New Insights into Adiponectin and Leptin Roles in Chronic Kidney Disease
Abstract
:1. Introduction
2. Adiponectin
2.1. Inflammation and Adiponectin
2.2. CVD Risk and Adiponectin
2.2.1. BMI
2.2.2. Vascular Calcification
2.2.3. Dyslipidemia
2.2.4. LVH
2.3. Mineral and Bone Disorder
2.4. Anemia
2.5. Adiponectin Isoforms
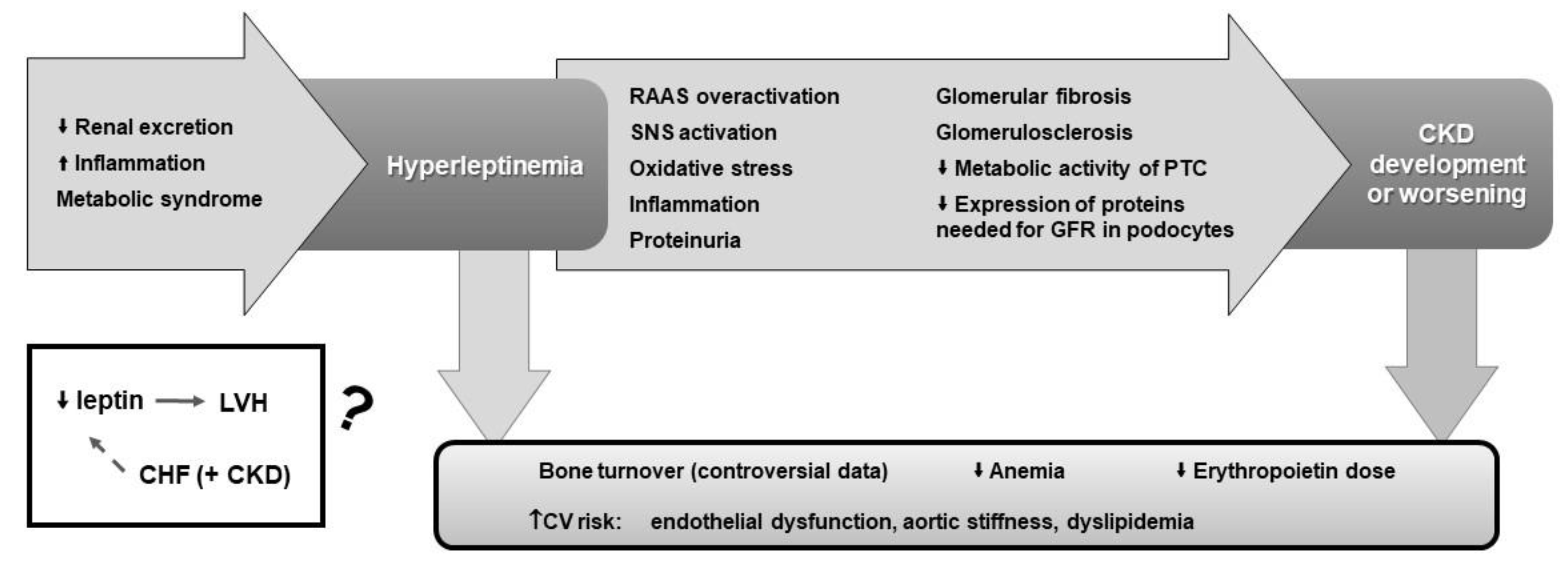
3. Leptin
3.1. Inflammation and Leptin
3.2. CVD Risk and Leptin
3.2.1. BMI
3.2.2. Vascular Complications
3.2.3. Dyslipidemia
3.2.4. LVH
3.3. Mineral and Bone Disorder
3.4. Anemia
4. Concluding Remarks
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Schlondorff, D.O. Overview of factors contributing to the pathophysiology of progressive renal disease. Kidney Int. 2008, 74, 860–866. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yamaguchi, J.; Tanaka, T.; Nangaku, M. Recent advances in understanding of chronic kidney disease. F1000Research 2015, 4, 1212. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Andrade-Oliveira, V.; Foresto-Neto, O.; Watanabe, I.K.M.; Zatz, R.; Câmara, N.O.S. Inflammation in Renal Diseases: New and Old Players. Front. Pharmacol. 2019, 10, 1192. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Raj, D.S.; Pecoits-Filho, R.; Kimmel, P.L. Inflammation in Chronic Kidney Disease. In Chronic Renal Disease; Kimmel, P.L., Rosenberg, M.E., Eds.; Academic Press: San Diego, CA, USA, 2015; pp. 199–212. [Google Scholar]
- Nehus, E. Obesity and chronic kidney disease. Curr. Opin. Pediatr. 2018, 30, 241–246. [Google Scholar] [CrossRef] [PubMed]
- Bikbov, B.; Purcell, C.A.; Levey, A.S.; Smith, M.; Abdoli, A.; Abebe, M.; Adebayo, O.M.; Afarideh, M.; Agarwal, S.K.; Agudelo-Botero, M.; et al. Global, regional, and national burden of chronic kidney disease, 1990–2017: A systematic analysis for the Global Burden of Disease Study 2017. Lancet 2020, 395, 709–733. [Google Scholar] [CrossRef] [Green Version]
- Lim, C.C.; Teo, B.W.; Tai, E.S.; Lim, S.C.; Chan, C.M.; Sethi, S.; Wong, T.Y.; Sabanayagam, C. Elevated Serum Leptin, Adiponectin and Leptin to Adiponectin Ratio Is Associated with Chronic Kidney Disease in Asian Adults. PLoS ONE 2015, 10, e0122009. [Google Scholar] [CrossRef] [Green Version]
- Liu, M.; Li, X.C.; Lu, L.; Cao, Y.; Sun, R.R.; Chen, S.; Zhang, P.Y. Cardiovascular disease and its relationship with chronic kidney disease. Eur. Rev. Med. Pharmacol. Sci. 2014, 18, 2918–2926. [Google Scholar]
- Coimbra, S.; Reis, F.; Nunes, S.R.R.P.; Viana, S.; Valente, M.J.; Rocha, S.; Catarino, C.; Rocha-Pereira, P.; Bronze-Da-Rocha, E.; Sameiro-Faria, M.; et al. The Protective Role of Adiponectin for Lipoproteins in End-Stage Renal Disease Patients: Relationship with Diabetes and Body Mass Index. Oxidative Med. Cell. Longev. 2019, 2019, 3021785. [Google Scholar] [CrossRef] [Green Version]
- Parida, S.; Siddharth, S.; Sharma, D. Adiponectin, Obesity, and Cancer: Clash of the Bigwigs in Health and Disease. Int. J. Mol. Sci. 2019, 20, 2519. [Google Scholar] [CrossRef] [Green Version]
- Magkos, F.; Sidossis, L.S. Recent advances in the measurement of adiponectin isoform distribution. Curr. Opin. Clin. Nutr. Metab. Care 2007, 10, 571–575. [Google Scholar] [CrossRef]
- Wang, Z.V.; Scherer, P.E. Adiponectin, the past two decades. J. Mol. Cell Biol. 2016, 8, 93–100. [Google Scholar] [CrossRef] [PubMed]
- Yamauchi, T.; Kamon, J.; Minokoshi, Y.; Ito, Y.; Waki, H.; Uchida, S.; Yamashita, S.; Noda, M.; Kita, S.; Ueki, K.; et al. Adiponectin stimulates glucose utilization and fatty-acid oxidation by activating AMP-activated protein kinase. Nat. Med. 2002, 8, 1288–1295. [Google Scholar] [CrossRef] [PubMed]
- Mao, X.; Kikani, C.K.; Riojas, R.A.; Langlais, P.; Wang, L.; Ramos, F.J.; Fang, Q.; Christ-Roberts, C.Y.; Hong, J.Y.; Kim, R.Y.; et al. APPL1 binds to adiponectin receptors and mediates adiponectin signalling and function. Nat. Cell Biol. 2006, 8, 516–523. [Google Scholar] [CrossRef] [PubMed]
- Choi, H.M.; Doss, H.M.; Kim, K.S. Multifaceted Physiological Roles of Adiponectin in Inflammation and Diseases. Int. J. Mol. Sci. 2020, 21, 1219. [Google Scholar] [CrossRef] [Green Version]
- Liu, Z.; Gan, L.; Wu, T.; Feng, F.; Luo, D.; Gu, H.; Liu, S.; Sun, C. Adiponectin reduces ER stress-induced apoptosis through PPARα transcriptional regulation of ATF2 in mouse adipose. Cell Death Dis. 2016, 7, e2487. [Google Scholar] [CrossRef] [Green Version]
- Achari, A.E.; Jain, S.K. Adiponectin, a Therapeutic Target for Obesity, Diabetes, and Endothelial Dysfunction. Int. J. Mol. Sci. 2017, 18, 1321. [Google Scholar] [CrossRef] [Green Version]
