Roux-en-Y Gastric Bypass Modulates AMPK, Autophagy and Inflammatory Response in Leukocytes of Obese Patients
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Population
2.2. Clinical and Biochemical Determinations
2.3. Evaluation of Systemic Cytokines IL6 and IL1β
2.4. Isolation of Leukocytes from Blood Samples
2.5. Fluorescence Imaging of Mitochondrial Membrane Potential
2.6. Immunoblotting
2.7. Statistical Analysis
3. Results
3.1. Anthropometric and Biochemical Parameters following RYGB Surgery
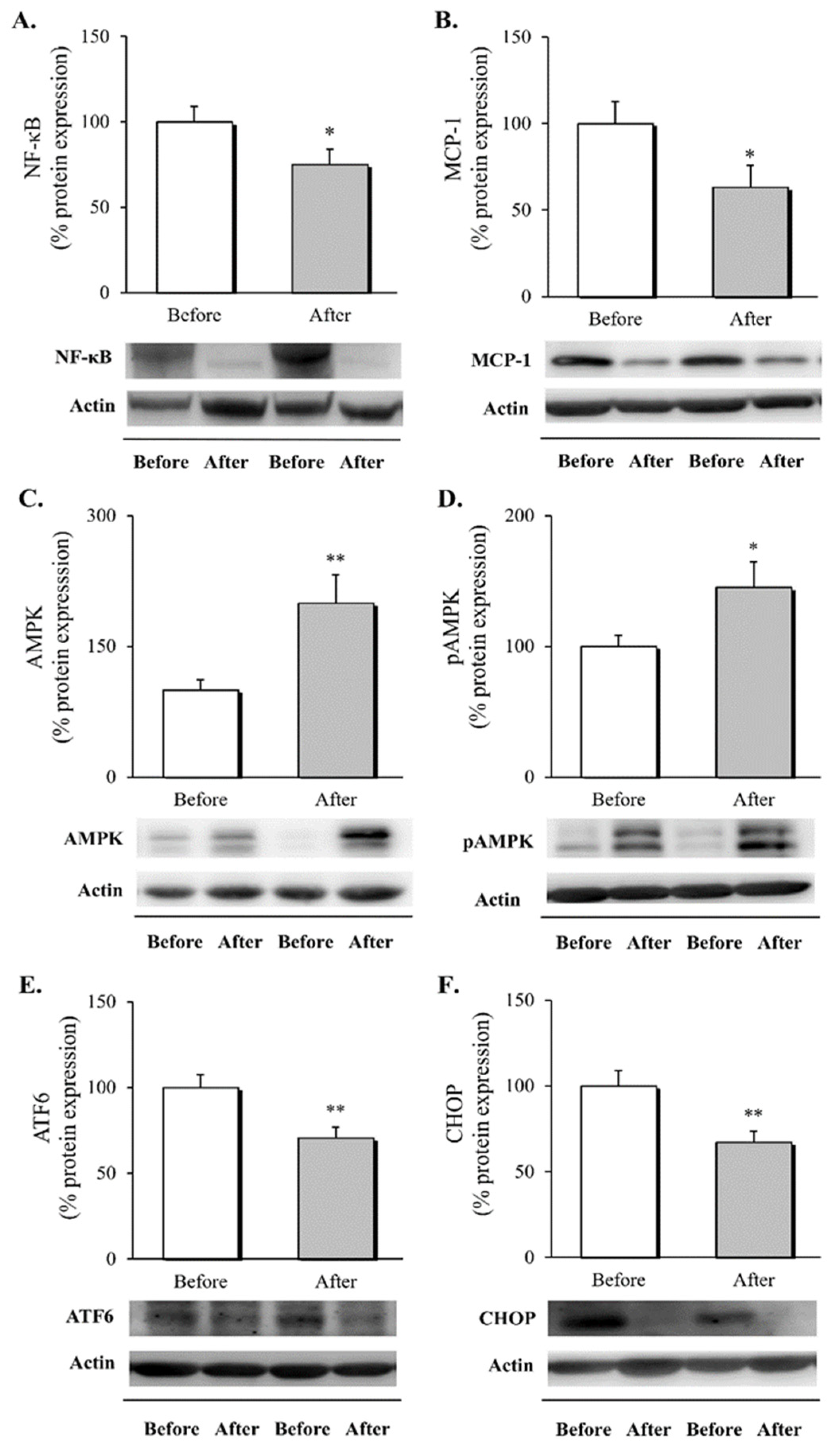
3.2. Inflammatory Response, AMPK Activation and ER Stress Markers in Leukocytes following RYGB Surgery
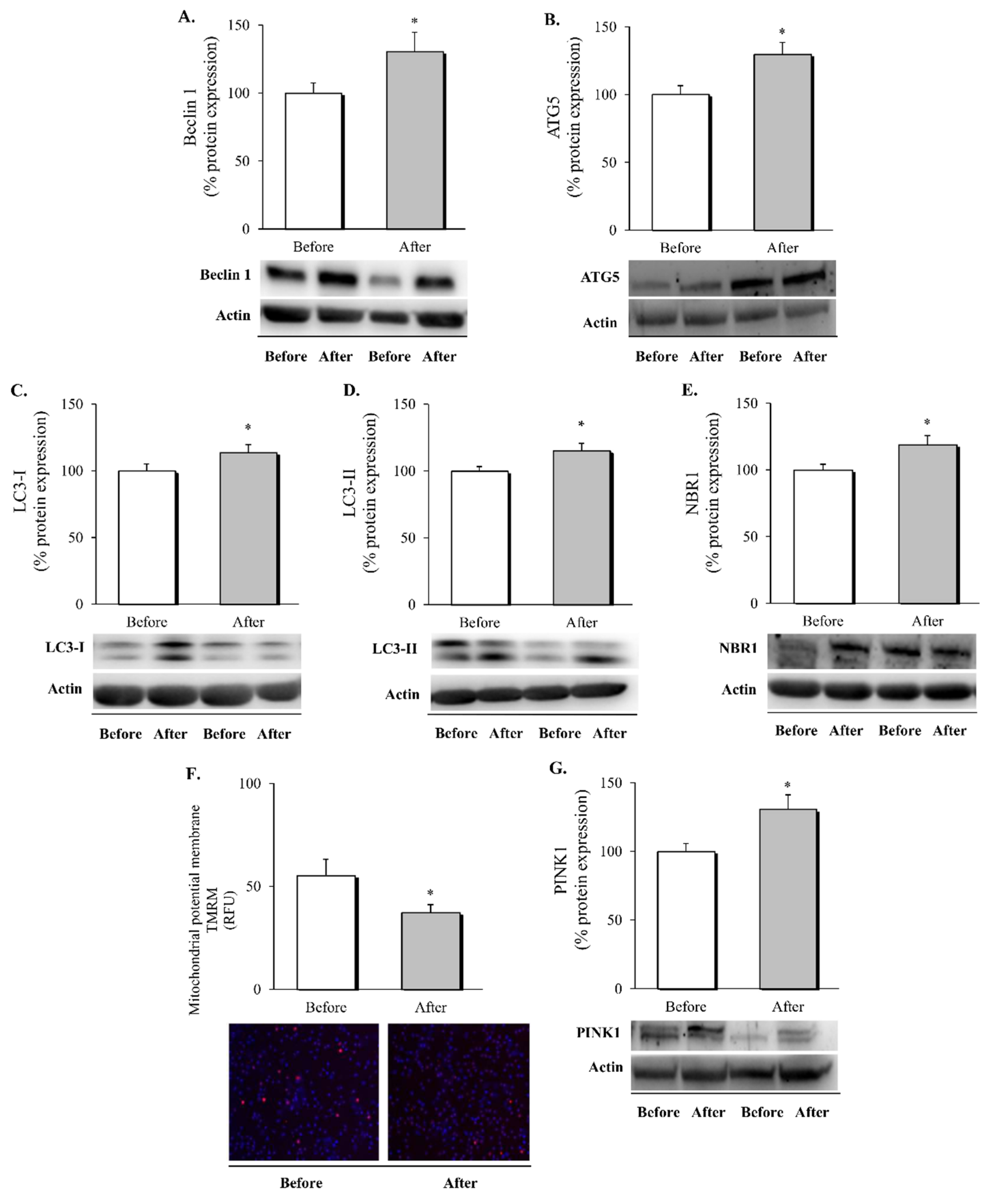
3.3. Autophagy and Mitophagy in Leukocytes following RYGB Surgery
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Blüher, M. Obesity: Global Epidemiology and Pathogenesis. Nat. Rev. Endocrinol. 2019, 15, 288–298. [Google Scholar] [CrossRef] [PubMed]
- Lorenzo, A.D. New Obesity Classification Criteria as a Tool for Bariatric Surgery Indication. WJG 2016, 22, 681. [Google Scholar] [CrossRef] [PubMed]
- Sjöström, L.; Narbro, K.; Sjöström, C.D.; Karason, K.; Larsson, B.; Wedel, H.; Lystig, T.; Sullivan, M.; Bouchard, C.; Carlsson, B.; et al. Effects of Bariatric Surgery on Mortality in Swedish Obese Subjects. N. Engl. J. Med. 2007, 357, 741–752. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rega-Kaun, G.; Kaun, C.; Jaegersberger, G.; Prager, M.; Hackl, M.; Demyanets, S.; Wojta, J.; Hohensinner, P.J. Roux-En-Y-Bariatric Surgery Reduces Markers of Metabolic Syndrome in Morbidly Obese Patients. Obes. Surg. 2020, 30, 391–400. [Google Scholar] [CrossRef] [Green Version]
- Farias, G.; Netto, B.D.M.; Boritza, K.; Bettini, S.C.; Vilela, R.M.; Dâmaso, A.R. Impact of Weight Loss on Inflammation State and Endothelial Markers Among Individuals with Extreme Obesity After Gastric Bypass Surgery: A 2-Year Follow-up Study. Obes. Surg. 2020, 30, 1881–1890. [Google Scholar] [CrossRef] [PubMed]
- Abad-Jiménez, Z.; López-Domènech, S.; Gómez-Abril, S.Á.; Periañez-Gómez, D.; de Marañón, A.M.; Bañuls, C.; Morillas, C.; Víctor, V.M.; Rocha, M. Effect of Roux-En-Y Bariatric Bypass Surgery on Subclinical Atherosclerosis and Oxidative Stress Markers in Leukocytes of Obese Patients: A One-Year Follow-Up Study. Antioxidants 2020, 9, 734. [Google Scholar] [CrossRef]
- Monteiro, R.; Azevedo, I. Chronic Inflammation in Obesity and the Metabolic Syndrome. Mediat. Inflamm. 2010, 2010, 1–10. [Google Scholar] [CrossRef]
