The Determinants of Liver Fibrosis in Patients with Nonalcoholic Fatty Liver Disease and Type 2 Diabetes Mellitus
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Participants
2.2. Sample and Clinical Data Collection
2.3. Liver Steatosis and Stiffness Measurement
2.4. Statistical Analysis
3. Results
3.1. Characteristics of Entire Cohort
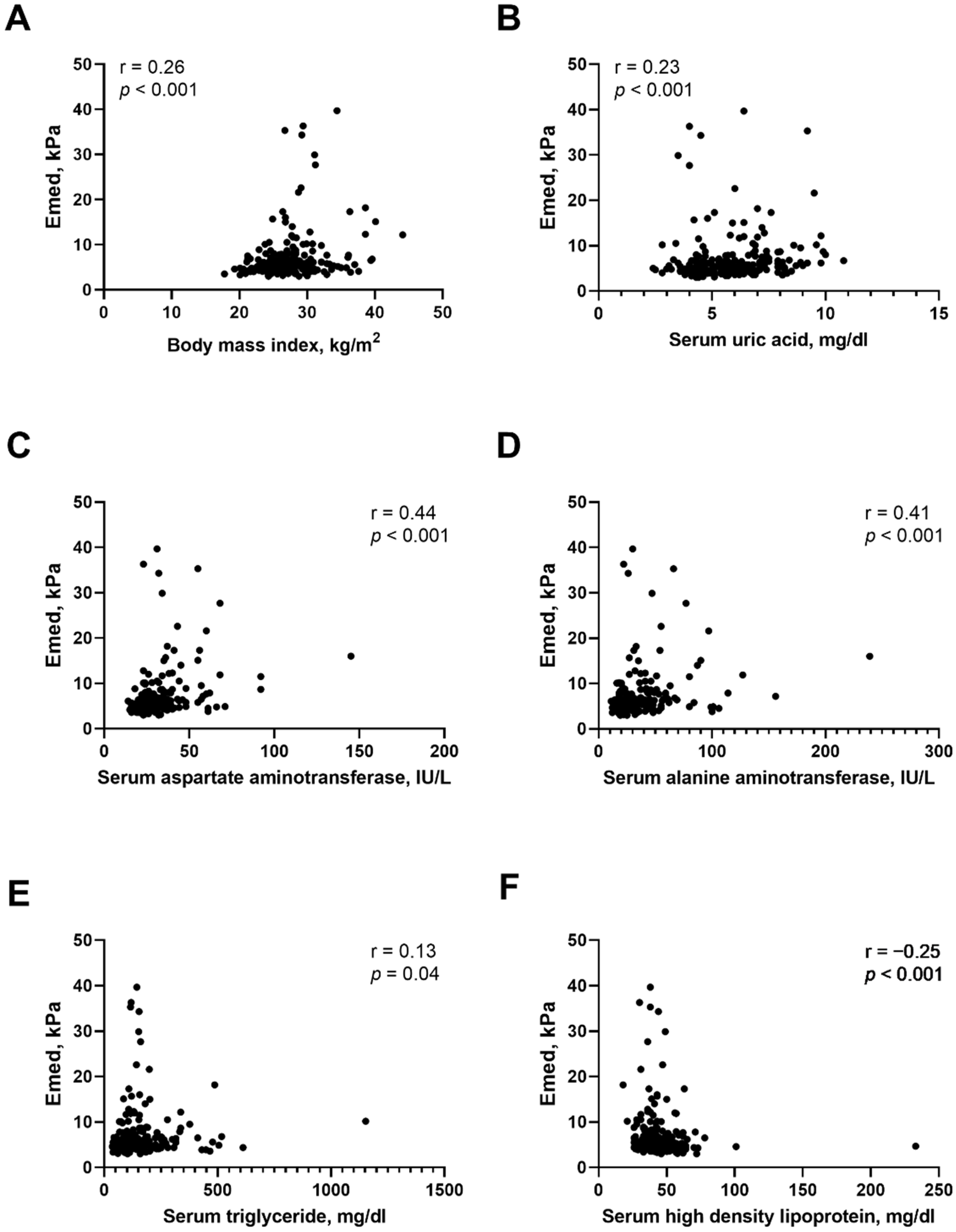
3.2. Determinants of Liver Stiffness in T2D Patients with NAFLD
3.3. Determinants of Risk of NASH in T2D Patients with NAFLD
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Vernon, G.; Baranova, A.; Younossi, Z.M. Systematic review: The epidemiology and natural history of non-alcoholic fatty liver disease and non-alcoholic steatohepatitis in adults. Aliment. Pharmacol. Ther. 2011, 34, 274–285. [Google Scholar] [CrossRef] [PubMed]
- Tiniakos, D.G.; Vos, M.B.; Brunt, E.M. Nonalcoholic fatty liver disease: Pathology and pathogenesis. Annu. Rev. Pathol. 2010, 5, 145–171. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Araujo, A.R.; Rosso, N.; Bedogni, G.; Tiribelli, C.; Bellentani, S. Global epidemiology of non-alcoholic fatty liver disease/non-alcoholic steatohepatitis: What we need in the future. Liver Int. Off. J. Int. Assoc. Study Liver 2018, 38 (Suppl. 1), 47–51. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wild, S.H.; Morling, J.R.; McAllister, D.A.; Kerssens, J.; Fischbacher, C.; Parkes, J.; Roderick, P.J.; Sattar, N.; Byrne, C.D.; on behalf of the Scottish and Southampton Diabetes and Liver Disease Group; et al. Type 2 diabetes and risk of hospital admission or death for chronic liver diseases. J. Hepatol. 2016, 64, 1358–1364. [Google Scholar] [CrossRef] [Green Version]
- Younossi, Z.M.; Koenig, A.B.; Abdelatif, D.; Fazel, Y.; Henry, L.; Wymer, M. Global epidemiology of nonalcoholic fatty liver disease-Meta-analytic assessment of prevalence, incidence, and outcomes. Hepatology 2016, 64, 73–84. [Google Scholar] [CrossRef] [Green Version]
- Lee, H.W.; Lee, J.S.; Kim, B.K.; Park, J.Y.; Kim, D.Y.; Ahn, S.H.; Kim, S.U. Evolution of liver fibrosis and steatosis markers in patients with type 2 diabetes after metformin treatment for 2years. J. Diabetes Complicat. 2021, 35, 107747. [Google Scholar] [CrossRef]
- Bhatt, H.B.; Smith, R.J. Fatty liver disease in diabetes mellitus. Hepatobiliary Surg. Nutr. 2015, 4, 101–108. [Google Scholar] [CrossRef]
- Targher, G.; Lonardo, A.; Byrne, C.D. Nonalcoholic fatty liver disease and chronic vascular complications of diabetes mellitus. Nature reviews. Endocrinology 2018, 14, 99–114. [Google Scholar] [CrossRef] [Green Version]
- Diehl, A.M.; Day, C. Cause, Pathogenesis, and Treatment of Nonalcoholic Steatohepatitis. N Engl. J. Med. 2017, 377, 2063–2072. [Google Scholar] [CrossRef] [PubMed]
- Alexander, M.; Loomis, A.K.; van der Lei, J.; Duarte-Salles, T.; Prieto-Alhambra, D.; Ansell, D.; Pasqua, A.; Lapi, F.; Rijnbeek, P.; Mosseveld, M.; et al. Risks and clinical predictors of cirrhosis and hepatocellular carcinoma diagnoses in adults with diagnosed NAFLD: Real-world study of 18 million patients in four European cohorts. BMC Med. 2019, 17, 95. [Google Scholar] [CrossRef]
