Efficacy of Arabic Coffee and Black Tea in Reducing Halitosis: A Randomized, Double-Blind, Controlled, Crossover Clinical Trial
Abstract
1. Introduction
2. Materials and Methods
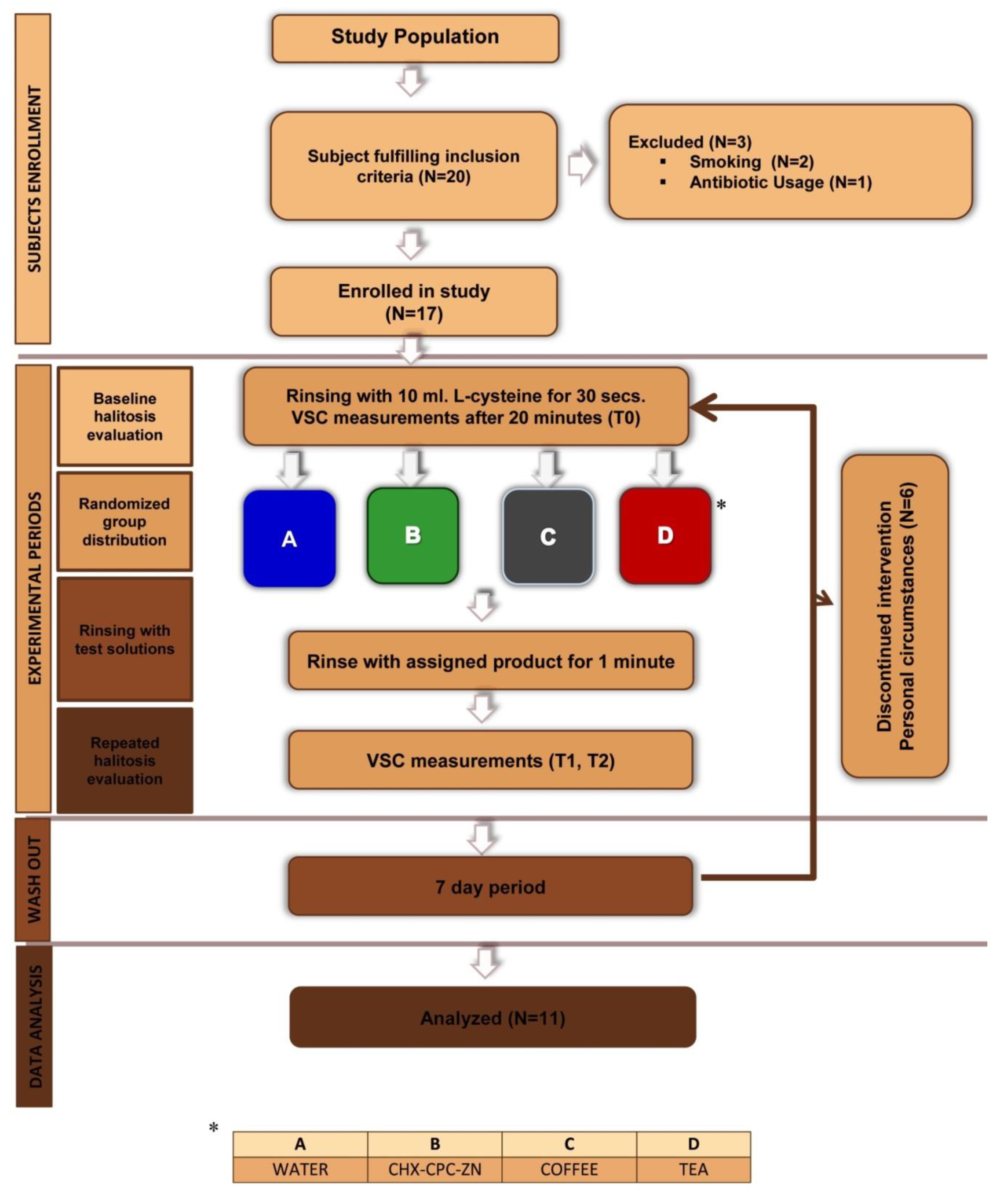
2.1. Study Design and Population
- Healthy male participants;
- Age 18 years or older;
- Absence of halitosis in exhaled air.
- Respiratory tract diseases;
- Tonsillitis;
- Stomach disorders;
- Antibiotic use in the previous three months;
- Being a smoker;
- Presence of fewer than 20 natural teeth.