- Galic, S.; Fullerton, M.D.; Schertzer, J.D.; Sikkema, S.; Marcinko, K.; Walkley, C.R.; Izon, D.; Honeyman, J.; Chen, Z.-P.; van Denderen, B.J.; et al. Hematopoietic AMPK Β1 Reduces Mouse Adipose Tissue Macrophage Inflammation and Insulin Resistance in Obesity. J. Clin. Investig. 2011, 121, 4903–4915. [Google Scholar] [CrossRef] [Green Version]
- Garcia, D.; Hellberg, K.; Chaix, A.; Wallace, M.; Herzig, S.; Badur, M.G.; Lin, T.; Shokhirev, M.N.; Pinto, A.F.M.; Ross, D.S.; et al. Genetic Liver-Specific AMPK Activation Protects against Diet-Induced Obesity and NAFLD. Cell Rep. 2019, 26, 192–208. [Google Scholar] [CrossRef] [Green Version]
- Li, Y.; Xu, S.; Mihaylova, M.M.; Zheng, B.; Hou, X.; Jiang, B.; Park, O.; Luo, Z.; Lefai, E.; Shyy, J.Y.-J.; et al. AMPK Phosphorylates and Inhibits SREBP Activity to Attenuate Hepatic Steatosis and Atherosclerosis in Diet-Induced Insulin-Resistant Mice. Cell Metab. 2011, 13, 376–388. [Google Scholar] [CrossRef] [Green Version]
- Chan, K.L.; Pillon, N.J.; Sivaloganathan, D.M.; Costford, S.R.; Liu, Z.; Théret, M.; Chazaud, B.; Klip, A. Palmitoleate Reverses High Fat-Induced Proinflammatory Macrophage Polarization via AMP-Activated Protein Kinase (AMPK). J. Biol. Chem. 2015, 290, 16979–16988. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xue, B.; Kahn, B.B. AMPK Integrates Nutrient and Hormonal Signals to Regulate Food Intake and Energy Balance through Effects in the Hypothalamus and Peripheral Tissues: AMPK Regulates Food Intake and Energy Balance. J. Physiol. 2006, 574, 73–83. [Google Scholar] [CrossRef] [PubMed]
- Mihaylova, M.M.; Shaw, R.J. The AMPK Signalling Pathway Coordinates Cell Growth, Autophagy and Metabolism. Nat. Cell Biol. 2011, 13, 1016–1023. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.; Moon, S.Y.; Kim, J.-S.; Baek, C.H.; Kim, M.; Min, J.Y.; Lee, S.K. Activation of AMP-Activated Protein Kinase Inhibits ER Stress and Renal Fibrosis. Am. J. Physiol. -Ren. Physiol. 2015, 308, F226–F236. [Google Scholar] [CrossRef]
- Hotamisligil, G.S. Endoplasmic Reticulum Stress and the Inflammatory Basis of Metabolic Disease. Cell 2010, 140, 900–917. [Google Scholar] [CrossRef] [Green Version]
- Zhang, K.; Kaufman, R.J. From Endoplasmic-Reticulum Stress to the Inflammatory Response. Nature 2008, 454, 455–462. [Google Scholar] [CrossRef] [Green Version]
