Alcohol Use Disorder: Neurobiology and Therapeutics
Abstract
1. Introduction
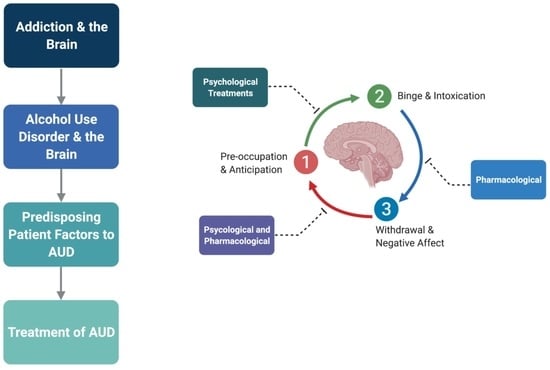
2. Addiction and the Brain
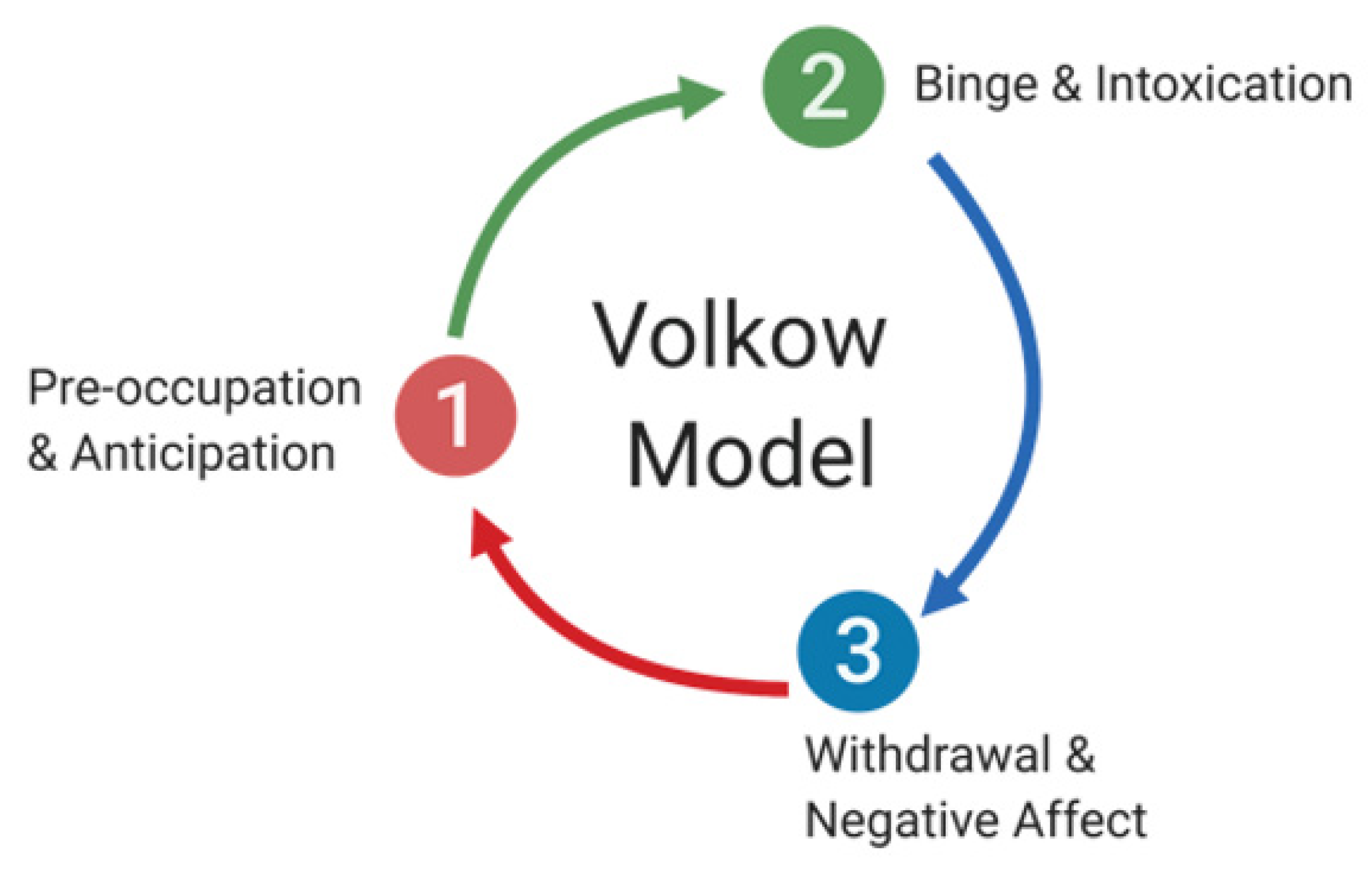
Stages of Addiction and Its Neuroanatomical Correlates
- (i).
- Binge/Intoxication Stage—Basal Ganglia
- (ii).
- Withdrawal/Negative Affect Stage—Amygdala
- (iii).
- Preoccupation/Anticipation Stage—Prefrontal Cortex
3. AUD and the Brain
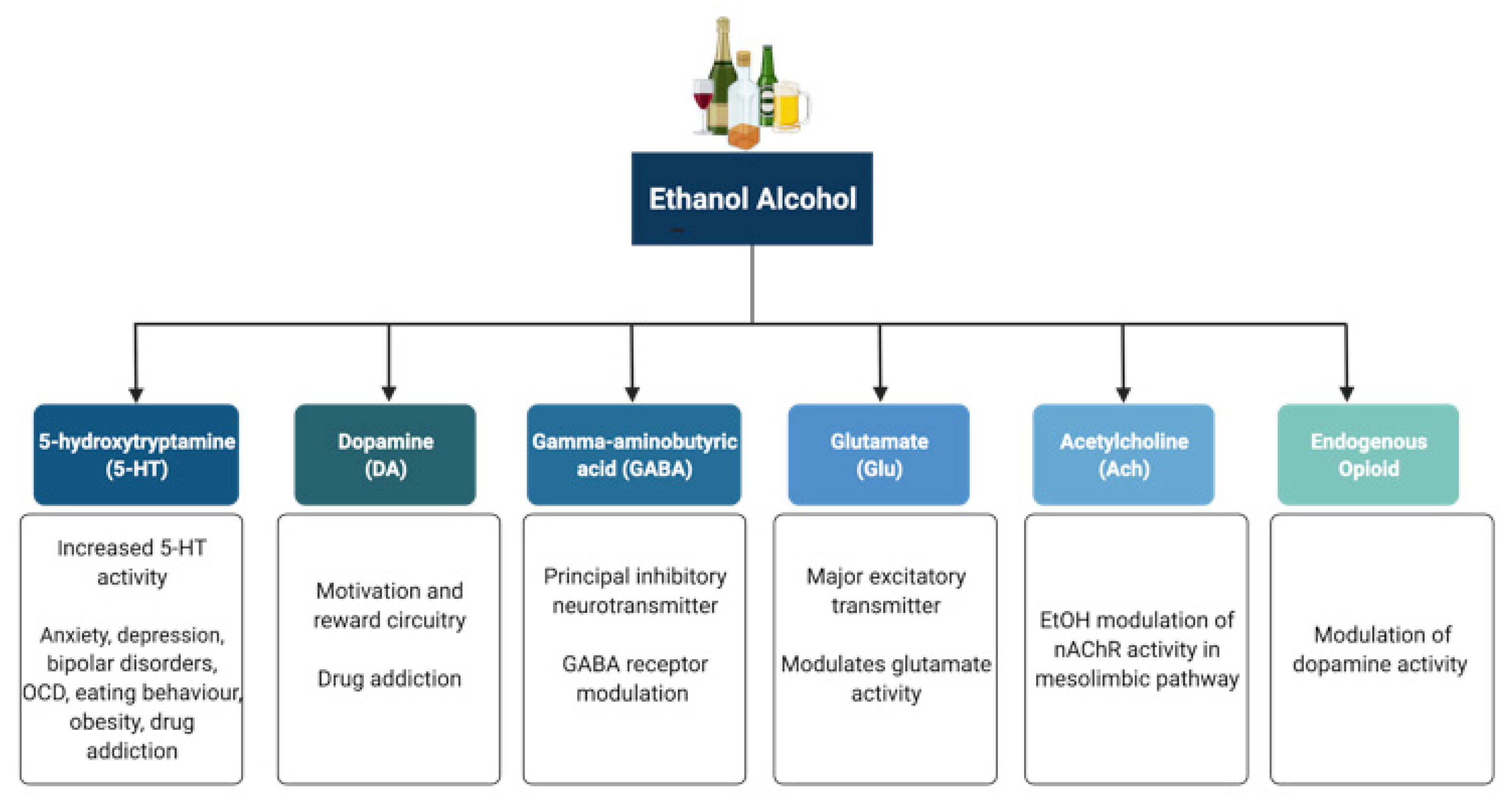
3.1. Effect of Alcohol on Neurotransmitter Systems
3.2. Effects of Alcohol on Other Synaptic Targets
4. Factors That Predispose Patients to AUD
5. Treatment of AUD
5.1. Psychological and Non-Pharmacological Therapies for AUD
5.2. Pharmacological Therapies for AUD
6. Conclusions and Future Directions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Koob, G.F.; Le Moal, M. Drug Abuse: Hedonic Homeostatic Dysregulation. Science 1997, 278, 52–58. [Google Scholar] [CrossRef]
- Brower, K.J.; Hall, J.M. Effects of age and alcoholism on sleep: A controlled study. J. Stud. Alcohol 2001, 62, 335–343. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Centers for Disease Control and Prevention. Report—Alcohol-Attributable Deaths, U.S., By Sex, Excessive Use; Alcohol Related Disease Impact (ARDI) Application; Centers for Disease Control and Prevention: Atlanta, GA, USA, 2013. Available online: https://nccd.cdc.gov/DPH_ARDI/Default/Report.aspx?T=AAM&P=f6d7eda7-036e-4553-9968-9b17ffad620e&R=d7a9b303-48e9-4440-bf47-070a4827e1fd&M=8E1C5233-5640-4EE8-9247-1ECA7DA325B9&F=&D (accessed on 12 June 2020).
- Substance Abuse and Mental Health Services Administration (SAMHSA). Results from the 2018 National Survey on Drug Use and Health: Detailed Tables, Sections 1–3; Substance Abuse and Mental Health Services Administration (SAMHSA), Center for Behavioral Health Statistics and Quality (CBHSQ): Rockville, MD, USA, 2018. Available online: https://www.samhsa.gov/data/sites/default/files/cbhsq-reports/NSDUHDetailedTabs2018R2/NSDUHDetailedTabs2018.pdf (accessed on 12 June 2020).
- World Health Organization. Global Status Report on Alcohol and Health 2014; World Health Organization: Geneve, Switzerland, 2014; pp. 1–392.
