Xenobiotic-Metabolizing Enzymes in Trematodes
Abstract
1. Introduction
2. Composition of the Xenobiotic-Metabolizing System in Trematodes
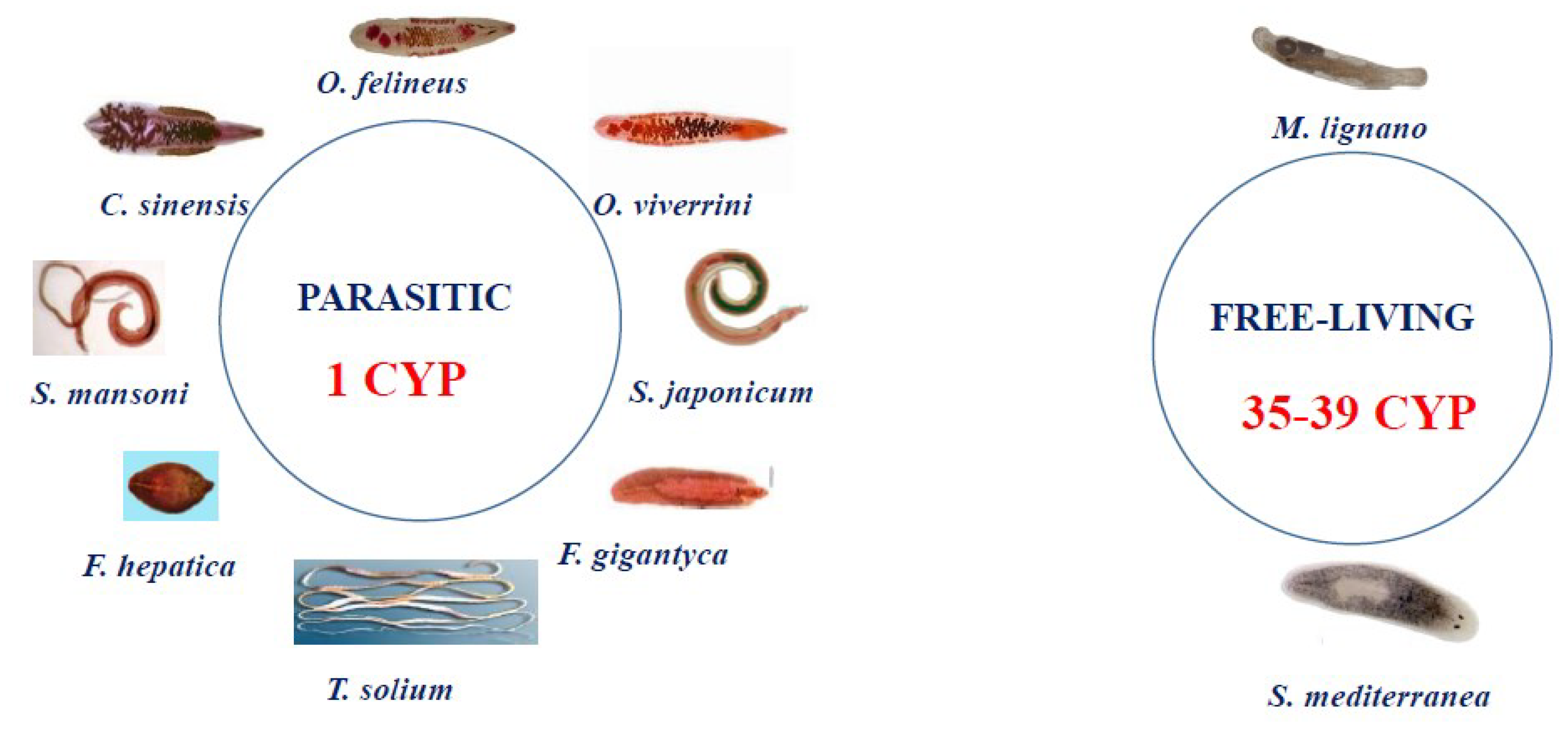
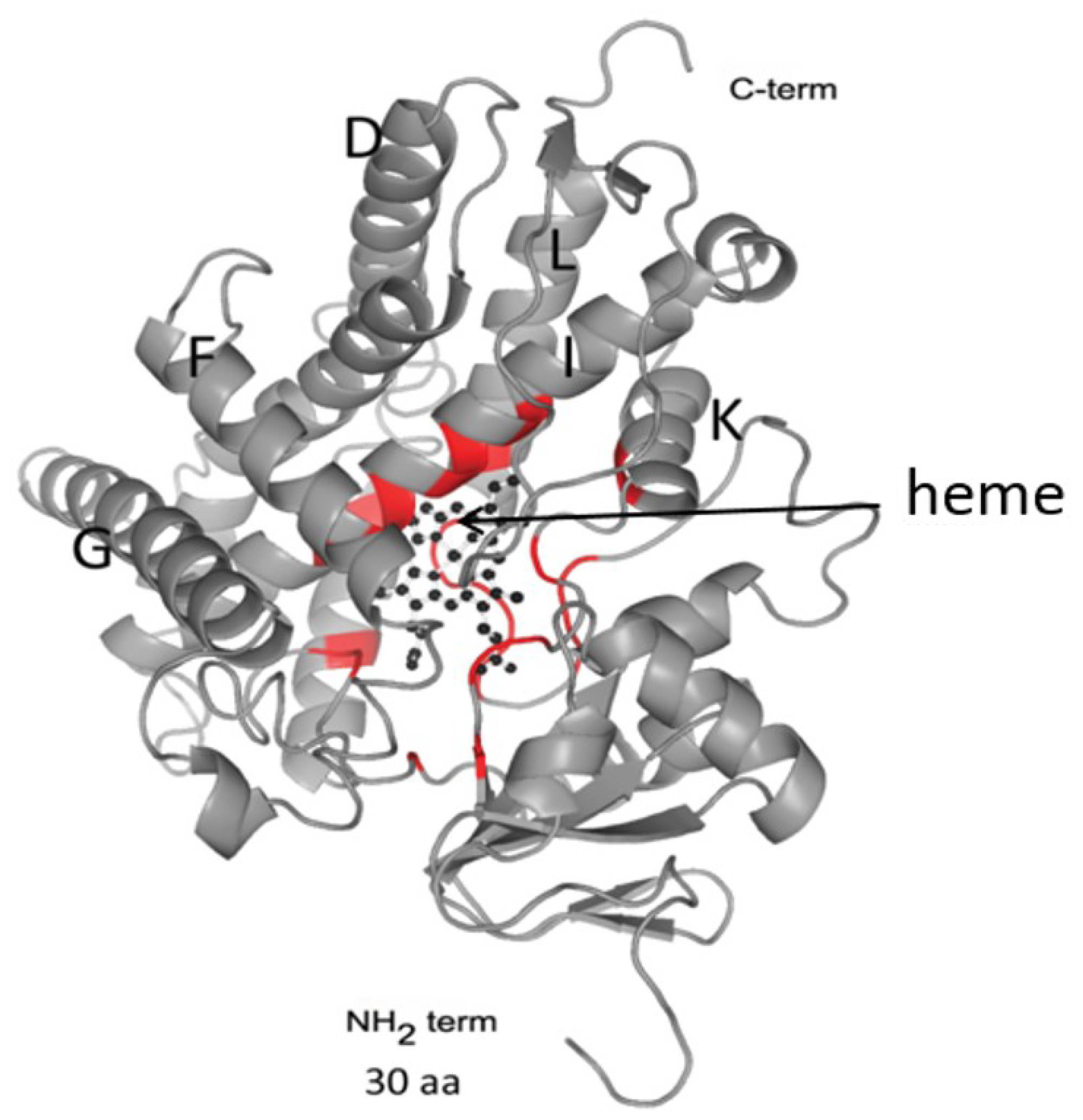
2.1. Phase I Enzymes
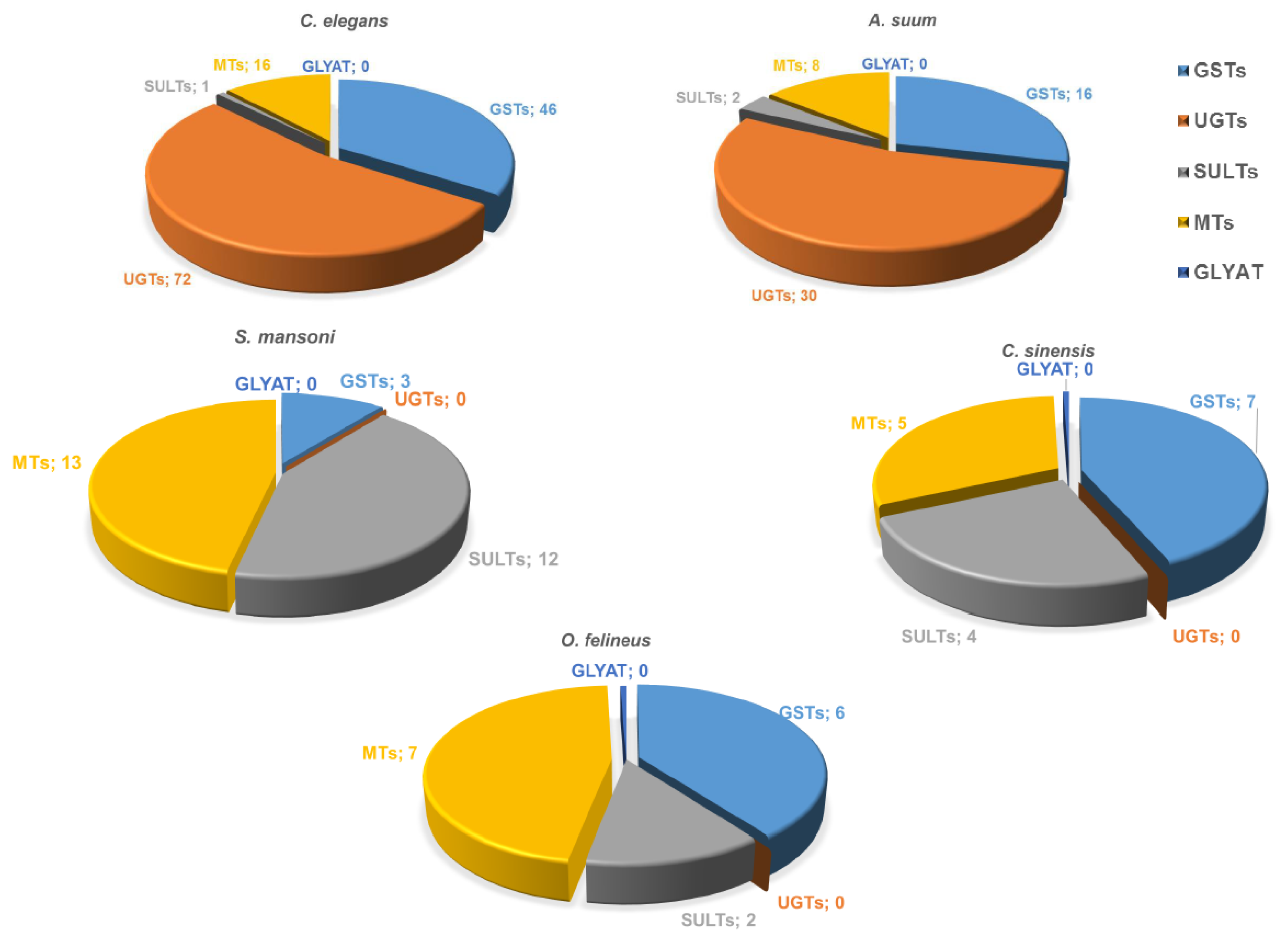
2.2. Phase II
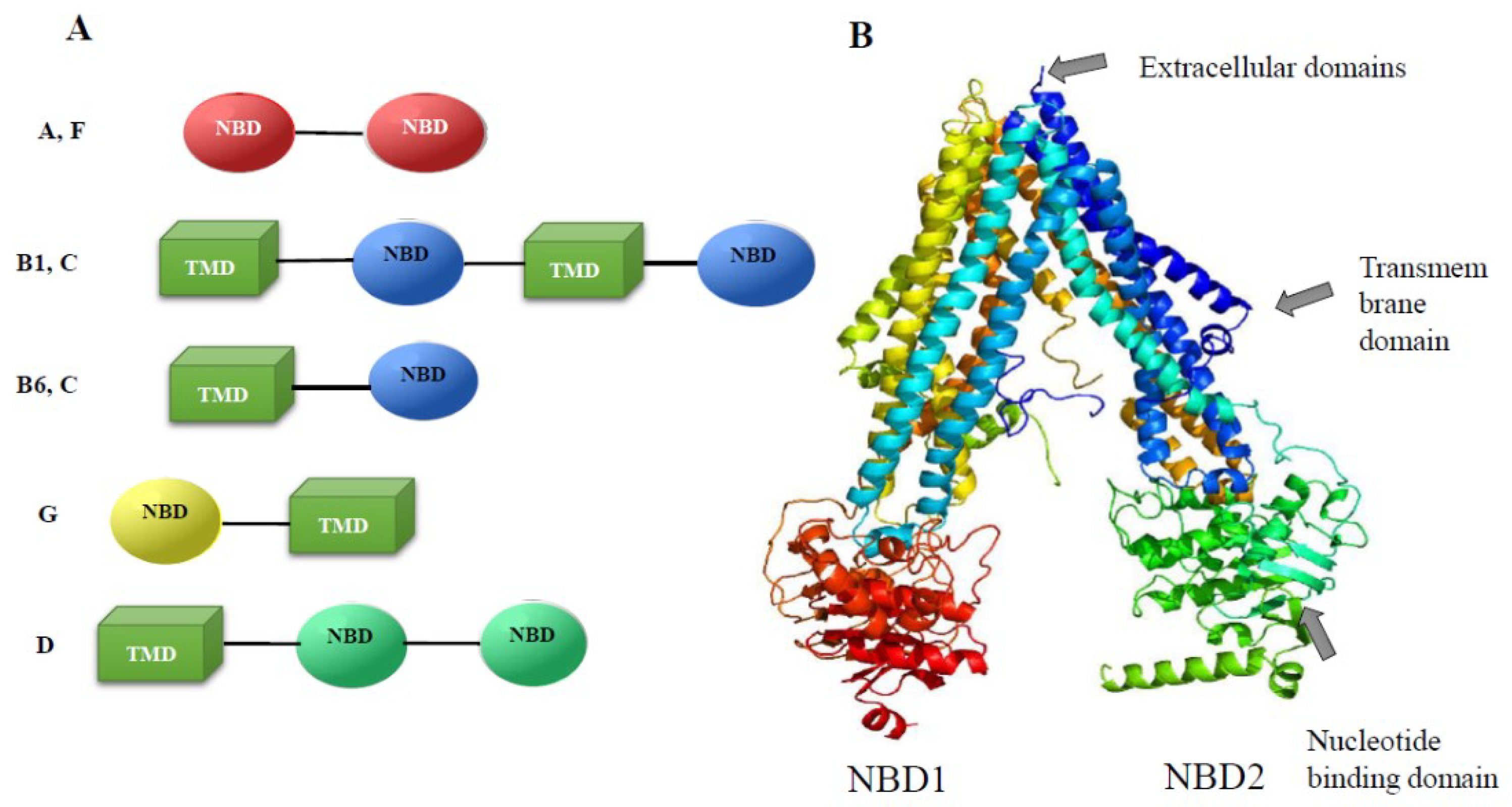
2.3. Identification of Phase III Genes
3. Functionality of Trematoda Xenobiotic-Metabolizing Enzymes: A Potential Link to Drug Resistance
3.1. Functionality of Phase I Enzymes
3.2. Functionality of Phase II Enzymes
3.3. Functionality of Phase III Proteins
4. Xenobiotic-Metabolizing Enzymes as Targets for Anthelmintic Therapy
4.1. Phase I Enzymes
4.2. Phase II Enzymes
4.3. Phase III Proteins
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Fürst, T.; Keiser, J.; Utzinger, J. Global burden of human food-borne trematodiasis: A systematic review and meta-analysis. Lancet Infect. Dis. 2012, 12, 210–221. [Google Scholar] [CrossRef] [PubMed]
- Cvilink, V.; Lamka, J.; Skálová, L. Xenobiotic metabolizing enzymes and metabolism of anthelminthics in helminths. Drug Metab. Rev. 2009, 41, 8–26. [Google Scholar] [CrossRef] [PubMed]
- Torres-Rivera, A.; Landa, A. Glutathione transferases from parasites: A biochemical view. Acta Trop. 2008, 105, 99–112. [Google Scholar] [CrossRef] [PubMed]
- Devine, C.; Brennan, G.P.; Lanusse, C.E.; Alvarez, L.I.; Trudgett, A.; Hoey, E.; Fairweather, I. Potentiation of triclabendazole action in vivo against a triclabendazole-resistant isolate of Fasciola hepatica following its co-administration with the metabolic inhibitor, ketoconazole. Vet. Parasitol. 2012, 184, 37–47. [Google Scholar] [CrossRef] [PubMed]
- Saeed, H.M.; Mostafa, M.H.; O’Connor, P.J.; Rafferty, J.A.; Doenhoff, M.J. Evidence for the presence of active cytochrome P450 systems in Schistosoma mansoni and Schistosoma haematobium adult worms. FEBS Lett. 2002, 519, 205–209. [Google Scholar] [CrossRef]
- Robinson, M.W.; Lawson, J.; Trudgett, A.; Hoey, E.M.; Fairweather, I. The comparative metabolism of triclabendazole sulphoxide by triclabendazole-susceptible and triclabendazole-resistant Fasciola hepatica. Parasitol. Res. 2004, 92, 205–210. [Google Scholar] [CrossRef]
- Solana, H.; Scarcella, S.; Virkel, G.; Ceriani, C.; Rodríguez, J.; Lanusse, C. Albendazole enantiomeric metabolism and binding to cytosolic proteins in the liver fluke Fasciola hepatica. Vet. Res. Commun. 2009, 33, 163–173. [Google Scholar] [CrossRef]
- Gouveia, M.J.; Pakharukova, M.Y.; Laha, T.; Sripa, B.; Maksimova, G.A.; Rinaldi, G.; Brindley, P.J.; Mordvinov, V.A.; Amaro, T.; Santos, L.L.; et al. Infection with Opisthorchis felineus induces intraepithelial neoplasia of the biliary tract in a rodent model. Carcinogenesis 2017, 38, 929–937. [Google Scholar] [CrossRef]