- Adams, C.J.; Kopp, M.C.; Larburu, N.; Nowak, P.R.; Ali, M.M.U. Structure and Molecular Mechanism of ER Stress Signaling by the Unfolded Protein Response Signal Activator IRE1. Front. Mol. Biosci. 2019, 6, 11. [Google Scholar] [CrossRef] [Green Version]
- Gregor, M.F.; Yang, L.; Fabbrini, E.; Mohammed, B.S.; Eagon, J.C.; Hotamisligil, G.S.; Klein, S. Endoplasmic Reticulum Stress Is Reduced in Tissues of Obese Subjects After Weight Loss. Diabetes 2009, 58, 693–700. [Google Scholar] [CrossRef] [Green Version]
- Ferraz-Bannitz, R.; Welendorf, C.R.; Coelho, P.O.; Salgado, W.; Nonino, C.B.; Beraldo, R.A.; Foss-Freitas, M.C. Bariatric Surgery Can Acutely Modulate ER-Stress and Inflammation on Subcutaneous Adipose Tissue in Non-Diabetic Patients with Obesity. Diabetol. Metab. Syndr. 2021, 13, 19. [Google Scholar] [CrossRef]
- Ohsumi, Y. Historical Landmarks of Autophagy Research. Cell Res. 2014, 24, 9–23. [Google Scholar] [CrossRef] [Green Version]
- Jin, S.M.; Lazarou, M.; Wang, C.; Kane, L.A.; Narendra, D.P.; Youle, R.J. Mitochondrial Membrane Potential Regulates PINK1 Import and Proteolytic Destabilization by PARL. J. Cell Biol. 2010, 191, 933–942. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kovsan, J.; Blüher, M.; Tarnovscki, T.; Klöting, N.; Kirshtein, B.; Madar, L.; Shai, I.; Golan, R.; Harman-Boehm, I.; Schön, M.R.; et al. Altered Autophagy in Human Adipose Tissues in Obesity. J. Clin. Endocrinol. Metab. 2011, 96, E268–E277. [Google Scholar] [CrossRef] [PubMed]
- Nuñez, C.E.; Rodrigues, V.S.; Gomes, F.S.; de Moura, R.F.; Victorio, S.C.; Bombassaro, B.; Chaim, E.A.; Pareja, J.C.; Geloneze, B.; Velloso, L.A.; et al. Defective Regulation of Adipose Tissue Autophagy in Obesity. Int. J. Obes. 2013, 37, 1473–1480. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Öst, A.; Svensson, K.; Ruishalme, I.; Brännmark, C.; Franck, N.; Krook, H.; Sandström, P.; Kjolhede, P.; Strålfors, P. Attenuated MTOR Signaling and Enhanced Autophagy in Adipocytes from Obese Patients with Type 2 Diabetes. Mol. Med. 2010, 16, 235–246. [Google Scholar] [CrossRef] [PubMed]
- Soussi, H.; Reggio, S.; Alili, R.; Prado, C.; Mutel, S.; Pini, M.; Rouault, C.; Clément, K.; Dugail, I. DAPK2 Downregulation Associates With Attenuated Adipocyte Autophagic Clearance in Human Obesity. Diabetes 2015, 64, 3452–3463. [Google Scholar] [CrossRef] [Green Version]