- Koehler, E.M.; Plompen, E.P.; Schouten, J.N.; Hansen, B.E.; Darwish Murad, S.; Taimr, P.; Leebeek, F.W.; Hofman, A.; Stricker, B.H.; Castera, L.; et al. Presence of diabetes mellitus and steatosis is associated with liver stiffness in a general population: The Rotterdam study. Hepatology 2016, 63, 138–147. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vilar-Gomez, E.; Calzadilla-Bertot, L.; Wai-Sun Wong, V.; Castellanos, M.; Aller-de la Fuente, R.; Metwally, M.; Eslam, M.; Gonzalez-Fabian, L.; Alvarez-Quinones Sanz, M.; Conde-Martin, A.F.; et al. Fibrosis Severity as a Determinant of Cause-Specific Mortality in Patients with Advanced Nonalcoholic Fatty Liver Disease: A Multi-National Cohort Study. Gastroenterology 2018, 155, 443–457.e417. [Google Scholar] [CrossRef] [PubMed]
- Kwok, R.; Choi, K.C.; Wong, G.L.; Zhang, Y.; Chan, H.L.; Luk, A.O.; Shu, S.S.; Chan, A.W.; Yeung, M.W.; Chan, J.C.; et al. Screening diabetic patients for non-alcoholic fatty liver disease with controlled attenuation parameter and liver stiffness measurements: A prospective cohort study. Gut 2016, 65, 1359–1368. [Google Scholar] [CrossRef] [PubMed]
- Younossi, Z.M.; Golabi, P.; de Avila, L.; Paik, J.M.; Srishord, M.; Fukui, N.; Qiu, Y.; Burns, L.; Afendy, A.; Nader, F. The global epidemiology of NAFLD and NASH in patients with type 2 diabetes: A systematic review and meta-analysis. J. Hepatol. 2019, 71, 793–801. [Google Scholar] [CrossRef]
- Mota, M.; Banini, B.A.; Cazanave, S.C.; Sanyal, A.J. Molecular mechanisms of lipotoxicity and glucotoxicity in nonalcoholic fatty liver disease. Metabolism 2016, 65, 1049–1061. [Google Scholar] [CrossRef] [Green Version]
- Lin, C.Y.; Adhikary, P.; Cheng, K. Cellular protein markers, therapeutics, and drug delivery strategies in the treatment of diabetes-associated liver fibrosis. Adv. Drug. Deliv. Rev. 2021, 174, 127–139. [Google Scholar] [CrossRef]
- Loomba, R.; Jain, A.; Diehl, A.M.; Guy, C.D.; Portenier, D.; Sudan, R.; Singh, S.; Faulkner, C.; Richards, L.; Hester, K.D.; et al. Validation of Serum Test for Advanced Liver Fibrosis in Patients with Nonalcoholic Steatohepatitis. Clin. Gastroenterol. Hepatol. Off. Clin. Pract. J. Am. Gastroenterol. Assoc. 2019, 17, 1867–1876.e1863. [Google Scholar] [CrossRef]
- Shima, T.; Sakai, K.; Oya, H.; Katayama, T.; Mitsumoto, Y.; Mizuno, M.; Kanbara, Y.; Okanoue, T. Diagnostic accuracy of combined biomarker measurements and vibration-controlled transient elastography (VCTE) for predicting fibrosis stage of non-alcoholic fatty liver disease. J. Gastroenterol. 2020, 55, 100–112. [Google Scholar] [CrossRef]
- Radaelli, M.G.; Martucci, F.; Perra, S.; Accornero, S.; Castoldi, G.; Lattuada, G.; Manzoni, G.; Perseghin, G. NAFLD/NASH in patients with type 2 diabetes and related treatment options. J. Endocrinol. Investig. 2018, 41, 509–521. [Google Scholar] [CrossRef]
