Influences of Lavender Essential Oil Inhalation on Stress Responses during Short-Duration Sleep Cycles: A Pilot Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Subjects
2.2. Aromatherapy
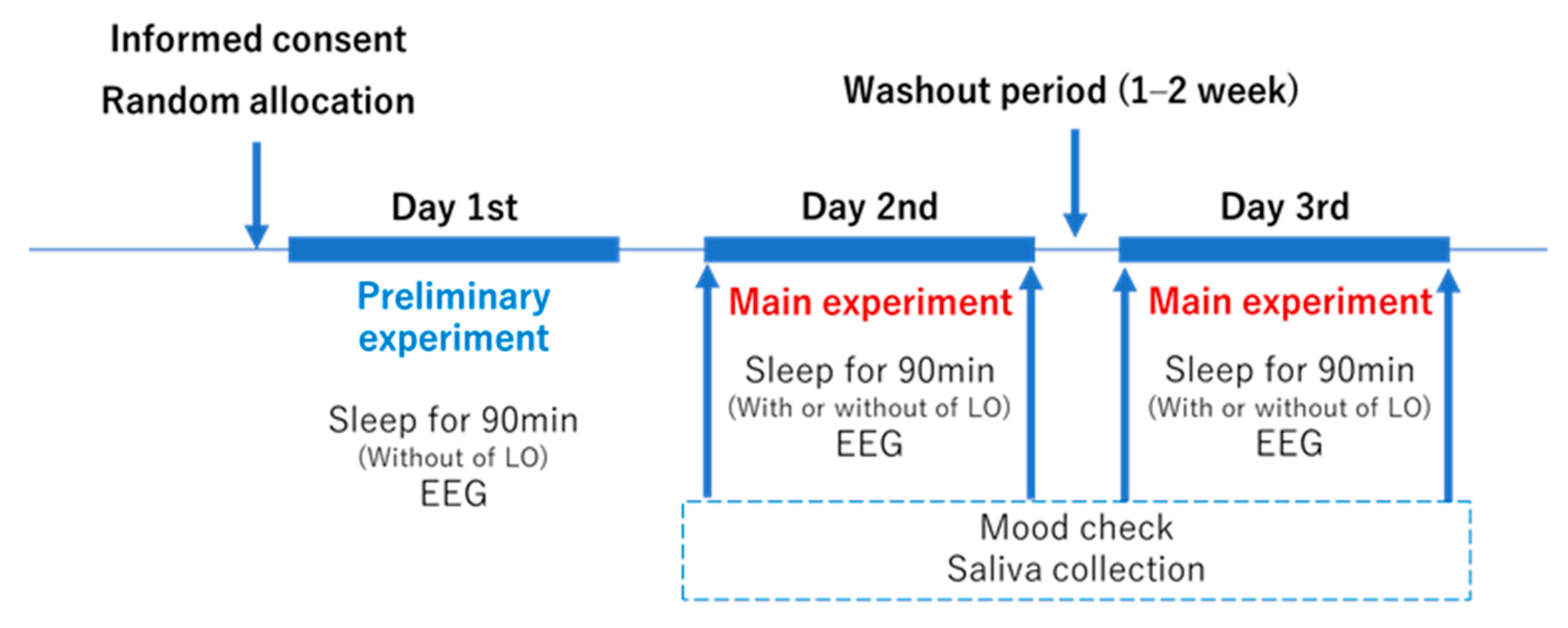
2.3. Study Design
2.4. Condition of Sleep
2.5. Evaluation of Stress
2.5.1. Objective Index: Measurements of Salivary Stress Markers
2.5.2. Subjective Index: The UWIST Mood Adjective Checklist
2.6. Statistical Analysis
3. Results
3.1. Salivary Stress Markers
3.2. UWIST Mood Adjective Checklists
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Alanko, K. Aromatherapists. In Handbook of Occupational Dermatology; Springer: New York, NK, USA, 2000; pp. 811–813. [Google Scholar]
- Ozkaraman, A.; Dügüm, Ö.; Özen Yılmaz, H.; Usta Yesilbalkan, Ö. Aromatherapy: The effect of lavender on anxiety and sleep quality in patients treated with chemotherapy. Clin. J. Oncol. Nurs. 2018, 22, 203–210. [Google Scholar] [CrossRef]
- Chen, M.C.; Fang, S.H.; Fang, L. The effects of aromatherapy in relieving symptoms related to job stress among nurses. J. Nurs. Pract. 2015, 21, 87–93. [Google Scholar] [CrossRef] [PubMed]
- Schneider, R.; Singer, N.; Singer, T. Medical aromatherapy revisited—Basic mechanisms, critique, and a new development. Hum. Psychopharmacol. 2019, 34, e2683. [Google Scholar] [CrossRef] [PubMed]
- Donatello, N.N.; Emer, A.A.; Salm, D.C.; Ludtke, D.D.; Bordignon, S.A.S.R.; Ferreira, J.K.; Salgado, A.S.I.; Venzke, D.; Bretanha, L.C.; Micke, G.A. Lavandula angustifolia essential oil inhalation reduces mechanical hyperalgesia in a model of inflammatory and neuropathic pain: The involvement of opioid and cannabinoid receptors. J. Neuroimmunol. 2020, 340, 577145. [Google Scholar] [CrossRef] [PubMed]
- Jianu, C.; Pop, G.; TGruia, A.; Horhat, F.G. Chemical composition and antimicrobial activity of essential oils of lavender (Lavandula angustifolia) and lavandin (Lavandula × intermedia) grown in Western Romania. Int. J. Agric. Biol. 2013, 15, 772–776. [Google Scholar]
- Hajhashemi, V.; Ghannadi, A.; Sharif, B. Anti-inflammatory and analgesic properties of the leaf extracts and essential oil of Lavandula angustifolia Mill. J. Ethnopharmacol. 2003, 89, 67–71. [Google Scholar] [CrossRef]
- Yamaguchi, M.; Tahara, Y.; Kosaka, S. Influence of concentration of fragrances on salivary alpha-amylase. Int. J. Cosmet. Sci. 2009, 31, 391–395. [Google Scholar] [CrossRef]
- Yousefi, M.; Shabunin, S.V.; Vatnikov, Y.A.; Kulikov, E.V.; Adineh, H.; Hamidi, M.K.; Hoseini, S.M. Effects of lavender (Lavandula angustifolia) extract inclusion in diet on growth performance, innate immunity, immune-related gene expression, and stress response of common carp, Cyprinus carpio. Aquaculture 2020, 515, 734588. [Google Scholar] [CrossRef]