- Chatterjee, T.; Pattanayak, R.; Ukil, A.; Chowdhury, S.; Bhattacharyya, M. Autophagy Protects Peripheral Blood Mononuclear Cells against Inflammation, Oxidative and Nitrosative Stress in Diabetic Dyslipidemia. Free. Radic. Biol. Med. 2019, 143, 309–323. [Google Scholar] [CrossRef]
- Rovira-Llopis, S.; Díaz-Morales, N.; Bañuls, C.; Blas-García, A.; Polo, M.; López-Domenech, S.; Jover, A.; Rocha, M.; Hernández-Mijares, A.; Víctor, V.M. Is Autophagy Altered in the Leukocytes of Type 2 Diabetic Patients? Antioxid. Redox Signal. 2015, 23, 1050–1056. [Google Scholar] [CrossRef]
- Gero, D.; Favre, L.; Allemann, P.; Fournier, P.; Demartines, N.; Suter, M. Laparoscopic Roux-En-Y Gastric Bypass Improves Lipid Profile and Decreases Cardiovascular Risk: A 5-Year Longitudinal Cohort Study of 1048 Patients. Obes. Surg. 2018, 28, 805–811. [Google Scholar] [CrossRef]
- Adams, T.D.; Davidson, L.E.; Litwin, S.E.; Kolotkin, R.L.; LaMonte, M.J.; Pendleton, R.C.; Strong, M.B.; Vinik, R.; Wanner, N.A.; Hopkins, P.N.; et al. Health Benefits of Gastric Bypass Surgery After 6 Years. JAMA 2012, 308, 1122. [Google Scholar] [CrossRef]
- Benotti, P.N.; Wood, G.C.; Carey, D.J.; Mehra, V.C.; Mirshahi, T.; Lent, M.R.; Petrick, A.T.; Still, C.; Gerhard, G.S.; Hirsch, A.G. Gastric Bypass Surgery Produces a Durable Reduction in Cardiovascular Disease Risk Factors and Reduces the Long-Term Risks of Congestive Heart Failure. JAHA 2017, 6. [Google Scholar] [CrossRef]
- Mosallaei, M.; Ehtesham, N.; Rahimirad, S.; Saghi, M.; Vatandoost, N.; Khosravi, S. PBMCs: A New Source of Diagnostic and Prognostic Biomarkers. Arch. Physiol. Biochem. 2020, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Cortez, E.; Neves, F.A.; Bernardo, A.F.; Stumbo, A.C.; Carvalho, L.; Garcia-Souza, É.; Sichieri, R.; Moura, A.S. Lymphocytes Mitochondrial Physiology as Biomarker of Energy Metabolism during Fasted and Fed Conditions. Sci. World J. 2012, 2012, 1–7. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Asghar, A.; Sheikh, N. Role of Immune Cells in Obesity Induced Low Grade Inflammation and Insulin Resistance. Cell. Immunol. 2017, 315, 18–26. [Google Scholar] [CrossRef] [PubMed]
- Kjøbsted, R.; Pedersen, A.J.T.; Hingst, J.R.; Sabaratnam, R.; Birk, J.B.; Kristensen, J.M.; Højlund, K.; Wojtaszewski, J.F.P. Intact Regulation of the AMPK Signaling Network in Response to Exercise and Insulin in Skeletal Muscle of Male Patients with Type 2 Diabetes: Illumination of AMPK Activation in Recovery from Exercise. Diabetes 2016, 65, 1219–1230. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Luo, T.; Nocon, A.; Fry, J.; Sherban, A.; Rui, X.; Jiang, B.; Xu, X.J.; Han, J.; Yan, Y.; Yang, Q.; et al. AMPK Activation by Metformin Suppresses Abnormal Extracellular Matrix Remodeling in Adipose Tissue and Ameliorates Insulin Resistance in Obesity. Diabetes 2016, 65, 2295–2310. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gauthier, M.-S.; O’Brien, E.L.; Bigornia, S.; Mott, M.; Cacicedo, J.M.; Xu, X.J.; Gokce, N.; Apovian, C.; Ruderman, N. Decreased AMP-Activated Protein Kinase Activity Is Associated with Increased Inflammation in Visceral Adipose Tissue and with Whole-Body Insulin Resistance in Morbidly Obese Humans. Biochem. Biophys. Res. Commun. 2011, 404, 382–387. [Google Scholar] [CrossRef] [Green Version]
- Fritzen, A.M.; Lundsgaard, A.-M.; Jordy, A.B.; Poulsen, S.K.; Stender, S.; Pilegaard, H.; Astrup, A.; Larsen, T.M.; Wojtaszewski, J.F.P.; Richter, E.A.; et al. New Nordic Diet–Induced Weight Loss Is Accompanied by Changes in Metabolism and AMPK Signaling in Adipose Tissue. J. Clin. Endocrinol. Metab. 2015, 100, 3509–3519. [Google Scholar] [CrossRef]
- Xu, X.J.; Apovian, C.; Hess, D.; Carmine, B.; Saha, A.; Ruderman, N. Improved Insulin Sensitivity 3 Months After RYGB Surgery Is Associated With Increased Subcutaneous Adipose Tissue AMPK Activity and Decreased Oxidative Stress. Diabetes 2015, 64, 3155–3159. [Google Scholar] [CrossRef] [Green Version]
- Angelini, G.; Castagneto Gissey, L.; Del Corpo, G.; Giordano, C.; Cerbelli, B.; Severino, A.; Manco, M.; Basso, N.; Birkenfeld, A.L.; Bornstein, S.R.; et al. New Insight into the Mechanisms of Ectopic Fat Deposition Improvement after Bariatric Surgery. Sci. Rep. 2019, 9, 17315. [Google Scholar] [CrossRef] [Green Version]
- García-Prieto, C.F.; Gil-Ortega, M.; Vega-Martín, E.; Ramiro-Cortijo, D.; Martín-Ramos, M.; Bordiú, E.; Sanchez-Pernaute, A.; Torres, A.; Aránguez, I.; Fernández-Alfonso, M.; et al. Beneficial Effect of Bariatric Surgery on Abnormal MMP-9 and AMPK Activities: Potential Markers of Obesity-Related CV Risk. Front. Physiol. 2019, 10, 553. [Google Scholar] [CrossRef]
- Yang, Z.; Kahn, B.B.; Shi, H.; Xue, B. Macrophage A1 AMP-Activated Protein Kinase (A1AMPK) Antagonizes Fatty Acid-Induced Inflammation through SIRT1. J. Biol. Chem. 2010, 285, 19051–19059. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bułdak, Ł.; Machnik, G.; Bułdak, R.J.; Łabuzek, K.; Bołdys, A.; Okopień, B. Exenatide and Metformin Express Their Anti-Inflammatory Effects on Human Monocytes/Macrophages by the Attenuation of MAPKs and NFκB Signaling. Naunyn-Schmiedeberg’s Arch. Pharmacol. 2016, 389, 1103–1115. [Google Scholar] [CrossRef] [PubMed]
