CD24 Is a Potential Immunotherapeutic Target for Mantle Cell Lymphoma
Abstract
1. Introduction
2. Materials and Methods
2.1. Reagents
2.2. Cell Lines and Culture Conditions
2.3. Primary Samples
2.4. Isolation of Immune Cells and MCL Blasts
2.5. Flow Cytometry Stainings
2.6. Macrophage-Mediated Phagocytosis Assay
2.7. Flow Cytometry-Based Trogocytosis Assay
2.8. Generation and Validation of F(ab’)2 Fragments
2.9. mRNA and Survival Data Analysis
2.10. Statistical Analysis
3. Results
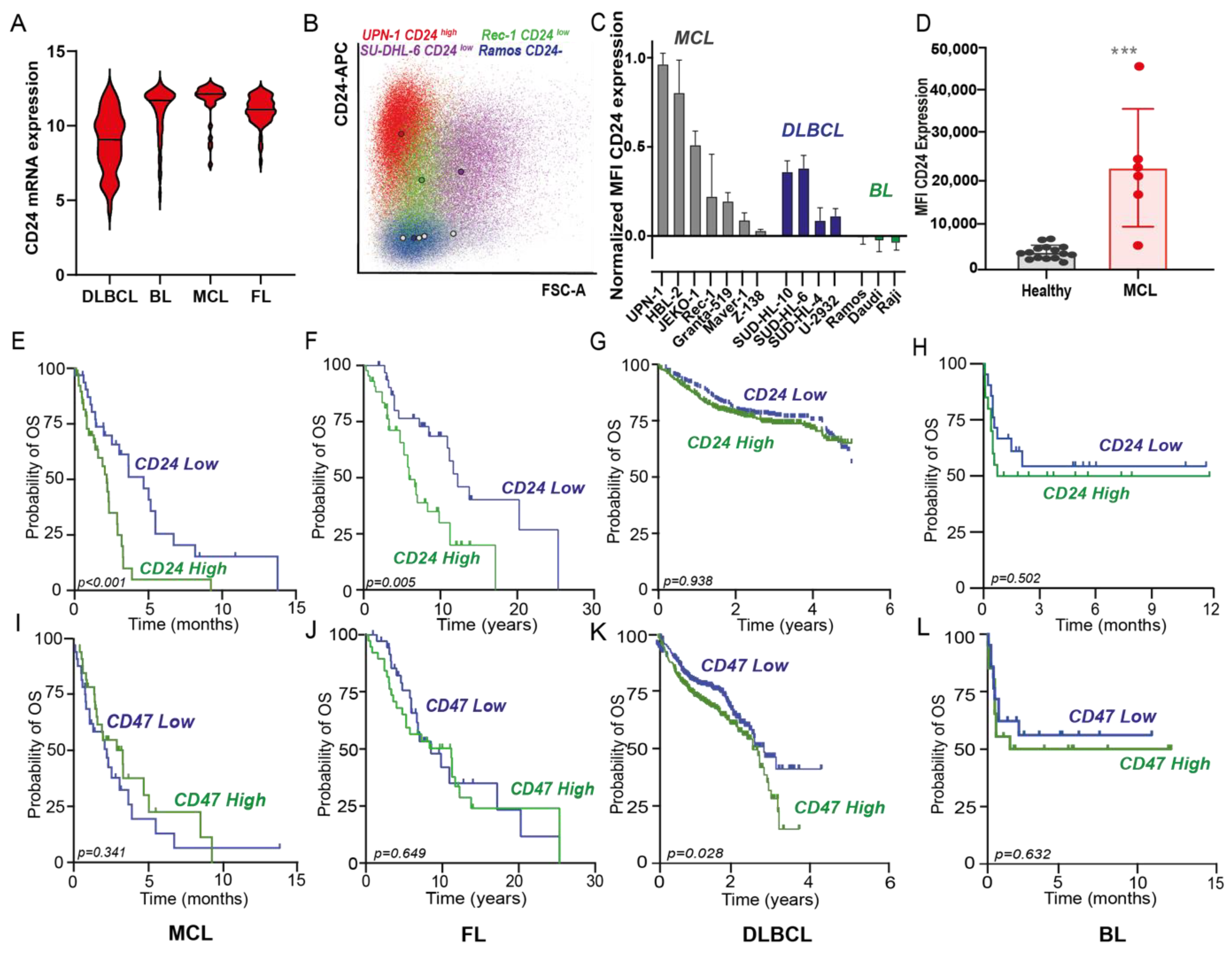
3.1. CD24 Is Expressed in Several B-Cell Lymphomas, Being Most Highly Expressed in MCL, Where It Correlates with Poor Prognosis in Contrast to Hallmark Immune Checkpoint CD47
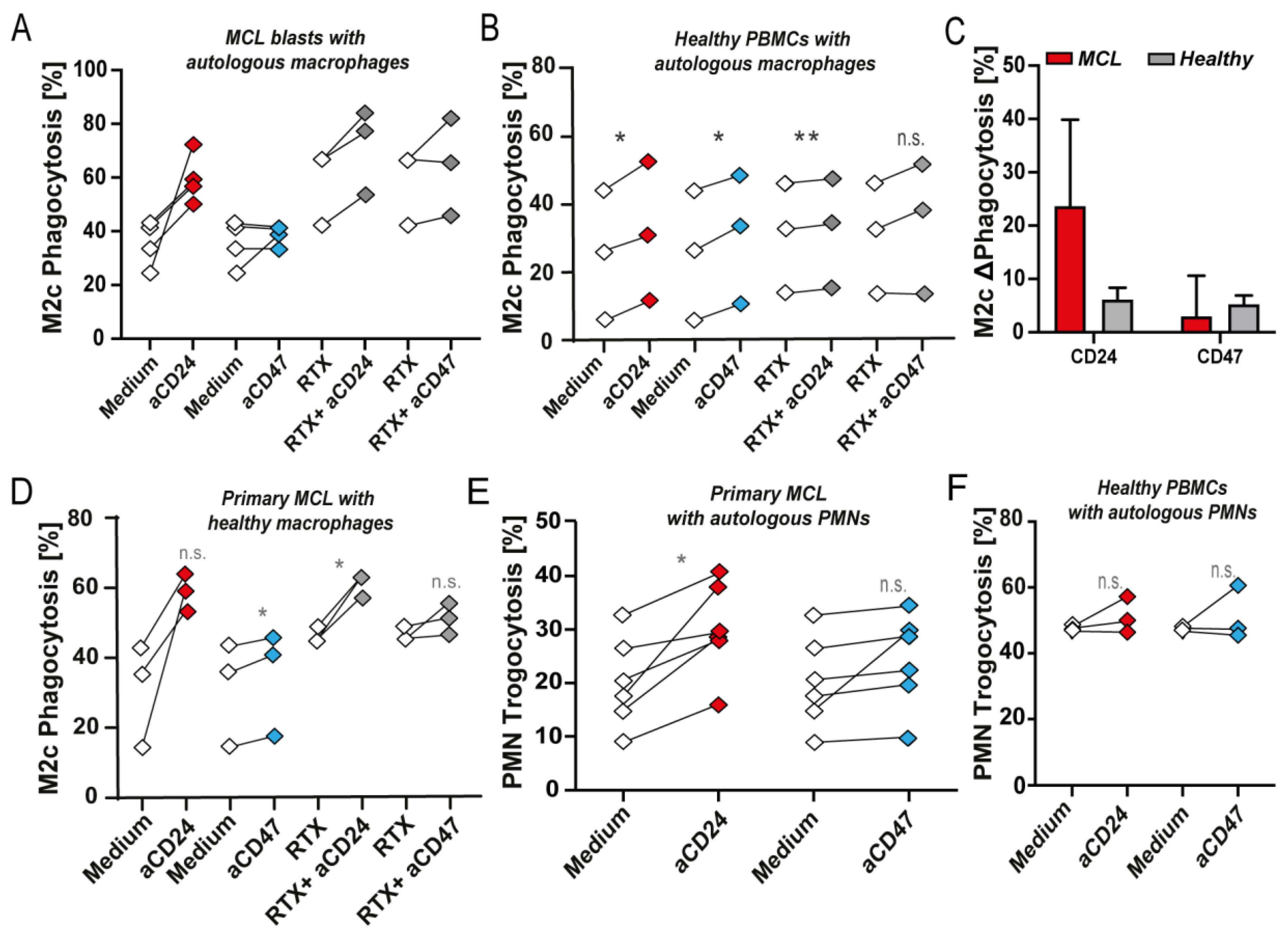
3.2. CD24 Is a Target for Reactivation of Phagocytosis in MCL, with a Superior Effect Than CD47 Antibody Treatment
3.3. CD24 mAb Treatment Increased Phagocytosis of Primary MCL Blasts by Autologous Macrophages and PMNs, but Did Not Induce High Level of Phagocytosis of Healthy Cells
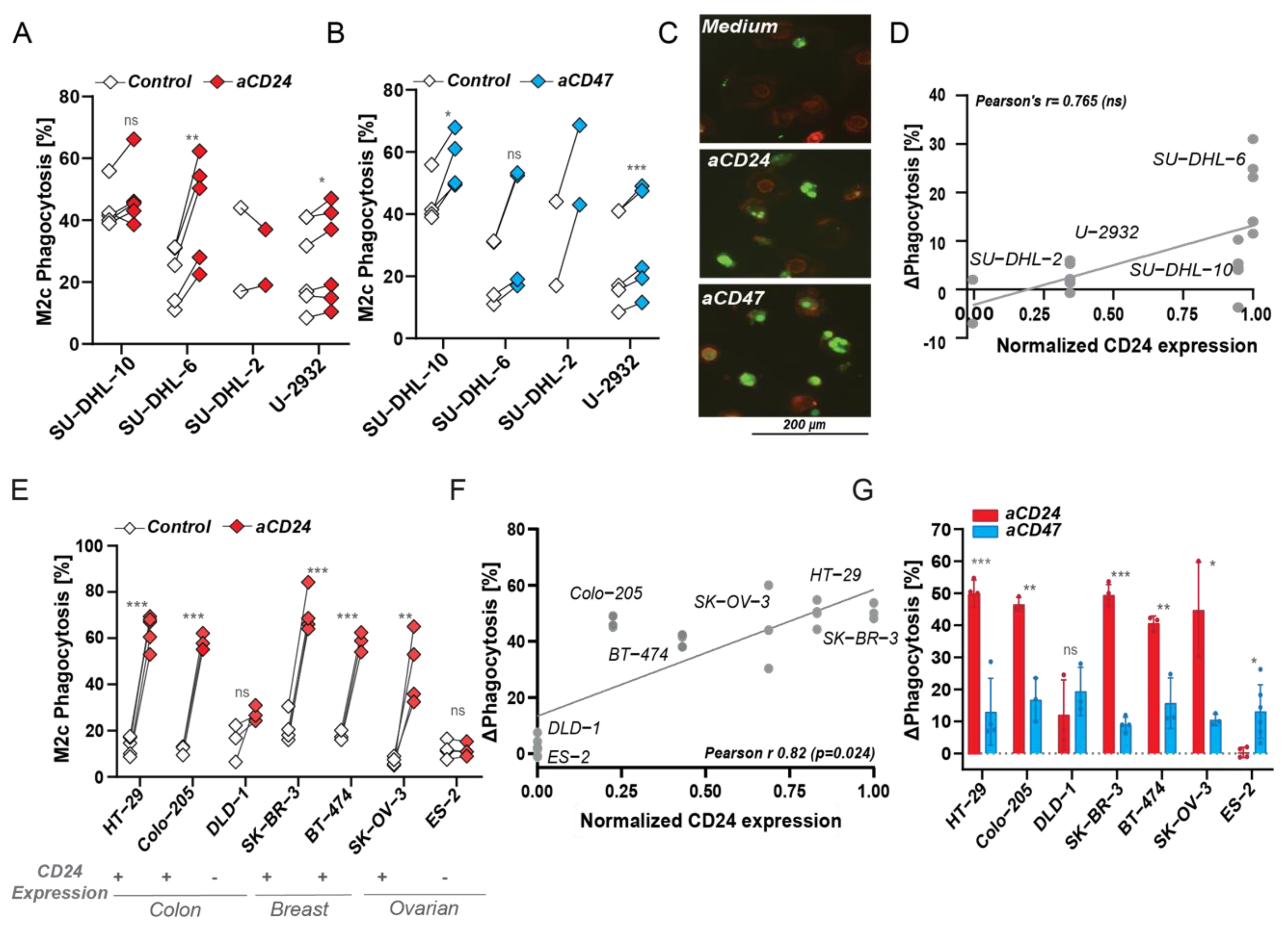
3.4. CD24 Antibody-Mediated Phagocytosis Is Superior to CD47 Checkpoint Targeting in MCL and Carcinoma, but Not in DLBCL
3.5. Induction of Phagocytosis of CD24 Expressing Cells Is Only in Part an Effect of Breaking CD24-Siglec-10 ‘Don’t Eat Me’ Signaling
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Robert, C. A decade of immune-checkpoint inhibitors in cancer therapy. Nat. Commun. 2020, 11, 3801. [Google Scholar] [CrossRef] [PubMed]
- Lentz, R.W.; Colton, M.D.; Mitra, S.S.; Messersmith, W.A. Innate Immune Checkpoint Inhibitors: The Next Breakthrough in Medical Oncology? Mol. Cancer Ther. 2021, 20, 961–974. [Google Scholar] [CrossRef] [PubMed]
- Cendrowicz, E.; Sas, Z.; Bremer, E.; Rygiel, T.P. The Role of Macrophages in Cancer Development and Therapy. Cancers 2021, 13, 1946. [Google Scholar] [CrossRef] [PubMed]
- Tong, B.; Wang, M. CD47 is a novel potent immunotherapy target in human malignancies: Current studies and future promises. Future Oncol. 2018, 14, 2179–2188. [Google Scholar] [CrossRef]
- Cendrowicz, E.; Jacob, L.; Greenwald, S.; Tamir, A.; Pecker, I.; Tabakman, R.; Ghantous, L.; Tamir, L.; Kahn, R.; Avichzer, J.; et al. DSP107 combines inhibition of CD47/SIRPalpha axis with activation of 4-1BB to trigger anti-cancer immunity. J. Exp. Clin. Cancer Res. 2022, 41, 97. [Google Scholar] [CrossRef]