2.2. Study Protocol
2.3. Experimental Phase
2.4. VSC Assessment
2.5. Statistical Tests
2.5.1. Study Power
2.5.2. Statistical Analysis
3. Results
3.1. Impact of Arabic Coffee on Halitosis
3.2. Impact of Black Tea on Halitosis
3.3. Impact of CHX-CPC-Zn Mouthwash on Halitosis
3.4. Impact of Water on Halitosis
3.5. Mouthwashes Intergroup Analysis
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- American Academy of Periodontology. Glossary of Periodontal Terms; American Academy of Periodontology: Chicago, IL, USA, 2001. [Google Scholar]
- Scully, C.; Greenman, J. Halitosis (breath odor). Periodontology 2000 2008, 48, 66–75. [Google Scholar] [CrossRef]
- Scully, C.; Greenman, J. Halitology (breath odour: Aetiopathogenesis and management). Oral Dis. 2012, 18, 333–345. [Google Scholar] [CrossRef]
- Alzoman, H. The association between periodontal diseases and halitosis among Saudi patients. Saudi Dent. J. 2021, 33, 34–38. [Google Scholar] [CrossRef]
- Yaegaki, K.; Coil, J.M. Examination, classification, and treatment of halitosis; clinical perspectives. J. Canadian Dent. Assoc. 2000, 66, 257–261. [Google Scholar]
- Yaegaki, K.; Brunette, D.M.; Tangerman, A.; Choe, Y.-S.; Winkel, E.G.; Ito, S.; Kitano, T.; Ii, H.; Calenic, B.; Ishkitiev, N.; et al. Standardization of clinical protocols in oral malodor research. J. Breath Res. 2012, 6, 017101. [Google Scholar] [CrossRef] [PubMed]
- Oshiro, A.; Zaitsu, T.; Ueno, M.; Kawaguchi, Y. Characterization of oral bacteria in the tongue coating of patients with halitosis using 16S rRNA analysis. Acta Odontol. Scand. 2020, 78, 541–546. [Google Scholar] [CrossRef] [PubMed]
- Slot, D.E.; De Geest, S.; Van Der Weijden, F.A.; Quirynen, M. Treatment of oral malodour. Medium-term efficacy of mechanical and/or chemical agents: A systematic review. J. Clin. Periodontol. 2015, 42, S303–S316. [Google Scholar] [CrossRef] [PubMed]
- Sood, P.; Devi, A.M.; Narang, R.; Swathi, V.; Makkar, D.K. Comparative Efficacy of Oil Pulling and Chlorhexidine on Oral Malodor: A Randomized Controlled Trial. J. Clin. Diagn. Res. 2014, 8, ZC18–ZC21. [Google Scholar] [CrossRef]
- Carvalho, M.D.; Tabchoury, C.; Cury, J.A.; Toledo, S.; Nogueira-Filho, G.R. Impact of mouthrinses on morning bad breath in healthy subjects. J. Clin. Periodontol. 2004, 31, 85–90. [Google Scholar] [CrossRef]