- Boß, M.; Newbatt, Y.; Gupta, S.; Collins, I.; Brüne, B.; Namgaladze, D. AMPK-Independent Inhibition of Human Macrophage ER Stress Response by AICAR. Sci. Rep. 2016, 6, 32111. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Alhusaini, S.; McGee, K.; Schisano, B.; Harte, A.; McTernan, P.; Kumar, S.; Tripathi, G. Lipopolysaccharide, High Glucose and Saturated Fatty Acids Induce Endoplasmic Reticulum Stress in Cultured Primary Human Adipocytes: Salicylate Alleviates This Stress. Biochem. Biophys. Res. Commun. 2010, 397, 472–478. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sharma, N.K.; Das, S.K.; Mondal, A.K.; Hackney, O.G.; Chu, W.S.; Kern, P.A.; Rasouli, N.; Spencer, H.J.; Yao-Borengasser, A.; Elbein, S.C. Endoplasmic Reticulum Stress Markers Are Associated with Obesity in Nondiabetic Subjects. J. Clin. Endocrinol. Metab. 2008, 93, 4532–4541. [Google Scholar] [CrossRef] [PubMed]
- López-Domènech, S.; Abad-Jiménez, Z.; Iannantuoni, F.; de Marañón, A.M.; Rovira-Llopis, S.; Morillas, C.; Bañuls, C.; Víctor, V.M.; Rocha, M. Moderate Weight Loss Attenuates Chronic Endoplasmic Reticulum Stress and Mitochondrial Dysfunction in Human Obesity. Mol. Metab. 2019, 19, 24–33. [Google Scholar] [CrossRef]
- Kim, J.; Kundu, M.; Viollet, B.; Guan, K.-L. AMPK and MTOR Regulate Autophagy through Direct Phosphorylation of Ulk1. Nat. Cell Biol. 2011, 13, 132–141. [Google Scholar] [CrossRef] [Green Version]
- Egan, D.F.; Shackelford, D.B.; Mihaylova, M.M.; Gelino, S.; Kohnz, R.A.; Mair, W.; Vasquez, D.S.; Joshi, A.; Gwinn, D.M.; Taylor, R.; et al. Phosphorylation of ULK1 (HATG1) by AMP-Activated Protein Kinase Connects Energy Sensing to Mitophagy. Science 2011, 331, 456–461. [Google Scholar] [CrossRef] [Green Version]
- Weerasekara, V.K.; Panek, D.J.; Broadbent, D.G.; Mortenson, J.B.; Mathis, A.D.; Logan, G.N.; Prince, J.T.; Thomson, D.M.; Thompson, J.W.; Andersen, J.L. Metabolic-Stress-Induced Rearrangement of the 14-3-3ζ Interactome Promotes Autophagy via a ULK1- and AMPK-Regulated 14-3-3ζ Interaction with Phosphorylated Atg9. Mol. Cell Biol. 2014, 34, 4379–4388. [Google Scholar] [CrossRef] [Green Version]
- Kim, J.; Kim, Y.C.; Fang, C.; Russell, R.C.; Kim, J.H.; Fan, W.; Liu, R.; Zhong, Q.; Guan, K.-L. Differential Regulation of Distinct Vps34 Complexes by AMPK in Nutrient Stress and Autophagy. Cell 2013, 152, 290–303. [Google Scholar] [CrossRef] [Green Version]
