Rapid Drop in Coronary Heart Disease Mortality in Czech Male Population—What Was Actually behind It?
Abstract
1. Introduction
2. Materials and Methods
2.1. Epidemiological Data
2.2. Biochemistry
2.3. Analysis of Adipose Tissue
2.4. Analysis of Fatty Acid Composition
2.5. Statistics
3. Results
3.1. Epidemiological, Economical, and Experimental Data
3.1.1. Treatment of Atherosclerosis Risk Factors and Acute Myocardial Infarction
3.1.2. Economic Change and Atherosclerosis Risk Factors
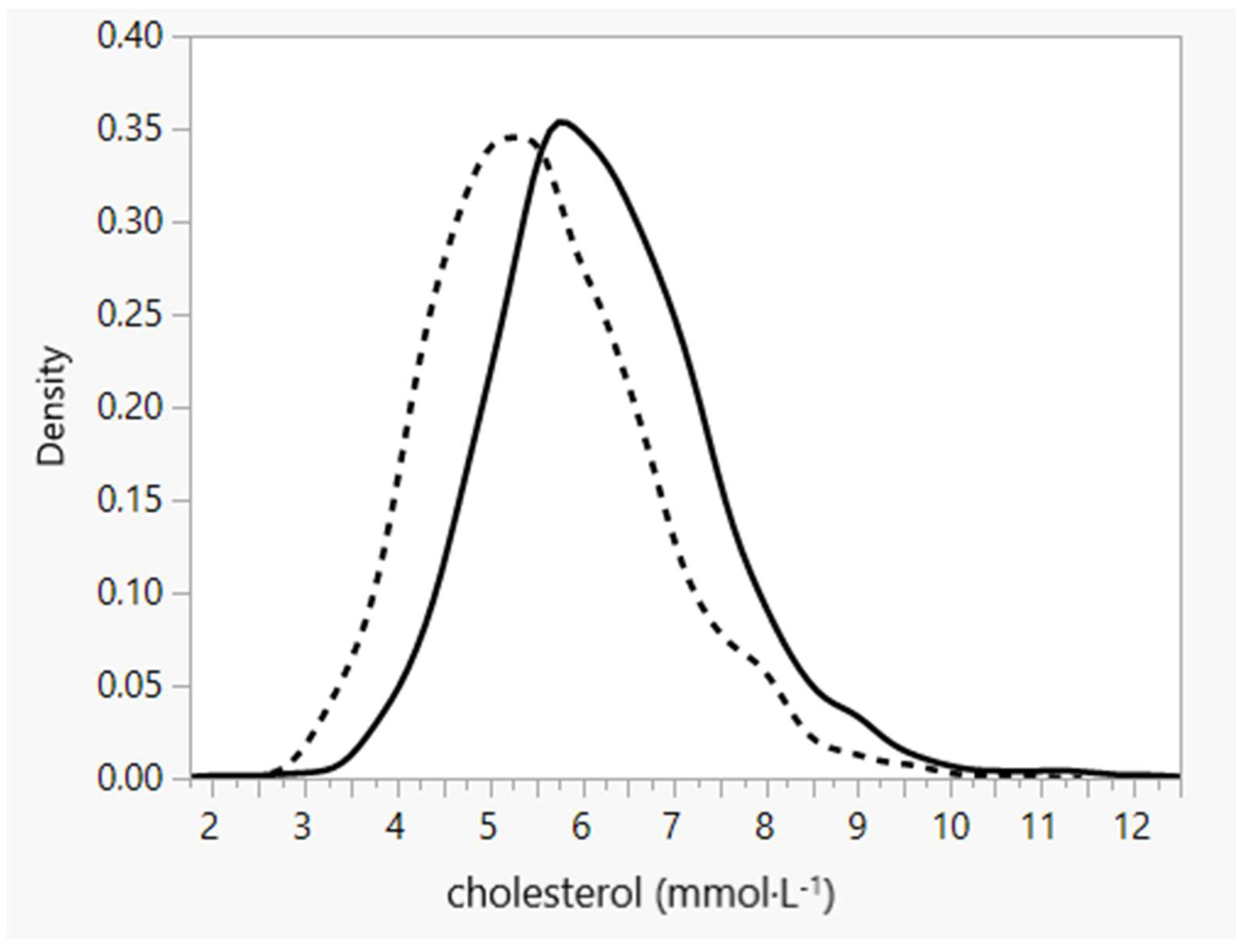
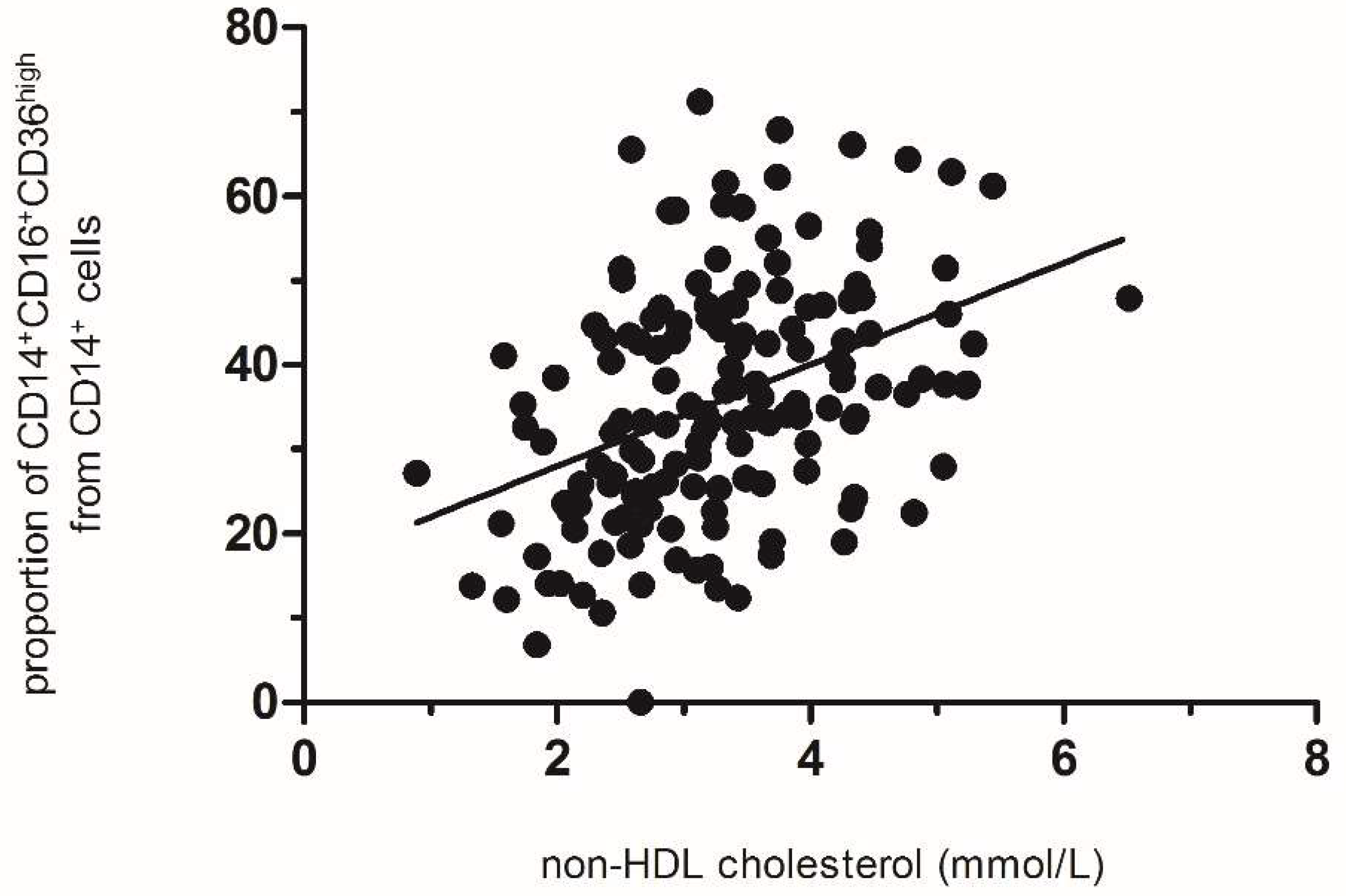
3.1.3. ProInflammatory Macrophages in Visceral Adipose Tissue and Cholesterolemia
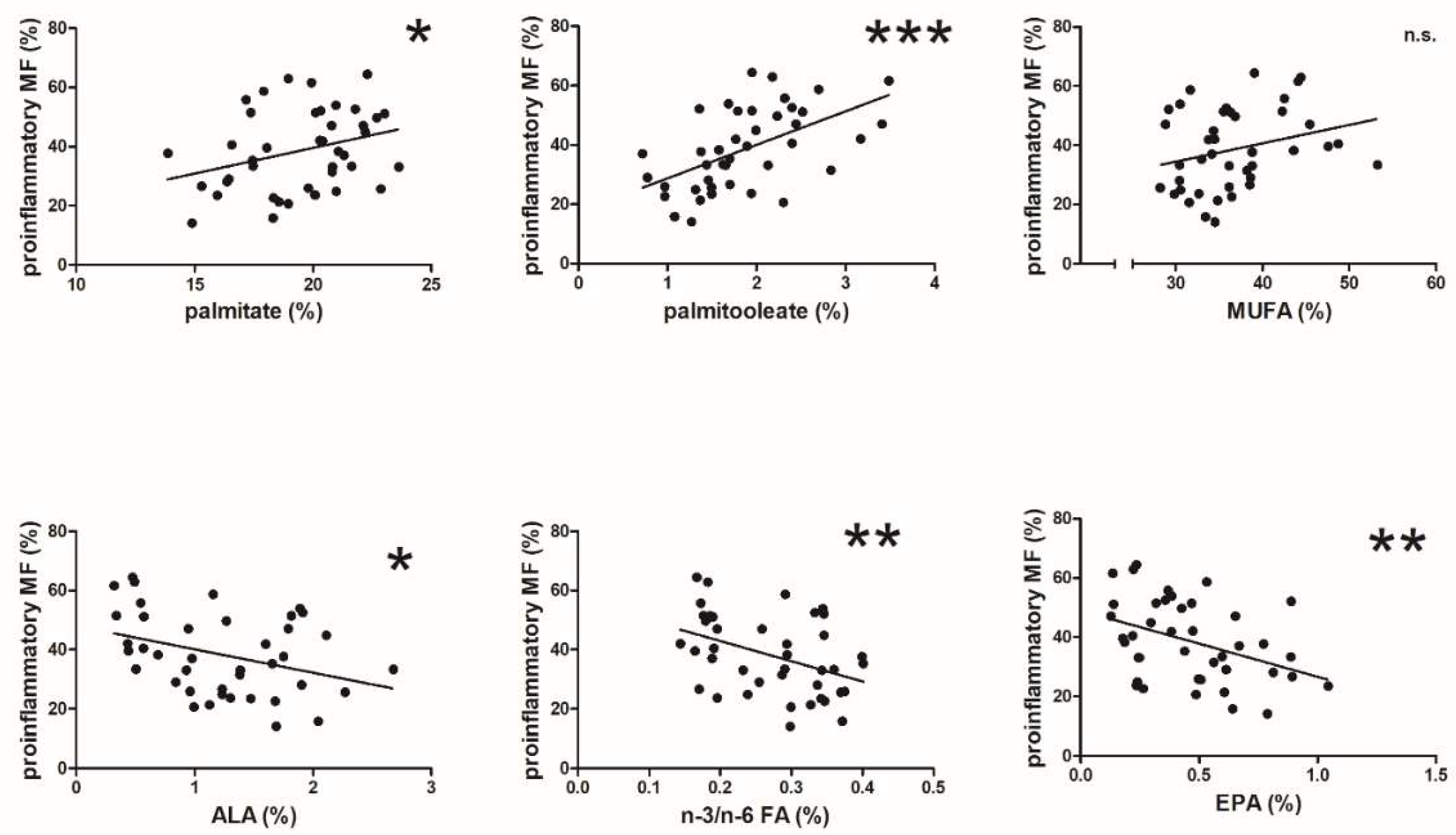
3.1.4. The Proportion of Proinflammatory Adipose Tissue Macrophages and Fatty Acid Composition
4. Discussion
4.1. CHD Mortality Changes and Cholesterol
4.2. CHD and Inflammation
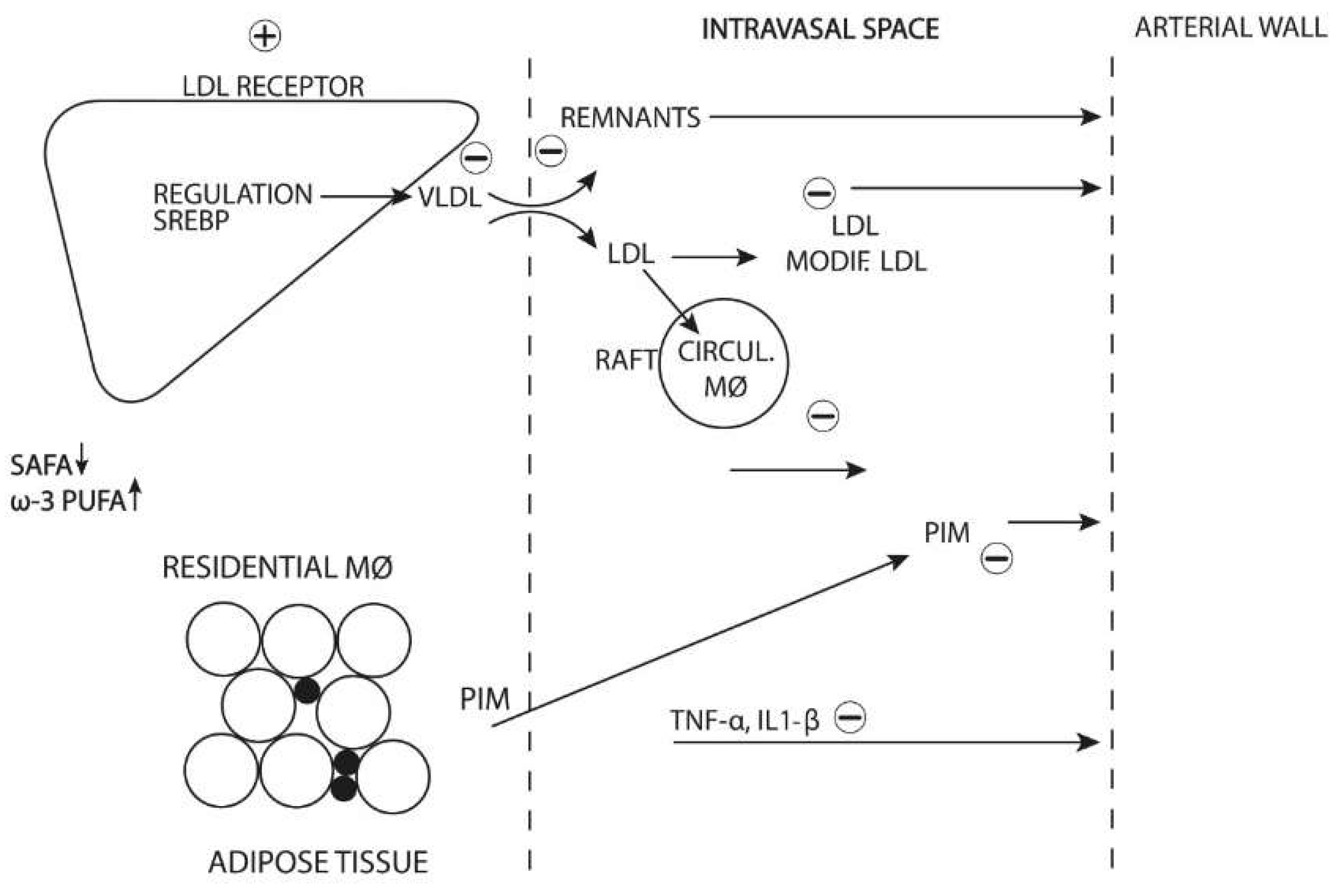
4.3. Combined Effect
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
References
- Rosamond, W.; Flegal, K.; Friday, G.; Furie, K.; Go, A.; Greenlund, K.; Haase, N.; Ho, M.; Howard, V.; Kissela, B.; et al. Heart disease and stroke statistics—2007 update: A report from the American Heart Association Statistics Committee and Stroke Statistics Subcommittee. Circulation 2007, 115, e69–e171. [Google Scholar] [CrossRef] [PubMed]
