Endothelial Dysfunction, Erectile Deficit and Cardiovascular Disease: An Overview of the Pathogenetic Links
Abstract
1. Introduction
2. Physiology of Erection and Physiopathology of Vasculogenic ED
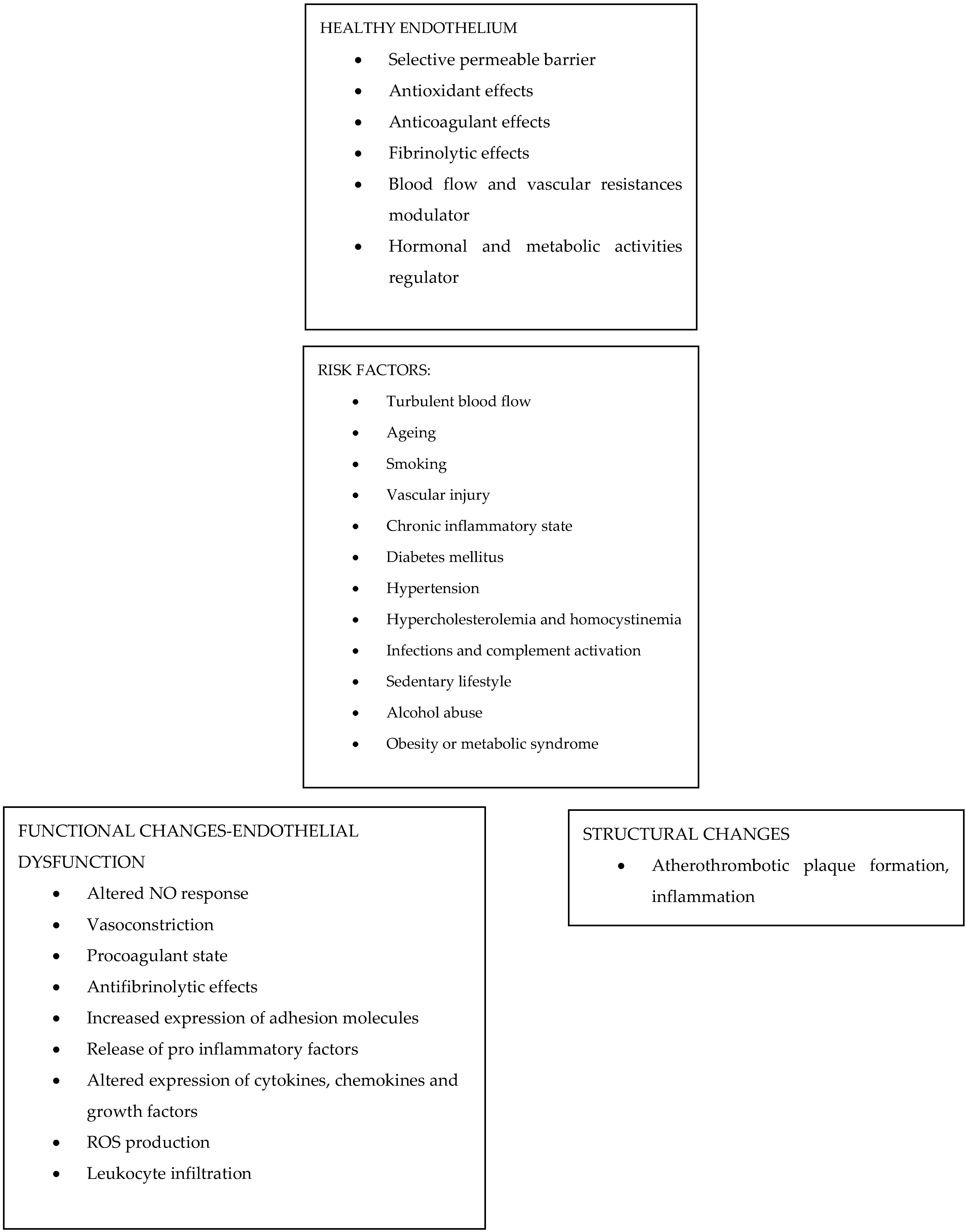
3. The Endothelium: Summary of Its Functions
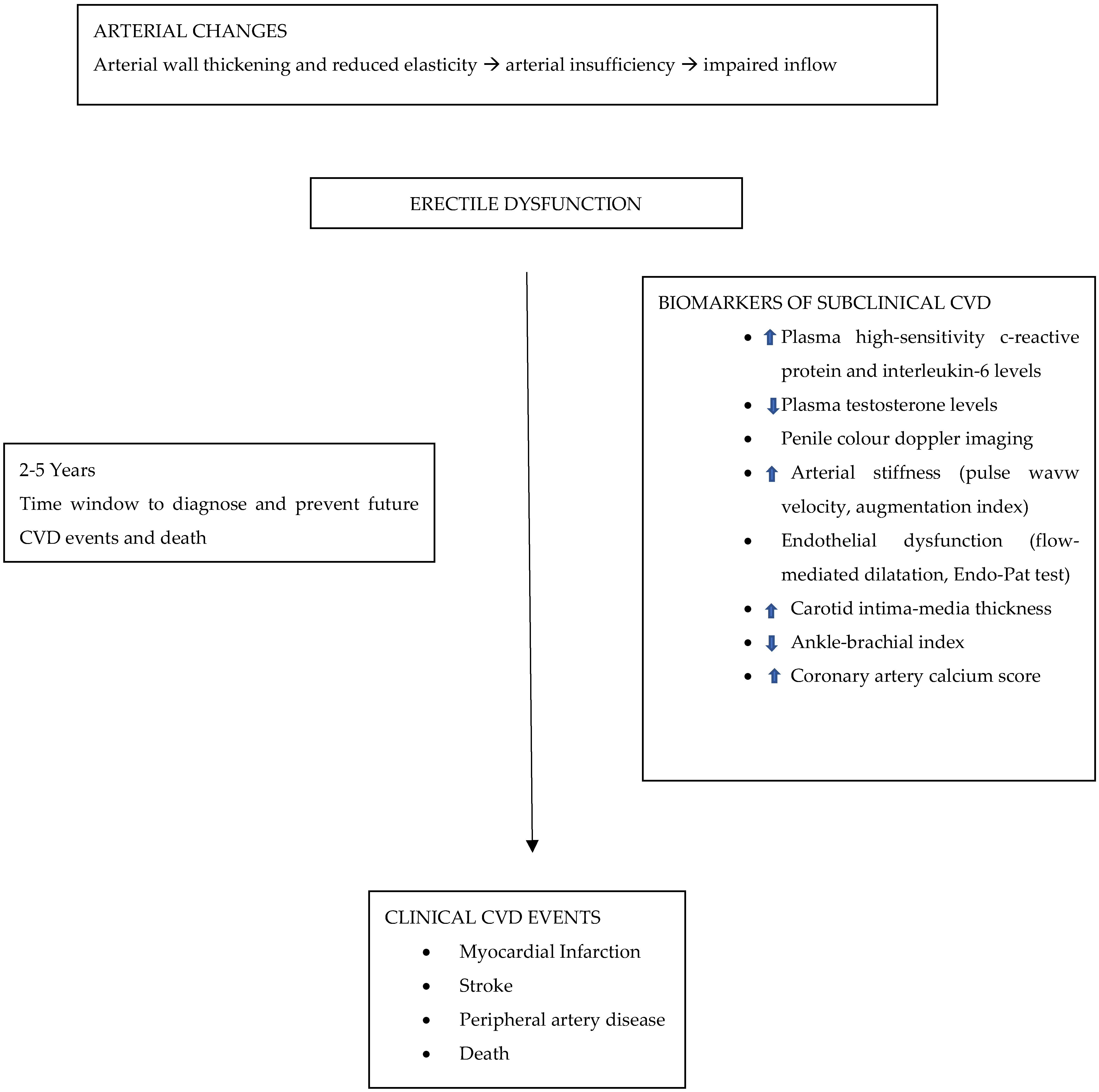
4. Endothelial Dysfunction and ED
5. ED and CVD
6. ED Therapeutic Approach
7. Cardiologic Assessment Recommendations for Patients Affected by ED
8. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Selvin, E.; Burnett, A.L.; Platz, E.A. Prevalence and risk factors for erectile dysfunction in the US. Am. J. Med. 2007, 120, 151–157. [Google Scholar] [CrossRef] [PubMed]
- Wylie, K. Erectile dysfunction. Adv. Psychosom. Med. 2008, 29, 33–49. [Google Scholar] [PubMed]
- Laumann, E.O.; Nicolosi, A.; Glasser, D.B.; Paik, A.; Gingell, C.; Moreira, E.; Wang, T. GSSAB Investigators’ Group. Sexual problems among women and men aged 40–80 y: Prevalence and correlates identified in the Global Study of Sexual Attitudes and Behaviors. Int. J. Impot. Res. 2005, 17, 39–57. [Google Scholar] [CrossRef] [PubMed]
- Nicolosi, A.; Laumann, E.O.; Glasser, D.B.; Moreira EDJr Paik, A.; Gingell, C. Global Study of Sexual Attitudes and Behaviors Investigators’ Group. Sexual behavior and sexual dysfunctions after age 40: The global study of sexual attitudes and behaviors. Urology 2004, 64, 991–997. [Google Scholar] [CrossRef] [PubMed]
- Corona, G.; Lee, D.M.; Forti, G.; O’Connor, D.B.; Maggi, M.; O’Neill, T.W.; Pendleton, N.; Bartfai, G.; Boonen, S.; Casanueva, F.F.; et al. EMAS Study Group. Age-related changes in general and sexual health in middle-aged and older men: Results from the European Male Ageing Study (EMAS). J. Sex. Med. 2010, 7, 1362–1380. [Google Scholar] [CrossRef] [PubMed]
- Feldman, H.A.; Goldstein, I.; Hatzichristou, D.G.; Krane, R.J.; McKinlay, J.B. Impotence and its medical and psychosocial correlates: Results of the Massachusetts Male Aging Study. J. Urol. 1994, 151, 54–61. [Google Scholar] [CrossRef]
- Rastrelli, G.; Maggi, M. Erectile dysfunction in fit and healthy young men: Psychological or pathological? Transl. Androl. Urol. 2017, 6, 79–90. [Google Scholar] [CrossRef]
- Donatucci, C.F.; Lue, T.F. Erectile dysfunction in men under 40: Etiology and treatment choice. Int. J. Impot. Res. 1993, 5, 97–103. [Google Scholar] [PubMed]
- Giuliano, F. Neurophysiology of Erection and Ejaculation. J. Sex. Med. 2011, 8, 310–315. [Google Scholar] [CrossRef]