- Shield, K.D.; Parry, C.; Rehm, J. Chronic Diseases and Conditions Related to Alcohol Use. Alcohol Res. Curr. Rev. 2014, 35, 155–171. [Google Scholar]
- Day, E.; Rudd, J.H.F. Alcohol use disorders and the heart. Addiction 2019, 114, 1670–1678. [Google Scholar] [CrossRef] [PubMed]
- Leong, C.; Bolton, J.M.; Ekuma, O.; Prior, H.J.; Singal, D.; Nepon, J.; Konrad, G.; Paillé, M.; Finlayson, G.; Nickel, N. Association of alcohol use disorder on alcohol-related cancers, diabetes, ischemic heart disease and death: A population-based, matched cohort study. Addiction 2021, 117, 368–381. [Google Scholar] [CrossRef]
- Reynolds, K.; Lewis, B.L.; Nolen, J.D.L.; Kinney, G.; Sathya, B.; He, J. Alcohol Consumption and Risk of Stroke. JAMA 2003, 289, 579–588. [Google Scholar] [CrossRef] [PubMed]
- Roerecke, M.; Rehm, J. Chronic heavy drinking and ischaemic heart disease: A systematic review and meta-analysis. Open Hear. 2014, 1, e000135. [Google Scholar] [CrossRef]
- Xi, B.; Veeranki, S.P.; Zhao, M.; Ma, C.; Yan, Y.; Mi, J. Relationship of Alcohol Consumption to All-Cause, Cardiovascular, and Cancer-Related Mortality in U.S. Adults. J. Am. Coll. Cardiol. 2017, 70, 913–922. [Google Scholar] [CrossRef]
- Buchanan, R.; Sinclair, J.M.A. Alcohol use disorder and the liver. Addiction 2020, 116, 1270–1278. [Google Scholar] [CrossRef]
- Crabb, D.W.; Im, G.Y.; Szabo, G.; Mellinger, J.L.; Lucey, M.R. Diagnosis and Treatment of Alcohol-Associated Liver Diseases: 2019 Practice Guidance from the American Association for the Study of Liver Diseases. Hepatology 2020, 71, 306–333. [Google Scholar] [CrossRef]
- Haas, S.L.; Ye, W.; Löhr, M. Alcohol consumption and digestive tract cancer. Curr. Opin. Clin. Nutr. Metab. Care 2012, 15, 457–467. [Google Scholar] [CrossRef] [PubMed]
- Seitz, H.K.; Maurer, B.; Stickel, F. Alcohol Consumption and Cancer of the Gastrointestinal Tract. Dig. Dis. 2005, 23, 297–303. [Google Scholar] [CrossRef] [PubMed]
- Majumder, S.; Chari, S.T. Chronic pancreatitis. Lancet 2016, 387, 1957–1966. [Google Scholar] [CrossRef]
- Yang, A.L.; Vadhavkar, S.; Singh, G.; Omary, M.B. Epidemiology of Alcohol-Related Liver and Pancreatic Disease in the United States. Arch. Intern. Med. 2008, 168, 649–656. [Google Scholar] [CrossRef]
- Gorky, J.; Schwaber, J. The role of the gut–brain axis in alcohol use disorders. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 2015, 65, 234–241. [Google Scholar] [CrossRef]
- Leclercq, S.; Schwarz, M.; Delzenne, N.M.; Stärkel, P.; de Timary, P. Alterations of kynurenine pathway in alcohol use disorder and abstinence: A link with gut microbiota, peripheral inflammation and psychological symptoms. Transl. Psychiatry 2021, 11, 1–9. [Google Scholar] [CrossRef]
- Foran, H.M.; O’Leary, K.D. Alcohol and intimate partner violence: A meta-analytic review. Clin. Psychol. Rev. 2008, 28, 1222–1234. [Google Scholar] [CrossRef]
- Boles, S.M.; Miotto, K. Substance abuse and violence: A review of the literature. Aggress. Violent Behav. 2003, 8, 155–174. [Google Scholar] [CrossRef]
- Sher, L. Alcohol consumption and suicide. QJM 2005, 99, 57–61. [Google Scholar] [CrossRef]
- Butt, P.; Beirness, D.; Gliksman, L.; Paradis, C.; Stockwell, T. Alcohol and Health in Canada: A Summary of Evidence and Guidelines for Low-Risk Drinking; Canadian Centre on Substance Abuse: Ottawa, ON, Canada, 2011; pp. 1–66. [Google Scholar]
- Koob, G.F.; Volkow, N.D. Neurobiology of addiction: A neurocircuitry analysis. Lancet Psychiatry 2016, 3, 760–773. [Google Scholar] [CrossRef]
- Koob, G.F.; Le Moal, M. Drug addiction, dysregulation of reward, and allostasis. Neuropsychopharmacology 2001, 24, 97–129. [Google Scholar] [CrossRef]
- Robison, A.; Nestler, E.J. Transcriptional and epigenetic mechanisms of addiction. Nat. Rev. Neurosci. 2011, 12, 623–637. [Google Scholar] [CrossRef] [PubMed]
- Maze, I.; Nestler, E.J. The epigenetic landscape of addiction. Ann. N. Y. Acad. Sci. 2011, 1216, 99–113. [Google Scholar] [CrossRef] [PubMed]
- Koob, G.F.; Buck, C.L.; Cohen, A.; Edwards, S.; Park, P.E.; Schlosburg, J.E.; Schmeichel, B.; Vendruscolo, L.F.; Wade, C.L.; Whitfield, T.W.; et al. Addiction as a stress surfeit disorder. Neuropsychopharmacology 2013, 76, 370–382. [Google Scholar] [CrossRef]
- Moeller, F.G.; Barratt, E.S.; Dougherty, D.M.; Schmitz, J.; Swann, A.C. Psychiatric Aspects of Impulsivity. Am. J. Psychiatry 2001, 158, 1783–1793. [Google Scholar] [CrossRef]
- Berlin, G.S.; Hollander, E. Compulsivity, impulsivity, and the DSM-5 process. CNS Spectr. 2013, 19, 62–68. [Google Scholar] [CrossRef]
- Koob, G.F.; Le Moal, M. Addiction and the Brain Antireward System. Annu. Rev. Psychol. 2008, 59, 29–53. [Google Scholar] [CrossRef]
- Goldstein, R.Z.; Volkow, N.D. Drug Addiction and Its Underlying Neurobiological Basis: Neuroimaging Evidence for the Involvement of the Frontal Cortex. Am. J. Psychiatry 2002, 159, 1642–1652. [Google Scholar] [CrossRef]
- Mitchell, J.M.; O’Neil, J.P.; Janabi, M.; Marks, S.M.; Jagust, W.J.; Fields, H.L. Alcohol Consumption Induces Endogenous Opioid Release in the Human Orbitofrontal Cortex and Nucleus Accumbens. Sci. Transl. Med. 2012, 4, 116ra6. [Google Scholar] [CrossRef]
- Volkow, N.D.; Wang, G.-J.; Telang, F.; Fowler, J.S.; Logan, J.; Jayne, M.; Ma, Y.; Pradhan, K.; Wong, C. Profound Decreases in Dopamine Release in Striatum in Detoxified Alcoholics: Possible Orbitofrontal Involvement. J. Neurosci. 2007, 27, 12700–12706. [Google Scholar] [CrossRef]
- Koob, G.F.; Roberts, A.J.; Schulteis, G.; Parsons, L.H.; Heyser, C.J.; Hyytia, P.; Merlo-Pich, E.; Weiss, F. Neurocircuitry Targets in Ethanol Reward and Dependence. Alcohol. Clin. Exp. Res. 1998, 22, 3–9. [Google Scholar] [CrossRef] [PubMed]
- Caine, S.B.; Thomsen, M.; Gabriel, K.; Berkowitz, J.S.; Gold, L.H.; Koob, G.F.; Tonegawa, S.; Zhang, J.; Xu, M. Lack of Self-Administration of Cocaine in Dopamine D1 Receptor Knock-Out Mice. J. Neurosci. 2007, 27, 13140–13150. [Google Scholar] [CrossRef] [PubMed]
- Zweifel, L.S.; Parker, J.G.; Lobb, C.J.; Rainwater, A.; Wall, V.Z.; Fadok, J.P.; Darvas, M.; Kim, M.J.; Mizumori, S.J.Y.; Paladini, C.A.; et al. Disruption of NMDAR-dependent burst firing by dopamine neurons provides selective assessment of phasic dopamine-dependent behavior. Proc. Natl. Acad. Sci. USA 2009, 106, 7281–7288. [Google Scholar] [CrossRef] [PubMed]
- Volkow, N.D.; Fowler, J.S.; Wang, G.-J. The addicted human brain: Insights from imaging studies. J. Clin. Investig. 2003, 111, 1444–1451. [Google Scholar] [CrossRef]
- Covey, D.P.; Roitman, M.F.; Garris, P.A. Illicit dopamine transients: Reconciling actions of abused drugs. Trends Neurosci. 2014, 37, 200–210. [Google Scholar] [CrossRef]
- Belin, D.; Jonkman, S.; Dickinson, A.; Robbins, T.; Everitt, B. Parallel and interactive learning processes within the basal ganglia: Relevance for the understanding of addiction. Behav. Brain Res. 2009, 199, 89–102. [Google Scholar] [CrossRef]
- Lüscher, C.; Malenka, R.C. Drug-Evoked Synaptic Plasticity in Addiction: From Molecular Changes to Circuit Remodeling. Neuron 2011, 69, 650–663. [Google Scholar] [CrossRef]
- Kalivas, P.W. The glutamate homeostasis hypothesis of addiction. Nat. Rev. Neurosci. 2009, 10, 561–572. [Google Scholar] [CrossRef]
- Cunningham, K.A.; Anastasio, N.C. Serotonin at the nexus of impulsivity and cue reactivity in cocaine addiction. Neuropharmacology 2013, 76, 460–478. [Google Scholar] [CrossRef]
- Everitt, B.J.; Belin, D.; Economidou, D.; Pelloux, Y.; Dalley, J.; Robbins, T. Neural mechanisms underlying the vulnerability to develop compulsive drug-seeking habits and addiction. Philos. Trans. R. Soc. B Biol. Sci. 2008, 363, 3125–3135. [Google Scholar] [CrossRef]
- Dani, J.A.; Heinemann, S. Molecular and Cellular Aspects Minireview of Nicotine Abuse. Neuron 1996, 16, 905–908. [Google Scholar] [CrossRef]
- Vashchinkina, E.; Panhelainen, A.; Aitta-Aho, T.; Korpi, E.R. GABAA receptor drugs and neuronal plasticity in reward and aversion: Focus on the ventral tegmental area. Front. Pharmacol. 2014, 5, 256. [Google Scholar] [CrossRef] [PubMed]
- Koob, G.F.; Le Moal, M. Plasticity of reward neurocircuitry and the ‘dark side’ of drug addiction. Nat. Neurosci. 2005, 8, 1442–1444. [Google Scholar] [CrossRef] [PubMed]
- Paterson, N.E.; Myers, C.; Markou, A. Effects of repeated withdrawal from continuous amphetamine administration on brain reward function in rats. Psychopharmacology 2000, 152, 440–446. [Google Scholar] [CrossRef] [PubMed]
- Schulteis, G.; Markou, A.; Cole, M.; Koob, G.F. Decreased brain reward produced by ethanol withdrawal. Proc. Natl. Acad. Sci. USA 1995, 92, 5880–5884. [Google Scholar] [CrossRef] [PubMed]
- Garavan, H.; Pankiewicz, J.; Bloom, A.; Cho, J.-K.; Sperry, L.; Ross, T.; Salmeron, B.J.; Risinger, R.; Kelley, D.; Stein, E.A. Cue-Induced Cocaine Craving: Neuroanatomical Specificity for Drug Users and Drug Stimuli. Am. J. Psychiatry 2000, 157, 1789–1798. [Google Scholar] [CrossRef]
- Rossetti, Z.L.; Hmaidan, Y.; Gessa, G.L. Marked inhibition of mesolimbic dopamine release: A common feature of ethanol, morphine, cocaine and amphetamine abstinence in rats. Eur. J. Pharmacol. 1992, 221, 227–234. [Google Scholar] [CrossRef]
- Weiss, F.; Hurd, Y.L.; Ungerstedt, U.; Markou, A.; Plotsky, P.M.; Koob, G.F. Neurochemical Correlates of Cocaine and Ethanol Self-Administration. Ann. N. Y. Acad. Sci. 1992, 654, 220–241. [Google Scholar] [CrossRef]
