreatic beta cells. Mol. Asp. Med. 2016, 49, 49–77. [Google Scholar] [CrossRef]
- Chapple, S.J.; Cheng, X.; Mann, G.E. Effects of 4-hydroxynonenal on vascular endothelial and smooth muscle cell redox signaling and function in health and disease. Redox Biol. 2013, 1, 319–331. [Google Scholar] [CrossRef]
- Harry, R.S.; Hiatt, L.A.; Kimmel, D.W.; Carney, C.K.; Halfpenny, K.C.; Cliffel, D.E.; Wright, D.W. Metabolic Impact of 4-Hydroxynonenal on Macrophage-Like RAW 264.7 Function and Activation. Chem. Res. Toxicol. 2012, 25, 1643–1651. [Google Scholar] [CrossRef][Green Version]
- Bocci, V.A.; Zanardi, I.; Travagli, V. Ozone acting on human blood yields a hormetic dose-response relationship. J. Transl. Med. 2011, 9, 66. [Google Scholar] [CrossRef] [PubMed]
- Shinriki, N.; Suzuki, T.; Takama, K.; Fukunaga, K.; Ohgiya, S.; Kubota, K.; Miura, T. Susceptibilities of Plasma Antioxi-dants and Erythrocyte Constituents to Low Levels of Ozone. Haematologia (Budap) 1998, 29, 229–239. [Google Scholar]
- Gatbonton-Schwager, T.N.; Sadhukhan, S.; Zhang, G.-F.; Letterio, J.J.; Tochtrop, G.P. Identification of a negative feedback loop in biological oxidant formation fegulated by 4-hydroxy-2-(E)-nonenal. Redox Biol. 2014, 2, 755–763. [Google Scholar] [CrossRef] [PubMed]
- Bocci, V.; Aldinucci, C.; Mosci, F.; Carraro, F.; Valacchi, G. Ozonation of Human Blood Induces a Remarkable Upregulation of Heme Oxygenase-1 and Heat Stress Protein-70. Mediat. Inflamm. 2007, 2007, 26785. [Google Scholar] [CrossRef] [PubMed]
- Hull, T.D.; Boddu, R.; Guo, L.; Tisher, C.C.; Traylor, A.M.; Patel, B.; Joseph, R.; Prabhu, S.D.; Suliman, H.B.; Piantadosi, C.A.; et al. Heme oxygenase-1 regulates mitochondrial quality control in the heart. JCI Insight 2016, 1, e85817. [Google Scholar] [CrossRef]
- Korski, K.I.; Kubli, D.A.; Wang, B.J.; Khalafalla, F.G.; Monsanto, M.M.; Firouzi, F.; Echeagaray, O.H.; Kim, T.; Adamson, R.M.; Dembitsky, W.P.; et al. Hypoxia Prevents Mitochondrial Dysfunction and Senescence in Human c-Kit+ Cardiac Progenitor Cells. Stem. Cells 2019, 37, 555–567. [Google Scholar] [CrossRef]
- Grieb, P.; Swiatkiewicz, M.; Prus, K.; Rejdak, K. Hypoxia may be a determinative factor in COVID-19 progression. Curr. Res. Pharmacol. Drug Discov. 2021, 2, 100030. [Google Scholar] [CrossRef]
- Bansal, S.; Biswas, G.; Avadhani, N.G. Mitochondria-targeted heme oxygenase-1 induces oxidative stress and mitochondrial dysfunction in macrophages, kidney fibroblasts and in chronic alcohol hepatotoxicity. Redox Biol. 2013, 2, 273–283. [Google Scholar] [CrossRef]
- Shen, H.-H.; Wang, C.-J.; Zhang, X.-Y.; Sheng, Y.-R.; Yang, S.-L.; Zheng, Z.-M.; Shi, J.-L.; Qiu, X.-M.; Xie, F.; Li, M.-Q. HIF1A-induced heme oxygenase 1 promotes the survival of decidual stromal cells against excess heme-mediated oxidative stress. Reproduction 2022, 163, 33–43. [Google Scholar] [CrossRef]
- Agani, F.H.; Puchowicz, M.; Chavez, J.C.; Pichiule, P.; LaManna, J. Role of nitric oxide in the regulation of HIF-1α expression during hypoxia. Am. J. Physiol. Physiol. 2002, 283, C178–C186. [Google Scholar] [CrossRef]