- Young, A.; Jonski, G.; Rölla, G. A study of triclosan and its solubilizers as inhibitors of oral malodour. J. Clin. Periodontol. 2002, 29, 1078–1081. [Google Scholar] [CrossRef]
- Blom, T.; Slot, D.; Quirynen, M.; Van Der Weijden, G. The effect of mouthrinses on oral malodor: A systematic review. Int. J. Dent. Hyg. 2012, 10, 209–222. [Google Scholar] [CrossRef]
- Alsaffar, D.; Alzoman, H. Efficacy of antioxidant mouthwash in the reduction of halitosis: A randomized, double blind, controlled crossover clinical trial. J. Dent. Sci. 2020. [Google Scholar] [CrossRef]
- Almas, K.; Al-Hawish, A.; Al-Khamis, W. Oral Hygiene Practices, Smoking Habits, and Self-Perceived Oral Malodor among Dental Students. J. Contemp. Dent. Pract. 2003, 4, 77–90. [Google Scholar] [CrossRef] [PubMed]
- Özen, A.E.; Bibiloni, M.D.M.; Pons, A.; Tur, J.A. Fluid intake from beverages across age groups: A systematic review. J. Hum. Nutr. Diet. 2014, 28, 417–442. [Google Scholar] [CrossRef] [PubMed]
- Tsou, S.-H.; Hu, S.-W.; Yang, J.-J.; Yan, M.; Lin, Y.-Y. Potential Oral Health Care Agent from Coffee against Virulence Factor of Periodontitis. Nutrients 2019, 11, 2235. [Google Scholar] [CrossRef] [PubMed]
- Gov, Y.; Sterer, N.; Rosenberg, M. In vitro effect of coffee on oral malodor-related parameters. J. Breath Res. 2010, 4, 026004. [Google Scholar] [CrossRef]
- Siddiqui, I.A.; Afaq, F.; Adhami, V.M.; Ahmad, N.; Mukhtar, H. Antioxidants of the Beverage Tea in Promotion of Human Health. Antioxid. Redox Signal. 2004, 6, 571–582. [Google Scholar] [CrossRef]
- Pohl, P.; Szymczycha-Madeja, A.; Welna, M. Direct ICP-OES multielement analysis of infused black and green teas and chemical fractionation of selected essential and non-essential elements prior to evaluation of their bioavailability and classification of teas by pattern recognition. Arab. J. Chem. 2020, 13, 1955–1965. [Google Scholar] [CrossRef]
- Chan, E.W.; Tie, P.P.; Soh, E.Y.; Law, Y.P. Antioxidant and antibacterial properties of green, black, and herbal teas of Camellia sinensis. Pharmacogn. Res. 2011, 3, 266–272. [Google Scholar] [CrossRef]
