The Cardiovascular System in Space: Focus on In Vivo and In Vitro Studies
Abstract
1. Introduction
2. Spaceflight Hazards
2.1. Isolation and Confinement
- Isolation and confinement are examples of psychological stressors. They induce a variety of psychological and physical effects, such as motivational decline, fatigue, somatic complaints (i.e., insomnia and headaches), and social tensions [6]. Two studies have correlated long-term isolation and confinement with strained crew relations, heightened friction, and social conflicts [7,8]. Further psychological stressors are the limited possibilities for rescue, high-risk conditions, and sleep disruptions [9].
- Multicultural and multinational factors are seen as important psychosocial stressors that have been related to preflight, inflight, and postflight incidences. National cultures influence several aspects of crew performance, such as communication, technology interfacing, religion and holidays, habitat aesthetics and work, management, and leadership style [10]. In addition, psychosocial stressors that have been identified are interpersonal tensions between crew and/or ground stations, disruptions in family life, and crew factors such as age, personality, etc.
- Space human factors involve the study of the interface between humans and technology. This discipline applies the principles by which humans interact with intelligent machines/tools. Several principles to take into account include hand–eye coordination, cognition, information processing, and memory capabilities. Other human factors are high/low levels of workload, danger, and risks associated with equipment failure or malfunctions [9].
- The habitability of the spacecraft refers to a multitude of areas, including architecture, clothing, crew displays/interfaces, housekeeping, hygiene, lighting, and privacy, among others [11]. The integration and support of human, technological, environmental, and mission elements promote crew performance, safety, and physical and psychological health during long-duration spaceflight. Other stressors related to the habitability are chronic exposure to vibrations/noise, limited sleep facilities, and isolation from support systems [6].
2.2. Space Radiation
2.3. Microgravity
3. Space Analogues
3.1. Isolation and Confinement Analogues
3.2. Radiation Analogues
3.3. Microgravity Analogues
3.3.1. In Vivo Microgravity Analogues
3.3.2. In Vitro Microgravity Analogues
4. Cardiovascular Diseases Related to Microgravity (In Vivo)
4.1. Cardiac System Adaptations and Remodelling
4.1.1. Haemodynamic Adaptations
4.1.2. Structural Adaptations
4.1.3. Cardiac Arrhythmias
4.2. Vascular System Adaptations and Remodelling
4.2.1. Haemodynamic Adaptations
4.2.2. Structural Adaptations
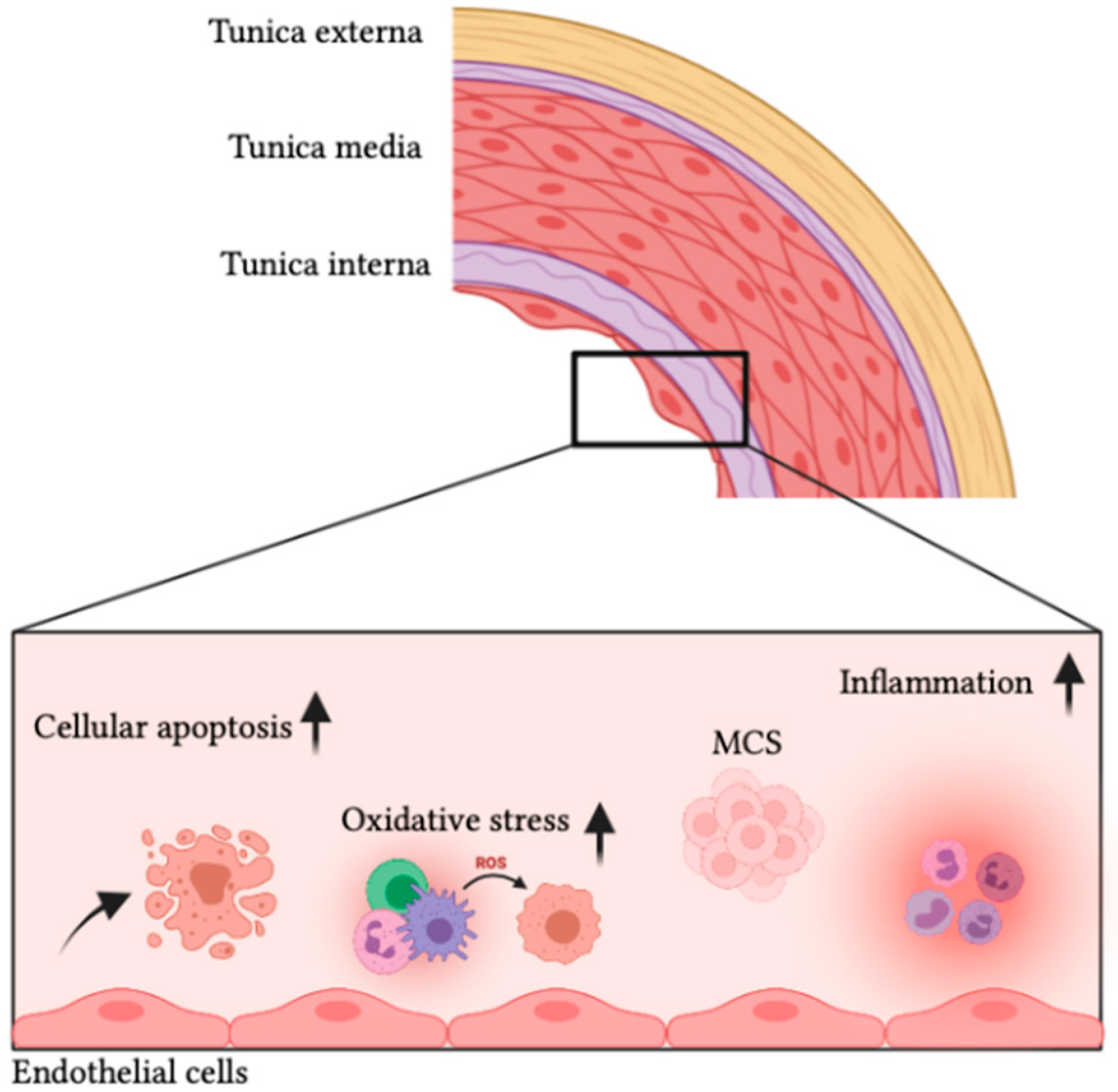
4.3. Endothelial Dysfunctions and Atherosclerosis
4.4. Blood Composition
4.5. Autonomic Cardiovascular System Adaptations
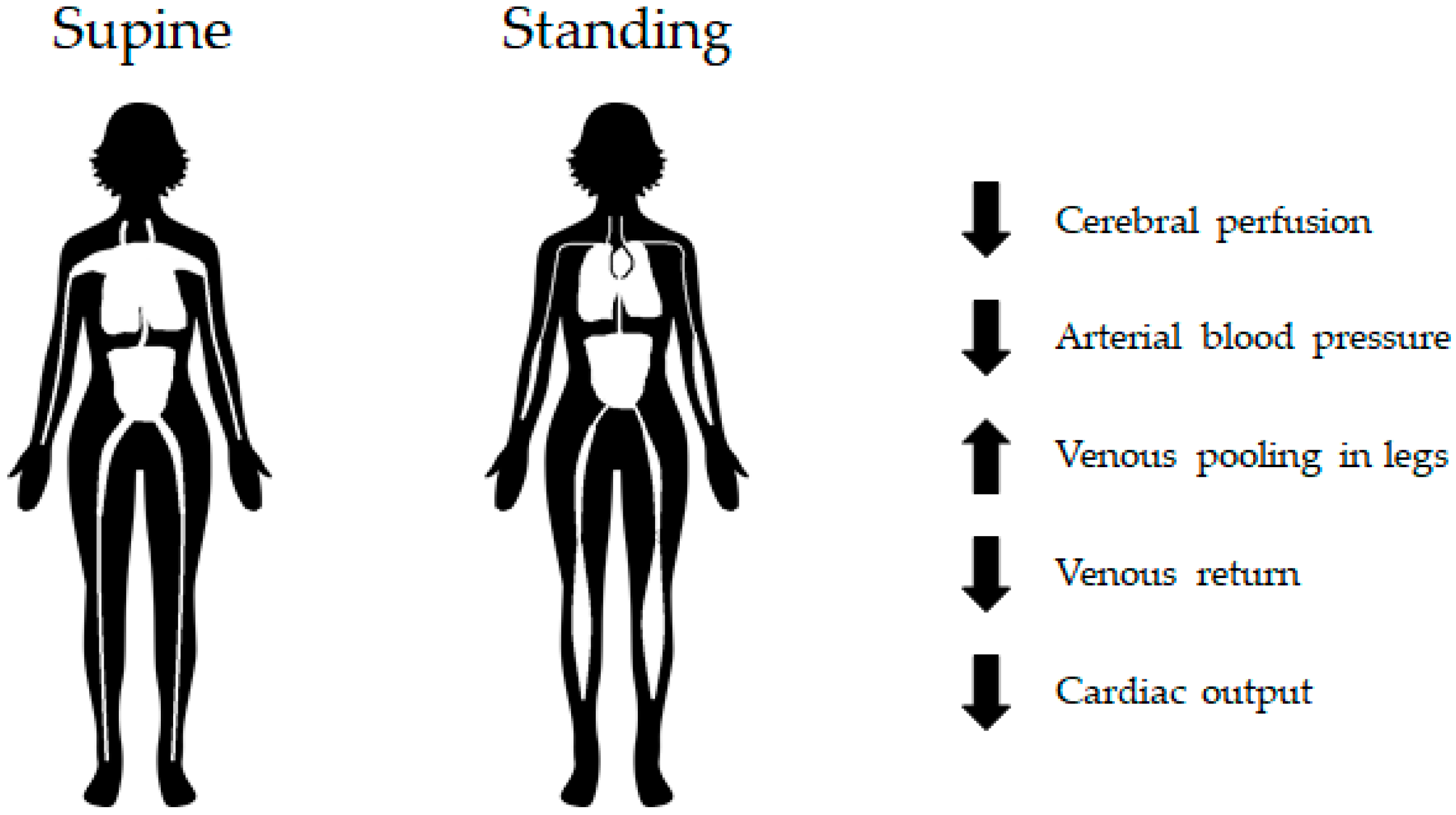
4.6. Orthostatic Intolerance
5. Cellular and Molecular Adaptation of Cardiovascular Cells to Spaceflight (In Vitro)
5.1. Fibroblasts
5.2. Vascular Smooth Muscle Cells
5.3. Endothelial Cells
5.3.1. Spheroid Formation and Angiogenesis
5.3.2. Endothelial Processes
5.3.3. Arterial Stiffness
5.4. Cardiomyocytes
5.5. Stem Cells
Endothelial Progenitor Cells
6. Conclusions
7. Materials and Methods
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| ANP | Atrial natriuretic peptide |
| AP | Arterial pressure |
| AS | Atherosclerosis |
| APC | Atrial premature contraction |
| CM | Conditioned media |
| CO | Cardiac output |
| CPC | Cardiovascular progenitor cell |
| CVD | Cardiovascular disease |
| CVP | Central venous pressure |
| DLR | Deutsches Zentrum für Luft- und Raumfahrt |
| DWI | Dry water immersion |
| EC | Endothelial cells |
| ECM | Extracellular matrix |
| EPC | Endothelial progenitor cell |
| EPO | Erythropoietin |
| ESA | European Space Agency |
| FN | Fibronectin |
| GCR | Galactic cosmic rays |
| Gy | Gray |
| HBR | Horizontal bedrest |
| HDBR | Head-down bed rest |
| HF | High frequency |
| HR | Heart rate |
| hPSC | Human pluripotent stem cells |
| HRV | Heart rate variability |
| HUVEC | Human umbilical vein endothelial cell |
| IF | Intermediate filament |
| IL | Interleukin |
| IMT | Intima media thickness |
| ISS | International Space Station |
| JAMIC | Japan Microgravity Center |
| JAXA | Japan Aerospace Exploration Agency |
| LDL | Low-density lipoprotein |
| LEO | Low Earth orbit |
| LF | Low frequency |
| LM | Laminin |
| MAP | Mean arterial pressure |
| MCS | Multicellular spheroids |
| ME | Microgravity followed by normal conditions |
| MeV | Megaelectronvolt |
| mSv | Millisieverts |
| MSC | Mesenchymal stem cell |
| MT | Microtubules |
| NASA | National Aeronautics and Space Administration |
| NEEMO | NASA’s Extreme Environment Mission Operation |
| NGAL | Neutrophil gelatinase associated lipocalin |
| Ni | Nickel |
| NO | Nitric oxide |
| OI | Orthostatic intolerance |
| QQc | Quality and quantity culture |
| RANTES | Regulated on activation normal T-cell expressed and secreted |
| RBC | Red blood cell |
| RE | Earth radius |
| RhoA | Ras homolog family member A |
| RICVD | Radiation induced cardiovascular disease |
| ROS | Reactive oxygen species |
| RPM | Random positioning machine |
| SAA | South Atlantic Anomaly |
| SV | Stroke volume |
| Sv | Sieverts |
| SVR | Systemic vascular resistance |
| TPR | Total peripheral resistance |
| VDCC | Voltage-dependent Ca2+ channels |
| VEGF | Vascular endothelial growth factor |
| VPC | Ventricular premature contraction |
| VSMC | Vascular smooth muscle cell |
| WI | Water immersion |
| ZARM | Center of Applied Science and Microgravity (Bremen, Germany) |
| µg | Microgravity |
| 2D | Two-dimensional |
| 3D | Three-dimensional |
References