- Soltis, A.R.; Kennedy, N.J.; Xin, X.; Zhou, F.; Ficarro, S.B.; Yap, Y.S.; Matthews, B.J.; Lauffenburger, D.A.; White, F.M.; Marto, J.A.; et al. Hepatic Dysfunction Caused by Consumption of a High-Fat Diet. Cell Rep. 2017, 21, 3317–3328. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yang, L.; Li, P.; Fu, S.; Calay, E.S.; Hotamisligil, G.S. Defective Hepatic Autophagy in Obesity Promotes ER Stress and Causes Insulin Resistance. Cell Metab. 2010, 11, 467–478. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ma, N.; Ma, R.; Tang, K.; Li, X.; He, B. Roux-En-Y Gastric Bypass in Obese Diabetic Rats Promotes Autophagy to Improve Lipid Metabolism through MTOR/P70S6K Signaling Pathway. J. Diabetes Res. 2020, 2020, 4326549. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, T.; Liu, J.; Tong, Q.; Lin, L. SIRT3 Acts as a Positive Autophagy Regulator to Promote Lipid Mobilization in Adipocytes via Activating AMPK. Int. J. Mol. Sci. 2020, 21, 372. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Deretic, V.; Saitoh, T.; Akira, S. Autophagy in Infection, Inflammation and Immunity. Nat. Rev. Immunol. 2013, 13, 722–737. [Google Scholar] [CrossRef] [PubMed]
- Dai, X.; Zhao, W.; Zhan, J.; Zeng, S.; Ran, D.; Zhang, H.; Song, Z.; Song, K.H.; Wu, L. B Cells Present Skewed Profile and Lose the Function of Supporting T Cell Inflammation after Roux-En-Y Gastric Bypass. Int. Immunopharmacol. 2017, 43, 16–22. [Google Scholar] [CrossRef] [PubMed]
- Laker, R.C.; Drake, J.C.; Wilson, R.J.; Lira, V.A.; Lewellen, B.M.; Ryall, K.A.; Fisher, C.C.; Zhang, M.; Saucerman, J.J.; Goodyear, L.J.; et al. Ampk Phosphorylation of Ulk1 Is Required for Targeting of Mitochondria to Lysosomes in Exercise-Induced Mitophagy. Nat. Commun. 2017, 8, 548. [Google Scholar] [CrossRef]
- Nguyen, T.N.; Padman, B.S.; Lazarou, M. Deciphering the Molecular Signals of PINK1/Parkin Mitophagy. Trends Cell Biol. 2016, 26, 733–744. [Google Scholar] [CrossRef]
- Sacks, J.; Mulya, A.; Fealy, C.E.; Huang, H.; Mosinski, J.D.; Pagadala, M.R.; Shimizu, H.; Batayyah, E.; Schauer, P.R.; Brethauer, S.A.; et al. Effect of Roux-En-Y Gastric Bypass on Liver Mitochondrial Dynamics in a Rat Model of Obesity. Physiol. Rep. 2018, 6, e13600. [Google Scholar] [CrossRef]
| Parameters | Before | After |
|---|---|---|
| n (females %) | 43 (83.7) | |
| Age (years) | 45.1 ± 11.4 | |