- Liu, Z.; Bruins, M.E.; Ni, L.; Vincken, J.-P. Green and Black Tea Phenolics: Bioavailability, Transformation by Colonic Microbiota, and Modulation of Colonic Microbiota. J. Agric. Food Chem. 2018, 66, 8469–8477. [Google Scholar] [CrossRef]
- Anita, P.; Sivasamy, S.; Kumar, P.D.M.; Balan, I.N.; Ethiraj, S. In vitro antibacterial activity of Camellia sinensis extract against cariogenic microorganisms. J. Basic Clin. Pharm. 2015, 6, 35–39. [Google Scholar] [CrossRef]
- Zeng, Q.C.; Wu, A.Z.; Pika, J. The effect of green tea extract on the removal of sulfur-containing oral malodor volatiles in vitro and its potential application in chewing gum. J. Breath Res. 2010, 4, 036005. [Google Scholar] [CrossRef]
- Lodhia, P.; Yaegaki, K.; Khakbaznejad, A.; Imai, T.; Sato, T.; Tanaka, T.; Murata, T.; Kamoda, T. Effect of Green Tea on Volatile Sulfur Compounds in Mouth Air. J. Nutr. Sci. Vitaminol. 2008, 54, 89–94. [Google Scholar] [CrossRef]
- Aizawa, F.; Kishi, M.; Moriya, T.; Takahashi, M.; Inaba, D.; Yonemitsu, M. The analysis of characteristics of elderly people with high VSC level. Oral Dis. 2005, 11, 80–82. [Google Scholar] [CrossRef] [PubMed]
- Silness, J.; Löe, H. Periodontal Disease in Pregnancy II. Correlation between Oral Hygiene and Periodontal Condition. Acta Odontol. Scand. 1964, 22, 121–135. [Google Scholar] [CrossRef]
- Löe, H.; Silness, J. Periodontal Disease in Pregnancy I. Prevalence and Severity. Acta Odontol. Scand. 1963, 21, 533–551. [Google Scholar] [CrossRef]
- Kleinberg, I.; Codipilly, D. Cysteine challenge testing: A powerful tool for examining oral malodour processes and treatments in vivo. Int. Dent. J. 2002, 52, 221–228. [Google Scholar] [CrossRef] [PubMed]
- Reyes, C.M.; Cornelis, M.C. Caffeine in the Diet: Country-Level Consumption and Guidelines. Nutrients 2018, 10, 1772. [Google Scholar] [CrossRef] [PubMed]
- Mendes, L.; Coimbra, J.; Pereira, A.L.; Resende, M.; Pinto, M.G. Comparative effect of a new mouthrinse containing chlorhexidine, triclosan and zinc on volatile sulphur compounds: A randomized, crossover, double-blind study. Int. J. Dent. Hyg. 2016, 14, 202–208. [Google Scholar] [CrossRef]
- Duangjai, A.; Suphrom, N.; Wungrath, J.; Ontawong, A.; Nuengchamnong, N.; Yosboonruang, A. Comparison of antioxidant, antimicrobial activities and chemical profiles of three coffee (Coffea arabica L.) pulp aqueous extracts. Integr. Med. Res. 2016, 5, 324–331. [Google Scholar] [CrossRef]