- Meck, J.V.; Reyes, C.J.; Perez, S.A.; Goldberger, A.L.; Ziegler, M.G. Marked Exacerbation of Orthostatic Intolerance after Long- vs. Short-Duration Spaceflight in Veteran Astronauts. Psychosom. Med. 2001, 63, 865–873. [Google Scholar] [CrossRef]
- Vernice, N.A.; Meydan, C.; Afshinnekoo, E.; Mason, C.E. Long-Term Spaceflight and the Cardiovascular System. Precis. Clin. Med. 2020, 3, 284–291. [Google Scholar] [CrossRef] [PubMed]
- DiFrancesco, J.M.; Olson, J.M. The Economics of Microgravity Research. NPJ Microgravity 2015, 1, 15001. [Google Scholar] [CrossRef] [PubMed]
- Grimm, D.; Egli, M.; Krüger, M.; Riwaldt, S.; Corydon, T.J.; Kopp, S.; Wehland, M.; Wise, P.; Infanger, M.; Mann, V.; et al. Tissue Engineering under Microgravity Conditions-Use of Stem Cells and Specialized Cells. Stem Cells Dev. 2018, 27, 787–804. [Google Scholar] [CrossRef]
- Grimm, D.; Wehland, M.; Corydon, T.J.; Richter, P.; Prasad, B.; Bauer, J.; Egli, M.; Kopp, S.; Lebert, M.; Krüger, M. The Effects of Microgravity on Differentiation and Cell Growth in Stem Cells and Cancer Stem Cells. Stem Cells Transl. Med. 2020, 9, 882–894. [Google Scholar] [CrossRef]
- Choukér, A.; Stahn, A.C. COVID-19—The Largest Isolation Study in History: The Value of Shared Learnings from Spaceflight Analogs. NPJ Microgravity 2020, 6, 1–7. [Google Scholar] [CrossRef]
- Materassi, M.; Forte, B.; Coster, A.J.; Skone, S. (Eds.) The Dynamical Ionosphere: A Systems Approach to Ionospheric Irregularity, 1st ed.; Elsevier: Waltham, MA, USA, 2019; ISBN 978-0-12-814782-5. [Google Scholar]
- Furukawa, S.; Nagamatsu, A.; Nenoi, M.; Fujimori, A.; Kakinuma, S.; Katsube, T.; Wang, B.; Tsuruoka, C.; Shirai, T.; Nakamura, A.J.; et al. Space Radiation Biology for “Living in Space”. BioMed Res. Int. 2020, 2020, 4703286. [Google Scholar] [CrossRef]
- Morphew, E. Psychological and Human Factors in Long Duration Spaceflight. MJM 2020, 6. [Google Scholar] [CrossRef]
- Dachev, T.P.; Bankov, N.G.; Tomov, B.T.; Matviichuk, Y.N.; Dimitrov, P.G.; Häder, D.-P.; Horneck, G. Overview of the ISS Radiation Environment Observed during the ESA EXPOSE-R2 Mission in 2014-2016: ISS Radiation Environment. Space Weather 2017, 15, 1475–1489. [Google Scholar] [CrossRef]
- Seedhouse, E. Space Radiation and Astronaut Safety; Springer Briefs in Space Development; Springer International Publishing: Cham, Switzerland, 2018; ISBN 978-3-319-74614-2. [Google Scholar]
- Nicogossian, A.E.; Williams, R.S.; Huntoon, C.L.; Doarn, C.R.; Polk, J.D.; Schneider, V.S. Space Physiology and Medicine: From Evidence to Practice, 4th ed.; Springer: New York, NY, USA, 2016; ISBN 978-1-4939-6652-3. [Google Scholar]
- Lockwood, M.; Hapgood, M. The Rough Guide to the Moon and Mars. Astron Geophys. 2007, 48, 6.11–6.17. [Google Scholar] [CrossRef]
- Brenner, D.J.; Doll, R.; Goodhead, D.T.; Hall, E.J.; Land, C.E.; Little, J.B.; Lubin, J.H.; Preston, D.L.; Preston, R.J.; Puskin, J.S.; et al. Cancer Risks Attributable to Low Doses of Ionizing Radiation: Assessing What We Really Know. Proc. Natl. Acad. Sci. USA 2003, 100, 13761–13766. [Google Scholar] [CrossRef]
- Delp, M.D.; Charvat, J.M.; Limoli, C.L.; Globus, R.K.; Ghosh, P. Apollo Lunar Astronauts Show Higher Cardiovascular Disease Mortality: Possible Deep Space Radiation Effects on the Vascular Endothelium. Sci. Rep. 2016, 6, 29901. [Google Scholar] [CrossRef]
- Kunz, H.; Quiriarte, H.; Simpson, R.J.; Ploutz-Snyder, R.; McMonigal, K.; Sams, C.; Crucian, B. Alterations in Hematologic Indices during Long-Duration Spaceflight. BMC Hematol. 2017, 17, 12. [Google Scholar] [CrossRef]
- Shykoff, B.E.; Farhi, L.E.; Olszowka, A.J.; Pendergast, D.R.; Rokitka, M.A.; Eisenhardt, C.G.; Morin, R.A. Cardiovascular Response to Submaximal Exercise in Sustained Microgravity. J. Appl. Physiol. 1996, 81, 26–32. [Google Scholar] [CrossRef] [PubMed]
- Norsk, P.; Damgaard, M.; Petersen, L.; Gybel, M.; Pump, B.; Gabrielsen, A.; Christensen, N.J. Vasorelaxation in Space. Hypertension 2006, 47, 69–73. [Google Scholar] [CrossRef] [PubMed]
- Prisk, G.K.; Guy, H.J.; Elliott, A.R.; Deutschman, R.A.; West, J.B. Pulmonary Diffusing Capacity, Capillary Blood Volume, and Cardiac Output during Sustained Microgravity. J. Appl. Physiol. 1993, 75, 15–26. [Google Scholar] [CrossRef]
- Norsk, P.; Asmar, A.; Damgaard, M.; Christensen, N.J. Fluid Shifts, Vasodilatation and Ambulatory Blood Pressure Reduction during Long Duration Spaceflight: Vasodilatation and Ambulatory Blood Pressure during Spaceflight. J. Physiol. 2015, 593, 573–584. [Google Scholar] [CrossRef]
- Blomqvist, G.C. Regulation of the Systemic Circulation at Microgravity and during Readaptation to 1G. Med. Sci. Sports Exerc. 1996, 28, 9–13. [Google Scholar] [CrossRef]
- Mulvagh, S.L.; Charles, J.B.; Riddle, J.M.; Rehbein, T.L.; Bungo, M.W. Echocardiographic Evaluation of the Cardiovascular Effects of Short-Duration Spaceflight. J. Clin. Pharmacol. 1991, 31, 1024–1026. [Google Scholar] [CrossRef] [PubMed]
- Perhonen, M.A.; Franco, F.; Lane, L.D.; Buckey, J.C.; Blomqvist, C.G.; Zerwekh, J.E.; Peshock, R.M.; Weatherall, P.T.; Levine, B.D. Cardiac Atrophy after Bed Rest and Spaceflight. J. Appl. Physiol. 2001, 91, 645–653. [Google Scholar] [CrossRef]
- Fritsch-Yelle, J.M.; Charles, J.B.; Jones, M.M.; Wood, M.L. Microgravity Decreases Heart Rate and Arterial Pressure in Humans. J. Appl. Physiol. 1996, 80, 910–914. [Google Scholar] [CrossRef] [PubMed]
- Buckey, J.C.; Gaffney, F.A.; Lane, L.D.; Levine, B.D.; Watenpaugh, D.E.; Wright, S.J.; Yancy, C.W.; Meyer, D.M.; Blomqvist, C.G. Central Venous Pressure in Space. J. Appl. Physiol. 1996, 81, 19–25. [Google Scholar] [CrossRef] [PubMed]
- Videbaek, R.; Norsk, P. Atrial Distension in Humans during Microgravity Induced by Parabolic Flights. J. Appl. Physiol. 1997, 83, 1862–1866. [Google Scholar] [CrossRef] [PubMed]
- Hargens, A.R.; Richardson, S. Cardiovascular Adaptations, Fluid Shifts, and Countermeasures Related to Space Flight. Respir. Physiol. Neurobiol. 2009, 169, S30–S33. [Google Scholar] [CrossRef] [PubMed]