- Mihai, S.; Codrici, E.; Popescu, I.D.; Enciu, A.-M.; Albulescu, L.; Necula, L.G.; Mambet, C.; Anton, G.; Tanase, C. Inflammation-Related Mechanisms in Chronic Kidney Disease Prediction, Progression, and Outcome. J. Immunol. Res. 2018, 2018, 2180373. [Google Scholar] [CrossRef] [Green Version]
- Kamimura, M.A.; Canziani, M.E.; Sanches, F.R.; Velludo, C.M.; Carrero, J.J.; Bazanelli, A.P.; Draibe, S.A.; Cuppari, L. Variations in adiponectin levels in patients with chronic kidney disease: A prospective study of 12 months. J. Bras. Nefrol. 2012, 34, 259–265. [Google Scholar] [CrossRef] [Green Version]
- Song, S.H.; Oh, T.R.; Choi, H.S.; Kim, C.S.; Ma, S.K.; Oh, K.H.; Ahn, C.; Kim, S.W.; Bae, E.H. High serum adiponectin as a biomarker of renal dysfunction: Results from the KNOW-CKD study. Sci. Rep. 2020, 10, 5598. [Google Scholar] [CrossRef] [Green Version]
- Axelsson, J. Obesity in Chronic Kidney Disease: Good or Bad? Blood Purif. 2008, 26, 23–29. [Google Scholar] [CrossRef]
- Martinez Cantarin, M.P.; Keith, S.W.; Waldman, S.A.; Falkner, B. Adiponectin receptor and adiponectin signaling in human tissue among patients with end-stage renal disease. Nephrol. Dial. Transplant. 2014, 29, 2268–2277. [Google Scholar] [CrossRef] [PubMed]
- Markaki, A.; Psylinakis, E.; Spyridaki, A. Adiponectin and end-stage renal disease. Hormones 2016, 15, 345–354. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Menon, V.; Li, L.; Wang, X.; Greene, T.; Balakrishnan, V.; Madero, M.; Pereira, A.A.; Beck, G.J.; Kusek, J.W.; Collins, A.J.; et al. Adiponectin and Mortality in Patients with Chronic Kidney Disease. J. Am. Soc. Nephrol. 2006, 17, 2599–2606. [Google Scholar] [CrossRef] [Green Version]
- Rhee, C.M.; Nguyen, D.V.; Moradi, H.; Brunelli, S.M.; Dukkipati, R.; Jing, J.; Nakata, T.; Kovesdy, C.P.; Brent, G.A.; Kalantar-Zadeh, K. Association of Adiponectin With Body Composition and Mortality in Hemodialysis Patients. Am. J. Kidney Dis. 2015, 66, 313–321. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kim, J.H.; Han, J.M.; Kim, H.; Lee, K.-B.; Chung, W.; Kim, Y.-S.; Park, S.K.; Chae, D.W.; Ahn, C.; Oh, K.H.; et al. Low serum adiponectin level is associated with better physical health-related quality of life in chronic kidney disease. Sci. Rep. 2021, 11, 10928. [Google Scholar] [CrossRef]
- Choi, S.H.; Ku, E.J.; Hong, E.S.; Lim, S.; Kim, K.W.; Moon, J.H.; Kim, K.M.; Park, Y.J.; Park, K.S.; Jang, H.C. High serum adiponectin concentration and low body mass index are significantly associated with increased all-cause and cardiovascular mortality in an elderly cohort, “adiponectin paradox”: The Korean Longitudinal Study on Health and Aging (KLoSHA). Int. J. Cardiol. 2015, 183, 91–97. [Google Scholar] [CrossRef]
- Hyun, Y.Y.; Lee, K.B.; Oh, K.H.; Ahn, C.; Park, S.K.; Chae, D.W.; Yoo, T.H.; Cho, K.H.; Kim, Y.S.; Hwang, Y.H. Serum adiponectin and protein–energy wasting in predialysis chronic kidney disease. Nutrition 2016, 33, 254–260. [Google Scholar] [CrossRef]
- Markaki, A.; Grammatikopoulou, M.G.; Venihaki, M.; Kyriazis, J.; Perakis, K.; Stylianou, K. Associations of adiponectin and leptin levels with protein-energy wasting, in end stage renal disease patients. Endocrinol. Nutr. 2016, 63, 449–457. [Google Scholar] [CrossRef]
- Kaynar, K.; Kural, B.V.; Ulusoy, S.; Cansiz, M.; Akcan, B.; Misir, N.; Yaman, S.; Kaya, N. Is there any interaction of resistin and adiponectin levels with protein-energy wasting among patients with chronic kidney disease. Hemodial. Int. 2014, 18, 153–162. [Google Scholar] [CrossRef]
- Alam, A.; Molnar, M.Z.; Czira, M.E.; Rudas, A.; Ujszaszi, A.; Kalantar-Zadeh, K.; Rosivall, L.; Mucsi, I. Serum Adiponectin Levels and Mortality after Kidney Transplantation. Clin. J. Am. Soc. Nephrol. 2013, 8, 460–467. [Google Scholar] [CrossRef] [Green Version]
- Rao, M.; Li, L.; Tighiouart, H.; Jaber, B.L.; Pereira, B.J.G.; Balakrishnan, V.S.; the HEMO Study Group. Plasma adiponectin levels and clinical outcomes among haemodialysis patients. Nephrol. Dial. Transplant. 2008, 23, 2619–2628. [Google Scholar] [CrossRef] [PubMed]
- Abdallah, E.; Waked, E.; Nabil, M.; El-Bendary, O. Adiponectin and Cardiovascular Outcomes among Hemodialysis Patients. Kidney Blood Press. Res. 2012, 35, 247–253. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Wei, L.-L.; Zhang, R.-P.; Han, C.-W.; Cao, Y. Globular adiponectin inhibits osteoblastic differentiation of vascular smooth muscle cells through the PI3K/AKT and Wnt/β-catenin pathway. J. Mol. Histol. 2021, 52, 1067–1080. [Google Scholar] [CrossRef]