- Gouveia, M.J.; Santos, J.; Brindley, P.J.; Rinaldi, G.; Lopes, C.; Santos, L.L.; Da Costa, J.M.; Vale, N. Estrogen-like metabolites and DNA-adducts in urogenital schistosomiasis-associated bladder cancer. Cancer Lett. 2015, 359, 226–232. [Google Scholar] [CrossRef]
- Ortiz de Montellano, P.R. (Ed.) Cytochrome P450: Structure, Mechanism, and Biochemistry, 3rd ed.; Kluwer Academic: Amsterdam, The Netherlands, 2005; 912p. [Google Scholar]
- Matoušková, P.; Vokřál, I.; Lamka, J.; Skálová, L. The Role of Xenobiotic-Metabolizing Enzymes in Anthelmintic Deactivation and Resistance in Helminths. Trends Parasitol. 2016, 32, 481–491. [Google Scholar] [CrossRef]
- Pakharukova, M.Y.; Ershov, N.I.; Vorontsova, E.V.; Katokhin, A.V.; Merkulova, T.I.; Mordvinov, V.A. Cytochrome P450 in fluke Opisthorchis felineus: Identification and characterization. Mol. Biochem. Parasitol. 2012, 181, 190–194. [Google Scholar] [CrossRef]
- Pakharukova, M.Y.; Vavilin, V.A.; Sripa, B.; Laha, T.; Brindley, P.J.; Mordvinov, V.A. Functional Analysis of the Unique Cytochrome P450 of the Liver Fluke Opisthorchis felineus. PLoS Negl. Trop. Dis. 2015, 9, e0004258. [Google Scholar] [CrossRef]
- Ershov, N.I.; Mordvinov, V.A.; Prokhortchouk, E.B.; Pakharukova, M.Y.; Gunbin, K.V.; Ustyantsev, K.; Genaev, M.A.; Blinov, A.G.; Mazur, A.; Boulygina, E.; et al. New insights from Opisthorchis felineus genome: Update on genomics of the epidemiologically important liver flukes. BMC Genom. 2019, 20, 399. [Google Scholar] [CrossRef]
- Ziniel, P.D.; Karumudi, B.; Barnard, A.H.; Fisher, E.M.; Thatcher, G.R.; Podust, L.M.; Williams, D.L. The Schistosoma mansoni Cytochrome P450 (CYP3050A1) Is Essential for Worm Survival and Egg Development. PLoS Negl. Trop. Dis. 2015, 9, e0004279, Erratum in PLoS Negl. Trop. Dis. 2016, 10, e0004439. [Google Scholar] [CrossRef]
- Li, Y.; Feng, Y.; Wang, X.; Cui, J.; Deng, X.; Zhang, X. Adaptation of pine wood nematode Bursaphelenchus xylophilus to β-pinene stress. BMC Genom. 2020, 21, 478. [Google Scholar] [CrossRef]
- Sheehan, D.; Meade, G.; Foley, V.M.; Dowd, C.A. Structure, function and evolution of glutathione transferases: Implications for classification of non-mammalian members of an ancient enzyme superfamily. Biochem. J. 2001, 360 Pt 1, 1–16. [Google Scholar] [CrossRef]
- Hayes, J.D.; McLellan, L.I. Glutathione and glutathione-dependent enzymes represent a co-ordinately regulated defence against oxidative stress. Free Radic. Res. 1999, 31, 273–300. [Google Scholar] [CrossRef]
- Armstrong, R.N. Mechanistic diversity in a metalloenzyme superfamily. Biochemistry 2000, 39, 13625–13632. [Google Scholar] [CrossRef]
