Non-Myelofibrosis Chronic Myeloproliferative Neoplasm Patients Show Better Seroconversion Rates after SARS-CoV-2 Vaccination Compared to Other Hematologic Diseases: A Multicentric Prospective Study of KroHem
Abstract
1. Introduction
2. Materials and Methods
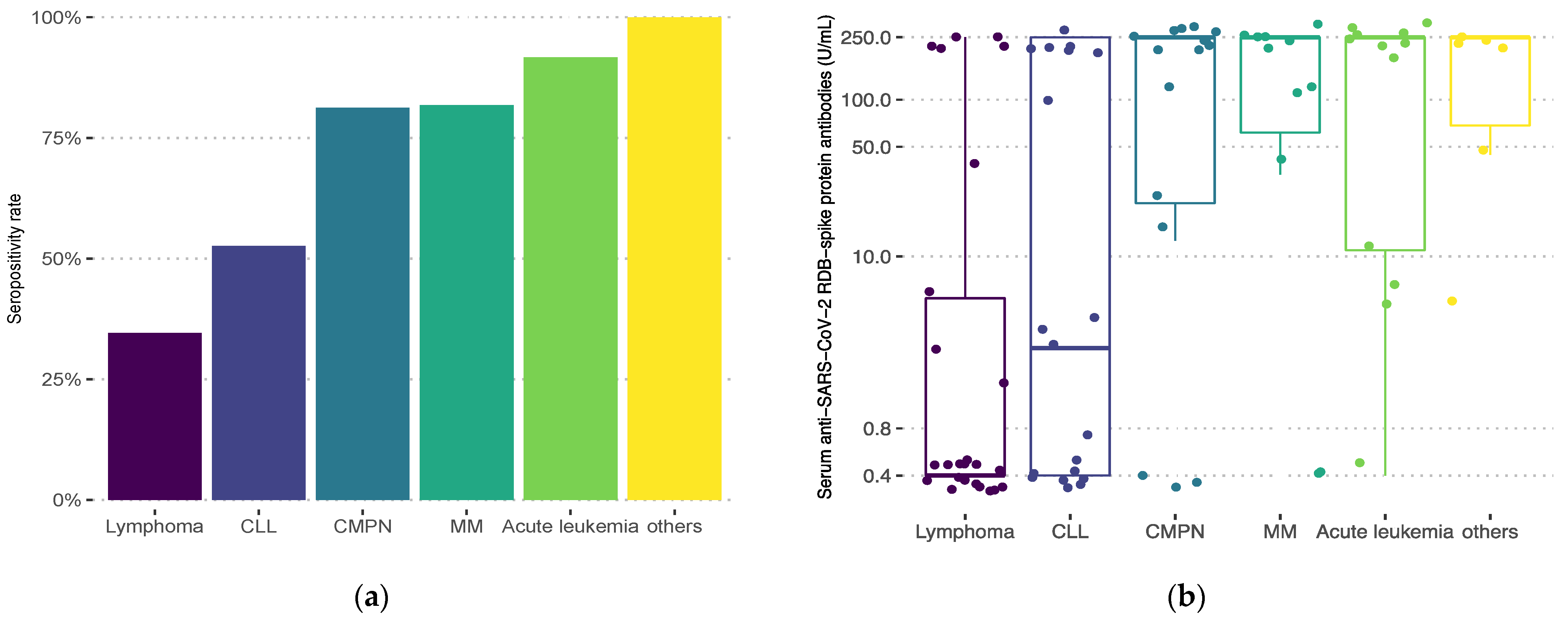
3. Results
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Vijenthira, A.; Gong, I.Y.; Fox, T.A.; Booth, S.; Cook, G.; Fattizzo, B.; Martín-Moro, F.; Razanamahery, J.; Riches, J.C.; Zwicker, J.; et al. Outcomes of patients with hematologic malignancies and COVID-19: A systematic review and meta-analysis of 3377 patients. Blood 2020, 136, 2881–2892. [Google Scholar] [CrossRef] [PubMed]
- Passamonti, F.; Cattaneo, C.; Arcaini, L.; Bruna, R.; Cavo, M.; Merli, F.; Angelucci, E.; Krampera, M.; Cairoli, R.; Della Porta, M.G.; et al. Clinical characteristics and risk factors associated with COVID-19 severity in patients with haematological malignancies in Italy: A retrospective, multicentre, cohort study. Lancet Haematol. 2020, 7, e737–e745. [Google Scholar] [CrossRef]
- García-Suárez, J.; de la Cruz, J.; Cedillo, Á.; Llamas, P.; Duarte, R.; Jiménez-Yuste, V.; Hernández-Rivas, J.Á.; Gil-Manso, R.; Kwon, M.; Sánchez-Godoy, P.; et al. Impact of hematologic malignancy and type of cancer therapy on COVID-19 severity and mortality: Lessons from a large population-based registry study. J. Hematol. Oncol. 2020, 13, 133. [Google Scholar] [CrossRef] [PubMed]
- Pagano, L.; Salmanton-García, J.; Marchesi, F.; Busca, A.; Corradini, P.; Hoenigl, M.; Klimko, N.; Koehler, P.; Pagliuca, A.; Passamonti, F.; et al. COVID-19 infection in adult patients with hematological malignancies: A European Hematology Association Survey (EPICOVIDEHA). J. Hematol. Oncol. 2021, 14, 168. [Google Scholar] [CrossRef] [PubMed]
- Polack, F.P.; Thomas, S.J.; Kitchin, N.; Absalon, J.; Gurtman, A.; Lockhart, S.; Perez, J.L.; Pérez Marc, G.; Moreira, E.D.; Zerbini, C.; et al. Safety and Efficacy of the BNT162b2 mRNA COVID-19 Vaccine. N. Engl. J. Med. 2020, 383, 2603–2615. [Google Scholar] [CrossRef] [PubMed]
- Baden, L.R.; El Sahly, H.M.; Essink, B.; Kotloff, K.; Frey, S.; Novak, R.; Diemert, D.; Spector, S.A.; Rouphael, N.; Creech, C.B.; et al. Efficacy and Safety of the mRNA-1273 SARS-CoV-2 Vaccine. N. Engl. J. Med. 2021, 384, 403–416. [Google Scholar] [CrossRef]
- Voysey, M.; Costa Clemens, S.A.; Madhi, S.A.; Weckx, L.Y.; Folegatti, P.M.; Aley, P.K.; Angus, B.; Baillie, V.L.; Barnabas, S.L.; Bhorat, Q.E.; et al. Single-dose administration and the influence of the timing of the booster dose on immunogenicity and efficacy of ChAdOx1 nCoV-19 (AZD1222) vaccine: A pooled analysis of four randomised trials. Lancet 2021, 397, 881–891. [Google Scholar] [CrossRef]
