Clinical Data on Canabinoids: Translational Research in the Treatment of Autism Spectrum Disorders
Abstract
:1. Introduction
2. Materials and Methods
3. Results
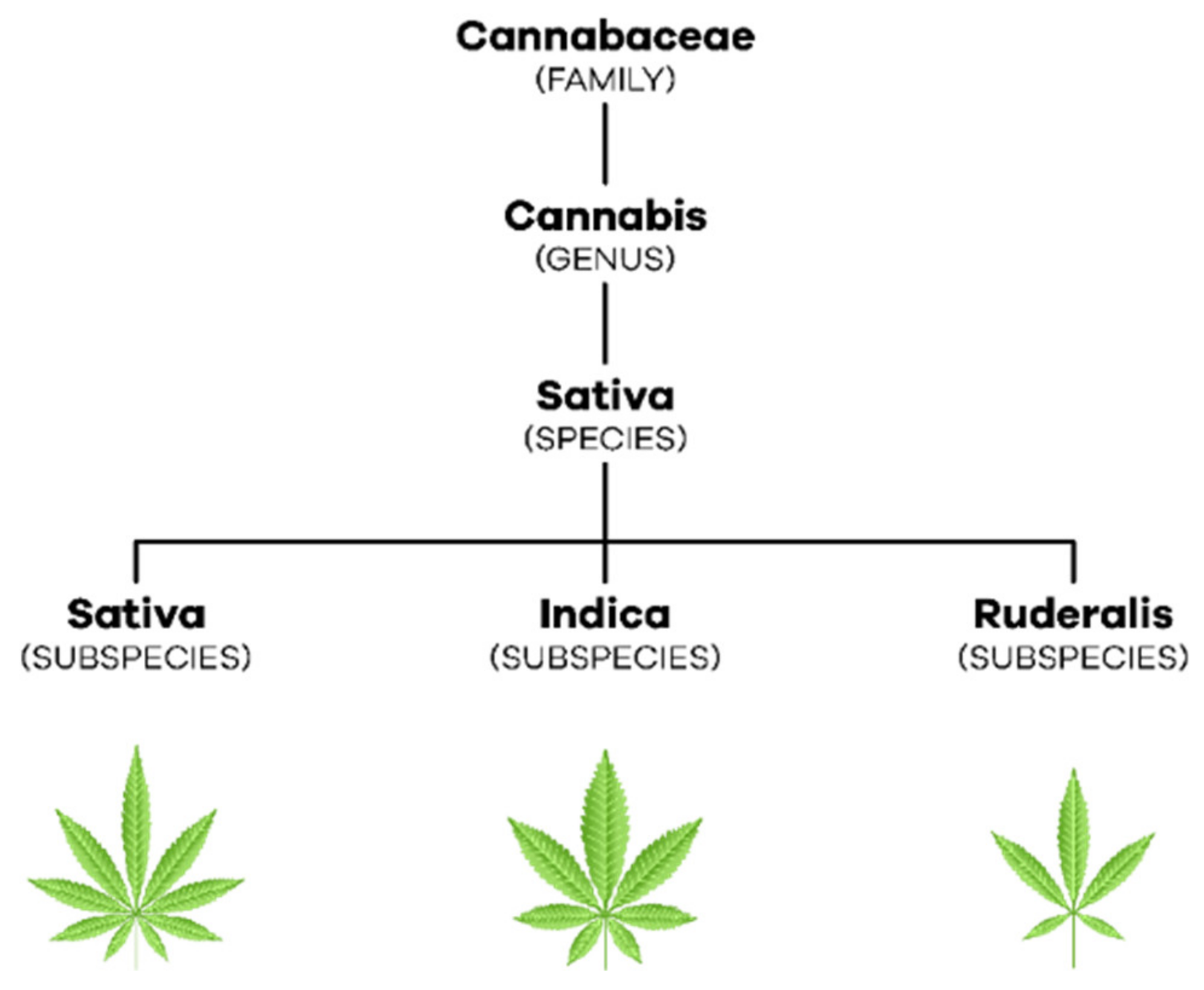
3.1. Cannabis, Flos
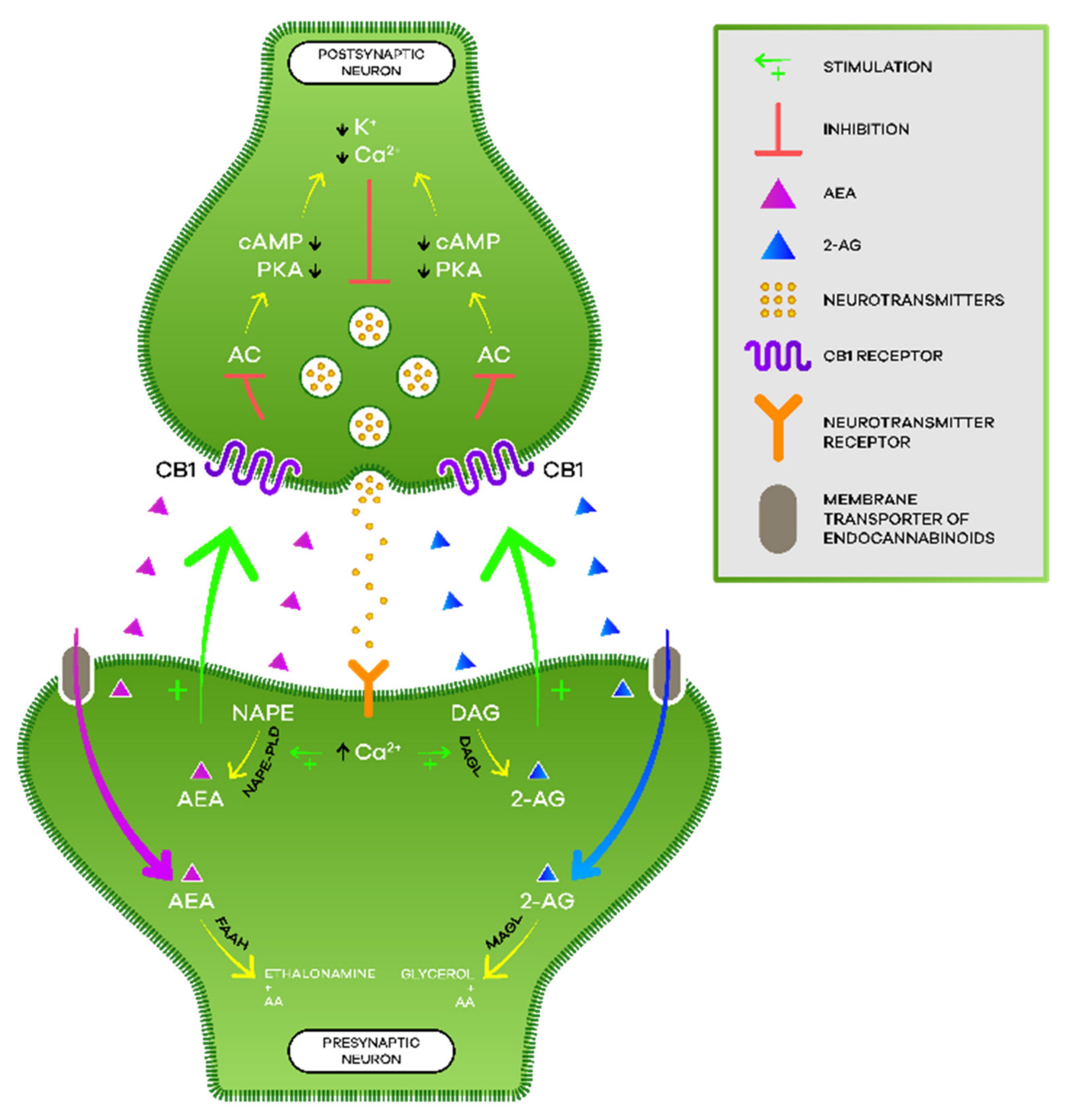
3.2. Endocannabinoid System
3.2.1. Cannabinoid Receptors
3.2.2. Endocannabinoids
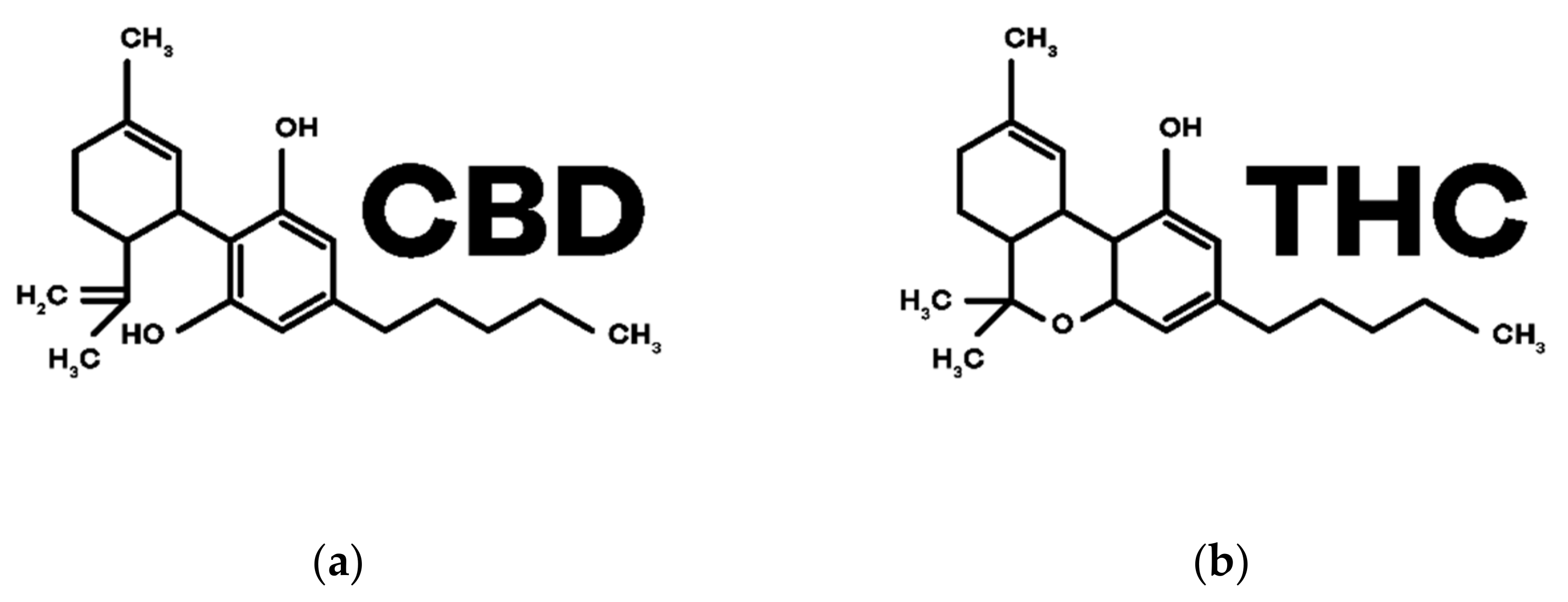
3.2.3. Phytocannabinoids
3.3. Physiological and Therapeutic Effects of Cannabinoids
3.4. Autism Spectrum Disorder
3.4.1. Etiology
3.4.2. Treatment
3.5. Application of the Cannabinoid Therapy in Autism Spectrum Disorders
3.5.1. Preliminary Clinical Studies
3.5.2. Clinical Trials
3.6. Adverse Effects of Cannabis
3.7. Approved Drugs
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
Abbreviations
| 2-AG | 2-Arachidonylglycerol; |
| 5-HT | Serotonin; |
| AA | Arachidonic Acid; |
| AC | Adenylate Cyclase; |
| ADHD | Attention Deficit Hyperactivity Disorder; |
| AEA | N-Arachidonoylethanolamide; |
| APSI | Autism Parenting Stress Index; |
| ASD | Autism Spectrum Disorder; |
| ATP | Adenosine Triphosphate; |
| cAMP | Cyclic Adenosine Monophosphate; |
| CBD | Cannabidiol; |
| CBDV | Cannabidivarin; |
| CGI-I | Clinical Global Impression—Improvement; |
| CNP | Peripheral Nervous System; |
| CNS | Central Nervous System; |
| CNVs | Copy Number Variations; |
| CVS | Cardiovascular System; |
| DAG | Diacylglycerol; |
| DAGL | Diacylglycerol Lipase; |
| EU | European Union; |
| FAAH | Fatty Acid Amide Hydrolase; |
| fALFF | Fractional Amplitude of Low-Frequency Fluctuations; |
| FC | Functional Brain Connectivity; |
| FDA | Food and Drug Administration; |
| GABA | Gamma-Aminobutyric Acid; |
| GPCRs | Orphan G Protein-Coupled Receptors; |
| HSQ-ASD | Home Situations Questionnaire—Autism Spectrum Disorder; |