- Brophy, P.M.; Barrett, J. Glutathione transferase in helminths. Parasitology 1990, 100 Pt 2, 345–349. [Google Scholar] [CrossRef]
- Hayes, J.D.; Flanagan, J.U.; Jowsey, I.R. Glutathione transferases. Annu. Rev. Pharmacol. Toxicol. 2005, 45, 51–88. [Google Scholar] [CrossRef]
- Kim, J.G.; Ahn, C.S.; Kim, S.H.; Bae, Y.A.; Kwon, N.Y.; Kang, I.; Yang, H.J.; Sohn, W.M.; Kong, Y. Clonorchis sinensis omega-class glutathione transferases play major roles in the protection of the reproductive system during maturation and the response to oxidative stress. Parasites Vectors 2016, 9, 337. [Google Scholar] [CrossRef] [PubMed]
- Han, Y.H.; Hong, S.J.; Cheong, H.K.; Chung, Y.J. Crystal structures of 26kDa Clonorchis sinensis glutathione S-transferase reveal zinc binding and putative metal binding. Biochem. Biophys. Res. Commun. 2013, 438, 457–461. [Google Scholar] [CrossRef] [PubMed]
- Johnson, K.A.; Angelucci, F.; Bellelli, A.; Hervé, M.; Fontaine, J.; Tsernoglou, D.; Capron, A.; Trottein, F.; Brunori, M. Crystal structure of the 28 kDa glutathione S-transferase from Schistosoma haematobium. Biochemistry 2003, 42, 10084–10094. [Google Scholar] [CrossRef] [PubMed]
- Brophy, P.M.; Pritchard, D.I. Parasitic helminth glutathione S-transferases: An update on their potential as targets for immuno-and chemotherapy. Exp. Parasitol. 1994, 79, 89–96. [Google Scholar] [CrossRef] [PubMed]
- Laing, R.; Kikuchi, T.; Martinelli, A.; Tsai, I.J.; Beech, R.N.; Redman, E.; Holroyd, N.; Bartley, D.J.; Beasley, H.; Britton, C.; et al. The genome and transcriptome of Haemonchus contortus, a key model parasite for drug and vaccine discovery. Genome Biol. 2013, 14, R88. [Google Scholar] [CrossRef] [PubMed]
- Lindblom, T.H.; Dodd, A.K. Xenobiotic detoxification in the nematode Caenorhabditis elegans. J. Exp. Zool. 2006, 305A, 720–729. [Google Scholar] [CrossRef]
- Xu, C.; Li, C.Y.; Kong, A.T. Induction of Phase I, II and III Drug Metabolism/Transport by Xenobiotics. Arch. Pharm. Res. 2005, 28, 249–268. [Google Scholar] [CrossRef]
- Dassa, E.; Bouige, P. The ABC of ABCS: A phylogenetic and functional classification of ABC systems in living organisms. Res. Microbiol. 2001, 152, 211–229. [Google Scholar] [CrossRef]
- Juliano, R.L.; Ling, V. Surface glycoprotein modulating drug permeability in Chinese hamster ovary cell mutants. Biochim. Biophys. Acta 1976, 455, 152–162. [Google Scholar] [CrossRef]
- Greenberg, R.M. ABC multidrug transporters in schistosomes and other parasitic flatworms. Parasitol. Int. 2013, 62, 647–653. [Google Scholar] [CrossRef]
- Mordvinov, V.A.; Ershov, N.I.; Pirozhkova, D.S.; Pakharukov, Y.V.; Pakharukova, M.Y. ABC transporters in the liver fluke Opisthorchis felineus. Mol. Biochem. Parasitol. 2017, 216, 60–68. [Google Scholar] [CrossRef] [PubMed]