- Weiss, F.; Parsons, L.H.; Schulteis, G.; Hyytiä, P.; Lorang, M.T.; Bloom, F.E.; Koob, G.F. Ethanol Self-Administration Restores Withdrawal-Associated Deficiencies in Accumbal Dopamine and 5-Hydroxytryptamine Release in Dependent Rats. J. Neurosci. 1996, 16, 3474–3485. [Google Scholar] [CrossRef]
- Weiss, F.; Markou, A.; Lorang, M.T.; Koob, G.F. Basal extracellular dopamine levels in the nucleus accumbens are decreased during cocaine withdrawal after unlimited-access self-administration. Brain Res. 1992, 593, 314–318. [Google Scholar] [CrossRef]
- Martinez, D.; Narendran, R.; Foltin, R.W.; Slifstein, M.; Hwang, D.-R.; Broft, A.; Huang, Y.; Cooper, T.B.; Fischman, M.W.; Kleber, H.D.; et al. Amphetamine-Induced Dopamine Release: Markedly Blunted in Cocaine Dependence and Predictive of the Choice to Self-Administer Cocaine. Am. J. Psychiatry 2007, 164, 622–629. [Google Scholar] [CrossRef] [PubMed]
- Volkow, N.D.; Tomasi, D.; Wang, G.J.; Logan, J.; Alexoff, D.; Jayne, M.; Fowler, J.S.; Wong, C.; Yin, P.; Du, C. Stimulant-induced dopamine increases are markedly blunted in active cocaine abusers. Mol. Psychiatry 2014, 19, 1037–1043. [Google Scholar] [CrossRef] [PubMed]
- Volkow, N.D.; Wang, G.-J.; Fowler, J.S.; Logan, J.; Gatley, S.J.; Hitzemann, R.; Chen, A.D.; Dewey, S.L.; Pappas, N. Decreased striatal dopaminergic responsiveness in detoxified cocaine-dependent subjects. Nature 1997, 386, 830–833. [Google Scholar] [CrossRef] [PubMed]
- Melis, M.; Spiga, S.; Diana, M. The Dopamine Hypothesis of Drug Addiction: Hypodopaminergic State. Int. Rev. Neurobiol. 2005, 63, 101–154. [Google Scholar] [CrossRef] [PubMed]
- Williams, S.B.; Yorgason, J.T.; Nelson, A.C.; Lewis, N.; Nufer, T.M.; Edwards, J.G.; Steffensen, S.C. Glutamate Transmission to Ventral Tegmental Area GABA Neurons Is Altered by Acute and Chronic Ethanol. Alcohol. Clin. Exp. Res. 2018, 42, 2186–2195. [Google Scholar] [CrossRef]
- Steffensen, S.C.; Bradley, K.D.; Hansen, D.M.; Wilcox, J.D.; Wilcox, R.S.; Allison, D.W.; Merrill, C.; Edwards, J.G. The role of connexin-36 gap junctions in alcohol intoxication and consumption. Synapse 2010, 65, 695–707. [Google Scholar] [CrossRef] [PubMed]
- Minkowski, C.P.; Epstein, D.; Frost, J.J.; Gorelick, D.A. Differential response to IV carfentanil in chronic cocaine users and healthy controls. Addict. Biol. 2010, 17, 149–155. [Google Scholar] [CrossRef] [PubMed]
- Davidson, M.; Shanley, B.; Wilce, P. Increased NMDA-induced excitability during ethanol withdrawal: A behavioural and histological study. Brain Res. 1995, 674, 91–96. [Google Scholar] [CrossRef]
- Dahchour, A.; De Witte, P.; Bolo, N.; Nédélec, J.-F.; Muzet, M.; Durbin, P.; Macher, J.-P. Central effects of acamprosate: Part 1. Acamprosate blocks the glutamate increase in the nucleus accumbens microdialysate in ethanol withdrawn rats. Psychiatry Res. Neuroimaging 1998, 82, 107–114. [Google Scholar] [CrossRef]
- Delfs, J.M.; Zhu, Y.; Druhan, J.P.; Aston-Jones, G. Noradrenaline in the ventral forebrain is critical for opiate withdrawal-induced aversion. Nature 2000, 403, 430–434. [Google Scholar] [CrossRef]
- Sommer, W.; Rimondini, R.; Hansson, A.C.; Hipskind, P.A.; Gehlert, D.R.; Barr, C.S.; Heilig, M. Upregulation of Voluntary Alcohol Intake, Behavioral Sensitivity to Stress, and Amygdala Crhr1 Expression Following a History of Dependence. Biol. Psychiatry 2008, 63, 139–145. [Google Scholar] [CrossRef] [PubMed]
- Zorrilla, E.P.; Valdez, G.R.; Weiss, F. Changes in levels of regional CRF-like-immunoreactivity and plasma corticosterone during protracted drug withdrawal in dependent rats. Psychopharmacology 2001, 158, 374–381. [Google Scholar] [CrossRef] [PubMed]
- Valdez, G.R.; Sabino, V.; Koob, G.F. Increased Anxiety-Like Behavior and Ethanol Self-Administration in Dependent Rats: Reversal via Corticotropin-Releasing Factor-2 Receptor Activation. Alcohol. Clin. Exp. Res. 2004, 28, 865–872. [Google Scholar] [CrossRef] [PubMed]
- Funk, C.K.; O’Dell, L.E.; Crawford, E.F.; Koob, G.F. Corticotropin-Releasing Factor within the Central Nucleus of the Amygdala Mediates Enhanced Ethanol Self-Administration in Withdrawn, Ethanol-Dependent Rats. J. Neurosci. 2006, 26, 11324–11332. [Google Scholar] [CrossRef] [PubMed]
- Romach, M.K.; Sellers, E. Management of the Alcohol withdrawal Syndrome. Annu. Rev. Med. 1991, 42, 323–340. [Google Scholar] [CrossRef]
- Walker, B.M.; Rasmussen, D.D.; Raskind, M.A.; Koob, G.F. α1-noradrenergic receptor antagonism blocks dependence-induced increases in responding for ethanol. Alcohol 2008, 42, 91–97. [Google Scholar] [CrossRef]
- Hölter, S.M.; Henniger, M.S.; Lipkowski, A.W.; Spanagel, R. Kappa-opioid receptors and relapse-like drinking in long-term ethanol-experienced rats. Psychopharmacology 2000, 153, 93–102. [Google Scholar] [CrossRef]
- Walker, B.M.; Koob, G.F. Pharmacological Evidence for a Motivational Role of κ-Opioid Systems in Ethanol Dependence. Neuropsychopharmacology 2007, 33, 643–652. [Google Scholar] [CrossRef]
- Land, B.B.; Bruchas, M.R.; Lemos, J.; Xu, M.; Melief, E.J.; Chavkin, C. The Dysphoric Component of Stress Is Encoded by Activation of the Dynorphin -Opioid System. J. Neurosci. 2008, 28, 407–414. [Google Scholar] [CrossRef]
- Pandey, S.C. The gene transcription factor cyclic AMP-responsive element binding protein: Role in positive and negative affective states of alcohol addiction. Pharmacol. Ther. 2004, 104, 47–58. [Google Scholar] [CrossRef]
- Economidou, D.; Cippitelli, A.; Stopponi, S.; Braconi, S.; Clementi, S.; Ubaldi, M.; Martin-Fardon, R.; Weiss, F.; Massi, M.; Ciccocioppo, R. Activation of Brain NOP Receptors Attenuates Acute and Protracted Alcohol Withdrawal Symptoms in the Rat. Alcohol. Clin. Exp. Res. 2011, 35, 747–755. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Sidhpura, N.; Parsons, L.H. Endocannabinoid-mediated synaptic plasticity and addiction-related behavior. Neuropharmacology 2011, 61, 1070–1087. [Google Scholar] [CrossRef] [PubMed]
- Roy, A.; Pandey, S.C. The Decreased Cellular Expression of Neuropeptide Y Protein in Rat Brain Structures during Ethanol Withdrawal after Chronic Ethanol Exposure. Alcohol. Clin. Exp. Res. 2002, 26, 796–803. [Google Scholar] [CrossRef] [PubMed]
- Gilpin, N.W.; Misra, K.; Koob, G.F. Neuropeptide Y in the central nucleus of the amygdala suppresses dependence-induced increases in alcohol drinking. Pharmacol. Biochem. Behav. 2008, 90, 475–480. [Google Scholar] [CrossRef]
- Thorsell, A.; Slawecki, C.J.; Ehlers, C.L. Effects of Neuropeptide Y on Appetitive and Consummatory Behaviors Associated With Alcohol Drinking in Wistar Rats With a History of Ethanol Exposure. Alcohol. Clin. Exp. Res. 2005, 29, 584–590. [Google Scholar] [CrossRef]
- Thorsell, A.; Repunte-Canonigo, V.; O’Dell, L.E.; Chen, S.A.; King, A.R.; Lekic, D.; Koob, G.F.; Sanna, P.P. Viral vector-induced amygdala NPY overexpression reverses increased alcohol intake caused by repeated deprivations in Wistar rats. Brain 2007, 130, 1330–1337. [Google Scholar] [CrossRef][Green Version]
- Ciccocioppo, R.; Economidou, D.; Fedeli, A.; Angeletti, S.; Weiss, F.; Heilig, M.; Massi, M. Attenuation of ethanol self-administration and of conditioned reinstatement of alcohol-seeking behaviour by the antiopioid peptide nociceptin/orphanin FQ in alcohol-preferring rats. Psychopharmacology 2003, 172, 170–178. [Google Scholar] [CrossRef]
- Basavarajappa, B.S.; Ninan, I.; Arancio, O. Acute ethanol suppresses glutamatergic neurotransmission through endocannabinoids in hippocampal neurons. J. Neurochem. 2008, 107, 1001–1013. [Google Scholar] [CrossRef]
- Tan, H.; Lauzon, N.M.; Bishop, S.F.; Bechard, M.A.; LaViolette, S.R. Integrated Cannabinoid CB1 Receptor Transmission within the Amygdala-Prefrontal Cortical Pathway Modulates Neuronal Plasticity and Emotional Memory Encoding. Cereb. Cortex 2009, 20, 1486–1496. [Google Scholar] [CrossRef]
- Chiu, C.; Puente, N.; Grandes, P.; Castillo, P.E. Dopaminergic Modulation of Endocannabinoid-Mediated Plasticity at GABAergic Synapses in the Prefrontal Cortex. J. Neurosci. 2010, 30, 7236–7248. [Google Scholar] [CrossRef]
- Azad, S.C.; Monory, K.; Marsicano, G.; Cravatt, B.F.; Lutz, B.; Zieglgänsberger, W.; Rammes, G. Circuitry for Associative Plasticity in the Amygdala Involves Endocannabinoid Signaling. J. Neurosci. 2004, 24, 9953–9961. [Google Scholar] [CrossRef]
- Peters, K.Z.; Oleson, E.B.; Cheer, J.F. A brain on cannabinoids: The role of dopamine release in reward seeking and addiction. Cold Spring Harb. Perspect. Med. 2021, 11, a1039305. [Google Scholar] [CrossRef]
- Volkow, N.D. Addiction, a Disease of Compulsion and Drive: Involvement of the Orbitofrontal Cortex. Cereb. Cortex 2000, 10, 318–325. [Google Scholar] [CrossRef] [PubMed]
- Volkow, N.D.; Wang, G.-J.; Fowler, J.S.; Telang, F. Overlapping neuronal circuits in addiction and obesity: Evidence of systems pathology. Philos. Trans. R. Soc. B Biol. Sci. 2008, 363, 3191–3200. [Google Scholar] [CrossRef] [PubMed]
- Kober, H.; Lacadie, C.M.; Wexler, B.E.; Malison, R.T.; Sinha, R.; Potenza, M.N. Brain Activity during Cocaine Craving and Gambling Urges: An fMRI Study. Neuropsychopharmacology 2015, 41, 628–637. [Google Scholar] [CrossRef] [PubMed]
- Risinger, R.C.; Salmeron, B.J.; Ross, T.; Amen, S.L.; Sanfilipo, M.; Hoffmann, R.G.; Bloom, A.S.; Garavan, H.; Stein, E.A. Neural correlates of high and craving during cocaine self-administration using BOLD fMRI. NeuroImage 2005, 26, 1097–1108. [Google Scholar] [CrossRef] [PubMed]