- Tomac, I.; Šeruga, M.; Labuda, J. Evaluation of antioxidant activity of chlorogenic acids and coffee extracts by an electrochemical DNA-based biosensor. Food Chem. 2020, 325, 126787. [Google Scholar] [CrossRef]
- Tasew, T.; Mekonnen, Y.; Gelana, T.; Redi-Abshiro, M.; Chandravanshi, B.S.; Ele, E.; Mohammed, A.M.; Mamo, H. In Vitro Antibacterial and Antioxidant Activities of Roasted and Green Coffee Beans Originating from Different Regions of Ethiopia. Int. J. Food Sci. 2020, 2020, 1–8. [Google Scholar] [CrossRef]
- Daglia, M.; Papetti, A.; Grisoli, P.; Aceti, C.; Spini, V.; Dacarro, C.; Gazzani, G. Isolation, Identification, and Quantification of Roasted Coffee Antibacterial Compounds. J. Agric. Food Chem. 2007, 55, 10208–10213. [Google Scholar] [CrossRef] [PubMed]
- Mehta, V.V.; Rajesh, G.; Rao, A.; Shenoy, R.; Mithun Pai, B.H. Antimicrobial Efficacy of Punica granatum mesocarp, Nelumbo nucifera Leaf, Psidium guajava Leaf and Coffea Canephora Extract on Common Oral Pathogens: An In-vitro Study. J. Clin. Diagn. Res. JCDR 2014, 8, 65–68. [Google Scholar]
- Bharath, N.; Sowmya, N.K.; Mehta, D.S. Determination of antibacterial activity of green coffee bean extract on periodontogenic bacteria like Porphyromonas gingivalis, Prevotella intermedia, Fusobacterium nucleatum and Aggregatibacter actinomycetemcomitans: An in vitro study. Contemp. Clin. Dent. 2015, 6, 166–169. [Google Scholar] [CrossRef]
- Ghazi, A.M.; Na’Ma, A.J.; Kshash, Q.H.; Jasim, N.S. Study of the Antibacterial Activity of Elettaria Cardamomum Extracts on the Growth of Some Gingivitis Inducing Bacteria in Culture Media. Int. J. Drug Deliv. Technol. 2020, 10, 38–45. [Google Scholar] [CrossRef]
- Souissi, M.; Azelmat, J.; Chaieb, K.; Grenier, D. Antibacterial and anti-inflammatory activities of cardamom (Elettaria cardamomum) extracts: Potential therapeutic benefits for periodontal infections. Anaerobe 2020, 61, 102089. [Google Scholar] [CrossRef] [PubMed]
- Erawati, S.; Rahardjo, A.; Pintauli, S. Clinical Efficacy of a New Mouthwash Containing Essential Oil of Cardamom in Reducing Volatile Sulphur Coumpounds Concentration. Int. J. Clin. Prev. Dent. 2014, 10, 237–242. [Google Scholar] [CrossRef]