- Bradley, B.F.; Starkey, N.J.; Brown, S.L.; Lea, R.W. Anxiolytic effects of Lavandula angustifolia odour on the Mongolian gerbil elevated plus maze. J. Ethnopharmacol. 2007, 111, 517–525. [Google Scholar] [CrossRef]
- Tomi, K.; Kitao, M.; Murakami, H.; Matsumura, Y.; Hayashi, T. Classification of lavender essential oils: Sedative effects of Lavandula oils. J. Essent. Oil Res. 2018, 30, 56–68. [Google Scholar] [CrossRef]
- Field, T.; Field, T.; Cullen, C.; Largie, S.; Diego, M.; Schanberg, S.; Kuhn, C. Lavender bath oil reduces stress and crying and enhances sleep in very young infants. Early Hum. Dev. 2008, 84, 399–401. [Google Scholar] [CrossRef]
- Fismer, K.L.; Pilkington, K. Lavender and sleep: A systematic review of the evidence. Eur. J. Integr. Med. 2012, 4, e436–e447. [Google Scholar] [CrossRef]
- Goel, N.; Kim, H.; Lao, R.P. An olfactory stimulus modifies nighttime sleep in young men and women. Chronobiol. Int. 2005, 22, 889–904. [Google Scholar] [CrossRef]
- Arzi, A.; Sela, L.; Green, A.; Givaty, G.; Dagan, Y.; Sobel, N. The influence of odorants on respiratory patterns in sleep. Chem. Senses 2010, 35, 31–40. [Google Scholar] [CrossRef] [PubMed]
- Hublin, C.; Kaprio, J.; Partinen, M.; Koskenvuo, M. Insufficient sleep—A population-based study in adults. Sleep 2001, 24, 392–400. [Google Scholar] [CrossRef]
- Basner, M.; Fomberstein, K.M.; Razavi, F.M.; Banks, S.; William, J.H.; Rosa, R.R.; Dinges, D.F. American time use survey: Sleep time and its relationship to waking activities. Sleep 2007, 30, 1085–1095. [Google Scholar] [CrossRef] [PubMed]
- McEwen, B.S.; Karatsoreos, I.N. Sleep Deprivation and Circadian Disruption: Stress, Allostasis, and Allostatic Load. Sleep Med. Clin. 2015, 10, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Van Dongen, H.P.; Maislin, G.; Mullington, J.M.; Dinges, D.F. The cumulative cost of additional wakefulness: Dose–response effects on neurobehavioral functions and sleep physiology from chronic sleep restriction and total sleep deprivation. Sleep 2003, 26, 117–126. [Google Scholar] [CrossRef] [PubMed]
- Milner, C.E.; Cote, K.A. Benefits of napping in healthy adults: Impact of nap length, time of day, age, and experience with napping. J. Sleep Res. 2009, 18, 272–281. [Google Scholar] [CrossRef] [PubMed]
- Vgontzas, A.N.; Pejovic, S.; Zoumakis, E.; Lin, H.M.; Bixler, E.O.; Basta, M.; Fang, J.; Sarrigiannidis, A.; Chrousos, G.P. Daytime napping after a night of sleep loss decreases sleepiness, improves performance, and causes beneficial changes in cortisol and interleukin-6 secretion. Am. J. Physiol. Endocrinol. Metab. 2007, 292, E253–E261. [Google Scholar] [CrossRef] [PubMed]
- Hayashi, M.; Ito, S.; Hori, T. The effects of a 20-min nap at noon on sleepiness, performance and EEG activity. Int. J. Psychophysiol. 1999, 32, 173–180. [Google Scholar] [CrossRef]