- Eddowes, P.J.; Sasso, M.; Allison, M.; Tsochatzis, E.; Anstee, Q.M.; Sheridan, D.; Guha, I.N.; Cobbold, J.F.; Deeks, J.J.; Paradis, V.; et al. Accuracy of FibroScan Controlled Attenuation Parameter and Liver Stiffness Measurement in Assessing Steatosis and Fibrosis in Patients with Nonalcoholic Fatty Liver Disease. Gastroenterology 2019, 156, 1717–1730. [Google Scholar] [CrossRef] [Green Version]
- de Ledinghen, V.; Vergniol, J.; Capdepont, M.; Chermak, F.; Hiriart, J.B.; Cassinotto, C.; Merrouche, W.; Foucher, J.; Brigitte le, B. Controlled attenuation parameter (CAP) for the diagnosis of steatosis: A prospective study of 5323 examinations. J. Hepatol. 2014, 60, 1026–1031. [Google Scholar] [CrossRef] [PubMed]
- Kao, W.Y.; Chang, I.W.; Chen, C.L.; Su, C.W.; Fang, S.U.; Tang, J.H.; Chang, C.C.; Chang, Y.J.; Wang, W. Fibroscan-Based Score to Predict Significant Liver Fibrosis in Morbidly Obese Patients with Nonalcoholic Fatty Liver Disease. Obes. Surg. 2020, 30, 1249–1257. [Google Scholar] [CrossRef] [PubMed]
- Newsome, P.N.; Sasso, M.; Deeks, J.J.; Paredes, A.; Boursier, J.; Chan, W.-K.; Yusuf, Y.; Czernichow, S.; Zheng, M.-H.; Vincent, W.-S.; et al. FibroScan-AST (FAST) score for the non-invasive identification of patients with non-alcoholic steatohepatitis with significant activity and fibrosis: A prospective derivation and global validation study. Lancet Gastroenterol. Hepatol. 2020, 5, 362–373. [Google Scholar] [CrossRef] [Green Version]
- Hideki, F.; Shinya, F.; Masaru, E.; Sawako, U.; Tatsuo, K.; Akihiro, T.; Yuji, N.; Shingo, T.; Naoki, N.; Norifumi, K. The FibroScan-aspartate aminotransferase score can stratify the disease severity in a Japanese cohort with fatty liver diseases. Sci. Rep. 2021, 11, 13844. [Google Scholar] [CrossRef]
- Zhao, H.; Song, X.; Li, Z.; Wang, X. Risk factors associated with nonalcohol fatty liver disease and fibrosis among patients with type 2 diabetes mellitus. Medicine 2018, 97, e12356. [Google Scholar] [CrossRef]
- Tada, T.; Toyoda, H.; Sone, Y.; Yasuda, S.; Miyake, N.; Kumada, T.; Tanaka, J. Type 2 diabetes mellitus: A risk factor for progression of liver fibrosis in middle-aged patients with non-alcoholic fatty liver disease. J. Gastroenterol. Hepatol. 2019, 34, 2011–2018. [Google Scholar] [CrossRef]
- Lomonaco, R.; Godinez Leiva, E.; Bril, F.; Shrestha, S.; Mansour, L.; Budd, J.; Portillo Romero, J.; Schmidt, S.; Chang, K.L.; Samraj, G.; et al. Advanced Liver Fibrosis Is Common in Patients with Type 2 Diabetes Followed in the Outpatient Setting: The Need for Systematic Screening. Diabetes Care 2021, 44, 399–406. [Google Scholar] [CrossRef]
- Ciardullo, S.; Monti, T.; Perseghin, G. High Prevalence of Advanced Liver Fibrosis Assessed by Transient Elastography among U.S. Adults with Type 2 Diabetes. Diabetes Care 2021, 44, 519–525. [Google Scholar] [CrossRef]
- Chen, K.; Sng, W.K.; Quah, J.H.; Liu, J.; Chong, B.Y.; Lee, H.K.; Wang, X.F.; Tan, N.C.; Chang, P.E.; Tan, H.C.; et al. Clinical spectrum of non-alcoholic fatty liver disease in patients with diabetes mellitus. PLoS ONE 2020, 15, e0236977. [Google Scholar] [CrossRef]