- Watenpaugh, D.E.; Buckey, J.C.; Lane, L.D.; Gaffney, F.A.; Levine, B.D.; Moore, W.E.; Wright, S.J.; Blomqvist, C.G. Effects of Spaceflight on Human Calf Hemodynamics. J. Appl. Physiol. 2001, 90, 1552–1558. [Google Scholar] [CrossRef]
- Norsk, P. Adaptation of the Cardiovascular System to Weightlessness: Surprises, Paradoxes and Implications for Deep Space Missions. Acta Physiol. 2020, 228. [Google Scholar] [CrossRef]
- Migeotte, P.-F.; Prisk, G.K.; Paiva, M. Microgravity Alters Respiratory Sinus Arrhythmia and Short-Term Heart Rate Variability in Humans. Am. J. Physiol. Heart Circ. Physiol. 2003, 284, H1995–H2006. [Google Scholar] [CrossRef][Green Version]
- Garrett-Bakelman, F.E.; Darshi, M.; Green, S.J.; Gur, R.C.; Lin, L.; Macias, B.R.; McKenna, M.J.; Meydan, C.; Mishra, T.; Nasrini, J.; et al. The NASA Twins Study: A Multidimensional Analysis of a Year-Long Human Spaceflight. Science 2019, 364, eaau8650. [Google Scholar] [CrossRef] [PubMed]
- Arbeille, P.; Provost, R.; Zuj, K. Carotid and Femoral Arterial Wall Distensibility during Long-Duration Spaceflight. Aerosp. Med. Hum. Perform. 2017, 88, 924–930. [Google Scholar] [CrossRef]
- Hughson, R.L.; Helm, A.; Durante, M. Heart in Space: Effect of the Extraterrestrial Environment on the Cardiovascular System. Nat. Rev. Cardiol. 2018, 15, 167–180. [Google Scholar] [CrossRef]
- Hughson, R.L.; Robertson, A.D.; Arbeille, P.; Shoemaker, J.K.; Rush, J.W.E.; Fraser, K.S.; Greaves, D.K. Increased Postflight Carotid Artery Stiffness and Inflight Insulin Resistance Resulting from 6-Mo Spaceflight in Male and Female Astronauts. Am. J. Physiol. Heart Circ. Physiol. 2016, 310, H628–H638. [Google Scholar] [CrossRef] [PubMed]
- Navasiolava, N.; Yuan, M.; Murphy, R.; Robin, A.; Coupé, M.; Wang, L.; Alameddine, A.; Gauquelin-Koch, G.; Gharib, C.; Li, Y.; et al. Vascular and Microvascular Dysfunction Induced by Microgravity and Its Analogs in Humans: Mechanisms and Countermeasures. Front. Physiol. 2020, 11, 952. [Google Scholar] [CrossRef] [PubMed]
- Iwase, S.; Nishimura, N.; Tanaka, K.; Mano, T. Effects of Microgravity on Human Physiology. In Beyond LEO—Human Health Issues for Deep Space Exploration [Working Title]; IntechOpen: London, UK, 2020. [Google Scholar]
- Pavy-Le Traon, A.; Heer, M.; Narici, M.V.; Rittweger, J.; Vernikos, J. From Space to Earth: Advances in Human Physiology from 20 Years of Bed Rest Studies (1986–2006). Eur. J. Appl. Physiol. 2007, 101, 143–194. [Google Scholar] [CrossRef] [PubMed]
- Aubert, A.E.; Beckers, F.; Verheyden, B. Cardiovascular Function and Basics of Physiology in Microgravity. Acta Cardiol. 2005, 129–151. [Google Scholar] [CrossRef] [PubMed]
- Watenpaugh, D.E. Fluid Volume Control during Short-Term Space Flight and Implications for Human Performance. J. Exp. Biol. 2001, 204, 3209–3215. [Google Scholar] [CrossRef]
- Ertl, A.C.; Diedrich, A.; Biaggioni, I.; Levine, B.D.; Robertson, R.M.; Cox, J.F.; Zuckerman, J.H.; Pawelczyk, J.A.; Ray, C.A.; Buckey, J.C.; et al. Human Muscle Sympathetic Nerve Activity and Plasma Noradrenaline Kinetics in Space. J. Physiol. 2002, 538, 321–329. [Google Scholar] [CrossRef]
- Pagel, J.I.; Choukèr, A. Effects of Isolation and Confinement on Humans-Implications for Manned Space Explorations. J. Appl. Physiol. 2016, 120, 1449–1457. [Google Scholar] [CrossRef]
- Xia, N.; Li, H. Loneliness, Social Isolation, and Cardiovascular Health. Antioxid. Redox Signal. 2018, 28, 837–851. [Google Scholar] [CrossRef]
- Harrison, A.A.; Clearwater, Y.A.; McKay, C.P. From Antarctica to Outer Space: Life in Isolation and Confinement; Springer: New York, NY, USA, 1991; ISBN 978-1-4612-3012-0. [Google Scholar]
- Sandal, G.M.; Endresen, I.M.; Vaernes, R.; Ursin, H. Personality and Coping Strategies During Submarine Missions. Mil. Psychol. 1999, 11, 381–404. [Google Scholar] [CrossRef]
- Vigo, D.E.; Tuerlinckx, F.; Ogrinz, B.; Wan, L.; Simonelli, G.; Bersenev, E.; Van den Bergh, O.; Aubert, A.E. Circadian Rhythm of Autonomic Cardiovascular Control During Mars500 Simulated Mission to Mars. Aviat. Space Environ. Med. 2013, 84, 1023–1028. [Google Scholar] [CrossRef]
- Baatout, S. Chapter 10 Space Analogs on Earth for Human (in Vivo) Studies. In Proceedings of the Life Sciences in Space, Leuven, Belgium, 1 May 2020. [Google Scholar]
- Koutnik, A.P.; Favre, M.E.; Noboa, K.; Sanchez-Gonzalez, M.A.; Moss, S.E.; Goubran, B.; Ari, C.; Poff, A.M.; Rogers, C.Q.; DeBlasi, J.M.; et al. Human Adaptations to Multiday Saturation on NASA NEEMO. Front. Physiol. 2021, 11, 610000. [Google Scholar] [CrossRef]
- Cuomo, J.R.; Sharma, G.K.; Conger, P.D.; Weintraub, N.L. Novel Concepts in Radiation-Induced Cardiovascular Disease. WJC 2016, 8, 504. [Google Scholar] [CrossRef]
- Darby, S.C.; Ewertz, M.; McGale, P.; Bennet, A.M.; Blom-Goldman, U.; Brønnum, D.; Correa, C.; Cutter, D.; Gagliardi, G.; Gigante, B.; et al. Risk of Ischemic Heart Disease in Women after Radiotherapy for Breast Cancer. N. Engl. J. Med. 2013, 368, 987–998. [Google Scholar] [CrossRef] [PubMed]
- Kashcheev, V.V.; Chekin, S.Y.; Karpenko, S.V.; Maksioutov, M.A.; Menyaylo, A.N.; Tumanov, K.A.; Kochergina, E.V.; Kashcheeva, P.V.; Gorsky, A.I.; Shchukina, N.V.; et al. Radiation Risk of Cardiovascular Diseases in the Cohort of Russian Emergency Workers of the Chernobyl Accident. Health Phys. 2017, 113, 23–29. [Google Scholar] [CrossRef]
- Boerma, M. Space Radiation and Cardiovascular Disease Risk. WJC 2015, 7, 882. [Google Scholar] [CrossRef] [PubMed]
- Preston, D.L.; Shimizu, Y.; Pierce, D.A.; Suyama, A.; Mabuchi, K. Studies of Mortality of Atomic Bomb Survivors. Report 13: Solid Cancer and Noncancer Disease Mortality: 1950–1997. Radiat. Res. 2003, 160, 381–407. [Google Scholar] [CrossRef]
- ESA Space Exploration Strategy; Strategic Planning and Outreach Office of the ESA Directorate of Human Spaceflight and Operations: Noordwijk, The Netherlands, 2015.