- Jalil, A.R.; Andrechak, J.C.; Discher, D.E. Macrophage checkpoint blockade: Results from initial clinical trials, binding analyses, and CD47-SIRPalpha structure-function. Antib. Ther. 2020, 3, 80–94. [Google Scholar] [CrossRef]
- Sikic, B.I.; Lakhani, N.; Patnaik, A.; Shah, S.A.; Chandana, S.R.; Rasco, D.; Colevas, A.D.; O’Rourke, T.; Narayanan, S.; Papadopoulos, K.; et al. First-in-Human, First-in-Class Phase I Trial of the Anti-CD47 Antibody Hu5F9-G4 in Patients With Advanced Cancers. J. Clin. Oncol. 2019, 37, 946–953. [Google Scholar] [CrossRef]
- Tseng, D.; Volkmer, J.P.; Willingham, S.B.; Contreras-Trujillo, H.; Fathman, J.W.; Fernhoff, N.B.; Seita, J.; Inlay, M.A.; Weiskopf, K.; Miyanishi, M.; et al. Anti-CD47 antibody-mediated phagocytosis of cancer by macrophages primes an effective antitumor T-cell response. Proc. Natl. Acad. Sci. USA 2013, 110, 11103–11108. [Google Scholar] [CrossRef]
- Advani, R.; Flinn, I.; Popplewell, L.; Forero, A.; Bartlett, N.L.; Ghosh, N.; Kline, J.; Roschewski, M.; LaCasce, A.; Collins, G.P.; et al. CD47 Blockade by Hu5F9-G4 and Rituximab in Non-Hodgkin’s Lymphoma. N. Engl. J. Med. 2018, 379, 1711–1721. [Google Scholar] [CrossRef]
- Barkal, A.A.; Weiskopf, K.; Kao, K.S.; Gordon, S.R.; Rosental, B.; Yiu, Y.Y.; George, B.M.; Markovic, M.; Ring, N.G.; Tsai, J.M.; et al. Engagement of MHC class I by the inhibitory receptor LILRB1 suppresses macrophages and is a target of cancer immunotherapy. Nat. Immunol. 2018, 19, 76–84. [Google Scholar] [CrossRef]
- Chen, H.M.; van der Touw, W.; Wang, Y.S.; Kang, K.; Mai, S.; Zhang, J.; Alsina-Beauchamp, D.; Duty, J.A.; Mungamuri, S.K.; Zhang, B.; et al. Blocking immunoinhibitory receptor LILRB2 reprograms tumor-associated myeloid cells and promotes antitumor immunity. J. Clin. Investig. 2018, 128, 5647–5662. [Google Scholar] [CrossRef] [PubMed]
- Yin, S.S.; Gao, F.H. Molecular Mechanism of Tumor Cell Immune Escape Mediated by CD24/Siglec-10. Front. Immunol. 2020, 11, 1324. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Ma, H.; Zhang, J.; Zhu, L.; Wang, C.; Yang, Y. Unraveling the roles of CD44/CD24 and ALDH1 as cancer stem cell markers in tumorigenesis and metastasis. Sci. Rep. 2017, 7, 13856. [Google Scholar] [CrossRef] [PubMed]
