Prothrombin Time-International Normalized Ratio Predicts the Outcome of Atrial Fibrillation Patients Taking Rivaroxaban
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Study Outcomes
2.3. Covariates
2.4. Statistical Analysis
3. Results
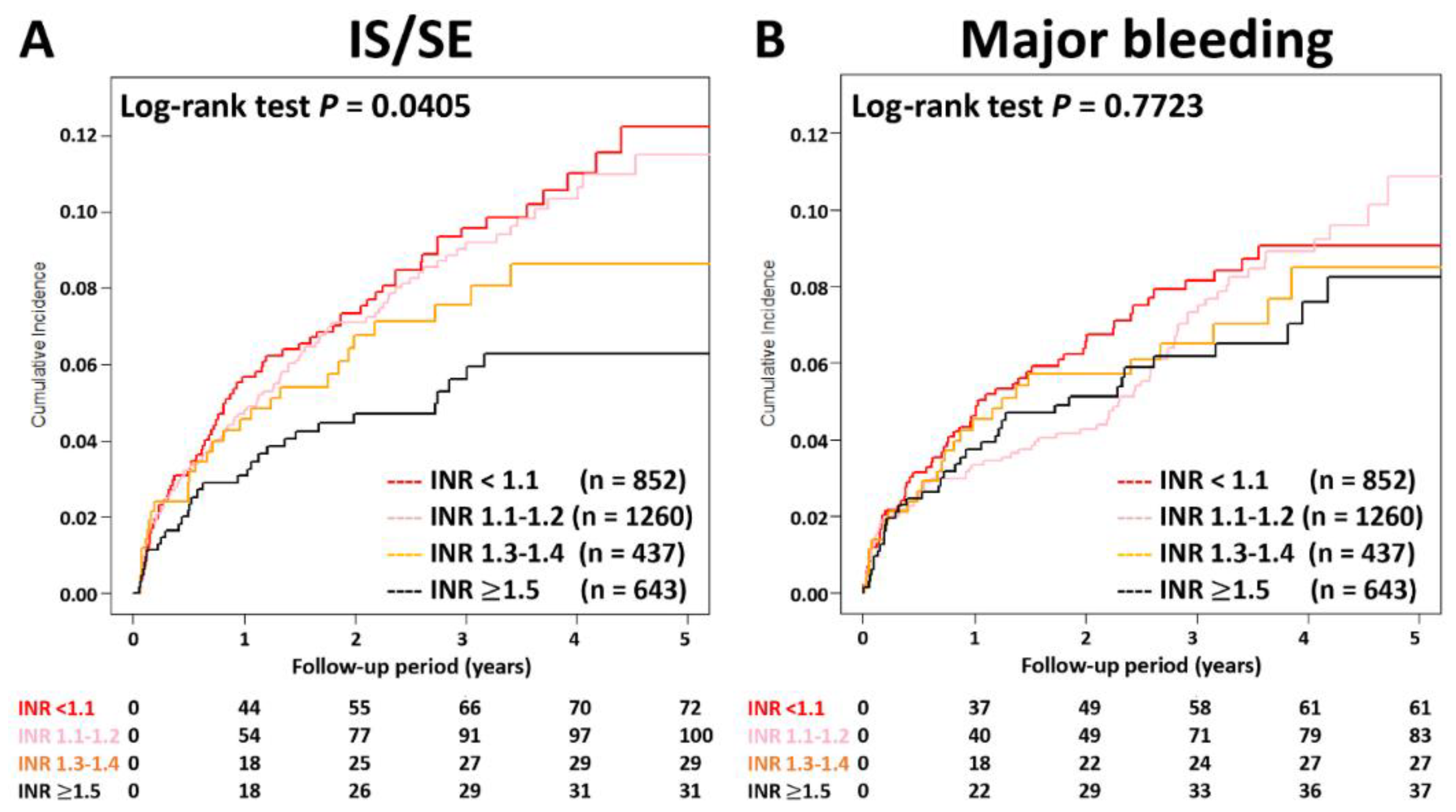
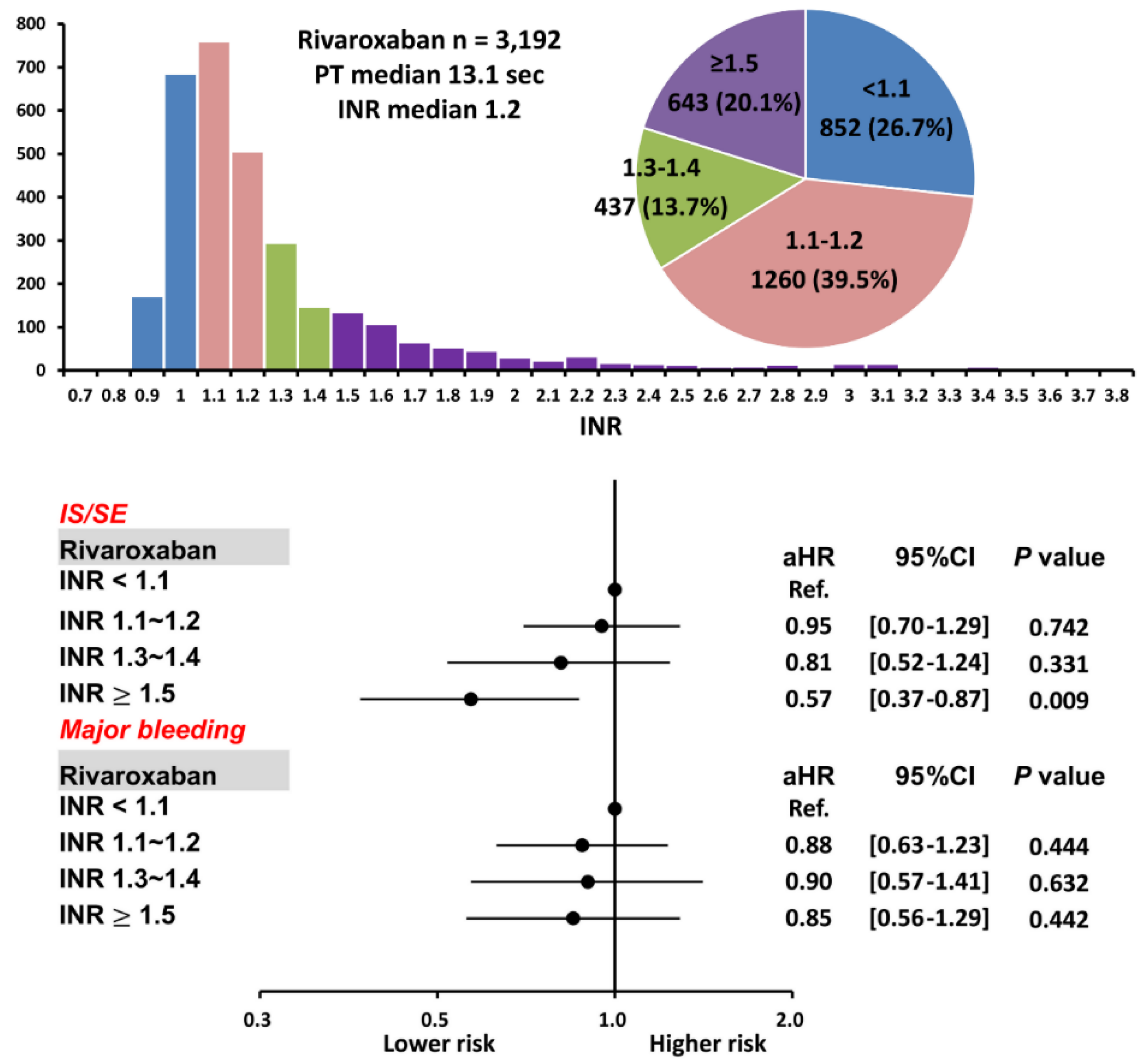
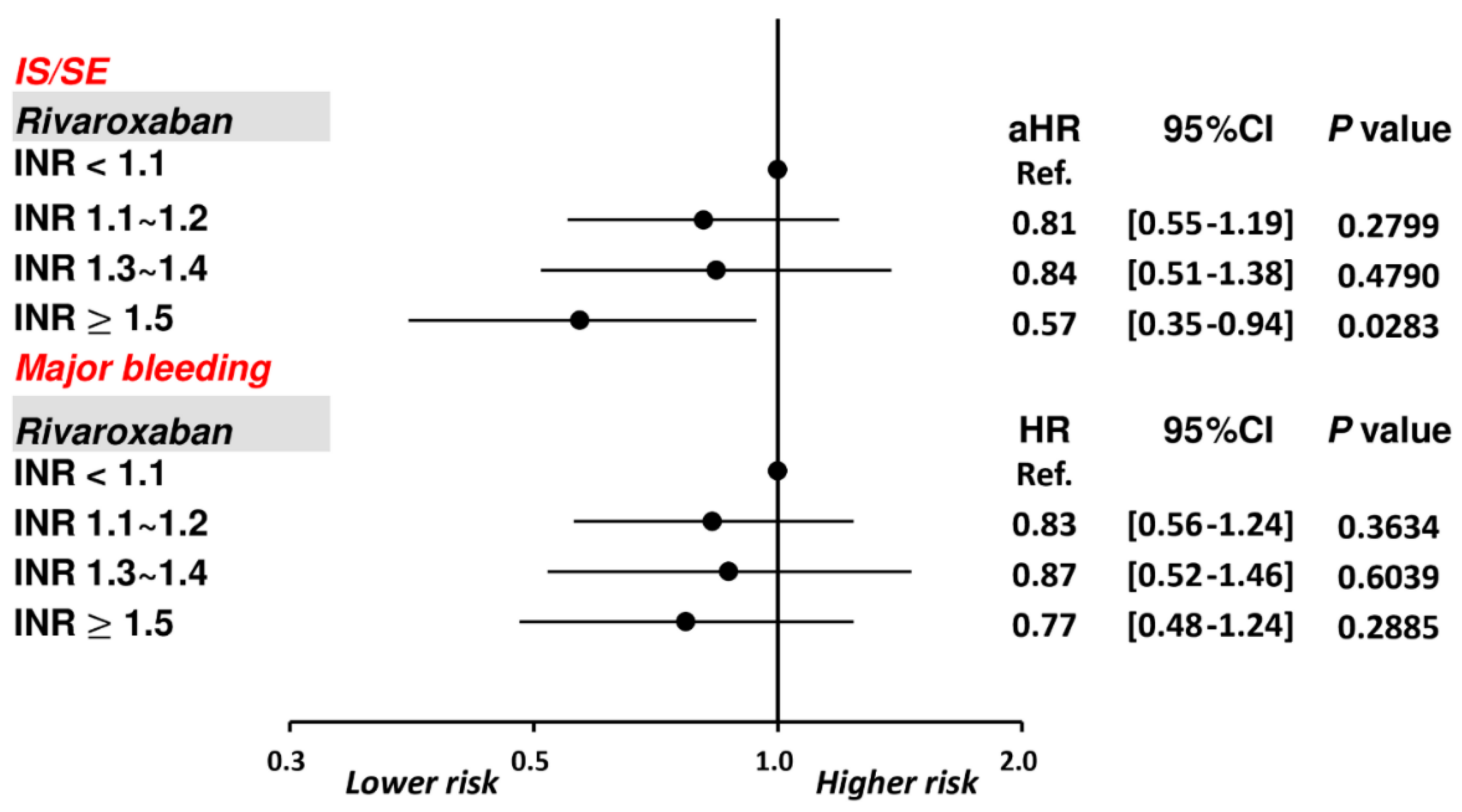
3.1. Patients Taking Rivaroxaban with Different INRs
3.2. “On-Label Dosing” of Rivaroxaban with Different INRs
3.3. Patients Taking Dabigatran with Different aPTT Ratios
4. Discussion
4.1. Factors Associated with the INR Values after Taking Rivaroxaban
4.2. The Role of PT-INR in Patients Taking Rivaroxaban
4.3. The Role of aPTT in Patients Taking Dabigatran
4.4. Clinical Implications
4.5. Study Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Chao, T.F.; Liu, C.J.; Tuan, T.C.; Chen, T.J.; Hsieh, M.H.; Lip, G.Y.; Chen, S.A. Lifetime Risks, Projected Numbers, and Adverse Outcomes in Asian Patients with Atrial Fibrillation: A Report From the Taiwan Nationwide AF Cohort Study. Chest 2018, 153, 453–466. [Google Scholar] [CrossRef] [PubMed]
- Steffel, J.; Verhamme, P.; Potpara, T.S.; Albaladejo, P.; Antz, M.; Desteghe, L.; Haeusler, K.G.; Oldgren, J.; Reinecke, H.; Roldan-Schilling, V.; et al. The 2018 European Heart Rhythm Association Practical Guide on the use of non-vitamin K antagonist oral anticoagulants in patients with atrial fibrillation. Eur. Heart. J. 2018, 39, 1330–1393. [Google Scholar] [CrossRef] [PubMed]
- Wańkowicz, P.; Nowacki, P.; Gołąb-Janowska, M. Risk factors for ischemic stroke in patients with non-valvular atrial fibrillation and therapeutic international normalized ratio range. Arch. Med. Sci. 2019, 15, 1217–1222. [Google Scholar] [CrossRef] [PubMed]
- Eikelboom, J.W.; Quinlan, D.J.; Hirsh, J.; Connolly, S.J.; Weitz, J.I. Laboratory Monitoring of Non-Vitamin K Antagonist Oral Anticoagulant Use in Patients with Atrial Fibrillation: A Review. JAMA Cardiol. 2017, 2, 566–574. [Google Scholar] [CrossRef] [PubMed]
- Reilly, P.A.; Lehr, T.; Haertter, S.; Connolly, S.J.; Yusuf, S.; Eikelboom, J.W.; Ezekowitz, M.D.; Nehmiz, G.; Wang, S.; Wallentin, L.; et al. The effect of dabigatran plasma concentrations and patient characteristics on the frequency of ischemic stroke and major bleeding in atrial fibrillation patients: The RE-LY Trial (Randomized Evaluation of Long-Term Anticoagulation Therapy). J. Am. Coll. Cardiol. 2014, 63, 321–328. [Google Scholar] [CrossRef]