- Deng, L.; Meng, W.; Li, D.; Qiu, D.; Wang, S.; Liu, H. The Effect of Ozone on Hypoxia, Hemolysis and Morphological Change of Blood from Patients with Aortic Dissection (AD): A Preliminary in Vitro Experiment of Ozonated Autohemo-therapy for Treating AD. Am. J. Transl. Res. 2018, 10, 1829–1840. [Google Scholar]
- Tian, M.; Liu, W.; Li, X.; Zhao, P.; Shereen, M.A.; Zhu, C.; Huang, S.; Liu, S.; Yu, X.; Yue, M.; et al. HIF-1α promotes SARS-CoV-2 infection and aggravates inflammatory responses to COVID-19. Signal Transduct. Target. Ther. 2021, 6, 308. [Google Scholar] [CrossRef]
- Dunn, L.L.; Kong, S.M.; Tumanov, S.; Chen, W.; Cantley, J.; Ayer, A.; Maghzal, G.J.; Midwinter, R.G.; Chan, K.H.; Ng, M.K.; et al. Hmox1 (Heme Oxygenase-1) Protects Against Ischemia-Mediated Injury via Stabilization of HIF-1α (Hypoxia-Inducible Factor-1α). Arter. Thromb. Vasc. Biol. 2020, 41, 317–330. [Google Scholar] [CrossRef]
- Papandreou, I.; Cairns, R.A.; Fontana, L.; Lim, A.L.; Denko, N.C. HIF-1 mediates adaptation to hypoxia by actively downregulating mitochondrial oxygen consumption. Cell Metab. 2006, 3, 187–197. [Google Scholar] [CrossRef] [PubMed]
- Pagé, E.L.; Chan, D.A.; Giaccia, A.J.; Levine, M.; Richard, D.E. Hypoxia-inducible Factor-1α Stabilization in Nonhypoxic Conditions: Role of Oxidation and Intracellular Ascorbate Depletion. Mol. Biol. Cell 2008, 19, 86–94. [Google Scholar] [CrossRef] [PubMed]
- Jay, R.R.; Howard, R. A Plausible “Penny” Costing Effective Treatment for Corona Virus-Ozone Therapy. J. Infect. Dis. Epidemiol. 2020, 6, 113. [Google Scholar] [CrossRef]
- Fernández-Cuadros, M.E.; Albaladejo-Florín, M.J.; Peña-Lora, D.; Álava-Rabasa, S.; Pérez-Moro, O.S. Ozone (O3) and SARS-CoV-2: Physiological Bases and Their Therapeutic Possibilities According to COVID-19 Evolutionary Stage. SN Compr. Clin. Med. 2020, 2, 1094–1102. [Google Scholar] [CrossRef]
- Manjunath, S.N.; Sakar, M.; Katapadi, M.; Balakrishna, R.G. Recent case studies on the use of ozone to combat coronavirus: Problems and perspectives. Environ. Technol. Innov. 2020, 21, 101313. [Google Scholar] [CrossRef]
- Yilmaz, N.; Eren, E.; Oz, C. COVID-19 and Ozone. Cyprus. J. Med. Sci. 2021, 5, 365–372. [Google Scholar] [CrossRef]
- Yousefi, B.; Banihashemian, S.Z.; Feyzabadi, Z.K.; Hasanpour, S.; Kokhaei, P.; Abdolshahi, A.; Emadi, A.; Eslami, M. Potential Therapeutic Effect of Oxygen-Ozone in Controlling of COVID-19 Disease. Med. Gas. Res. 2022, 12, 33–40. [Google Scholar] [CrossRef]
- Gu, Y.Q. Suggestion for a Long-Term Effective Disinfection to Eliminate the COVID-19 Epidemic. BAOJ Microbiol. 2022, 6, 1004. [Google Scholar]
- Bocci, V.; Valacchi, G. Nrf2 activation as target to implement therapeutic treatments. Front. Chem. 2015, 3, 4. [Google Scholar] [CrossRef] [PubMed]
- Falero-Perez, J.; Song, Y.-S.; Sorenson, C.M.; Sheibani, N. CYP1B1: A key regulator of redox homeostasis. Trends Cell Mol. Biol. 2018, 13, 27–45. [Google Scholar] [PubMed]
- Wang, G.; Xiao, B.; Deng, J.; Gong, L.; Li, Y.; Li, J.; Zhong, Y. The Role of Cytochrome P450 Enzymes in COVID-19 Pathogenesis and Therapy. Front. Pharmacol. 2022, 13, 791922. [Google Scholar] [CrossRef] [PubMed]