- Salaria, S.; Means, A.; Revetta, F.; Idrees, K.; Liu, E.; Shi, C. Expression of CD24, a Stem Cell Marker, in Pancreatic and Small Intestinal Neuroendocrine Tumors. Am. J. Clin. Pathol. 2015, 144, 642–648. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Yang, W.; Kim, D.; Kim, D.K.; Choi, K.U.; Suh, D.S.; Kim, J.H. Therapeutic Strategies for Targeting Ovarian Cancer Stem Cells. Int. J. Mol. Sci. 2021, 22, 5059. [Google Scholar] [CrossRef]
- Overdevest, J.B.; Knubel, K.H.; Duex, J.E.; Thomas, S.; Nitz, M.D.; Harding, M.A.; Smith, S.C.; Frierson, H.F.; Conaway, M.; Theodorescu, D. CD24 expression is important in male urothelial tumorigenesis and metastasis in mice and is androgen regulated. Proc. Natl. Acad. Sci. USA 2012, 109, E3588–E3596. [Google Scholar] [CrossRef]
- Altevogt, P.; Sammar, M.; Huser, L.; Kristiansen, G. Novel insights into the function of CD24: A driving force in cancer. Int. J. Cancer 2021, 148, 546–559. [Google Scholar] [CrossRef]
- Fang, X.; Zheng, P.; Tang, J.; Liu, Y. CD24: From A to Z. Cell Mol. Immunol. 2010, 7, 100–103. [Google Scholar] [CrossRef]
- Barkal, A.A.; Brewer, R.E.; Markovic, M.; Kowarsky, M.; Barkal, S.A.; Zaro, B.W.; Krishnan, V.; Hatakeyama, J.; Dorigo, O.; Barkal, L.J.; et al. CD24 signalling through macrophage Siglec-10 is a target for cancer immunotherapy. Nature 2019, 572, 392–396. [Google Scholar] [CrossRef]
- Kwon, M.J.; Han, J.; Seo, J.H.; Song, K.; Jeong, H.M.; Choi, J.S.; Kim, Y.J.; Lee, S.H.; Choi, Y.L.; Shin, Y.K. CD24 Overexpression Is Associated with Poor Prognosis in Luminal A and Triple-Negative Breast Cancer. PLoS ONE 2015, 10, e0139112. [Google Scholar] [CrossRef]
- Zhang, P.; Zheng, P.; Liu, Y. Amplification of the CD24 Gene Is an Independent Predictor for Poor Prognosis of Breast Cancer. Front. Genet. 2019, 10, 560. [Google Scholar] [CrossRef] [PubMed]
- Tarhriz, V.; Bandehpour, M.; Dastmalchi, S.; Ouladsahebmadarek, E.; Zarredar, H.; Eyvazi, S. Overview of CD24 as a new molecular marker in ovarian cancer. J. Cell Physiol. 2019, 234, 2134–2142. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.L.; Guo, C.R.; Su, W.Y.; Chen, Y.X.; Xu, J.; Fang, J.Y. CD24 Overexpression Related to Lymph Node Invasion and Poor Prognosis of Colorectal Cancer. Clin. Lab. 2018, 64, 497–505. [Google Scholar] [CrossRef] [PubMed]