- Mueck, W.; Stampfuss, J.; Kubitza, D.; Becka, M. Clinical pharmacokinetic and pharmacodynamic profile of rivaroxaban. Clin. Pharmacokinet. 2014, 53, 1–16. [Google Scholar] [CrossRef]
- Zhang, H.; Liu, Z.; Mu, G.; Wang, Z.; Zhou, S.; Xie, Q.; Ma, L.; Wang, Z.; Hu, K.; Gong, Y.; et al. Diagnostic performance of coagulation indices for direct oral anticoagulant concentration. Thromb. Res. 2020, 195, 171–179. [Google Scholar] [CrossRef]
- Liu, Z.; Zhang, H.; Xie, Q.; Mu, G.; Zhou, S.; Wang, Z.; Wang, Z.; Jiang, J.; Xiang, Q.; Cui, Y. Different Coagulation Indicators in Predicting Clinical Outcomes for Patients with Direct Oral Anticoagulants: A Systematic Review and Meta-analysis. Clin. Ther. 2020, 42, 2066–2081.e9. [Google Scholar] [CrossRef]
- Blann, A.D.; Lip, G.Y. Laboratory monitoring of the non-vitamin K oral anticoagulants. J. Am. Coll. Cardiol. 2014, 64, 1140–1142. [Google Scholar] [CrossRef]
- Testa, S.; Legnani, C.; Tripodi, A.; Paoletti, O.; Pengo, V.; Abbate, R.; Bassi, L.; Carraro, P.; Cini, M.; Paniccia, R.; et al. Poor comparability of coagulation screening test with specific measurement in patients receiving direct oral anticoagulants: Results from a multicenter/multiplatform study. J. Thromb. Haemost. 2016, 14, 2194–2201. [Google Scholar] [CrossRef]
- Tripodi, A.; Chantarangkul, V.; Legnani, C.; Testa, S.; Tosetto, A. Interlaboratory variability in the measurement of direct oral anticoagulants: Results from the external quality assessment scheme. J. Thromb. Haemost. 2018, 16, 565–570. [Google Scholar] [CrossRef]
- Garcia, D.; Barrett, Y.C.; Ramacciotti, E.; Weitz, J.I. Laboratory assessment of the anticoagulant effects of the next generation of oral anticoagulants. J. Thromb. Haemost. 2013, 11, 245–252. [Google Scholar] [CrossRef]
- Cuker, A.; Siegal, D.M.; Crowther, M.A.; Garcia, D.A. Laboratory measurement of the anticoagulant activity of the non-vitamin K oral anticoagulants. J. Am. Coll. Cardiol. 2014, 64, 1128–1139. [Google Scholar] [CrossRef]
- Liu, T.; Hui, J.; Hou, Y.Y.; Zou, Y.; Jiang, W.P.; Yang, X.J.; Wang, X.H. Meta-Analysis of Efficacy and Safety of Low-Intensity Warfarin Therapy for East Asian Patients with Nonvalvular Atrial Fibrillation. Am. J. Cardiol. 2017, 120, 1562–1567. [Google Scholar] [CrossRef]
- Eikelboom, J.W.; Hirsh, J. Monitoring unfractionated heparin with the aPTT: Time for a fresh look. Thromb. Haemost. 2006, 96, 547–552. [Google Scholar]