- Afaq, F.; Abu Zaid, M.; Pelle, E.; Khan, N.; Syed, D.N.; Matsui, M.S.; Maes, D.; Mukhtar, H. Aryl Hydrocarbon Receptor Is an Ozone Sensor in Human Skin. J. Investig. Dermatol. 2009, 129, 2396–2403. [Google Scholar] [CrossRef]
- Jacob, A.; Hartz, A.M.; Potin, S.; Coumoul, X.; Yousif, S.; Scherrmann, J.-M.; Bauer, B.; Declèves, X. Aryl hydrocarbon receptor-dependent upregulation of Cyp1b1 by TCDD and diesel exhaust particles in rat brain microvessels. Fluids Barriers CNS 2011, 8, 23. [Google Scholar] [CrossRef]
- Chirumbolo, S. The Role of Quercetin, Flavonols and Flavones in Modulating Inflammatory Cell Function. Inflamm. Allergy-Drug Targets 2010, 9, 263–285. [Google Scholar] [CrossRef]
- Kvarantan, A.; Balta, V.; Zarkovic, N.; Horvat, T.; Vukovic, T.; Zarkovic, K.; Kalogjera, L. Association between aryl hydrocarbon receptor and 4-hydroxynonenal in oxidative stress-mediated chronic rhinosinusitis with nasal polyps. Eur. J. Inflamm. 2021, 19. [Google Scholar] [CrossRef]
- Larigot, L.; Benoit, L.; Koual, M.; Tomkiewicz, C.; Barouki, R.; Coumoul, X. Aryl Hydrocarbon Receptor and Its Diverse Ligands and Functions: An Exposome Receptor. Annu. Rev. Pharmacol. Toxicol. 2022, 62, 383–404. [Google Scholar] [CrossRef]
- Hwang, H.J.; Dornbos, P.; Steidemann, M.; Dunivin, T.K.; Rizzo, M.; LaPres, J.J. Mitochondrial-targeted aryl hydrocarbon receptor and the impact of 2,3,7,8-tetrachlorodibenzo-p-dioxin on cellular respiration and the mitochondrial proteome. Toxicol. Appl. Pharmacol. 2016, 304, 121–132. [Google Scholar] [CrossRef]
- Dwier, M.; Michalek, R.; Saloupis, P.; McDonnell, D.; Malek, G. Oxidized Lipids Activate Aryl Hydrocarbon Receptor (AhR) and Differentially Regulate Metabolic Pathways in Retinal Pigment Epithelial Cells (RPE). Invest. Ophtalmol. Vis. Sci. 2011, 52, 3328. [Google Scholar]
- Mao, K.; Chen, S.; Chen, M.; Ma, Y.; Wang, Y.; Huang, B.; He, Z.; Zeng, Y.; Hu, Y.; Sun, S.; et al. Nitric oxide suppresses NLRP3 inflammasome activation and protects against LPS-induced septic shock. Cell Res. 2013, 23, 201–212. [Google Scholar] [CrossRef] [PubMed]
- Laskin, D.L.; Sunil, V.; Guo, Y.; E Heck, D.; Laskin, J.D. Increased nitric oxide synthase in the lung after ozone inhalation is associated with activation of NF-kappa B. Environ. Health Perspect. 1998, 106, 1175–1178. [Google Scholar] [CrossRef] [PubMed]
- Hogg, N.; Kalyanaraman, B. Nitric oxide and lipid peroxidation. Biochim. Biophys. Acta 1999, 1411, 378–384. [Google Scholar] [CrossRef]
- Anderson, E.J.; A Katunga, L.; Willis, M.S. Mitochondria as a source and target of lipid peroxidation products in healthy and diseased heart. Clin. Exp. Pharmacol. Physiol. 2011, 39, 179–193. [Google Scholar] [CrossRef] [PubMed]
- Ceban, F.; Ling, S.; Lui, L.M.; Lee, Y.; Gill, H.; Teopiz, K.M.; Rodrigues, N.B.; Subramaniapillai, M.; Di Vincenzo, J.D.; Cao, B.; et al. Fatigue and cognitive impairment in Post-COVID-19 Syndrome: A systematic review and meta-analysis. Brain Behav. Immun. 2021, 101, 93–135. [Google Scholar] [CrossRef]