| MAGL | Monoacylglycerol Lipase; |
| MAPK | Mitogen-Activated Protein Kinase; |
| NAPE | N-Acyl-Phosphatidylethanolamine; |
| NAPE-PLD | N-Acyl-Phosphatidylethanolamine-Specific Phospholipase D; |
| PKA | Protein Kinase A; |
| PPARγs | Nuclear Receptors Activated by Peroxisome Proliferators Type γ; |
| Receptor CB1 | Cannabinoid Receptor Type 1; |
| Receptor CB2 | Cannabinoid Receptor Type 2; |
| SNPs | Single Nucleotide Polymorphisms; |
| SRS-2 | Social Responsiveness Scale—Second Edition; |
| THC | ∆9-Tetrahydrocannabinol; |
| Trp | Tryptophan; |
| TRPV1s | Transient Receptors Potential Cation Channel Subfamily V Member 1. |
References
- Klumpers, L.E.; Thacker, D.L. A brief background on cannabis: From plant to medical indications. J. AOAC Int. 2019, 102, 412–420. [Google Scholar] [CrossRef] [PubMed]
- Pollio, A. The Name of Cannabis: A Short Guide for Nonbotanists. Cannabis Cannabinoid Res. 2016, 1, 234–238. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bonini, S.A.; Premoli, M.; Tambaro, S.; Kumar, A.; Maccarinelli, G.; Memo, M.; Mastinu, A. Cannabis sativa: A comprehensive ethnopharmacological review of a medicinal plant with a long history. J. Ethnopharmacol. 2018, 227, 300–315. [Google Scholar] [CrossRef] [PubMed]
- Poleg, S.; Golubchik, P.; Offen, D.; Weizman, A. Cannabidiol as a suggested candidate for treatment of autism spectrum disorder. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 2018, 8, 90–96. [Google Scholar] [CrossRef]
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, 5th ed.; American Psychiatric Association: Arlington, VA, USA, 2013; pp. 50–59. [Google Scholar]
- Lord, C.; Elsabbagh, M.; Baird, G.; Veenstra-Vanderweele, J. Autism spectrum disorder. Lancet 2018, 392, 508–520. [Google Scholar] [CrossRef]