- Durante, M.; Reitz, G.; Angerer, O. Space Radiation Research in Europe: Flight Experiments and Ground-Based Studies. Radiat. Environ. Biophys. 2010, 49, 295–302. [Google Scholar] [CrossRef]
- Herranz, R.; Anken, R.; Boonstra, J.; Braun, M.; Christianen, P.C.M.; de Geest, M.; Hauslage, J.; Hilbig, R.; Hill, R.J.A.; Lebert, M.; et al. Ground-Based Facilities for Simulation of Microgravity: Organism-Specific Recommendations for Their Use, and Recommended Terminology. Astrobiology 2013, 13, 1–17. [Google Scholar] [CrossRef]
- NASA. NASA Analogs Field Testing: VIIP and Psychological—Envihab Research (VaPER). Available online: https://www.nasa.gov/sites/default/files/thumbnails/image/rsl_studie_10_09_2015_021.jpg (accessed on 27 September 2021).
- Nassef, M.Z.; Kopp, S.; Wehland, M.; Melnik, D.; Sahana, J.; Krüger, M.; Corydon, T.J.; Oltmann, H.; Schmitz, B.; Schütte, A.; et al. Real Microgravity Influences the Cytoskeleton and Focal Adhesions in Human Breast Cancer Cells. Int. J. Mol. Sci. 2019, 20, 3156. [Google Scholar] [CrossRef]
- Watenpaugh, D.E. Analogs of Microgravity: Head-down Tilt and Water Immersion. J. Appl. Physiol. 2016, 120, 904–914. [Google Scholar] [CrossRef] [PubMed]
- Cromwell, R.L.; Scott, J.M.; Downs, M.; Yarbough, P.O.; Zanello, S.B.; Ploutz-Snyder, L. Overview of the NASA 70-Day Bed Rest Study. Med. Sci. Sports Exerc. 2018, 50, 1909–1919. [Google Scholar] [CrossRef]
- Hargens, A.R.; Vico, L. Long-Duration Bed Rest as an Analog to Microgravity. J. Appl. Physiol. 2016, 120, 891–903. [Google Scholar] [CrossRef] [PubMed]
- Kramer, A.; Gollhofer, A.; Armbrecht, G.; Felsenberg, D.; Gruber, M. How to Prevent the Detrimental Effects of Two Months of Bed-Rest on Muscle, Bone and Cardiovascular System: An RCT. Sci. Rep. 2017, 7, 13177. [Google Scholar] [CrossRef]
- Amirova, L.; Navasiolava, N.; Rukavishvikov, I.; Gauquelin-Koch, G.; Gharib, C.; Kozlovskaya, I.; Custaud, M.-A.; Tomilovskaya, E. Cardiovascular System Under Simulated Weightlessness: Head-Down Bed Rest vs. Dry Immersion. Front. Physiol. 2020, 11, 395. [Google Scholar] [CrossRef] [PubMed]
- Tomilovskaya, E.; Shigueva, T.; Sayenko, D.; Rukavishnikov, I.; Kozlovskaya, I. Dry Immersion as a Ground-Based Model of Microgravity Physiological Effects. Front. Physiol. 2019, 10, 284. [Google Scholar] [CrossRef]
- Weber, B.; Schätzle, S.; Riecke, C. Comparing the Effects of Space Flight and Water Immersion on Sensorimotor Performance. In Proceedings of the Human Factors and Ergonomics Society Europe Chapter 2017 Annual Conference, Rome, Italy, 28–30 September 2018. [Google Scholar]
- Tomilovskaya, E.; Amirova, L.; Nosikova, I.; Rukavishnikov, I.; Chernogorov, R.; Lebedeva, S.; Saveko, A.; Ermakov, I.; Ponomarev, I.; Zelenskaya, I.; et al. The First Female Dry Immersion (NAIAD-2020): Design and Specifics of a 3-Day Study. Front. Physiol. 2021, 12, 661959. [Google Scholar] [CrossRef]
- Ulbrich, C.; Wehland, M.; Pietsch, J.; Aleshcheva, G.; Wise, P.; van Loon, J.; Magnusson, N.; Infanger, M.; Grosse, J.; Eilles, C.; et al. The Impact of Simulated and Real Microgravity on Bone Cells and Mesenchymal Stem Cells. BioMed Res. Int. 2014, 2014, 928507. [Google Scholar] [CrossRef]
- Schwarz, R.P.; Goodwin, T.J.; Wolf, D.A. Cell Culture for Three-Dimensional Modeling in Rotating-Wall Vessels: An Application of Simulated Microgravity. J. Tissue Cult. Methods 1992, 14, 51–57. [Google Scholar] [CrossRef]
- Hammond, T.G.; Hammond, J.M. Optimized Suspension Culture: The Rotating-Wall Vessel. Am. J. Physiol. Ren. Physiol. 2001, 281, F12–F25. [Google Scholar] [CrossRef]
- Grimm, D.; Wehland, M.; Pietsch, J.; Aleshcheva, G.; Wise, P.; van Loon, J.; Ulbrich, C.; Magnusson, N.E.; Infanger, M.; Bauer, J. Growing Tissues in Real and Simulated Microgravity: New Methods for Tissue Engineering. Tissue Eng. Part B Rev. 2014, 20, 555–566. [Google Scholar] [CrossRef] [PubMed]
- Newcombe, F.C. Limitations of the Klinostat as an Instrument for Scientific Research. Science 1904, 20, 376–379. [Google Scholar] [CrossRef]
- Cogoli, M. The Fast Rotating Clinostat: A History of Its Use in Gravitational Biology and a Comparison of Ground-Based and Flight Experiment Results. ASGSB Bull. 1992, 5, 59–67. [Google Scholar]
- Hemmersbach, R.; Strauch, S.M.; Seibt, D.; Schuber, M. Comparative Studies on Gravisensitive Protists on Ground (2D and 3D Clinostats) and in Microgravity. Microgravity Sci. Technol. 2006, 18, 257–259. [Google Scholar] [CrossRef]
- Yuge, D.L.; Kajiume, T.; Tahara, H.; Kawahara, Y.; Umeda, C.; Yoshimoto, R.; Wu, S.-L.; Yamaoka, K.; Asashima, M.; Kataoka, K.; et al. Microgravity Potentiates Stem Cell Proliferation While Sustaining the Capability of Differentiation. Available online: https://www.liebertpub.com/doi/abs/10.1089/scd.2006.15.921 (accessed on 18 May 2021).