- Kannel, W.B.; Thom, T.J. Declining cardiovascular mortality. Circulation 1984, 70, 331–336. [Google Scholar] [CrossRef] [PubMed]
- Capewell, S.; Morrison, C.E.; McMurray, J.J. Contribution of modern cardiovascular treatment and risk factor changes to the decline in coronary heart disease mortality in Scotland between 1975 and 1994. Heart 1999, 81, 380–386. [Google Scholar] [CrossRef] [PubMed]
- Ford, E.S.; Ajani, U.A.; Croft, J.B.; Critchley, J.A.; Labarthe, D.R.; Kottke, T.E.; Giles, W.H.; Capewell, S. Explaining the decrease in U.S. deaths from coronary disease, 1980–2000. N. Engl. J. Med. 2007, 356, 2388–2398. [Google Scholar] [CrossRef] [PubMed]
- Björck, L.; Rosengren, A.; Bennett, K.; Lappas, G.; Capewell, S. Modelling the decreasing coronary heart disease mortality in Sweden between 1986 and 2002. Eur. Heart J. 2009, 30, 1046–1056. [Google Scholar] [CrossRef]
- Moore, K.J.; Tabas, I. Macrophages in the pathogenesis of atherosclerosis. Cell 2011, 145, 341–355. [Google Scholar] [CrossRef]
- Hansson, G.K. Atherosclerosis--an immune disease: The Anitschkov Lecture 2007. Atherosclerosis 2009, 202, 2–10. [Google Scholar] [CrossRef]
- von Ehr, A.; Bode, C.; Hilgendorf, I. Macrophages in Atheromatous Plaque Developmental Stages. Front. Cardiovasc. Med. 2022, 9, 865367. [Google Scholar] [CrossRef]
- Ridker, P.M.; Buring, J.E.; Cook, N.R.; Rifai, N. C-reactive protein, the metabolic syndrome, and risk of incident cardiovascular events: An 8-year follow-up of 14 719 initially healthy American women. Circulation 2003, 107, 391–397. [Google Scholar] [CrossRef]