- Lu, Y.; Bian, Y.; Wang, Y.; Bai, R.; Wang, J.; Xiao, C. Globular adiponectin reduces vascular calcification via inhibition of ER-stress-mediated smooth muscle cell apoptosis. Int. J. Clin. Exp. Pathol. 2015, 8, 2545–2554. [Google Scholar] [PubMed]
- Lu, Y.; Ma, Y.; Wang, R.; Sun, J.; Guo, B.; Wei, R.; Jia, Y. Adiponectin inhibits vascular smooth muscle cell calcification induced by beta-glycerophosphate through JAK2/STAT3 signaling pathway. J. Biosci. 2019, 44, 86. [Google Scholar] [CrossRef] [PubMed]
- Aoqui, C.; Cuppari, L.; Kamimura, M.A.; Canziani, M.E. Increased visceral adiposity is associated with coronary artery calcification in male patients with chronic kidney disease. Eur. J. Clin. Nutr. 2013, 67, 610–614. [Google Scholar] [CrossRef]
- Alderson, H.V.; Ritchie, J.P.; Green, D.; Chiu, D.; Kalra, P.A. Potential for Biomarkers of Chronic Kidney Disease-Mineral Bone Disorder to Improve Patient Care. Nephron Clin. Pract. 2013, 124, 141–150. [Google Scholar] [CrossRef]
- Golembiewska, E.; Qureshi, A.R.; Dai, L.; Lindholm, B.; Heimbürger, O.; Söderberg, M.; Brismar, T.B.; Ripsweden, J.; Barany, P.; Johnson, R.J.; et al. Copeptin is independently associated with vascular calcification in chronic kidney disease stage 5. BMC Nephrol. 2020, 21, 43. [Google Scholar] [CrossRef]
- Markaki, A.; Kyriazis, J.; Stylianou, K.; Fragkiadakis, G.A.; Perakis, K.; Margioris, A.N.; Ganotakis, E.S.; Daphnis, E. The Role of Serum Magnesium and Calcium on the Association between Adiponectin Levels and All-Cause Mortality in End-Stage Renal Disease Patients. PLoS ONE 2012, 7, e52350. [Google Scholar] [CrossRef]
- Kim, C.S.; Bae, E.H.; Ma, S.K.; Park, S.K.; Lee, J.Y.; Chung, W.; Lee, K.; Kim, Y.H.; Oh, K.-H.; Ahn, C.; et al. Association of serum adiponectin concentration with aortic arterial stiffness in chronic kidney disease: From the KNOW-CKD study. Clin. Exp. Nephrol. 2016, 21, 608–616. [Google Scholar] [CrossRef]
- Sakura, T.; Okuno, S.; Nishio, E.; Norimine, K.; Ishimura, E.; Yamakawa, T.; Shoji, S.; Inaba, M. The association of serum adiponectin with abdominal aortic calcification in Japanese male hemodialysis patients: A cross-sectional observational study. Sci. Rep. 2017, 7, 6434. [Google Scholar] [CrossRef] [PubMed]
- Hayashi, M.; Shibata, R.; Takahashi, H.; Ishii, H.; Aoyama, T.; Kasuga, H.; Yamada, S.; Ohashi, K.; Maruyama, S.; Matsuo, S.; et al. Association of Adiponectin with Carotid Arteriosclerosis in Predialysis Chronic Kidney Disease. Am. J. Nephrol. 2011, 34, 249–255. [Google Scholar] [CrossRef] [PubMed]
- Lin, S.-C.; Wu, T.-J.; Wu, D.-A.; Hsu, B.-G. Hypoadiponectinemia is associated with aortic stiffness in nondialysis diabetic patients with stage 3–5 chronic kidney disease. Vascular 2022, 30, 384–391. [Google Scholar] [CrossRef] [PubMed]
- Kaysen, G.A. Lipid and Lipoprotein Metabolism in Chronic Kidney Disease. J. Ren. Nutr. 2009, 19, 73–77. [Google Scholar] [CrossRef]
- Ribeiro, S.; Faria Mdo, S.; Silva, G.; Nascimento, H.; Rocha-Pereira, P.; Miranda, V.; Vieira, E.; Santos, R.; Mendonça, D.; Quintanilha, A.; et al. Oxidized low-density lipoprotein and lipoprotein(a) levels in chronic kidney disease patients under hemodialysis: Influence of adiponectin and of a polymorphism in the apolipoprotein(a) gene. Hemodial. Int. 2012, 16, 481–490. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.Y.; Bae, E.H.; Ma, S.K.; Chae, D.W.; Choi, K.H.; Kim, Y.-S.; Hwang, Y.-H.; Ahn, C.; Kim, S.W. Association of serum adiponectin level with albuminuria in chronic kidney disease patients. Clin. Exp. Nephrol. 2015, 20, 443–449. [Google Scholar] [CrossRef]
- Abe, Y.; Eto, S.; Matsumae, T.; Ogahara, S.; Murata, T.; Watanabe, M.; Nakashima, H.; Saito, T. The proportion and metabolic effects of adiponectin multimeric isoforms in patients with chronic kidney disease on maintenance hemodialysis. Ren. Fail. 2010, 32, 849–854. [Google Scholar] [CrossRef] [Green Version]
- Poniku, A.; Bajraktari, G.; Elezi, S.; Ibrahimi, P.; Henein, M.Y. Adiponectin correlates with body mass index and to a lesser extent with left ventricular mass in dialysis patients. Cardiol. J. 2018, 25, 501–511. [Google Scholar] [CrossRef] [Green Version]