- Kay, R.; Rosten, P.M.; Humphries, R.K. CD24, a signal transducer modulating B cell activation responses, is a very short peptide with a glycosyl phosphatidylinositol membrane anchor. J. Immunol. 1991, 147, 1412–1416. [Google Scholar] [PubMed]
- Lingwood, D.; Simons, K. Lipid rafts as a membrane-organizing principle. Science 2010, 327, 46–50. [Google Scholar] [CrossRef] [PubMed]
- Aigner, S.; Ruppert, M.; Hubbe, M.; Sammar, M.; Sthoeger, Z.; Butcher, E.C.; Vestweber, D.; Altevogt, P. Heat stable antigen (mouse CD24) supports myeloid cell binding to endothelial and platelet P-selectin. Int. Immunol. 1995, 7, 1557–1565. [Google Scholar] [CrossRef]
- Myung, J.H.; Gajjar, K.A.; Pearson, R.M.; Launiere, C.A.; Eddington, D.T.; Hong, S. Direct measurements on CD24-mediated rolling of human breast cancer MCF-7 cells on E-selectin. Anal. Chem. 2011, 83, 1078–1083. [Google Scholar] [CrossRef]
- Kristiansen, G.; Machado, E.; Bretz, N.; Rupp, C.; Winzer, K.J.; Konig, A.K.; Moldenhauer, G.; Marme, F.; Costa, J.; Altevogt, P. Molecular and clinical dissection of CD24 antibody specificity by a comprehensive comparative analysis. Lab. Investig. 2010, 90, 1102–1116. [Google Scholar] [CrossRef]
- Zhou, Q.; Wu, Y.; Nielsen, P.J.; Liu, Y. Homotypic interaction of the heat-stable antigen is not responsible for its co-stimulatory activity for T cell clonal expansion. Eur. J. Immunol. 1997, 27, 2524–2528. [Google Scholar] [CrossRef]
- Suzuki, T.; Kiyokawa, N.; Taguchi, T.; Sekino, T.; Katagiri, Y.U.; Fujimoto, J. CD24 induces apoptosis in human B cells via the glycolipid-enriched membrane domains/rafts-mediated signaling system. J. Immunol. 2001, 166, 5567–5577. [Google Scholar] [CrossRef]
- Mensah, F.F.K.; Armstrong, C.W.; Reddy, V.; Bansal, A.S.; Berkovitz, S.; Leandro, M.J.; Cambridge, G. CD24 Expression and B Cell Maturation Shows a Novel Link With Energy Metabolism: Potential Implications for Patients With Myalgic Encephalomyelitis/Chronic Fatigue Syndrome. Front. Immunol. 2018, 9, 2421. [Google Scholar] [CrossRef] [PubMed]