- Dean, R.C.; Lue, T.F. Physiology of penile erection and pathophysiology of erectile dysfunction. Urol. Clin. N. Am. 2005, 32, 379–395. [Google Scholar] [CrossRef]
- Panchatsharam, P.K.; Durland, J.; Zito, P.M. Physiology, Erection. In StatPearls [Internet]; StatPearls Publishing: Treasure Island, FL, USA, 2022. [Google Scholar]
- Cartledge, J.; Minhas, S.; Eardley, I. The role of nitric oxide in penile erection. Expert Opin. Pharmacother. 2001, 2, 95–107. [Google Scholar] [CrossRef] [PubMed]
- Cines, D.B.; Pollak, E.S.; A Buck, C.; Loscalzo, J.; A Zimmerman, G.; McEver, R.P.; Pober, J.S.; Wick, T.; A Konkle, B.; Schwartz, B.S.; et al. Endothelial cells in physiology and in the pathophysiology of vascular disorders. Blood 1998, 91, 3527–3561. [Google Scholar] [PubMed]
- Esmon, C.T. The endothelial cell protein C receptor. Thromb. Haemost. 2000, 83, 639–643. [Google Scholar] [CrossRef] [PubMed]
- Ignarro, L.J.; Byrns, R.E.; Buga, G.M.; Wood, K.S. Endothelium-derived relaxing factor from pulmonary artery and vein possesses pharmacologic and chemical properties identical to those of nitric oxide radical. Circ. Res. 1987, 61, 866–879. [Google Scholar] [CrossRef] [PubMed]
- Alom-Ruiz, S.P.; Anilkumar, N.; Shah, A.M. Reactive oxygen species and endothelial activation. Antioxid. Redox Signal. 2008, 10, 1089–1100. [Google Scholar] [CrossRef]
- Pober, J.S. Endothelial activation: Intracellular signaling pathways. Arthritis Res. Ther. 2002, 4 (Suppl. 3), S109–S116. [Google Scholar] [CrossRef][Green Version]
- Blann, A.D.; Lip, G.Y.H. The endothelium in atherothrombotic disease: Assessment of function, mechanisms and clinical implications. Blood Coag. Fibrinolysis 1998, 9, 297–306. [Google Scholar] [CrossRef]
- Pober, J.S. Cytokine-mediated activation of vascular endothelium. Am. J. Pathol. 1988, 133, 426–433. [Google Scholar] [PubMed]
- Endemann, D.H.; Schiffrin, E.L. Endothelial dysfunction. J. Am. Soc. Nephrol. 2004, 15, 1983–1992. [Google Scholar] [CrossRef]
- Costa, C.; Virag, R. The Endothelial–Erectile Dysfunction Connection: An Essential Update. J. Sex. Med. 2009, 6, 2390–2404. [Google Scholar] [CrossRef] [PubMed]
- Egashira, K.; Suzuki, S.; Hirooka, Y.; Kai, H.; Sugimachi, M.; Imaizumi, T.; Takeshita, A. Impaired endothelium-dependent vasodilation of large epicardial and resistance coronary arteries in patients with essential hypertension. Different responses to acetylcholine and substance P. Hypertension 1995, 25, 201–206. [Google Scholar] [CrossRef]
- Tostes, R.C.; Carneiro, F.S.; Lee, A.J.; Giachini, F.R.; Leite, R.; Osawa, Y.; Webb, R.C. Cigarette smoking and erectile dysfunction: Focus on NO bioavailability and ROS generation. J. Sex. Med. 2008, 5, 1284–1295. [Google Scholar] [CrossRef] [PubMed]
