Adverse Events and Tolerability of Combined Durvalumab and Tremelimumab versus Durvalumab Alone in Solid Cancers: A Systematic Review and Meta-Analysis
Abstract
:1. Introduction
2. Materials and Methods
2.1. Search Strategy
2.2. Data Extraction
2.3. Primary Outcomes
2.4. Statistical Analysis
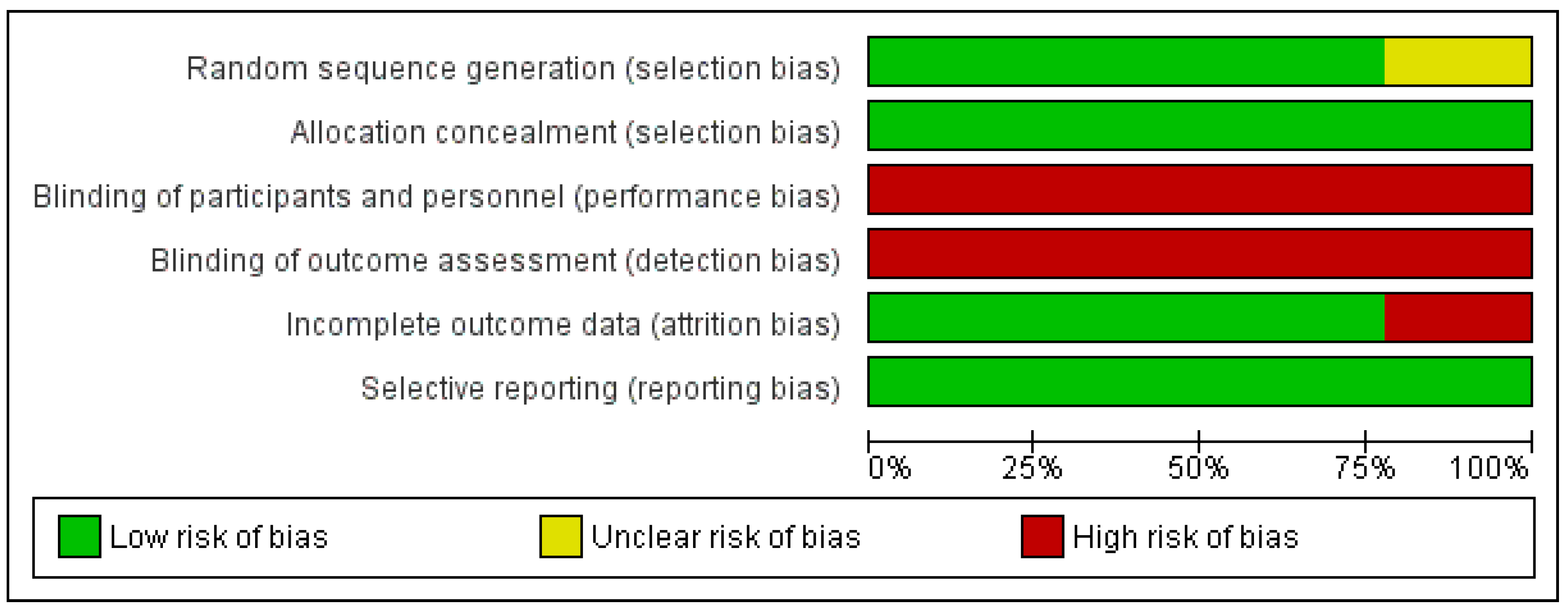
2.5. Risk of Bias Assessment
3. Results
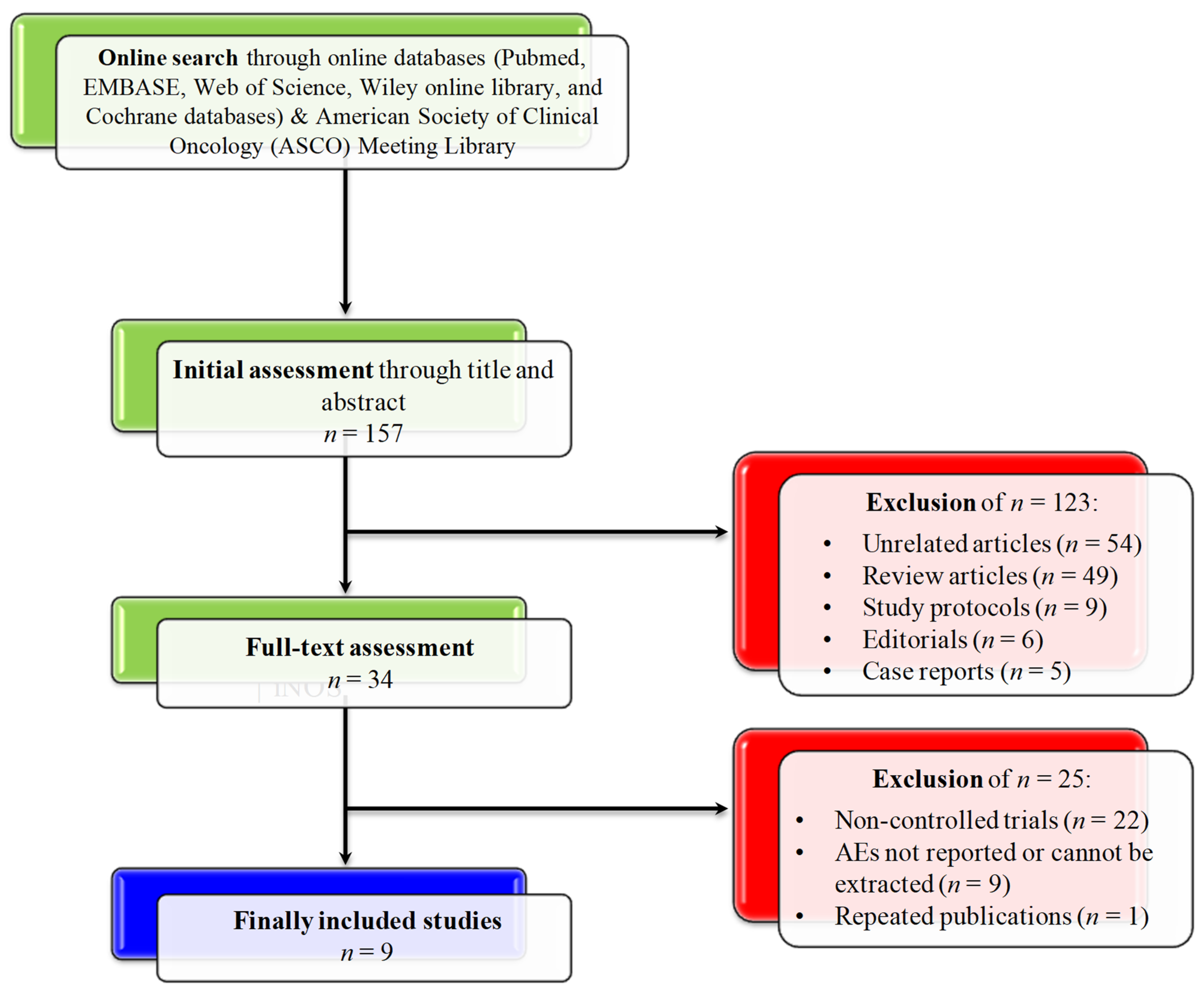
3.1. Search Results
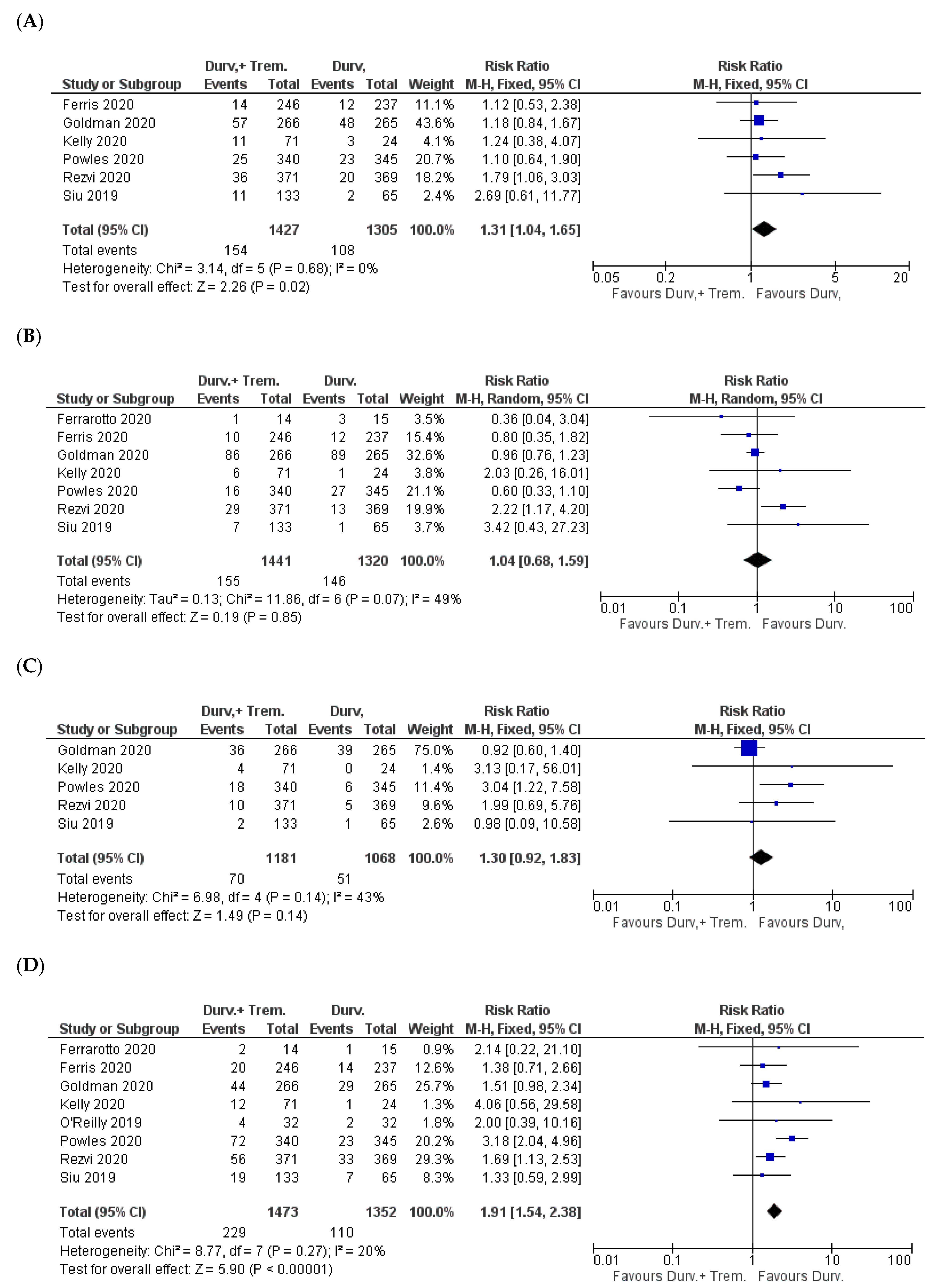
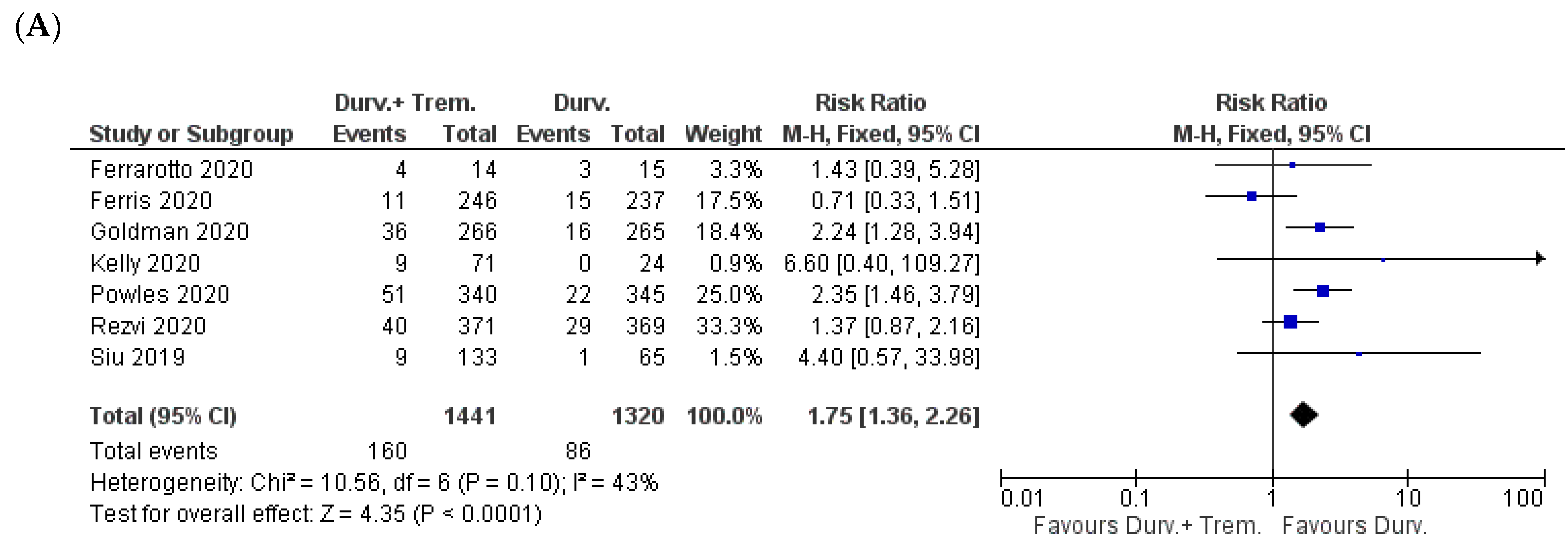
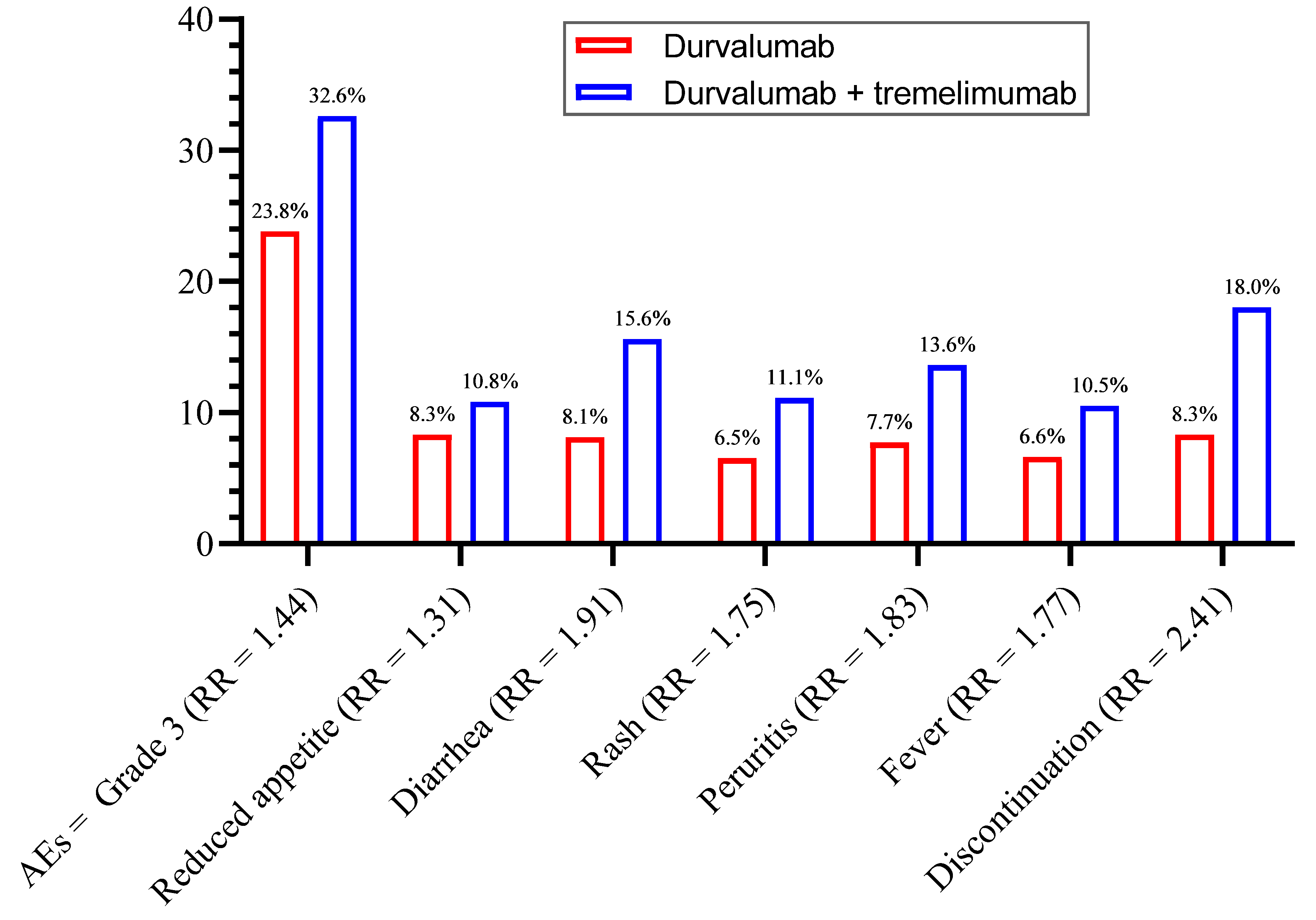
3.2. Overall Incidence of Side Effects
3.3. Gastrointestinal Side Effects
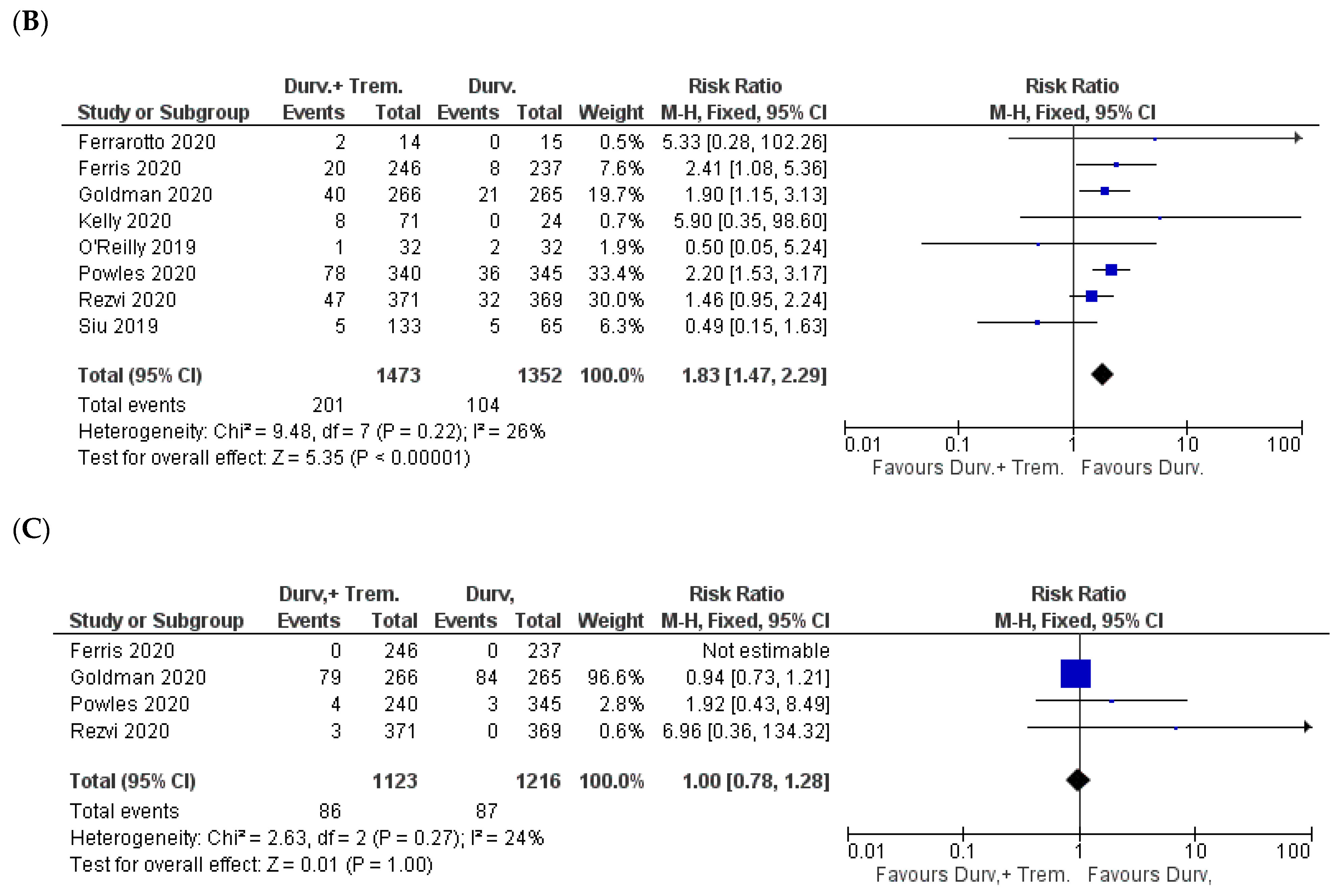
3.4. Dermal Manifestations
3.5. Hematological Side Effects
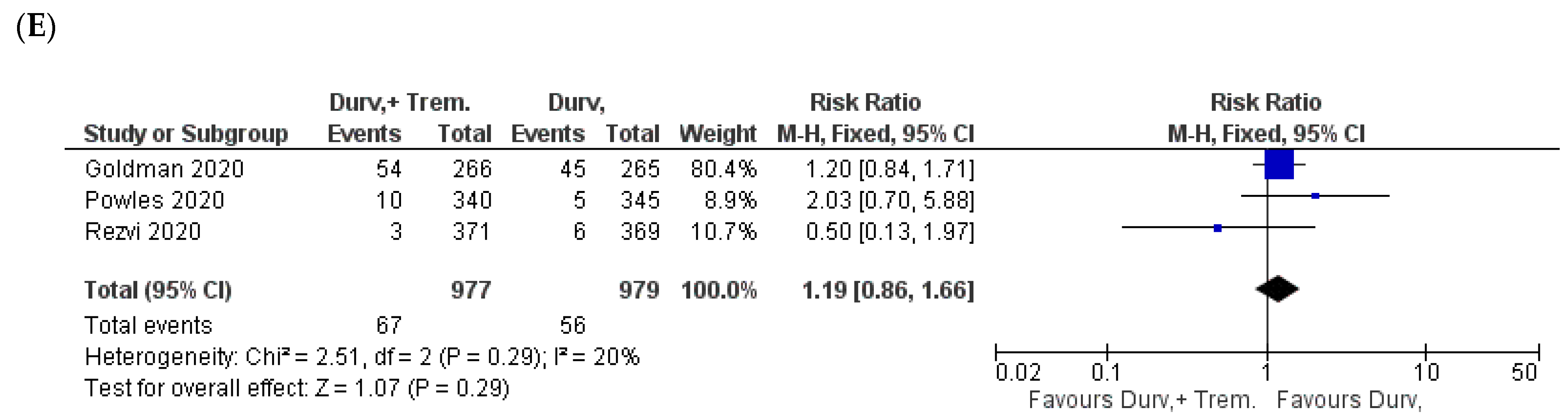
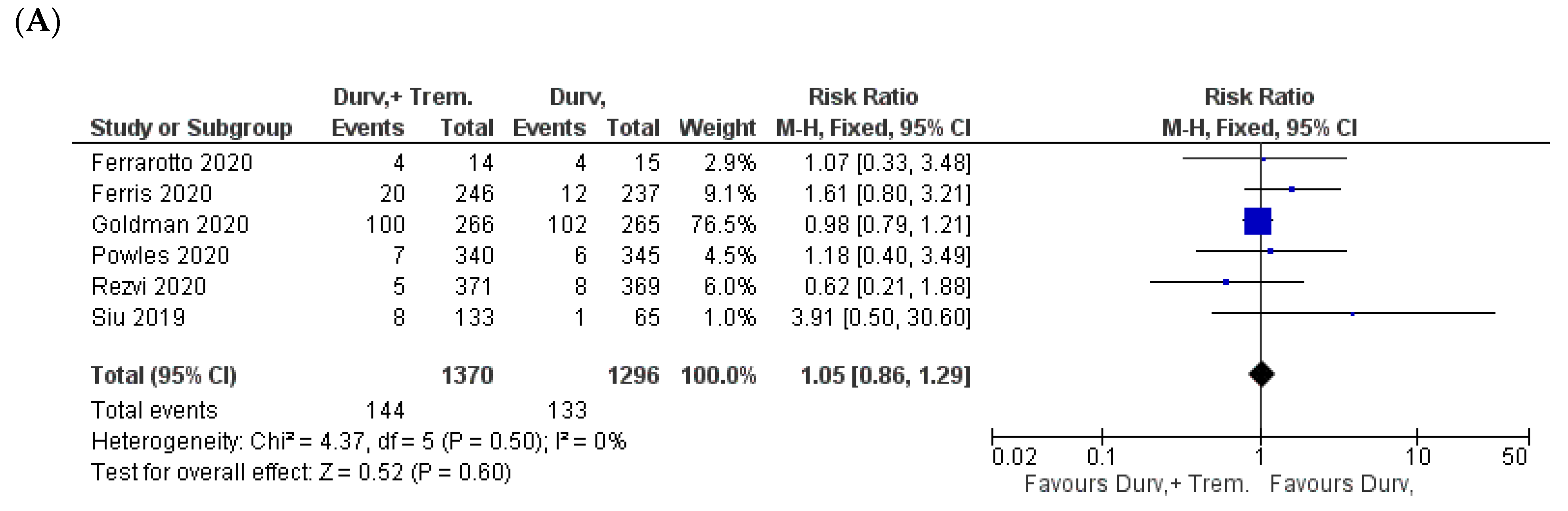
3.6. Metabolic and Endocrine Manifestations
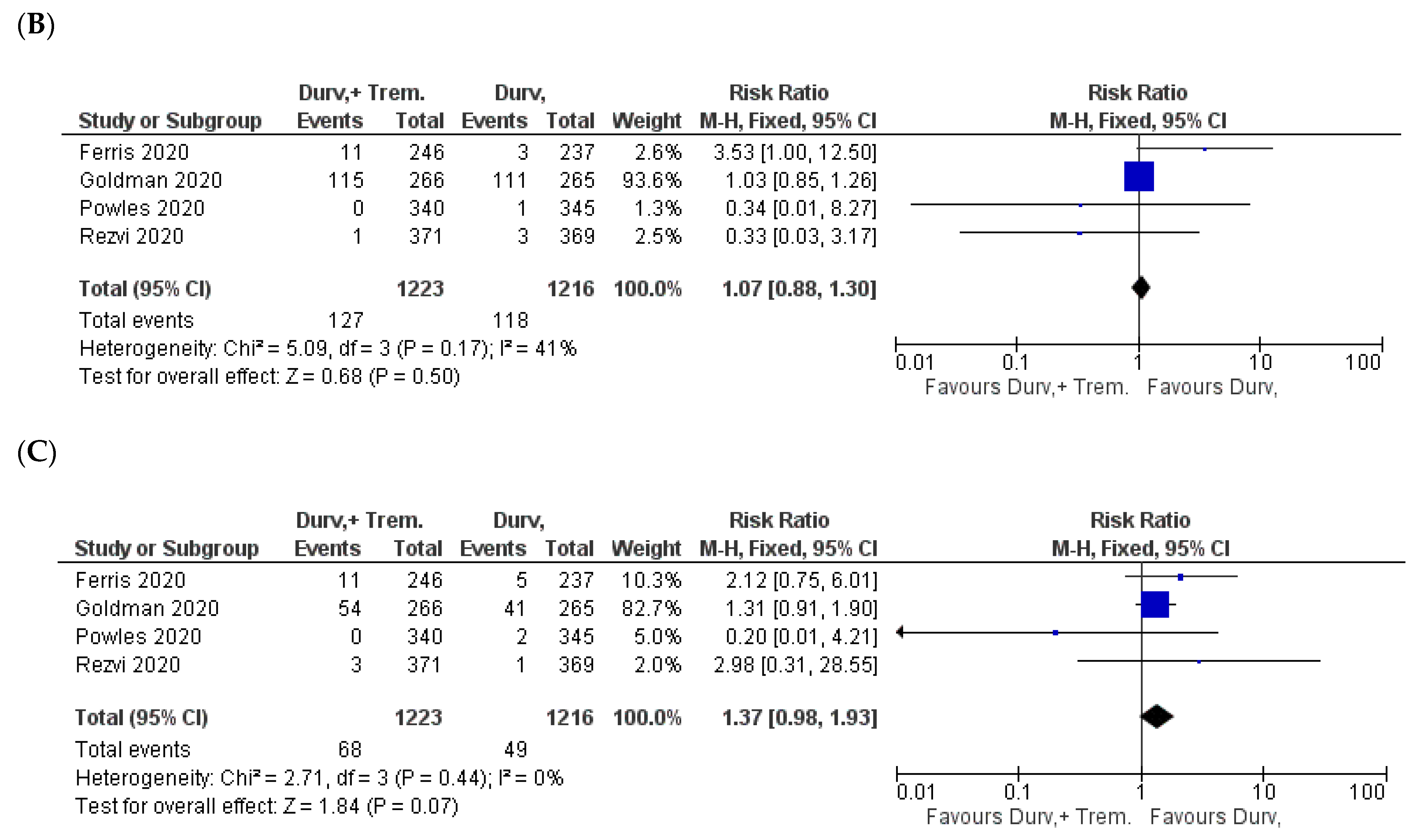
3.7. General Manifestations
3.8. Treatment Discontinuation and Mortality
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Fahmy, O.; Khairul-Asri, M.G.; Stenzl, A.; Gakis, G. The current status of checkpoint inhibitors in metastatic bladder cancer. Clin. Exp. Metastasis 2016, 33, 629–635. [Google Scholar] [CrossRef] [PubMed]
- Fahmy, O.; Alhakamy, N.A.; Khairul-Asri, M.G.; Ahmed, O.A.A.; Fahmy, U.A.; Fresta, C.G.; Caruso, G. Oncological response and predictive biomarkers for the checkpoint inhibitors in castration-resistant metastatic prostate cancer: A systematic review and meta-analysis. J. Pers. Med. 2021, 12, 8. [Google Scholar] [CrossRef] [PubMed]
- Fahmy, O.; Alhakamy, N.A.; Rizg, W.Y.; Bagalagel, A.; Alamoudi, A.J.; Aldawsari, H.M.; Khateb, A.M.; Eldakhakhny, B.M.; Fahmy, U.A.; Abdulaal, W.H.; et al. Updates on molecular and biochemical development and progression of prostate cancer. J. Clin. Med. 2021, 10, 5127. [Google Scholar] [CrossRef] [PubMed]
- Shsm, H.; Fahmy, U.A.; Alhakamy, N.A.; Khairul-Asri, M.G.; Fahmy, O. Neoadjuvant therapy using checkpoint inhibitors before radical cystectomy for muscle invasive bladder cancer: A systematic review. J. Pers. Med. 2021, 11, 1195. [Google Scholar] [CrossRef]
- Wills, S.; Hochmuth, L.K.; Bauer, K.S., Jr.; Deshmukh, R. Durvalumab: A newly approved checkpoint inhibitor for the treatment of urothelial carcinoma. Curr. Probl. Cancer 2019, 43, 181–194. [Google Scholar] [CrossRef]
- Syn, N.L.; Teng, M.W.L.; Mok, T.S.K.; Soo, R.A. De-novo and acquired resistance to immune checkpoint targeting. Lancet Oncol. 2017, 18, e731–e741. [Google Scholar] [CrossRef]