- Volkow, N.D.; Wang, G.-J.; Ma, Y.; Fowler, J.S.; Wong, C.; Ding, Y.-S.; Hitzemann, R.; Swanson, J.M.; Kalivas, P. Activation of Orbital and Medial Prefrontal Cortex by Methylphenidate in Cocaine-Addicted Subjects But Not in Controls: Relevance to Addiction. J. Neurosci. 2005, 25, 3932–3939. [Google Scholar] [CrossRef]
- Jasinska, A.J.; Stein, E.A.; Kaiser, J.; Naumer, M.J.; Yalachkov, Y. Factors modulating neural reactivity to drug cues in addiction: A survey of human neuroimaging studies. Neurosci. Biobehav. Rev. 2013, 38, 1–16. [Google Scholar] [CrossRef]
- Volkow, N.D.; Wang, G.-J.; Fowler, J.S.; Tomasi, D.; Telang, F. Addiction: Beyond dopamine reward circuitry. Proc. Natl. Acad. Sci. USA 2011, 108, 15037–15042. [Google Scholar] [CrossRef]
- George, O.; Sanders, C.; Freiling, J.; Grigoryan, E.; Vu, S.; Allen, C.D.; Crawford, E.; Mandyam, C.D.; Koob, G.F. Recruitment of medial prefrontal cortex neurons during alcohol withdrawal predicts cognitive impairment and excessive alcohol drinking. Proc. Natl. Acad. Sci. USA 2012, 109, 18156–18161. [Google Scholar] [CrossRef]
- Rando, K.; Hong, K.-I.; Bhagwagar, Z.; Li, C.-S.R.; Bergquist, K.; Guarnaccia, J.; Sinha, R. Association of Frontal and Posterior Cortical Gray Matter Volume with Time to Alcohol Relapse: A Prospective Study. Am. J. Psychiatry 2011, 168, 183–192. [Google Scholar] [CrossRef] [PubMed]
- Faingold, C.; N’Gouemo, P.; Riaz, A. Ethanol and neurotransmitter interactions—From molecular to integrative effects. Prog. Neurobiol. 1998, 55, 509–535. [Google Scholar] [CrossRef]
- Valenzuela, C.F.; Jotty, K. Mini-review: Effects of ethanol on GABAA receptor-mediated neurotransmission in the cere-bellar cortex—Recent advances. Cerebellum 2015, 14, 438–446. [Google Scholar] [CrossRef]
- Valenzuela, C.F.; Puglia, M.P.; Zucca, S. Focus On: Neurotransmitter Systems. Alcohol Res. Health 2011, 34, 106. [Google Scholar] [PubMed]
- Lovinger, D.M.; Roberto, M. Synaptic Effects Induced by Alcohol. In Behavioral Neurobiology of Alcohol Addiction; Springer: Berlin/Heidelberg, Germany, 2010; pp. 31–86. [Google Scholar] [CrossRef]
- Gingrich, J.A.; Hen, R. Dissecting the role of the serotonin system in neuropsychiatric disorders using knockout mice. Psychopharmacology 2001, 155, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Murphy, D.L.; Lerner, A.; Rudnick, G.; Lesch, K.P. Serotonin transporter: Gene, genetic disorders, and pharmacogenetics. Mol. Interv. 2004, 4, 109–123. [Google Scholar] [CrossRef] [PubMed]
- Jun, S.B.; Ikeda, S.R.; Sung, J.E.; Lovinger, D.M. Ethanol induces persistent potentiation of 5-HT3 receptor-stimulated GABA release at synapses on rat hippocampal CA1 neurons. Neuropharmacology 2020, 184, 108415. [Google Scholar] [CrossRef] [PubMed]
- Sung, K.-W.; Engel, S.R.; Allan, A.M.; Lovinger, D.M. 5-HT3 receptor function and potentiation by alcohols in frontal cortex neurons from transgenic mice overexpressing the receptor. Neuropharmacology 2000, 39, 2346–2351. [Google Scholar] [CrossRef]
- Enoch, M.-A.; Gorodetsky, E.; Hodgkinson, C.; Roy, A.; Goldman, D. Functional genetic variants that increase synaptic serotonin and 5-HT3 receptor sensitivity predict alcohol and drug dependence. Mol. Psychiatry 2010, 16, 1139–1146. [Google Scholar] [CrossRef]
- Heinz, A.; Mann, K.; Weinberger, D.R.; Goldman, D. Serotonergic Dysfunction, Negative Mood States, and Response to Alcohol. Alcohol. Clin. Exp. Res. 2001, 25, 487–495. [Google Scholar] [CrossRef]
- McHugh, R.K.; Hofmann, S.G.; Asnaani, A.; Sawyer, A.T.; Otto, M.W. The serotonin transporter gene and risk for alcohol dependence: A meta-analytic review. Drug Alcohol Depend. 2010, 108, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Seneviratne, C.; Huang, W.; Ait-Daoud, N.; Li, M.D.; Johnson, B.A. Characterization of a Functional Polymorphism in the 3′ UTR of SLC6A4 and its Association with Drinking Intensity. Alcohol. Clin. Exp. Res. 2009, 33, 332–339. [Google Scholar] [CrossRef] [PubMed]
- Seneviratne, C.; Franklin, J.; Beckett, K.; Ma, J.Z.; Ait-Daoud, N.; Payne, T.L.; Johnson, B.A.; Li, M.D. Association, interaction, and replication analysis of genes encoding serotonin transporter and 5-HT3 receptor subunits A and B in alcohol dependence. Hum. Genet. 2013, 132, 1165–1176. [Google Scholar] [CrossRef] [PubMed]
- Weiss, F.; Lorang, M.T.; Bloom, F.E.; Koob, G.F. Oral alcohol self-administration stimulates dopamine release in the rat nucleus accumbens: Genetic and motivational determinants. J. Pharmacol. Exp. Ther. 1993, 267, 250–258. [Google Scholar]
- Melendez, R.I.; Rodd-Henricks, Z.A.; Engleman, E.A.; Li, T.-K.; McBride, W.J.; Murphy, J.M. Microdialysis of Dopamine in the Nucleus Accumbens of Alcohol-Preferring (P) Rats during Anticipation and Operant Self-Administration of Ethanol. Alcohol. Clin. Exp. Res. 2002, 26, 318–325. [Google Scholar] [CrossRef]
- Brodie, M.S.; Shefner, S.A.; Dunwiddie, T.V. Ethanol increases the firing rate of dopamine neurons of the rat ventral tegmental area in vitro. Brain Res. 1990, 508, 65–69. [Google Scholar] [CrossRef]
- Franke, B.; Schellekens, A.F.A.; Ellenbroek, B.; Cools, A.; De Jong, C.A.J.; Buitelaar, J.K.; Verkes, R.-J. Reduced Dopamine Receptor Sensitivity as an Intermediate Phenotype in Alcohol Dependence and the Role of the COMT Val158Met and DRD2 Taq1A Genotypes. Arch. Gen. Psychiatry 2012, 69, 339–348. [Google Scholar] [CrossRef]
- Mignini, F.; Napolioni, V.; Codazzo, C.; Carpi, F.M.; Vitali, M.; Romeo, M.; Ceccanti, M. DRD2/ANKK1 TaqIA and SLC6A3 VNTR polymorphisms in alcohol dependence: Association and gene–gene interaction study in a population of Central Italy. Neurosci. Lett. 2012, 522, 103–107. [Google Scholar] [CrossRef]
- Smith, L.; Watson, M.; Gates, S.; Ball, D.; Foxcroft, D. Meta-Analysis of the Association of the Taq1A Polymorphism with the Risk of Alcohol Dependency: A HuGE Gene-Disease Association Review. Am. J. Epidemiology 2007, 167, 125–138. [Google Scholar] [CrossRef]
- Vasconcelos, A.C.C.G.; Neto, E.D.S.R.; Pinto, G.; Yoshioka, F.K.N.; Motta, F.J.N.; Vasconcelos, D.F.P.; Canalle, R. Association Study of theSLC6A3VNTR (DAT) andDRD2/ANKK1Taq1A Polymorphisms with Alcohol Dependence in a Population from Northeastern Brazil. Alcohol. Clin. Exp. Res. 2015, 39, 205–211. [Google Scholar] [CrossRef]
- Xu, M.; Lin, Z. Genetic influences of dopamine transport gene on alcohol dependence: A pooled analysis of 13 studies with 2483 cases and 1753 controls. Prog. Neuro-Psychopharmacology Biol. Psychiatry 2010, 35, 1255–1260. [Google Scholar] [CrossRef] [PubMed]
- Enoch, M.-A.; Waheed, J.F.; Harris, C.R.; Albaugh, B.; Goldman, D. Sex Differences in the Influence of COMT Val158Met on Alcoholism and Smoking in Plains American Indians. Alcohol. Clin. Exp. Res. 2006, 30, 399–406. [Google Scholar] [CrossRef] [PubMed]
- Tiihonen, J.; Hallikainen, T.; Lachman, H.; Saito, T.; Volavka, J.; Kauhanen, J.; Salonen, J.T.; Ryynänen, O.-P.; Koulu, M.; Karvonen, M.K.; et al. Association between the functional variant of the catechol-O-methyltransferase (COMT) gene and type 1 alcoholism. Mol. Psychiatry 1999, 4, 286–289. [Google Scholar] [CrossRef] [PubMed]
- Foroud, T.; Wetherill, L.F.; Dick, D.M.; Hesselbrock, V.; Nurnberger, J.; Kramer, J.; Tischfield, J.; Schuckit, M.; Bierut, L.J.; Xuei, X.; et al. Lack of Association of Alcohol Dependence and Habitual Smoking with Catechol-O-methyltransferase. Alcohol. Clin. Exp. Res. 2007, 31, 1773–1779. [Google Scholar] [CrossRef] [PubMed]
- Pascale, E.; Lucarelli, M. Alcohol Addiction: A Molecular Biology Perspective. Curr. Med. Chem. 2015, 22, 670–684. [Google Scholar] [CrossRef]
- Bergen, A.W.; Kokoszka, J.; Peterson, R.; Long, J.C.; Virkkunen, M.; Linnoila, M.; Goldman, D. μ opioid receptor gene variants: Lack of association with alcohol dependence. Mol. Psychiatry 1997, 2, 490–494. [Google Scholar] [CrossRef]
- Zhang, Y.; Wang, D.; Johnson, A.D.; Papp, A.C.; Sadée, W. Allelic Expression Imbalance of Human mu Opioid Receptor (OPRM1) Caused by Variant A118G. J. Biol. Chem. 2005, 280, 32618–32624. [Google Scholar] [CrossRef]
- Ramchandani, V.A.; Umhau, J.; Pavon, F.J.; Ruiz-Velasco, V.; Margas, W.; Sun, H.; Damadzic, R.; Eskay, R.; Schoor, M.; Thorsell, A.; et al. A genetic determinant of the striatal dopamine response to alcohol in men. Mol. Psychiatry 2010, 16, 809–817. [Google Scholar] [CrossRef]
- Kranzler, H.R.; Gelernter, J.; O’Malley, S.; Hernandez-Avila, C.A.; Kaufman, D. Association of alcohol or other drug dependence with alleles of the mu opioid receptor gene (OPRM1). Alcohol. Clin. Exp. Res. 1998, 22, 1359–1362. [Google Scholar] [CrossRef]
- Arias, A.; Feinn, R.; Kranzler, H.R. Association of an Asn40Asp (A118G) polymorphism in the μ-opioid receptor gene with substance dependence: A meta-analysis. Drug Alcohol Depend. 2006, 83, 262–268. [Google Scholar] [CrossRef]
- Rouvinen-Lagerström, N.; Lahti, J.; Alho, H.; Kovanen, L.; Aalto, M.; Partonen, T.; Silander, K.; Sinclair, D.; Räikkönen, K.; Eriksson, J.G.; et al. µ-Opioid Receptor Gene (OPRM1) Polymorphism A118G: Lack of Association in Finnish Populations with Alcohol Dependence or Alcohol Consumption. Alcohol Alcohol. 2013, 48, 519–525. [Google Scholar] [CrossRef] [PubMed]
- Ashenhurst, J.R.; Bujarski, S.; Ray, L.A. Delta and kappa opioid receptor polymorphisms influence the effects of naltrexone on subjective responses to alcohol. Pharmacol. Biochem. Behav. 2012, 103, 253–259. [Google Scholar] [CrossRef] [PubMed]