- Wang, C.L.; Wu, V.C.; Kuo, C.F.; Chu, P.H.; Tseng, H.J.; Wen, M.S.; Chang, S.H. Efficacy and Safety of Non-Vitamin K Antagonist Oral Anticoagulants in Atrial Fibrillation Patients with Impaired Liver Function: A Retrospective Cohort Study. J. Am. Heart Assoc. 2018, 7, e009263. [Google Scholar] [CrossRef]
- Pamukcu, B.; Lip, G.Y.; Lane, D.A. Simplifying stroke risk stratification in atrial fibrillation patients: Implications of the CHA2DS2-VASc risk stratification scores. Age Ageing 2010, 39, 533–535. [Google Scholar] [CrossRef]
- Pisters, R.; Lane, D.A.; Nieuwlaat, R.; de Vos, C.B.; Crijns, H.J.; Lip, G.Y. A novel user-friendly score (HAS-BLED) to assess 1-year risk of major bleeding in patients with atrial fibrillation: The Euro Heart Survey. Chest 2010, 138, 1093–1100. [Google Scholar] [CrossRef]
- Hori, M.; Matsumoto, M.; Tanahashi, N.; Momomura, S.; Uchiyama, S.; Goto, S.; Izumi, T.; Koretsune, Y.; Kajikawa, M.; Kato, M.; et al. Rivaroxaban vs. warfarin in Japanese patients with atrial fibrillation—The J-ROCKET AF study. Circ. J. Off. J. Jpn. Circ. Soc. 2012, 76, 2104–2111. [Google Scholar]
- Patel, M.R.; Mahaffey, K.W.; Garg, J.; Pan, G.; Singer, D.E.; Hacke, W.; Breithardt, G.; Halperin, J.L.; Hankey, G.J.; Piccini, J.P.; et al. Rivaroxaban versus warfarin in nonvalvular atrial fibrillation. N. Engl. J. Med. 2011, 365, 883–891. [Google Scholar] [CrossRef]
- Huang, J.-H.; Lin, Y.-K.; Chung, C.-C.; Hsieh, M.-H.; Chiu, W.-C.; Chen, Y.-J. Factors That Determine the Prothrombin Time in Patients with Atrial Fibrillation Receiving Rivaroxaban. Clin. Appl. Thromb. 2018, 24, 188S–193S. [Google Scholar] [CrossRef] [PubMed]
- Okada, A.; Sugano, Y.; Nagai, T.; Takashio, S.; Honda, S.; Asaumi, Y.; Aiba, T.; Noguchi, T.; Kusano, K.F.; Ogawa, H.; et al. Prognostic Value of Prothrombin Time International Normalized Ratio in Acute Decompensated Heart Failure—A Combined Marker of Hepatic Insufficiency and Hemostatic Abnormality. Circ. J. 2016, 80, 913–923. [Google Scholar] [CrossRef] [PubMed]
- Correale, M.; Tarantino, N.; Petrucci, R.; Tricarico, L.; Laonigro, I.; Di Biase, M.; Brunetti, N.D. Liver disease and heart failure: Back and forth. Eur. J. Intern. Med. 2018, 48, 25–34. [Google Scholar] [CrossRef] [PubMed]
- Ofek, F.; Bar Chaim, S.; Kronenfeld, N.; Ziv-Baran, T.; Berkovitch, M. International Normalized Ratio Is Significantly Elevated with Rivaroxaban and Apixaban Drug Therapies: A Retrospective Study. Clin. Ther. 2017, 39, 1003–1010. [Google Scholar] [CrossRef]