- Lopez-Beltran, A.; Cimadamore, A.; Blanca, A.; Massari, F.; Vau, N.; Scarpelli, M.; Cheng, L.; Montironi, R. Immune checkpoint inhibitors for the treatment of bladder cancer. Cancers 2021, 13, 131. [Google Scholar] [CrossRef]
- Fitzpatrick, O.; Naidoo, J. Immunotherapy for stage iii nsclc: Durvalumab and beyond. Lung Cancer 2021, 12, 123–131. [Google Scholar] [CrossRef]
- Al-Salama, Z.T. Durvalumab: A review in extensive-stage sclc. Target Oncol. 2021, 16, 857–864. [Google Scholar] [CrossRef]
- Ribas, A. Clinical development of the anti-ctla-4 antibody tremelimumab. Semin Oncol 2010, 37, 450–454. [Google Scholar] [CrossRef]
- Martins, F.; Sofiya, L.; Sykiotis, G.P.; Lamine, F.; Maillard, M.; Fraga, M.; Shabafrouz, K.; Ribi, C.; Cairoli, A.; Guex-Crosier, Y.; et al. Adverse effects of immune-checkpoint inhibitors: Epidemiology, management and surveillance. Nat. Rev. Clin. Oncol. 2019, 16, 563–580. [Google Scholar] [CrossRef] [PubMed]
- Chikuma, S.; Abbas, A.K.; Bluestone, J.A. B7-independent inhibition of t cells by ctla-4. J. Immunol. 2005, 175, 177–181. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Brzostek, J.; Gascoigne, N.R.; Rybakin, V. Cell type-specific regulation of immunological synapse dynamics by b7 ligand recognition. Front Immunol 2016, 7, 24. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Keilholz, U. Ctla-4: Negative regulator of the immune response and a target for cancer therapy. J. Immunother. 2008, 31, 431–439. [Google Scholar] [CrossRef]
- Ribas, A. Tumor immunotherapy directed at pd-1. N Engl J Med 2012, 366, 2517–2519. [Google Scholar] [CrossRef] [Green Version]
- Sabbatino, F.; Conti, V.; Liguori, L.; Polcaro, G.; Corbi, G.; Manzo, V.; Tortora, V.; Carlomagno, C.; Vecchione, C.; Filippelli, A.; et al. Molecules and mechanisms to overcome oxidative stress inducing cardiovascular disease in cancer patients. Life 2021, 11, 105. [Google Scholar] [CrossRef]
- Teppo, H.R.; Soini, Y.; Karihtala, P. Reactive oxygen species-mediated mechanisms of action of targeted cancer therapy. Oxid. Med. Cell Longev. 2017, 2017, 1485283. [Google Scholar] [CrossRef]
- Caruso, G.; Fresta, C.G.; Grasso, M.; Santangelo, R.; Lazzarino, G.; Lunte, S.M.; Caraci, F. Inflammation as the common biological link between depression and cardiovascular diseases: Can carnosine exert a protective role? Curr. Med. Chem. 2020, 27, 1782–1800. [Google Scholar] [CrossRef]
- Remon, J.; Passiglia, F.; Ahn, M.J.; Barlesi, F.; Forde, P.M.; Garon, E.B.; Gettinger, S.; Goldberg, S.B.; Herbst, R.S.; Horn, L.; et al. Immune checkpoint inhibitors in thoracic malignancies: Review of the existing evidence by an iaslc expert panel and recommendations. J. Thorac. Oncol. 2020, 15, 914–947. [Google Scholar] [CrossRef]