- Nishikawa, M.; Ohgushi, H.; Tamai, N.; Osuga, K.; Uemura, M.; Yoshikawa, H.; Myoui, A. The Effect of Simulated Microgravity by Three-Dimensional Clinostat on Bone Tissue Engineering. Cell Transpl. 2005, 14, 829–835. [Google Scholar] [CrossRef]
- Van Loon, J.J.W.A. Some History and Use of the Random Positioning Machine, RPM, in Gravity Related Research. Adv. Space Res. 2007, 39, 1161–1165. [Google Scholar] [CrossRef]
- Wuest, S.; Richard, S.; Kopp, S.; Grimm, D.; Egli, M. Simulated Microgravity: Critical Review on the Use of Random Positioning Machines for Mammalian Cell Culture. BioMed Res. Int. 2015, 2015, 971474. [Google Scholar] [CrossRef] [PubMed]
- Brungs, S.; Petrat, G.; von der Wiesche, M.; Anken, R.; Kolanus, W.; Hemmersbach, R. Simulating Parabolic Flight like G-Profiles on Ground—A Combination of Centrifuge and Clinostat. Microgravity Sci. Technol. 2016, 28, 231–235. [Google Scholar] [CrossRef]
- MSFC. NASA—Drop Everything! Available online: https://www.nasa.gov/audience/foreducators/k-4/features/F_Drop_Everything.html (accessed on 18 May 2021).
- ZARM. ZARM: Bremen Drop Tower. Available online: https://www.zarm.uni-bremen.de/en/drop-tower/general-information.html (accessed on 18 May 2021).
- Cross, M. Technology: Japanese Space Research Comes down to Earth. Available online: https://www.newscientist.com/article/mg12717352-900-technology-japanese-space-research-comes-down-to-earth/ (accessed on 18 May 2021).
- Liu, T.Y.; Wu, Q.P.; Sun, B.Q.; Han, F.T. Microgravity Level Measurement of the Beijing Drop Tower Using a Sensitive Accelerometer. Sci. Rep. 2016, 6, 31632. [Google Scholar] [CrossRef] [PubMed]
- Haber, F.; Haber, H. Possible Methods of Producing the Gravity-Free State for Medical Research. J. Aviat. Med. 1950, 21, 395–400. [Google Scholar]
- Pletser, V. Short Duration Microgravity Experiments in Physical and Life Sciences during Parabolic Flights: The First 30 ESA Campaigns. Acta Astronaut. 2004, 55, 829–854. [Google Scholar] [CrossRef]
- Sawada, H.; Ui, K.; Mori, M.; Yamamoto, H.; Hayashi, R.; Matunaga, S.; Ohkami, Y. Micro-Gravity Experiment of a Space Robotic Arm Using Parabolic Flight. Adv. Robot. 2012, 18, 247–267. [Google Scholar] [CrossRef]
- De Witt, J.K.; Perusek, G.P.; Lewandowski, B.E.; Gilkey, K.M.; Savina, M.C.; Samorezov, S.; Edwards, W.B. Locomotion in Simulated and Real Microgravity: Horizontal Suspension vs. Parabolic Flight. Aviat. Space Environ. Med. 2010, 81, 1092–1099. [Google Scholar] [CrossRef] [PubMed]
- Martin, D.S.; Lee, S.M.C.; Matz, T.P.; Westby, C.M.; Scott, J.M.; Stenger, M.B.; Platts, S.H. Internal Jugular Pressure Increases during Parabolic Flight. Physiol. Rep. 2016, 4, e13068. [Google Scholar] [CrossRef] [PubMed]
- Carr, C.E.; Bryan, N.C.; Saboda, K.N.; Bhattaru, S.A.; Ruvkun, G.; Zuber, M.T. Acceleration Profiles and Processing Methods for Parabolic Flight. NPJ Microgravity 2018, 4, 1–5. [Google Scholar] [CrossRef]
- Mukai, C.N.; Lathers, C.M.; Charles, J.B.; Bennett, B.S.; Igarashi, M.; Patel, S. Acute Hemodynamic Responses to Weightlessness during Parabolic Flight. J. Clin. Pharmacol. 1991, 31, 993–1000. [Google Scholar] [CrossRef] [PubMed]
- DeLombard, R.; Hrovat, K.; Kelly, E.M.; Humphreys, B. Interpreting the International Space Station Microgravity Environment; AIAA Paper 2005-0727; AIAA: Reno, NV, USA, 2005; pp. 1–12. [Google Scholar]
- DLR International Space Station (ISS) Status on Completion. Available online: https://www.dlr.de/content/en/articles/missions-projects/iss/iss-data-modules.html (accessed on 7 April 2021).
- Witze, A. Astronauts Have Conducted Nearly 3000 Science Experiments Aboard the ISS. Nature. 2020. Available online: https://www.nature.com/articles/d41586-020-03085-8 (accessed on 9 December 2021).
- NASA Reference Guide to The International Space Station—Utilization Edition. 2015. Available online: https://www.nasa.gov/sites/default/files/atoms/files/np-2015-05-022-jsc-iss-guide-2015-update-111015-508c.pdf (accessed on 9 December 2021).
- Kovo, Y. Bion-M1. Available online: http://www.nasa.gov/ames/research/space-biosciences/bion-m1 (accessed on 27 May 2021).
- Kazakova, A.E.; Abrashkin, V.I.; Stratilatov, N.V.; Smirnov, N.N. Bion-M Satellite—The Unique Special-Purpose Laboratory. In Proceedings of the 57th International Astronautical Congress, Valencia, Spain, 2 October 2006; American Institute of Aeronautics and Astronautics: Valencia, Spain, 2006. [Google Scholar]
- Sychev, V.N.; Ilyin, E.A.; Yarmanova, E.N.; Rakov, D.V.; Ushakov, I.B.; Kirilin, A.N.; Orlov, O.I.; Grigoriev, A.I. The BION-M1 project: Overview and first results. Aviakosm. Ekolog. Med. 2014, 48, 7–14. [Google Scholar] [PubMed]
- Herault, S.; Fomina, G.; Alferova, I.; Kotovskaya, A.; Poliakov, V.; Arbeille, P. Cardiac, Arterial and Venous Adaptation to Weightlessness during 6-Month MIR Spaceflights with and without Thigh Cuffs (Bracelets). Eur. J. Appl. Physiol. 2000, 81, 384–390. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, K.; Nishimura, N.; Kawai, Y. Adaptation to Microgravity, Deconditioning, and Countermeasures. J. Physiol. Sci 2017, 67, 271–281. [Google Scholar] [CrossRef]
- Summers, R.L.; Martin, D.S.; Meck, J.V.; Coleman, T.G. Mechanism of Spaceflight-Induced Changes in Left Ventricular Mass. Am. J. Cardiol. 2005, 95, 1128–1130. [Google Scholar] [CrossRef]
- May, C.; Borowski, A.; Martin, D.; Popovic, Z.; Negishi, K.; Hussan, J.R.; Gladding, P.; Hunter, P.; Iskovitz, I.; Kassemi, M.; et al. Affect of microgravity on cardiac shape: Comparison of pre- and in-flight data to mathematical modeling. J. Am. Coll. Cardiol. 2014, 63, A1096. [Google Scholar] [CrossRef][Green Version]
- Anzai, T.; Frey, M.A.; Nogami, A. Cardiac Arrhythmias during Long-Duration Spaceflights. J. Arrhythmia 2014, 30, 139–149. [Google Scholar] [CrossRef]
- Edyvean, J.; Estenne, M.; Paiva, M.; Engel, L.A. Lung and Chest Wall Mechanics in Microgravity. J. Appl. Physiol. 1991, 71, 1956–1966. [Google Scholar] [CrossRef] [PubMed]
- Nicogossian, A.E.; Charles, J.B.; Bungo, M.W.; Leach-Huntoon, C.S.; Nicgossian, A.E. Cardiovascular Function in Space Flight. Acta Astronaut 1991, 24, 323–328. [Google Scholar] [CrossRef]