- Koenig, W.; Sund, M.; Fröhlich, M.; Fischer, H.G.; Löwel, H.; Döring, A.; Hutchinson, W.L.; Pepys, M.B. C-Reactive protein, a sensitive marker of inflammation, predicts future risk of coronary heart disease in initially healthy middle-aged men: Results from the MONICA (Monitoring Trends and Determinants in Cardiovascular Disease) Augsburg Cohort Study, 1984 to 1992. Circulation 1999, 99, 237–242. [Google Scholar] [CrossRef]
- Weisberg, S.P.; McCann, D.; Desai, M.; Rosenbaum, M.; Leibel, R.L.; Ferrante, A.W. Obesity is associated with macrophage accumulation in adipose tissue. J. Clin. Investig. 2003, 112, 1796–1808. [Google Scholar] [CrossRef] [PubMed]
- Christen, T.; Trompet, S.; Rensen, P.C.N.; Willems van Dijk, K.; Lamb, H.J.; Jukema, J.W.; Rosendaal, F.R.; le Cessie, S.; de Mutsert, R. The role of inflammation in the association between overall and visceral adiposity and subclinical atherosclerosis. Nutr. Metab. Cardiovasc. Dis. 2019, 29, 728–735. [Google Scholar] [CrossRef] [PubMed]
- Poledne, R.; Kralova Lesna, I.; Kralova, A.; Fronek, J.; Cejkova, S. The relationship between non-HDL cholesterol and macrophage phenotypes in human adipose tissue. J. Lipid Res. 2016, 57, 1899–1905. [Google Scholar] [CrossRef] [PubMed]