- Baxi, S.; Yang, A.; Gennarelli, R.L.; Khan, N.; Wang, Z.; Boyce, L.; Korenstein, D. Immune-related adverse events for anti-pd-1 and anti-pd-l1 drugs: Systematic review and meta-analysis. BMJ 2018, 360, k793. [Google Scholar] [CrossRef] [Green Version]
- Thakker, R.A.; Lee, M.A.; Albaeni, A.; Elbadawi, A.; Suthar, K.H.; Perez, C.; Sonstein, L.K.; Farr, N.M.; Venkatesan, R.; Khalife, W.; et al. Clinical characteristics and outcomes in immune checkpoint inhibitor therapy-associated myocarditis. Cardiol. Res. 2021, 12, 270–278. [Google Scholar] [CrossRef] [PubMed]
- Ferrarotto, R.; Bell, D.; Rubin, M.L.; Hutcheson, K.A.; Johnson, J.M.; Goepfert, R.P.; Phan, J.; Elamin, Y.Y.; Torman, D.K.; Warneke, C.L.; et al. Impact of neoadjuvant durvalumab with or without tremelimumab on cd8(+) tumor lymphocyte density, safety, and efficacy in patients with oropharynx cancer: Ciao trial results. Clin. Cancer Res. 2020, 26, 3211–3219. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ferris, R.L.; Haddad, R.; Even, C.; Tahara, M.; Dvorkin, M.; Ciuleanu, T.E.; Clement, P.M.; Mesia, R.; Kutukova, S.; Zholudeva, L.; et al. Durvalumab with or without tremelimumab in patients with recurrent or metastatic head and neck squamous cell carcinoma: Eagle, a randomized, open-label phase iii study. Ann. Oncol. 2020, 31, 942–950. [Google Scholar] [CrossRef]
- Goldman, J.W.; Dvorkin, M.; Chen, Y.; Reinmuth, N.; Hotta, K.; Trukhin, D.; Statsenko, G.; Hochmair, M.J.; Özgüroğlu, M.; Ji, J.H.; et al. Durvalumab, with or without tremelimumab, plus platinum-etoposide versus platinum-etoposide alone in first-line treatment of extensive-stage small-cell lung cancer (caspian): Updated results from a randomised, controlled, open-label, phase 3 trial. Lancet Oncol. 2021, 22, 51–65. [Google Scholar] [CrossRef]
- Kelly, R.J.; Lee, J.; Bang, Y.J.; Almhanna, K.; Blum-Murphy, M.; Catenacci, D.V.T.; Chung, H.C.; Wainberg, Z.A.; Gibson, M.K.; Lee, K.W.; et al. Safety and efficacy of durvalumab and tremelimumab alone or in combination in patients with advanced gastric and gastroesophageal junction adenocarcinoma. Clin. Cancer Res. 2020, 26, 846–854. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- O’Reilly, E.M.; Oh, D.Y.; Dhani, N.; Renouf, D.J.; Lee, M.A.; Sun, W.; Fisher, G.; Hezel, A.; Chang, S.C.; Vlahovic, G.; et al. Durvalumab with or without tremelimumab for patients with metastatic pancreatic ductal adenocarcinoma: A phase 2 randomized clinical trial. JAMA Oncol. 2019, 5, 1431–1438. [Google Scholar] [CrossRef]