- Shapira, S.; Kazanov, D.; Mdah, F.; Yaakobi, H.; Herishanu, Y.; Perry, C.; Avivi, I.; Itchaki, G.; Shacham-Abulafia, A.; Raanani, P.; et al. Feasibly of CD24/CD11b as a Screening Test for Hematological Malignancies. J. Pers. Med. 2021, 11, 724. [Google Scholar] [CrossRef] [PubMed]
- Lancaster-Shorts, K.; Pirruccello, S. CD24 Expression in Follicular Lymphoma: An Alternative B-Cell Marker in Therapy Selected, Recurrent Lymphoma. Grad. Med. Educ. Res. J. 2020, 2, 65. [Google Scholar] [CrossRef]
- Jain, P.; Wang, M. Mantle cell lymphoma: 2019 update on the diagnosis, pathogenesis, prognostication, and management. Am. J. Hematol. 2019, 94, 710–725. [Google Scholar] [CrossRef] [PubMed]
- Maddocks, K. Update on mantle cell lymphoma. Blood 2018, 132, 1647–1656. [Google Scholar] [CrossRef] [PubMed]
- Hanel, W.; Epperla, N. Emerging therapies in mantle cell lymphoma. J. Hematol. Oncol. 2020, 13, 79. [Google Scholar] [CrossRef] [PubMed]
- Susanibar-Adaniya, S.; Barta, S.K. 2021 Update on Diffuse large B cell lymphoma: A review of current data and potential applications on risk stratification and management. Am. J. Hematol. 2021, 96, 617–629. [Google Scholar] [CrossRef] [PubMed]
- Qiao, L.Y.; Li, H.B.; Zhang, Y.; Shen, D.; Liu, P.; Che, Y.Q. CD24 Contributes to Treatment Effect in ABC-DLBCL Patients with R-CHOP Resistance. Pharmgenom. Pers. Med. 2021, 14, 591–599. [Google Scholar] [CrossRef]
- Ma, M.C.J.; Tadros, S.; Bouska, A.; Heavican, T.; Yang, H.; Deng, Q.; Moore, D.; Akhter, A.; Hartert, K.; Jain, N.; et al. Subtype-specific and co-occurring genetic alterations in B-cell non-Hodgkin lymphoma. Haematologica 2022, 107, 690–701. [Google Scholar] [CrossRef]
- Blenk, S.; Engelmann, J.C.; Pinkert, S.; Weniger, M.; Schultz, J.; Rosenwald, A.; Muller-Hermelink, H.K.; Muller, T.; Dandekar, T. Explorative data analysis of MCL reveals gene expression networks implicated in survival and prognosis supported by explorative CGH analysis. BMC Cancer 2008, 8, 106. [Google Scholar] [CrossRef]
- Glas, A.M.; Kersten, M.J.; Delahaye, L.J.; Witteveen, A.T.; Kibbelaar, R.E.; Velds, A.; Wessels, L.F.; Joosten, P.; Kerkhoven, R.M.; Bernards, R.; et al. Gene expression profiling in follicular lymphoma to assess clinical aggressiveness and to guide the choice of treatment. Blood 2005, 105, 301–307. [Google Scholar] [CrossRef] [PubMed]