- McCall, L.I.; El Aroussi, A.; Choi, J.Y.; Vieira, D.F.; De Muylder, G.; Johnston, J.B.; Chen, S.; Kellar, D.; Siqueira-Neto, J.L.; Roush, W.R.; et al. Targeting Ergosterol biosynthesis in Leishmania donovani: Essentiality of sterol 14 alpha-demethylase. PLoS Negl. Trop. Dis. 2015, 9, e0003588. [Google Scholar] [CrossRef]
- Verma, S.; Mehta, A.; Shaha, C. CYP5122A1, a novel cytochrome P450 is essential for survival of Leishmania donovani. PLoS ONE 2011, 6, e25273. [Google Scholar] [CrossRef] [PubMed]
- Rottiers, V.; Motola, D.L.; Gerisch, B.; Cummins, C.L.; Nishiwaki, K.; Mangelsdorf, D.J.; Antebi, A. Hormonal control of C. elegans dauer formation and life span by a Rieske-like oxygenase. Dev. Cell 2006, 10, 473–482. [Google Scholar] [CrossRef]
- Kulas, J.; Schmidt, C.; Rothe, M.; Schunck, W.H.; Menzel, R. Cytochrome P450-dependent metabolism of eicosapentaenoic acid in the nematode Caenorhabditis elegans. Arch. Biochem. Biophys. 2008, 472, 65–75. [Google Scholar] [CrossRef] [PubMed]
- Shukla, R.; Chetri, P.B.; Sonkar, A.; Pakharukova, M.Y.; Mordvinov, V.A.; Tripathi, T. Identification of novel natural inhibitors of Opisthorchis felineus cytochrome P450 using structure-based screening and molecular dynamic simulation. J. Biomol. Struct. Dyn. 2018, 36, 3541–3556. [Google Scholar] [CrossRef]
- Sabat, J.; Stuehr, D.J.; Yeh, S.R.; Rousseau, D.L. Characterization of the Proximal Ligand in the P420 Form of Inducible Nitric Oxide Synthase. J. Am. Chem. Soc. 2009, 131, 12186–12192. [Google Scholar] [CrossRef][Green Version]
- Sun, L.; Wang, Z.; Jiang, H.; Tan, X.; Huang, Z. Novel Conformational Transitions of Human Cytochrome P450 2C8 during Thermal and Acid-induced Unfolding. Chin. J. Chem. 2010, 28, 1491–1502. [Google Scholar] [CrossRef]
- Driscoll, M.D.; McLean, K.J.; Cheesman, M.R.; Jowitt, T.A.; Howard, M.; Carroll, P.; Parish, T.; Munro, A.W. Expression and characterization of Mycobacterium tuberculosis CYP144: Common themes and lessons learned in the M. tuberculosis P450 enzyme family. Biochim. Biophys. Acta 2011, 1814, 76–87. [Google Scholar] [CrossRef]
- Dunford, A.J.; McLean, K.J.; Sabri, M.; Seward, H.E.; Heyes, D.J.; Scrutton, N.S.; Munro, A.W. Rapid P450 Heme Iron Reduction by Laser Photoexcitation of Mycobacterium tuberculosis CYP121 and CYP51B1. J. Biol. Chem. 2007, 282, 24816–24824. [Google Scholar] [CrossRef]
- Mordvinov, V.A.; Shilov, A.G.; Pakharukova, M.Y. Anthelmintic activity of cytochrome P450 inhibitors miconazole and clotrimazole: In-vitro effect on the liver fluke Opisthorchis felineus. Int. J. Antimicrob. Agents 2017, 50, 97–100. [Google Scholar] [CrossRef]