- Woodruff, A.E.; Wovkulich, M.M.; Mogle, B.; Hassan, A.K. Association between prothrombin time and bleeding in hospitalized patients receiving rivaroxaban. Am. J. Health Pharm. 2018, 75, 1783–1789. [Google Scholar] [CrossRef]
- Kawabata, M.; Goya, M.; Takahashi, Y.; Maeda, S.; Yagishita, A.; Sekigawa, M.; Akiyoshi, K.; Yamamoto, T.; Koyama, T.; Hirao, K. Excessive Prolongation of Coagulation Time During Treatment with Direct Oral Anticoagulants in Patients With Nonvalvular Atrial Fibrillation. Can. J. Cardiol. 2019, 35, 736–743. [Google Scholar] [CrossRef]
- Kawabata, M.; Yokoyama, Y.; Sasano, T.; Hachiya, H.; Tanaka, Y.; Yagishita, A.; Sugiyama, K.; Nakamura, T.; Suzuki, M.; Isobe, M.; et al. Bleeding events and activated partial thromboplastin time with dabigatran in clinical practice. J. Cardiol. 2013, 62, 121–126. [Google Scholar] [CrossRef][Green Version]
- Wańkowicz, P.; Staszewski, J.; Dębiec, A.; Nowakowska-Kotas, M.; Szylińska, A.; Rotter, I. Ischemic Stroke Risk Factors in Patients with Atrial Fibrillation Treated with New Oral Anticoagulants. J. Clin. Med. 2021, 10, 1223. [Google Scholar] [CrossRef]


| Rivaroxaban | |||||
|---|---|---|---|---|---|
| INR < 1.1 (n = 852) | INR = 1.1~1.2 (n = 1260) | INR = 1.3~1.4 (n = 437) | INR ≥ 1.5 (n = 643) | p Value (ANOVA) | |
| Coagulation test | |||||
| PT (s), median value [IQR] | 10.7 [10.4–11.1] | 12.4 [11.9–13.1] * | 14.6 [14.0–15.3] *,† | 19.7 [17.4–25.8] v,*,† | <0.01 |
| INR, median value | 1.01 [1.00–1.05] | 1.15 [1.10–1.20] * | 1.32 [1.30–1.40] *,† | 1.80 [1.60–2.30] v,*,† | <0.01 |
| Rivaroxaban Dosage | |||||
| Rivaroxaban 10 mg once daily, n (%) | 391 (45.89%) | 538 (42.7%) | 149 (34.1%) *,† | 199 (30.95%) *,† | <0.01 |
| Rivaroxaban 15 mg once daily, n (%) | 410 (48.12%) | 635 (50.4%) | 242 (55.38%) * | 370 (57.54%) *,† | <0.01 |
| Rivaroxaban 20 mg once daily, n (%) | 51 (5.99%) | 87 (6.9%) | 46 (10.53%) *,† | 74 (11.51%) *,† | <0.01 |
| Patients following ROCKET AF dose criteria, n (%) | 104 (12.21%) | 179 (14.21%) | 79 (18.08%) * | 134 (20.84%) *,† | <0.01 |
| Patients following with J-ROCKET AF dose criteria, n (%) | 448 (52.58%) | 666 (52.86%) | 244 (55.84%) | 349 (54.28%) | 0.66 |
| Patients following ROCKET AF or J-ROCKET AF dose criteria, n (%) | 552 (64.79%) | 845 (67.06%) | 323 (73.91%) *,† | 483 (75.12%) *,† | <0.01 |
| Patients taking off-label underdose, n (%) | 288 (33.8%) | 396 (31.43%) | 101 (23.11%) *,† | 140 (21.77%) *,† | <0.01 |