- Young, N.; Findling, R.L. An update on pharmacotherapy for autism spectrum disorder in children and adolescents. Curr. Opin. Psychiatry 2015, 28, 91–101. [Google Scholar] [CrossRef]
- Sharma, S.R.; Gonda, X.; Tarazi, F.I. Autism Spectrum Disorder: Classification, diagnosis and therapy. Pharmacol. Ther. 2018, 190, 91–104. [Google Scholar] [CrossRef]
- Loss, C.M.; Teodoro, L.; Rodrigues, G.D.; Moreira, L.R.; Peres, F.F.; Zuardi, A.W.; Crippa, J.A.; Hallak, J.E.C.; Abílio, V.C. Is Cannabidiol during Neurodevelopment a Promising Therapy for Schizophrenia and Autism Spectrum Disorders? Front. Pharmacol. 2021, 11, 2461. [Google Scholar] [CrossRef]
- Bundesinstitut für Arzneimittel und Medizinprodukte. German Pharmacopoeia, Cannabis, Flor–Monograph. Cannabisblüten Cannabis Flos; BAnz AT 24.04.2018 B5; Bekanntmachung einer Mitteilung zum Deutschen Arzneibuch; Bundesinstitut für Arzneimittel und Medizinprodukte: Bonn, Germany, 2018.
- Mcpartland, J.M. Cannabis Systematics at the Levels of Family, Genus, and Species. Cannabis Cannabinoid Res. 2018, 3, 203–212. [Google Scholar] [CrossRef] [Green Version]
- Kinghorn, A.D.; Falk, H.; Gibbons, S.; Kobayashi, J. 103-Phytocannabinoids-Unraveling the Complex Chemistry and Pharmacology of Cannabis sativa. In Progress in the Chemistry of Organic Natural Products; Springer Nature: Cham, Switzerland, 2017. [Google Scholar]
- Gould, J. The Cannabis Crop. Nature 2015, 525, S2–S3. [Google Scholar] [CrossRef] [Green Version]
- European Medicines Agency; Herbal Medicinal Products Committee. Guideline on Good Agricultural and Collection Practice (GACP) for Starting Materials of Herbal Origin. 2006. Available online: https://www.ema.europa.eu/en/good-agricultural-collection-practice-starting-materials-herbal-origin (accessed on 17 January 2022).