- Poznanski, P.; Lesniak, A.; Korostynski, M.; Szklarczyk, K.; Lazarczyk, M.; Religa, P.; Bujalska-Zadrozny, M.; Sadowski, B.; Sacharczuk, M. Delta-opioid receptor antagonism leads to excessive ethanol consumption in mice with enhanced activity of the endogenous opioid system. Neuropharmacology 2017, 118, 90–101. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.C.; Hinrichs, A.L.; Stock, H.; Budde, J.; Allen, R.; Bertelsen, S.; Kwon, J.M.; Wu, W.; Dick, D.M.; Rice, J.; et al. Evidence of common and specific genetic effects: Association of the muscarinic acetylcholine receptor M2 (CHRM2) gene with alcohol dependence and major depressive syndrome. Hum. Mol. Genet. 2004, 13, 1903–1911. [Google Scholar] [CrossRef] [PubMed]
- Luo, X.; Kranzler, H.R.; Zuo, L.; Wang, S.; Blumberg, H.; Gelernter, J. CHRM2 gene predisposes to alcohol dependence, drug dependence and affective disorders: Results from an extended case–control structured association study. Hum. Mol. Genet. 2005, 14, 2421–2434. [Google Scholar] [CrossRef]
- Sherva, R.; Kranzler, H.R.; Yu, Y.; Logue, M.W.; Poling, J.; Arias, A.J.; Anton, R.F.; Oslin, D.; Farrer, L.A.; Gelernter, J. Variation in Nicotinic Acetylcholine Receptor Genes is Associated with Multiple Substance Dependence Phenotypes. Neuropsychopharmacology 2010, 35, 1921–1931. [Google Scholar] [CrossRef]
- Wolfe, S. The Synaptic Interactions of Alcohol and the Endogenous Cannabinoid System. Alcohol Res. Curr. Rev. 2022, 42, 3. [Google Scholar] [CrossRef]
- Rubio, M.; De Miguel, R.; Fernández-Ruiz, J.; Gutiérrez-López, M.D.; Carai, M.A.; Ramos, J.A. Effects of a short-term exposure to alcohol in rats on FAAH enzyme and CB1 receptor in different brain areas. Drug Alcohol Depend. 2009, 99, 354–358. [Google Scholar] [CrossRef]
- Rubio, M.; McHugh, D.; Fernández-Ruiz, J.; Bradshaw, H.; Walker, J.M. Short-term exposure to alcohol in rats affects brain levels of anandamide, other N-acylethanolamines and 2-arachidonoyl-glycerol. Neurosci. Lett. 2007, 421, 270–274. [Google Scholar] [CrossRef][Green Version]
- Ferrer, B.; Bermúdez-Silva, F.J.; Bilbao, A.; Alvarez-Jaimes, L.; Sanchez-Vera, I.; Giuffrida, A.; Serrano, A.; Baixeras, E.; Khaturia, S.; Navarro, M.; et al. Regulation of brain anandamide by acute administration of ethanol. Biochem. J. 2007, 404, 97–104. [Google Scholar] [CrossRef]
- Vinod, K.; Yalamanchili, R.; Xie, S.; Cooper, T.; Hungund, B. Effect of chronic ethanol exposure and its withdrawal on the endocannabinoid system. Neurochem. Int. 2006, 49, 619–625. [Google Scholar] [CrossRef] [PubMed]
- Ceccarini, J.; Casteels, C.; Koole, M.; Bormans, G.; Van Laere, K. Transient changes in the endocannabinoid system after acute and chronic ethanol exposure and abstinence in the rat: A combined PET and microdialysis study. Eur. J. Pediatr. 2013, 40, 1582–1594. [Google Scholar] [CrossRef] [PubMed]
- Ortiz, S.; Oliva, J.M.; Rial, S.P.; Palomo, T.; Manzanares, J. Chronic ethanol consumption regulates cannabinoid CB1 receptor gene expression in selected regions of rat brain. Alcohol Alcohol. 2004, 39, 88–92. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Cippitelli, A.; Bilbao, A.; Hansson, A.C.; Del Arco, I.; Sommer, W.; Heilig, M.; Massi, M.; Bermúdez-Silva, F.J.; Navarro, M.; Ciccocioppo, R.; et al. Cannabinoid CB1 receptor antagonism reduces conditioned reinstatement of ethanol-seeking behavior in rats. Eur. J. Neurosci. 2005, 21, 2243–2251. [Google Scholar] [CrossRef]
- Devaud, L.L.; Fritschy, J.-M.; Sieghart, W.; Morrow, A.L. Bidirectional Alterations of GABAA Receptor Subunit Peptide Levels in Rat Cortex during Chronic Ethanol Consumption and Withdrawal. J. Neurochem. 2002, 69, 126–130. [Google Scholar] [CrossRef]
- Mhatre, M.C.; Pena, G.; Sieghart, W.; Ticku, M.K. Antibodies Specific for GABAA Receptor alpha Subunits Reveal that Chronic Alcohol Treatment Down-Regulates alpha-Subunit Expression in Rat Brain Regions. J. Neurochem. 1993, 61, 1620–1625. [Google Scholar] [CrossRef]
- Enoch, M.-A. The role of GABAA receptors in the development of alcoholism. Pharmacol. Biochem. Behav. 2008, 90, 95–104. [Google Scholar] [CrossRef]
- Hodge, C.W.; Mehmert, K.K.; Kelley, S.P.; McMahon, T.; Haywood, A.; Olive, F.; Wang, D.; Sanchez-Perez, A.M.; Messing, R. Supersensitivity to allosteric GABAA receptor modulators and alcohol in mice lacking PKCε. Nat. Neurosci. 1999, 2, 997–1002. [Google Scholar] [CrossRef]
- Qi, Z.-H.; Song, M.; Wallace, M.; Wang, D.; Newton, P.; McMahon, T.; Chou, W.-H.; Zhang, C.; Shokat, K.M.; Messing, R. Protein Kinase C epsilon Regulates gamma-Aminobutyrate Type A Receptor Sensitivity to Ethanol and Benzodiazepines through Phosphorylation of gamma2 Subunits. J. Biol. Chem. 2007, 282, 33052–33063. [Google Scholar] [CrossRef]
- Maiya, R.; McMahon, T.; Wang, D.; Kanter, B.; Gandhi, D.; Chapman, H.L.; Miller, J.; Messing, R.O. Selective chemical genetic inhibition of protein kinase C epsilon reduces ethanol consumption in mice. Neuropharmacology 2016, 107, 40–48. [Google Scholar] [CrossRef]
- Besheer, J.; Lepoutre, V.; Mole, B.; Hodge, C.W. GABAA receptor regulation of voluntary ethanol drinking requires PKCε. Synapse 2006, 60, 411–419. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Burkhardt, J.M.; Adermark, L. Locus of onset and subpopulation specificity of in vivo ethanol effect in the reciprocal ventral tegmental area–nucleus accumbens circuit. Neurochem. Int. 2014, 76, 122–130. [Google Scholar] [CrossRef] [PubMed]
- Diaz, M.R.; Valenzuela, C.F. Sensitivity of GABAergic Tonic Currents to Acute Ethanol in Cerebellar Granule Neurons is Not Age- or δ Subunit-Dependent in Developing Rats. Alcohol. Clin. Exp. Res. 2016, 40, 83–92. [Google Scholar] [CrossRef]
- Liang, J.; Zhang, N.; Cagetti, E.; Houser, C.R.; Olsen, R.W.; Spigelman, I. Chronic Intermittent Ethanol-Induced Switch of Ethanol Actions from Extrasynaptic to Synaptic Hippocampal GABAA Receptors. J. Neurosci. 2006, 26, 1749–1758. [Google Scholar] [CrossRef] [PubMed]
- Jia, F.; Chandra, D.; Homanics, G.E.; Harrison, N.L. Ethanol Modulates Synaptic and Extrasynaptic GABAA Receptors in the Thalamus. J. Pharmacol. Exp. Ther. 2008, 326, 475–482. [Google Scholar] [CrossRef] [PubMed]
- Edenberg, H.J.; Dick, D.M.; Xuei, X.; Tian, H.; Almasy, L.; Bauer, L.O.; Crowe, R.R.; Goate, A.; Hesselbrock, V.; Jones, K.; et al. Variations in GABRA2, Encoding the α2 Subunit of the GABAA Receptor, Are Associated with Alcohol Dependence and with Brain Oscillations. Am. J. Hum. Genet. 2004, 74, 705–714. [Google Scholar] [CrossRef] [PubMed]
- Agrawal, A.; Edenberg, H.J.; Foroud, T.; Bierut, L.J.; Dunne, G.; Hinrichs, A.L.; Nurnberger, J.I.; Crowe, R.; Kuperman, S.; Schuckit, M.A.; et al. Association of GABRA2 with Drug Dependence in the Collaborative Study of the Genetics of Alcoholism Sample. Behav. Genet. 2006, 36, 640–650. [Google Scholar] [CrossRef]
- Covault, J.; Gelernter, J.; Hesselbrock, V.; Nellissery, M.; Kranzler, H.R. Allelic and haplotypic association ofGABRA2 with alcohol dependence. Am. J. Med Genet. 2004, 129B, 104–109. [Google Scholar] [CrossRef]
- Fehr, C.; Sander, T.; Tadic, A.; Lenzen, K.P.; Anghelescu, I.; Klawe, C.; Dahmen, N.; Schmidt, L.G.; Szegedi, A. Confirmation of association of the GABRA2 gene with alcohol dependence by subtype-specific analysis. Psychiatr. Genet. 2006, 16, 9–17. [Google Scholar] [CrossRef]
- Roberto, M.; Varodayan, F.P. Synaptic targets: Chronic alcohol actions. Neuropharmacology 2017, 122, 85–99. [Google Scholar] [CrossRef]
- Cozzoli, D.K.; Goulding, S.P.; Zhang, P.W.; Xiao, B.; Hu, J.-H.; Ary, A.W.; Obara, I.; Rahn, A.; Abou-Ziab, H.; Tyrrel, B.; et al. Binge Drinking Upregulates Accumbens mGluR5-Homer2-PI3K Signaling: Functional Implications for Alcoholism. J. Neurosci. 2009, 29, 8655–8668. [Google Scholar] [CrossRef] [PubMed]
- Schroeder, J.P.; Spanos, M.; Stevenson, J.R.; Besheer, J.; Salling, M.; Hodge, C.W. Cue-induced reinstatement of alcohol-seeking behavior is associated with increased ERK1/2 phosphorylation in specific limbic brain regions: Blockade by the mGluR5 antagonist MPEP. Neuropharmacology 2008, 55, 546–554. [Google Scholar] [CrossRef] [PubMed]
- Ding, Z.-M.; Ingraham, C.M.; Rodd, Z.A.; McBride, W.J. Alcohol drinking increases the dopamine-stimulating effects of ethanol and reduces D2 auto-receptor and group II metabotropic glutamate receptor function within the posterior ventral tegmental area of alcohol preferring (P) rats. Neuropharmacology 2016, 109, 41–48. [Google Scholar] [CrossRef] [PubMed]
- Dildy, J.E.; Leslie, S.W. Ethanol inhibits NMDA-induced increases in free intracellular Ca2+ in dissociated brain cells. Brain Res. 1989, 499, 383–387. [Google Scholar] [CrossRef]
- Hoffman, P.L.; Rabe, C.S.; Moses, F.; Tabakoff, B. N-Methyl-D-Aspartate Receptors and Ethanol: Inhibition of Calcium Flux and Cyclic GMP Production. J. Neurochem. 1989, 52, 1937–1940. [Google Scholar] [CrossRef] [PubMed]
- Lovinger, D.M.; White, G.; Weight, F.F. Ethanol Inhibits NMDA-Activated Ion Current in Hippocampal Neurons. Science 1989, 243, 1721–1724. [Google Scholar] [CrossRef]
- Läck, A.K.; Diaz, M.; Chappell, A.; DuBois, D.W.; McCool, B.A. Chronic Ethanol and Withdrawal Differentially Modulate Pre- and Postsynaptic Function at Glutamatergic Synapses in Rat Basolateral Amygdala. J. Neurophysiol. 2007, 98, 3185–3196. [Google Scholar] [CrossRef]
- Jin, C.; Smothers, C.T.; Woodward, J.J. Enhanced Ethanol Inhibition of Recombinant N-methyl-D-aspartate Receptors by Magnesium: Role of NR3A Subunits. Alcohol. Clin. Exp. Res. 2008, 32, 1059–1066. [Google Scholar] [CrossRef]