| Patients taking off-label overdose, n (%) | 3 (0.35%) | 6 (0.48%) | 4 (0.92%) | 5 (0.78%) | 0.50 |
| Baseline characteristics | |||||
| Age, yrs | 74.03 ± 10.12 | 75.24 ± 10.2 * | 75.19 ± 10.81 | 74.36 ± 10.67 | 0.03 |
| Female, n (%) | 393 (46.13%) | 496 (39.37%) * | 172 (39.36%) * | 286 (44.48%) † | 0.01 |
| CHA2DS2-VASc | 3.67 ± 1.63 | 3.63 ± 1.63 | 3.61 ± 1.6 | 3.6 ± 1.59 | 0.75 |
| HAS-BLED | 2.91 ± 1.23 | 2.9 ± 1.23 | 2.76 ± 1.16 *,† | 2.66 ± 1.19 *,†,v | <0.01 |
| Past medical history, n (%) | |||||
| Chronic lung disease | 271 (31.81%) | 440 (34.92%) | 135 (30.89%) | 184 (28.62%) † | 0.04 |
| Chronic liver disease | 199 (23.36%) | 303 (24.05%) | 82 (18.76%) † | 116 (18.04%) *,† | <0.01 |
| Congestive heart failure | 92 (10.8%) | 144 (11.43%) | 60 (13.73%) | 104 (16.17%) *,† | <0.01 |
| Hypertension | 668 (78.4%) | 953 (75.63%) | 335 (76.66%) | 484 (75.27%) | 0.43 |
| Hyperlipidemia | 385 (45.19%) | 565 (44.84%) | 187 (42.79%) | 258 (40.12%) † | 0.18 |
| Diabetes mellitus | 330 (38.73%) | 462 (36.67%) | 164 (37.53%) | 199 (30.95%) *,†,v | 0.01 |
| Previous stroke | 169 (19.84%) | 248 (19.68%) | 72 (16.48%) | 135 (21%) | 0.32 |
| Previous TIA | 14 (1.64%) | 31 (2.46%) | 14 (3.2%) | 11 (1.71%) | 0.22 |
| Ischemic heart disease | 103 (12.09%) | 153 (12.14%) | 53 (12.13%) | 70 (10.89%) | 0.86 |
| Gout | 139 (16.31%) | 230 (18.25%) | 88 (20.14%) | 108 (16.8%) | 0.32 |
| Peripheral artery disease | 3 (0.35%) | 3 (0.24%) | 1 (0.23%) | 0 (0%) | 0.55 |
| Malignancy | 132 (15.49%) | 223 (17.7%) | 89 (20.37%) * | 108 (16.8%) | 0.17 |
| Baseline laboratory data | |||||
| Hemoglobin, g/dL | 12.87 ± 2.12 | 12.7 ± 2.21 * | 12.31 ± 2.41 *,† | 12.51 ± 2.37 * | <0.01 |
| Platelet, × 1000/Ul | 204.11 ± 68.11 | 197.79 ± 70.32 | 187.88 ± 78.57 *,† | 195.89 ± 74.53 | 0.01 |
| eGFR, mL/min/1.73 m2 | 75.19 ± 31.48 | 75.97 ± 31.89 | 75.44 ± 35.1 | 71.99 ± 30.86 †,v | 0.08 |
| ALT, U/L | 27.79 ± 22.9 | 27.18 ± 20.16 | 27.06 ± 21.3 | 27.31 ± 23.86 | 0.93 |
| Height, cm | 160.16 ± 8.38 | 160.60 ± 8.89 | 160.78 ± 9.10 | 160.40 ± 9.46 | 0.71 |
| Body weight, kg | 64.35 ± 12.90 | 65.03 ± 13.95 | 64.30 ± 13.76 | 63.60 ± 13.33 | 0.27 |
| Baseline medications, n (%) | |||||
| Use of NSAIDs | 131 (15.38%) | 187 (14.84%) | 66 (15.1%) | 97 (15.09%) | 0.99 |
| Use of PPI | 140 (16.43%) | 159 (12.62%) * | 56 (12.81%) | 81 (12.6%) * | 0.06 |
| Use of ACEI/ARB | 465 (54.58%) | 720 (57.14%) | 247 (56.52%) | 385 (59.88%) * | 0.24 |
