Factors Associated with SARS-CoV-2 Infection Risk among Healthcare Workers of an Italian University Hospital
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Setting and Population
2.2. Data Collection
2.3. Statistical Analysis
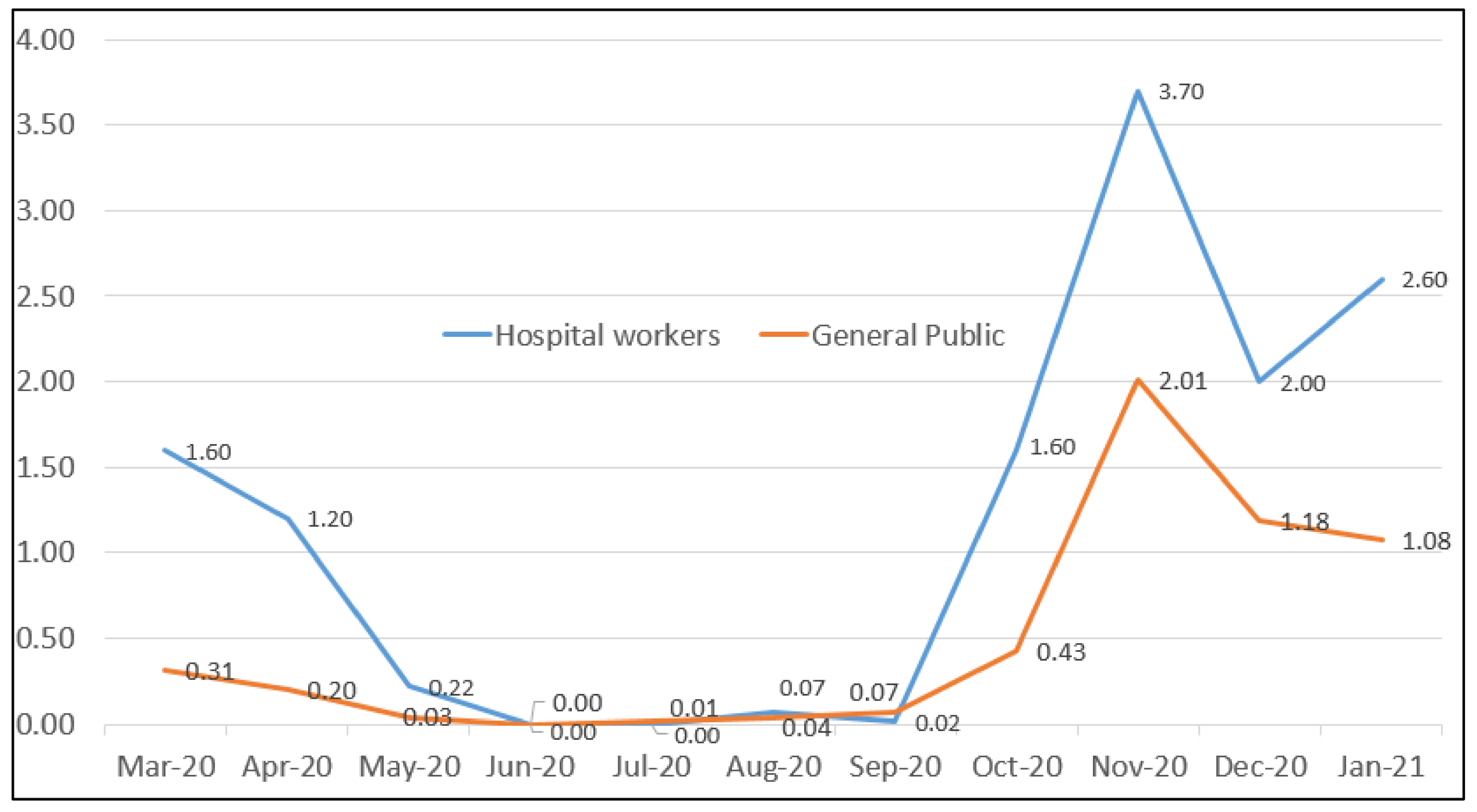
3. Results
3.1. Association of SARS-CoV-2 Infection with the Variables Studied
3.2. Results of the Multiple Logistic Regression Analysis
4. Discussion
Limitations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Iversen, K.; Bundgaard, H.; Hasselbalch, R.B.; Kristensen, J.H.; Nielsen, P.B.; Pries-Heje, M.; Knudsen, A.D.; Christensen, C.E.; Fogh, K.; Norsk, J.B.; et al. Risk of COVID-19 in health-care workers in Denmark: An observational cohort study. Lancet Infect. Dis. 2020, 20, 1401–1408. [Google Scholar] [CrossRef]
- Modenese, A.; Gobba, F. Increased Risk of COVID-19-Related Deaths among General Practitioners in Italy. Healthcare 2020, 8, 155. [Google Scholar] [CrossRef] [PubMed]
- Shah, A.S.V.; Wood, R.; Gribben, C.; Caldwell, D.; Bishop, J.; Weir, A.; Kennedy, S.; Reid, M.; Smith-Palmer, A.; Goldberg, D.; et al. Risk of hospital admission with coronavirus disease 2019 in healthcare workers and their households: Nationwide linkage cohort study. BMJ 2020, 371, m3582. [Google Scholar] [CrossRef] [PubMed]