- Pullukcu, H.; Ertem, E.; Karaca, Y.; Yamazhan, T.; Sertoz, R.Y.; Altuglu, İ. Efficacy of accelerated hepatitis B vaccination program in patients being actively treated for hematologic malignancies. Int. J. Infect. Dis. 2008, 12, 166–170. [Google Scholar] [CrossRef][Green Version]
- La Torre, G. Influenza and pneumococcal vaccination in hematological malignancies: A systematic review of efficacy, effectiveness and safety. Mediterr. J. Hematol. Infect. Dis. 2016, 8, e2016044. [Google Scholar] [CrossRef][Green Version]
- Mullane, K.M.; Morrison, V.A.; Camacho, L.H.; Arvin, A.; McNeil, S.A.; Durrand, J.; Campbell, B.; Su, S.-C.; Chan, I.S.F.; Parrino, J.; et al. Safety and efficacy of inactivated varicella zoster virus vaccine in immunocompromised patients with malignancies: A two-arm, randomised, double-blind, phase 3 trial. Lancet Infect. Dis. 2019, 19, 1001–1012. [Google Scholar] [CrossRef]
- Abdul-Jawad, S.; Baù, L.; Alaguthurai, T.; del Molino del Barrio, I.; Laing, A.G.; Hayday, T.S.; Monin, L.; Muñoz-Ruiz, M.; McDonald, L.; Francos Quijorna, I.; et al. Acute Immune Signatures and Their Legacies in Severe Acute Respiratory Syndrome Coronavirus-2 Infected Cancer Patients. Cancer Cell 2021, 39, 257–275.e6. [Google Scholar] [CrossRef] [PubMed]
- Herzog Tzarfati, K.; Gutwein, O.; Apel, A.; Rahimi-Levene, N.; Sadovnik, M.; Harel, L.; Benveniste-Levkovitz, P.; Bar Chaim, A.; Koren-Michowitz, M. BNT162b2 COVID-19 vaccine is significantly less effective in patients with hematologic malignancies. Am. J. Hematol. 2021, 96, 1195–1203. [Google Scholar] [CrossRef] [PubMed]
- Agha, M.E.; Blake, M.; Chilleo, C.; Wells, A.; Haidar, G. Suboptimal Response to Coronavirus Disease 2019 Messenger RNA Vaccines in Patients With Hematologic Malignancies: A Need for Vigilance in the Postmasking Era. Open Forum Infect. Dis. 2021, 8, ofab353. [Google Scholar] [CrossRef] [PubMed]
- Rotterdam, J.; Thiaucourt, M.; Weiss, C.; Schwaab, J.; Reiter, A.; Kreil, S.; Steiner, L.; Fenchel, S.; Popp, H.D.; Hofmann, W.-K.; et al. Definition of factors associated with negative antibody response after COVID-19 vaccination in patients with hematological diseases. Ann. Hematol. 2022, 101, 1825–1834. [Google Scholar] [CrossRef]
- Malard, F.; Gaugler, B.; Gozlan, J.; Bouquet, L.; Fofana, D.; Siblany, L.; Eshagh, D.; Adotevi, O.; Laheurte, C.; Ricard, L.; et al. Weak immunogenicity of SARS-CoV-2 vaccine in patients with hematologic malignancies. Blood Cancer J. 2021, 11, 142. [Google Scholar] [CrossRef]
- Roche. 2022. Elecsys® Anti-SARS-CoV-2 S. Material Number 09289267190, Method Sheet 2022-07, V3.0. Material Number 09289275190, Method Sheet 2022-06, V4.0. Available online: https://diagnostics.roche.com/content/dam/diagnostics/Blueprint/en/pdf/cps/factsheet-elecsys-anti-sars-cov-2-s-mc--05522.pdf (accessed on 29 October 2022).