| Use of loop diuretics | 230 (27%) | 456 (36.19%) * | 157 (35.93%) * | 251 (39.04%) * | <0.01 |
| Use of amiodarone | 206 (24.18%) | 264 (20.95%) | 74 (16.93%) * | 125 (19.44%) * | 0.01 |
| Use of dronedarone | 24 (2.82%) | 44 (3.49%) | 19 (4.35%) | 23 (3.58%) | 0.55 |
| Use of quinidine | 0 (0%) | 2 (0.16%) | 1 (0.23%) | 2 (0.31%) | 0.48 |
| Use of beta-blocker | 462 (54.23%) | 716 (56.83%) | 250 (57.21%) | 395 (61.43%) * | 0.05 |
| Use of diltiazem | 161 (18.9%) | 278 (22.06%) | 94 (21.51%) | 110 (17.11%) | 0.05 |
| Use of verapamil | 40 (4.69%) | 65 (5.16%) | 21 (4.81%) | 23 (3.58%) | 0.49 |
| Use of digoxin | 136 (15.96%) | 235 (18.65%) | 92 (21.05%) * | 164 (25.51%) *,† | <0.01 |
| Use of statin | 280 (32.86%) | 437 (34.68%) | 150 (34.32%) | 212 (32.97%) | 0.80 |
| Use of Azi-/Clari-/Erythromycin | 15 (1.76%) | 36 (2.86%) | 6 (1.37%) | 9 (1.4%) † | 0.08 |
| Use of Itraconzaole | 1 (0.12%) | 1 (0.08%) | 0 (0%) | 0 (0%) | 0.30 |
| Use of cyclosporin | 3 (0.35%) | 1 (0.08%) | 2 (0.46%) | 1 (0.16%) | 0.75 |
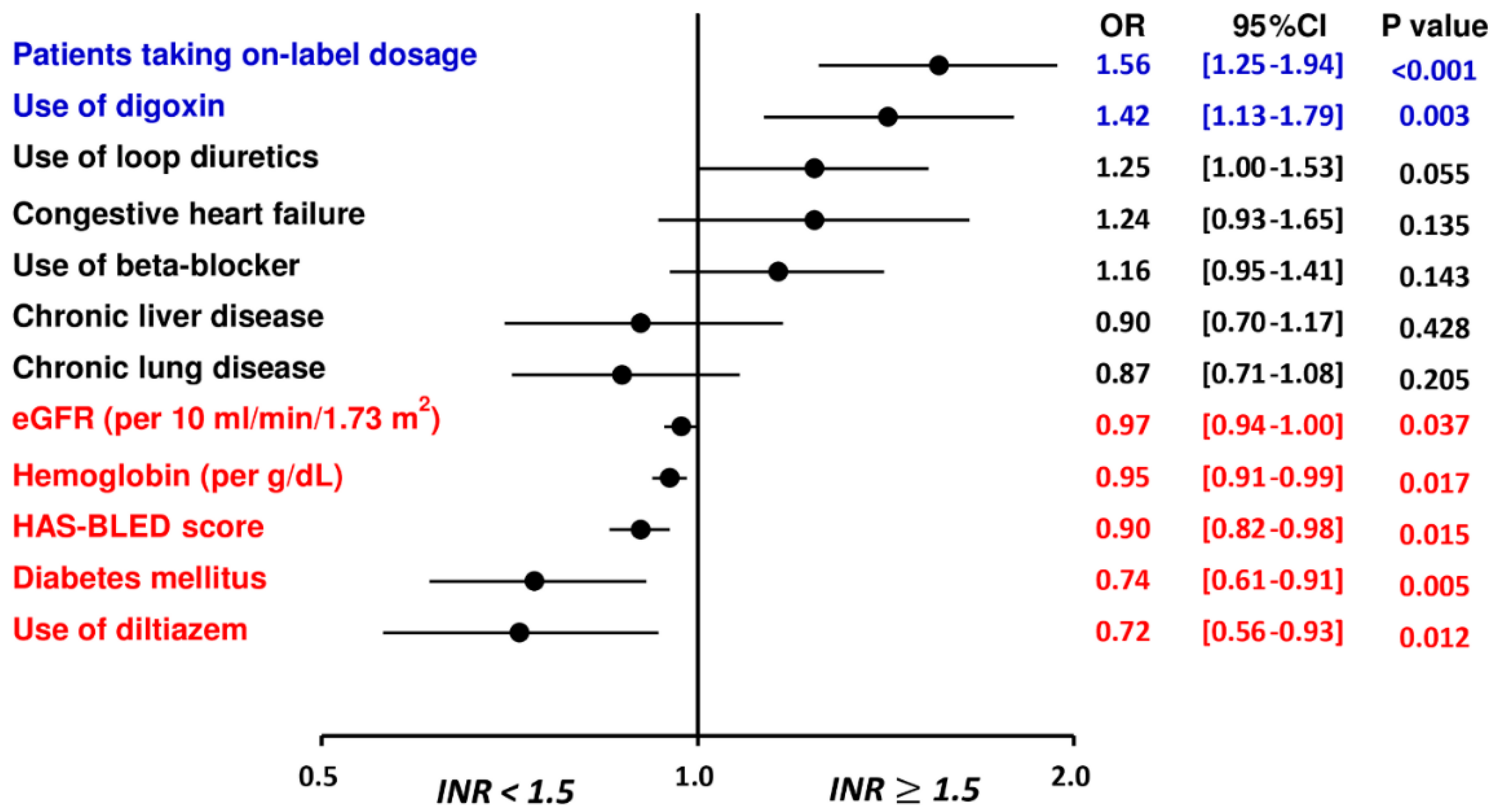
| Univariate Odds Ratio (OR) | Multivariate Odds Ratio (OR) | |||
|---|---|---|---|---|
| OR (95% CI) | p Value | OR (95% CI) | p Value | |
| Patients treated with on-label dosing of rivaroxaban | 1.45 (1.19–1.77) | <0.01 | 1.56 (1.25–1.94) | <0.01 |
| Age | 1.00 (0.99–1.00) | 0.31 | ||
| Female | 1.12 (0.94–1.34) | 0.19 | ||
| Body weight | 0.99 (0.99–1.00) | 0.11 | ||
| CHA2DS2-VASc score | 0.97 (0.92–1.03) | 0.32 | ||
| HAS-BLED score | 0.86 (0.80–0.93) | <0.01 | 0.90 (0.82–0.98) | 0.02 |
| Chronic lung disease | 0.81 (0.67–0.98) | 0.03 | 0.87 (0.71–1.08) | 0.21 |
| Chronic liver disease | 0.74 (0.59–0.92) | <0.01 | 0.90 (0.70–1.17) | 0.43 |
| Congestive heart failure | 1.47 (1.15–1.87) | <0.01 | 1.24 (0.93–1.65) | 0.14 |
| Diabetes mellitus | 0.75 (0.62–0.90) | <0.01 | 0.74 (0.61–0.91) | <0.01 |
| Hemoglobin, per g/dL | 0.95 (0.91–0.99) | 0.02 | 0.95 (0.91–0.99) | 0.02 |
| Platelet, per 10,000/uL | 1.00 (1.00–1.00) | 0.53 | ||
| eGFR, per 10 mL/min/1.73 m2 | 0.96 (0.93–0.99) | <0.01 | 0.97 (0.94–1.00) | 0.04 |
| Use of loop diuretics | 1.30 (1.08–1.55) | <0.01 | 1.24 (1.00–1.53) | 0.06 |
| Use of amiodarone | 0.89 (0.72–1.11) | 0.29 | ||
| Use of beta-blocker | 1.25 (1.05–1.49) | 0.01 | 1.16 (0.95–1.41) | 0.14 |
| Use of diltiazem | 0.78 (0.62–0.98) | 0.03 | 0.72 (0.56–0.93) | 0.01 |
| Use of digoxin | 1.54 (1.26–1.89) | <0.01 | 1.42 (1.13–1.79) | <0.01 |
| Dabigatran | |||||