- Planchard, D.; Reinmuth, N.; Orlov, S.; Fischer, J.R.; Sugawara, S.; Mandziuk, S.; Marquez-Medina, D.; Novello, S.; Takeda, Y.; Soo, R.; et al. Arctic: Durvalumab with or without tremelimumab as third-line or later treatment of metastatic non-small-cell lung cancer. Ann. Oncol. 2020, 31, 609–618. [Google Scholar] [CrossRef] [Green Version]
- Moher, D.; Shamseer, L.; Clarke, M.; Ghersi, D.; Liberati, A.; Petticrew, M.; Shekelle, P.; Stewart, L.A. Preferred reporting items for systematic review and meta-analysis protocols (prisma-p) 2015 statement. Syst. Rev. 2015, 4, 1. [Google Scholar] [CrossRef] [Green Version]
- Caruso, G.; Godos, J.; Castellano, S.; Micek, A.; Murabito, P.; Galvano, F.; Ferri, R.; Grosso, G.; Caraci, F. The therapeutic potential of carnosine/anserine supplementation against cognitive decline: A systematic review with meta-analysis. Biomedicines 2021, 9, 253. [Google Scholar] [CrossRef]
- Powles, T.; van der Heijden, M.S.; Castellano, D.; Galsky, M.D.; Loriot, Y.; Petrylak, D.P.; Ogawa, O.; Park, S.H.; Lee, J.L.; De Giorgi, U.; et al. Durvalumab alone and durvalumab plus tremelimumab versus chemotherapy in previously untreated patients with unresectable, locally advanced or metastatic urothelial carcinoma (danube): A randomised, open-label, multicentre, phase 3 trial. Lancet Oncol. 2020, 21, 1574–1588. [Google Scholar] [CrossRef]
- Rizvi, N.A.; Cho, B.C.; Reinmuth, N.; Lee, K.H.; Luft, A.; Ahn, M.J.; van den Heuvel, M.M.; Cobo, M.; Vicente, D.; Smolin, A.; et al. Durvalumab with or without tremelimumab vs standard chemotherapy in first-line treatment of metastatic non-small cell lung cancer: The mystic phase 3 randomized clinical trial. JAMA Oncol. 2020, 6, 661–674. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Siu, L.L.; Even, C.; Mesía, R.; Remenar, E.; Daste, A.; Delord, J.P.; Krauss, J.; Saba, N.F.; Nabell, L.; Ready, N.E.; et al. Safety and efficacy of durvalumab with or without tremelimumab in patients with pd-l1-low/negative recurrent or metastatic hnscc: The phase 2 condor randomized clinical trial. JAMA Oncol. 2019, 5, 195–203. [Google Scholar] [CrossRef] [PubMed]
- Parikh, R.B.; Min, E.J.; Wileyto, E.P.; Riaz, F.; Gross, C.P.; Cohen, R.B.; Hubbard, R.A.; Long, Q.; Mamtani, R. Uptake and survival outcomes following immune checkpoint inhibitor therapy among trial-ineligible patients with advanced solid cancers. JAMA Oncol. 2021, 7, 1843. [Google Scholar] [CrossRef] [PubMed]