- Farina, V.H.; De Lima, A.P.; Balducci, I.; Brandão, A.A.H. Effects of the medicinal plants Curcuma zedoaria and Camellia sinensis on halitosis control. Braz. Oral Res. 2012, 26, 523–529. [Google Scholar] [CrossRef]
- Young, A.; Jonski, G.; Rölla, G. The oral anti-volatile sulphur compound effects of zinc salts and their stability constants. Eur. J. Oral Sci. 2002, 110, 31–34. [Google Scholar] [CrossRef] [PubMed]
- Van Steenberghe, D.; Avontroodt, P.; Peeters, W.; Pauwels, M.; Coucke, W.; Lijnen, A.; Quirynen, M. Effect of Different Mouthrinses on Morning Breath. J. Periodontol. 2001, 72, 1183–1191. [Google Scholar] [CrossRef]
- Roldán, S.; Winkel, E.G.; Herrera, D.; Sanz, M.; Van Winkelhoff, A.J. The effects of a new mouthrinse containing chlorhexidine, cetylpyridinium chloride and zinc lactate on the microflora of oral halitosis patients: A dual-centre, double-blind placebo-controlled study. J. Clin. Periodontol. 2003, 30, 427–434. [Google Scholar] [CrossRef] [PubMed]
- Dadamio, J.; Laleman, I.; De Geest, S.; Vancauwenberghe, F.; DeKeyser, C.; Coucke, W.; Quirynen, M. Usefulness of a new malodour-compound detection portable device in oral malodour diagnosis. J. Breath Res. 2013, 7, 46005. [Google Scholar] [CrossRef]
- Kakoei, S.; Barkhori, F.; Mirzazadeh, A.; Mohammadi, M.; Gholamhoseinian, A. Influence of menstrual cycle and salivary ß-estradiol on volatile sulfur compound. J. Oral Health Oral Epidemiol. 2012, 1, 41–45. [Google Scholar]
- Calil, C.; Lima, P.; Bernardes, C.; Groppo, F.; Bado, F.; Marcondes, F. Influence of gender and menstrual cycle on volatile sulphur compounds production. Arch. Oral Biol. 2008, 53, 1107–1112. [Google Scholar] [CrossRef] [PubMed]
| Variables | Mean ± SD |
|---|---|
| Age | 22.2(±4.1) |
| PI | 1.05 (±0.77) |
| GI | 1.26 (±0.74) |
| PD | 2.1 (±0.92) |
| Intervention | Mean (SD) | p-Value | ||
|---|---|---|---|---|
| To | T1 | T2 | ||
| H2S | ||||
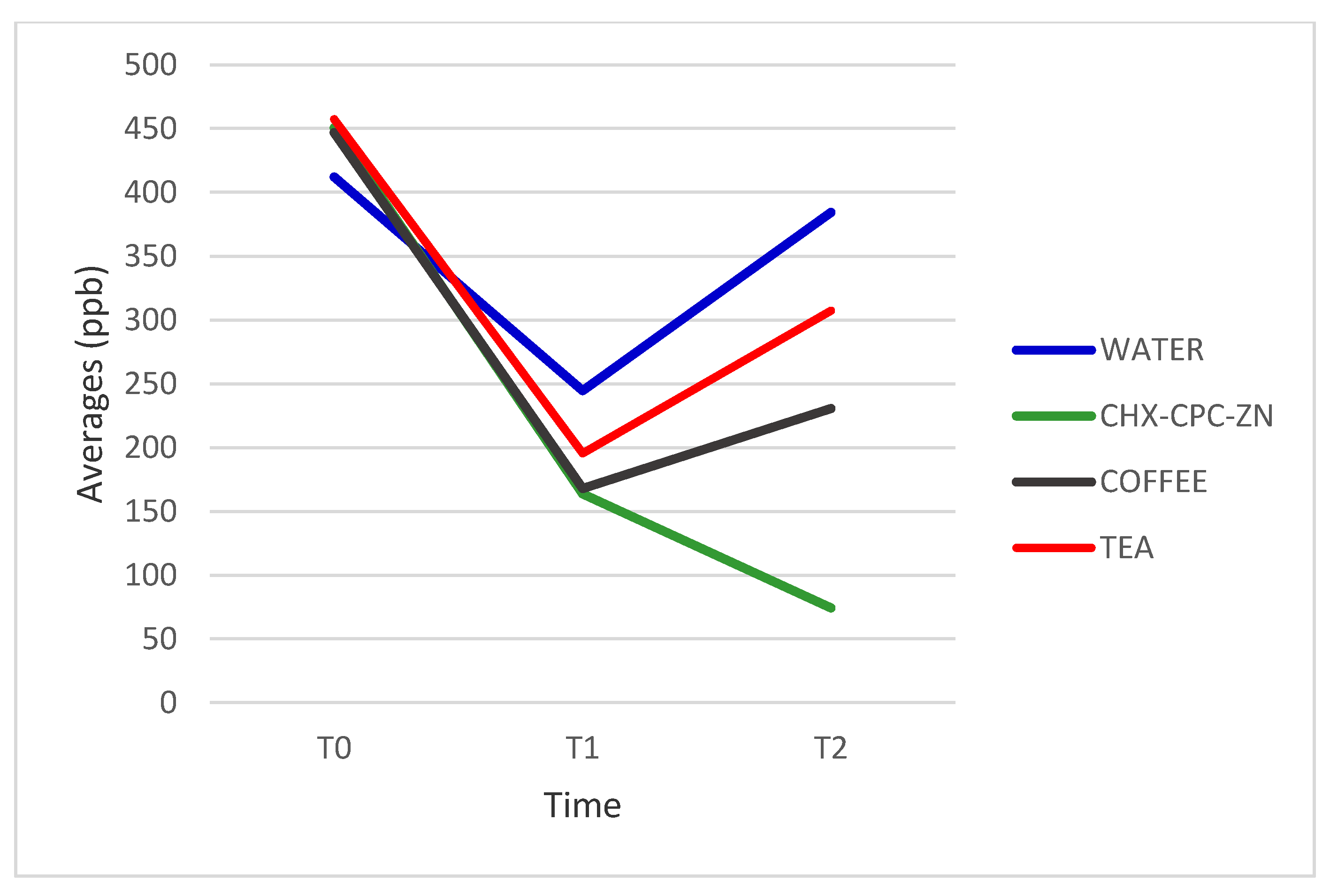
| WATER | 412.18 (148.53) | 244.54 (95.28) | 384.36 (161.89) | 0.710 |
| CHX-CPC-ZN | 450.54 (291.35) | 163.72 (178.95) | 74.27 (72.21) | 0.000 * |
| COFFEE | 446.82 (169.65) | 167.82 (88.41) | 230.54 (128.82) | 0.000 * |