- Maroon, J.; Bost, J. Review of the neurological benefits of phytocannabinoids. Surg. Neurol. Int. 2018, 9, 91. [Google Scholar] [CrossRef] [PubMed]
- Lu, D.; Potter, D.E. Cannabinoids and the Cannabinoid Receptors: An Overview. In Handbook of Cannabis and Related Pathologies: Biology, Pharmacology, Diagnosis, and Treatment; Elsevier Inc.: Amsterdam, The Netherlands, 2017. [Google Scholar]
- Zou, S.; Kumar, U. Cannabinoid receptors and the endocannabinoid system: Signaling and function in the central nervous system. Int. J. Mol. Sci. 2018, 19, 833. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Brigida, A.L.; Schultz, S.; Cascone, M.; Antonucci, N.; Siniscalco, D. Endocannabinod signal dysregulation in autism spectrum disorders: A correlation link between inflammatory state and Neuro-Immune alterations. Int. J. Mol. Sci. 2017, 18, 1425. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Araujo, D.J.; Tjoa, K.; Saijo, K. The Endocannabinoid System as a Window into Microglial Biology and Its Relationship to Autism. Front. Cell. Neurosci. 2019, 13, 424. [Google Scholar] [CrossRef]
- Cohen, K.; Weizman, A.; Weinstein, A. Positive and Negative Effects of Cannabis and Cannabinoids on Health. Clin. Pharmacol. Ther. 2019, 105, 1139–1147. [Google Scholar] [CrossRef]
- Loprinzi, P.D.; Zou, L.; Li, H. The endocannabinoid system as a potential mechanism through which exercise influences episodic memory function. Brain Sci. 2019, 9, 112. [Google Scholar] [CrossRef] [Green Version]
- Chonhofen, P.; Bristot, I.J.; Crippa, J.A.; Hallak, J.E.C.; Zuardi, A.W.; Parsons, R.B.; Klamt, F. Cannabinoid-Based Therapies and Brain Development: Potential Harmful Effect of Early Modulation of the Endocannabinoid System. CNS Drugs 2018, 32, 697–712. [Google Scholar] [CrossRef] [Green Version]
- Aran, A.; Cayam-Rand, D. Medical cannabis in children. Rambam Maimonides Med. J. 2020, 11, e0003. [Google Scholar] [CrossRef] [Green Version]
- Appendino, G. The early history of cannabinoid research. Rend. Lincei 2020, 31, 919–929. [Google Scholar] [CrossRef]
- Pisanti, S.; Malfitno, M.; Ciaglia, E.; Ranieri, R.; Cuomo, G.; Abate, M.; Faggiana, G.; Proto, M.C.; Fiore, D.; Laezza, C.; et al. Cannabidiol: State of the art and new challenges for therapeutic applications. Pharmacol. Ther. 2017, 175, 133–150. [Google Scholar] [CrossRef]
- Devinsky, O.; Cilio, M.R.; Cross, H.; Fernandez-Ruiz, J.; French, J.; Hill, C.; Katz, R.; Marzo, V.D.; Jutras-Aswad, D.; Notcutt, W.G.; et al. Cannabidiol: Pharmacology and potential therapeutic role in epilepsy and other neuropsychiatric disorders. Epilepsia 2014, 55, 791–802. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gu, B. Cannabidiol provides viable treatment opportunity for multiple neurological pathologies of autism spectrum disorder. Glob. Drugs Ther. 2017, 2, 1–4. [Google Scholar] [CrossRef] [Green Version]
- Fraguas-Sánchez, A.I.; Torres-Suárez, A.I. Medical Use of Cannabinoids. Drugs 2018, 78, 1665–1703. [Google Scholar] [CrossRef] [PubMed]
- White, C.M. A Review of Human Studies Assessing Cannabidiol’s (CBD) Therapeutic Actions and Potential. J. Clin. Pharmacol. 2019, 59, 923–934. [Google Scholar] [CrossRef] [PubMed]
- Bridgeman, M.B.; Abazia, D.T. Medicinal Cannabis: History, Pharmacology, and Implications for the Acute Care Setting. Pharm. Ther. 2017, 42, 180–188. [Google Scholar]
- Stasiłowicz, A.; Tomala, A.; Podolak, I.; Cielecka-Piontek, J. Cannabis sativa L. As a natural drug meeting the criteria of a multitarget approach to treatment. Int. J. Mol. Sci. 2021, 22, 778. [Google Scholar] [CrossRef]
- Breijyeh, Z.; Jubeh, B.; Bufo, S.A.; Karaman, R.; Scrano, L. Cannabis: A Toxin-Producing Plant with Potential Therapeutic Uses. Toxins 2021, 13, 117. [Google Scholar] [CrossRef]
- Oberbarnscheidt, T.; Miller, N.S. The Impact of Cannabidiol on Psychiatric and Medical Conditions. J. Clin. Med. Res. 2020, 12, 393–403. [Google Scholar] [CrossRef]
- Goyal, H.; Singla, U.; Gupta, U.; May, E. Role of cannabis in digestive disorders. Eur. J. Gastroenterol. Hepatol. 2017, 29, 135–142. [Google Scholar] [CrossRef]
- Babson, K.A.; Sottile, J.; Morabito, D. Cannabis, Cannabinoids, and Sleep: A Review of the Literature. Curr. Psychiatry Rep. 2017, 19, 23. [Google Scholar] [CrossRef]
- Robertson, C.E.; Baron-Cohen, S. Sensory perception in autism. Nat. Rev. Neurosci. 2017, 18, 671–684. [Google Scholar] [CrossRef] [PubMed]
- Chung, S.; Son, J.-W. Visual Perception in Autism Spectrum Disorder: A Review of Neuroimaging Studies. J. Korean Acad. Child Adolesc. Psychiatry 2020, 31, 105–120. [Google Scholar] [CrossRef] [PubMed]
- Mukherjee, S.B. Autism Spectrum Disorders—Diagnosis and Management. Indian J. Pediatrics 2017, 84, 307–314. [Google Scholar] [CrossRef] [PubMed]
- Pretzsch, C.M.; Freyberg, J.; Voinescu, B.; Lythgoe, D.; Horder, J.; Mendez, M.A.; Wichers, R.; Ajram, L.; Ivin, G.; Heasman, M.; et al. Effects of cannabidiol on brain excitation and inhibition systems; a randomised placebo-controlled single dose trial during magnetic resonance spectroscopy in adults with and without autism spectrum disorder. Neuropsychopharmacology 2019, 44, 1398–1405. [Google Scholar] [CrossRef] [Green Version]
- Pretzsch, C.M.; Freyberg, J.; Voinescu, B.; Lythgoe, D.; Horder, J.; Mendez, M.A.; Wichers, R.; Ajram, L.; Ivin, G.; Heasman, M.; et al. Effects of cannabidivarin (CBDV) on brain excitation and inhibition systems in adults with and without Autism Spectrum Disorder (ASD): A single dose trial during magnetic resonance spectroscopy. Transl. Psychiatry 2019, 9, 313. [Google Scholar] [CrossRef] [Green Version]
- Zou, M.; Liu, Y.; Xie, S.; Wang, L.; Li, D.; Li, L.; Wang, F.; Zhang, Y.; Xia, W.; Sun, C.; et al. Alterations of the endocannabinoid system and its therapeutic potential in autism spectrum disorder. Open Biol. 2021, 11, 200306. [Google Scholar] [CrossRef]
- Lukito, S.; Norman, L.; Carlisi, C.; Radua, J.; Hart, H.; Simonoff, E.; Rubia, K. Comparative meta-analyses of brain structural and functional abnormalities during cognitive control in attention-deficit/hyperactivity disorder and autism spectrum disorder. Psychol. Med. 2020, 50, 894–919. [Google Scholar] [CrossRef]
- Eapen, V.; Nicholls, L.; Spagnol, V.; Mathew, N.E. Current status of biological treatment options in Autism Spectrum Disorder. Asian J. Psychiatry 2017, 30, 1–10. [Google Scholar] [CrossRef]
- Masi, A.; Demayo, M.M.; Glozier, N.; Guastella, A.J. An Overview of Autism Spectrum Disorder, Heterogeneity and Treatment Options. Neurosci. Bull. 2017, 33, 183–193. [Google Scholar] [CrossRef] [Green Version]
- Iglesias-Vázquez, L.; van Ginkel Riba, G.; Arija, V.; Canals, J. Composition of Gut Microbiota in Children with Autism Spectrum Disorder: A Systematic Review and Meta-Analysis. Nutrients 2020, 12, 792. [Google Scholar] [CrossRef] [Green Version]
- Yoon, S.H.; Choi, J.; Lee, W.J.; Do, J.T. Genetic and Epigenetic Etiology Underlying Autism Spectrum Disorder. J. Clin. Med. 2020, 9, 966. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Loomes, R.; Hull, L.; Mandy, W.P.L. What Is the Male-to-Female Ratio in Autism Spectrum Disorder? A Systematic Review and Meta-Analysis. J. Am. Acad. Child Adolesc. Psychiatry 2017, 56, 466–474. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- RISPERDAL (risperidone)-Prescribing Information. Available online: https://www.accessdata.fda.gov/drugsatfda_docs/label/2009/020272s056,020588s044,021346s033,021444s03lbl.pdf (accessed on 22 May 2021).