- Sudre, C.H.; Murray, B.; Varsavsky, T.; Graham, M.S.; Penfold, R.S.; Bowyer, R.C.; Pujol, J.C.; Klaser, K.; Antonelli, M.; Canas, L.S.; et al. Attributes and predictors of long COVID. Nat. Med. 2021, 27, 626–631. [Google Scholar] [CrossRef] [PubMed]
- Merad, M.; Blish, C.A.; Sallusto, F.; Iwasaki, A. The immunology and immunopathology of COVID-19. Science 2022, 375, 1122–1127. [Google Scholar] [CrossRef]
- Andrade, B.S.; Siqueira, S.; Soares, W.D.A.; Rangel, F.D.S.; Santos, N.; Freitas, A.D.S.; da Silveira, P.R.; Tiwari, S.; Alzahrani, K.; Góes-Neto, A.; et al. Long-COVID and Post-COVID Health Complications: An Up-to-Date Review on Clinical Conditions and Their Possible Molecular Mechanisms. Viruses 2021, 13, 700. [Google Scholar] [CrossRef]
- Evans, R.A.; Leavy, O.C.; Richardson, M.; Elneima, O.; McCauley, H.J.C.; Shikotra, A.; Singapuri, A.; Sereno, M.; Saunders, R.M.; Harris, V.C.; et al. Clinical characteristics with inflammation profiling of long COVID and association with 1-year recovery following hospitalisation in the UK: A prospective observational study. Lancet Respir. Med. 2022, 10, 761–775. [Google Scholar] [CrossRef]
- Weinstock, L.B.; Brook, J.B.; Walters, A.S.; Goris, A.; Afrin, L.B.; Molderings, G.J. Mast cell activation symptoms are prevalent in Long-COVID. Int. J. Infect. Dis. 2021, 112, 217–226. [Google Scholar] [CrossRef] [PubMed]
- Kappelmann, N.; Dantzer, R.; Khandaker, G.M. Interleukin-6 as potential mediator of long-term neuropsychiatric symptoms of COVID-19. Psychoneuroendocrinology 2021, 131, 105295. [Google Scholar] [CrossRef]
- Phetsouphanh, C.; Darley, D.R.; Wilson, D.B.; Howe, A.; Munier, C.M.L.; Patel, S.K.; Juno, J.A.; Burrell, L.M.; Kent, S.J.; Dore, G.J.; et al. Immunological dysfunction persists for 8 months following initial mild-to-moderate SARS-CoV-2 infection. Nat. Immunol. 2022, 23, 210–216. [Google Scholar] [CrossRef]
- Doykov, I.; Hällqvist, J.; Gilmour, K.C.; Grandjean, L.; Mills, K.; Heywood, W.E. ‘The long tail of Covid-19’-The detection of a prolonged inflammatory response after a SARS-CoV-2 infection in asymptomatic and mildly affected patients. F1000Research 2021, 9, 1349. [Google Scholar] [CrossRef] [PubMed]
- Chang, J.D.S.; Lu, H.-S.; Chang, Y.-F.; Wang, D. Ameliorative effect of ozone on cytokine production in mice injected with human rheumatoid arthritis synovial fibroblast cells. Rheumatol. Int. 2004, 26, 142–151. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Sánchez, G.; Schwartz, A.; Di Donna, V. Potential Cytoprotective Activity of Ozone Therapy in SARS-CoV-2/COVID-19. Antioxidants 2020, 9, 389. [Google Scholar] [CrossRef]
- Varesi, A.; Chirumbolo, S.; Ricevuti, G. Oxygen–ozone treatment and COVID-19: Antioxidants targeting endothelia lead the scenery. Intern. Emerg. Med. 2021, 17, 593–596. [Google Scholar] [CrossRef]
- Wei, A.; Feng, H.; Jia, X.-M.; Tang, H.; Liao, Y.-Y.; Li, B.-R. Ozone therapy ameliorates inflammation and endometrial injury in rats with pelvic inflammatory disease. Biomed. Pharmacother. 2018, 107, 1418–1425. [Google Scholar] [CrossRef]