- Poledne, R.; Malinska, H.; Kubatova, H.; Fronek, J.; Thieme, F.; Kauerova, S.; Lesna, I.K. Polarization of Macrophages in Human Adipose Tissue is Related to the Fatty Acid Spectrum in Membrane Phospholipids. Nutrients 2019, 12, 8. [Google Scholar] [CrossRef]
- Majesky, M.W. Developmental basis of vascular smooth muscle diversity. Arterioscler. Thromb. Vasc. Biol. 2007, 27, 1248–1258. [Google Scholar] [CrossRef]
- Libby, P.; Ridker, P.M.; Hansson, G.K. Progress and challenges in translating the biology of atherosclerosis. Nature 2011, 473, 317–325. [Google Scholar] [CrossRef]
- Geovanini, G.R.; Libby, P. Atherosclerosis and inflammation: Overview and updates. Clin. Sci. 2018, 132, 1243–1252. [Google Scholar] [CrossRef]
- Ridker, P.M.; Everett, B.M.; Thuren, T.; MacFadyen, J.G.; Chang, W.H.; Ballantyne, C.; Fonseca, F.; Nicolau, J.; Koenig, W.; Anker, S.D.; et al. Antiinflammatory Therapy with Canakinumab for Atherosclerotic Disease. N. Engl. J. Med. 2017, 377, 1119–1131. [Google Scholar] [CrossRef]
- Poledne, R.; Skodová, Z. Changes in nutrition, cholesterol concentration, and cardiovascular disease mortality in the Czech population in the past decade. Nutrition 2000, 16, 785–786. [Google Scholar] [CrossRef]
- Kralova Lesna, I.; Poledne, R.; Fronek, J.; Kralova, A.; Sekerkova, A.; Thieme, F.; Pitha, J. Macrophage subsets in the adipose tissue could be modified by sex and the reproductive age of women. Atherosclerosis 2015, 241, 255–258. [Google Scholar] [CrossRef]