- ABILIFY (aripiprazol)-Prescribing Information. Available online: https://www.accessdata.fda.gov/drugsatfda_docs/label/2014/021436s038,021713s030,021729s022,021866s023lbl.pdf (accessed on 22 May 2021).
- Chakrabarti, B.; Persico, A.; Battista, N.; Maccarrone, M. Endocannabinoid Signaling in Autism. Neurotherapeutics 2015, 12, 837–847. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Karhson, D.S.; Krasinska, K.M.; Dallaire, J.A.; Libove, R.A.; Phillips, J.M.; Chien, A.S.; Garner, J.P.; Hardan, A.Y.; Parker, K.J. Plasma anandamide concentrations are lower in children with autism spectrum disorder. Mol. Autism 2018, 9, 18. [Google Scholar] [CrossRef] [PubMed]
- Aran, A.; Eylon, M.; Harel, M.; Polianski, L.; Nemirovski, A.; Tepper, S.; Schnapp, A.; Cassuto, H.; Wattad, N.; Tam, J. Lower circulating endocannabinoid levels in children with autism spectrum disorder. Mol. Autism 2019, 10, 2. [Google Scholar] [CrossRef]
- Wei, D.; Lee, D.Y.; Cox, C.D.; Karsten, C.A.; Penagarikano, O.; Geschwind, D.H.; Gall, C.M.; Piomelli, D. Endocannabinoid signaling mediates oxytocin-driven social reward. Proc. Natl. Acad. Sci. USA 2015, 112, 14084–14089. [Google Scholar] [CrossRef] [Green Version]
- Schultz, S.; Siniscalco, D. Endocannabinoid system involvement in autism spectrum disorder: An overview with potential therapeutic applications. AIMS Mol. Sci. 2019, 6, 27–37. [Google Scholar] [CrossRef]
- Siniscalco, D.; Sapone, A.; Giordano, C.; Cirillo, A.; de Magistris, L.; Rossi, F.; Fasano, A.; Bradstreet, J.J.; Maione, S.; Antonucci, N. Cannabinoid receptor type 2, but not type 1, is up-regulated in peripheral blood mononuclear cells of children affected by autistic disorders. J. Autism Dev. Disord. 2013, 43, 2686–2695. [Google Scholar] [CrossRef]
- Cooper, R.E.; Williams, E.; Seegobin, S.; Tye, C.; Kuntsi, J.; Asherson, P. Cannabinoids in attention-deficit/hyperactivity disorder: A randomised-controlled trial. Eur. Neuropsychopharmacol. 2017, 27, 795–808. [Google Scholar] [CrossRef]
- Mostafavi, M.; Gaitanis, J. Autism Spectrum Disorder and Medical Cannabis: Review and Clinical Experience. Semin. Pediatric Neurol. 2020, 35, 100833. [Google Scholar] [CrossRef]
- Kurz, R.; Blaas, K. Use of dronabinol (delta-9-THC) in autism: A prospective single-case-study with an early infantile autistic child. Cannabinoids 2010, 5, 4–6. [Google Scholar]
- Ponton, J.A.; Smyth, K.; Soumbasis, E.; Llanos, S.A.; Lewis, M.; Meerholz, W.A.; Tanguay, R.L. A pediatric patient with autism spectrum disorder and epilepsy using cannabinoid extracts as complementary therapy: A case report. J. Med. Case Rep. 2020, 14, 162. [Google Scholar] [CrossRef] [PubMed]
- Bar-Lev Schleider, L.; Mechoulam, R.; Saban, N.; Meiri, G.; Novack, V. Real life Experience of Medical Cannabis Treatment in Autism: Analysis of Safety and Efficacy. Sci. Rep. 2019, 9, 200. [Google Scholar] [CrossRef] [PubMed]
- Aran, A.; Cassuto, H.; Lubotzky, A.; Wattad, N.; Hazan, E. Brief Report: Cannabidiol-Rich Cannabis in Children with Autism Spectrum Disorder and Severe Behavioral Problems—A Retrospective Feasibility Study. J. Autism Dev. Disord. 2019, 49, 1284–1288. [Google Scholar] [CrossRef] [PubMed]
- Fleury-Teixeira, P.; Caixeta, F.V.; Da Silva, L.C.R.; Brasil-Neto, J.P.; Malcher-Lopes, R. Effects of cbd-enriched cannabis sativa extract on autism spectrum disorder symptoms: An observational study of 18 participants undergoing compassionate use. Front. Neurol. 2018, 10, 1145. [Google Scholar] [CrossRef] [Green Version]
- Pretzsch, C.M.; Freyberg, J.; Voinescu, B.; Lythgoe, D.; Horder, J.; Mendez, M.A.; Wichers, R.; Ajram, L.; Ivin, G.; Heasman, M.; et al. The effect of cannabidiol (CBD) on low-frequency activity and functional connectivity in the brain of adults with and without autism spectrum disorder (ASD). J. Psychopharmacol. 2019, 33, 1141–1148. [Google Scholar] [CrossRef]
- Kuester, G.; Vergara, K.; Ahumada, A.; Gazmuri, A.M. Oral cannabis extracts as a promising treatment for the core symptoms of autism spectrum disorder: Preliminary experience in Chilean patients. J. Neurol. Sci. 2017, 381, 932–933. [Google Scholar] [CrossRef]
- Barchel, D.; Stolar, O.; De-Haan, T.; Ziv-Baran, T.; Saban, N.; Fuchs, D.O.; Koren, G.; Berkovitch, M. Oral cannabidiol use in children with autism spectrum disorder to treat related symptoms and Co-morbidities. Front. Pharmacol. 2019, 9, 1521. [Google Scholar] [CrossRef]
- Mcvige, J.; Headd, V.; Alwaidy, M.; Lis, D.; Kaur, D.; Albert, B.; Mechtler, L. Medical cannabis in the treatment of patients with autism spectrum disorder. Neurology 2020, 94 (Suppl. 15), 1648. [Google Scholar]
- EPIDIOLEX (cannabidiol) Oral Solution-Prescribing Information. Available online: https://www.accessdata.fda.gov/drugsatfda_docs/label/2018/210365lbl.pdf (accessed on 29 May 2021).