- Alam, M.S.; Kamrul-Hasan, A.B.M.; Kalam, S.T.; Rahman, S.M.M.; Hoque, M.I.; Islam, M.B.; Paul, A.K. Liver Stiffness Measurement by Using Transient Elastography in Bangladeshi Patients with Type 2 Diabetes Mellitus and Ultrasonography-Diagnosed Nonalcoholic Fatty Liver Disease. Diabetes Metab. Syndr Obes. 2021, 14, 3089–3096. [Google Scholar] [CrossRef]
- Pitisuttithum, P.; Chan, W.K.; Piyachaturawat, P.; Imajo, K.; Nakajima, A.; Seki, Y.; Kasama, K.; Kakizaki, S.; Fan, J.G.; Song, M.J.; et al. Predictors of advanced fibrosis in elderly patients with biopsy-confirmed nonalcoholic fatty liver disease: The GOASIA study. BMC Gastroenterol. 2020, 20, 88. [Google Scholar] [CrossRef] [PubMed]
- Lai, L.L.; Wan Yusoff, W.N.I.; Vethakkan, S.R.; Nik Mustapha, N.R.; Mahadeva, S.; Chan, W.K. Screening for non-alcoholic fatty liver disease in patients with type 2 diabetes mellitus using transient elastography. J. Gastroenterol. Hepatol. 2019, 34, 1396–1403. [Google Scholar] [CrossRef] [PubMed]
- Cvitanovic Tomas, T.; Moskon, M.; Mraz, M.; Rozman, D. Computational Modelling of Liver Metabolism and its Applications in Research and the Clinics. Acta Chim. Slov. 2018, 65, 253–265. [Google Scholar] [CrossRef] [PubMed]
- Tuong, T.T.K.; Tran, D.K.; Phu, P.Q.T.; Hong, T.N.D.; Dinh, T.C.; Chu, D.T. Non-Alcoholic Fatty Liver Disease in Patients with Type 2 Diabetes: Evaluation of Hepatic Fibrosis and Steatosis Using Fibroscan. Diagnostics 2020, 10, 159. [Google Scholar] [CrossRef] [Green Version]
- Chiang, D.J.; Pritchard, M.T.; Nagy, L.E. Obesity, diabetes mellitus, and liver fibrosis. Am. J. Physiol. Gastrointest Liver Physiol. 2011, 300, G697–G702. [Google Scholar] [CrossRef] [Green Version]
- Tolman, K.G.; Fonseca, V.; Dalpiaz, A.; Tan, M.H. Spectrum of liver disease in type 2 diabetes and management of patients with diabetes and liver disease. Diabetes Care 2007, 30, 734–743. [Google Scholar] [CrossRef] [Green Version]
- Makri, E.; Goulas, A.; Polyzos, S.A. Epidemiology, Pathogenesis, Diagnosis and Emerging Treatment of Nonalcoholic Fatty Liver Disease. Arch. Med. Res. 2021, 52, 25–37. [Google Scholar] [CrossRef]
- Kumar, V.; Mahato, R.I. Delivery and targeting of miRNAs for treating liver fibrosis. Pharm. Res. 2015, 32, 341–361. [Google Scholar] [CrossRef]
- Kuchay, M.S.; Martinez-Montoro, J.I.; Choudhary, N.S.; Fernandez-Garcia, J.C.; Ramos-Molina, B. Non-Alcoholic Fatty Liver Disease in Lean and Non-Obese Individuals: Current and Future Challenges. Biomedicines 2021, 9, 1346. [Google Scholar] [CrossRef]
- Zou, Z.Y.; Wong, V.W.; Fan, J.G. Epidemiology of nonalcoholic fatty liver disease in non-obese populations: Meta-analytic assessment of its prevalence, genetic, metabolic, and histological profiles. J. Dig. Dis. 2020, 21, 372–384. [Google Scholar] [CrossRef]
- Ye, Q.; Zou, B.; Yeo, Y.H.; Li, J.; Huang, D.Q.; Wu, Y.; Yang, H.; Liu, C.; Kam, L.Y.; Tan, X.X.E.; et al. Global prevalence, incidence, and outcomes of non-obese or lean non-alcoholic fatty liver disease: A systematic review and meta-analysis. The lancet. Gastroenterol. Hepatol. 2020, 5, 739–752. [Google Scholar] [CrossRef]