- Tahmasebi, S.; Qasim, M.T.; Krivenkova, M.V.; Zekiy, A.O.; Thangavelu, L.; Aravindhan, S.; Izadi, M.; Jadidi-Niaragh, F.; Ghaebi, M.; Aslani, S.; et al. The effects of oxygen–ozone therapy on regulatory T-cell responses in multiple sclerosis patients. Cell Biol. Int. 2021, 45, 1498–1509. [Google Scholar] [CrossRef]
- Díaz-Resendiz, K.J.G.; Benitez-Trinidad, A.B.; Covantes-Rosales, C.E.; Toledo-Ibarra, G.A.; Ortiz-Lazareno, P.C.; Girón-Pérez, D.A.; Bueno-Durán, A.Y.; Pérez-Díaz, D.A.; Barcelos-García, R.G.; Girón-Pérez, M.I. Loss of mitochondrial membrane potential (Δ Ψ m ) in leucocytes as post-COVID-19 sequelae. J. Leukoc. Biol. 2022, 112, 23–29. [Google Scholar] [CrossRef]
- Galam, L.; Failla, A.; Soundararajan, R.; Lockey, R.F.; Kolliputi, N. 4-Hydroxynonenal regulates mitochondrial function in human small airway epithelial cells. Oncotarget 2015, 6, 41508–41521. [Google Scholar] [CrossRef] [PubMed]
- Rai, P.; Janardhan, K.S.; Meacham, J.; Madenspacher, J.H.; Lin, W.-C.; Karmaus, P.W.F.; Martinez, J.; Li, Q.-Z.; Yan, M.; Zeng, J.; et al. IRGM1 links mitochondrial quality control to autoimmunity. Nat. Immunol. 2021, 22, 312–321. [Google Scholar] [CrossRef] [PubMed]
- Paul, B.D.; Lemle, M.D.; Komaroff, A.L.; Snyder, S.H. Redox imbalance links COVID-19 and myalgic encephalomyelitis/chronic fatigue syndrome. Proc. Natl. Acad. Sci. USA 2021, 118, e2024358118. [Google Scholar] [CrossRef] [PubMed]
- Vollbracht, C.; Kraft, K. Oxidative Stress and Hyper-Inflammation as Major Drivers of Severe COVID-19 and Long COVID: Implications for the Benefit of High-Dose Intravenous Vitamin C. Front. Pharmacol. 2022, 13, 899198. [Google Scholar] [CrossRef]
- Brodin, P.; Casari, G.; Townsend, L.; O’Farrelly, C.; Tancevski, I.; Löffler-Ragg, J.; Mogensen, T.H.; Casanova, J.L.; Abel, L.; Aiuti, A.; et al. Studying severe long COVID to understand post-infectious disorders beyond COVID-19. Nat. Med. 2022, 28, 879–882. [Google Scholar] [CrossRef]
- Clavo, B.; Rodríguez-Esparragón, F.; Rodríguez-Abreu, D.; Martínez-Sánchez, G.; Llontop, P.; Aguiar-Bujanda, D.; Fernández-Pérez, L.; Santana-Rodríguez, N. Modulation of Oxidative Stress by Ozone Therapy in the Prevention and Treatment of Chemotherapy-Induced Toxicity: Review and Prospects. Antioxidants 2019, 8, 588. [Google Scholar] [CrossRef]
- Galiè, M.; Costanzo, M.; Nodari, A.; Boschi, F.; Calderan, L.; Mannucci, S.; Covi, V.; Tabaracci, G.; Malatesta, M. Mild ozonisation activates antioxidant cell response by the Keap1/Nrf2 dependent pathway. Free. Radic. Biol. Med. 2018, 124, 114–121. [Google Scholar] [CrossRef]
- Mohamed, M.S.; Johansson, A.; Jonsson, J.; Schiöth, H.B. Dissecting the Molecular Mechanisms Surrounding Post-COVID-19 Syndrome and Neurological Features. Int. J. Mol. Sci. 2022, 23, 4275. [Google Scholar] [CrossRef]
- Ercegovac, M.; Asanin, M.; Savic-Radojevic, A.; Ranin, J.; Matic, M.; Djukic, T.; Coric, V.; Jerotic, D.; Todorovic, N.; Milosevic, I.; et al. Antioxidant Genetic Profile Modifies Probability of Developing Neurological Sequelae in Long-COVID. Antioxidants 2022, 11, 954. [Google Scholar] [CrossRef]