- Ferrari, D.; Clementi, N.; Spanò, S.M.; Albitar-Nehme, S.; Ranno, S.; Colombini, A.; Criscuolo, E.; Di Resta, C.; Tomaiuolo, R.; Viganó, M.; et al. Harmonization of six quantitative SARS-CoV-2 serological assays using sera of vaccinated subjects. Clin. Chim. Acta 2021, 522, 144–151. [Google Scholar] [CrossRef]
- Zou, G. A Modified Poisson Regression Approach to Prospective Studies with Binary Data. Am. J. Epidemiol. 2004, 159, 702–706. [Google Scholar] [CrossRef]
- Mulligan, M.J.; Lyke, K.E.; Kitchin, N.; Absalon, J.; Gurtman, A.; Lockhart, S.; Neuzil, K.; Raabe, V.; Bailey, R.; Swanson, K.A.; et al. Phase I/II study of COVID-19 RNA vaccine BNT162b1 in adults. Nature 2020, 586, 589–593. [Google Scholar] [CrossRef]
- Jackson, L.A.; Anderson, E.J.; Rouphael, N.G.; Roberts, P.C.; Makhene, M.; Coler, R.N.; McCullough, M.P.; Chappell, J.D.; Denison, M.R.; Stevens, L.J.; et al. An mRNA Vaccine against SARS-CoV-2—Preliminary Report. N. Engl. J. Med. 2020, 383, 1920–1931. [Google Scholar] [CrossRef] [PubMed]
- Folegatti, P.M.; Ewer, K.J.; Aley, P.K.; Angus, B.; Becker, S.; Belij-Rammerstorfer, S.; Bellamy, D.; Bibi, S.; Bittaye, M.; Clutterbuck, E.A.; et al. Safety and immunogenicity of the ChAdOx1 nCoV-19 vaccine against SARS-CoV-2: A preliminary report of a phase 1/2, single-blind, randomised controlled trial. Lancet 2020, 396, 467–478. [Google Scholar] [CrossRef]
- Hughes, A.; Clarson, J.; Tang, C.; Vidovic, L.; White, D.L.; Hughes, T.P.; Yong, A.S.M. CML patients with deep molecular responses to TKI have restored immune effectors and decreased PD-1 and immune suppressors. Blood 2017, 129, 1166–1176. [Google Scholar] [CrossRef] [PubMed]
- Breccia, M.; Abruzzese, E.; Accurso, V.; Attolico, I.; Barulli, S.; Bergamaschi, M.; Binotto, G.; Bocchia, M.; Bonifacio, M.; Caocci, G.; et al. COVID-19 infection in chronic myeloid leukaemia after one year of the pandemic in Italy. A Campus CML report. Br. J. Haematol. 2022, 196, 559–565. [Google Scholar] [CrossRef] [PubMed]
- Harrington, P.; de Lavallade, H.; Doores, K.J.; O’Reilly, A.; Seow, J.; Graham, C.; Lechmere, T.; Radia, D.; Dillon, R.; Shanmugharaj, Y.; et al. Single dose of BNT162b2 mRNA vaccine against SARS-CoV-2 induces high frequency of neutralising antibody and polyfunctional T-cell responses in patients with myeloproliferative neoplasms. Leukemia 2021, 35, 3573–3577. [Google Scholar] [CrossRef] [PubMed]
- Barbui, T.; Vannucchi, A.M.; Alvarez-Larran, A.; Iurlo, A.; Masciulli, A.; Carobbio, A.; Ghirardi, A.; Ferrari, A.; Rossi, G.; Elli, E.; et al. High mortality rate in COVID-19 patients with myeloproliferative neoplasms after abrupt withdrawal of ruxolitinib. Leukemia 2021, 35, 485–493. [Google Scholar] [CrossRef] [PubMed]
- Salisbury, R.A.; Curto-Garcia, N.; O’Sullivan, J.; Chen, F.; Polzella, P.; Godfrey, A.L.; Russell, J.; Knapper, S.; Willan, J.; Frewin, R.; et al. Results of a national UK physician reported survey of COVID-19 infection in patients with a myeloproliferative neoplasm. Leukemia 2021, 35, 2424–2430. [Google Scholar] [CrossRef]