- Phipps, M.; Wattacheril, J. Non-alcoholic fatty liver disease (NAFLD) in non-obese individuals. Frontline Gastroenterol. 2020, 11, 478–483. [Google Scholar] [CrossRef] [PubMed]
- Yen, F.S.; Lai, J.N.; Wei, J.C.; Chiu, L.T.; Hwu, C.M.; Hou, M.C.; Hsu, C.C. Sulfonylureas may be useful for glycemic management in patients with diabetes and liver cirrhosis. PLoS ONE 2020, 15, e0243783. [Google Scholar] [CrossRef] [PubMed]
- Scheen, A.J. Pharmacokinetic and toxicological considerations for the treatment of diabetes in patients with liver disease. Expert Opin. Drug Metab. Toxicol. 2014, 10, 839–857. [Google Scholar] [CrossRef]
- Almahmoud, M.H.; Al Khawaja, N.M.; Alkinani, A.; Khader, Y.; Ajlouni, K.M. Prevalence of fatty liver disease and its associated factors among Jordanian patients with type 2 diabetes mellitus: A cross-sectional study. Ann. Med. Surg. 2021, 68, 102677. [Google Scholar] [CrossRef]
- Nascimbeni, F.; Aron-Wisnewsky, J.; Pais, R.; Tordjman, J.; Poitou, C.; Charlotte, F.; Bedossa, P.; Poynard, T.; Clement, K.; Ratziu, V.; et al. Statins, antidiabetic medications and liver histology in patients with diabetes with non-alcoholic fatty liver disease. BMJ Open Gastroenterol. 2016, 3, e000075. [Google Scholar] [CrossRef]
- Khunti, K.; Chatterjee, S.; Gerstein, H.C.; Zoungas, S.; Davies, M.J. Do sulphonylureas still have a place in clinical practice? Lancet Diabetes Endocrinol 2018, 6, 821–832. [Google Scholar] [CrossRef]
- Takeshita, Y.; Kanamori, T.; Tanaka, T.; Kaikoi, Y.; Kita, Y.; Takata, N.; Iida, N.; Arai, K.; Yamashita, T.; Harada, K.; et al. Study Protocol for Pleiotropic Effects and Safety of Sodium-Glucose Cotransporter 2 Inhibitor Versus Sulfonylurea in Patients with Type 2 Diabetes and Nonalcoholic Fatty Liver Disease. Diabetes Ther. 2020, 11, 549–560. [Google Scholar] [CrossRef] [Green Version]
- Siddiqui, M.T.; Amin, H.; Garg, R.; Chadalavada, P.; Al-Yaman, W.; Lopez, R.; Singh, A. Medications in type-2 diabetics and their association with liver fibrosis. World J. Gastroenterol. 2020, 26, 3249–3259. [Google Scholar] [CrossRef]
- Giannini, E.G.; Testa, R.; Savarino, V. Liver enzyme alteration: A guide for clinicians. CMAJ 2005, 172, 367–379. [Google Scholar] [CrossRef] [Green Version]
- Sanyal, D.; Mukherjee, P.; Raychaudhuri, M.; Ghosh, S.; Mukherjee, S.; Chowdhury, S. Profile of liver enzymes in non-alcoholic fatty liver disease in patients with impaired glucose tolerance and newly detected untreated type 2 diabetes. Indian J. Endocrinol. Metab. 2015, 19, 597–601. [Google Scholar] [CrossRef] [PubMed]
- Bril, F.; McPhaul, M.J.; Caulfield, M.P.; Clark, V.C.; Soldevilla-Pico, C.; Firpi-Morell, R.J.; Lai, J.; Shiffman, D.; Rowland, C.M.; Cusi, K. Performance of Plasma Biomarkers and Diagnostic Panels for Nonalcoholic Steatohepatitis and Advanced Fibrosis in Patients with Type 2 Diabetes. Diabetes Care 2020, 43, 290–297. [Google Scholar] [CrossRef] [PubMed]