- Kralova Lesna, I.; Kralova, A.; Cejkova, S.; Fronek, J.; Petras, M.; Sekerkova, A.; Thieme, F.; Janousek, L.; Poledne, R. Characterisation and comparison of adipose tissue macrophages from human subcutaneous, visceral and perivascular adipose tissue. J. Transl. Med. 2016, 14, 208. [Google Scholar] [CrossRef] [PubMed]
- Malinska, H.; Hüttl, M.; Oliyarnyk, O.; Bratova, M.; Kazdova, L. Conjugated linoleic acid reduces visceral and ectopic lipid accumulation and insulin resistance in chronic severe hypertriacylglycerolemia. Nutrition 2015, 31, 1045–1051. [Google Scholar] [CrossRef] [PubMed]
- Eder, K. Gas chromatographic analysis of fatty acid methyl esters. J. Chromatogr. B Biomed. Appl. 1995, 671, 113–131. [Google Scholar] [CrossRef]
- Zatoński, W.A. Epidemiological analysis of health situation development in Europe and its causes until 1990. Ann. Agric. Environ. Med. 2011, 18, 194–202. [Google Scholar] [PubMed]
- Vojacek, J. Development of the Czech intervention cardiology and its comparison with Europe. Interv. Akut. Kardiol. 2003, 2, 152–155. (In Czech) [Google Scholar]
- Paukner, K.; Králová Lesná, I.; Poledne, R. Cholesterol in the Cell Membrane-An Emerging Player in Atherogenesis. Int. J. Mol. Sci. 2022, 23, 533. [Google Scholar] [CrossRef]
- Barrett, T.J. Macrophages in Atherosclerosis Regression. Arterioscler. Thromb. Vasc. Biol. 2020, 40, 20–33. [Google Scholar] [CrossRef]