- Illups, K.L. Erectile dysfunction as a marker for vascular disease. Curr. Urol. Rep. 2005, 6, 439–444. [Google Scholar] [CrossRef] [PubMed]
- Munzel, T.; Heitzer, T.; Harrison, D.G. The physiology and pathophysiology of the nitric oxide/superoxide system. Herz 1997, 22, 158–172. [Google Scholar] [CrossRef]
- Esposito, K.; Ciotola, M.; Sasso, F.C.; Cozzolino, D.; Saccomanno, F.; Assaloni, R.; Ceriello, A.; Giugliano, D. Effect of a single high-fat meal on endothelial function in patients with the metabolic syndrome: Role of tumor necrosis factor-alpha. Nutr. Metab. Cardiovasc. Dis. 2007, 17, 274–279. [Google Scholar] [CrossRef]
- Trepels, T.; Zeiher, A.M.; Fichtlscherer, S. The endothelium and inflammation. Endothelium 2006, 13, 423–429. [Google Scholar] [CrossRef]
- Neumann, P.; Gertzberg, N.; Johnson, A. TNF-alpha induces a decrease in eNOS promoter activity. Am. J. Physiol.-Lung Cell Mol. Physiol. 2004, 286, L452–L459. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Peppel, K.; Sivashanmugam, P.; Orman, E.S.; Brian, L.; Exum, S.T.; Freedman, N.J. Expression of tumor necrosis factor receptor-1 in arterial wall cells promotes atherosclerosis. Arterioscler. Thromb. Vasc. Biol. 2007, 27, 1087–1094. [Google Scholar] [CrossRef]
- Petrache, I.; Crow, M.T.; Neuss, M.; Garcia, J.G. Central involvement of Rho family GTPases in TNFalpha-mediated bovine pulmonary endothelial cell apoptosis. Biochem. Biophys. Res. Commun. 2003, 306, 244–249. [Google Scholar] [CrossRef]
- Torella, D.; Ellison, G.M.; Torella, M.; Vicinanza, C.; Aquila, I.; Iaconetti, C.; Scalise, M.; Marino, F.; Henning, B.J.; Lewis, F.C.; et al. Carbonic anhydrase activation is associated with worsened pathological remodeling in human ischemic diabetic cardiomyopathy. J. Am. Heart Assoc. 2014, 3, e000434. [Google Scholar] [CrossRef]
- Feldman, H.A.; Johannes, C.B.; Derby, C.A.; Kleinman, K.; Mohr, B.A.; Araujo, A.B.; McKinlay, J.B. Erectile Dysfunction and Coronary Risk Factors: Prospective Results from the Massachusetts Male Aging Study. Prev. Med. 2000, 30, 328–338. [Google Scholar] [CrossRef] [PubMed]
- Fung, M.M.; Bettencourt, R.; Barrett-Connor, E. Heart disease risk factors predict erectile dysfunction 25 years later: The Rancho Bernardo Study. J. Am. Coll. Cardiol. 2004, 43, 1405–1411. [Google Scholar] [CrossRef] [PubMed]
- Jackson, G. Erectile dysfunction and cardiovascular disease. Int. J. Clin. Pract. 1999, 53, 363–368. [Google Scholar] [PubMed]