- Schumann, G.; Johann, M.; Frank, J.; Preuss, U.; Dahmen, N.; Laucht, M.; Rietschel, M.; Rujescu, D.; Lourdusamy, A.; Clarke, T.-K.; et al. Systematic Analysis of Glutamatergic Neurotransmission Genes in Alcohol Dependence and Adolescent Risky Drinking Behavior. Arch. Gen. Psychiatry 2008, 65, 826–838. [Google Scholar] [CrossRef]
- Karpyak, V.M.; Geske, J.R.; Colby, C.L.; Mrazek, D.A.; Biernacka, J.M. Genetic variability in the NMDA-dependent AMPA trafficking cascade is associated with alcohol dependence. Addict. Biol. 2011, 17, 798–806. [Google Scholar] [CrossRef]
- Abrahao, K.P.; Ariwodola, O.J.; Butler, T.R.; Rau, A.R.; Skelly, M.J.; Carter, E.; Alexander, N.P.; McCool, B.A.; Souza-Formigoni, M.L.O.; Weiner, J.L. Locomotor sensitization to ethanol impairs NMDA receptor-dependent synaptic plasticity in the nucleus accumbens and increases ethanol self-administration. J. Neurosci. 2013, 33, 4834–4842. [Google Scholar] [CrossRef] [PubMed]
- Läck, A.; Christian, D.; Diaz, M.; McCool, B. Chronic ethanol and withdrawal effects on kainate receptor–mediated excitatory neurotransmission in the rat basolateral amygdala. Alcohol 2009, 43, 25–33. [Google Scholar] [CrossRef][Green Version]
- Bell, R.L.; Hauser, S.R.; McClintick, J.; Rahman, S.; Edenberg, H.J.; Szumlinski, K.K.; McBride, W.J. Ethanol-Associated Changes in Glutamate Reward Neurocircuitry: A Minireview of Clinical and Preclinical Genetic Findings. Prog. Mol. Biol. Transl. Sci. 2015, 137, 41–85. [Google Scholar] [CrossRef] [PubMed]
- Zorumski, C.F.; Mennerick, S.; Izumi, Y. Acute and chronic effects of ethanol on learning-related synaptic plasticity. Alcohol 2013, 48, 1–17. [Google Scholar] [CrossRef]
- McCool, B.A. Ethanol modulation of synaptic plasticity. Neuropharmacology 2011, 61, 1097–1108. [Google Scholar] [CrossRef] [PubMed]
- Fontaine, C.J.; Patten, A.R.; Sickmann, H.M.; Helfer, J.L.; Christie, B.R. Effects of pre-natal alcohol exposure on hippocampal synaptic plasticity: Sex, age and methodological considerations. Neurosci. Biobehav. Rev. 2016, 64, 12–34. [Google Scholar] [CrossRef] [PubMed]
- Abrahao, K.; Salinas, A.; Lovinger, D.M. Alcohol and the Brain: Neuronal Molecular Targets, Synapses, and Circuits. Neuron 2017, 96, 1223–1238. [Google Scholar] [CrossRef]
- Mulholland, P.J.; Hopf, F.W.; Bukiya, A.N.; Martin, G.E.; Liu, J.; Dopico, A.M.; Bonci, A.; Treistman, S.N.; Chandler, L.J. Sizing up Ethanol-Induced Plasticity: The Role of Small and Large Conductance Calcium-Activated Potassium Channels. Alcohol. Clin. Exp. Res. 2009, 33, 1125–1135. [Google Scholar] [CrossRef]
- Mulholland, P.J.; Becker, H.C.; Woodward, J.J.; Chandler, L.J. Small Conductance Calcium-Activated Potassium Type 2 Channels Regulate Alcohol-Associated Plasticity of Glutamatergic Synapses. Biol. Psychiatry 2011, 69, 625–632. [Google Scholar] [CrossRef]
- Nimitvilai, S.; Lopez, M.F.; Mulholland, P.J.; Woodward, J.J. Chronic Intermittent Ethanol Exposure Enhances the Excitability and Synaptic Plasticity of Lateral Orbitofrontal Cortex Neurons and Induces a Tolerance to the Acute Inhibitory Actions of Ethanol. Neuropsychopharmacology 2015, 41, 1112–1127. [Google Scholar] [CrossRef]
- Kuniishi, H.; Ichisaka, S.; Matsuda, S.; Futora, E.; Harada, R.; Hata, Y. Chronic inactivation of the orbitofrontal cortex increases anxiety-like behavior and impulsive aggression, but decreases depression-like behavior in rats. Front. Behav. Neurosci. 2017, 10, 250. [Google Scholar] [CrossRef] [PubMed]
- Dopico, A.M.; Bukiya, A.N.; Martin, G.E. Ethanol modulation of mammalian BK channels in excitable tissues: Molecular targets and their possible contribution to alcohol-induced altered behavior. Front. Physiol. 2014, 5, 466. [Google Scholar] [CrossRef] [PubMed]
- Bodhinathan, K.; Slesinger, P.A. Molecular mechanism underlying ethanol activation of G-protein–gated inwardly rectifying potassium channels. Proc. Natl. Acad. Sci. USA 2013, 110, 18309–18314. [Google Scholar] [CrossRef] [PubMed]
- Lüscher, C.; Slesinger, P.A. Emerging roles for G protein-gated inwardly rectifying potassium (GIRK) channels in health and disease. Nat. Rev. Neurosci. 2010, 11, 301–315. [Google Scholar] [CrossRef]
- Montesinos, J.; Pascual, M.; Millán-Esteban, D.; Guerri, C. Binge-like ethanol treatment in adolescence impairs autophagy and hinders synaptic maturation: Role of TLR4. Neurosci. Lett. 2018, 682, 85–91. [Google Scholar] [CrossRef]
- Jury, N.J.; Pollack, G.A.; Ward, M.J.; Bezek, J.L.; Ng, A.J.; Pinard, C.R.; Bergstrom, H.; Holmes, A. Chronic Ethanol during Adolescence Impacts Corticolimbic Dendritic Spines and Behavior. Alcohol. Clin. Exp. Res. 2017, 41, 1298–1308. [Google Scholar] [CrossRef]
- Lee, K.; Dunwiddie, T.; Deitrich, R.; Lynch, G.; Hoffer, B. Chronic ethanol consumption and hippocampal neuron dendritic spines: A morphometric and physiological analysis. Exp. Neurol. 1981, 71, 541–549. [Google Scholar] [CrossRef]
- Romero, A.M.; Renau-Piqueras, J.; Marin, M.P.; Timoneda, J.; Berciano, M.T.; Lafarga, M.; Esteban-Pretel, G. Chronic Alcohol Alters Dendritic Spine Development in Neurons in Primary Culture. Neurotox. Res. 2013, 24, 532–548. [Google Scholar] [CrossRef]
- Inomata, K.; Nasu, F.; Tanaka, H. Decreased density of synaptic formation in the frontal cortex of neonatal rats exposed to ethanol in utero. Int. J. Dev. Neurosci. 1987, 5, 455–457. [Google Scholar] [CrossRef]
- Uys, J.D.; McGuier, N.S.; Gass, J.T.; Griffin, W.C.; Ball, L.; Mulholland, P.J. Chronic intermittent ethanol exposure and withdrawal leads to adaptations in nucleus accumbens core postsynaptic density proteome and dendritic spines. Addict. Biol. 2015, 21, 560–574. [Google Scholar] [CrossRef]
- Chandler, L.J.; Carpenter-Hyland, E.; Hendricson, A.W.; Maldve, R.E.; Morrisett, R.A.; Zhou, F.C.; Sari, Y.; Bell, R.; Szumlinski, K.K. Structural and Functional Modifications in Glutamateric Synapses Following Prolonged Ethanol Exposure. Alcohol. Clin. Exp. Res. 2006, 30, 368–376. [Google Scholar] [CrossRef] [PubMed]
- Sharma, R.; Engemann, S.C.; Sahota, P.; Thakkar, M.M. Effects of Ethanol on Extracellular Levels of Adenosine in the Basal Forebrain: An In Vivo Microdialysis Study in Freely Behaving Rats. Alcohol. Clin. Exp. Res. 2010, 34, 813–818. [Google Scholar] [CrossRef] [PubMed]
- Hughes, V.; Richardson, M.J.E.; Wall, M.J. Acute ethanol exposure has bidirectional actions on the endogenous neuromodulator adenosine in rat hippocampus. J. Cereb. Blood Flow Metab. 2018, 175, 1471–1485. [Google Scholar] [CrossRef] [PubMed]
- Pany, S.; Das, J. Alcohol binding in the C1 (C1A+C1B) domain of protein kinase C epsilon. Biochim. Biophys. Acta (BBA)—Gen. Subj. 2015, 1850, 2368–2376. [Google Scholar] [CrossRef]
- Ron, D.; Barak, S. Molecular mechanisms underlying alcohol-drinking behaviours. Nat. Rev. Neurosci. 2016, 17, 576–591. [Google Scholar] [CrossRef]
- Yoshimura, M.; Pearson, S.; Kadota, Y.; Gonzalez, C.E. Identification of Ethanol Responsive Domains of Adenylyl Cyclase. Alcohol. Clin. Exp. Res. 2006, 30, 1824–1832. [Google Scholar] [CrossRef]
- Mayfield, R.D.; Harris, R.A.; Schuckit, M.A. Genetic factors influencing alcohol dependence. J. Cereb. Blood Flow Metab. 2008, 154, 275–287. [Google Scholar] [CrossRef]
- Edenberg, H.J. The Genetics of Alcohol Metabolism: Role of Alcohol Dehydrogenase and Aldehyde Dehydrogenase Variants. Alcohol Res. Health 2007, 30, 5–13. [Google Scholar]
- Ji, Y.B.; Tae, K.; Ahn, T.H.; Lee, S.H.; Kim, K.R.; Park, C.W.; Park, B.L.; Shin, H.D. ADH1B and ALDH2 polymorphisms and their associations with increased risk of squamous cell carcinoma of the head and neck in the Korean population. Oral Oncol. 2011, 47, 583–587. [Google Scholar] [CrossRef]
- Kim, J.-I.; Ganesan, S.; Luo, S.X.; Wu, Y.-W.; Park, E.J.; Huang, E.J.; Chen, L.; Ding, J.B. Aldehyde dehydrogenase 1a1 mediates a GABA synthesis pathway in midbrain dopaminergic neurons. Science 2015, 350, 102–106. [Google Scholar] [CrossRef]
- Blomeyer, D.; Treutlein, J.; Esser, G.; Schmidt, M.H.; Schumann, G.; Laucht, M. Interaction between CRHR1 Gene and Stressful Life Events Predicts Adolescent Heavy Alcohol Use. Biol. Psychiatry 2008, 63, 146–151. [Google Scholar] [CrossRef] [PubMed]
- Treutlein, J.; Kissling, C.; Frank, J.; Wiemann, S.; Dong, L.; Depner, M.; Saam, C.; Lascorz, J.; Soyka, M.; Preuss, U.W.; et al. Genetic association of the human corticotropin releasing hormone receptor 1 (CRHR1) with binge drinking and alcohol intake patterns in two independent samples. Mol. Psychiatry 2006, 11, 594–602. [Google Scholar] [CrossRef]
- Tidey, J.W.; Monti, P.M.; Rohsenow, D.J.; Gwaltney, C.J.; Miranda, R., Jr.; McGeary, J.E.; MacKillop, J.; Swift, R.M.; Abrams, D.B.; Shiffman, S.; et al. Moderators of Naltrexone’s Effects on Drinking, Urge, and Alcohol Effects in Non-Treatment-Seeking Heavy Drinkers in the Natural Environment. Alcohol. Clin. Exp. Res. 2007, 32, 58–66. [Google Scholar] [CrossRef] [PubMed]
- Anton, R.F.; Oroszi, G.; Omalley, S.S.; Couper, D.; Swift, R.M.; Pettinati, H.M.; Goldman, D. An Evaluation of μ-Opioid Receptor (OPRM1) as a Predictor of Naltrexone Response in the Treatment of Alcohol Dependence. Arch. Gen. Psychiatry 2008, 65, 135–144. [Google Scholar] [CrossRef] [PubMed]
- Chamorro, A.-J.; Marcos, M.; Mirón-Canelo, J.-A.; Pastor, I.; González-Sarmiento, R.; Laso, F.-J. Association of µ-opioid receptor (OPRM1) gene polymorphism with response to naltrexone in alcohol dependence: A systematic review and meta-analysis. Addict. Biol. 2012, 17, 505–512. [Google Scholar] [CrossRef] [PubMed]