- Gao, M.; Piernas, C.; Astbury, N.M.; Hippisley-Cox, J.; O’Rahilly, S.; Aveyard, P.; Jebb, S.A. Associations between body-mass index and COVID-19 severity in 6·9 million people in England: A prospective, community-based, cohort study. Lancet Diabetes Endocrinol. 2021, 9, 350–359. [Google Scholar] [CrossRef]
- Moazzami, B.; Chaichian, S.; Kasaeian, A.; Djalalinia, S.; Akhlaghdoust, M.; Eslami, M.; Broumand, B. Metabolic risk factors and risk of Covid-19: A systematic review and meta-analysis. PLoS ONE 2020, 15, e0243600. [Google Scholar] [CrossRef]
- Rizza, S.; Coppeta, L.; Grelli, S.; Ferrazza, G.; Chiocchi, M.; Vanni, G.; Bonomo, O.C.; Bellia, A.; Andreoni, M.; Magrini, A.; et al. High body mass index and night shift work are associated with COVID-19 in health care workers. J. Endocrinol. Investig. 2021, 44, 1097–1101. [Google Scholar] [CrossRef]
- Thakur, B.; Dubey, P.; Benitez, J.; Torres, J.P.; Reddy, S.; Shokar, N.; Aung, K.; Mukherjee, D.; Dwivedi, A.K. A systematic review and meta-analysis of geographic differences in comorbidities and associated severity and mortality among individuals with COVID-19. Sci. Rep. 2021, 11, 8562. [Google Scholar] [CrossRef]
- U.S. Centers for Disease Control and Prevention (CDC). People with Certain Medical Conditions. Available online: https://www.cdc.gov/coronavirus/2019-ncov/need-extra-precautions/people-with-medical-conditions.html (accessed on 1 November 2021).