- Dhand, R.; Sohal, H. Good sleep, bad sleep! The role of daytime naps in healthy adults. Curr. Opin. Pulm. Med. 2006, 12, 379–382. [Google Scholar] [CrossRef] [PubMed]
- Mednick, S.; Nakayama, K.; Stickgold, R. Sleep-dependent learning: A nap is as good as a night. Nat. Neurosci. 2003, 6, 697–698. [Google Scholar] [CrossRef]
- Yamada, T.; Hara, K.; Shojima, N.; Yamauchi, T.; Kadowaki, T. Daytime napping and the risk of cardiovascular disease and all-cause mortality: A prospective study and dose-response meta-analysis. Sleep 2015, 38, 1945–1953. [Google Scholar] [CrossRef] [PubMed]
- Soyka, M. Treatment of benzodiazepine dependence. N. Engl. J. Med. 2017, 376, 1147–1157. [Google Scholar] [CrossRef]
- Vickers, A. Why aromatherapy works (even if it doesn’t) and why we need less research. Br. J. Gen. Pract. 2000, 50, 444–445. [Google Scholar] [PubMed]
- Zhan, H.; Zuo, T.; Tao, R.; Chang, C. Robust tunicate cellulose nanocrystal/palygorskite nanorod membranes for multifunctional oil/water emulsion separation. ACS Sustain. Chem. Eng. 2018, 6, 10833–10840. [Google Scholar] [CrossRef]
- Chien, L.W.; Cheng, S.L.; Liu, C.F. The effect of lavender aromatherapy on autonomic nervous system in midlife women with insomnia. Evid. Based Complement. Alternat. Med. 2012, 2012, 740813. [Google Scholar] [CrossRef]
- Matsumoto, T.; Asakura, H.; Hayashi, T. Does lavender aromatherapy alleviate premenstrual emotional symptoms?: A randomized crossover trial. BioPsychoSoc. Med. 2013, 7, 12. [Google Scholar] [CrossRef]
- Donelli, D.; Antonelli, M.; Bellinazzi, C.; Gensini, G.F.; Firenzuoli, F. Effects of lavender on anxiety: A systematic review and meta-analysis. Phytomedicine 2019, 65, 153099. [Google Scholar] [CrossRef]
- Gu, S.; Wang, F.; Patel, N.P.; Bourgeois, J.A.; Huang, J.H. A model for basic emotions using observations of behavior in drosophila. Front Psychol. 2019, 10, 781. [Google Scholar] [CrossRef] [PubMed]
- Shirasawa, S.; Ishida, T.; Hakoda, Y.; Haraguchi, M. The effects of energetic arousal on memory search. Jpn. J. Psychon. Sci. 1999, 17, 93–99, (In Japanese, abstract in English). [Google Scholar]
- Matthews, G.; Jones, D.M.; Chamberlain, A.G. Refining the measurement of mood: The UWIST mood adjective checklist. Br. J. Psychol. 1990, 81, 17–42. [Google Scholar] [CrossRef]
- Głębocka, A. Stress and dehumanizing behaviors of medical staff toward patients. Adv. Exp. Med. Biol. 2019, 1133, 97–104. [Google Scholar]
- Pokorski, M.; Suchorzynska, A. psychobehavioral effects of meditation. Adv. Exp. Med. Biol. 2018, 1023, 85–91. [Google Scholar] [PubMed]
- Biernacka, M.; Jakubowska-Winecka, A.; Biernacki, M. The role of emotional control in the regulation of mood in parents of children with mucopolysaccharidosis. Stress Health 2014, 30, 253–258. [Google Scholar] [CrossRef] [PubMed]