- Mansour, A.; Mohajeri-Tehrani, M.R.; Samadi, M.; Gerami, H.; Qorbani, M.; Bellissimo, N.; Poustchi, H.; Hekmatdoost, A. Risk factors for non-alcoholic fatty liver disease-associated hepatic fibrosis in type 2 diabetes patients. Acta Diabetol. 2019, 56, 1199–1207. [Google Scholar] [CrossRef] [PubMed]
- Mofrad, P.; Contos, M.J.; Haque, M.; Sargeant, C.; Fisher, R.A.; Luketic, V.A.; Sterling, R.K.; Shiffman, M.L.; Stravitz, R.T.; Sanyal, A.J. Clinical and histologic spectrum of nonalcoholic fatty liver disease associated with normal ALT values. Hepatology 2003, 37, 1286–1292. [Google Scholar] [CrossRef] [PubMed]
- Sberna, A.L.; Bouillet, B.; Rouland, A.; Brindisi, M.C.; Nguyen, A.; Mouillot, T.; Duvillard, L.; Denimal, D.; Loffroy, R.; Verges, B.; et al. European Association for the Study of the Liver (EASL), European Association for the Study of Diabetes (EASD) and European Association for the Study of Obesity (EASO) clinical practice recommendations for the management of non-alcoholic fatty liver disease: Evaluation of their application in people with Type 2 diabetes. Diabet. Med. 2018, 35, 368–375. [Google Scholar] [CrossRef]
| Entire Cohort (n = 226) | F0-F1 (n = 176) | F2-F4 (n = 50) | p-Value | |
|---|---|---|---|---|
| Age, year | 62.1 ± 10.7 | 61.8 ± 10.5 | 63.1 ± 11.3 | 0.46 |
| Sex (male), % | 51.8 | 50.6 | 56.0 | 0.71 |
| Smoke, % | 26.1 | 25.0 | 30.0 | 0.48 |
| Alcohol, % | 19.9 | 18.8 | 24.0 | 0.41 |
| Heart disease, % | 34.5 | 35.2 | 32.0 | 0.67 |
| Hypertension, % | 65.0 | 63.6 | 70.0 | 0.41 |
| Hyperlipidemia, % | 81.0 | 82.4 | 76.0 | 0.31 |
| T2D duration, year | 10.0 ± 7.8 | 10.4 ± 7.6 | 8.6 ± 8.3 | 0.21 |
| Body Mass Index, kg/m2 | 27.3 ± 4.1 | 26.7 ± 3.8 | 29.2 ± 4.5 | 0.001 |
| Body Mass Index ≥25 kg/m2 | 68.6 | 63.6 | 86.0 | 0.003 |
| Emed, kPa | 5.3 (4.4,6.8) | 4.8 (4.3,5.8) | 10.4 (8.2,15.8) | <0.001 |
| CAP, dB/m | 294.5 ± 39.3 | 291.4 ± 38.4 | 305.4 ± 40.7 | 0.03 |
| FIB-4 | 1.5 ± 0.8 | 1.4 ± 0.6 | 2.0 ± 1.0 | <0.001 |
| FAST score | 0.25 ± 0.20 | 0.17 ± 0.12 | 0.50 ± 0.21 | <0.001 |
| FAST score ≥0.35, % | 22.6 | 8.0 | 74.0 | <0.001 |
| Medication | ||||
| Sulfonylurea (yes vs. no) | 47.3 | 43.8 | 60.0 | 0.04 |
| DPP4 inhibitor (yes vs. no) | 64.2 | 61.4 | 74.0 | 0.10 |
| Metformin (yes vs. no) | 81.0 | 82.4 | 76.0 | 0.31 |
| Thiazolidinedione (yes vs.no) | 38.9 | 42.6 | 26.0 | 0.03 |
| Insulin (yes vs. no) | 13.3 | 12.5 | 16.0 | 0.52 |
| Statin (yes vs. no) | 46.9 | 47.7 | 44.0 | 0.64 |
| Laboratory parameters | ||||
| Creatinine, mg/dL | 1.0 ± 0.5 | 1.0 ± 0.4 | 1.0 ± 0.6 | 0.25 |
| Hemoglobin, g/dL | 13.7 ± 1.6 | 13.7 ± 1.5 | 13.7 ± 1.8 | 0.76 |
| Albumin, g/dL | 4.6± 0.3 | 4.6 ± 0.3 | 4.6 ± 0.2 | 0.88 |
| Uric acid, mg/dL | 5.8 ± 1.6 | 5.7 ± 1.5 | 6.4 ± 1.8 | 0.003 |
| AST, IU/L | 30.6 ± 14.8 | 27.4 ± 9.8 | 41.8 ± 22.4 | <0.001 |
| ALT, IU/L | 34.8 ± 25.9 | 30.7 ± 19.9 | 49.3 ± 37.2 | <0.001 |
| Cholesterol, mg/dL | 167.7 ± 39.7 | 167.9 ± 39.4 | 167.0 ± 41.2 | 0.89 |
| Triglyceride, mg/dL | 127 (95,179) | 123 (91,178) | 148 (108,181) | 0.09 |
| HDL-C, mg/dL | 44.7 ± 16.9 | 45.9 ± 18.2 | 40.3 ± 10.3 | 0.006 |
| LDL-C, mg/dL | 94.2 ± 33.1 | 93.6 ± 31.7 | 96.0 ± 38.1 | 0.69 |