- Çelebi, G.; Pişkin, N.; Çelik Bekleviç, A.; Altunay, Y.; Salcı Keleş, A.; Tüz, M.A.; Altınsoy, B.; Hacıseyitoğlu, D. Specific risk factors for SARS-CoV-2 transmission among health care workers in a university hospital. Am. J. Infect. Control 2020, 48, 1225–1230. [Google Scholar] [CrossRef] [PubMed]
- Jespersen, S.; Mikkelsen, S.; Greve, T.; Kaspersen, K.A.; Tolstrup, M.; Boldsen, J.K.; Redder, J.D.; Nielsen, K.; Abildgaard, A.M.; Kolstad, H.A.; et al. SARS-CoV-2 seroprevalence survey among 17,971 healthcare and administrative personnel at hospitals, pre-hospital services, and specialist practitioners in the Central Denmark Region. Clin. Infect. Dis. 2020, ciaa1471. [Google Scholar] [CrossRef]
- Kantele, A.; Lääveri, T.; Kareinen, L.; Pakkanen, S.H.; Blomgren, K.; Mero, S.; Patjas, A.; Virtanen, J.; Uusitalo, R.; Lappalainen, M.; et al. SARS-CoV-2 infections among healthcare workers at Helsinki University Hospital, Finland, spring 2020: Serosurvey, symptoms and risk factors. Travel Med. Infect. Dis. 2021, 39, 101949. [Google Scholar] [CrossRef] [PubMed]
- Poletti, P.; Tirani, M.; Cereda, D.; Guzzetta, G.; Trentini, F.; Marziano, V.; Toso, C.; Piatti, A.; Piccarreta, R.; Melegaro, A.; et al. Seroprevalence of and Risk Factors Associated With SARS-CoV-2 Infection in Health Care Workers During the Early COVID-19 Pandemic in Italy. JAMA Netw. Open 2021, 4, e2115699. [Google Scholar] [CrossRef] [PubMed]
- Piccoli, L.; Ferrari, P.; Piumatti, G.; Jovic, S.; Rodriguez, B.F.; Mele, F.; Giacchetto-Sasselli, I.; Terrot, T.; Silacci-Fregni, C.; Cameroni, E.; et al. Risk assessment and seroprevalence of SARS-CoV-2 infection in healthcare workers of COVID-19 and non-COVID-19 hospitals in Southern Switzerland. Lancet Reg. Health Eur. 2021, 1, 100013. [Google Scholar] [CrossRef] [PubMed]
- Vimercati, L.; Stefanizzi, P.; De Maria, L.; Caputi, A.; Cavone, D.; Quarato, M.; Gesualdo, L.; Lopalco, P.L.; Migliore, G.; Sponselli, S.; et al. Large-scale IgM and IgG SARS-CoV-2 serological screening among healthcare workers with a low infection prevalence based on nasopharyngeal swab tests in an Italian university hospital: Perspectives for public health. Environ. Res. 2021, 195, 110793. [Google Scholar] [CrossRef]
- Gagneux-Brunon, A.; Pelissier, C.; Gagnaire, J.; Pillet, S.; Pozzetto, B.; Botelho-Nevers, E.; Berthelot, P. SARS-CoV-2 infection: Advocacy for training and social distancing in healthcare settings. J. Hosp. Infect. 2020, 106, 610–612. [Google Scholar] [CrossRef] [PubMed]
- Griswold, D.P.; Gempeler, A.; Kolias, A.; Hutchinson, P.J.; Rubiano, A.M. Personal protective equipment for reducing the risk of COVID-19 infection among health care workers involved in emergency trauma surgery during the pandemic: An umbrella review. J. Trauma Acute Care Surg. 2021, 90, e72–e80. [Google Scholar] [CrossRef]
- Nguyen, L.H.; Drew, D.A.; Graham, M.S.; Joshi, A.D.; Guo, C.G.; Ma, W.; Mehta, R.S.; Sikavi, D.R.; Lo, C.H.; Kwon, S.; et al. Risk of COVID-19 among frontline healthcare workers and the general community: A prospective cohort study. medRxiv 2020, arXiv:2020.04.29.20084111. [Google Scholar] [CrossRef]
- Maidstone, R.; Anderson, S.G.; Ray, D.W.; Rutter, M.K.; Durrington, H.J.; Blaikley, J.F. Shift work is associated with positive COVID-19 status in hospitalised patients. Thorax 2021, 76, 601–606. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Jiang, X.; Huang, Q.; Wang, H.; Gurarie, D.; Ndeffo-Mbah, M.; Fan, F.; Fu, P.; Horn, M.A.; Mondal, A.; et al. Risk factors of SARS-CoV-2 infection in healthcare workers: A retrospective study of a nosocomial outbreak. Sleep Med. X 2020, 2, 100028. [Google Scholar] [CrossRef]
- Istituto Superiore di Sanità. Epidemia. COVID-19 Integrated Surveillance Data in Italy. Available online: https://www.epicentro.iss.it/en/coronavirus/sars-cov-2-dashboard (accessed on 2 November 2021).