- O’Kane, P.D.; Jackson, G. Erectile dysfunction: Is there silent obstructive coronary artery disease? Int. J. Clin. Pract. 2001, 55, 219–220. [Google Scholar]
- Kloner, R.A.; Mullin, S.H.; Shook, T.; Matthews, R.; Mayeda, G.; Burstein, S.; Peled, H.; Pollick, C.; Choudhary, R.; Rosen, R.; et al. Erectile dysfunction in the cardiac patient: How common and should we treat? J. Urol. 2003, 170, S46–S50. [Google Scholar] [CrossRef]
- Solomon, H.; Man, J.W.; Wierzbicki, A.S.; Jackson, G. Relation of erectile dysfunction to angiographic coronary artery disease. Am. J. Cardiol. 2003, 91, 230–231. [Google Scholar] [CrossRef]
- Montorsi, F.; Briganti, A.; Salonia, A.; Rigatti, P.; Margonato, A.; Macchi, A.; Galli, S.; Ravagnani, P.M.; Montorsi, P. Erectile dysfunction prevalence, time of onset and association with risk factors in 300 consecutive patients with acute chest pain and angiographically documented coronary artery disease. Eur. Urol. 2003, 44, 360–365. [Google Scholar] [CrossRef]
- Montorsi, P.; Ravagnani, P.M.; Galli, S.; Rotatori, F.; Briganti, A.; Salonia, A.; Dehò, F.; Montorsi, F. Common grounds for erectile dysfunction and coronary artery disease. Curr. Opin. Urol. 2004, 14, 361–365. [Google Scholar] [CrossRef]
- Pritzker, M. The penile stress test: A window to the hearts of man? Circulation 1999, 100 (Suppl. 1), 350–353. [Google Scholar]
- Vlachopoulos, C.; Rokkas, K.; Ioakeimidis, N.; Aggeli, C.; Michaelides, A.; Roussakis, G.; Fassoulakis, C.; Askitis, A.; Stefanadis, C. Prevalence of asymptomatic coronary artery disease in men with vasculogenic erectile dysfunction: A prospective angiographic study. Eur. Urol. 2005, 48, 996–1003. [Google Scholar] [CrossRef]
- Banks, E.; Joshy, G.; Abhayaratna, W.P.; Kritharides, L.; Macdonald, P.S.; Korda, R.J.; Chalmers, J.P. Erectile dysfunction severity as a risk marker for cardiovascular disease hospitalisation and all-cause mortality: A prospective cohort study. PLoS Med. 2013, 10, e1001372. [Google Scholar] [CrossRef] [PubMed]
- Montorsi, P.; Ravagnani, P.M.; Galli, S.; Rotatori, F.; Veglia, F.; Briganti, A.; Salonia, A.; Dehò, F.; Rigatti, P.; Montorsi, F.; et al. Association between erectile dysfunction and coronary artery disease—Role of coronary clinical presentation and extent of coronary vessels involvement: The COBRA trial. Eur. Heart J. 2006, 27, 2632–2639. [Google Scholar] [PubMed]
- Yaman, O.; Gulpinar, O.; Hasan, T.; Ozdol, C.; Ertas, F.S.; Ozgenci, E. Erectile dysfunction may predict coronary artery disease: Relationship between coronary artery calcium scoring and erectile dysfunction severity. Int. Urol. Nephrol. 2008, 40, 117–123. [Google Scholar] [CrossRef] [PubMed]