- Demontis, G.C.; Germani, M.M.; Caiani, E.G.; Barravecchia, I.; Passino, C.; Angeloni, D. Human Pathophysiological Adaptations to the Space Environment. Front. Physiol. 2017, 8, 547. [Google Scholar] [CrossRef] [PubMed]
- Arbeille, P.; Provost, R.; Zuj, K. Carotid and Femoral Artery Intima-Media Thickness During 6 Months of Spaceflight. Aerosp. Med. Hum. Perform. 2016, 87, 449–453. [Google Scholar] [CrossRef]
- Lee, S.M.C.; Ribeiro, L.C.; Martin, D.S.; Zwart, S.R.; Feiveson, A.H.; Laurie, S.S.; Macias, B.R.; Crucian, B.E.; Krieger, S.; Weber, D.; et al. Arterial Structure and Function during and after Long-Duration Spaceflight. J. Appl. Physiol. 2020, 129, 108–123. [Google Scholar] [CrossRef] [PubMed]
- Coupé, M.; Fortrat, J.O.; Larina, I.; Gauquelin-Koch, G.; Gharib, C.; Custaud, M.A. Cardiovascular Deconditioning: From Autonomic Nervous System to Microvascular Dysfunctions. Respir. Physiol. Neurobiol. 2009, 169, S10–S12. [Google Scholar] [CrossRef]
- Grossman, S.C.; Porth, C.M. Porth’s Pathophysiology Concepts of Altered Health States; Wolters Kluwer Lippincott Williams & Wilkins: Philadelphia, PA, USA, 2014; ISBN 978-1-4511-4600-4. [Google Scholar]
- Versari, D.; Daghini, E.; Virdis, A.; Ghiadoni, L.; Taddei, S. Endothelium-Dependent Contractions and Endothelial Dysfunction in Human Hypertension: Endothelium and Hypertension. Br. J. Pharmacol. 2009, 157, 527–536. [Google Scholar] [CrossRef]
- Félétou, M.; Vanhoutte, P.M. Endothelial Dysfunction: A Multifaceted Disorder (The Wiggers Award Lecture). Am. J. Physiol. Heart Circ. Physiol. 2006, 291, H985–H1002. [Google Scholar] [CrossRef]
- Haller, H. Endothelial Function: General Considerations. Drugs 1997, 53, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Sena, C.M.; Leandro, A.; Azul, L.; Seiça, R.; Perry, G. Vascular Oxidative Stress: Impact and Therapeutic Approaches. Front. Physiol. 2018, 9, 1668. [Google Scholar] [CrossRef]
- Avolio, A.; Butlin, M.; Liu, Y.-Y.; Viegas, K.; Avadhanam, B.; Lindesay, G. Regulation of arterial stiffness: Cellular, molecular and neurogenic mechanisms. ARTRES 2011, 5, 122. [Google Scholar] [CrossRef]
- Leach, C.S.; Johnson, P.C.; Cintron, N.M. The Endocrine System in Space Flight. Acta Astronaut. 1988, 17, 161–166. [Google Scholar] [CrossRef]
- Zieman, S.J.; Melenovsky, V.; Kass, D.A. Mechanisms, Pathophysiology, and Therapy of Arterial Stiffness. ATVB 2005, 25, 932–943. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, K.; Okumura, H.; Guo, R.; Naruse, K. Effect of Oxidative Stress on Cardiovascular System in Response to Gravity. Int. J. Mol. Sci. 2017, 18, 1426. [Google Scholar] [CrossRef] [PubMed]
- Hansson, G.K. Inflammation, Atherosclerosis, and Coronary Artery Disease. N. Engl. J. Med. 2005, 352, 1685–1695. [Google Scholar] [CrossRef] [PubMed]
- Wang, G.; Jacquet, L.; Karamariti, E.; Xu, Q. Origin and Differentiation of Vascular Smooth Muscle Cells: Smooth Muscle Cell Origin and Differentiation. J. Physiol. 2015, 593, 3013–3030. [Google Scholar] [CrossRef]
- Udden, M.M.; Driscoll, T.B.; Gibson, L.A.; Patton, C.S.; Pickett, M.H.; Jones, J.B.; Nachtman, R.; Allebban, Z.; Ichiki, A.T.; Lange, R.D. Blood Volume and Erythropoiesis in the Rat during Spaceflight. Aviat. Space Environ. Med. 1995, 66, 557–561. [Google Scholar]
- Alfrey, C.P.; Udden, M.M.; Huntoon, C.L.; Driscoll, T. Destruction of Newly Released Red Blood Cells in Space Flight. Med. Sci. Sports Exerc. 1996, 28, 42–44. [Google Scholar] [CrossRef]
- Shaffer, F.; Ginsberg, J.P. An Overview of Heart Rate Variability Metrics and Norms. Front. Public Health 2017, 5, 258. [Google Scholar] [CrossRef]
- Goldberger, A.L.; Bungo, M.W.; Baevsky, R.M.; Bennett, B.S.; Rigney, D.R.; Mietus, J.E.; Nikulina, G.A.; Charles, J.B. Heart Rate Dynamics during Long-Term Space Flight: Report on Mir Cosmonauts. Am. Heart J. 1994, 128, 202–204. [Google Scholar] [CrossRef]
- Xu, D.; Shoemaker, J.K.; Blaber, A.P.; Arbeille, P.; Fraser, K.; Hughson, R.L. Reduced Heart Rate Variability during Sleep in Long-Duration Spaceflight. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2013, 305, R164–R170. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Larsen, P.N.; Moesgaard, F.; Madsen, P.; Pedersen, M.; Secher, N.H. Subcutaneous Oxygen and Carbon Dioxide Tensions during Head-up Tilt-Induced Central Hypovolaemia in Humans. Scand. J. Clin. Lab. Investig. 1996, 56, 17–24. [Google Scholar] [CrossRef]
- Pocock, G.; Richards, C.D. Human Physiology: The Basis of Medicine, 2nd ed.; Oxford Core Texts; Oxford University Press: Oxford, UK, 2004; ISBN 978-0-19-858527-5. [Google Scholar]
- Fu, Q.; Shibata, S.; Hastings, J.L.; Platts, S.H.; Hamilton, D.M.; Bungo, M.W.; Stenger, M.B.; Ribeiro, C.; Adams-Huet, B.; Levine, B.D. Impact of Prolonged Spaceflight on Orthostatic Tolerance during Ambulation and Blood Pressure Profiles in Astronauts. Circulation 2019, 140, 729–738. [Google Scholar] [CrossRef] [PubMed]
- Buckey, J.C.; Lane, L.D.; Levine, B.D.; Watenpaugh, D.E.; Wright, S.J.; Moore, W.E.; Gaffney, F.A.; Blomqvist, C.G. Orthostatic Intolerance after Spaceflight. J. Appl. Physiol. 1996, 81, 7–18. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.M.C.; Feiveson, A.H.; Stein, S.; Stenger, M.B.; Platts, S.H. Orthostatic Intolerance After ISS and Space Shuttle Missions. Aerosp. Med. Hum. Perform. 2015, 86, 54–67. [Google Scholar] [CrossRef]
- Bondar, R.L. The Neurolab Spacelab Mission: Neuroscience Research in Space: Results from the STS-90, Neurolab Spacelab Mission. Arch. Neurol. 2005, 62, 1314. [Google Scholar] [CrossRef]
- Convertino, V.A.; Cooke, W.H. Vascular Functions in Humans Following Cardiovascular Adaptations to Spaceflight. Acta Astronaut. 2007, 60, 259–266. [Google Scholar] [CrossRef]
- Beck, M.; Tabury, K.; Moreels, M.; Jacquet, P.; Van Oostveldt, P.; De Vos, W.H.; Baatout, S. Simulated Microgravity Decreases Apoptosis in Fetal Fibroblasts. Int. J. Mol. Med. 2012, 30, 309–313. [Google Scholar] [CrossRef]
- Beck, M.; Moreels, M.; Quintens, R.; Abou-El-Ardat, K.; El-Saghire, H.; Tabury, K.; Michaux, A.; Janssen, A.; Neefs, M.; Van Oostveldt, P.; et al. Chronic Exposure to Simulated Space Conditions Predominantly Affects Cytoskeleton Remodeling and Oxidative Stress Response in Mouse Fetal Fibroblasts. Int. J. Mol. Med. 2014, 34, 606–615. [Google Scholar] [CrossRef]