- Home-ClinicalTrials.gov. Available online: https://clinicaltrials.gov/ (accessed on 5 June 2021).
- Cannabinoids for Behavioral Problems in Children with ASD (NCT02956226). Available online: https://clinicaltrals.gov/ct2/show/study/NCT02956226 (accessed on 5 June 2021).
- Shifting Brain Excitation-Inhibition Balance in Autism Spectrum Disorder (NCT03537950). Available online: https://clinicaltrials.gov/ct2/show/study/NCT03537950 (accessed on 5 June 2021).
- Medical Cannabis Registry and Pharmacology (NCT03699527). Available online: https://clinicaltrals.gov/ct2/show/study/NCT03699527 (accessed on 5 June 2021).
- Safety and Tolerability of Cannabidivarin (CBDV) in Children and Young Adults with Autism Spectrum Disorder (NCT03849456). Available online: https://clinicaltrials.gov/ct2/show/study/NCT03849456 (accessed on 5 June 2021).
- Cannabidivarin (CBDV), vs. Placebo in Children with Autism Spectrum Disorder (ASD) (NCT03202303). Available online: https://clinicaltrials.gov/ct2/show/study/NCT03202303 (accessed on 5 June 2021).
- Trial of Cannabidiol to Treat Severe Behavior Problems in Children with Autism (NCT04517799). Available online: https://clinicaltrials.gov/ct2/show/study/NCT04517799 (accessed on 5 June 2021).
- Cannabidiol for ASD Open Trial (NCT03900923). Available online: https://clinicaltrials.gov/ct2/show/study/NCT03900923 (accessed on 5 June 2021).
- Trial to Investigate the Safety and Efficacy of Cannabidiol Oral Solution (GWP42003-P; CBD-OS) in Children and Adolescents with Autism Spectrum Disorder (NCT04745026). Available online: https://clinicaltrals.gov/ct2/show/study/NCT04745026 (accessed on 5 June 2021).
- CASCADE: Cannabidiol Study in Children with Autism Spectrum Disorder (NCT04520685). Available online: https://clinicaltrials.gov/ct2/show/study/NCT04520685 (accessed on 5 June 2021).
- Aran, A.; Harel, M.; Cassuto, H.; Polyansky, L.; Schnapp, A.; Wattad, N.; Shmueli, D.; Golan, D.; Castellanos, F.X. Cannabinoid treatment for autism: A proof-of-concept randomized trial. Mol. Autism 2021, 12, 6. [Google Scholar] [CrossRef]
- Sachs, J.; Mcglade, E.; Yurgelun-Todd, D. Safety and Toxicology of Cannabinoids. Neurotherapeutics 2015, 12, 735–746. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Campbell, C.T.; Phillips, M.S.; Manasco, K. Cannabinoids in Pediatrics. J. Pediatric Pharmacol. Ther. 2017, 22, 176–185. [Google Scholar] [CrossRef] [PubMed]
- Medical Use of Cannabis and Cannabinoids: Questions and Answers for Policymaking. Available online: http://www.emcdda.europa.eu/system/files/publications/10171/20185584_TD0618186ENN_PDF.pdf (accessed on 12 June 2021).
- CESAMET (nabilone) Capsules, for Oral Administration-Charateristics. Available online: https://www.accessdata.fda.gov/drugsatfda_docs/label/2006/018677s011lbl.pdf (accessed on 12 June 2021).
- MARINOL (dronabinol) Capsules, for Oral Use-Prescribing Information. Available online: https://www.accessdata.fda.gov/drugsatfda_docs/label/2017/018651s029lbl.pdf (accessed on 12 June 2021).
- SYNDROS (dronabinol) Oral Solution-Prescribing Information. Available online: https://www.accessdata.fda.gov/drugsatfda_docs/label/2017/205525s003lbl.pdf (accessed on 12 June 2021).
- Sativex Oralmucosal Spray. Available online: https://www.medicines.org.uk/emc/product/602/smpc#gref (accessed on 12 June 2021).