- Russo, G.I.; Musso, N.; Romano, A.; Caruso, G.; Petralia, S.; Lanzanò, L.; Broggi, G.; Camarda, M. The role of dielectrophoresis for cancer diagnosis and prognosis. Cancers 2021, 14, 198. [Google Scholar] [CrossRef] [PubMed]
- Patel, S.P.; Kurzrock, R. Pd-l1 expression as a predictive biomarker in cancer immunotherapy. Mol. Cancer Ther. 2015, 14, 847–856. [Google Scholar] [CrossRef] [Green Version]
- Hotte, S.; Winquist, E.; Chi, K.; Ellard, S.; Sridhar, S.; Emmenegger, U.; Salim, M.; Iqbal, N.; Canil, C.; Kollmannsberger, C. Cctg ind 232: A phase ii study of durvalumab with or without tremelimumab in patients with metastatic castration resistant prostate cancer (mcrpc). Ann. Oncol. 2019, 30, v885. [Google Scholar] [CrossRef]
- Kwon, E.D.; Drake, C.G.; Scher, H.I.; Fizazi, K.; Bossi, A.; Van den Eertwegh, A.J.; Krainer, M.; Houede, N.; Santos, R.; Mahammedi, H. Ipilimumab versus placebo after radiotherapy in patients with metastatic castration-resistant prostate cancer that had progressed after docetaxel chemotherapy (ca184-043): A multicentre, randomised, double-blind, phase 3 trial. Lancet Oncol. 2014, 15, 700–712. [Google Scholar] [CrossRef] [Green Version]
- Karzai, F.; VanderWeele, D.; Madan, R.A.; Owens, H.; Cordes, L.M.; Hankin, A.; Couvillon, A.; Nichols, E.; Bilusic, M.; Beshiri, M.L.; et al. Activity of durvalumab plus olaparib in metastatic castration-resistant prostate cancer in men with and without DNA damage repair mutations. J. Immunother. Cancer 2018, 6, 141. [Google Scholar] [CrossRef]
- Calabrò, L.; Morra, A.; Giannarelli, D.; Amato, G.; D’Incecco, A.; Covre, A.; Lewis, A.; Rebelatto, M.C.; Danielli, R.; Altomonte, M.; et al. Tremelimumab combined with durvalumab in patients with mesothelioma (nibit-meso-1): An open-label, non-randomised, phase 2 study. Lancet Respir. Med. 2018, 6, 451–460. [Google Scholar] [CrossRef]
- Calabrò, L.; Morra, A.; Fonsatti, E.; Cutaia, O.; Amato, G.; Giannarelli, D.; Di Giacomo, A.M.; Danielli, R.; Altomonte, M.; Mutti, L.; et al. Tremelimumab for patients with chemotherapy-resistant advanced malignant mesothelioma: An open-label, single-arm, phase 2 trial. Lancet Oncol. 2013, 14, 1104–1111. [Google Scholar] [CrossRef]
- Intlekofer, A.M.; Thompson, C.B. At the bench: Preclinical rationale for ctla-4 and pd-1 blockade as cancer immunotherapy. J. Leukoc. Biol. 2013, 94, 25–39. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wei, S.C.; Levine, J.H.; Cogdill, A.P.; Zhao, Y.; Anang, N.A.S.; Andrews, M.C.; Sharma, P.; Wang, J.; Wargo, J.A.; Pe’er, D.; et al. Distinct cellular mechanisms underlie anti-ctla-4 and anti-pd-1 checkpoint blockade. Cell 2017, 170, 1120–1133.e1117. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Leighl, N.B.; Redman, M.W.; Rizvi, N.; Hirsch, F.R.; Mack, P.C.; Schwartz, L.H.; Wade, J.L.; Irvin, W.J.; Reddy, S.C.; Crawford, J.; et al. Phase ii study of durvalumab plus tremelimumab as therapy for patients with previously treated anti-pd-1/pd-l1 resistant stage iv squamous cell lung cancer (lung-map substudy s1400f, nct03373760). J. Immunother. Cancer 2021, 9, e002973. [Google Scholar] [CrossRef] [PubMed]
- Pires da Silva, I.; Ahmed, T.; Reijers, I.L.M.; Weppler, A.M.; Betof Warner, A.; Patrinely, J.R.; Serra-Bellver, P.; Allayous, C.; Mangana, J.; Nguyen, K.; et al. Ipilimumab alone or ipilimumab plus anti-pd-1 therapy in patients with metastatic melanoma resistant to anti-pd-(l)1 monotherapy: A multicentre, retrospective, cohort study. Lancet Oncol. 2021, 22, 836–847. [Google Scholar] [CrossRef]
- Schoenfeld, J.D.; Giobbie-Hurder, A.; Ranasinghe, S.; Kao, K.Z.; Lako, A.; Tsuji, J.; Liu, Y.; Brennick, R.C.; Gentzler, R.D.; Lee, C.; et al. Durvalumab plus tremelimumab alone or in combination with low-dose or hypofractionated radiotherapy in metastatic non-small-cell lung cancer refractory to previous pd(l)-1 therapy: An open-label, multicentre, randomised, phase 2 trial. Lancet Oncol. 2022, 23, 279–291. [Google Scholar] [CrossRef]
- Smolarz, B.; Makowska, M.; Romanowicz, H. Pharmacogenetics of drug-resistant epilepsy (review of literature). Int. J. Mol. Sci. 2021, 22, 11696. [Google Scholar] [CrossRef]


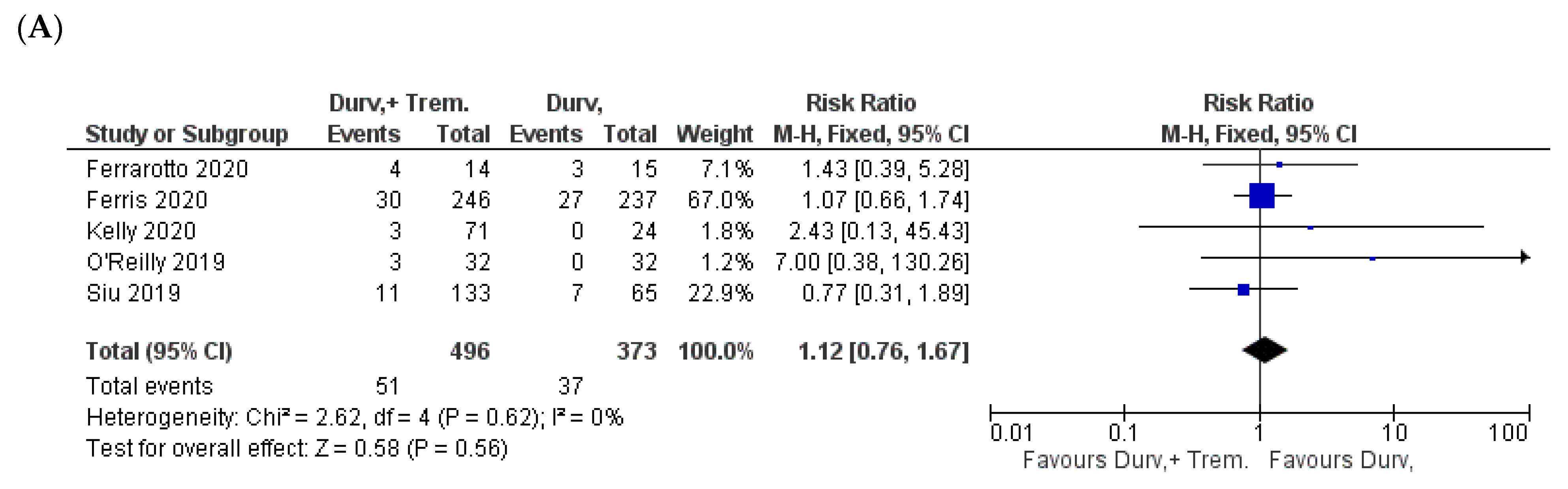

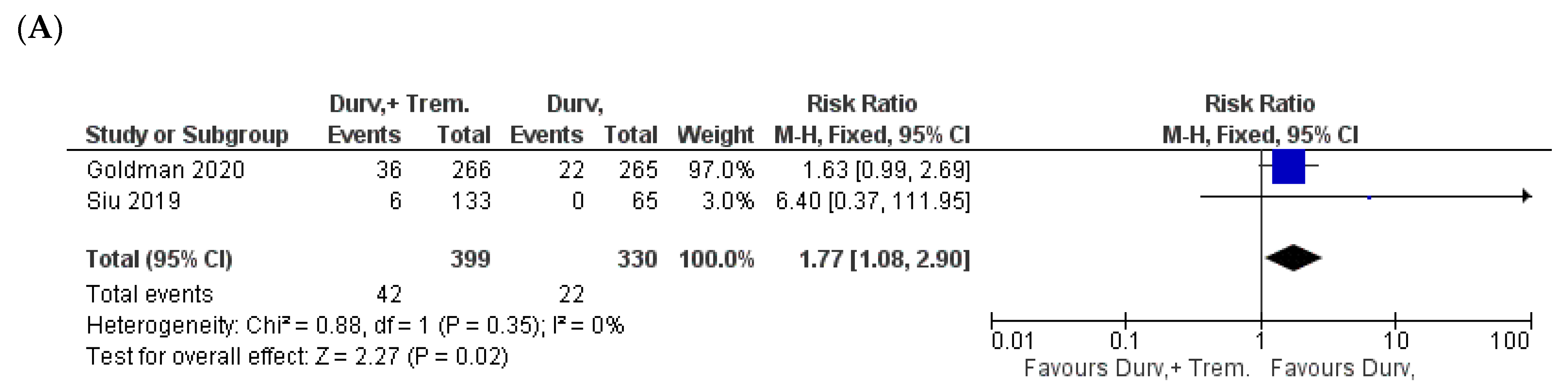
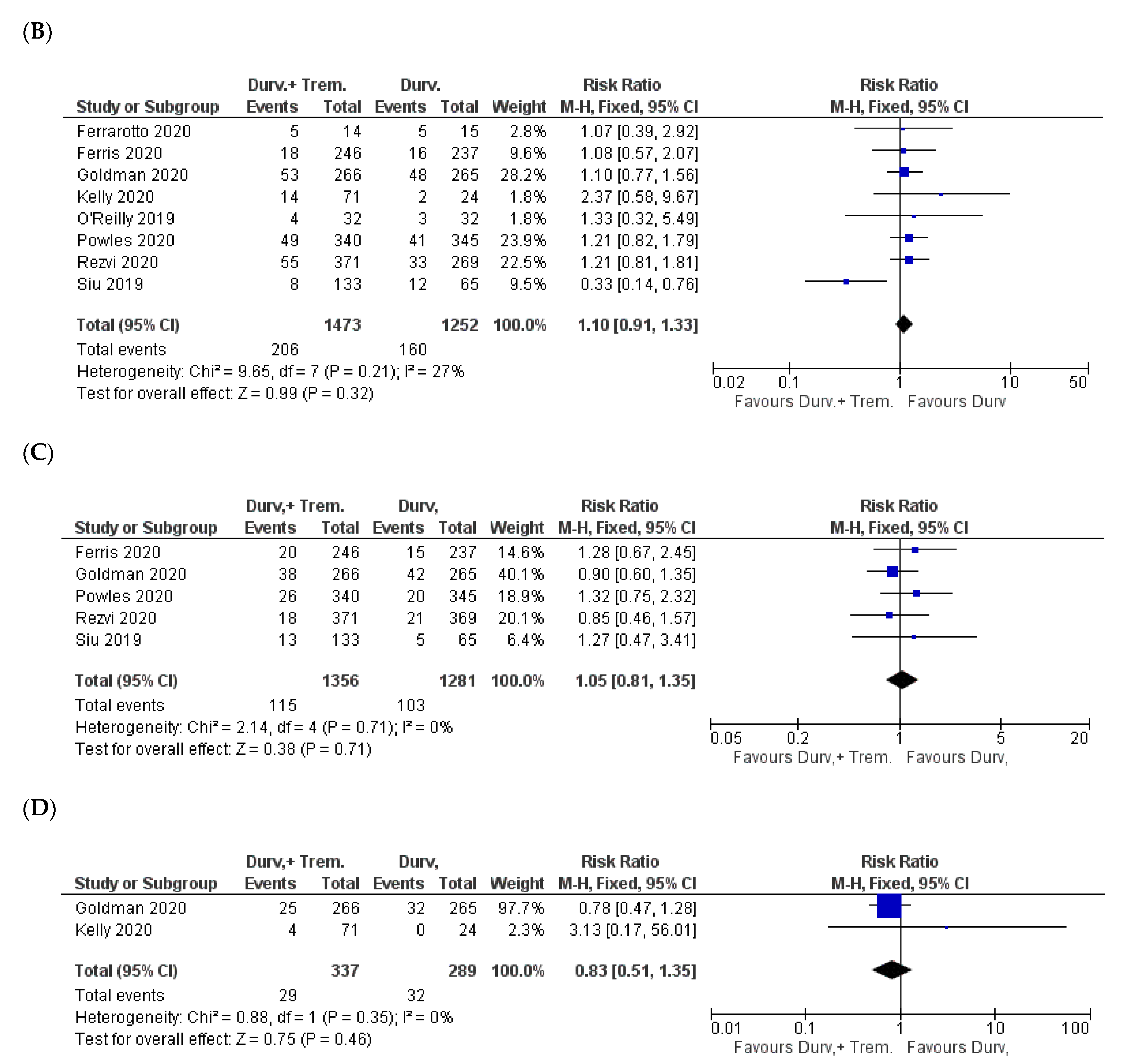
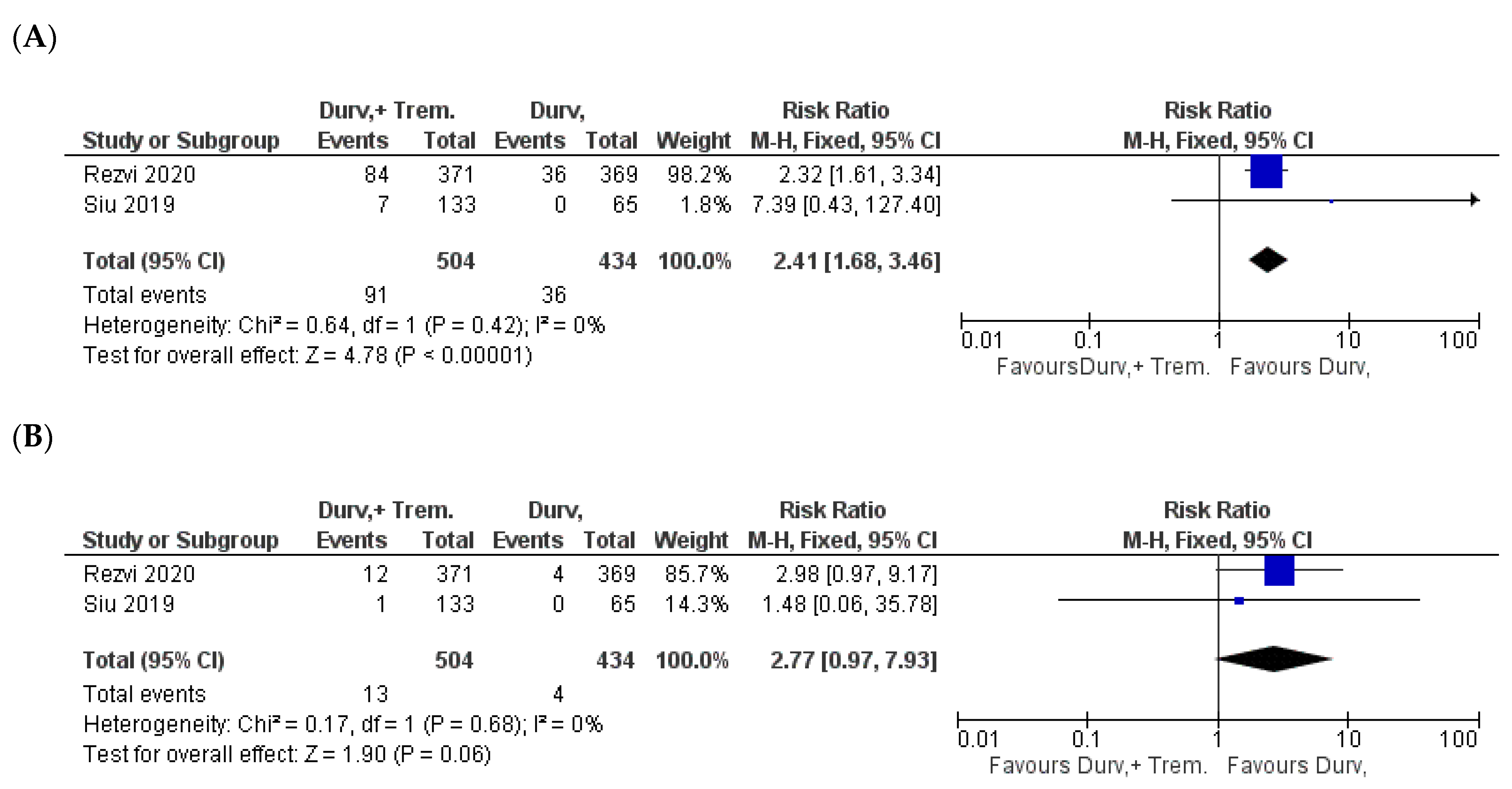
| Study | NCT ID/Trial Name | Phase and Status | Timeframe | Patient Criteria | Sample Size (D + T vs. D) | Doses | Outcome |
|---|---|---|---|---|---|---|---|
| Ferrarotto 2020 [22] | NCT03144778 (CIAO trial) | Phase I, randomized, open-label, single institution | Jul. 2017–Feb. 2019 | Newly diagnosed stage II-IVA oropharynx cancer or locoregionally recurrent oropharynx cancer amenable to resection | 14 15 | Two cycles of intravenous D 1500 mg ± T 75 mg on day 1 of a 28-day cycle | D + T did not increase CD8+ TIL density more than D alone |
| Ferris 2020 [23] | NCT02369874 (EAGLE) | Phase III, randomized, open label, multicenter | Nov. 2015–Jul. 2017 | Recurrent or metastatic head and neck squamous cell carcinoma | 246 236 | D (10 mg/kg every 2 weeks (q2w)), D + T (D 20 mg/kg every 4 weeks (q4w) + T 1 mg/kg q4w up to four doses, followed by D 10 mg/kg q2w) | Combining D with T did not show improvement over D activity |