- Maneikis, K.; Šablauskas, K.; Ringelevičiūtė, U.; Vaitekėnaitė, V.; Čekauskienė, R.; Kryžauskaitė, L.; Naumovas, D.; Banys, V.; Pečeliūnas, V.; Beinortas, T.; et al. Immunogenicity of the BNT162b2 COVID-19 mRNA vaccine and early clinical outcomes in patients with haematological malignancies in Lithuania: A national prospective cohort study. Lancet Haematol. 2021, 8, e583–e592. [Google Scholar] [CrossRef]
- Guglielmelli, P.; Mazzoni, A.; Maggi, L.; Kiros, S.T.; Zammarchi, L.; Pilerci, S.; Rocca, A.; Spinicci, M.; Borella, M.; Bartoloni, A.; et al. Impaired response to first SARS-CoV-2 dose vaccination in myeloproliferative neoplasm patients receiving ruxolitinib. Am. J. Hematol. 2021, 96, E408–E410. [Google Scholar] [CrossRef]
- Cattaneo, D.; Bucelli, C.; Cavallaro, F.; Consonni, D.; Iurlo, A. Impact of diagnosis and treatment on response to COVID-19 vaccine in patients with BCR-ABL1-negative myeloproliferative neoplasms. A single-center experience. Blood Cancer J. 2021, 11, 185. [Google Scholar] [CrossRef]
- Caocci, G.; Mulas, O.; Mantovani, D.; Costa, A.; Galizia, A.; Barabino, L.; Greco, M.; Murru, R.; La Nasa, G. Ruxolitinib does not impair humoral immune response to COVID-19 vaccination with BNT162b2 mRNA COVID-19 vaccine in patients with myelofibrosis. Ann. Hematol. 2022, 101, 929–931. [Google Scholar] [CrossRef]
- Elli, E.M.; Baratè, C.; Mendicino, F.; Palandri, F.; Palumbo, G.A. Mechanisms Underlying the Anti-inflammatory and Immunosuppressive Activity of Ruxolitinib. Front. Oncol. 2019, 9, 1186. [Google Scholar] [CrossRef]
- Larsen, T.S.; Christensen, J.H.; Hasselbalch, H.C.; Pallisgaard, N. The JAK2 V617F mutation involves B- and T-lymphocyte lineages in a subgroup of patients with Philadelphia-chromosome negative chronic myeloproliferative disorders. Br. J. Haematol. 2007, 136, 745–751. [Google Scholar] [CrossRef] [PubMed]
- Tefferi, A.; Pardanani, A. Serious Adverse Events During Ruxolitinib Treatment Discontinuation in Patients With Myelofibrosis. Mayo Clin. Proc. 2011, 86, 1188–1191. [Google Scholar] [CrossRef] [PubMed]
- Han, M.K.; Antila, M.; Ficker, J.H.; Gordeev, I.; Guerreros, A.; Bernus, A.L.; Roquilly, A.; Sifuentes-Osornio, J.; Tabak, F.; Teijeiro, R.; et al. Ruxolitinib in addition to standard of care for the treatment of patients admitted to hospital with COVID-19 (RUXCOVID): A randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Rheumatol. 2022, 4, e351–e361. [Google Scholar] [CrossRef]
- von Lilienfeld-Toal, M.; Vehreschild, J.J.; Cornely, O.; Pagano, L.; Compagno, F.; Hirsch, H.H. Frequently asked questions regarding SARS-CoV-2 in cancer patients—Recommendations for clinicians caring for patients with malignant diseases. Leukemia 2020, 34, 1487–1494. [Google Scholar] [CrossRef] [PubMed]