| Diseases and Symptoms | Therapeutic Potential of Cannabinoids |
|---|---|
| Alzheimer’s Disease | Anti-inflammatory Neuroprotector Antioxidant |
| Parkinson’s Disease | |
| Huntington’s Disease | |
| Multiple Sclerosis | Antispastic Analgesic |
| Epilepsy | Anticonvulsant |
| Tourette’s Syndrome | Improvement of symptomatology |
| Cancer | Analgesic Antiemetic Appetite stimulator Antitumor |
| Glaucoma | Intraocular pression reduction |
| Inflammatory Bowel Diseases | Anti-inflammatory Healing |
| Schizophrenia | Antipsychotic |
| Sleep Disorders | Decrease sleep latency and nocturnal awakenings Sedative |
| Pain | Analgesic |
| Post-Traumatic Stress Disorder | Anxiolytic |
| Nausea and Vomiting | Antiemetic |
| Anorexia | Appetite stimulator |
| Title | Publication Year | Type of Study | Number of Participants | Average Age | Administrated Treatment | Daily Dose | Duration of Treatment and Follow-Up | Outcomes (% of Decrease of Symptoms) | Adverse Effects (Number of Participants) | Reference |
|---|---|---|---|---|---|---|---|---|---|---|
| Use of dronabinol (delta-9-THC) in autism: A prospective single-case-study with an early infantile autistic child | 2010 | Individual Case Study | 1 | 6 years old | Dronabinol dissolved in sesame oil | Initial: 1 drop in the morning (0.62 mg) Final: 1 drop in the morning, 1 drop in the middle of the day, and 3 drops in the evening (3.62 mg) | Treatment: 6 months Follow-up: initial and after 6 months | Irritability: 55.5% Lethargy: 26.7% Stereotyped behaviour: 33.3% Hyperactivity: 56.2% Inappropriate speech: 28.6% | NR | [58] |
| Oral cannabis extracts as a promising treatment for the core symptoms of autism spectrum disorder: Preliminary experience in Chilean patients | 2017 | Case Series Study | 21 | 9 years old and 10 months | 15 participants: extract with CBD:THC (1:1) 4 participants: extract with high CBD content 2 participants: extract with high THC content | NR | Treatment: 3 to 12 months Follow-up: after 6, 7 months, on average | CGI-I e APSI: 66.7% Central symptoms of ASDs: >50%, in at least one of the symptoms Sensory disturbances, food acceptance, sleep disturbances, and seizures: >50% | Agitation: 2 Irritability: 1 | [64] |
| Brief Report: Cannabidiol-Rich Cannabis in Children with Autism Spectrum Disorder and Severe Behavioral Problems—A Retrospective Feasibility Study | 2018 | Retrospective Cohort Study | 60 | 11 years old and 8 months | Plant extract with CBD:THC (20:1), dissolved in olive oil | 2 administrations per day (1.8 ± 1.6 mg/kg/day of CBD and 0.22 ± 0.14 mg/kg/day of THC) 3 administrations per day (3.8 ± 2.6 mg/kg/day of CBD and 0.29 ± 0.22 mg/kg/day of THC) | Treatment: 7 to 13 months Follow-up: continuous | Behaviour: 61% Anxiety: 39% Communication: 47% HSQ-ASD: 29% APSI: 33% | Hypervigilance: 8 Agitation: 5 Irritability: 5 Decrease appetite: 5 | [61] |
| Oral Cannabidiol Use in Children with Autism Spectrum Disorder to Treat Related Symptoms and Co-morbidities | 2019 | Prospective Cohort Study | 53 | 11 years old | Oil with 30% of cannabinoids, CBD:THC (1:20) | 16 mg/kg of CBD (maximum of 600 mg daily) 0.8 mg/kg of THC (maximum of 40 mg daily) | Treatment: 66 days, on average Follow-up: biweekly telephone interviews | Self-injury and rage attacks: 67.6% Hyperactivity: 68.4% Sleep disorders: 71.4% Anxiety: 47.1% Overall improvement: 74.5% | Somnolence: 12 Decrease appetite: 6 Appetite increase: 4 | [65] |
| Real-life experience of medical cannabis treatment in autism: analysis of safety and efficacy | 2019 | Prospective Cohort Study | 188 | 12 years old and 9 months | 66 participants: Oil with 30% of CBD and 1.5% of THC 46 participants with insomnias: Oil with 3% of THC | 66 participants: 3 administrations per day (79.5 ± 6.5 mg of CBD and 4.0 ± 3.0 mg of THC, per dose) 46 participants with insomnia: 1 administration at evening (5.0 ± 4.5 mg of THC, per dose) | Treatment: 6 months Follow-up: after 1 and 6 months | Quality of life: 35.5% Mood: 21.5% Ability to perform activities of daily living: 16.5% Sleep quality: 21.4% Concentration: 14% Agitation: 91% Rage attacks: 90.3% Seizures: 15.4% (total improvement: 84.6%) | Agitation: 6 Somnolence: 3 Psychoactive effects: 3 Appetite increase: 3 Digestion problems: 3 Dry mouth: 2 Decrease appetite: 2 | [60] |
| Effects of CBD-Enriched Cannabis sativa Extract on Autism Spectrum Disorder Symptoms: An Observational Study of 18 Participants Undergoing Compassionate Use | 2019 | Prospective Cohort Study | 18 | 10 years old and 9 months | Plant extract dosed in CBD:THC (75:1), administered as oral capsules | 2 administrations per day (4,55 mg/kg/day of CBD and 0,06 mg/kg/day of THC, on average) | Treatment: 6 to 9 months Follow-up: initial and monthly | ADHD: 86.7% Behavioural disorders: 73.3% Motor deficits: 83.3% Autonomy deficits: 66.7% Communication and social interaction deficits: 73.3% Cognitive deficits: 86.7% Sleep disorders: 100% Seizures: 60% (total improvement: 40%) | Somnolence: 3 Irritability: 3 Diarrhea: 1 Appetite increase: 1 Conjunctival hyperemia: 1 Increased body temperature: 1 Nocturia: 2 | [62] |
| Medical Cannabis in the Treatment of Patients with Autism Spectrum Disorder | 2020 | Case Series Study | 20 | NR | Medicinal cannabis | NR | NR | Improvement in: All points of ACGIC Epilepsy Pain Sleep Mood Aggressiveness Concentration | Unspecified adverse effects: 3 | [66] |
| A pediatric patient with autism spectrum disorder using cannabinoid extracts as complementary therapy: a case report | 2020 | Individual Case Study | 1 | 15 years old | CBE dosed in CBD:THC (20:1), dissolved in olive oil | Administração de 0.2 mL duas vezes ao dia (4 mg de CBD e 0.2 mg de THC, por dose) | Follow-up: contínuo de 6 meses | Improvement in: Quality of life Behavioral and communicative symptoms Anxiety Sleep disorders Weight gain | NR | [59] |
| Autism Spectrum Disorder and Medical Cannabis: Review and Clinical Experience | 2020 | Retrospective Cohort Study | 32 | NR | Medicinal or Hemp-based Cannabis Products | NR | NR | Epilepsy: 91% Aggressiveness: 60% | Worsening of obsessive-compulsive and repetitive behavior, insomnia or mania: 4 | [57] |
| Effects of cannabidiol on brain excitation and inhibition systems; a randomized placebo-controlled single-dose trial during magnetic resonance spectroscopy in adults with and without autism spectrum disorder. | 2019 | Case- Control Study | 34, of which: 17 with ASD 17 without ASD (control) | 31 years old and 3 months | Liquid oral dose of CBD or Placebo | 600 mg of CBD or Placebo | Treatment: 2 doses separated by, at least, 13 days Follow-up: at the time of administration and after 2 h | CBD increases glutamate in the basal ganglia and decreases in the prefrontal cortex in participants with ASD and control CBD decreases GABA in basal ganglia and prefrontal cortex in ASD subjects and increases in control subjects | NR | [39] |