- EUROSTAT Statistics Explained. Healthcare Personnel Statistics—Nursing and Caring Professionals. Available online: https://ec.europa.eu/eurostat/statistics-explained/index.php?title=Healthcare_personnel_statistics_-_nursing_and_caring_professionals#Healthcare_personnel (accessed on 1 November 2021).
- EUROSTAT Data Browser. Health Personnel (Excluding Nursing and Caring Professionals). Available online: https://ec.europa.eu/eurostat/databrowser/view/hlth_rs_prs1/default/table?lang=en (accessed on 1 November 2021).
- CoronaVirus COVID-19. Dati Provincia di Modena. Available online: http://www.pangoo.it/coronavirus/?t=province&r=Modena&data=y#table (accessed on 1 November 2021).
- Modenese, A.; Mazzoli, T.; Berselli, N.; Ferrari, D.; Bargellini, A.; Borella, P.; Filippini, T.; Marchesi, I.; Paduano, S.; Vinceti, M.; et al. Frequency of Anti-SARS-CoV-2 Antibodies in Various Occupational Sectors in an Industrialized Area of Northern Italy from May to October 2020. Int. J. Environ. Res. Public Health 2021, 18, 7948. [Google Scholar] [CrossRef] [PubMed]
- Lopez Bernal, J.; Andrews, N.; Gower, C.; Robertson, C.; Stowe, J.; Tessier, E.; Simmons, R.; Cottrell, S.; Roberts, R.; O’Doherty, M.; et al. Effectiveness of the Pfizer-BioNTech and Oxford-AstraZeneca vaccines on covid-19 related symptoms, hospital admissions, and mortality in older adults in England: Test negative case-control study. BMJ 2021, 373, n1088. [Google Scholar] [CrossRef]
- Modenese, A.; Paduano, S.; Bargellini, A.; Bellucci, R.; Marchetti, S.; Bruno, F.; Grazioli, P.; Vivoli, R.; Gobba, F. Neutralizing Anti-SARS-CoV-2 Antibody Titer and Reported Adverse Effects, in a Sample of Italian Nursing Home Personnel after Two Doses of the BNT162b2 Vaccine Administered Four Weeks Apart. Vaccines 2021, 9, 652. [Google Scholar] [CrossRef] [PubMed]
- Simons, D.; Shahab, L.; Brown, J.; Perski, O. The association of smoking status with SARS-CoV-2 infection; hospitalization and mortality from COVID-19: A living rapid evidence review with Bayesian meta-analyses (version 7). Addiction 2021, 116, 1319–1368. [Google Scholar] [CrossRef] [PubMed]
- Vardavas, C.I.; Nikitara, K. COVID-19 and smoking: A systematic review of the evidence. Tob. Induc. Dis. 2020, 18, 20. [Google Scholar] [CrossRef] [PubMed]
- Gülsen, A.; König, I.R.; Jappe, U.; Drömann, D. Effect of comorbid pulmonary disease on the severity of COVID-19: A systematic review and meta-analysis. Respirology 2021, 26, 552–565. [Google Scholar] [CrossRef] [PubMed]
- Mendes, N.F.; Jara, C.P.; Mansour, E.; Araújo, E.P.; Velloso, L.A. Asthma and COVID-19: A systematic review. Allergy Asthma Clin. Immunol. 2021, 17, 5. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Ao, G.; Qi, X.; Xie, B. The association between COVID-19 and asthma: A systematic review and meta-analysis. Clin. Exp. Allergy 2020, 50, 1274–1277. [Google Scholar] [CrossRef]