- Jarrott, B.; Head, R.; Pringle, K.G.; Lumbers, E.R.; Martin, J.H. “LONG COVID”—A hypothesis for understanding the biological basis and pharmacological treatment strategy. Pharmacol. Res. Perspect. 2022, 10, e00911. [Google Scholar] [CrossRef]
- Crook, H.; Raza, S.; Nowell, J.; Young, M.; Edison, P. Long covid—mechanisms, risk factors, and management. BMJ 2021, 374, n1648. [Google Scholar] [CrossRef]
- Di Girolamo, F.G.; Fiotti, N.; Sisto, U.G.; Nunnari, A.; Colla, S.; Mearelli, F.; Vinci, P.; Schincariol, P.; Biolo, G. Skeletal Muscle in Hypoxia and Inflammation: Insights on the COVID-19 Pandemic. Front. Nutr. 2022, 9, 865402. [Google Scholar] [CrossRef]
- Karaarslan, F.; Güneri, F.D.; Kardeş, S. Long COVID: Rheumatologic/musculoskeletal symptoms in hospitalized COVID-19 survivors at 3 and 6 months. Clin. Rheumatol. 2021, 41, 289–296. [Google Scholar] [CrossRef] [PubMed]
- Khan, S.A.; Seyam, O.; Smith, N.L.; Reid, I.; Gandhi, J.; Jiang, W. Clinical utility of ozone therapy for musculoskeletal disorders. Med. Gas. Res. 2018, 8, 103–110. [Google Scholar] [CrossRef] [PubMed]
- De Sire, A.; Agostini, F.; Lippi, L.; Mangone, M.; Marchese, S.; Cisari, C.; Bernetti, A.; Invernizzi, M. Oxygen–Ozone Therapy in the Rehabilitation Field: State of the Art on Mechanisms of Action, Safety and Effectiveness in Patients with Musculoskeletal Disorders. Biomolecules 2021, 11, 356. [Google Scholar] [CrossRef] [PubMed]
- De Sire, A.; Marotta, N.; Ferrillo, M.; Agostini, F.; Sconza, C.; Lippi, L.; Respizzi, S.; Giudice, A.; Invernizzi, M.; Ammendolia, A. Oxygen-Ozone Therapy for Reducing Pro-Inflammatory Cytokines Serum Levels in Musculoskeletal and Temporomandibular Disorders: A Comprehensive Review. Int. J. Mol. Sci. 2022, 23, 2528. [Google Scholar] [CrossRef]
- Akkawi, I. Ozone therapy for musculoskeletal disorders Current concepts. Acta Biomed. 2020, 91, e2020191. [Google Scholar] [CrossRef]
- Tirelli, U.; Franzini, M.; Valdenassi, L.; Pandolfi, S.; Berretta, M.; Ricevuti, G.; Chirumbolo, S. Patients with Myalgic Encephalomyelitis/Chronic Fatigue Syndrome (ME/CFS) Greatly Improved Fatigue Symptoms When Treated with Oxygen-Ozone Autohemotherapy. J. Clin. Med. 2021, 11, 29. [Google Scholar] [CrossRef]
- Tirelli, U.; Cirrito, C.; Pavanello, M.; Piasentin, C.; Lleshi, A.; Taibi, R. Ozone therapy in 65 patients with fibromyalgia: An effective therapy. Eur. Rev. Med. Pharmacol. Sci. 2019, 23, 1786–1788. [Google Scholar] [CrossRef]
- Li, Y.; Feng, X.; Ren, H.; Huang, H.; Wang, Y.; Yu, S. Low-Dose Ozone Therapy Improves Sleep Quality in Patients with Insomnia and Coronary Heart Disease by Elevating Serum BDNF and GABA. Bull. Exp. Biol. Med. 2021, 170, 493–498. [Google Scholar] [CrossRef]
- Moghimi, N.; Di Napoli, M.; Biller, J.; Siegler, J.E.; Shekhar, R.; McCullough, L.D.; Harkins, M.S.; Hong, E.; Alaouieh, D.A.; Mansueto, G.; et al. The Neurological Manifestations of Post-Acute Sequelae of SARS-CoV-2 infection. Curr. Neurol. Neurosci. Rep. 2021, 21, 44. [Google Scholar] [CrossRef] [PubMed]