| Goldman 2020 [24] | NCT03043872 (CASPIAN) | Phase III, randomized, open label, multicenter | Mar. 2017–May. 2018 | Treatment-naive, histologically or cytologically documented extensive-stage small-cell lung cancer | 266 265 | Patients in the immunotherapy groups received four cycles of platinum–etoposide + D 1500 mg ± T 75 mg every 3 weeks, followed by maintenance D 1500 mg every 4 weeks. Patients in the D + T + platinum–etoposide group received one additional dose of T 75 mg after platinum–etoposide (up to five doses) | Addition of T to D plus platinum–etoposide did not significantly improve outcomes vs. platinum–etoposide |
| Kelly 2020 [25] | NCT02340975 | Phase 1b/II, randomized, open label, multicenter | Mar. 2015–Jan. 2018 | Metastatic/recurrent gastric or gastroesophageal junction cancer | 71 24 | D 20 mg/kg + T 1 mg/kg Q4W for four cycles, followed by D 10 mg/kg Q2W. Patients in arm B received D monotherapy (10 mg/kg) Q2W | Response rates were low regardless of monotherapy or combination strategies |
| Planchard 2020 [27] | NCT02352948 (ARCTIC) | Phase III, randomized, open label, multicenter | Jan. 2015–Sep. 2016 | Metastatic NSCLC | 173 62 | D + T (12 weeks D 20 mg/kg) + T 1 mg/kg q4w then 34 weeks vs. D 10 mg/kg q2w D (up to 12 months 10 mg/kg every 2 weeks (q2w)) | The efficacy of D + T was comparable with that of D monotherapy, suggesting a limited contribution of T |
| Powles 2020 [30] | NCT02516241 (DANUBE) | Phase III, randomized, open label, multicenter | Nov. 2015–Mar. 2017 | Untreated patients with unresectable, locally advanced, or metastatic urothelial carcinoma | 340 345 | D monotherapy (at a fixed dose of 1500 mg, administered intravenously every 4 weeks); the combination of D (1500 mg) and T (75 mg), both administered intravenously every 4 weeks for up to four doses, followed by D maintenance monotherapy (1500 mg, administered intravenously every 4 weeks) | Combination treatment suggests that T has activity in this disease when given in combination with D, but it also increases toxicity |
| Rezvi 2020 [31] | NCT02453282 (MYSTIC) | Phase III, randomized, open label, multicenter | Jul. 2015–Jun. 2016 | Metastatic NSCLC | 371 369 | D (20 mg/kg every 4 weeks) plus T (1 mg/kg every 4 weeks, up to 4 doses), D (20 mg/kg every 4 weeks) | D + T combination was associated with a higher rate of AEs, leading to discontinuation of D |