- Zoccali, C.; Benedetto, F.A.; Mallamaci, F.; Tripepi, G.; Giacone, G.; Cataliotti, A.; Seminara, G.; Stancanelli, B.; Malatino, L.S. Prognostic Value of Echocardiographic Indicators of Left Ventricular Systolic Function in Asymptomatic Dialysis Patients. J. Am. Soc. Nephrol. 2004, 15, 1029–1037. [Google Scholar] [CrossRef] [Green Version]
- Komaba, H.; Igaki, N.; Goto, S.; Yokota, K.; Takemoto, T.; Hirosue, Y.; Goto, T. Adiponectin Is Associated with Brain Natriuretic Peptide and Left Ventricular Hypertrophy in Hemodialysis Patients with Type 2 Diabetes Mellitus. Nephron Clin. Pract. 2007, 107, c103–c108. [Google Scholar] [CrossRef]
- Ayerden Ebinç, F.; Ebinç, H.; Derici, U.; Aral, A.; Aybay, C.; Taçoy, G.; Koç, E.; Mutluay, R.; Reis, K.A.; Erten, Y.; et al. The relationship between adiponectin levels and proinflammatory cytokines and left ventricular mass in dialysis patients. J. Nephrol. 2009, 22, 216–223. [Google Scholar] [PubMed]
- Lee, Y.; Kim, B.K.; Lim, Y.-H.; Kim, M.K.; Choi, B.Y.; Shin, J. The Relationship between Adiponectin and Left Ventricular Mass Index Varies with the Risk of Left Ventricular Hypertrophy. PLoS ONE 2013, 8, e70246. [Google Scholar] [CrossRef] [PubMed]
- Lo, M.M.; Salisbury, S.; Scherer, P.E.; Furth, S.L.; Warady, B.A.; Mitsnefes, M.M. Serum adiponectin complexes and cardiovascular risk in children with chronic kidney disease. Pediatr. Nephrol. 2011, 26, 2009–2017. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Okuno, S.; Ishimura, E.; Norimine, K.; Tsuboniwa, N.; Kagitani, S.; Yamakawa, K.; Sato, K.K.; Hayashi, T.; Shoji, S.; Nishizawa, Y.; et al. Serum adiponectin and bone mineral density in male hemodialysis patients. Osteoporos. Int. 2011, 23, 2027–2035. [Google Scholar] [CrossRef]
- Kuźniewski, M.; Fedak, D.; Dumnicka, P.; Kapusta, M.; Stępień, E.; Chowaniec, E.; Krzanowska, K.; Krzanowski, M.; Chmiel, G.; Solnica, B.; et al. Carboxylated and intact osteocalcin predict adiponectin concentration in hemodialyzed patients. Ren. Fail. 2016, 38, 451–457. [Google Scholar] [CrossRef] [Green Version]
- Rutkowski, J.M.; Pastor, J.; Sun, K.; Park, S.K.; Bobulescu, I.A.; Chen, C.T.; Moe, O.W.; Scherer, P.E. Adiponectin alters renal calcium and phosphate excretion through regulation of klotho expression. Kidney Int. 2017, 91, 324–337. [Google Scholar] [CrossRef] [Green Version]
- Hyun, Y.Y.; Kim, H.; Oh, Y.K.; Oh, K.-H.; Ahn, C.; Sung, S.A.; Choi, K.H.; Kim, S.W.; Lee, K.-B. High fibroblast growth factor 23 is associated with coronary calcification in patients with high adiponectin: Analysis from the KoreaN cohort study for Outcome in patients With Chronic Kidney Disease (KNOW-CKD) study. Nephrol. Dial. Transplant. 2018, 34, 123–129. [Google Scholar] [CrossRef]
- Spoto, B.; Pizzini, P.; Tripepi, G.; Mallamaci, F.; Zoccali, C. Circulating adiponectin modifies the FGF23 response to vitamin D receptor activation: A post hoc analysis of a double-blind, randomized clinical trial. Nephrol. Dial. Transplant. 2018, 33, 1764–1769. [Google Scholar] [CrossRef]
- Kohno, K.; Narimatsu, H.; Shiono, Y.; Suzuki, I.; Kato, Y.; Sho, R.; Otani, K.; Ishizawa, K.; Yamashita, H.; Kubota, I.; et al. High Serum Adiponectin Level Is a Risk Factor for Anemia in Japanese Men: A Prospective Observational Study of 1029 Japanese Subjects. PLoS ONE 2016, 11, e0165511. [Google Scholar] [CrossRef] [Green Version]
- Tsuboi, A.; Watanabe, M.; Kazumi, T.; Fukuo, K. Anemia and Reduced Renal Function are Independent Predictors of Elevated Serum Adiponectin in Elderly Women. J. Atheroscler. Thromb. 2013, 20, 568–574. [Google Scholar] [CrossRef] [Green Version]
- Kim, H.; Yun, H.-R.; Park, S.; Jhee, J.H.; Park, J.T.; Yoo, T.-H.; Lee, K.-B.; Kim, Y.-H.; Sung, S.-A.; Lee, J.; et al. High serum adiponectin is associated with anemia development in chronic kidney disease: The results from the KNOW-CKD study. Cytokine 2018, 103, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Aso, Y.; Suganuma, R.; Wakabayashi, S.; Hara, K.; Nakano, T.; Suetsugu, M.; Matsumoto, S.; Nakamachi, T.; Takebayashi, K.; Morita, K.; et al. Anemia is associated with an elevated serum level of high-molecular-weight adiponectin in patients with type 2 diabetes independently of renal dysfunction. Transl. Res. 2009, 154, 175–182. [Google Scholar] [CrossRef] [PubMed]