- Terpos, E.; Gavriatopoulou, M.; Fotiou, D.; Giatra, C.; Asimakopoulos, I.; Dimou, M.; Sklirou, A.D.; Ntanasis-Stathopoulos, I.; Darmani, I.; Briasoulis, A.; et al. Poor Neutralizing Antibody Responses in 132 Patients with CLL, NHL and HL after Vaccination against SARS-CoV-2: A Prospective Study. Cancers 2021, 13, 4480. [Google Scholar] [CrossRef] [PubMed]
- Ghione, P.; Gu, J.J.; Attwood, K.; Torka, P.; Goel, S.; Sundaram, S.; Mavis, C.; Johnson, M.; Thomas, R.; McWhite, K.; et al. Impaired humoral responses to COVID-19 vaccination in patients with lymphoma receiving B-cell–directed therapies. Blood 2021, 138, 811–814. [Google Scholar] [CrossRef]
- Pimpinelli, F.; Marchesi, F.; Piaggio, G.; Giannarelli, D.; Papa, E.; Falcucci, P.; Pontone, M.; Di Martino, S.; Laquintana, V.; La Malfa, A.; et al. Fifth-week immunogenicity and safety of anti-SARS-CoV-2 BNT162b2 vaccine in patients with multiple myeloma and myeloproliferative malignancies on active treatment: Preliminary data from a single institution. J. Hematol. Oncol. 2021, 14, 81. [Google Scholar] [CrossRef]
- Van Oekelen, O.; Gleason, C.R.; Agte, S.; Srivastava, K.; Beach, K.F.; Aleman, A.; Kappes, K.; Mouhieddine, T.H.; Wang, B.; Chari, A.; et al. Highly variable SARS-CoV-2 spike antibody responses to two doses of COVID-19 RNA vaccination in patients with multiple myeloma. Cancer Cell 2021, 39, 1028–1030. [Google Scholar] [CrossRef]
- Redjoul, R.; Le Bouter, A.; Beckerich, F.; Fourati, S.; Maury, S. Antibody response after second BNT162b2 dose in allogeneic HSCT recipients. Lancet 2021, 398, 298–299. [Google Scholar] [CrossRef]
- Maillard, A.; Redjoul, R.; Klemencie, M.; Labussière Wallet, H.; Le Bourgeois, A.; D’Aveni, M.; Huynh, A.; Berceanu, A.; Marchand, T.; Chantepie, S.; et al. Antibody response after 2 and 3 doses of SARS-CoV-2 mRNA vaccine in allogeneic hematopoietic cell transplant recipients. Blood 2022, 139, 134–137. [Google Scholar] [CrossRef]
- Piñana, J.L.; López-Corral, L.; Martino, R.; Montoro, J.; Vazquez, L.; Pérez, A.; Martin-Martin, G.; Facal-Malvar, A.; Ferrer, E.; Pascual, M.; et al. SARS-CoV-2-reactive antibody detection after SARS-CoV-2 vaccination in hematopoietic stem cell transplant recipients: Prospective survey from the Spanish Hematopoietic Stem Cell Transplantation and Cell Therapy Group. Am. J. Hematol. 2022, 97, 30–42. [Google Scholar] [CrossRef] [PubMed]
- Emary, K.R.W.; Golubchik, T.; Aley, P.K.; Ariani, C.V.; Angus, B.; Bibi, S.; Blane, B.; Bonsall, D.; Cicconi, P.; Charlton, S.; et al. Efficacy of ChAdOx1 nCoV-19 (AZD1222) vaccine against SARS-CoV-2 variant of concern 202012/01 (B.1.1.7): An exploratory analysis of a randomised controlled trial. Lancet 2021, 397, 1351–1362. [Google Scholar] [CrossRef]
- Lopez Bernal, J.; Andrews, N.; Gower, C.; Gallagher, E.; Simmons, R.; Thelwall, S.; Stowe, J.; Tessier, E.; Groves, N.; Dabrera, G.; et al. Effectiveness of COVID-19 Vaccines against the B.1.617.2 (Delta) Variant. N. Engl. J. Med. 2021, 385, 585–594. [Google Scholar] [CrossRef] [PubMed]