|---|---|---|---|---|---|
| aPTT Ratio < 1.1 (n = 305) | aPTT Ratio = 1.1~1.2 (n = 234) | aPTT Ratio = 1.3~1.4 (n = 195) | aPTT Ratio ≥ 1.5 (n = 224) | p Value (ANOVA) | |
| Coagulation test | |||||
| aPPT (s), median value [IQR] | 27.9 [26.4–29.1] | 33.4 [32.2–34.8] * | 38.9 [37.5–40.5] *,† | 48.1 [44.8–54.1] v,*,† | <0.01 |
| aPTT ratio, median value | 1.00 [0.94–1.04] | 1.19 [1.15–1.24] * | 1.39 [1.34–1.45] *,† | 1.72 [1.60–1.93] v,*,† | <0.01 |
| Dabigatran Dosage | |||||
| Dabigatran 110 mg twice daily, n (%) | 254 (83%) | 197 (84%) | 165 (85%) | 181 (81%) | 0.72 |
| Dabigatran 150 mg twice daily, n (%) | 51 (17%) | 37 (16%) | 30 (15%) | 43 (19%) | 0.72 |
| Baseline characteristics | |||||
| Age, yrs | 72.39 ± 10.19 | 72.02 ± 9.82 | 74.20 ± 10.30 | 75.03 ± 10.13 *,† | <0.01 |
| Female, n (%) | 113 (37%) | 76 (32%) | 62 (32%) | 91 (41%) | 0.08 |
| CHA2DS2-VASc | 3.10 ± 1.32 | 3.09 ± 1.43 | 3.35 ± 1.52 | 3.39 ± 1.40 | 0.02 |
| HAS-BLED | 2.83 ± 1.16 | 2.73 ± 1.17 | 2.86 ± 1.26 | 2.88 ± 1.09 | 0.56 |
| Past medical history, n (%) | |||||
| Chronic lung disease | 91 (30%) | 68 (29%) | 56 (29%) | 72 (32%) | 0.86 |
| Chronic liver disease | 54 (18%) | 53 (23%) | 40 (21%) | 35 (16%) | 0.23 |
| Congestive heart failure | 19 (6%) | 13 (6%) | 17 (9%) | 18 (8%) | 0.52 |
| Hypertension | 238 (78%) | 184 (79%) | 142 (73%) | 167 (75%) | 0.41 |
| Hyperlipidemia | 138 (45%) | 112 (48%) | 88 (45%) | 99 (44%) | 0.88 |
| Diabetes mellitus | 120 (39%) | 75 (32%) | 71 (36%) | 89 (40%) | 0.27 |
| Previous stroke | 76 (25%) | 64 (27%) | 64 (33%) | 68 (30%) | 0.24 |
| Previous TIA | 7 (2%) | 11 (4%) | 11 (6%) | 10 (4%) | 0.28 |
| Ischemic heart disease | 25 (8%) | 13 (10%) | 13 (7%) | 24 (11%) | 0.43 |
| Gout | 47 (15%) | 38 (12%) | 38 (19%) | 43 (19%) | 0.13 |
| Peripheral artery disease | 1 (0%) | 1 (0%) | 1 (1%) | 0 (0%) | 0.57 |
| Malignancy | 53 (17%) | 28 (15%) | 28 (14%) | 38 (17%) | 0.72 |
| Baseline laboratory data | |||||
| Hemoglobin, g/dL | 13.25 ± 2.20 | 13.71 ± 1.91 | 13.51 ± 1.99 | 12.96 ± 2.15 † | <0.01 |
| Platelet, × 1000/Ul | 202.18 ± 65.80 | 203.17 ± 58.84 | 207.76 ± 77.75 | 207.84 ± 81.07 | 0.76 |
| eGFR, ml/min/1.73 m2 | 79.28 ± 31.13 | 80.66 ± 24.10 | 76.69 ± 24.86 | 74.90 ± 29.79 | 0.12 |
| ALT, U/L | 25.99 ± 29.96 | 26.73 ± 20.28 | 26.17 ± 19.41 | 24.73 ± 17.63 | 0.75 |
| Height, cm | 161.56 ± 9.38 | 162.51 ± 8.11 | 162.28 ± 8.30 | 159.60 ± 8.87 † | 0.01 |
| Body weight, kg | 65.61 ± 13.22 | 68.32 ± 15.69 | 66.41 ± 11.14 | 64.44 ± 13.69 | 0.06 |
