Short-Term Impact of Video-Assisted Thoracoscopic Surgery on Lung Function, Physical Function, and Quality of Life
Abstract
1. Introduction
2. Methods
2.1. Study Design
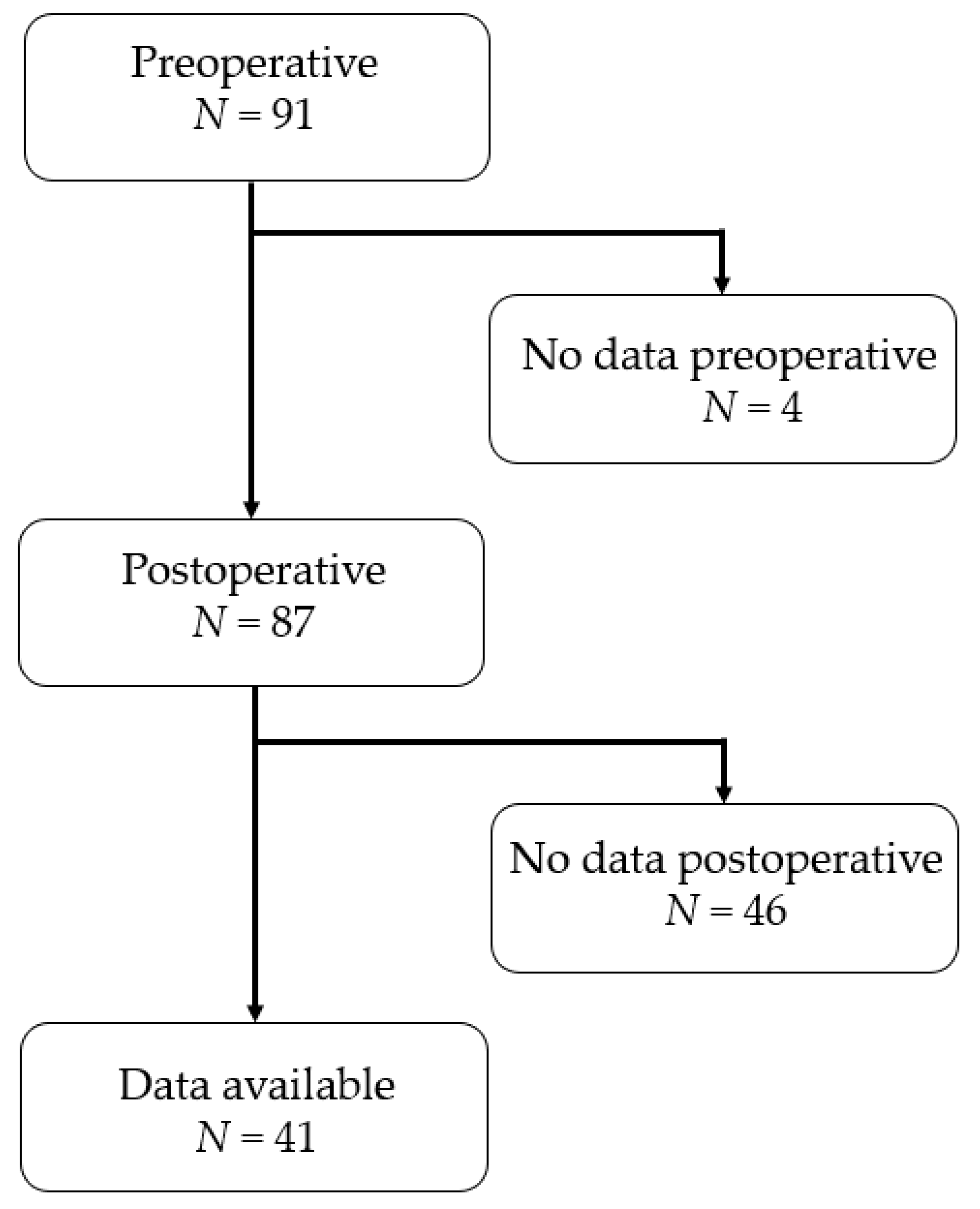
2.2. Patients and Methods
2.3. Video-Assisted Thoracoscopic Surgery
2.4. Rehabilitation Program
2.5. Lung Function
2.6. Physical Function
2.7. Health-Related Quality of Life
2.8. Statistical Analysis
3. Results
3.1. Socio-Demographic and Clinical Characteristics
3.2. Differences in Lung Function before and after VATS
3.3. Differences in Physical Function before and after VATS
3.4. Differences in Health-Related QOL before and after VATS
3.5. Factors Affecting Postoperative QOL
4. Discussion
Study Limitations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Jemal, A.; Bray, F.; Center, M.M.; Ferlay, J.; Ward, E.; Forman, D. Global cancer statistics. CA Cancer J. Clin. 2011, 61, 69–90. [Google Scholar] [CrossRef] [PubMed]
- Ferlay, J.; Shin, H.R.; Bray, F.; Forman, D.; Mathers, C.; Parkin, D.M. Estimates of worldwide burden of cancer in 2008: GLOBOCAN 2008. Int. J. Cancer 2010, 127, 2893–2917. [Google Scholar] [CrossRef] [PubMed]
- Grogan, E.L.; Jones, D.R. VATS lobectomy is better than open thoracotomy: What is the evidence for short-term outcomes? Thorac. Surg Clin. 2008, 18, 249–258. [Google Scholar] [CrossRef] [PubMed]
- Cheng, D.; Downey, R.J.; Kernstine, K.; Stanbridge, R.; Shennib, H.; Wolf, R.; Ohtsuka, T.; Schmid, R.; Waller, D.; Fernando, H.; et al. Video-assisted thoracic surgery in lung cancer resection: A meta-analysis and systematic review of controlled trials. Innovations 2007, 2, 261–292. [Google Scholar] [CrossRef]
- Kaseda, S.; Aoki, T.; Hangai, N.; Shimizu, K. Better pulmonary function and prognosis with video-assisted thoracic surgery than with thoracotomy. Ann. Thorac. Surg. 2000, 70, 1644–1646. [Google Scholar] [CrossRef]
- Brunelli, A.; Thomas, C.; Dinesh, P.; Lumb, A. Enhanced recovery pathway versus standard care in patients undergoing video-assisted thoracoscopic lobectomy. J. Thorac. Cardiovasc. Surg. 2017, 154, 2084–2090. [Google Scholar] [CrossRef]
- Agostini, P.; Lugg, S.T.; Adams, K.; Smith, T.; Kalkat, M.; Rajesh, P.B.; Steyn, R.S.; Naidu, B.; Rushton, A.; Bishay, E. Video-assisted thoracoscopic lobectomy: Which patients require postoperative physiotherapy? Physiotherapy 2020, 106, 87–93. [Google Scholar] [CrossRef]