- Dave, S.S.; Fu, K.; Wright, G.W.; Lam, L.T.; Kluin, P.; Boerma, E.J.; Greiner, T.C.; Weisenburger, D.D.; Rosenwald, A.; Ott, G.; et al. Molecular diagnosis of Burkitt’s lymphoma. N. Engl. J. Med. 2006, 354, 2431–2442. [Google Scholar] [CrossRef] [PubMed]
- Bouwstra, R.; He, Y.; de Boer, J.; Kooistra, H.; Cendrowicz, E.; Fehrmann, R.S.N.; Ammatuna, E.; Zu Eulenburg, C.; Nijland, M.; Huls, G.; et al. CD47 Expression Defines Efficacy of Rituximab with CHOP in Non-Germinal Center B-cell (Non-GCB) Diffuse Large B-cell Lymphoma Patients (DLBCL), but Not in GCB DLBCL. Cancer Immunol. Res. 2019, 7, 1663–1671. [Google Scholar] [CrossRef] [PubMed]
- Fadok, V.A.; de Cathelineau, A.; Daleke, D.L.; Henson, P.M.; Bratton, D.L. Loss of phospholipid asymmetry and surface exposure of phosphatidylserine is required for phagocytosis of apoptotic cells by macrophages and fibroblasts. J. Biol. Chem. 2001, 276, 1071–1077. [Google Scholar] [CrossRef] [PubMed]
- VanDerMeid, K.R.; Elliott, M.R.; Baran, A.M.; Barr, P.M.; Chu, C.C.; Zent, C.S. Cellular Cytotoxicity of Next-Generation CD20 Monoclonal Antibodies. Cancer Immunol. Res. 2018, 6, 1150–1160. [Google Scholar] [CrossRef] [PubMed]
- Invivogen. <review-ImmunoglobulinG-invivogen.pdf>. 2011. Available online: https://www.invivogen.com/review-antibody-generation (accessed on 29 April 2022).
- Waldmann, T.A. Monoclonal antibodies in diagnosis and therapy. Science 1991, 252, 1657–1662. [Google Scholar] [CrossRef] [PubMed]
- Benkerrou, M.; Jais, J.P.; Leblond, V.; Durandy, A.; Sutton, L.; Bordigoni, P.; Garnier, J.L.; Le Bidois, J.; Le Deist, F.; Blanche, S.; et al. Anti-B-cell monoclonal antibody treatment of severe posttransplant B-lymphoproliferative disorder: Prognostic factors and long-term outcome. Blood 1998, 92, 3137–3147. [Google Scholar] [CrossRef]
- Chen, G.Y.; Tang, J.; Zheng, P.; Liu, Y. CD24 and Siglec-10 selectively repress tissue damage-induced immune responses. Science 2009, 323, 1722–1725. [Google Scholar] [CrossRef]
- Forgione, R.E.; Di Carluccio, C.; Guzman-Caldentey, J.; Gaglione, R.; Battista, F.; Chiodo, F.; Manabe, Y.; Arciello, A.; Del Vecchio, P.; Fukase, K.; et al. Unveiling Molecular Recognition of Sialoglycans by Human Siglec-10. iScience 2020, 23, 101231. [Google Scholar] [CrossRef]