- Kaiser, D.R.; Billups, K.; Mason, C.; Wetterling, R.; Lundberg, J.L.; Bank, A.J. Impaired brachial artery endothelium dependent and -independent vasodilatation in men with erectile dysfunction and no other clinical cardiovascular disease. J. Am. Coll. Cardiol. 2004, 43, 179–184. [Google Scholar] [CrossRef][Green Version]
- Vlachopoulos, C.; Ioakeimidis, N.; Rokkas, K.; Angelis, A.; Terentes-Printzios, D.; Stefanadis, C.; Tousoulis, D. Acute effect of sildenafil on inflammatory markers/mediators in patients with vasculogenic erectile dysfunction. Int. J. Cardiol. 2015, 182, 98–101. [Google Scholar] [CrossRef]
- Carrizzo, A.; Izzo, C.; Forte, M.; Sommella, E.; Di Pietro, P.; Venturini, E.; Ciccarelli, M.; Galasso, G.; Rubattu, S.; Campiglia, P.; et al. A Novel Promising Frontier for Human Health: The Beneficial Effects of Nutraceuticals in Cardiovascular Diseases. Int. J. Mol. Sci.vol. 2020, 21, 8706. [Google Scholar] [CrossRef]
- Carrizzo, A.; Moltedo, O.; Damato, A.; Martinello, K.; Di Pietro, P.; Oliveti, M.; Acernese, F.; Giugliano, G.; Izzo, R.; Sommella, E.; et al. New Nutraceutical Combination Reduces Blood Pressure and Improves Exercise Capacity in Hypertensive Patients Via a Nitric Oxide-Dependent Mechanism. J. Am. Hear. Assoc. 2020, 9, 5. [Google Scholar] [CrossRef]
- Di Pietro, P.; Lizio, R.; Izzo, C.; Visco, V.; Damato, A.; Venturini, E.; de Lucia, M.; Galasso, G.; Migliarino, S.; Rasile, B.; et al. A Novel Combination of High-Load Omega-3 Lysine Complex (AvailOm®) and Anthocyanins Exerts Beneficial Cardiovascular Effects. Antioxidants 2022, 11, 896. [Google Scholar] [CrossRef]
- Salvatore, T.; Pafundi, P.C.; Galiero, R.; Rinaldi, L.; Caturano, A.; Vetrano, E.; Aprea, C.; Albanese, G.; Di Martino, A.; Ricozzi, C.; et al. Can Metformin Exert as an Active Drug on Endothelial Dysfunction in Diabetic Subjects? Biomedicines 2020, 9, 3. [Google Scholar] [CrossRef]
- Salvatore, T.; Caturano, A.; Galiero, R.; Di Martino, A.; Albanese, G.; Vetrano, E.; Sardu, C.; Marfella, R.; Rinaldi, L.; Sasso, F.C. Cardiovascular Benefits from Gliflozins: Effects on Endothelial Function. Biomedicines 2021, 9, 1356. [Google Scholar] [CrossRef]
- Ambrosino, P.; Grassi, G.; Maniscalco, M. Endothelial Dysfunction: From a Pathophysiological Mechanism to a Potential Therapeutic Target. Biomedicines 2022, 10, 78. [Google Scholar] [CrossRef] [PubMed]