- Grajfoner, D.; Harte, E.; Potter, L.M.; McGuigan, N. The effect of dog-assisted Intervention on student well-being, mood, and anxiety. Int. J. Environ. Res. Public Health 2017, 14, 483. [Google Scholar] [CrossRef]
- Stolarski, M.; Matthews, G. Time perspectives predict mood states and satisfaction with life over and above personality. Curr. Psychol. 2016, 35, 516–526. [Google Scholar] [CrossRef]
- O’Connor, P.J. Mental energy: Assessing the mood dimension. Nutr. Rev. 2006, 64, S7–S9. [Google Scholar] [CrossRef]
- Smith, S.M.; Vale, W.W. The role of the hypothalamic-pituitary-adrenal axis in neuroendocrine responses to stress. Dialogues Clin. Neurosci. 2006, 8, 383–395. [Google Scholar]
- Gerra, G.; Zaimovic, A.; Mascetti, G.G.; Gardini, S.; Zambelli, U.; Timpano, M.; Raggi, M.A.; Brambilla, F. Neuroendocrine responses to experimentally induced psychological stress in healthy humans. Psychoneuroendocrinology 2001, 26, 91–107. [Google Scholar] [CrossRef]
- Den, R.; Toda, M.; Nagasawa, S.; Kitamura, K.; Morimoto, K. Circadian rhythm of human salivary chromogranin A. Biomed. Res. 2007, 28, 57–60. [Google Scholar] [CrossRef] [PubMed]
- Edwards, S.; Clow, A.; Evans, P.; Hucklebridge, F. Exploration of the awakening cortisol response in relation to diurnal cortisol secretory activity. Life Sci. 2001, 68, 2093–2103. [Google Scholar] [CrossRef]
- Speirs, R.L.; Herring, J.; Cooper, W.D.; Hardy, C.C.; Hind, C.R. The influence of sympathetic activity and isoprenaline on the secretion of amylase from the human parotid gland. Arch. Oral Biol. 1974, 19, 747–752. [Google Scholar] [CrossRef]
- Nater, U.M.; Rohleder, N. Salivary alpha-amylase as a non-invasive biomarker for the sympathetic nervous system: Current state of research. Psychoneuroendocrinology 2009, 34, 486–496. [Google Scholar] [CrossRef]
- Blaschko, H.; Comline, R.S.; Schneider, F.H.; Silver, M.; Smith, A.D. Secretion of a chromaffin granule protein, chromogranin, from the adrenal gland after splanchnic stimulation. Nature 1967, 215, 58–59. [Google Scholar] [CrossRef] [PubMed]
- Kawada, S.; Fukusaki, C.; Ohtani, M.; Kobayashi, K. Effects of hyperoxic inhalation on psychological stress-induced salivary biomarkers. Biomed. Res. 2009, 30, 245–249. [Google Scholar] [CrossRef]
- Ugawa, Y.; Nishigawa, G.; Maruo, Y.; Suwaki, M.; Minagi, S. Salivary stress biomarker levels during speech in patients with maxillectomy defect. Head Neck 2011, 33, 620–626. [Google Scholar] [CrossRef]
- Engleman, H.M.; Martin, S.E.; Deary, I.J.; Douglas, N.J. Effect of CPAP therapy on daytime function in patients with mild sleep apnoea/hypopnoea syndrome. Thorax 1997, 52, 114–119. [Google Scholar] [CrossRef]