- Xue, J.-H.; Zhang, L.-F.; Ma, J.; Xie, M.-J. Differential Regulation of L-Type Ca2+ Channels in Cerebral and Mesenteric Arteries after Simulated Microgravity in Rats and Its Intervention by Standing. Am. J. Physiol. Heart Circ. Physiol. 2007, 293, H691–H701. [Google Scholar] [CrossRef]
- Kang, H.; Fan, Y.; Sun, A.; Jia, X.; Deng, X. Simulated Microgravity Exposure Modulates the Phenotype of Cultured Vascular Smooth Muscle Cells. Cell Biochem. Biophys. 2013, 66, 121–130. [Google Scholar] [CrossRef]
- Owens, G.K.; Kumar, M.S.; Wamhoff, B.R. Molecular Regulation of Vascular Smooth Muscle Cell Differentiation in Development and Disease. Physiol. Rev. 2004, 84, 767–801. [Google Scholar] [CrossRef] [PubMed]
- Beamish, J.A.; He, P.; Kottke-Marchant, K.; Marchant, R.E. Molecular Regulation of Contractile Smooth Muscle Cell Phenotype: Implications for Vascular Tissue Engineering. Tissue Eng. Part B Rev. 2010, 16, 467–491. [Google Scholar] [CrossRef]
- Zhang, L.-F. Vascular Adaptation to Microgravity: What Have We Learned? J. Appl. Physiol. 2001, 91, 2415–2430. [Google Scholar] [CrossRef] [PubMed]
- Matchkov, V.V.; Kudryavtseva, O.; Aalkjaer, C. Intracellular Ca2+ Signalling and Phenotype of Vascular Smooth Muscle Cells: Intracellular Ca2+ and Smooth Muscle Cell Phenotype. Basic Clin. Pharmacol. Toxicol. 2012, 110, 42–48. [Google Scholar] [CrossRef] [PubMed]
- El-Rahman, R.R.A.; Harraz, O.F.; Brett, S.E.; Anfinogenova, Y.; Mufti, R.E.; Goldman, D.; Welsh, D.G. Identification of L- and T-Type Ca2+ Channels in Rat Cerebral Arteries: Role in Myogenic Tone Development. Am. J. Physiol. Heart Circ. Physiol. 2013, 304, H58–H71. [Google Scholar] [CrossRef]
- Zhang, B.; Chen, L.; Bai, Y.; Song, J.; Cheng, J.; Ma, H.; Ma, J.; Xie, M. MiR-137 and Its Target T-type CaV3.1 Channel Modulate Dedifferentiation and Proliferation of Cerebrovascular Smooth Muscle Cells in Simulated Microgravity Rats by Regulating Calcineurin/NFAT Pathway. Cell Prolif. 2020, 53, e12774. [Google Scholar] [CrossRef]
- Delp, M.D.; Colleran, P.N.; Wilkerson, M.K.; McCurdy, M.R.; Muller-Delp, J. Structural and Functional Remodeling of Skeletal Muscle Microvasculature Is Induced by Simulated Microgravity. Am. J. Physiol. Heart Circ. Physiol. 2000, 278, H1866–H1873. [Google Scholar] [CrossRef]
- Zhang, L.F.; Ma, J.; Mao, Q.W.; Yu, Z.B. Plasticity of Arterial Vasculature during Simulated Weightlessness and Its Possible Role in the Genesis of Postflight Orthostatic Intolerance. J. Gravit. Physiol. 1997, 4, P97–P100. [Google Scholar] [PubMed]
- Kapitonova, M.Y.; Muid, S.; Froemming, G.R.A.; Yusoff, W.N.W.; Othman, S.; Ali, A.M.; Nawawi, H.M. Real Space Flight Travel Is Associated with Ultrastructural Changes, Cytoskeletal Disruption and Premature Senescence of HUVEC. Malays. J. Pathol. 2012, 34, 103–113. [Google Scholar]
- Kapitonova, M.Y.; Kuznetsov, S.L.; Froemming, G.R.A.; Muid, S.; Nor-Ashikin, M.N.K.; Otman, S.; Shahir, A.R.M.; Nawawi, H. Effects of Space Mission Factors on the Morphology and Function of Endothelial Cells. Bull. Exp. Biol. Med. 2013, 154, 796–801. [Google Scholar] [CrossRef]
- Versari, S.; Longinotti, G.; Barenghi, L.; Maier, J.A.M.; Bradamante, S. The Challenging Environment on Board the International Space Station Affects Endothelial Cell Function by Triggering Oxidative Stress through Thioredoxin Interacting Protein Overexpression: The ESA-SPHINX Experiment. FASEB J. 2013, 27, 4466–4475. [Google Scholar] [CrossRef] [PubMed]
- Maier, J.A.M.; Cialdai, F.; Monici, M.; Morbidelli, L. The Impact of Microgravity and Hypergravity on Endothelial Cells. BioMed Res. Int. 2015, 2015, 434803. [Google Scholar] [CrossRef] [PubMed]
- Sato, M.; Ohashi, T. Biorheological Views of Endothelial Cell Responses to Mechanical Stimuli. Biorheology 2005, 42, 421–441. [Google Scholar]
- Pietsch, J.; Gass, S.; Nebuloni, S.; Echegoyen, D.; Riwaldt, S.; Baake, C.; Bauer, J.; Corydon, T.; Egli, M.; Infanger, M.; et al. Three-Dimensional Growth of Human Endothelial Cells in an Automated Cell Culture Experiment Container during the SpaceX CRS-8 ISS Space Mission—The SPHEROIDS Project—ClinicalKey. Available online: https://www-clinicalkey-com.ez.statsbiblioteket.dk:12048/#!/content/playContent/1-s2.0-S014296121730073X?returnurl=null&referrer=null (accessed on 26 November 2021).
- Krüger, M.; Pietsch, J.; Bauer, J.; Kopp, S.; Carvalho, D.; Baatout, S.; Moreels, M.; Melnik, D.; Wehland, M.; Egli, M.; et al. Growth of Endothelial Cells in Space and in Simulated Microgravity—A Comparison on the Secretory Level. Cell. Physiol. Biochem. 2019, 52, 1039–1060. [Google Scholar] [CrossRef] [PubMed]
- Dittrich, A.; Grimm, D.; Sahana, J.; Bauer, J.; Krüger, M.; Infanger, M.; Magnusson, N.E. Key Proteins Involved in Spheroid Formation and Angiogenesis in Endothelial Cells after Long-Term Exposure to Simulated Microgravity. CPB 2018, 45, 429–445. [Google Scholar] [CrossRef]
- Eilenberg, W.; Stojkovic, S.; Piechota-Polanczyk, A.; Kaun, C.; Rauscher, S.; Gröger, M.; Klinger, M.; Wojta, J.; Neumayer, C.; Huk, I.; et al. Neutrophil Gelatinase-Associated Lipocalin (NGAL) Is Associated with Symptomatic Carotid Atherosclerosis and Drives Pro-Inflammatory State In Vitro. Eur. J. Vasc. Endovasc. Surg. 2016, 51, 623–631. [Google Scholar] [CrossRef]
- Suffee, N.; Hlawaty, H.; Meddahi-Pelle, A.; Maillard, L.; Louedec, L.; Haddad, O.; Martin, L.; Laguillier, C.; Richard, B.; Oudar, O.; et al. RANTES/CCL5-Induced pro-Angiogenic Effects Depend on CCR1, CCR5 and Glycosaminoglycans. Angiogenesis 2012, 15, 727–744. [Google Scholar] [CrossRef]
- Morbidelli, L.; Monici, M.; Marziliano, N.; Cogoli, A.; Fusi, F.; Waltenberger, J.; Ziche, M. Simulated Hypogravity Impairs the Angiogenic Response of Endothelium by Up-Regulating Apoptotic Signals. Biochem. Biophys. Res. Commun. 2005, 334, 491–499. [Google Scholar] [CrossRef]
- Wen, Q.; Janmey, P.A. Polymer Physics of the Cytoskeleton. Curr. Opin. Solid State Mater. Sci. 2011, 15, 177–182. [Google Scholar] [CrossRef]
- Carlsson, S.I.M.; Bertilaccio, M.T.S.; Ballabio, E.; Maier, J.A.M. Endothelial Stress by Gravitational Unloading: Effects on Cell Growth and Cytoskeletal Organization. Biochim. Biophys. Acta Mol. Cell Res. 2003, 1642, 173–179. [Google Scholar] [CrossRef] [PubMed]