- Vestergaard, N.; Søgaard, P.; Torp-Pedersen, C.; Aasbjerg, K. Relationship between treatment of erectile dysfunction and future risk of cardiovascular disease: A nationwide cohort study. Eur. J. Prev. Cardiol. 2017, 24, 1498–1505. [Google Scholar] [CrossRef] [PubMed]
- Nehra, A.; Jackson, G.; Miner, M.; Billups, K.L.; Burnett, A.L.; Buvat, J.; Carson, C.C.; Cunningham, G.R.; Ganz, P.; Goldstein, I.; et al. The Princeton III Consensus recommendations for the management of erectile dysfunction and cardiovascular disease. Mayo Clin. Proc. 2012, 87, 766–778. [Google Scholar] [CrossRef] [PubMed]
- Hippisley-Cox, J.; Coupland, C.; Brindle, P. Development and validation of QRISK3 risk prediction algorithms to estimate future risk of cardiovascular disease: Prospective cohort study. BMJ 2017, 357, j2099. [Google Scholar] [CrossRef] [PubMed]
- Inman, B.; Sauver, J.L.S.; Jacobson, D.J.; McGree, M.E.; Nehra, A.; Lieber, M.M.; Roger, V.L.; Jacobsen, S. A population-based, longitudinal study of erectile dysfunction and future coronary artery disease. Mayo Clin. Proc. 2009, 84, 108–113. [Google Scholar] [CrossRef] [PubMed]
- Billups, K.L.; Miner, M.M.; Wierzbicki, A.S.; Jackson, G. Gender-based cardiometabolic risk evaluation in minority and non-minority men grading the evidence of non-traditional determinants of cardiovascular risk. Int. J. Clin. Pract. 2010, 65, 134–147. [Google Scholar] [CrossRef]
- Lee, G.K.; Lee, L.C.; Liu, C.W.; Lim, S.L.; Shi, L.M.; Ong, H.Y.; Lim, Y.-T.; Yeo, T.-C. Framingham risk score inadequately predicts cardiac risk in young patients presenting with a first myocardial infarction. Ann. Acad. Med. Singap. 2010, 39, 163. [Google Scholar]
- Araujo, A.B.; Hall, S.A.; Ganz, P.; Chiu, G.R.; Rosen, R.C.; Kupelian, V.; Travison, T.G.; McKinlay, J.B. Does erectile dysfunction contribute to cardiovascular disease risk prediction beyond the Framingham Risk Score? J. Am. Coll. Cardiol. 2010, 55, 350–356. [Google Scholar] [CrossRef] [PubMed]
- Gupta, B.P.; Murad, M.H.; Clifton, M.M.; Prokop, L.; Nehra, A.; Kopecky, S.L. The effect of lifestyle modification and cardiovascular risk factor reduction on erectile dysfunction: A systematic review and meta-analysis. Arch. Intern. Med. 2011, 171, 1797–1803. [Google Scholar] [CrossRef]
- Fang, S.C.; Rosen, R.C.; Vita, J.A.; Ganz, P.; Kupelian, V. Changes in erectile dysfunction over time in relation to Framingham cardiovascular risk in the Boston Area Community Health (BACH) Survey. J. Sex. Med. 2015, 12, 100–108. [Google Scholar] [CrossRef]