| Weight (kg) | 108.7 ± 15.6 | 79.2 ± 13.0 *** |
| BMI (kg/m2) | 39.6 ± 4.9 | 29.2 ± 4.4 *** |
| Waist (cm) | 115.0 ± 10.2 | 88.6 ± 11.5 *** |
| EWL (%) | 79.1 ± 30.6 | |
| SBP (mmHg) | 133.2 ± 15.6 | 123.8 ± 17.3 ** |
| DBP (mmHg) | 81.4 ± 10.7 | 73.9 ± 9.8 ** |
| Glucose (mg/dL) | 98.7 ± 26.3 | 86.0 ± 12.3 *** |
| Insulin (μU/mL) | 14.6 ± 7.8 | 7.1 ± 3.2 *** |
| HOMA-IR | 3.8 ± 3.5 | 1.5 ± 0.7 *** |
| HbA1c (%) | 5.5 ± 0.7 | 5.2 ± 0.5 *** |
| TC (mg/dL) | 187.0 ± 33.6 | 166.9 ± 27.7 ** |
| HDLc (mg/dL) | 47.0 ± 8.9 | 55.1 ± 10.0 *** |
| LDLc (mg/dL) | 122.6 ± 41.4 | 96.4 ± 21.4 *** |
| TG (mg/dL) | 96 (74, 143) | 78 (55, 100) ** |
| hsCRP (mg/L) | 3.7 (1.7, 6.3) | 0.6 (0.3, 1.2) *** |
| IL6 (pg/mL) | 4.0 ± 3.0 | 3.3 ± 2.3 * |
| IL1β (pg/mL) | 1.2 ± 0.9 | 1.0 ± 0.7 * |
| Treatment | ||
| Hypertension % (n) | 34.8 (15) | 9.3 (4) |
| Hyperlipidemia % (n) | 23.3 (10) | 9.3 (4) |
| T2D % (n) | 30.2 (13) | 4.6 (2) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Abad-Jiménez, Z.; López-Domènech, S.; García-Gargallo, C.; Vezza, T.; Gómez-Abril, S.Á.; Morillas, C.; Díaz-Pozo, P.; Falcón, R.; Bañuls, C.; Víctor, V.M.; et al. Roux-en-Y Gastric Bypass Modulates AMPK, Autophagy and Inflammatory Response in Leukocytes of Obese Patients. Biomedicines 2022, 10, 430. https://doi.org/10.3390/biomedicines10020430
Abad-Jiménez Z, López-Domènech S, García-Gargallo C, Vezza T, Gómez-Abril SÁ, Morillas C, Díaz-Pozo P, Falcón R, Bañuls C, Víctor VM, et al. Roux-en-Y Gastric Bypass Modulates AMPK, Autophagy and Inflammatory Response in Leukocytes of Obese Patients. Biomedicines. 2022; 10(2):430. https://doi.org/10.3390/biomedicines10020430
Chicago/Turabian StyleAbad-Jiménez, Zaida, Sandra López-Domènech, Celia García-Gargallo, Teresa Vezza, Segundo Ángel Gómez-Abril, Carlos Morillas, Pedro Díaz-Pozo, Rosa Falcón, Celia Bañuls, Víctor M. Víctor, and et al. 2022. "Roux-en-Y Gastric Bypass Modulates AMPK, Autophagy and Inflammatory Response in Leukocytes of Obese Patients" Biomedicines 10, no. 2: 430. https://doi.org/10.3390/biomedicines10020430
APA StyleAbad-Jiménez, Z., López-Domènech, S., García-Gargallo, C., Vezza, T., Gómez-Abril, S. Á., Morillas, C., Díaz-Pozo, P., Falcón, R., Bañuls, C., Víctor, V. M., & Rocha, M. (2022). Roux-en-Y Gastric Bypass Modulates AMPK, Autophagy and Inflammatory Response in Leukocytes of Obese Patients. Biomedicines, 10(2), 430. https://doi.org/10.3390/biomedicines10020430