- Kralova Lesna, I.; Suchanek, P.; Brabcova, E.; Kovar, J.; Malinska, H.; Poledne, R. Effect of different types of dietary fatty acids on subclinical inflammation in humans. Physiol. Res./Acad. Sci. Bohemoslov. 2013, 62, 145–152. [Google Scholar] [CrossRef]
- Kawai, T.; Autieri, M.V.; Scalia, R. Adipose tissue inflammation and metabolic dysfunction in obesity. Am. J. Physiol. Cell Physiol. 2021, 320, C375–C391. [Google Scholar] [CrossRef]
- Engin, A.B. Adipocyte-Macrophage Cross-Talk in Obesity. Adv. Exp. Med. Biol. 2017, 960, 327–343. [Google Scholar] [CrossRef]
- Kralova Lesna, I.; Petras, M.; Cejkova, S.; Kralova, A.; Fronek, J.; Janousek, L.; Thieme, F.; Tyll, T.; Poledne, R. Cardiovascular disease predictors and adipose tissue macrophage polarization: Is there a link? Eur. J. Prev. Cardiol. 2018, 25, 328–334. [Google Scholar] [CrossRef] [PubMed]
- Ridker, P.M.; Cushman, M.; Stampfer, M.J.; Tracy, R.P.; Hennekens, C.H. Inflammation, aspirin, and the risk of cardiovascular disease in apparently healthy men. N. Engl. J. Med. 1997, 336, 973–979. [Google Scholar] [CrossRef] [PubMed]
- Tall, A.R.; Yvan-Charvet, L. Cholesterol, inflammation and innate immunity. Nat. Rev. Immunol. 2015, 15, 104–116. [Google Scholar] [CrossRef] [PubMed]