- Zou, Y.; Huang, D.; Jiang, Q.; Guo, Y.; Chen, C. The Vaccine Efficacy Against the SARS-CoV-2 Omicron: A Systemic Review and Meta-Analysis. Front. Public Health 2022, 10, 940956. [Google Scholar] [CrossRef] [PubMed]
- Obeid, M.; Suffiotti, M.; Pellaton, C.; Bouchaab, H.; Cairoli, A.; Salvadé, V.; Stevenel, C.; Hottinger, R.; Pythoud, C.; Coutechier, L.; et al. Humoral Responses Against Variants of Concern by COVID-19 mRNA Vaccines in Immunocompromised Patients. JAMA Oncol. 2022, 8, e220446. [Google Scholar] [CrossRef] [PubMed]
- Tenforde, M.W.; Patel, M.M.; Gaglani, M.; Ginde, A.A.; Douin, D.J.; Talbot, H.K.; Casey, J.D.; Mohr, N.M.; Zepeski, A.; McNeal, T.; et al. Effectiveness of a Third Dose of Pfizer-BioNTech and Moderna Vaccines in Preventing COVID-19 Hospitalization Among Immunocompetent and Immunocompromised Adults—United States, August–December 2021. MMWR. Morb. Mortal. Wkly. Rep. 2022, 71, 118–124. [Google Scholar] [CrossRef]
- Redjoul, R.; Le Bouter, A.; Parinet, V.; Fourati, S.; Maury, S. Antibody response after third BNT162b2 dose in recipients of allogeneic HSCT. Lancet Haematol. 2021, 8, e681–e683. [Google Scholar] [CrossRef]
- Herishanu, Y.; Rahav, G.; Levi, S.; Braester, A.; Itchaki, G.; Bairey, O.; Dally, N.; Shvidel, L.; Ziv-Baran, T.; Polliack, A.; et al. Efficacy of a third BNT162b2 mRNA COVID-19 vaccine dose in patients with CLL who failed standard 2-dose vaccination. Blood 2022, 139, 678–685. [Google Scholar] [CrossRef]
- Re, D.; Seitz-Polski, B.; Brglez, V.; Carles, M.; Graça, D.; Benzaken, S.; Liguori, S.; Zahreddine, K.; Delforge, M.; Bailly-Maitre, B.; et al. Humoral and cellular responses after a third dose of SARS-CoV-2 BNT162b2 vaccine in patients with lymphoid malignancies. Nat. Commun. 2022, 13, 864. [Google Scholar] [CrossRef]
- Monin, L.; Laing, A.G.; Muñoz-Ruiz, M.; McKenzie, D.R.; del Molino del Barrio, I.; Alaguthurai, T.; Domingo-Vila, C.; Hayday, T.S.; Graham, C.; Seow, J.; et al. Safety and immunogenicity of one versus two doses of the COVID-19 vaccine BNT162b2 for patients with cancer: Interim analysis of a prospective observational study. Lancet Oncol. 2021, 22, 765–778. [Google Scholar] [CrossRef]
- Westhoff, T.H.; Seibert, F.S.; Anft, M.; Blazquez-Navarro, A.; Skrzypczyk, S.; Doevelaar, A.; Hölzer, B.; Paniskaki, K.; Dolff, S.; Wilde, B.; et al. Correspondence on ‘SARS-CoV-2 vaccination in rituximab-treated patients: Evidence for impaired humoral but inducible cellular immune response’. Ann. Rheum. Dis. 2021, 80, e162. [Google Scholar] [CrossRef] [PubMed]
- Mrak, D.; Tobudic, S.; Koblischke, M.; Graninger, M.; Radner, H.; Sieghart, D.; Hofer, P.; Perkmann, T.; Haslacher, H.; Thalhammer, R.; et al. SARS-CoV-2 vaccination in rituximab-treated patients: B cells promote humoral immune responses in the presence of T-cell-mediated immunity. Ann. Rheum. Dis. 2021, 80, 1345–1350. [Google Scholar] [CrossRef] [PubMed]