- Ridker, P.M. High-sensitivity C-reactive protein: Potential adjunct for global risk assessment in the primary prevention of cardiovascular disease. Circulation 2001, 103, 1813–1818. [Google Scholar] [CrossRef] [PubMed]
- Ridker, P.M. C-reactive protein and the prediction of cardiovascular events among those at intermediate risk: Moving an inflammatory hypothesis toward consensus. J. Am. Coll. Cardiol. 2007, 49, 2129–2138. [Google Scholar] [CrossRef] [PubMed]
- Ferrandis-Cortes, C.; Martínez-Jabaloyas, J.M.; Díez-Calzadilla, N.A.; Hernández-Medina, J.A.; Chuan-Nuez, P. Cardiovascular risk assessment using high-sensitivity C-reactive protein in patients with erectile dysfunction. Urol. Int. 2013, 91, 187–191. [Google Scholar] [CrossRef] [PubMed]
- Laughlin, G.A.; Barrett-Connor, E.; Bergstrom, J. Low serum testosterone and mortality in older men. J. Clin. Endocrinol. Metab. 2008, 93, 68–75. [Google Scholar] [CrossRef] [PubMed]
- Haring, R.; Völzke, H.; Steveling, A.; Krebs, A.; Felix, S.B.; Schöfl, C.; Dörr, M.; Nauck, M.; Wallaschofski, H. Low serum testosterone levels are associated with increased risk of mortality in a population-based cohort of men aged 20-79. Eur. Heart J. 2010, 31, 1494–1501. [Google Scholar] [CrossRef] [PubMed]
- Jackson, G.; Boon, N.; Eardley, I.; Kirby, M.; Dean, J.; Hackett, G.; Montorsi, P.; Montorsi, F.; Vlachopoulos, C.; Kloner, R.; et al. Erectile dysfunction and coronary artery disease prediction: Evidence-based guidance and consensus. Int. J. Clin. Pract. 2010, 64, 848–857. [Google Scholar] [CrossRef] [PubMed]
- Corona, G.G.; Rastrelli, G.; Di Pasquale, G.; Sforza, A.; Mannucci, E.; Maggi, M. Endogenous Testosterone Levels and Cardiovascular Risk: Meta-Analysis of Observational Studies. J. Sex. Med. 2018, 15, 1260–1271. [Google Scholar] [CrossRef] [PubMed]
- Caminiti, G.; Volterrani, M.; Iellamo, F.; Marazzi, G.; Massaro, R.; Miceli, M.; Mammi, C.; Piepoli, M.; Fini, M.; Rosano, G.M.C. Effect of long-acting testosterone treatment on functional exercise capacity, skeletal muscle performance, insulin resistance, and baroreflex sensitivity in elderly patients with chronic heart failure a double-blind, placebocontrolled, randomized study. J. Am. Coll. Cardiol. 2009, 54, 919–927. [Google Scholar] [CrossRef]
- Vlachopoulos, C.; Jackson, G.; Stefanadis, C.; Montorsi, P. Erectile dysfunction in the cardiovascular patient. Eur. Heart J. 2013, 34, 2034–2046. [Google Scholar] [CrossRef]
- Dahabreh, I.J.; Paulus, J.K. Association of episodic physical and sexual activity with triggering of acute cardiac events: Systematic review and meta-analysis. Jama 2011, 305, 1225–1233. [Google Scholar] [CrossRef]