| HbA1C, % | 6.9 (6.4,7.9) | 6.9 (6.4,7.8) | 7.6 (6.5,8.5) | 0.07 |
| Crude OR (95%Cl) | p-Value | Adjusted OR (95%Cl) | p-Value | |
|---|---|---|---|---|
| Clinical data | ||||
| Age, year | 1.01 (0.98–1.04) | 0.44 | 1.06 (1.01–1.11) | 0.02 |
| Sex (female vs. male) | 0.85 (0.46–1.57) | 0.60 | 1.34 (0.57–3.11) | 0.50 |
| T2D duration, year | 0.97 (0.92–1.02) | 0.18 | - | - |
| Body mass index, kg/m2 | 1.15 (1.06–1.24) | <0.001 | 1.14 (1.02–1.26) | 0.02 |
| Smoke (yes vs. no) | 1.29 (0.64–2.57) | 0.48 | - | - |
| Alcohol (yes vs. no) | 1.237 (0.65–2.90) | 0.41 | - | - |
| Heart disease (yes vs. no) | 0.87 (0.44–1.69) | 0.67 | - | - |
| Hyperlipidemia (yes vs. no) | 0.68 (0.32–1.44) | 0.31 | - | - |
| Hypertension (yes vs. no) | 1.33 (0.68–2.63) | 0.41 | - | - |
| Sulfonylurea (yes vs. no) | 1.93 (1.02–3.66) | 0.04 | 2.83 (1.22–6.57) | 0.02 |
| DPP4 inhibitor (yes vs. no) | 1.79 (0.89–3.61) | 0.10 | - | - |
| Metfotmin (yes vs. no) | 0.68 (0.32–1.44) | 0.31 | - | - |
| Thiazolidinedione (yes vs. no) | 0.47 (0.24–0.95) | 0.04 | 0.65 (0.27–1.56) | 0.34 |
| Insulin (yes vs. no) | 1.33 (0.55–3.21) | 0.52 | - | - |
| Statin (yes vs. no) | 0.86 (0.46–1.62) | 0.64 | - | - |
| Laboratory data | ||||
| CAP, dB/m | 1.01 (1.00–1.02) | 0.03 | 1.01 (0.99–1.02) | 0.15 |
| Creatinine, mg/dL | 1.51 (0.82–2.77) | 0.18 | - | - |
| Hemoglobin, g/dL | 1.04 (0.85–1.27) | 0.73 | - | - |
| Albumin, g/dL | 1.08 (0.41–2.88) | 0.88 | - | - |
| Uric acid, mg/dL | 1.34 (1.10–1.64) | 0.004 | 1.20 (0.94–1.53) | 0.15 |
| AST, IU/L | 1.07 (1.04–1.10) | <0.001 | 1.12 (1.05–1.19) | <0.001 |
| ALT, IU/L | 1.03 (1.01–1.04) | <0.001 | 0.98 (0.95–1.01) | 0.24 |
| Cholesterol, mg/dL | 1.00 (0.99–1.01) | 0.89 | - | - |
| Log (Triglyceride) | 3.36 (0.90–12.48) | 0.07 | - | - |
| HDL-C, mg/dL | 0.96 (0.93–0.99) | 0.01 | 0.97 (0.93–1.01) | 0.17 |
| LDL-C, mg/dL | 1.00 (0.99–1.01) | 0.66 | - | - |
| HbA1C, % | 1.00 (0.93–1.07) | 0.96 | - | - |
| Age ≥65 Years | Age <65 Years | BMI ≥25 kg/m2 | BMI <25 kg/m2 | |||||
|---|---|---|---|---|---|---|---|---|
| Adjusted OR (95%Cl) | p-Value | Adjusted OR (95%Cl) | p-Value | Adjusted OR (95%Cl) | p-Value | Adjusted OR (95%Cl) | p-Value | |
| Age, year | - | - | - | - | 1.02 (0.98–1.07) | 0.25 | 1.15 (0.97–1.36) | 0.19 |
| Sex(female vs. male) | 2.30 (0.56–9.40) | 0.25 | 1.05 (0.34–3.23) | 0.94 | 1.01 (0.42–2.43) | 0.98 | 43.56 (0.65–2942.19) | 0.08 |
| Body mass index, kg/m2 | 1.41 (1.08–1.84) | 0.01 | 1.08 (0.96–1.22) | 0.21 | - | - | - | - |
| Sulfonylurea (yes vs. no) | 11.37 (2.06–62.88) | 0.005 | 1.64 (0.55–4.84) | 0.37 | 2.80 (1.14–6.84) | 0.02 | 0.89 (0.06–14.49) | 0.94 |
| Thiazolidinedione (yes vs. no) | 0.47 (0.11–1.97) | 0.30 | 0.43 (0.13–1.45) | 0.17 | 0.47 (0.18–1.23) | 0.13 | 1.99 (0.13–30.60) | 0.62 |
| Uric acid, mg/dL | 1.39 (0.92–2.10) | 0.12 | 1.26 (0.88–1.80) | 0.21 | 1.22 (0.93–1.60) | 0.15 | 2.68 (1.01–7.11) | 0.04 |
| AST | 1.32 (1.10–1.59) | 0.003 | 1.10 (1.02–1.18) | 0.02 | 1.11 (1.04–1.18) | 0.002 | 1.60 (1.01–2.52) | 0.04 |