- Xiao, N.; Zhu, X.; Li, K.; Chen, Y.; Liu, X.; Xu, B.; Lei, M.; Xu, J.; Sun, H.C. Blocking siglec-10(hi) tumor-associated macrophages improves anti-tumor immunity and enhances immunotherapy for hepatocellular carcinoma. Exp. Hematol. Oncol. 2021, 10, 36. [Google Scholar] [CrossRef]
- Smith, S.C.; Oxford, G.; Wu, Z.; Nitz, M.D.; Conaway, M.; Frierson, H.F.; Hampton, G.; Theodorescu, D. The metastasis-associated gene CD24 is regulated by Ral GTPase and is a mediator of cell proliferation and survival in human cancer. Cancer Res. 2006, 66, 1917–1922. [Google Scholar] [CrossRef] [PubMed]
- Nielsen, P.J.; Lorenz, B.; Muller, A.M.; Wenger, R.H.; Brombacher, F.; Simon, M.; von der Weid, T.; Langhorne, W.J.; Mossmann, H.; Kohler, G. Altered erythrocytes and a leaky block in B-cell development in CD24/HSA-deficient mice. Blood 1997, 89, 1058–1067. [Google Scholar] [CrossRef] [PubMed]
- Ayre, D.C.; Christian, S.L. CD24: A Rheostat That Modulates Cell Surface Receptor Signaling of Diverse Receptors. Front. Cell Dev. Biol. 2016, 4, 146. [Google Scholar] [CrossRef] [PubMed]
- Pallegar, N.K.; Ayre, D.C.; Christian, S.L. Repression of CD24 surface protein expression by oncogenic Ras is relieved by inhibition of Raf but not MEK or PI3K. Front. Cell Dev. Biol. 2015, 3, 47. [Google Scholar] [CrossRef]
- Bretz, N.P.; Salnikov, A.V.; Perne, C.; Keller, S.; Wang, X.; Mierke, C.T.; Fogel, M.; Erbe-Hofmann, N.; Schlange, T.; Moldenhauer, G.; et al. CD24 controls Src/STAT3 activity in human tumors. Cell Mol. Life Sci. 2012, 69, 3863–3879. [Google Scholar] [CrossRef]
- Lan, H.; Li, W.; Jiang, H.; Yang, Y.; Zheng, X. Intracellular signaling transduction pathways triggered by a well-known anti-GHR monoclonal antibody, Mab263, in vitro and in vivo. Int. J. Mol. Sci. 2014, 15, 20538–20554. [Google Scholar] [CrossRef]
- Tchernychev, B.; Furie, B.; Furie, B.C. Peritoneal macrophages express both P-selectin and PSGL-1. J. Cell Biol. 2003, 163, 1145–1155. [Google Scholar] [CrossRef]