- Marasco, V.; Carniti, C.; Guidetti, A.; Farina, L.; Magni, M.; Miceli, R.; Calabretta, L.; Verderio, P.; Ljevar, S.; Serpenti, F.; et al. T-cell immune response after mRNA SARS-CoV-2 vaccines is frequently detected also in the absence of seroconversion in patients with lymphoid malignancies. Br. J. Haematol. 2022, 196, 548–558. [Google Scholar] [CrossRef] [PubMed]
- Rogers, T.F.; Zhao, F.; Huang, D.; Beutler, N.; Burns, A.; He, W.; Limbo, O.; Smith, C.; Song, G.; Woehl, J.; et al. Isolation of potent SARS-CoV-2 neutralizing antibodies and protection from disease in a small animal model. Science 2020, 369, 956–963. [Google Scholar] [CrossRef] [PubMed]
| Variable | All Patients (N = 90) | Non-Responders (n = 32) | Responders (n = 58) | p Value |
|---|---|---|---|---|
| Age (years), median (IQR) | 64.4 (53.5–71.5) | 71 (66.3–75.9) | 59.7 (49–66.8) | <0.001 |
| Age > 67 years, No. (%) | 35 (38.9%) | 23 (71.9%) | 12 (20.7%) | <0.001 |
| Sex (female), No. (%) | 41 (45.6%) | 17 (53.1%) | 24 (41.4%) | 0.284 |
| Time from second dose to antibody testing (days), median (IQR) | 19 (14–27) | 19 (13–26) | 19 (14–27) | 0.923 |
| Vaccine type, No. (%) | overall p value 0.412 | |||
| BNT162b2 (Pfizer-BioNTech) | 65 (72.2%) | 21 (65.6%) | 44 (75.9%) | |
| ChAdOx1 nCoV-19 (Oxford-AstraZeneca) | 16 (17.8%) | 8 (25%) | 8 (13.8%) | |
| mRNA-1273 (Moderna) | 9 (10%) | 3 (9.4%) | 6 (10.3%) | |
| Diagnosis, No. (%) |
overall p value 0.002 | |||
| Non-Hodgkin’s lymphoma | 24 (26.7%) | 17 (53.1%) | 7 (12.1%) | <0.001 |
| Chronic lymphocytic leukemia | 19 (21.1%) | 9 (28.1%) | 10 (17.2%) | 0.226 |
| Acute leukemia | 12 (13.3%) | 1 (3.1%) | 11 (19.0%) | 0.050 |
| Chronic myeloid leukemia | 7 (7.8%) | 0 | 7 (12.1%) | 0.042 |
| Other chronic myeloproliferative neoplasms | 9 (10%) | 3 (9.4%) | 6 (10.3%) | 0.883 |
| Multiple myeloma/amyloidosis | 11 (12.2%) | 2 (6.3%) | 9 (15.5%) | 0.199 |
| Hodgkin’s lymphoma | 2 (2.2%) | 0 | 2 (3.4%) | 0.288 |
| Myelodysplastic syndrome | 3 (3.3%) | 0 | 3 (5.2%) | 0.191 |
| Non-malignant disorders ° | 3 (3.3%) | 0 | 3 (5.2%) | 0.191 |
| Time from diagnosis to second dose (months), median (IQR) * | 36.6 (17.4–82.9) | 39.0 (11.4–73.8) | 35.8 (18.5–86.9) | 0.448 |
| Total serum IgG < 7.0 g/L, No. (%) + | 19 (29.7%) | 8 (33.3%) | 11 (27.5%) | 0.832 |
| In active treatment, No. (%) | 56 (62.2%) | 28 (87.5%) | 28 (48.3%) | <0.001 |
| Prior therapy, No. (%) | overall p value 0.706 | |||
| None | 6 (6.7%) | 1 (3.1%) | 5 (8.6%) | |
| 1st line | 42 (46.7%) | 14 (43.8%) | 28 (48.3%) | |
| 2nd line | 25 (27.8%) | 10 (31.3%) | 15 (25.9%) | |
| ≥3rd line | 17 (18.9%) | 7 (21.9%) | 10 (17.2%) | |
| HSCT, No. (%) § | overall p value < 0.001 | |||
| >1 year prior | 10 (11.1%) | 1 (3.1%) | 9 (15.5%) | 0.012 |