- Oslin, D.W.; Berrettini, W.; Kranzler, H.R.; Pettinati, H.; Gelernter, J.; Volpicelli, J.R.; O’Brien, C.P. A Functional Polymorphism of the μ-Opioid Receptor Gene is Associated with Naltrexone Response in Alcohol-Dependent Patients. Neuropsychopharmacology 2003, 28, 1546–1552. [Google Scholar] [CrossRef] [PubMed]
- Wagner, F.A.; Anthony, J.C. From first drug use to drug dependence: Developmental periods of risk for dependence upon marijuana, cocaine, and alcohol. Prim. Care Companion J. Clin. Psychiatry 2002, 4, 33. [Google Scholar] [CrossRef]
- Spear, L.P. Adolescent Neurodevelopment. J. Adolesc. Health 2013, 52, S7–S13. [Google Scholar] [CrossRef]
- Grant, B.F.; Stinson, F.S.; Harford, T.C. Age at onset of alcohol use and DSM-IV alcohol abuse and dependence: A 12-year follow-up. J. Subst. Abus. 2001, 13, 493–504. [Google Scholar] [CrossRef]
- Volkow, N.D. Altered pathways: Drug abuse and age of onset. Addict. Prof. 2006, 26, 29. [Google Scholar]
- Hingson, R.W.; Heeren, T.; Winter, M.R. Age of Alcohol-Dependence Onset: Associations with Severity of Dependence and Seeking Treatment. Pediatrics 2006, 118, e755–e763. [Google Scholar] [CrossRef] [PubMed]
- King, K.M.; Chassin, L. A Prospective Study of the Effects of Age of Initiation of Alcohol and Drug Use on Young Adult Substance Dependence. J. Stud. Alcohol Drugs 2007, 68, 256–265. [Google Scholar] [CrossRef] [PubMed]
- Hanson, K.L.; Medina, K.L.; Padula, C.B.; Tapert, S.F.; Brown, S.A. Impact of Adolescent Alcohol and Drug Use on Neuropsychological Functioning in Young Adulthood: 10-Year Outcomes. J. Child Adolesc. Subst. Abus. 2011, 20, 135–154. [Google Scholar] [CrossRef] [PubMed]
- Squeglia, L.M.; Spadoni, A.D.; Infante, M.A.; Myers, M.G.; Tapert, S.F. Initiating moderate to heavy alcohol use predicts changes in neuropsychological functioning for adolescent girls and boys. Psychol. Addict. Behav. 2009, 23, 715–722. [Google Scholar] [CrossRef]
- King, M.; Lipsky, M.S. Clinical implications of aging. Dis.-A-Mon. 2015, 61, 467–474. [Google Scholar] [CrossRef]
- Ryan, M.; Merrick, E.L.; Hodgkin, D.; Horgan, C.M.; Garnick, D.W.; Panas, L.; Ritter, G.; Blow, F.C.; Saitz, R. Drinking Patterns of Older Adults with Chronic Medical Conditions. J. Gen. Intern. Med. 2013, 28, 1326–1332. [Google Scholar] [CrossRef]
- Qato, D.M.; Manzoor, B.S.; Lee, T.A. Drug-Alcohol Interactions in Older U.S. Adults. J. Am. Geriatr. Soc. 2015, 63, 2324–2331. [Google Scholar] [CrossRef]
- Moore, A.A.; Whiteman, E.J.; Ward, K.T. Risks of combined alcohol/medication use in older adults. Am. J. Geriatr. Pharmacother. 2007, 5, 64–74. [Google Scholar] [CrossRef]
- Hawkins, J.D.; Catalano, R.F.; Miller, J.Y. Risk and protective factors for alcohol and other drug problems in adolescence and early adulthood: Implications for substance abuse prevention. Psychol. Bull. 1992, 112, 64–105. [Google Scholar] [CrossRef]
- Sloboda, Z.; Glantz, M.D.; Tarter, R.E. Revisiting the Concepts of Risk and Protective Factors for Understanding the Etiology and Development of Substance Use and Substance Use Disorders: Implications for Prevention. Subst. Use Misuse 2012, 47, 944–962. [Google Scholar] [CrossRef]
- Grant, B.F.; Goldstein, R.B.; Saha, T.D.; Chou, S.P.; Jung, J.; Zhang, H.; Pickering, R.P.; Ruan, W.J.; Smith, S.M.; Huang, B.; et al. Epidemiology of DSM-5 alcohol use disorder results from the national epidemiologic survey on alcohol and related conditions III. JAMA Psychiatry 2015, 72, 757–766. [Google Scholar] [CrossRef] [PubMed]
- Kohn, R.; Saxena, S.; Levav, I.; Saraceno, B. The treatment gap in mental health care. Bull. World Health Organ 2004, 82, 858–866. [Google Scholar] [PubMed]
- Kranzler, H.R.; Soyka, M. Diagnosis and Pharmacotherapy of Alcohol Use Disorder. JAMA 2018, 320, 815–824. [Google Scholar] [CrossRef] [PubMed]
- Park-Lee, E.; Lipari, R.N.; Hedden, S.L.; Kroutil, L.A.; Porter, J.D. Receipt of Services for Substance Use and Mental Health Issues among Adults: Results from the 2016 National Survey on Drug Use and Health. In CBHSQ Data Review; Substance Abuse and Mental Health Services Administration: Rockville, MD, USA, 2012. [Google Scholar]
- Carvalho, A.F.; Heilig, M.; Perez, A.; Probst, C.; Rehm, J. Alcohol use disorders. Lancet 2019, 394, 781–792. [Google Scholar] [CrossRef]
- McCrady, B.S. Treatment Interventions for Women with Alcohol Use Disorder. Alcohol Res. Curr. Rev. 2020, 40, 2020. [Google Scholar] [CrossRef]
- Hasin, D.S.; Grant, B.F. Major Depression in 6050 Former Drinkers. Arch. Gen. Psychiatry 2002, 59, 794–800. [Google Scholar] [CrossRef]
- Smedslund, G.; Berg, R.C.; Hammerstrøm, K.T.; Steiro, A.; Leiknes, K.A.; Dahl, H.M.; Karlsen, K. Motivational interviewing for substance abuse. Campbell Syst. Rev. 2011, 7, 1–126. [Google Scholar]
- Weiss, R.D.; O’Malley, S.S.; Hosking, J.D.; LoCastro, J.S.; Swift, R.; for the COMBINE Study Research Group. Do Patients with Alcohol Dependence Respond to Placebo? Results from the COMBINE Study. J. Stud. Alcohol Drugs 2008, 69, 878–884. [Google Scholar] [CrossRef]
- Bien, T.H.; Miller, W.R.; Tonigan, J.S. Brief interventions for alcohol problems: A review. Addiction 1993, 88, 315–336. [Google Scholar] [CrossRef]
- McHugh, R.K.; Hearon, B.A.; Otto, M. Cognitive Behavioral Therapy for Substance Use Disorders. Psychiatr. Clin. N. Am. 2010, 33, 511–525. [Google Scholar] [CrossRef]
- Dutra, L.; Stathopoulou, G.; Basden, S.L.; Leyro, T.M.; Powers, M.; Otto, M.W. A Meta-Analytic Review of Psychosocial Interventions for Substance Use Disorders. Am. J. Psychiatry 2008, 165, 179–187. [Google Scholar] [CrossRef] [PubMed]
- Kelly, J.F.; Humphreys, K.; Ferri, M. Alcoholics Anonymous and other 12-step programs for alcohol use disorder. Cochrane Database Syst. Rev. 2020, 2020, CD012880. [Google Scholar] [CrossRef]
- Zindel, L.R.; Kranzler, H.R. Pharmacotherapy of Alcohol Use Disorders: Seventy-Five Years of Progress. J. Stud. Alcohol Drugs Suppl. 2014, 75, 79–88. [Google Scholar] [CrossRef] [PubMed]
- Ambrogne, J.A. Reduced-risk drinking as a treatment goal: What clinicians need to know. J. Subst. Abus. Treat. 2002, 22, 45–53. [Google Scholar] [CrossRef]
- Soravia, L.M.; Schläfli, K.; Stutz, S.; Rösner, S.; Moggi, F. Resistance to Temptation: The Interaction of External and Internal Control on Alcohol Use during Residential Treatment for Alcohol Use Disorder. Alcohol. Clin. Exp. Res. 2015, 39, 2209–2214. [Google Scholar] [CrossRef]
- Gossop, M.; Stewart, D.; Marsden, J. Attendance at Narcotics Anonymous and Alcoholics Anonymous meetings, frequency of attendance and substance use outcomes after residential treatment for drug dependence: A 5-year follow-up study. Addiction 2007, 103, 119–125. [Google Scholar] [CrossRef] [PubMed]
- Lieber, C.S. Alcohol-nutrition interaction: 1984 update. Alcohol 1984, 1, 151–157. [Google Scholar] [CrossRef]
- Lieber, C.S. Role of Oxidative Stress and Antioxidant Therapy in Alcoholic and Nonalcoholic Liver Diseases. Adv. Pharmacol. 1996, 38, 601–628. [Google Scholar] [CrossRef]
- Lieber, C.S. Relationships Between Nutrition, Alcohol Use, and Liver Disease. Alcohol Res. Health 2003, 27, 220. [Google Scholar]
- Lieber, C.S. S-Adenosyl-l-methionine: Its role in the treatment of liver disorders. Am. J. Clin. Nutr. 2002, 76, 1183S–1187S. [Google Scholar] [CrossRef]
- Chantar, M.L.M.; García-Trevijano, E.R.; Latasa, M.U.; Pérez-Mato, I.; Del Pino, M.M.S.; Corrales, F.J.; Avila, M.; Mato, J.M. Importance of a deficiency in S-adenosyl-l-methionine synthesis in the pathogenesis of liver injury. Am. J. Clin. Nutr. 2002, 76, 1177S–1182S. [Google Scholar] [CrossRef]
- Videla, L.A.; Valenzuela, A. Alcohol ingestion, liver glutathione and lipoperoxidation: Metabolic interrelations and pathological implications. Life Sci. 1982, 31, 2395–2407. [Google Scholar] [CrossRef]
- Devi, S.L.; Anuradha, C.V. Mitochondrial damage, cytotoxicity and apoptosis in iron-potentiated alcoholic liver fibrosis: Amelioration by taurine. Amino Acids 2009, 38, 869–879. [Google Scholar] [CrossRef] [PubMed]
- McCarty, M.F. Nutraceutical strategies for ameliorating the toxic effects of alcohol. Med Hypotheses 2013, 80, 456–462. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, A.; Hobara, N.; Kobayashi, M.; Nakatsukasa, H.; Nagashima, H. Lowering of Blood Acetaldehyde but Not Ethanol Concentrations by Pantethine following Alcohol Ingestion: Different Effects in Flushing and Nonflushing Subjects. Alcohol. Clin. Exp. Res. 1985, 9, 272–276. [Google Scholar] [CrossRef]
- Masterton, G.S.; Plevris, J.N.; Hayes, P.C. Review article: Omega-3 fatty acids—A promising novel therapy for non-alcoholic fatty liver disease. Aliment. Pharmacol. Ther. 2009, 31, 679–692. [Google Scholar] [CrossRef]
- Pauluci, R.; Noto, A.R.; Curado, D.F.; Siqueira-Campos, M.J.; Bezerra, A.G.; Galduróz, J.C.F. Omega-3 for the Prevention of Alcohol Use Disorder Relapse: A Placebo-Controlled, Randomized Clinical Trial. Front. Psychiatry 2022, 13, 826448. [Google Scholar] [CrossRef]
- Wang, M.; Zhang, X.; Ma, L.-J.; Feng, R.-B.; Yan, C.; Su, H.; He, C.; Kang, J.X.; Liu, B.; Wan, J.-B. Omega-3 polyunsaturated fatty acids ameliorate ethanol-induced adipose hyperlipolysis: A mechanism for hepatoprotective effect against alcoholic liver disease. Biochim. Biophys. Acta (BBA)—Mol. Basis Dis. 2017, 1863, 3190–3201. [Google Scholar] [CrossRef]
- Mishra, B.R.; Nizamie, S.H.; Das, B.; Praharaj, S.K. Efficacy of repetitive transcranial magnetic stimulation in alcohol dependence: A sham-controlled study. Addiction 2010, 105, 49–55. [Google Scholar] [CrossRef]