| Baseline medications, n (%) | |||||
| Use of NSAIDs | 47 (15%) | 29 (12%) | 33 (17%) | 35 (16%) | 0.59 |
| Use of PPI | 38 (12%) | 19 (8%) | 18 (9%) | 22 (10%) | 0.38 |
| Use of ACEI/ARB | 161 (53%) | 122 (52%) | 110 (56%) | 120 (54%) | 0.83 |
| Use of loop diuretics | 70 (23%) | 43 (18%) | 43 (22%) | 54 (24%) | 0.47 |
| Use of amiodarone | 65 (21%) | 56 (24%) | 33 (17%) | 42 (19%) | 0.29 |
| Use of dronedarone | 4 (1%) | 4 (2%) | 0 (0%) | 1 (0%) | 0.22 |
| Use of quinidine | 0 (0%) | 0 (0%) | 0 (0%) | 0 (0%) | 1.00 |
| Use of beta-blocker | 170 (56%) | 120 (51%) | 97 (51%) | 127 (57%) | 0.38 |
| Use of diltiazem | 54 (18%) | 39 (17%) | 31 (16%) | 48 (21%) | 0.45 |
| Use of verapamil | 8 (3%) | 6 (3%) | 8 (4%) | 11 (5%) | 0.41 |
| Use of digoxin | 34 (11%) | 40 (17%) | 30 (15%) * | 36 (16%) | 0.21 |
| Use of statin | 108 (35%) | 91 (39%) | 72 (37%) | 84 (38%) | 0.87 |
| Use of Azi-/Clari-/Erythromycin | 6 (2%) | 5 (2%) | 2 (1%) | 4 (2%) | 0.83 |
| Use of Itraconzaole | 0 (0%) | 0 (0%) | 0 (0%) | 0 (0%) | 1.00 |
| Use of cyclosporin | 1 (0%) | 0 (0%) | 0 (0%) | 0 (0%) | 0.54 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chao, T.-F.; Chan, Y.-H.; Tsai, P.-C.; Lee, H.-F.; Chang, S.-H.; Kuo, C.-T.; Lip, G.Y.H.; Chen, S.-A.; Yeh, Y.-H. Prothrombin Time-International Normalized Ratio Predicts the Outcome of Atrial Fibrillation Patients Taking Rivaroxaban. Biomedicines 2022, 10, 3210. https://doi.org/10.3390/biomedicines10123210
Chao T-F, Chan Y-H, Tsai P-C, Lee H-F, Chang S-H, Kuo C-T, Lip GYH, Chen S-A, Yeh Y-H. Prothrombin Time-International Normalized Ratio Predicts the Outcome of Atrial Fibrillation Patients Taking Rivaroxaban. Biomedicines. 2022; 10(12):3210. https://doi.org/10.3390/biomedicines10123210
Chicago/Turabian StyleChao, Tze-Fan, Yi-Hsin Chan, Pei-Chien Tsai, Hsin-Fu Lee, Shang-Hung Chang, Chi-Tai Kuo, Gregory Y. H. Lip, Shih-Ann Chen, and Yung-Hsin Yeh. 2022. "Prothrombin Time-International Normalized Ratio Predicts the Outcome of Atrial Fibrillation Patients Taking Rivaroxaban" Biomedicines 10, no. 12: 3210. https://doi.org/10.3390/biomedicines10123210
APA StyleChao, T.-F., Chan, Y.-H., Tsai, P.-C., Lee, H.-F., Chang, S.-H., Kuo, C.-T., Lip, G. Y. H., Chen, S.-A., & Yeh, Y.-H. (2022). Prothrombin Time-International Normalized Ratio Predicts the Outcome of Atrial Fibrillation Patients Taking Rivaroxaban. Biomedicines, 10(12), 3210. https://doi.org/10.3390/biomedicines10123210