- AlGusbi, S.; Krücken, J.; Ramünke, S.; von Samson-Himmelstjerna, G.; Demeler, J. Analysis of putative inhibitors of anthelmintic resistance mechanisms in cattle gastrointestinal nematodes. Int. J. Parasitol. 2014, 44, 647–658. [Google Scholar] [CrossRef]
- Prchal, L.; Bártíková, H.; Bečanová, A.; Jirásko, R.; Vokřál, I.; Stuchlíková, L.; Skálová, L.; Kubíček, V.; Lamka, J.; Trejtnar, F.; et al. Biotransformation of anthelmintics and the activity of drug-metabolizing enzymes in the tapeworm Moniezia expansa. Parasitology 2015, 142, 648–659. [Google Scholar] [CrossRef]
- Hernandez, E.P.; Kusakisako, K.; Talactac, M.R.; Galay, R.L.; Hatta, T.; Fujisaki, K.; Tsuji, N.; Tanaka, T. Glutathione S-transferases play a role in the detoxification of flumethrin and chlorpyrifos in Haemaphysalis longicornis. Parasites Vectors 2018, 11, 460. [Google Scholar] [CrossRef]
- Kalita, J.; Shukla, R.; Shukla, H.; Gadhave, K.; Giri, R.; Tripathi, T. Comprehensive analysis of the catalytic and structural properties of a mu-class glutathione s-transferase from Fasciola gigantica. Sci. Rep. 2017, 7, 17547. [Google Scholar] [CrossRef]
- Mourão Mde, M.; Dinguirard, N.; Franco, G.R.; Yoshino, T.P. Role of the endogenous antioxidant system in the protection of Schistosoma mansoni primary sporocysts against exogenous oxidative stress. PLoS Negl. Trop. Dis. 2009, 3, e550. [Google Scholar] [CrossRef]
- Tang, C.L.; Zhou, H.H.; Zhu, Y.W.; Huang, J.; Wang, G.B. Glutathione S-transferase influences the fecundity of Schistosoma japonicum. Acta Trop. 2019, 191, 8–12. [Google Scholar] [CrossRef]
- Vokřál, I.; Jirásko, R.; Stuchlíková, L.; Bártíková, H.; Szotáková, B.; Lamka, J.; Várady, M.; Skálová, L. Biotransformation of albendazole and activities of selected detoxification enzymes in Haemonchus contortus strains susceptible and resistant to anthelmintics. Vet. Parasitol. 2013, 196, 373–381. [Google Scholar] [CrossRef]
- Stasiuk, S.J.; MacNevin, G.; Workentine, M.L.; Gray, D.; Redman, E.; Bartley, D.; Morrison, A.; Sharma, N.; Colwell, D.; Ro, D.K.; et al. Similarities and differences in the biotransformation and transcriptomic responses of Caenorhabditis elegans and Haemonchus contortus to five different benzimidazole drugs. Int. J. Parasitol. Drugs Drug Resist. 2019, 11, 13–29. [Google Scholar] [CrossRef]
- Kotze, A.C.; Ruffell, A.P.; Ingham, A.B. Phenobarbital induction and chemical synergism demonstrate the role of UDP-glucuronosyltransferases in detoxification of naphthalophos by Haemonchus contortus larvae. Antimicrob. Agents Chemother. 2014, 58, 7475–7483. [Google Scholar] [CrossRef]
- Cioli, D.; Pica-Mattoccia, L.; Moroni, R. Schistosoma mansoni: Hycanthone/oxamniquine resistance is controlled by a single autosomal recessive gene. Exp. Parasitol. 1992, 75, 425–432. [Google Scholar] [CrossRef] [PubMed]