- Nomori, H.; Ohtsuka, T.; Horio, H.; Naruke, T.; Suemasu, K. Difference in the impairment of vital capacity and 6-minute walking after a lobectomy performed by thoracoscopic surgery, an anterior limited thoracotomy, an anteroaxillary thoracotomy, and a posterolateral thoracotomy. Surg. Today 2003, 33, 7–12. [Google Scholar] [CrossRef]
- Podsiadlo, D.; Richardson, S. The timed “Up & Go”: A test of basic functional mobility for frail elderly persons. J. Am. Geriatr. Soc. 1991, 39, 142–148. [Google Scholar]
- Jones, C.J.; Rikli, R.E.; Beam, W.C. A 30-s chair-stand test as a measure of lower body strength in community-residing older adults. Res. Q Exerc. Sport. 1999, 70, 113–119. [Google Scholar] [CrossRef]
- Aaronson, N.K.; Ahmedzai, S.; Bergman, B.; Bullinger, M.; Cull, A.; Duez, N.J.; Filiberti, A.; Flechtner, H.; Fleishman, S.B.; de Haes, J.C.; et al. The European Organization for Research and Treatment of Cancer QLQ-C30: A quality-of-life instrument for use in international clinical trials in oncology. J. Natl. Cancer Inst. 1993, 85, 365–376. [Google Scholar] [CrossRef] [PubMed]
- Kubota, M.; Kobayashi, H.; Quanjer, P.H.; Omori, H.; Tatsumi, K.; Kanazawa, M. Reference values for spirometry, including vital capacity, in Japanese adults calculated with the LMS method and compared with previous values. Respir. Investig. 2014, 52, 242–250. [Google Scholar] [CrossRef]
- Sawabata, N.; Nagayasu, T.; Kadota, Y.; Goto, T.; Horio, H.; Mori, T.; Yamashita, S.; Iwasaki, A. Risk assessment of lung resection for lung cancer according to pulmonary function: Republication of systematic review and proposals by guideline committee of the Japanese association for chest surgery 2014. Gen. Thorac. Cardiovasc. Surg. 2015, 63, 14–21. [Google Scholar] [CrossRef] [PubMed]
- Montazeri, A.; Gillis, C.R.; McEwen, J. Quality of life in patients with lung cancer: A review of literature from 1970 to 1995. Chest 1998, 113, 467–481. [Google Scholar] [PubMed]
- Abendstein, H.; Nordgren, M.; Boysen, M.; Jannert, M.; Silander, E.; Ahlner-Elmqvist, M.; Hammerlid, E.; Bjordal, K. Quality of life and head and neck cancer: A 5 year prospective study. Laryngoscope 2005, 115, 2183–2192. [Google Scholar] [CrossRef]