- Deravi, N.; Fathi, M.; Vakili, K.; Yaghoobpoor, S.; Pirzadeh, M.; Mokhtari, M.; Fazel, T.; Ahsan, E.; Ghaffari, S. SARS-CoV-2 infection in patients with diabetes mellitus and hypertension: A systematic review. Rev. Cardiovasc. Med. 2020, 21, 385–397. [Google Scholar] [CrossRef] [PubMed]
- Tian, Y.; Qiu, X.; Wang, C.; Zhao, J.; Jiang, X.; Niu, W.; Huang, J.; Zhang, F. Cancer associates with risk and severe events of COVID-19: A systematic review and meta-analysis. Int. J. Cancer 2021, 148, 363–374. [Google Scholar] [CrossRef] [PubMed]
- Hughes, M.M.; Groenewold, M.R.; Lessem, S.E.; Xu, K.; Ussery, E.N.; Wiegand, R.E.; Qin, X.; Do, T.; Thomas, D.; Tsai, S.; et al. Update: Characteristics of health care personnel with COVID19—United States, February 12–July 16, 2020. MMWR Morb. Mortal. Wkly. Rep. 2020, 69, 1364–1368. [Google Scholar] [CrossRef]
- Jacob, J.T.; Baker, J.M.; Fridkin, S.K.; Lopman, B.A.; Steinberg, J.P.; Christenson, R.H.; King, B.; Leekha, S.; O’Hara, L.M.; Rock, P.; et al. Risk Factors Associated With SARS-CoV-2 Seropositivity Among US Health Care Personnel. JAMA Netw. Open 2021, 4, e211283. [Google Scholar] [CrossRef]
- Self, W.H.; Tenforde, M.W.; Stubblefield, W.B.; Feldstein, L.R.; Steingrub, J.S.; Shapiro, N.I.; Ginde, A.A.; Prekker, M.E.; Brown, S.M.; Peltan, I.D.; et al. Seroprevalence of SARS-CoV-2 among frontline health care personnel in a multistate hospital network—13 academic medical centers, April–June 2020. MMWR Morb. Mortal. Wkly. Rep. 2020, 69, 1221–1226. [Google Scholar] [CrossRef] [PubMed]
- Yoshikawa, M.; Asaba, K. Educational Attainment Decreases the Risk of COVID-19 Severity in the European Population: A Two-Sample Mendelian Randomization Study. Front. Public Health 2021, 9, 673451. [Google Scholar] [CrossRef] [PubMed]
- Loef, B.; van Baarle, D.; van der Beek, A.J.; Sanders, E.A.M.; Bruijning-Verhagen, P.; Proper, K.I. Shift Work and Respiratory Infections in Health-Care Workers. Am. J. Epidemiol. 2019, 188, 509–517. [Google Scholar] [CrossRef] [PubMed]
- Munter, G.; Levi-Vineberg, T.; Sylvetsky, N. Vitamin D deficiency among physicians: A comparison between hospitalists and community-based physicians. Osteoporos. Int. 2015, 26, 1673–1676. [Google Scholar] [CrossRef]
- Benskin, L.L. A Basic Review of the Preliminary Evidence That COVID-19 Risk and Severity Is Increased in Vitamin D Deficiency. Front. Public Health 2020, 8, 513. [Google Scholar] [CrossRef] [PubMed]
- Meltzer, D.O.; Best, T.J.; Zhang, H.; Vokes, T.; Arora, V.M.; Solway, J. Association of Vitamin D Levels, Race/Ethnicity, and Clinical Characteristics With COVID-19 Test Results. JAMA Netw. Open 2021, 4, e214117. [Google Scholar] [CrossRef] [PubMed]