| ≤1 year prior | 19 (21.1%) | 1 (3.1%) | 18 (31%) | 0.071 |
| Anti-CD20 mAb therapy six months prior, No. (%) | 23 (25.6%) | 22 (68.8%) | 1 (1.7%) | <0.001 |
| Corticosteroid therapy six months prior, No. (%) | 17 (18.9%) | 6 (18.8%) | 11 (19%) | 0.980 |
| Prednisone equivalent dose (mg), median (IQR) | 133.3 (80–133.3) | 120 (80–120) | 133.3 (96.7–133.3) | 0.173 |
| Prednisone equivalent dose > 120 mg, No. (%) | 10 (11.1%) | 2 (6.3%) | 8 (13.8%) | 0.485 |
| Variable | Univariable | Multivariable | ||
|---|---|---|---|---|
| RR [95% CI] | p Value | RR [95% CI] | p Value | |
| Age > 67 years | 2.44 [1.51–3.92] | < 0.001 | 2.57 [1.45–4.57] | 0.001 |
| Non-Hodgkin’s lymphoma | 3.12 [1.86–5.21] | < 0.001 | ||
| Active treatment | 1.68 [1.26–2.26] | < 0.001 | ||
| HSCT ° | 0.14 [0.04–0.55] | 0.005 | ||
| HSCT > 1 year prior | 0.12 [0.02–0.83] | 0.031 | ||
| Anti-CD20 mAb therapy six months prior | 6.41 [3.59–11.43] | 0.002 | 4.88 [2.58–9.27] | < 0.001 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sertić, Z.; Lucijanić, M.; Bašić-Kinda, S.; Serventi Seiwerth, R.; Periša, V.; Sertić, D.; Coha, B.; Pulanić, D.; Perić, Z.; Desnica, L.; et al. Non-Myelofibrosis Chronic Myeloproliferative Neoplasm Patients Show Better Seroconversion Rates after SARS-CoV-2 Vaccination Compared to Other Hematologic Diseases: A Multicentric Prospective Study of KroHem. Biomedicines 2022, 10, 2892. https://doi.org/10.3390/biomedicines10112892
Sertić Z, Lucijanić M, Bašić-Kinda S, Serventi Seiwerth R, Periša V, Sertić D, Coha B, Pulanić D, Perić Z, Desnica L, et al. Non-Myelofibrosis Chronic Myeloproliferative Neoplasm Patients Show Better Seroconversion Rates after SARS-CoV-2 Vaccination Compared to Other Hematologic Diseases: A Multicentric Prospective Study of KroHem. Biomedicines. 2022; 10(11):2892. https://doi.org/10.3390/biomedicines10112892
Chicago/Turabian StyleSertić, Zrinka, Marko Lucijanić, Sandra Bašić-Kinda, Ranka Serventi Seiwerth, Vlatka Periša, Dubravka Sertić, Božena Coha, Dražen Pulanić, Zinaida Perić, Lana Desnica, and et al. 2022. "Non-Myelofibrosis Chronic Myeloproliferative Neoplasm Patients Show Better Seroconversion Rates after SARS-CoV-2 Vaccination Compared to Other Hematologic Diseases: A Multicentric Prospective Study of KroHem" Biomedicines 10, no. 11: 2892. https://doi.org/10.3390/biomedicines10112892
APA StyleSertić, Z., Lucijanić, M., Bašić-Kinda, S., Serventi Seiwerth, R., Periša, V., Sertić, D., Coha, B., Pulanić, D., Perić, Z., Desnica, L., Mikulić, M., Vodanović, M., Radman-Livaja, I., Šegulja, D., Rogić, D., Valković, T., Aurer, I., & Duraković, N. (2022). Non-Myelofibrosis Chronic Myeloproliferative Neoplasm Patients Show Better Seroconversion Rates after SARS-CoV-2 Vaccination Compared to Other Hematologic Diseases: A Multicentric Prospective Study of KroHem. Biomedicines, 10(11), 2892. https://doi.org/10.3390/biomedicines10112892