- Cioli, D.; Pica-Mattoccia, L.; Rosenberg, S.; Archer, S. Evidence for the mode of antischistosomal action of hycanthone. Life Sci. 1985, 37, 161–167. [Google Scholar] [CrossRef]
- Valentim, C.L.; Cioli, D.; Chevalier, F.D.; Cao, X.; Taylor, A.B.; Holloway, S.P.; Pica-Mattoccia, L.; Guidi, A.; Basso, A.; Tsai, I.J.; et al. Genetic and molecular basis of drug resistance and species-specific drug action in schistosome parasites. Science 2013, 342, 1385–1389. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Yu, H.; Lv, Y.; Bushley, K.E.; Wickham, J.D.; Gao, S.; Hu, S.; Zhao, L.; Sun, J. Gene family expansion of pinewood nematode to detoxify its host defence chemicals. Mol. Ecol. 2020, 29, 940–955. [Google Scholar] [CrossRef] [PubMed]
- Robinson, M.W.; Menon, R.; Donnelly, S.M.; Dalton, J.P.; Ranganathan, S. An integrated transcriptomics and proteomics analysis of the secretome of the helminth pathogen Fasciola hepatica: Proteins associated with invasion and infection of the mammalian host. Mol. Cell. Proteom. 2009, 8, 1891–1907. [Google Scholar] [CrossRef] [PubMed]
- Pakharukova, M.Y.; Zaparina, O.G.; Kovner, A.V.; Mordvinov, V.A. Inhibition of Opisthorchis felineus glutathione-dependent prostaglandin synthase by resveratrol correlates with attenuation of cholangiocyte neoplasia in a hamster model of opisthorchiasis. Int. J. Parasitol. 2019, 49, 963–973. [Google Scholar] [CrossRef]
- Lifschitz, A.; Ballent, M.; Lanusse, C. Macrocyclic lactones and cellular transport-related drug interactions: A perspective from in vitro assays to nematode control in the field. Curr. Pharm. Biotechnol. 2012, 13, 912–923. [Google Scholar] [CrossRef]
- Janssen, I.J.; Krücken, J.; Demeler, J.; von Samson-Himmelstjerna, G. Caenorhabditis elegans: Modest increase of susceptibility to ivermectin in individual P-glycoprotein loss-of-function strains. Exp. Parasitol. 2013, 134, 171–177. [Google Scholar] [CrossRef]
- James, C.E.; Davey, M.W. Increased expression of ABC transport proteins is associated with ivermectin resistance in the model nematode Caenorhabditis elegans. Int. J. Parasitol. 2009, 39, 213–220. [Google Scholar] [CrossRef]
- Dai, F.; Yoo, W.G.; Lee, J.Y.; Lu, Y.; Pak, J.H.; Sohn, W.M.; Hong, S.J. Multidrug resistance-associated protein 4 is a bile transporter of Clonorchis sinensis simulated by in silico docking. Parasites Vectors 2017, 10, 578. [Google Scholar] [CrossRef]
- Dai, F.; Yoo, W.G.; Lee, J.Y.; Lu, Y.; Pak, J.H.; Sohn, W.-M.; Hong, S.J. Molecular and structural characteristics of multidrug resistance-associated protein 7 in Chinese liver fluke Clonorchis sinensis. Parasitol. Res. 2017, 116, 953–962. [Google Scholar] [CrossRef] [PubMed]