- Moorthi, R.N.; Fadel, W.; Eckert, G.J.; Ponsler-Sipes, K.; Moe, S.M.; Lin, C. Bone marrow fat is increased in chronic kidney disease by magnetic resonance spectroscopy. Osteoporos. Int. 2015, 26, 1801–1807. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Naveiras, O.; Nardi, V.; Wenzel, P.L.; Hauschka, P.V.; Fahey, F.; Daley, G.Q. Bone-marrow adipocytes as negative regulators of the haematopoietic microenvironment. Nature 2009, 460, 259–263. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schober, F.; Neumeier, M.; Weigert, J.; Wurm, S.; Wanninger, J.; Schäffler, A.; Dada, A.; Liebisch, G.; Schmitz, G.; Aslanidis, C.; et al. Low molecular weight adiponectin negatively correlates with the waist circumference and monocytic IL-6 release. Biochem. Biophys. Res. Commun. 2007, 361, 968–973. [Google Scholar] [CrossRef]
- Neumeier, M.; Weigert, J.; Schäffler, A.; Wehrwein, G.; Müller-Ladner, U.; Schölmerich, J.; Wrede, C.; Buechler, C. Different effects of adiponectin isoforms in human monocytic cells. J. Leukoc. Biol. 2006, 79, 803–808. [Google Scholar] [CrossRef] [Green Version]
- Song, H.; Chan, J.; Rovin, B.H. Induction of chemokine expression by adiponectin in vitro is isoform dependent. Transl. Res. 2009, 154, 18–26. [Google Scholar] [CrossRef] [Green Version]
- Lee, K.-H.; Jeong, J.; Woo, J.; Lee, C.-H.; Yoo, C.-G. Globular Adiponectin Exerts a Pro-Inflammatory Effect via IκB/NF-κB Pathway Activation and Anti-Inflammatory Effect by IRAK-1 Downregulation. Mol. Cells 2018, 41, 762–770. [Google Scholar] [CrossRef]
- Hattori, Y.; Nakano, Y.; Hattori, S.; Tomizawa, A.; Inukai, K.; Kasai, K. High molecular weight adiponectin activates AMPK and suppresses cytokine-induced NF-κB activation in vascular endothelial cells. FEBS Lett. 2008, 582, 1719–1724. [Google Scholar] [CrossRef] [Green Version]
- Münzberg, H.; Morrison, C.D. Structure, production and signaling of leptin. Metabolism 2015, 64, 13–23. [Google Scholar] [CrossRef] [Green Version]
- Myers, M.G.; Cowley, M.A.; Münzberg, H. Mechanisms of Leptin Action and Leptin Resistance. Annu. Rev. Physiol. 2008, 70, 537–556. [Google Scholar] [CrossRef] [PubMed]
- Rahmouni, K.; Haynes, W.G.; Morgan, D.A.; Mark, A.L. Intracellular Mechanisms Involved in Leptin Regulation of Sympathetic Outflow. Hypertension 2003, 41, 763–767. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Harlan, S.M.; Rahmouni, K. PI3K signaling: A key pathway in the control of sympathetic traffic and arterial pressure by leptin. Mol. Metab. 2013, 2, 69–73. [Google Scholar] [CrossRef] [PubMed]
- La Cava, A. Leptin in inflammation and autoimmunity. Cytokine 2016, 98, 51–58. [Google Scholar] [CrossRef] [PubMed]
- Katsiki, N.; Mikhailidis, D.P.; Banach, M. Leptin, cardiovascular diseases and type 2 diabetes mellitus. Acta Pharmacol. Sin. 2018, 39, 1176–1188. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wolf, G.; Ziyadeh, F.N. Leptin and Renal Fibrosis. Contrib. Nephrol. 2006, 151, 175–183. [Google Scholar] [CrossRef]
- Korczynska, J.; Czumaj, A.; Chmielewski, M.; Swierczynski, J.; Sledzinski, T. The Causes and Potential Injurious Effects of Elevated Serum Leptin Levels in Chronic Kidney Disease Patients. Int. J. Mol. Sci. 2021, 22, 4685. [Google Scholar] [CrossRef]
- Alix, P.M.; Guebre-Egziabher, F.; Soulage, C.O. Leptin as an uremic toxin: Deleterious role of leptin in chronic kidney disease. Biochimie 2014, 105, 12–21. [Google Scholar] [CrossRef]
- Van Tellingen, A.; Grooteman, M.P.; Schoorl, M.; Ter Wee, P.M.; Bartels, P.C.; Schoorl, M.; Van Der Ploeg, T.; Nubé, M.J. Enhanced long-term reduction of plasma leptin concentrations by super-flux polysulfone dialysers. Nephrol. Dial. Transplant. 2004, 19, 1198–1203. [Google Scholar] [CrossRef]
- Kim, S.; Oh, K.H.; Chin, H.J.; Na, K.Y.; Kim, Y.S.; Chae, D.W.; Ahn, C.; Han, J.; Joo, K. Effective removal of leptin via hemodiafiltration with on-line endogenous reinfusion therapy. Clin. Nephrol. 2009, 72, 442–448. [Google Scholar] [CrossRef]
- Golembiewska, E.; Safranow, K.; Ciechanowski, K.; Bober, J.; Bogacka, A.; Stepniewska, J. Adipokines and parameters of peritoneal membrane transport in newly started peritoneal dialysis patients. Acta Biochim. Pol. 2013, 60, 617–621. [Google Scholar] [CrossRef] [PubMed]
- Aminzadeh, M.A.; Pahl, M.V.; Barton, C.H.; Doctor, N.S.; Vaziri, N.D. Human uraemic plasma stimulates release of leptin and uptake of tumour necrosis factor- in visceral adipocytes. Nephrol. Dial. Transplant. 2009, 24, 3626–3631. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ambarkar, M.; Pemmaraju, S.V.; Gouroju, S.; Manohar, S.M.; Bitla, A.R.; Yajamanam, N.; Vishnubhotla, S. Adipokines and their Relation to Endothelial Dysfunction in Patients with Chronic Kidney Disease. J. Clin. Diagn. Res. 2016, 10, BC04–BC08. [Google Scholar] [CrossRef] [PubMed]