| Effects of cannabidivarin (CBDV) on brain excitation and inhibition systems in adults with and without Autism Spectrum Disorder (ASD): a single dose trial during magnetic resonance spectroscopy | 2019 | Case- Control Study | 34, of which:17 with ASD 17 without ASD(control) | 31 years old and 3 months | Liquid oral dose of CBDV or Placebo | 600 mg of CBDV or Placebo | Treatment: 2 doses separated by, at least, 13 days Follow-up: at the time of administration and after 2 h | CBDV with no impact on GABA and glutamate levels in the prefrontal cortex CBDV alters basal ganglia glutamate levels negatively correlated with baseline glutamate levels | NR | [40] |
| The effect of cannabidiol (CBD) on low-frequency activity and functional connectivity in the brain of adults with and without autism spectrum disorder (ASD) | 2019 | Case- Control Study | 34, of which: 17 with ASD 17 without ASD (control) | 31 years old and 3 months | Liquid oral dose of CBD or Placebo | 600 mg of CBD or Placebo | Treatment: 2 doses separated by, at least, 13 days Follow-up: at the time of administration and after 2 h | CBD prominently increases fALFF in the cerebellar vermis and right fusiform gyrus in subjects with ASD CBD increases FC in the cerebellar vermis but not in the right fusiform gyrus | NR | [63] |
| Title | Identifier | Type of Study | State | Study Population | Number of Participants | Duration | Expected Completion Date | Results | Reference |
|---|---|---|---|---|---|---|---|---|---|
| Cannabinoids for Behavioural Problems in Autism Spectrum Disorder: A Double-Blind, Randomized, Placebo-controlled Trial with Crossover | NCT02956226 | Randomized Interventional Study (Clinical Trial) Quadruple mask Parallel | Complete | Patients with ASD and behavioral problems, between 5 and 21 years old | 150 | 3 months | 27 December 2018 | Published | [69] |
| Shifting Brain Excitation-Inhibition Balance Through the Endocannabinoid System in Men with Autism Spectrum Disorder (ASD) and in Healthy Controls | NCT03537950 | Randomized Interventional Study (Clinical Trial) Double mask Crossover | Unknown | Patients with ASD, between 18 and 50 years old, male | 38 | 5 to 6 months | 27 April 2019 | Not Published | [70] |
| Medical Cannabis Registry and Pharmacology | NCT03699527 | Observational Study (Prospective Cohort Study) | Complete | Patients with ASD under 21 years old | 119 | 5 years | 15 January 2020 | Not Published | [71] |
| Safety and Tolerability of GWP42006 in Children and Young Adults with Autism Spectrum Disorder | NCT03849456 | Interventional Study (Clinical trial) Open Label | Terminated | Patients with ASD, between 4 and 18 years old | 1 | 52 weeks | 26 May 2020 | Not Published | [72] |
| Cannabidivarin (CBDV) vs. Placebo in Children with Autism Spectrum Disorder (ASD) | NCT03202303 | Randomized Interventional Study (Clinical Trial) Parallel Double mask | In recruitment | Patients with ASD, between 5 and 18 years old | 100 | 12 weeks | 30 September 2021 | Not Published | [73] |
| A Double-Blind, Crossover Trial of Cannabidiol to Treat Severe Behavior Problems in Children with Autism | NCT04517799 | Randomized Interventional Study (Clinical Trial) Quadruple mask Crossover | In recruitment | Patients with ASD and severe behavioral problems, between 7 and 14 years old, male | 30 | 16 weeks | 31 December 2021 | Not Published | [74] |
| A Phase 2 Study of Cannabidiol as a New Treatment for Autism Spectrum Disorder in Children and Adolescents | NCT03900923 | Interventional Study (Clinical trial) Open Label | In recruitment | Patients with ASD, between 7 and 17 years old | 30 | 6 weeks | 1 July 2022 | Not Published | [75] |
| An Exploratory, Phase 2, Randomized, Double-blind, Placebo-controlled Trial to Investigate the Safety and Efficacy of Cannabidiol Oral Solution (GWP42003 P; CBD-OS) in Children and Adolescents with Autism Spectrum Disorder | NCT04745026 | Randomized Interventional Study (Clinical Trial) Quadruple mask Parallel | In recruitment | Patients with ASD, between 6 and 17 years old | 160 | 12 weeks | 30 March 2023 | Not Published | [76] |
| Cannabidiol Study in Children with Autism Spectrum Disorder (CASCADE): A Double-Blind, Placebo-Controlled Study to Investigate Efficacy and Safety of Cannabidiol in Children and Adolescents With Autism | NCT04520685 | Randomized Interventional Study (Clinical Trial) Triple mask Crossover | In recruitment | Patients with ASD, between 5 and 17 years old | 70 | 27 weeks | 1 June 2023 | Not Published | [77] |
| Affected Area | Adverse Effects |
|---|---|
| Cardiovascular System | Increase cardiovascular activity Tachycardia Systemic vasodilation Increased risk of myocardial infarction, cardiomyopathy, angina, and cardiorespiratory arrest |
| Respiratory System | Increased inflammation and airway resistance Destruction of lung tissue Increased risk of developing chronic bronchitis and pulmonary emphysema Increased risk of developing lung cancer |
| Mental Health | Anxiety and panic attacks Exacerbation or development of schizophrenia, bipolar disorder, and depression in vulnerable patients |
| Cognition | Change in sensory and temporal perception Short-term memory alteration Change in psychomotor function Dependency |
| Hormonal System, Fertility and Maternity | Anti-androgenic effects Reduction of pregnant weight gain Low-birth-weight new-borns |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Carreira, L.D.; Matias, F.C.; Campos, M.G. Clinical Data on Canabinoids: Translational Research in the Treatment of Autism Spectrum Disorders. Biomedicines 2022, 10, 796. https://doi.org/10.3390/biomedicines10040796
Carreira LD, Matias FC, Campos MG. Clinical Data on Canabinoids: Translational Research in the Treatment of Autism Spectrum Disorders. Biomedicines. 2022; 10(4):796. https://doi.org/10.3390/biomedicines10040796
Chicago/Turabian StyleCarreira, Laura D., Francisca C. Matias, and Maria G. Campos. 2022. "Clinical Data on Canabinoids: Translational Research in the Treatment of Autism Spectrum Disorders" Biomedicines 10, no. 4: 796. https://doi.org/10.3390/biomedicines10040796
APA StyleCarreira, L. D., Matias, F. C., & Campos, M. G. (2022). Clinical Data on Canabinoids: Translational Research in the Treatment of Autism Spectrum Disorders. Biomedicines, 10(4), 796. https://doi.org/10.3390/biomedicines10040796