- Bjordal, K.; de Graeff, A.; Fayers, P.M.; Hammerlid, E.; van Pottelsberghe, C.; Curran, D.; Ahlner-Elmqvist, M.; Maher, E.J.; Meyza, J.W.; Brédart, A.; et al. A 12 country field study of the EORTC QLQ-C30 (version 3.0) and the head and neck cancer specific module (EORTC QLQ-H&N35) in head and neck patients. EORTC Qual. Life Group. Eur. J. Cancer. 2000, 36, 1796–1807. [Google Scholar]
- Nomori, H.; Horio, H.; Naruke, T.; Suemasu, K. What is the advantage of a thoracoscopic lobectomy over a limited thoracotomy procedure for lung cancer surgery? Ann. Thorac. Surg. 2001, 72, 879–884. [Google Scholar] [CrossRef]
- Stéphan, F.; Boucheseiche, S.; Hollande, J.; Flahault, A.; Cheffi, A.; Bazelly, B.; Bonnet, F. Pulmonary complications following lung resection: A comprehensive analysis of incidence and possible risk factors. Chest 2000, 118, 1263–1270. [Google Scholar]
- Uramoto, H.; Nakanishi, R.; Fujino, Y.; Imoto, H.; Takenoyama, M.; Yoshimatsu, T.; Oyama, T.; Osaki, T.; Yasumoto, K. Prediction of pulmonary complications after a lobectomy in patients with non-small cell lung cancer. Thorax 2001, 56, 59–61. [Google Scholar] [CrossRef]
- Uzieblo, M.; Welsh, R.; Pursel, S.E.; Chmielewski, G.W. Incidence and significance of lobar atelectasis in thoracic surgical patients. Am. Surg. 2000, 66, 476–480. [Google Scholar]
- Korst, R.J.; Humphrey, C.B. Complete lobar collapse following pulmonary lobectomy. Its incidence, predisposing factors, and clinical ramifications. Chest 1997, 111, 1285–1289. [Google Scholar] [CrossRef] [PubMed]
- Abolhoda, A.; Liu, D.; Brooks, A.; Burt, M. Prolonged air leak following radical upper lobectomy: An analysis of incidence and possible risk factors. Chest 1998, 113, 1507–1510. [Google Scholar] [CrossRef] [PubMed]
- Donoghue, O.A.; Horgan, N.F.; Savva, G.M.; Cronin, H.; O’Regan, C.; Kenny, R.A. Association between timed up-and-go and memory, executive function, and processing speed. J. Am. Geriatr. Soc. 2012, 60, 1681–1686. [Google Scholar] [CrossRef] [PubMed]
- Hollands, K.L.; Hollands, M.A.; Zietz, D.; Wing, A.M.; Wright, C.; van Vliet, P. Kinematics of turning 180° during the timed up and go in stroke survivors with and without falls history. Neurorehabil. Neural Repair. 2010, 24, 358–367. [Google Scholar] [CrossRef] [PubMed]