- Noor, S.; Alam, F.; Fatima, S.S.; Khan, M.; Rehman, R. Role of Leptin and dyslipidemia in chronic kidney disease. Pak. J. Pharm. Sci. 2018, 31, 893–897. [Google Scholar]
- Dessie, G.; Ayelign, B.; Akalu, Y.; Shibabaw, T.; Molla, M.D. Effect of Leptin on Chronic Inflammatory Disorders: Insights to Therapeutic Target to Prevent Further Cardiovascular Complication. Diabetes Metab. Syndr. Obes. 2021, 14, 3307–3322. [Google Scholar] [CrossRef]
- Xu, F.-P.; Chen, M.-S.; Wang, Y.-Z.; Yi, Q.; Lin, S.-B.; Chen, A.F.; Luo, J.-D. Leptin Induces Hypertrophy via Endothelin-1–Reactive Oxygen Species Pathway in Cultured Neonatal Rat Cardiomyocytes. Circulation 2004, 110, 1269–1275. [Google Scholar] [CrossRef] [Green Version]
- He, W.-H.; Li, B.; Zhu, X.; Zhang, K.-H.; Li, B.-M.; Liu, Z.-J.; Liu, G.-Y.; Wang, J. The role and mechanism of NADPH oxidase in leptin-induced reactive oxygen species production in hepatic stellate cells. Zhonghua Gan Zang Bing Za Zhi 2010, 18, 849–854. [Google Scholar]
- Kovesdy, C.P.; Furth, S.L.; Zoccali, C.; World Kidney Day Steering Committee. Obesity and kidney disease: Hidden consequences of the epidemic. J. Ren. Care 2017, 43, 3–10. [Google Scholar] [CrossRef] [Green Version]
- Nehus, E.; Furth, S.; Warady, B.; Mitsnefes, M. Correlates of Leptin in Children with Chronic Kidney Disease. J. Pediatr. 2014, 165, 825–829. [Google Scholar] [CrossRef] [Green Version]
- Saluk, J.; Bansal, V.; Hoppensteadt, D.; Syed, D.; Abro, S.; Fareed, J. Prevalence of metabolic syndrome in patients with end stage renal disease and relevance of biomarkers. Int. Angiol. 2016, 35, 47–56. [Google Scholar] [CrossRef]
- Tsai, J.-P.; Tsai, C.-C.; Liu, H.-M.; Lee, C.-J.; Liou, H.-H.; Hsu, B.-G. Hyperleptinaemia positively correlated with metabolic syndrome in hemodialysis patients. Eur. J. Intern. Med. 2011, 22, e105–e109. [Google Scholar] [CrossRef] [PubMed]
- Zoccali, C.; Postorino, M.; Marino, C.; Pizzini, P.; Cutrupi, S.; Tripepi, G.; CREDIT Working Group. Waist circumference modifies the relationship between the adipose tissue cytokines leptin and adiponectin and all-cause and cardiovascular mortality in haemodialysis patients. J. Intern. Med. 2011, 269, 172–181. [Google Scholar] [CrossRef] [PubMed]
- Díez, J.; Bossola, M.; Fernández-Reyes, M.; Di Stasio, E.; Tazza, L.; Luciani, G.; Codoceo, R.; Iglesias, P.; Rodríguez, A.; González, E.; et al. Relationship between leptin and all-cause and cardiovascular mortality in chronic hemodialysis patients. Nefrología 2011, 31, 206–212. [Google Scholar] [CrossRef]
- Beberashvili, I.; Sinuani, I.; Azar, A.; Yasur, H.; Feldman, L.; Efrati, S.; Averbukh, Z.; Weissgarten, J. Nutritional and Inflammatory Status of Hemodialysis Patients in Relation to Their Body Mass Index. J. Ren. Nutr. 2009, 19, 238–247. [Google Scholar] [CrossRef]
- Delgado, C.; Chertow, G.M.; Kaysen, G.A.; Dalrymple, L.S.; Kornak, J.; Grimes, B.; Johansen, K.L. Associations of Body Mass Index and Body Fat With Markers of Inflammation and Nutrition Among Patients Receiving Hemodialysis. Am. J. Kidney Dis. 2017, 70, 817–825. [Google Scholar] [CrossRef]
- Kara, E.; Ahbap, E.; Sahutoglu, T.; Sakaci, T.; Basturk, T.; Koc, Y.; Sevinc, M.; Akgöl, C.; Ucar, Z.A.; Kayalar, A.O.; et al. Elevated serum leptin levels are associated with good nutritional status in non-obese chronic hemodialysis patients. Clin. Nephrol. 2015, 83, 147–153. [Google Scholar] [CrossRef] [PubMed]
- Risovic, I.; Vlatkovic, V.; Popovic-Pejicic, S.; Malešević, G. Relationship between serum leptin levels, non-cardiovascular risk factors and mortality in hemodialysis patients. Rom. J. Intern. Med. 2021, 59, 187–193. [Google Scholar] [CrossRef]
- Risović, I.; Vlatković, V.; Popović-Pejičić, S.; Trbojević-Stanković, J. Relationship between Leptin Level, Inflammation, and Volume Status in Maintenance Hemodialysis Patients. Ther. Apher. Dial. 2019, 23, 59–64. [Google Scholar] [CrossRef]
- Mitsides, N.; Cornelis, T.; Broers, N.J.H.; Diederen, N.M.P.; Brenchley, P.; Van Der Sande, F.M.; Schalkwijk, C.G.; Kooman, J.P.; Mitra, S. Extracellular overhydration linked with endothelial dysfunction in the context of inflammation in haemodialysis dependent chronic kidney disease. PLoS ONE 2017, 12, e0183281. [Google Scholar] [CrossRef] [Green Version]
- Lee, J.Y.; Kim, J.-S.; Yang, J.-W.; Choi, S.O.; Sohn, J.H.; Han, B.-G. Serum leptin level is associated with phase angle in CKD5 patients not undergoing dialysis. PLoS ONE 2018, 13, e0202055. [Google Scholar] [CrossRef]