- Langsted, A.; Nordestgaard, B.G. Nonfasting lipids, lipoproteins, and apolipoproteins in individuals with and without diabetes: 58 434 individuals from the Copenhagen General Population Study. Clin. Chem. 2011, 57, 482–489. [Google Scholar] [CrossRef] [PubMed]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Poledne, R.; Kralova, A.; Bartuskova, H.; Paukner, K.; Kauerova, S.; Fronek, J.; Lanska, V.; Kralova Lesna, I. Rapid Drop in Coronary Heart Disease Mortality in Czech Male Population—What Was Actually behind It? Biomedicines 2022, 10, 2871. https://doi.org/10.3390/biomedicines10112871
Poledne R, Kralova A, Bartuskova H, Paukner K, Kauerova S, Fronek J, Lanska V, Kralova Lesna I. Rapid Drop in Coronary Heart Disease Mortality in Czech Male Population—What Was Actually behind It? Biomedicines. 2022; 10(11):2871. https://doi.org/10.3390/biomedicines10112871
Chicago/Turabian StylePoledne, Rudolf, Anna Kralova, Hana Bartuskova, Karel Paukner, Sona Kauerova, Jiri Fronek, Vera Lanska, and Ivana Kralova Lesna. 2022. "Rapid Drop in Coronary Heart Disease Mortality in Czech Male Population—What Was Actually behind It?" Biomedicines 10, no. 11: 2871. https://doi.org/10.3390/biomedicines10112871
APA StylePoledne, R., Kralova, A., Bartuskova, H., Paukner, K., Kauerova, S., Fronek, J., Lanska, V., & Kralova Lesna, I. (2022). Rapid Drop in Coronary Heart Disease Mortality in Czech Male Population—What Was Actually behind It? Biomedicines, 10(11), 2871. https://doi.org/10.3390/biomedicines10112871