| Considered Variables | University Hospital Workers Studied N (% of the Whole Sample) | SARS-CoV-2 Infection Diagnosis | p | ||
|---|---|---|---|---|---|
| No Infection N (%) | Infection N (%) | ||||
| Gender | Females | 4168 (70.7) | 3650 (87.6) | 518 (12.4) | 0.05 |
| Male | 1729 (29.3) | 1482 (85.7) | 247 (14.3) | ||
| Age classes (years) | ≤30 | 1312 (22.3) | 1153 (87.9) | 159 (12.1) | 0.11 |
| 31–40 | 1512 (25.6) | 1313 (86.8) | 199 (13.2) | ||
| 41–50 | 1318 (22.4) | 1130 (85.7) | 188 (12.3) | ||
| 51-60 | 1329 (22.5) | 1150 (86.5) | 178 (13.5) | ||
| >60 | 426 (7.2) | 386 (90.6) | 41 (9.4) | ||
| BMI (missing data = 1468) | Underweight | 228 (5.1) | 209 (91.7) | 19 (8.3) | 0.004 |
| Normal weight | 2784 (62.9) | 2445 (87.8) | 339 (12.2) | ||
| Overweight | 1019 (23.0) | 868 (85.2) | 151 (14.8) | ||
| Obese | 398 (9.0) | 328 (82.4) | 70 (17.6) | ||
| Smoking habit (missing data = 3033) | Non smoker | 2864 (74.1) | 2475 (86.4) | 389 (13.6) | 0.51 |
| Ex-smoker | 274 (7.1) | 238 (86.9) | 36 (13.1) | ||
| Current smokers | 728 (18.8) | 641 (88.0) | 87 (12.0) | ||
| Job category | non-HCW | 622 (10.6) | 583 (93.7) | 39 (6.3) | <0.0001 |
| Nurses & Nurse aides | 2603 (44.1) | 2129 (81.8) | 474 (18.2) | ||
| Physicians | 1790 (30.3) | 1587 (88.7) | 203 (11.3) | ||
| Other HCW | 882 (15.0) | 833 (94.4) | 49 (5.6) | ||
| Nightshifts at work | No | 4639 (78.7) | 4149 (89.4) | 490 (10.6) | <0.0001 |
| Yes | 1258 (21.3) | 983 (78.1) | 275 (21.9) | ||
| Presence of reported diseases (any) | No | 4707 (79.8) | 4104 (87.2) | 603 (12.8) | 0.47 |
| Yes | 1190 (20.2) | 1028 (85.7) | 162 (14.3) | ||
| Cardiovascular diseases (including hypertension) | No | 5492 (93.1) | 4785 (87.1) | 707 (12.9) | 0.40 |
| Yes | 405 (6.9) | 347 (85.7) | 58 (14.3) | ||
| Hypertension | No | 5573 (94.5) | 4852 (87.1) | 721 (12.9) | 0.73 |
| Yes | 324 (5.5) | 280 (86.4) | 44 (13.6) | ||
| Diabetes | No | 5818 (98.7) | 5062 (87.0) | 756 (13.0) | 0.67 |
| Yes | 79 (1.3) | 70 (88.6) | 9 (11.4) | ||
| Neoplastic diseases | No | 5672 (96.2) | 4931 (86.9) | 741 (13.1) | 0.29 |
| Yes | 225 (3.8) | 201 (89.3) | 24 (10.7) | ||
| Chronic respiratory diseases | No | 5817 (98.6) | 5070 (87.2) | 747 (12.8) | 0.02 |
| Yes | 80 (1.4) | 62 (77.5) | 18 (22.5) | ||
| Autoimmune diseases | No | 5435 (922) | 4730 (87.0) | 705 (13.0) | 1.00 |
| Yes | 462 (7.8) | 402 (87.0) | 60 (13.0) | ||
| Studied Variables | Adjusted Odds Ratio (95% Confidence Intervals) | ||||
|---|---|---|---|---|---|
| Model 1 | p | Model 2 | P | ||
| Gender | Females | ref. | 0.77 | ref. | 0.76 |
| Male | 1.03 (0.83–1.29) | 1.04 (0.83–1.29) | |||
| Age classes (years) | ≤30 | ref. | 0.73 | ref. | 0.77 |