- Kingshott, R.N.; Cosway, R.J.; Deary, I.J.; Douglas, N.J. The effect of sleep fragmentation on cognitive processing using computerized topographic brain mapping. J. Sleep Res. 2000, 9, 353–357. [Google Scholar] [CrossRef][Green Version]
- Hossain, S.J.; Aoshima, H.; Koda, H.; Kiso, Y. Potentiation of the ionotropic GABA receptor response by whiskey fragrance. J. Agric. Food Chem. 2002, 50, 6828–6834. [Google Scholar] [CrossRef]
- Elisabetsky, E.; Marschner, J.; Souza, D.O. Effects of linalool on glutamatergic system in the rat cerebral cortex. Neurochem. Res. 1995, 20, 461–465. [Google Scholar] [CrossRef] [PubMed]
- Elisabetsky, E.; Brum, L.F.; Souza, D.O. Anticonvulsant properties of linalool in glutamate-related seizure models. Phytomedicine 1999, 6, 107–113. [Google Scholar] [CrossRef]
- Huang, L.; Abuhamdah, S.; Howes, M.J.R.; Dixon, C.L.; Elliot, M.S.; Ballard, C.; Holmes, C.; Burns, A.; Perry, E.K.; Francis, P.T. Pharmacological profile of essential oils derived from Lavandula angustifolia and Melissa officinalis with anti-agitation properties: Focus on ligand-gated channels. J. Pharm. Pharmacol. 2008, 60, 1515–1522. [Google Scholar] [CrossRef] [PubMed]
- Shi, Y.; Li, Y.; Yin, J.; Hu, H.; Xue, M.; Li, X.; Cheng, W.; Wang, Y.; Li, X.; Wang, Y.; et al. A novel sympathetic neuronal GABAergic signalling system regulates NE release to prevent ventricular arrhythmias after acute myocardial infarction. Acta Physiol. 2019, 227, e13315. [Google Scholar] [CrossRef] [PubMed]
- Angelone, T.; Mazza, R.; Cerra, M.C. Chromogranin-A: A multifaceted cardiovascular role in health and disease. Curr. Med. Chem. 2012, 19, 4042–4050. [Google Scholar] [CrossRef] [PubMed]
- Trinder, J.; Kleiman, J.; Carrington, M.; Smith, S.; Breen, S.; Tan, N.; Kim, Y. Autonomic activity during human sleep as a function of time and sleep stage. J. Sleep Res. 2001, 10, 253–264. [Google Scholar] [CrossRef] [PubMed]
- Tassi, P.; Muzet, A. Sleep inertia. Sleep Med. Rev. 2000, 4, 341–353. [Google Scholar] [CrossRef] [PubMed]

| (n = 9) | Before | After | p-Value | |
|---|---|---|---|---|
| Saliva cortisol (nmol/mg protein) | Control | 170.33 (31.89, 252.03) | 34.55 (17.04, 55.06) | 0.010 ** |
| LEO | 70.33 (68.41, 112.52) | 21.72 (19.54, 27.03) | 0.000 ** | |
| Saliva α-amylase (U/mg protein) | Control | 119.59 (82.56, 152.46) | 80.76 (76.87, 106.69) | 0.031 * |
| LEO | 104.64 (82.99, 126.49) | 63.79 (48.55, 80.51) | 0.001 ** | |
| Saliva CgA (pmol/mg protein) | Control | 4.39 (3.07, 8.07) | 9.61 (8.63, 14.60) | 0.143 |
| LEO | 8.32 (5.46, 10.06) | 12.95 (6.91, 17.39) | 0.230 | |
| (n = 9) | Before | After | Rate of Change(After/Before) | Before vs. After p-Value | Control vs. LEO p-Value | |
|---|---|---|---|---|---|---|
| Bright | Control | 2 (1, 3) | 1 (1, 3) | 1.00 (1.00, 1.00) | 1.000 | 0.750 |