- Kasinathan, R.S.; Sharma, L.K.; Cunningham, C.; Webb, T.R.; Greenberg, R.M. Inhibition or knockdown of ABC transporters enhances susceptibility of adult and juvenile schistosomes to Praziquantel. PLoS Negl. Trop. Dis. 2014, 8, e3265. [Google Scholar] [CrossRef] [PubMed]
- Pinto-Almeida, A.; Mendes, T.; Armada, A.; Belo, S.; Carrilho, E.; Viveiros, M.; Afonso, A. The Role of Efflux Pumps in Schistosoma mansoni Praziquantel Resistant Phenotype. PLoS ONE 2015, 10, e0140147. [Google Scholar] [CrossRef] [PubMed]
- Kusel, J.R.; McVeigh, P.; Thornhill, J.A. The schistosome excretory system: A key to regulation of metabolism, drug excretion and host interaction. Trends Parasitol. 2009, 25, 353–358. [Google Scholar] [CrossRef] [PubMed]
- Solana, M.V.; Domínguez, M.F.; Scarcella, S.; Radio, S.; Smircich, P.; Fernández, S.; Solana, H.; Tort, J.F. Different SNPs in Fasciola hepatica P-glycoprotein from diverse Latin American populations are not associated with Triclabendazole resistance. Mol. Biochem. Parasitol. 2018, 224, 57–60. [Google Scholar] [CrossRef]
- Elliott, T.P.; Spithill, T.W. The T687G SNP in a P-glycoprotein gene of Fasciola hepatica is not associated with resistance to triclabendazole in two resistant Australian populations. Mol. Biochem. Parasitol. 2014, 198, 45–47. [Google Scholar] [CrossRef] [PubMed]
- Gunatilleke, S.S.; Calvet, C.M.; Johnston, J.B.; Chen, C.K.; Erenburg, G.; Gut, J.; Engel, J.C.; Ang, K.K.; Mulvaney, J.; Chen, S.; et al. Diverse inhibitor chemotypes targeting Trypanosoma cruzi CYP51. PLoS Negl. Trop. Dis. 2012, 6, e1736. [Google Scholar] [CrossRef]
- Pakharukova, M.Y.; Pakharukov, Y.V.; Mordvinov, V.A. Effects of miconazole/clotrimazole and praziquantel combinations against the liver fluke Opisthorchis felineus in vivo and in vitro. Parasitol. Res. 2018, 117, 2327–2331. [Google Scholar] [CrossRef]
- Anandanarayanan, A.; Raina, O.K.; Lalrinkima, H.; Rialch, A.; Sankar, M.; Varghese, A. RNA interference in Fasciola gigantica: Establishing and optimization of experimental RNAi in the newly excysted juveniles of the fluke. PLoS Negl. Trop. Dis. 2017, 11, e0006109. [Google Scholar] [CrossRef]
- Cheng, P.C.; Lin, C.N.; Peng, S.Y.; Kang, T.F.; Lee, K.M. Combined IL-12 Plasmid and Recombinant SjGST Enhance the Protective and Anti-pathology Effect of SjGST DNA Vaccine Against Schistosoma japonicum. PLoS Negl. Trop. Dis. 2016, 10, e0004459. [Google Scholar] [CrossRef]
- Shuxian, L.; Yongkang, H.; Guangchen, S.; Xing-song, L.; Yuxin, X.; McManus, D.P. Anti-fecundity immunity to Schistosoma japonicum induced in Chinese water buffaloes (Bos buffelus) after vaccination with recombinant 26 kDa glutathione-S-transferase (reSjc26GST). Vet. Parasitol. 1997, 69, 39–47. [Google Scholar] [CrossRef] [PubMed]