| TEA | 457.27 (334.58) | 195.54 (154.27) | 307.27 (252.08) | 0.045 * |
| Methyl mercaptan | ||||
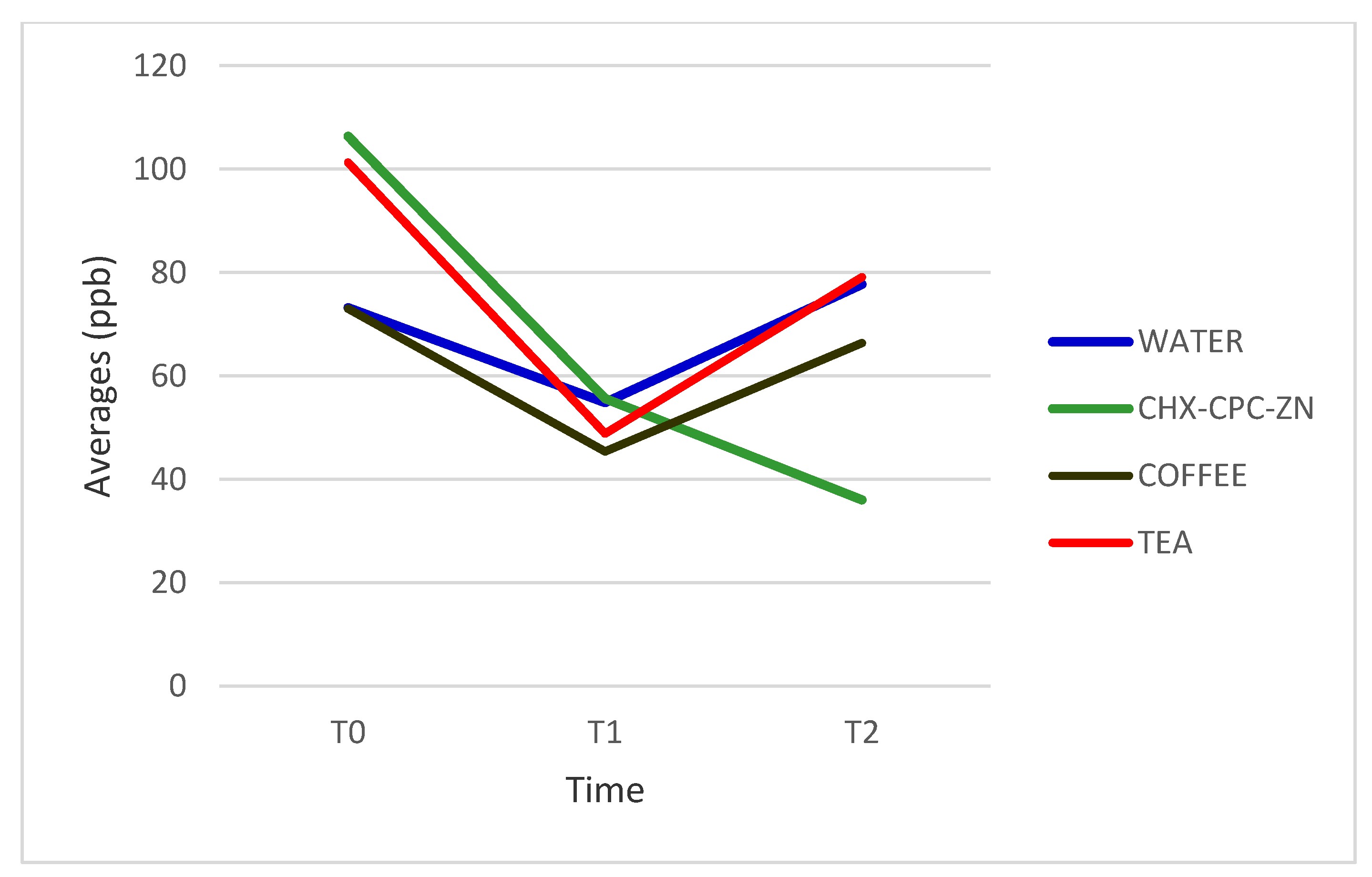
| WATER | 73.18 (44.61) | 54.82 (20.65) | 77.72 (38.96) | 0.306 |
| CHX-CPC-ZN | 106.36 (63.73) | 55.63 (38.87) | 36.0 (36.05) | 0.005 * |
| COFFEE | 73.0 (58.88) | 45.36 (52.87) | 66.36 (44.77) | 0.445 |
| TEA | 101.27 (67.35) | 48.82 (26.35) | 79.09 (27.03) | 0.033 * |
| Time | Mean (SD) | p-Value | |||
|---|---|---|---|---|---|
| WATER | CHX-CPC-ZN | COFFEE | TEA | ||
| H2S | |||||
| BASELINE | 412.18 (148.53) | 450.54 (291.35) | 446.82 (169.65) | 457.27 (334.58) | 0.975 |
| T1 | 244.54 (95.28) | 163.72 (178.95) | 167.82 (88.41) | 195.54 (154.27) | 0.481 |
| T2 | 384.36 (161.89) | 74.27 (72.21) | 230.54 (128.82) | 307.27(252.08) | 0.000 * |
| Methyl mercaptan | |||||
| T0 | 73.18 (44.61) | 106.36 (63.73) | 73.0 (58.88) | 101.27 (67.35) | 0.403 |
| T1 | 54.82 (20.65) | 55.63 (38.87) | 45.36 (52.87) | 48.82 (26.35) | 0.899 |
| T2 | 77.72 (38.96) | 36.0 (36.05) | 66.36 (44.77) | 79.09 (27.03) | 0.034 * |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alzoman, H.; Alzahrani, A.; Alwehaiby, K.; Alanazi, W.; AlSarhan, M. Efficacy of Arabic Coffee and Black Tea in Reducing Halitosis: A Randomized, Double-Blind, Controlled, Crossover Clinical Trial. Healthcare 2021, 9, 250. https://doi.org/10.3390/healthcare9030250
Alzoman H, Alzahrani A, Alwehaiby K, Alanazi W, AlSarhan M. Efficacy of Arabic Coffee and Black Tea in Reducing Halitosis: A Randomized, Double-Blind, Controlled, Crossover Clinical Trial. Healthcare. 2021; 9(3):250. https://doi.org/10.3390/healthcare9030250
Chicago/Turabian StyleAlzoman, Hamad, Ahmed Alzahrani, Khaled Alwehaiby, Waleed Alanazi, and Mohammed AlSarhan. 2021. "Efficacy of Arabic Coffee and Black Tea in Reducing Halitosis: A Randomized, Double-Blind, Controlled, Crossover Clinical Trial" Healthcare 9, no. 3: 250. https://doi.org/10.3390/healthcare9030250
APA StyleAlzoman, H., Alzahrani, A., Alwehaiby, K., Alanazi, W., & AlSarhan, M. (2021). Efficacy of Arabic Coffee and Black Tea in Reducing Halitosis: A Randomized, Double-Blind, Controlled, Crossover Clinical Trial. Healthcare, 9(3), 250. https://doi.org/10.3390/healthcare9030250