| O’Reilly 2019 [26] | NCT02558894 | Phase II, randomized, open label, multicenter | Nov. 2015–Mar. 2017 | Metastatic pancreatic ductal adenocarcinoma | 32 32 | D therapy (1500 mg every 4 weeks) plus T therapy (75 mg every 4 weeks) for 4 cycles followed by D therapy (1500 mg every 4 weeks) or D monotherapy (1500 mg every 4 weeks) for up to 12 months | The observed efficacy of D + T therapy and D monotherapy was reflective of a population of patients with mPDAC who had poor prognoses and rapidly progressing disease |
| Siu 2019 [32] | NCT02319044 (CONDOR) | Phase II, randomized, open label, multicenter | Apr. 2015–Mar. 2016 | Patients with PD-L1–low/negative recurrent or metastatic head and neck squamous cell carcinoma | 133 65 | D (20 mg/kg every 4 weeks) + T (1 mg/kg every 4 weeks) for four cycles, followed by D (10 mg/kg every 2 weeks), or D (10 mg/kg every 2 weeks) monotherapy, or T (10 mg/kg every 4 weeks for seven doses then every 12 weeks for two doses) monotherapy | Minimal observed difference between D and D + T |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fahmy, O.; Ahmed, O.A.A.; Khairul-Asri, M.G.; Alhakamy, N.A.; Alharbi, W.S.; Fahmy, U.A.; El-Moselhy, M.A.; Fresta, C.G.; Caruso, G.; Caraci, F. Adverse Events and Tolerability of Combined Durvalumab and Tremelimumab versus Durvalumab Alone in Solid Cancers: A Systematic Review and Meta-Analysis. Biomedicines 2022, 10, 1101. https://doi.org/10.3390/biomedicines10051101
Fahmy O, Ahmed OAA, Khairul-Asri MG, Alhakamy NA, Alharbi WS, Fahmy UA, El-Moselhy MA, Fresta CG, Caruso G, Caraci F. Adverse Events and Tolerability of Combined Durvalumab and Tremelimumab versus Durvalumab Alone in Solid Cancers: A Systematic Review and Meta-Analysis. Biomedicines. 2022; 10(5):1101. https://doi.org/10.3390/biomedicines10051101
Chicago/Turabian StyleFahmy, Omar, Osama A. A. Ahmed, Mohd Ghani Khairul-Asri, Nabil A. Alhakamy, Waleed S. Alharbi, Usama A. Fahmy, Mohamed A. El-Moselhy, Claudia G. Fresta, Giuseppe Caruso, and Filippo Caraci. 2022. "Adverse Events and Tolerability of Combined Durvalumab and Tremelimumab versus Durvalumab Alone in Solid Cancers: A Systematic Review and Meta-Analysis" Biomedicines 10, no. 5: 1101. https://doi.org/10.3390/biomedicines10051101
APA StyleFahmy, O., Ahmed, O. A. A., Khairul-Asri, M. G., Alhakamy, N. A., Alharbi, W. S., Fahmy, U. A., El-Moselhy, M. A., Fresta, C. G., Caruso, G., & Caraci, F. (2022). Adverse Events and Tolerability of Combined Durvalumab and Tremelimumab versus Durvalumab Alone in Solid Cancers: A Systematic Review and Meta-Analysis. Biomedicines, 10(5), 1101. https://doi.org/10.3390/biomedicines10051101