| 31–40 | 1.20 (0.92–1.56) | 0.17 | 1.19 (0.92–1.55) | 0.18 | |
| 41–50 | 1.06 (0.80–1.43) | 0.69 | 1.07 (0.79–1.43) | 0.67 | |
| 51–60 | 1.06 (0.77–1.46) | 0.71 | 1.08 (0.79–1.48) | 0.61 | |
| >60 | 1.06 (0.65–1.72) | 0.82 | 1.09 (0.68–1.75) | 0.72 | |
| BMI | Underweight | 0.76 (0.45–1.26) | 0.28 | 0.75 (0.45–1.25) | 0.27 |
| Normal weight | ref. | 0.41 | ref. | 0.05 | |
| Overweight | 1.28 (1.01–1.62) | 0.04 | 1.27 (1.00–1.61) | 0.05 | |
| Obese | 1.41 (1.01–1.95) | 0.04 | 1.38 (1.00–1.92) | 0.06 | |
| Smoking habit | Non smoker | ref. | 0.02 | ref. | 0.01 |
| Ex-smoker | 0.79 (0.54–1.16) | 0.24 | 0.78 (0.53–1.15) | 0.20 | |
| Current smokers | 0.70 (0.54–0.91) | 0.008 | 0.70 (0.54–0.91) | 0.006 | |
| Job role | Non-HCW | ref. | <0.0001 | ref. | <0.0001 |
| Nurses and Nurse aides | 2.67 (1.63–4.37) | <0.0001 | 2.74 (1.67–4.50) | <0.0001 | |
| Physicians | 1.91 (1.16–3.14) | 0.01 | 1.94 (1.17–3.19) | 0.01 | |
| Other HCW | 0.50 (0.27–0.93) | 0.03 | 0.50 (0.27–0.93) | 0.03 | |
| Nightshifts at work | No | ref. | <0.0001 | ref. | <0.0001 |
| Yes | 1.80 (1.41–2.30) | 1.78 (1.39–2.28) | |||
| Presence of reported diseases (any) | No | ref. | 0.34 | / | |
| Yes | 1.13 (0.88–1.45) | ||||
| Chronic respiratory diseases | No | / | ref. | 0.001 | |
| Yes | 3.15 (1.64–6.05) | ||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Modenese, A.; Casolari, L.; Rossi, G.; Della Vecchia, E.; Glieca, F.; D’Elia, C.; Garavini, D.; Righi, E.; Mariani, S.; Venturelli, L.; et al. Factors Associated with SARS-CoV-2 Infection Risk among Healthcare Workers of an Italian University Hospital. Healthcare 2021, 9, 1495. https://doi.org/10.3390/healthcare9111495
Modenese A, Casolari L, Rossi G, Della Vecchia E, Glieca F, D’Elia C, Garavini D, Righi E, Mariani S, Venturelli L, et al. Factors Associated with SARS-CoV-2 Infection Risk among Healthcare Workers of an Italian University Hospital. Healthcare. 2021; 9(11):1495. https://doi.org/10.3390/healthcare9111495
Chicago/Turabian StyleModenese, Alberto, Loretta Casolari, Giorgia Rossi, Elena Della Vecchia, Francesca Glieca, Cristiana D’Elia, Denise Garavini, Elena Righi, Stefania Mariani, Luca Venturelli, and et al. 2021. "Factors Associated with SARS-CoV-2 Infection Risk among Healthcare Workers of an Italian University Hospital" Healthcare 9, no. 11: 1495. https://doi.org/10.3390/healthcare9111495
APA StyleModenese, A., Casolari, L., Rossi, G., Della Vecchia, E., Glieca, F., D’Elia, C., Garavini, D., Righi, E., Mariani, S., Venturelli, L., Vivoli, D., & Gobba, F. (2021). Factors Associated with SARS-CoV-2 Infection Risk among Healthcare Workers of an Italian University Hospital. Healthcare, 9(11), 1495. https://doi.org/10.3390/healthcare9111495