| ALT | 0.96 (0.87–1.05) | 0.36 | 0.98 (0.95–1.02) | 0.28 | 0.98 (0.95–1.02) | 0.26 | 0.77 (0.55–1.08) | 0.13 |
| HDL-C, mg/dL | 1.01 (0.94–1.09) | 0.75 | 0.96 (0.90–1.02) | 0.16 | 0.98 (0.94–1.03) | 0.42 | 0.84 (0.66–1.07) | 0.15 |
| Crude OR (95%Cl) | p-Value | Adjusted OR (95%Cl) | p-Value | |
|---|---|---|---|---|
| Clinical data | ||||
| Age, year | 0.97 (0.94–0.99) | 0.04 | 1.07 (1.02–1.13) | 0.01 |
| Sex (female vs. male) | 1.20 (0.65–2.22) | 0.56 | 2.33 (0.88–6.15) | 0.09 |
| T2D duration, year | 0.96 (0.91–1.01) | 0.09 | - | - |
| Body mass index, kg/m2 | 1.20 (1.11–1.31) | <0.001 | 1.27 (1.13–1.44) | <0.001 |
| Smoke (yes vs. no) | 0.84 (0.41–1.74) | 0.63 | - | - |
| Alcohol (yes vs. no) | 1.14 (0.53–2.45) | 0.74 | - | - |
| Heart disease (yes vs. no) | 0.83 (0.431.62) | 0.59 | - | - |
| Hyperlipidemia (yes vs. no) | 1.13 (0.50–2.53) | 0.77 | - | - |
| Hypertension (yes vs. no) | 0.88 (0.46–1.68) | 0.70 | - | - |
| Sulfonylurea (yes vs. no) | 1.64 (0.87–3.08) | 0.12 | - | - |
| DPP4 inhibitor (yes vs. no) | 1.45 (0.74–2.86) | 0.28 | - | - |
| Metformin (yes vs. no) | 0.53 (0.25–1.09) | 0.09 | - | - |
| Thiazolidinedione (yes vs. no) | 0.35 (0.17–0.73) | 0.005 | 0.52 (1.09–1.18) | 0.22 |
| Insulin (yes vs. no) | 1.57 (0.67–3.69) | 0.30 | - | - |
| Statin (yes vs. no) | 0.60 (0.32–1.14) | 0.12 | - | - |
| Laboratory data | ||||
| Creatinine, mg/dL | 0.99 (0.51–1.95) | 0.99 | - | - |
| Hemoglobin, g/dL | 1.20 (0.97–1.49) | 0.09 | - | - |
| Albumin, g/dL | 1.08 (0.41–2.87) | 0.87 | - | - |
| Uric acid, mg/dL | 1.17 (0.96–1.42) | 0.12 | - | - |
| AST, IU/L | 1.29 (1.20–1.40) | <0.001 | - | - |
| ALT, IU/L | 1.10 (1.07–1.14) | <0.001 | 1.13 (1.09–1.18) | <0.001 |
| Cholesterol, mg/dL | 1.01 (0.99–1.01) | 0.17 | - | - |
| Log (Triglyceride) | 5.94 (1.57–22.40) | 0.009 | 3.36 (0.51–22.01) | 0.20 |
| HDL-C, mg/dL | 1.00 (0.99–1.02) | 0.77 | - | - |
| LDL-C, mg/dL | 1.01 (0.99–1.02) | 0.19 | - | - |
| HbA1C, % | 1.01 (0.95–1.08) | 0.69 | - | - |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dai, C.-Y.; Fang, T.-J.; Hung, W.-W.; Tsai, H.-J.; Tsai, Y.-C. The Determinants of Liver Fibrosis in Patients with Nonalcoholic Fatty Liver Disease and Type 2 Diabetes Mellitus. Biomedicines 2022, 10, 1487. https://doi.org/10.3390/biomedicines10071487
Dai C-Y, Fang T-J, Hung W-W, Tsai H-J, Tsai Y-C. The Determinants of Liver Fibrosis in Patients with Nonalcoholic Fatty Liver Disease and Type 2 Diabetes Mellitus. Biomedicines. 2022; 10(7):1487. https://doi.org/10.3390/biomedicines10071487
Chicago/Turabian StyleDai, Chia-Yen, Tzu-Jung Fang, Wei-Wen Hung, Hui-Ju Tsai, and Yi-Chun Tsai. 2022. "The Determinants of Liver Fibrosis in Patients with Nonalcoholic Fatty Liver Disease and Type 2 Diabetes Mellitus" Biomedicines 10, no. 7: 1487. https://doi.org/10.3390/biomedicines10071487
APA StyleDai, C.-Y., Fang, T.-J., Hung, W.-W., Tsai, H.-J., & Tsai, Y.-C. (2022). The Determinants of Liver Fibrosis in Patients with Nonalcoholic Fatty Liver Disease and Type 2 Diabetes Mellitus. Biomedicines, 10(7), 1487. https://doi.org/10.3390/biomedicines10071487