| LEO | 2 (1, 2) | 2 (1, 2) | 1.00 (1.00, 1.00) | 1.000 | ||
| Vigorous | Control | 2 (1, 3) | 1 (1, 2) | 1.00 (0.50, 1.00) | 0.375 | 0.375 |
| LEO | 2 (1, 2) | 2 (1, 2) | 1.00 (1.00, 1.00) | 1.000 | ||
| Sleepy | Control | 2 (2, 3) | 3 (2, 4) | 1.00 (1.00, 1.50) | 0.375 | 0.375 |
| LEO | 2 (2, 3) | 2 (2, 4) | 1.00 (1.00, 1.33) | 1.000 | ||
| Energetic | Control | 2 (2, 3) | 1 (1, 3) | 1.00 (0.50, 1.00) | 0.500 | 0.250 |
| LEO | 2 (1, 3) | 2 (1, 3) | 1.00 (1.00, 1.00) | 1.000 | ||
| Idle | Control | 4 (3, 4) | 4 (3, 4) | 1.00 (1.00, 1.00) | 1.000 | 1.000 |
| LEO | 4 (3, 4) | 3 (3, 4) | 1.00 (1.00, 1.00) | 0.500 | ||
| Industrious | Control | 2 (1, 3) | 1 (1, 2) | 1.00 (1.00, 1.00) | 0.500 | 0.375 |
| LEO | 2 (1, 3) | 2 (1, 2) | 1.00 (1.00, 1.00) | 1.000 | ||
| Unenterprising | Control | 4 (4, 4) | 4 (3, 4) | 1.00 (1.00, 1.00) | 1.000 | 1.000 |
| LEO | 4 (4, 4) | 4 (3, 4) | 1.00 (1.00, 1.00) | 1.000 | ||
| Passive | Control | 3 (3, 4) | 4 (3, 4) | 1.00 (1.00, 1.00) | 1.000 | 0.500 |
| LEO | 4 (2, 4) | 4 (3, 4) | 1.00 (1.00, 1.33) | 0.750 | ||
| Dull | Control | 2 (1, 3) | 3 (2, 4) | 1.00 (1.00, 1.33) | 0.125 | 0.500 |
| LEO | 3 (2, 3) | 3 (2, 4) | 1.00 (1.00, 1.50) | 0.375 | ||
| Active | Control | 2 (1, 3) | 2 (1, 4) | 1.00 (1.00, 1.33) | 0.500 | 0.500 |
| LEO | 2 (1, 3) | 2 (1, 3) | 1.00 (1.00, 1.00) | 1.000 | ||
| Energy arousal | Control | 25 (23, 28) | 26 (25, 26) | 1.00 (1.00, 1.17) | 0.438 | 0.742 |
| LEO | 25 (24, 29) | 25 (25, 27) | 1.00 (0.93, 1.07) | 0.984 | ||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yogi, W.; Tsukada, M.; Sato, Y.; Izuno, T.; Inoue, T.; Tsunokawa, Y.; Okumo, T.; Hisamitsu, T.; Sunagawa, M. Influences of Lavender Essential Oil Inhalation on Stress Responses during Short-Duration Sleep Cycles: A Pilot Study. Healthcare 2021, 9, 909. https://doi.org/10.3390/healthcare9070909
Yogi W, Tsukada M, Sato Y, Izuno T, Inoue T, Tsunokawa Y, Okumo T, Hisamitsu T, Sunagawa M. Influences of Lavender Essential Oil Inhalation on Stress Responses during Short-Duration Sleep Cycles: A Pilot Study. Healthcare. 2021; 9(7):909. https://doi.org/10.3390/healthcare9070909
Chicago/Turabian StyleYogi, Wakako, Mana Tsukada, Yosuke Sato, Takuji Izuno, Tatsuki Inoue, Yoshiki Tsunokawa, Takayuki Okumo, Tadashi Hisamitsu, and Masataka Sunagawa. 2021. "Influences of Lavender Essential Oil Inhalation on Stress Responses during Short-Duration Sleep Cycles: A Pilot Study" Healthcare 9, no. 7: 909. https://doi.org/10.3390/healthcare9070909
APA StyleYogi, W., Tsukada, M., Sato, Y., Izuno, T., Inoue, T., Tsunokawa, Y., Okumo, T., Hisamitsu, T., & Sunagawa, M. (2021). Influences of Lavender Essential Oil Inhalation on Stress Responses during Short-Duration Sleep Cycles: A Pilot Study. Healthcare, 9(7), 909. https://doi.org/10.3390/healthcare9070909