- Benton, M.J.; Alexander, J.L. Validation of functional fitness tests as surrogates for strength measurement in frail, older adults with chronic obstructive pulmonary disease. Am. J. Phys Med. Rehabil. 2009, 88, 579–583. [Google Scholar] [CrossRef]
- Demmy, T.L.; Nwogu, C. Is video-assisted thoracic surgery lobectomy better? Quality of life considerations. Ann. Thorac. Surg. 2008, 85, S719–S728. [Google Scholar] [CrossRef]
- Puri, V.; Meyers, B.F. Video-assisted thoracoscopic surgery lobectomy for lung cancer. Surg. Oncol. Clin. N. Am. 2013, 22, 27–38. [Google Scholar] [CrossRef]
| Comorbidities | N |
|---|---|
| Chronic obstructive pulmonary disease | |
| GOLD stage I | 9 |
| GOLD stage II | 7 |
| Hypertension | 16 |
| Mitral insufficiency | 10 |
| Aortic insufficiency | 7 |
| Diabetes | 7 |
| Angina pectoris | 6 |
| Ischemic heart disease | 5 |
| Dyslipidemia | 6 |
| Tricuspid regurgitation | 2 |
| Interstitial pneumonia | 1 |
| Congestive heart failure | 1 |
| Item | Preoperative | Postoperative | p Value | ||
|---|---|---|---|---|---|
| Mean (SD) | Median | Mean (SD) | Median | ||
| FVC in L (L) | 2.5 (0.6) | 2.4 | 1.7 (0.5) | 1.7 | p < 0.001 |
| FVC/pred (%) | 88.2 (15.1) | 87.0 | 62.2 (14.8) | 61.0 | p < 0.001 |
| FEV1 in L (L) | 1.9 (0.5) | 1.9 | 1.3 (0.3) | 1.2 | p < 0.001 |
| FEV1/FVC (%) | 77.6 (8.7) | 79.6 | 76.6 (8.9) | 78.9 | 0.367 |
| FEV1% pred (%) | 84.7 (18.1) | 84.0 | 58.3 (15.6) | 55.0 | p < 0.001 |
| TUG (s) | 6.2 (1.2) | 6.0 | 6.7 (1.3) | 7.0 | 0.006 |
| CS-30 (number of times) | 16.1 (4.4) | 16 | 15.1 (4.3) | 14 | 0.020 |
| Item | Preoperative | Postoperative | p-Value | ||
|---|---|---|---|---|---|
| Mean (SD) | Median | Mean (SD) | Median | ||
| Global health status/QOL | 68.9 (20.9) | 667 | 58.9 (20.8) | 50.0 | 0.003 |
| Physical function | 91.9 (9.9) | 93.3 | 82.0 (13.1) | 86.7 | p < 0.001 |
| Role function | 91.5 (16.3) | 100 | 72.9 (24.7) | 66.7 | 0.001 |
| Emotional function | 76.0 (20.3) | 75.0 | 78.0 (20.5) | 75.0 | 0.455 |
| Cognitive function | 84.6 (16.4) | 83.3 | 81.3 (18.0) | 83.3 | 0.344 |
| Social function | 91.5 (15.0) | 100 | 81.3 (22.7) | 83.3 | 0.005 |
| Fatigue | 17.1 (16.7) | 11.1 | 32.2 (19.8) | 33.3 | 0.001 |
| Nausea/vomiting | 0.8 (3.6) | 0 | 3.7 (10.2) | 0 | 0.084 |
| Pain | 7.3 (14.0) | 0 | 28.5 (16.8) | 33.3 | p < 0.001 |
| Dyspnea | 9.8 (17.1) | 0 | 31.7 (21.0) | 33.3 | p < 0.001 |