- Mondelli, V.; Pariante, C.M. What can neuroimmunology teach us about the symptoms of long-COVID? Oxf. Open Immunol. 2021, 2, iqab004. [Google Scholar] [CrossRef]
- Scassellati, C.; Ciani, M.; Galoforo, A.C.; Zanardini, R.; Bonvicini, C.; Geroldi, C. Molecular mechanisms in cognitive frailty: Potential therapeutic targets for oxygen-ozone treatment. Mech. Ageing Dev. 2020, 186, 111210. [Google Scholar] [CrossRef] [PubMed]
- Scassellati, C.; Galoforo, A.C.; Esposito, C.; Ciani, M.; Ricevuti, G.; Bonvicini, C. Promising Intervention Approaches to Potentially Resolve Neuroinflammation And Steroid Hormones Alterations in Alzheimer’s Disease and Its Neuropsychiatric Symptoms. Aging Dis. 2021, 12, 1337–1357. [Google Scholar] [CrossRef] [PubMed]
- Clavo, B.; Santana-Rodríguez, N.; Gutierrez, D.; Lopez, J.C.; Suarez, G.; Lopez, L.; Robaina, F.; Bocci, V. Long-Term Improvement in Refractory Headache Following Ozone Therapy. J. Altern. Complement. Med. 2013, 19, 453–458. [Google Scholar] [CrossRef]
- Haggiag, S.; Prosperini, L.; Stasolla, A.; Gerace, C.; Tortorella, C.; Gasperini, C. Ozone-induced encephalopathy: A novel iatrogenic entity. Eur. J. Neurol. 2021, 28, 2471–2478. [Google Scholar] [CrossRef]
- Rangel, K.; Cabral, F.O.; Lechuga, G.C.; Carvalho, J.P.R.S.; Villas-Bôas, M.H.S.; Midlej, V.; De-Simone, S.G. Potent Activity of a High Concentration of Chemical Ozone against Antibiotic-Resistant Bacteria. Molecules 2022, 27, 3998. [Google Scholar] [CrossRef]
- Roede, J.R.; Jones, D.P. Reactive species and mitochondrial dysfunction: Mechanistic significance of 4-hydroxynonenal. Environ. Mol. Mutagen. 2010, 51, 380–390. [Google Scholar] [CrossRef]
- Dodson, M.; Wani, W.Y.; Redmann, M.; Benavides, G.A.; Johnson, M.S.; Ouyang, X.; Cofield, S.S.; Mitra, K.; Darley-Usmar, V.; Zhang, J. Regulation of autophagy, mitochondrial dynamics, and cellular bioenergetics by 4-hydroxynonenal in primary neurons. Autophagy 2017, 13, 1828–1840. [Google Scholar] [CrossRef]

| Ref. | Study Type | Number of Participants | Clinical Condition | Treatment | Results | Statistically Significant |
|---|---|---|---|---|---|---|
| [42] | RCT | 60 patients (30 treated and 30 controls) | Mild to moderate COVID-19 | Rectal insufflation + minor autohemotherapy | ↓ CRP, LDH, ferritin ↑ SpO2 | No |
| [44] | PCS | 55 patients (37 treated and 18 controls) | Hospitalized | CT + major autohemotherapy (IV for seven consecutive days, 30 μg/mL) | ↓ ICU hospitalization ↓ Mortality risk | No |
| [47] | CR | 3 patients | Respiratory failure | Autohemotherapy (1–6 sessions) | ↓ Hypoxia ↓ LDH, CRP, D-dimer ↓ Mechanical ventilation ↓ FiO2 ↑ PaO2 | NA |
| [45] | CSS | 50 patients | Hospitalized in ICUs presenting ARDS | 4 cycles of autohemotherapy (SIOOT protocol), 30 μg/mL | ↓ CRP, LDH, ALT, IL-6, D-dimer ↑ SatO2% | Yes |
| [43] | PCS | 18 patients (9 treated and 9 controls) | Severe pneumonia | Autohemotherapy twice a day for 4 days (median), 40 μg/mL | ↑ Recovery rate ↓ Time to reduce CRP, D-dimer, ferritin, LDH | Yes |