- Kuhn, J.; Lenartz, D.; Huff, W.; Lee, S.; Koulousakis, A.; Klosterkoetter, J.; Sturm, V. Remission of alcohol dependency following deep brain stimulation of the nucleus accumbens: Valuable therapeutic implications? J. Neurol. Neurosurg. Psychiatry 2007, 78, 1152–1153. [Google Scholar] [CrossRef]
- Klauss, J.; Anders, Q.S.; Felippe, L.V.; Nitsche, M.A.; Nakamura-Palacios, E.M. Multiple Sessions of Transcranial Direct Current Stimulation (tDCS) Reduced Craving and Relapses for Alcohol Use: A Randomized Placebo-Controlled Trial in Alcohol Use Disorder. Front. Pharmacol. 2018, 9, 716. [Google Scholar] [CrossRef] [PubMed]
- Diana, M.; Bolloni, C.; Antonelli, M.; Di Giuda, D.; Cocciolillo, F.; Fattore, L.; Addolorato, G. Repetitive transcranial magnetic stimulation: Re-wiring the alcoholic human brain. Alcohol 2019, 74, 113–124. [Google Scholar] [CrossRef] [PubMed]
- Mark, T.L.; Kassed, C.A.; Vandivort-Warren, R.; Levit, K.R.; Kranzler, H.R. Alcohol and opioid dependence medications: Prescription trends, overall and by physician specialty. Drug Alcohol Depend. 2009, 99, 345–349. [Google Scholar] [CrossRef] [PubMed]
- Krampe, H.; Ehrenreich, H. Supervised Disulfiram as Adjunct to Psychotherapy in Alcoholism Treatment. Curr. Pharm. Des. 2010, 16, 2076–2090. [Google Scholar] [CrossRef]
- Fuller, R.K.; Branchey, L.; Brightwell, D.R.; Derman, R.M.; Emrick, C.D.; Iber, F.L.; James, K.E.; Lacoursiere, R.B.; Lee, K.K.; Lowenstam, I.; et al. Disulfiram Treatment of Alcoholism: A Veterans Administration Cooperative Study. JAMA 1986, 256, 1449–1455. [Google Scholar] [CrossRef] [PubMed]
- Suh, J.J.; Pettinati, H.M.; Kampman, K.M.; O’Brien, C.P. The Status of Disulfiram. J. Clin. Psychopharmacol. 2006, 26, 290–302. [Google Scholar] [CrossRef] [PubMed]
- Hald, J.; Jacobsen, E. A drug sensitising the organism to ethyl alcohol. Lancet 1948, 252, 1001–1004. [Google Scholar] [CrossRef]
- Ulrichsen, J.; Nielsen, M.K.; Ulrichsen, M. Disulfiram in severe alcohol-ism—An open controlled study. Nord. J. Psychiatry 2010, 64, 356–362. [Google Scholar] [CrossRef]
- Wright, C.; Moore, R.D. Disulfiram treatment of alcoholism. Am. J. Med. 1990, 88, 647–655. [Google Scholar] [CrossRef]
- Skinner, M.D.; Lahmek, P.; Pham, H.; Aubin, H.-J. Disulfiram Efficacy in the Treatment of Alcohol Dependence: A Meta-Analysis. PLoS ONE 2014, 9, e87366. [Google Scholar] [CrossRef]
- Alharbi, F.F.; El-Guebaly, N. The Relative Safety of Disulfiram. Addict. Disord. Their Treat. 2013, 12, 140–147. [Google Scholar] [CrossRef]
- O’Malley, S.S.; Jaffe, A.J.; Rode, S.; Rounsaville, B.J. Experience of a “slip” among alcoholics treated with naltrexone or placebo. Am. J. Psychiatry 1996, 153, 281–283. [Google Scholar] [CrossRef] [PubMed]
- Donoghue, K.; Elzerbi, C.; Saunders, R.; Whittington, C.; Pilling, S.; Drummond, C. The efficacy of acamprosate and naltrexone in the treatment of alcohol dependence, Europe versus the rest of the world: A meta-analysis. Addiction 2015, 110, 920–930. [Google Scholar] [CrossRef] [PubMed]
- Rösner, S.; Hackl-Herrwerth, A.; Leucht, S.; Vecchi, S.; Srisurapanont, M.; Soyka, M. Opioid Antagonists for Alcohol Dependence. Cochrane Database Syst Rev. 2010, 12, CD001867. [Google Scholar] [CrossRef]
- Rösner; Hackl-Herrwerth, A.; Leucht, S.; Lehert, P.; Vecchi, S.; Soyka, M. Acamprosate for Alcohol Dependence. Cochrane Database Syst Rev. 2010, 9, CD004332. [Google Scholar] [CrossRef]
- Fitzgerald, N.; Angus, K.; Elders, A.; De Andrade, M.; Raistrick, D.; Heather, N.; McCambridge, J. Weak evidence on nalmefene creates dilemmas for clinicians and poses questions for regulators and researchers. Addiction 2016, 111, 1477–1487. [Google Scholar] [CrossRef]
- Mann, K.; Torup, L.; Sørensen, P.; Gual, A.; Swift, R.; Walker, B.; van den Brink, W. Nalmefene for the management of alcohol dependence: Review on its pharmacology, mechanism of action and meta-analysis on its clinical efficacy. Eur. Neuropsychopharmacol. 2016, 26, 1941–1949. [Google Scholar] [CrossRef]
- Mason, B.J.; Salvato, F.R.; Williams, L.D.; Ritvo, E.C.; Cutler, R.B. A Double-blind, Placebo-Controlled Study of Oral Nalmefene for Alcohol Dependence. Arch. Gen. Psychiatry 1999, 56, 719–724. [Google Scholar] [CrossRef]
- Anton, R.F.; Pettinati, H.; Zweben, A.; Kranzler, H.R.; Johnson, B.; Bohn, M.J.; McCaul, M.E.; Anthenelli, R.; Salloum, I.; Galloway, G.; et al. A Multi-site Dose Ranging Study of Nalmefene in the Treatment of Alcohol Dependence. J. Clin. Psychopharmacol. 2004, 24, 421–428. [Google Scholar] [CrossRef]
- Karhuvaara, S.; Simojoki, K.; Virta, A.; Rosberg, M.; Löyttyniemi, E.; Nurminen, T.; Kallio, A.; Mäkelä, R. Targeted Nalmefene With Simple Medical Management in the Treatment of Heavy Drinkers: A Randomized Double-Blind Placebo-Controlled Multicenter Study. Alcohol. Clin. Exp. Res. 2007, 31, 1179–1187. [Google Scholar] [CrossRef]
- Brink, W.V.D.; Strang, J.; Gual, A.; Sørensen, P.; Jensen, T.J.; Mann, K. Safety and tolerability of as-needed nalmefene in the treatment of alcohol dependence: Results from the Phase III clinical programme. Expert Opin. Drug Saf. 2015, 14, 495–504. [Google Scholar] [CrossRef] [PubMed]
- Pierce, M.; Sutterland, A.; Beraha, E.M.; Morley, K.; Brink, W.V.D. Efficacy, tolerability, and safety of low-dose and high-dose baclofen in the treatment of alcohol dependence: A systematic review and meta-analysis. Eur. Neuropsychopharmacol. 2018, 28, 795–806. [Google Scholar] [CrossRef] [PubMed]
- Leung, J.G.; Hall-Flavin, D.; Nelson, S.; Schmidt, K.A.; Schak, K.M. The Role of Gabapentin in the Management of Alcohol Withdrawal and Dependence. Ann. Pharmacother. 2015, 49, 897–906. [Google Scholar] [CrossRef] [PubMed]
- Wiffen, P.J.; Derry, S.; Bell, R.F.; Rice, A.S.; Tölle, T.R.; Phillips, T.; Moore, R.A. Gabapentin for chronic neuropathic pain in adults (Review). Cochrane Database Syst. Rev. 2017, 6, CD007938. [Google Scholar] [CrossRef] [PubMed]
- McLean, M.J.; Bukhari, A.A.; Wamil, A.W. Effects of topiramate on sodium-dependent action-potential firing by mouse spinal cord neurons in cell culture. Epilepsia 2000, 41, 21–24. [Google Scholar] [CrossRef]
- White, H.; Brown, S.; Woodhead, J.H.; Skeen, G.A.; Wolf, H.H. Topiramate enhances GABA-mediated chloride flux and GABA-evoked chloride currents in murine brain neurons and increases seizure threshold. Epilepsy Res. 1997, 28, 167–179. [Google Scholar] [CrossRef]
- Shank, R.P.; Gardocki, J.F.; Vaught, J.L.; Davis, C.B.; Schupsky, J.J.; Raffa, R.B.; Dodgson, S.J.; Nortey, S.O.; Maryanoff, B.E. Topiramate: Preclinical Evaluation of a Structurally Novel Anticonvulsant. Epilepsia 1994, 35, 450–460. [Google Scholar] [CrossRef]
- Skradski, S.; White, H.S. Topiramate blocks kainate-evoked cobalt influx into cultured neurons. Epilepsia 2000, 41, 45–47. [Google Scholar] [CrossRef]
- Yilmaz, M.; Naziroǧlu, M.; Kutluhan, S.; Yilmaz, N.; Yürekli, V.A.; Vural, H. Topiramate modulates hippocampus NMDA receptors via brain Ca2+ homeostasis in pentylentetrazol-induced epilepsy of rats. J. Recept. Signal Transduct. 2011, 31, 173–179. [Google Scholar] [CrossRef]
- Qian, J.; Noebels, J.L. Topiramate alters excitatory synaptic transmission in mouse hippocampus. Epilepsy Res. 2003, 55, 225–233. [Google Scholar] [CrossRef]
- Zhang, X.; Velumian, A.A.; Jones, O.T.; Carlen, P.L. Modulation of high-voltage-activated calcium channels in dentate granule cells by topiramate. Epilepsia. 2000, 41, 52–60. [Google Scholar] [CrossRef] [PubMed]
- Langtry, H.D.; Gillis, J.C.; Davis, R. Topiramate. Drugs 1997, 54, 752–773. [Google Scholar] [CrossRef] [PubMed]
- Mula, M.; Cavanna, A.E.; Monaco, F. Psychopharmacology of topiramate: From epilepsy to bipolar disorder. Neuropsychiatr. Dis. Treat. 2006, 2, 475–488. [Google Scholar] [CrossRef] [PubMed]
- Blodgett, J.C.; Del Re, A.; Maisel, N.C.; Finney, J.W. A Meta-Analysis of Topiramate’s Effects for Individuals with Alcohol Use Disorders. Alcohol. Clin. Exp. Res. 2014, 38, 1481–1488. [Google Scholar] [CrossRef] [PubMed]
- Shank, R.P.; Gardocki, J.F.; Streeter, A.J.; Maryanoff, B.E. An Overview of the Preclinical Aspects of Topiramate: Pharmacology, Pharmacokinetics, and Mechanism of Action. Epilepsia 2000, 41, 3–9. [Google Scholar] [CrossRef]
- Soyka, M.; Müller, C.A. Pharmacotherapy of alcoholism—An update on approved and off-label medications. Expert Opin. Pharmacother. 2017, 18, 1187–1199. [Google Scholar] [CrossRef]
- Mason, B.J. Emerging pharmacotherapies for alcohol use disorder. Neuropharmacology 2017, 122, 244–253. [Google Scholar] [CrossRef]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yang, W.; Singla, R.; Maheshwari, O.; Fontaine, C.J.; Gil-Mohapel, J. Alcohol Use Disorder: Neurobiology and Therapeutics. Biomedicines 2022, 10, 1192. https://doi.org/10.3390/biomedicines10051192
Yang W, Singla R, Maheshwari O, Fontaine CJ, Gil-Mohapel J. Alcohol Use Disorder: Neurobiology and Therapeutics. Biomedicines. 2022; 10(5):1192. https://doi.org/10.3390/biomedicines10051192
Chicago/Turabian StyleYang, Waisley, Rohit Singla, Oshin Maheshwari, Christine J. Fontaine, and Joana Gil-Mohapel. 2022. "Alcohol Use Disorder: Neurobiology and Therapeutics" Biomedicines 10, no. 5: 1192. https://doi.org/10.3390/biomedicines10051192
APA StyleYang, W., Singla, R., Maheshwari, O., Fontaine, C. J., & Gil-Mohapel, J. (2022). Alcohol Use Disorder: Neurobiology and Therapeutics. Biomedicines, 10(5), 1192. https://doi.org/10.3390/biomedicines10051192