| Insomnia/sleep | 19.5 (31.6) | 0 | 30.9 (25.2) | 33.3 | 0.008 |
| Appetite loss | 10.6 (21.7) | 0 | 22.0 (26.5) | 0 | 0.022 |
| Constipation | 13.8 (22.3) | 0 | 22.8 (30.2) | 0 | 0.033 |
| Diarrhea | 5.7 (12.7) | 0 | 7.3 (14.0) | 0 | 0.527 |
| Financial hardship related to illness | 8.9 (15.0) | 0 | 13.8 (21.0) | 0 | 0.134 |
| Item | Included Variable † | β | Adjusted R2 | p Value |
|---|---|---|---|---|
| Global health status/QOL | Presence or absence of COPD | −13.813 | 0.085 | 0.036 |
| Physical | Presence or absence of COPD CS30 | −13.247 1.208 | 0.277 | 0.001 0.007 |
| Role | FVC/predicted after surgery | 0.769 | 0.195 | 0.003 |
| Cognitive | Recovery rate of postoperative VC | −39.300 | 0.148 | 0.008 |
| Dyspnea | FVC/predicted after surgery | −0.496 | 0.100 | 0.025 |
| Appetite loss | CS30 | −2.703 | 0.174 | 0.004 |
| Constipation | Recovery rate of postoperative VC Day of start walking after surgery | 88.390 35.801 | 0.269 | p < 0.001 0.010 |
| Diarrhea | Age | 0.650 | 0.182 | 0.003 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Akezaki, Y.; Nakata, E.; Tominaga, R.; Iwata, O.; Kawakami, J.; Tsuji, T.; Ueno, T.; Yamashita, M.; Sugihara, S. Short-Term Impact of Video-Assisted Thoracoscopic Surgery on Lung Function, Physical Function, and Quality of Life. Healthcare 2021, 9, 136. https://doi.org/10.3390/healthcare9020136
Akezaki Y, Nakata E, Tominaga R, Iwata O, Kawakami J, Tsuji T, Ueno T, Yamashita M, Sugihara S. Short-Term Impact of Video-Assisted Thoracoscopic Surgery on Lung Function, Physical Function, and Quality of Life. Healthcare. 2021; 9(2):136. https://doi.org/10.3390/healthcare9020136
Chicago/Turabian StyleAkezaki, Yoshiteru, Eiji Nakata, Ritsuko Tominaga, Orie Iwata, Juichi Kawakami, Tetsuya Tsuji, Tsuyoshi Ueno, Motohiro Yamashita, and Shinsuke Sugihara. 2021. "Short-Term Impact of Video-Assisted Thoracoscopic Surgery on Lung Function, Physical Function, and Quality of Life" Healthcare 9, no. 2: 136. https://doi.org/10.3390/healthcare9020136
APA StyleAkezaki, Y., Nakata, E., Tominaga, R., Iwata, O., Kawakami, J., Tsuji, T., Ueno, T., Yamashita, M., & Sugihara, S. (2021). Short-Term Impact of Video-Assisted Thoracoscopic Surgery on Lung Function, Physical Function, and Quality of Life. Healthcare, 9(2), 136. https://doi.org/10.3390/healthcare9020136