- Abe, M.; Okada, K.; Maruyama, T.; Maruyama, N.; Matsumoto, K.; Soma, M. Relationship Between Erythropoietin Responsiveness, Insulin Resistance, and Malnutrition-Inflammation-Atherosclerosis (Mia) Syndrome in Hemodialysis Patients with Diabetes. Int. J. Artif. Organs 2011, 34, 16–25. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Yao, J.; Yao, Y.; Boström, K.I. Contributions of the Endothelium to Vascular Calcification. Front. Cell Dev. Biol. 2021, 9, 620882. [Google Scholar] [CrossRef] [PubMed]
- Ding, N.; Liu, B.; Song, J.; Bao, S.; Zhen, J.; Lv, Z.; Wang, R. Leptin promotes endothelial dysfunction in chronic kidney disease through AKT/GSK3β and β-catenin signals. Biochem. Biophys. Res. Commun. 2016, 480, 544–551. [Google Scholar] [CrossRef] [PubMed]
- Liu, B.; Qiao, J.; Hu, J.; Fan, M.; Zhao, Y.; Su, H.; Wang, Z.; Yu, Q.; Ma, Q.; Li, Y.; et al. Leptin promotes endothelial dysfunction in chronic kidney disease by modulating the MTA1-mediated WNT/β-catenin pathway. Mol. Cell. Biochem. 2020, 473, 155–166. [Google Scholar] [CrossRef]
- Lu, J.-W.; Chi, P.-J.; Lin, Y.-L.; Wang, C.-H.; Hsu, B.-G. Serum leptin levels are positively associated with aortic stiffness in patients with chronic kidney disease stage 3–5. Adipocyte 2020, 9, 206–211. [Google Scholar] [CrossRef]
- Nemcsik, J.; Kiss, I.; Tislér, A. Arterial stiffness, vascular calcification and bone metabolism in chronic kidney disease. World J. Nephrol. 2012, 1, 25–34. [Google Scholar] [CrossRef] [Green Version]
- Kastarinen, H.; Kesäniemi, Y.A.; Ukkola, O. Leptin and lipid metabolism in chronic kidney failure. Scand. J. Clin. Lab. Investig. 2009, 69, 401–408. [Google Scholar] [CrossRef]
- Korczyńska, J.; Czumaj, A.; Chmielewski, M.; Sledziński, M.; Mika, A.; Sledziński, T. Increased Expression of the Leptin Gene in Adipose Tissue of Patients with Chronic Kidney Disease–The Possible Role of an Abnormal Serum Fatty Acid Profile. Metabolites 2020, 10, 98. [Google Scholar] [CrossRef] [Green Version]
- Scholze, A.; Rattensperger, D.; Zidek, W.; Tepel, M. Low Serum Leptin Predicts Mortality in Patients with Chronic Kidney Disease Stage 5. Obesity 2007, 15, 1617–1622. [Google Scholar] [CrossRef]
- Molnar, M.Z.; Nagy, K.; Remport, A.; Gaipov, A.; Fülöp, T.; Czira, M.E.; Kovesdy, C.P.; Mucsi, I.; Mathe, Z. Association Between Serum Leptin Level and Mortality in Kidney Transplant Recipients. J. Ren. Nutr. 2017, 27, 53–61. [Google Scholar] [CrossRef]
- Qin, Z.; Yang, Q.; Yang, M.; Han, M.; Xiong, Y.; Liao, R.; Su, B. Serum leptin concentration can predict cardiovascular outcomes and all-cause death in maintenance hemodialysis patients. Clin. Chim. Acta 2021, 520, 87–94. [Google Scholar] [CrossRef] [PubMed]
- Sweigert, P.J.; Bansal, V.K.; Hoppensteadt, D.A.; Saluk, J.L.; Syed, D.A.; Fareed, J. Inflammatory and Metabolic Syndrome Biomarker Analysis of Vascular Outcomes in End-stage Renal Disease. Int. J. Angiol. 2017, 26, 43–48. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Motyl, K.J.; Rosen, C.J. Understanding leptin-dependent regulation of skeletal homeostasis. Biochimie 2012, 94, 2089–2096. [Google Scholar] [CrossRef]
- Park, S.-H.; Jia, T.; Qureshi, A.R.; Barany, P.; Heimbürger, O.; Larsson, T.E.; Axelsson, J.; Stenvinkel, P.; Lindholm, B. Determinants and survival implications of low bone mineral density in end-stage renal disease patients. J. Nephrol. 2013, 26, 485–494. [Google Scholar] [CrossRef] [PubMed]
- Coen, G. Leptin and bone metabolism. J. Nephrol. 2004, 17, 187–189. [Google Scholar]
- Wang, C.-H.; Lai, Y.-H.; Lin, Y.-L.; Kuo, C.-H.; Syu, R.-J.; Chen, M.-C.; Hsu, B.-G. Increased Serum Leptin Level Predicts Bone Mineral Density in Hemodialysis Patients. Int. J. Endocrinol. 2020, 2020, 8451751. [Google Scholar] [CrossRef]
- Ghazali, A.; Grados, F.; Oprisiu, R.; Bunea, D.; Morinière, P.; El Esper, N.; El Esper, I.; Brazier, M.; Souberbielle, J.C.; Fournier, A.; et al. Bone mineral density directly correlates with elevated serum leptin in haemodialysis patients. Nephrol. Dial. Transplant. 2003, 18, 1882–1890. [Google Scholar] [CrossRef] [Green Version]
- de Oliveira, R.B.; Liabeuf, S.; Okazaki, H.; Lenglet, A.; Desjardins, L.; Lemke, H.-D.; Vanholder, R.; Choukroun, G.; Massy, Z.A.; European Uremic Toxin Work Group (EUTox). The clinical impact of plasma leptin levels in a cohort of chronic kidney disease patients. Clin. Kidney J. 2012, 6, 63–70. [Google Scholar] [CrossRef] [Green Version]