| [48] | CR | 2 patients | Mild fever, dyspnea | Major autohemotherapy (daily for 7 days), 20 μg/mL | ↓ CRP, LDH, IL-6 ↑ Recovery rate | NA |
| [49] | RCT | 28 patients (14 treated and 14 controls) | Hospitalized with severe COVID-19 | CT + autohemotherapy twice a day for 7 days | ↓ Need for ventilation = Inflammatory markers = Lymphocyte subpopulations | No |
| [50] | CCS | 28 patients (14 treated and 14 controls) | Bilateral COVID-19 pneumonia | Intra-rectal ozone daily for 8 days, 35 μg/mL | ↓ D-dimer, fibrinogen, urea, CRP, LDH, IL-6, ferritin ↓ Radiological pneumonitis ↓ Leukocytes ↑ Lymphocytes ↓ O2 supply | Yes |
| [51] | CR | 2 patients | Presenting hypoxia and requiring ventilation | Rectal ozone insufflation (1–2 sessions) | ↑ SpO2 | NA |
| [52] | RCCS | 235 healthcare workers (64 treated and 171 controls) | Healthy | COVID-19 standard prophylaxis + IV ozonized saline daily for 4 days/month | ↓ COVID-19 incidence | Yes |
| [53] | RCT | 92 patients (48 treated and 44 controls) | Mild to moderate pneumonia | CT + autohemotherapy daily for 3 days, 40 μg/mL | ↓ Leukocytes ↑ CRP = Mortality rate = Need for ventilation = Hospital stay | No |
| [54] | PCT | 10 patients | Moderate COVID-19 | CT + IV ozonized saline (200 mL daily for 8 days) | ↓ CRP, D-dimer, IL-6 ↑ SpO2/FiO2 | Yes |
| [55] | CCS | 60 patients (30 treated and 30 controls) | Mild to moderate pneumonia | CT + autohemotherapy daily for 3 days, 40 μg/mL | ↓ SIMEU clinical phenotype | Yes |
| [56] | RCT | 30 patients (15 treated and 15 controls) | CT + inhalation of nebulized ozone, 3 sessions of 10 min (0.2 ppm/session) every 5 min daily for 5 days) | ↓ Hospitalization time ↓ CRP = D-dimer, urea, LDH lymphocytes, leukocytes, platelets | Yes | |
| [57] | Multicenter CSS | 100 patients | PASC | Autohemotherapy (SIOOT protocol) | ↓ Fatigue | Yes |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chirumbolo, S.; Varesi, A.; Franzini, M.; Valdenassi, L.; Pandolfi, S.; Tirelli, U.; Esposito, C.; Ricevuti, G. The Mito-Hormetic Mechanisms of Ozone in the Clearance of SARS-CoV2 and in the COVID-19 Therapy. Biomedicines 2022, 10, 2258. https://doi.org/10.3390/biomedicines10092258
Chirumbolo S, Varesi A, Franzini M, Valdenassi L, Pandolfi S, Tirelli U, Esposito C, Ricevuti G. The Mito-Hormetic Mechanisms of Ozone in the Clearance of SARS-CoV2 and in the COVID-19 Therapy. Biomedicines. 2022; 10(9):2258. https://doi.org/10.3390/biomedicines10092258
Chicago/Turabian StyleChirumbolo, Salvatore, Angelica Varesi, Marianno Franzini, Luigi Valdenassi, Sergio Pandolfi, Umberto Tirelli, Ciro Esposito, and Giovanni Ricevuti. 2022. "The Mito-Hormetic Mechanisms of Ozone in the Clearance of SARS-CoV2 and in the COVID-19 Therapy" Biomedicines 10, no. 9: 2258. https://doi.org/10.3390/biomedicines10092258
APA StyleChirumbolo, S., Varesi, A., Franzini, M., Valdenassi, L., Pandolfi, S., Tirelli, U., Esposito, C., & Ricevuti, G. (2022). The Mito-Hormetic Mechanisms of Ozone in the Clearance of SARS-CoV2 and in the COVID-19 Therapy. Biomedicines, 10(9), 2258. https://doi.org/10.3390/biomedicines10092258