- Mbanefo, E.C.; Kumagai, T.; Kodama, Y.; Kurosaki, T.; Furushima-Shimogawara, R.; Cherif, M.S.; Mizukami, S.; Kikuchi, M.; Huy, N.T.; Ohta, N.; et al. Immunogenicity and anti-fecundity effect of nanoparticle coated glutathione S-transferase (SjGST) DNA vaccine against murine Schistosoma japonicum infection. Parasitol. Int. 2015, 64, 24–31. [Google Scholar] [CrossRef] [PubMed]
- Merrifield, M.; Hotez, P.J.; Beaumier, C.M.; Gillespie, P.; Strych, U.; Hayward, T.; Bottazzi, M.E. Advancing a vaccine to prevent human schistosomiasis. Vaccine 2016, 34, 2988–2991. [Google Scholar] [CrossRef] [PubMed]
- Savage, J.; Meaney, M.; Brennan, G.P.; Hoey, E.; Trudgett, A.; Fairweather, I. Disruption of vitellogenesis and spermatogenesis by triclabendazole (TCBZ) in a TCBZ-resistant isolate of Fasciola hepatica following incubation in vitro with a P-glycoprotein inhibitor. Parasitology 2014, 141, 1064–1079. [Google Scholar] [CrossRef] [PubMed]
- Savage, J.; Meaney, M.; Brennan, G.P.; Hoey, E.; Trudgett, A.; Fairweather, I. Effect of the P-glycoprotein inhibitor, R(+)-verapamil on the drug susceptibility of a triclabendazole-resistant isolate of Fasciola hepatica. Vet. Parasitol. 2013, 195, 72–86. [Google Scholar] [CrossRef] [PubMed]
- Vicente, B.; López-Abán, J.; Chaccour, J.; Hernández-Goenaga, J.; Nicolas, P.; Fernández-Soto, P.; Muro, A.; Chaccour, C. The effect of ivermectin alone and in combination with cobicistat or elacridar in experimental Schistosoma mansoni infection in mice. Sci. Rep. 2021, 11, 4476. [Google Scholar] [CrossRef] [PubMed]
- Vale, N.; Gouveia, M.J.; Botelho, M.; Sripa, B.; Suttiprapa, S.; Rinaldi, G.; Gomes, P.; Brindley, P.J.; Correia da Costa, J.M. Carcinogenic liver fluke Opisthorchis viverrini oxysterols detected by LC-MS/MS survey of soluble fraction parasite extract. Parasitol. Int. 2013, 62, 535–542. [Google Scholar] [CrossRef]
- Cavalieri, E.L.; Rogan, E.G. Unbalanced metabolism of endogenous estrogens in the etiology and prevention of human cancer. J. Steroid Biochem. Mol. Biol. 2011, 125, 169–180. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mordvinov, V.; Pakharukova, M. Xenobiotic-Metabolizing Enzymes in Trematodes. Biomedicines 2022, 10, 3039. https://doi.org/10.3390/biomedicines10123039
Mordvinov V, Pakharukova M. Xenobiotic-Metabolizing Enzymes in Trematodes. Biomedicines. 2022; 10(12):3039. https://doi.org/10.3390/biomedicines10123039
Chicago/Turabian StyleMordvinov, Viatcheslav, and Maria Pakharukova. 2022. "Xenobiotic-Metabolizing Enzymes in Trematodes" Biomedicines 10, no. 12: 3039. https://doi.org/10.3390/biomedicines10123039
APA StyleMordvinov, V., & Pakharukova, M. (2022). Xenobiotic-Metabolizing Enzymes in Trematodes. Biomedicines, 10(12), 3039. https://doi.org/10.3390/biomedicines10123039