- Crawford-Young, S.J. Effects of Microgravity on Cell Cytoskeleton and Embryogenesis. Int. J. Dev. Biol. 2006, 50, 183–191. [Google Scholar] [CrossRef]
- Grenon, S.M.; Jeanne, M.; Aguado-Zuniga, J.; Conte, M.S.; Hughes-Fulford, M. Effects of Gravitational Mechanical Unloading in Endothelial Cells: Association between Caveolins, Inflammation and Adhesion Molecules. Sci. Rep. 2013, 3, 1494. [Google Scholar] [CrossRef]
- Janmaleki, M.; Pachenari, M.; Seyedpour, S.M.; Shahghadami, R.; Sanati-Nezhad, A. Impact of Simulated Microgravity on Cytoskeleton and Viscoelastic Properties of Endothelial Cell. Sci. Rep. 2016, 6, 32418. [Google Scholar] [CrossRef] [PubMed]
- Infanger, M.; Kossmehl, P.; Shakibaei, M.; Baatout, S.; Witzing, A.; Grosse, J.; Bauer, J.; Cogoli, A.; Faramarzi, S.; Derradji, H.; et al. Induction of Three-Dimensional Assembly and Increase in Apoptosis of Human Endothelial Cells by Simulated Microgravity: Impact of Vascular Endothelial Growth Factor. Apoptosis 2006, 11, 749–764. [Google Scholar] [CrossRef]
- Grimm, D.; Infanger, M.; Westphal, K.; Ulbrich, C.; Pietsch, J.; Kossmehl, P.; Vadrucci, S.; Baatout, S.; Flick, B.; Paul, M.; et al. A Delayed Type of Three-Dimensional Growth of Human Endothelial Cells under Simulated Weightlessness. Tissue Eng. Part A 2009, 15, 2267–2275. [Google Scholar] [CrossRef]
- Tuday, E.C.; Nyhan, D.; Shoukas, A.A.; Berkowitz, D.E. Simulated Microgravity-Induced Aortic Remodeling. J. Appl. Physiol. 2009, 106, 2002–2008. [Google Scholar] [CrossRef]
- Arteaga-Solis, E.; Gayraud, B.; Ramirez, F. Elastic and Collagenous Networks in Vascular Diseases. Cell Struct. Funct. 2000, 25, 69–72. [Google Scholar] [CrossRef]
- Liu, C.; Zhong, G.; Zhou, Y.; Yang, Y.; Tan, Y.; Li, Y.; Gao, X.; Sun, W.; Li, J.; Jin, X.; et al. Alteration of Calcium Signalling in Cardiomyocyte Induced by Simulated Microgravity and Hypergravity. Cell Prolif. 2020, 53, e12783. [Google Scholar] [CrossRef] [PubMed]
- Xiong, J.; Li, Y.; Nie, J. Effects of Simulated Microgravity on Nitric Oxide Level in Cardiac Myocytes and Its Mechanism. Sci. China C Life Sci. 2003, 46, 302–309. [Google Scholar] [CrossRef] [PubMed]
- Baio, J.; Martinez, A.F.; Silva, I.; Hoehn, C.V.; Countryman, S.; Bailey, L.; Hasaniya, N.; Pecaut, M.J.; Kearns-Jonker, M. Cardiovascular Progenitor Cells Cultured Aboard the International Space Station Exhibit Altered Developmental and Functional Properties. NPJ Microgravity 2018, 4, 13. [Google Scholar] [CrossRef]
- Jha, R.; Wu, Q.; Singh, M.; Preininger, M.K.; Han, P.; Ding, G.; Cho, H.C.; Jo, H.; Maher, K.O.; Wagner, M.B.; et al. Simulated Microgravity and 3D Culture Enhance Induction, Viability, Proliferation and Differentiation of Cardiac Progenitors from Human Pluripotent Stem Cells. Sci. Rep. 2016, 6, 30956. [Google Scholar] [CrossRef] [PubMed]
- Tarantino, U.; Cariati, I.; Marini, M.; D’Arcangelo, G.; Tancredi, V.; Primavera, M.; Lundusi, R.; Gasbarra, E.; Scimeca, M. Effects of Simulated Microgravity on Muscle Stem Cells Activity. Cell Physiol. Biochem. 2020, 54, 736–747. [Google Scholar] [CrossRef] [PubMed]
- Xue, L.; Li, Y.; Chen, J. Duration of Simulated Microgravity Affects the Differentiation of Mesenchymal Stem Cells. Mol. Med. Rep. 2017, 15, 3011–3018. [Google Scholar] [CrossRef]
- Kong, L.; Wang, Y.; Wang, H.; Pan, Q.; Zuo, R.; Bai, S.; Zhang, X.; Lee, W.Y.; Kang, Q.; Li, G. Conditioned Media from Endothelial Progenitor Cells Cultured in Simulated Microgravity Promote Angiogenesis and Bone Fracture Healing. Stem Cell Res. 2021, 12, 47. [Google Scholar] [CrossRef]
- Ramaswamy, V.; Dirr, E.W.; Allen, J.B. The Effect of Simulated Microgravity on Differentiation of Porcine Blood-Derived Vascular Stem Cells. Stem Cells Dev. 2016, 25, 329–336. [Google Scholar] [CrossRef] [PubMed]
- Hagiwara, H.; Higashibata, A.; Ogawa, S.; Kanazawa, S.; Mizuno, H.; Tanaka, R. Effectiveness of Endothelial Progenitor Cell Culture under Microgravity for Improved Angiogenic Potential. Sci. Rep. 2018, 8, 14239. [Google Scholar] [CrossRef]

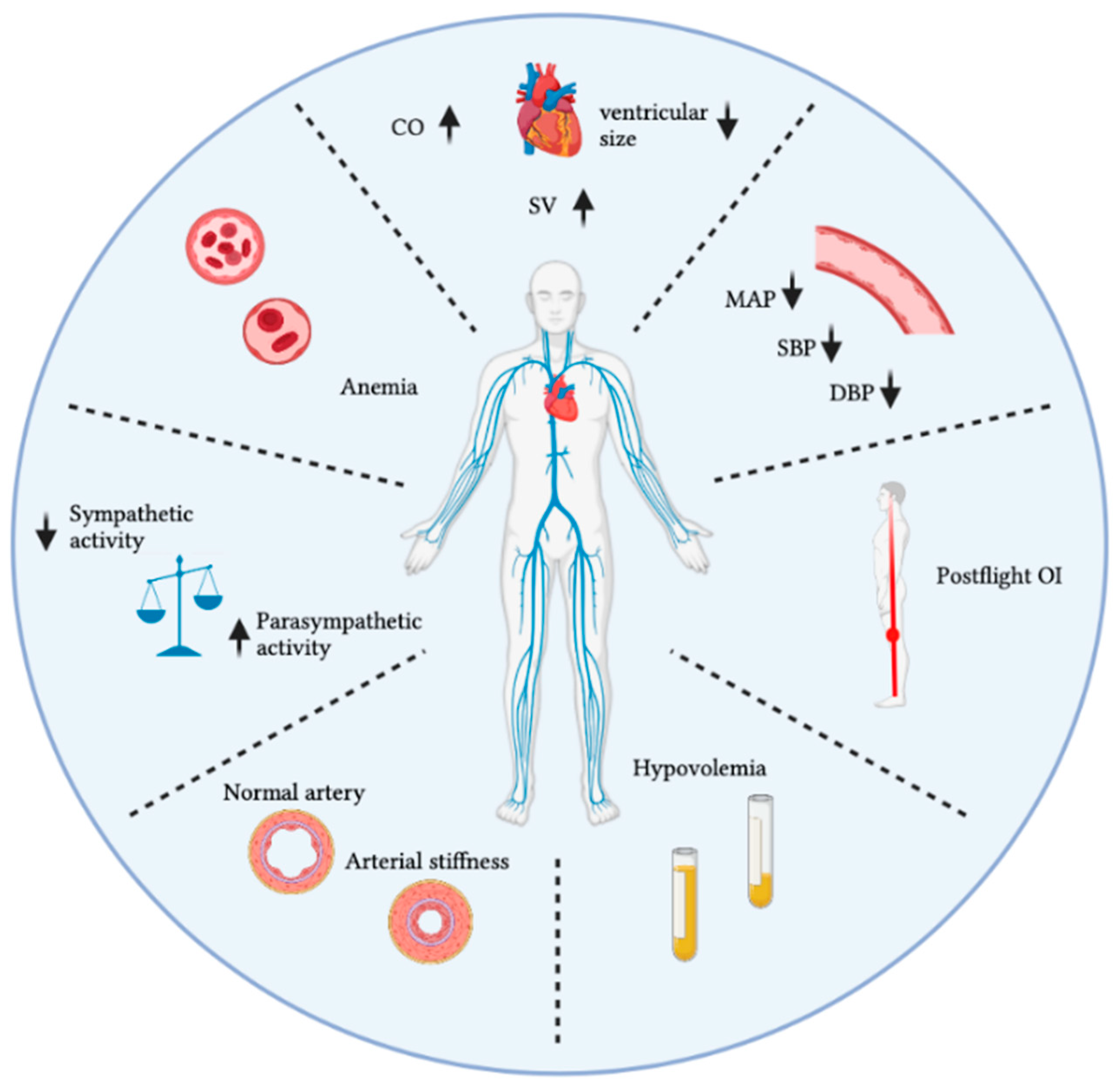
| Ref | Variables | Initial Reaction | ST Response | LT Response |
|---|---|---|---|---|
| [2] | Blood volume | ↑ | ↓ 10–15% | ↓ 10–15% |
| [2,16] | Haematocrit | ↑ | ↓ 10–15% | Unchanged or ↓ |
| [17,18,19,20] | CO | ↑ | ↑18–24% | ↑ 41% |
| [19,20] | SV | ↑ | ↑ 46% | ↑ 35% |
| [21,22,23] | Ventricular size | ↑ 20% | ↓ 10% | ↓ 10% |
| [24,25,26] | CVP | ↓ | ↓ | ↓ |
| [20,27,28,29] | MAP | Unchanged | Unchanged | ↓ 10 mmHg |
| [20,27,28,29] | SBP | Unchanged | Unchanged | ↓ 8 mmHg |
| [20,27,28,29] | DBP | Unchanged | ↓ 5 mmHg | ↓ 9 mmHg |
| [20,29] | SVR | Unchanged | Unchanged | ↓ 39% |
| [24,30] | HR | Unchanged | Unchanged or↓ | Unchanged or ↓ |
| [31,32] | cIMT | ↑ 10–12% (6 M) | ||
| ↑ 20% (1 Y) | ||||
| [31,32] | Femoral IMT | ↑ 10%–15% | ||
| [33,34,35] | Arterial stiffness | ↑ 17%–30% |
| Search Terms | Number of Articles |
|---|---|
| Microgravity AND Cardiovascular disease | 479 |
| Microgravity AND Radiation | 1103 |
| Radiation AND Cardiovascular disease | 31,676 |
| Radiation AND Microgravity AND Cardiovascular disease | 25 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Baran, R.; Marchal, S.; Garcia Campos, S.; Rehnberg, E.; Tabury, K.; Baselet, B.; Wehland, M.; Grimm, D.; Baatout, S. The Cardiovascular System in Space: Focus on In Vivo and In Vitro Studies. Biomedicines 2022, 10, 59. https://doi.org/10.3390/biomedicines10010059
Baran R, Marchal S, Garcia Campos S, Rehnberg E, Tabury K, Baselet B, Wehland M, Grimm D, Baatout S. The Cardiovascular System in Space: Focus on In Vivo and In Vitro Studies. Biomedicines. 2022; 10(1):59. https://doi.org/10.3390/biomedicines10010059
Chicago/Turabian StyleBaran, Ronni, Shannon Marchal, Sebastian Garcia Campos, Emil Rehnberg, Kevin Tabury, Bjorn Baselet, Markus Wehland, Daniela Grimm, and Sarah Baatout. 2022. "The Cardiovascular System in Space: Focus on In Vivo and In Vitro Studies" Biomedicines 10, no. 1: 59. https://doi.org/10.3390/biomedicines10010059
APA StyleBaran, R., Marchal, S., Garcia Campos, S., Rehnberg, E., Tabury, K., Baselet, B., Wehland, M., Grimm, D., & Baatout, S. (2022). The Cardiovascular System in Space: Focus on In Vivo and In Vitro Studies. Biomedicines, 10(1), 59. https://doi.org/10.3390/biomedicines10010059