- Ahmadi, F.; Salari, S.; Maziar, S.; Esfahanian, F.; Khazaeipour, Z.; Ranjbarnovin, N. Relationship between serum leptin levels and bone mineral density and bone metabolic markers in patients on hemodialysis. Saudi J. Kidney Dis. Transpl. 2013, 24, 41–47. [Google Scholar] [CrossRef]
- Jiang, Y.; Zhang, J.; Yuan, Y.; Zha, X.; Xing, C.; Shen, C.; Shen, Z.; Qin, C.; Zeng, M.; Yang, G.; et al. Association of Increased Serum Leptin with Ameliorated Anemia and Malnutrition in Stage 5 Chronic Kidney Disease Patients after Parathyroidectomy. Sci. Rep. 2016, 6, 27918. [Google Scholar] [CrossRef] [Green Version]
- Axelsson, J.; Qureshi, A.R.; Heimbürger, O.; Lindholm, B.; Stenvinkel, P.; Bárány, P. Body Fat Mass and Serum Leptin Levels Influence Epoetin Sensitivity in Patients With ESRD. Am. J. Kidney Dis. 2005, 46, 628–634. [Google Scholar] [CrossRef] [PubMed]
- Hung, S.-C.; Tung, T.-Y.; Yang, C.-S.; Tarng, D.-C. High-Calorie Supplementation Increases Serum Leptin Levels and Improves Response to rHuEPO in Long-Term Hemodialysis Patients. Am. J. Kidney Dis. 2005, 45, 1073–1083. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, K.; Kuragano, T.; Kimura, T.; Nanami, M.; Hasuike, Y.; Nakanishi, T. Interplay of adipocyte and hepatocyte: Leptin upregulates hepcidin. Biochem. Biophys. Res. Commun. 2018, 495, 1548–1554. [Google Scholar] [CrossRef]
- Uslu, S.; Kebapçi, N.; Kara, M.; Bal, C. Relationship between adipocytokines and cardiovascular risk factors in patients with type 2 diabetes mellitus. Exp. Ther. Med. 2012, 4, 113–120. [Google Scholar] [CrossRef]
- Abdella, N.A.; Mojiminiyi, O.A.; Moussa, M.A.; Zaki, M.; Al Mohammedi, H.; Al Ozairi, E.S.S.; Al Jebely, S. Plasma leptin concentration in patients with Type 2 diabetes: Relationship to cardiovascular disease risk factors and insulin resistance. Diabet. Med. 2005, 22, 278–285. [Google Scholar] [CrossRef] [PubMed]
- Asakawa, H.; Tokunaga, K.; Kawakami, F. Relationship of leptin level with metabolic disorders and hypertension in Japanese type 2 diabetes mellitus patients. J. Diabetes Complic. 2001, 15, 57–62. [Google Scholar] [CrossRef]
- Lopez-Jaramillo, P.; Gomez-Arbelaez, D.; Lopez-Lopez, J.; Lopez-Lopez, C.; Martinez-Ortega, J.; Gomez-Rodriguez, A.; Triana-Cubillos, S. The role of leptin/adiponectin ratio in metabolic syndrome and diabetes. Horm. Mol. Biol. Clin. Investig. 2014, 18, 37–45. [Google Scholar] [CrossRef] [Green Version]
- Kadowaki, T.; Yamauchi, T.; Kubota, N.; Hara, K.; Ueki, K.; Tobe, K. Adiponectin and adiponectin receptors in insulin resistance, diabetes, and the metabolic syndrome. J. Clin. Investig. 2006, 116, 1784–1792. [Google Scholar] [CrossRef] [Green Version]
- Iwashima, Y.; Katsuya, T.; Ishikawa, K.; Ouchi, N.; Ohishi, M.; Sugimoto, K.; Fu, Y.; Motone, M.; Yamamoto, K.; Matsuo, A.; et al. Hypoadiponectinemia Is an Independent Risk Factor for Hypertension. Hypertension 2004, 43, 1318–1323. [Google Scholar] [CrossRef] [Green Version]
- Dervisoglu, E.; Eraldemir, C.; Kalender, B.; Kir, H.M.; Caglayan, C. Adipocytokines Leptin and Adiponectin, and Measures of Malnutrition-Inflammation in Chronic Renal Failure: Is There a Relationship? J. Ren. Nutr. 2008, 18, 332–337. [Google Scholar] [CrossRef]



| Adiponectin | Leptin | |
|---|---|---|
| Main site of synthesis | Adipocyte | Adipocyte |
| Molecular weight | 30 kDa | 16 kDa |
| Receptors | AdipoR1, AdipoR2 and T-cadherin | LepR (six isoforms: a–f) |
| Biological actions related to CVD | Anti-inflammatory Antidyslipidemic Antidiabetic Changes in arterial stiffness | Regulation of food intake and insulin sensitivity Proinflammatory Changes in arterial stiffness |
| Circulating levels in ESRD | ↑↑ | ↑ |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Coimbra, S.; Rocha, S.; Valente, M.J.; Catarino, C.; Bronze-da-Rocha, E.; Belo, L.; Santos-Silva, A. New Insights into Adiponectin and Leptin Roles in Chronic Kidney Disease. Biomedicines 2022, 10, 2642. https://doi.org/10.3390/biomedicines10102642
Coimbra S, Rocha S, Valente MJ, Catarino C, Bronze-da-Rocha E, Belo L, Santos-Silva A. New Insights into Adiponectin and Leptin Roles in Chronic Kidney Disease. Biomedicines. 2022; 10(10):2642. https://doi.org/10.3390/biomedicines10102642
Chicago/Turabian StyleCoimbra, Susana, Susana Rocha, Maria João Valente, Cristina Catarino, Elsa Bronze-da-Rocha, Luís Belo, and Alice Santos-Silva. 2022. "New Insights into Adiponectin and Leptin Roles in Chronic Kidney Disease" Biomedicines 10, no. 10: 2642. https://doi.org/10.3390/biomedicines10102642