- Finas, D.; Huszar, M.; Agic, A.; Dogan, S.; Kiefel, H.; Riedle, S.; Gast, D.; Marcovich, R.; Noack, F.; Altevogt, P.; et al. L1 cell adhesion molecule (L1CAM) as a pathogenetic factor in endometriosis. Hum. Reprod. 2008, 23, 1053–1062. [Google Scholar] [CrossRef]
- Went, P.; Dellas, T.; Bourgau, C.; Maurer, R.; Augustin, F.; Tzankov, A.; Dirnhofer, S. [Expression profile and prognostic significance of CD24, p53 and p21 in lymphomas. A tissue microarray study of over 600 non-Hodgkin lymphomas]. Dtsch. Med. Wochenschr. 2004, 129, 2094–2099. [Google Scholar] [CrossRef]
- Yao, Y.; Xu, X.H.; Jin, L. Macrophage Polarization in Physiological and Pathological Pregnancy. Front. Immunol. 2019, 10, 792. [Google Scholar] [CrossRef]
- Porta, C.; Riboldi, E.; Ippolito, A.; Sica, A. Molecular and epigenetic basis of macrophage polarized activation. Semin. Immunol. 2015, 27, 237–248. [Google Scholar] [CrossRef] [PubMed]
- Iribarren, K.; Buque, A.; Mondragon, L.; Xie, W.; Levesque, S.; Pol, J.; Zitvogel, L.; Kepp, O.; Kroemer, G. Anticancer effects of anti-CD47 immunotherapy in vivo. Oncoimmunology 2019, 8, 1550619. [Google Scholar] [CrossRef] [PubMed]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Freile, J.Á.; Ustyanovska Avtenyuk, N.; Corrales, M.G.; Lourens, H.J.; Huls, G.; van Meerten, T.; Cendrowicz, E.; Bremer, E. CD24 Is a Potential Immunotherapeutic Target for Mantle Cell Lymphoma. Biomedicines 2022, 10, 1175. https://doi.org/10.3390/biomedicines10051175
Freile JÁ, Ustyanovska Avtenyuk N, Corrales MG, Lourens HJ, Huls G, van Meerten T, Cendrowicz E, Bremer E. CD24 Is a Potential Immunotherapeutic Target for Mantle Cell Lymphoma. Biomedicines. 2022; 10(5):1175. https://doi.org/10.3390/biomedicines10051175
Chicago/Turabian StyleFreile, Jimena Álvarez, Natasha Ustyanovska Avtenyuk, Macarena González Corrales, Harm Jan Lourens, Gerwin Huls, Tom van Meerten, Ewa Cendrowicz, and Edwin Bremer. 2022. "CD24 Is a Potential Immunotherapeutic Target for Mantle Cell Lymphoma" Biomedicines 10, no. 5: 1175. https://doi.org/10.3390/biomedicines10051175
APA StyleFreile, J. Á., Ustyanovska Avtenyuk, N., Corrales, M. G., Lourens, H. J., Huls, G., van Meerten, T., Cendrowicz, E., & Bremer, E. (2022). CD24 Is a Potential Immunotherapeutic Target for Mantle Cell Lymphoma. Biomedicines, 10(5), 1175. https://doi.org/10.3390/biomedicines10051175