- Drory, Y.; Shapira, I.; Fisman, E.Z.; Pines, A. Myocardial ischemia during sexual activity in patients with coronary artery disease. Am. J. Cardiol. 1995, 75, 835–837. [Google Scholar] [CrossRef]
- DeBusk, R.; Drory, Y.; Goldstein, I.; Jackson, G.; Kaul, S.; Kimmel, S.E.; Kostis, J.B.; Kloner, R.A.; Lakin, M.; Meston, C.M.; et al. Management of sexual dysfunction in patients with cardiovascular disease: Recommendations of the Princeton consensus panel. Am. J. Cardiol. 2000, 86, 175–181. [Google Scholar] [CrossRef]
- Chrysant, S.G. Antihypertensive therapy causes erectile dysfunction. Curr. Opin. Cardiol. 2015, 30, 383–390. [Google Scholar] [CrossRef] [PubMed]
- Vlachopoulos, C.; Ioakeimidis, N.; Stefanadis, C. Biomarkers, erectile dysfunction, and cardiovascular risk prediction: The latest of an evolving concept. Asian J. Androl. 2015, 17, 17–20. [Google Scholar] [CrossRef] [PubMed]
- Sainz, I.; Amaya, J.; Garcia, M. Erectile dysfunction in heart disease patients. Int. J. Impot. Res. 2004, 16 (Suppl. 2), S13–S17. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Barrett-Connor, E. Cardiovascular risk stratification and cardiovascular risk factors associated with erectile dysfunction: Assessing cardiovascular risk in men with erectile dysfunction. Clin. Cardiol. 2004, 27 (Suppl. 1), I8–I13. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
De Leonardis, F.; Colalillo, G.; Finazzi Agrò, E.; Miano, R.; Fuschi, A.; Asimakopoulos, A.D. Endothelial Dysfunction, Erectile Deficit and Cardiovascular Disease: An Overview of the Pathogenetic Links. Biomedicines 2022, 10, 1848. https://doi.org/10.3390/biomedicines10081848
De Leonardis F, Colalillo G, Finazzi Agrò E, Miano R, Fuschi A, Asimakopoulos AD. Endothelial Dysfunction, Erectile Deficit and Cardiovascular Disease: An Overview of the Pathogenetic Links. Biomedicines. 2022; 10(8):1848. https://doi.org/10.3390/biomedicines10081848
Chicago/Turabian StyleDe Leonardis, Federico, Gaia Colalillo, Enrico Finazzi Agrò, Roberto Miano, Andrea Fuschi, and Anastasios D. Asimakopoulos. 2022. "Endothelial Dysfunction, Erectile Deficit and Cardiovascular Disease: An Overview of the Pathogenetic Links" Biomedicines 10, no. 8: 1848. https://doi.org/10.3390/biomedicines10081848
APA StyleDe Leonardis, F., Colalillo, G., Finazzi Agrò, E., Miano, R., Fuschi, A., & Asimakopoulos, A. D. (2022). Endothelial Dysfunction, Erectile Deficit and Cardiovascular Disease: An Overview of the Pathogenetic Links. Biomedicines, 10(8), 1848. https://doi.org/10.3390/biomedicines10081848





