Nutritional Intake and Meal Composition of Patients Consuming Texture Modified Diets and Thickened Fluids: A Systematic Review and Meta-Analysis
Abstract
1. Introduction
2. Materials and Methods
2.1. Selection Criteria
2.2. Data Sources
2.3. Data Collection and Analysis
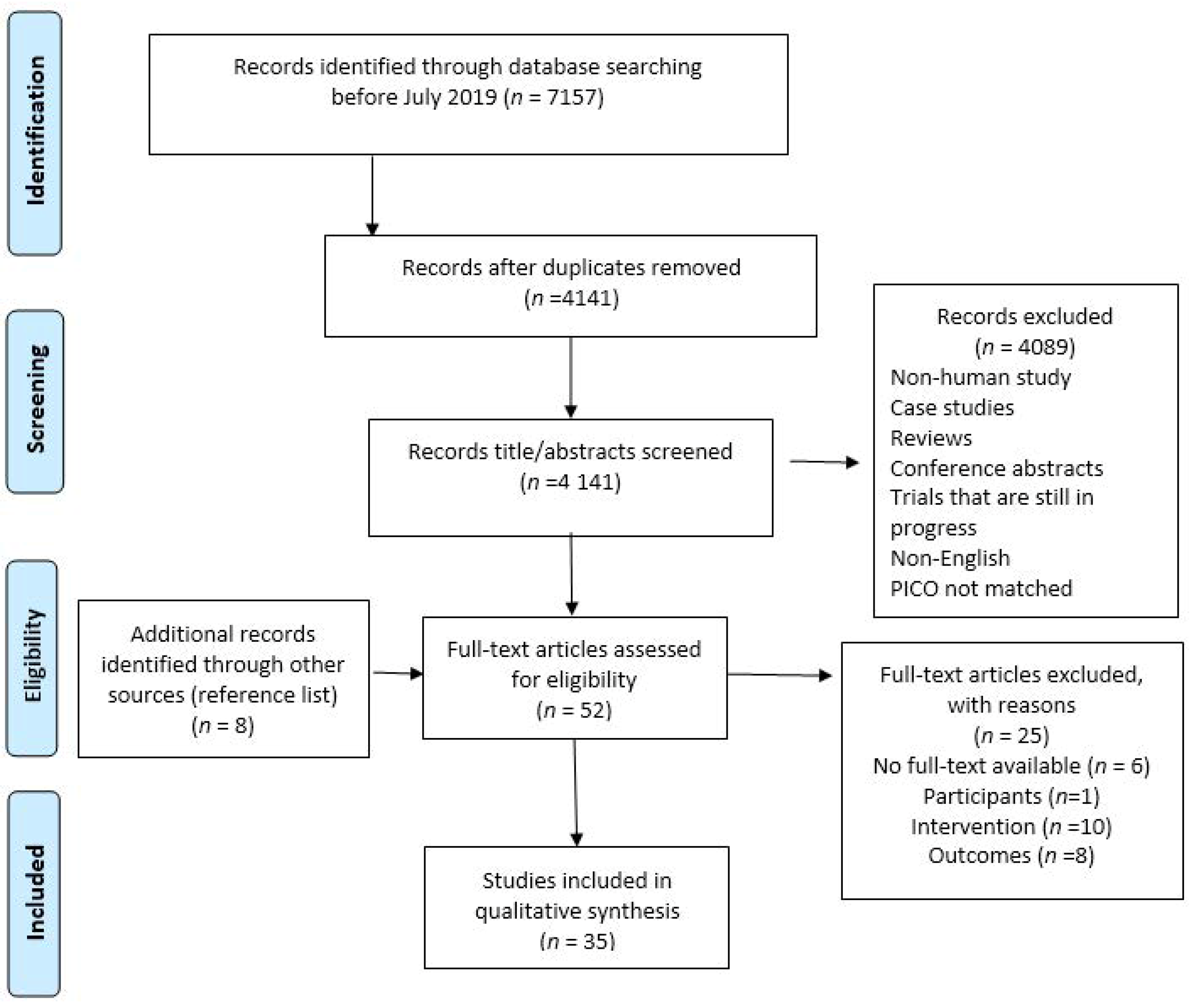
3. Results
3.1. Nutrition Intake
3.1.1. TMDs and Regular Diet
3.1.2. Thickened Fluids
3.1.3. Texture and/or Nutrition Enhanced TMDs and Traditional Cook-Fresh TMDs
3.2. Meal Consumption
3.2.1. TMDs and Regular Diet
3.2.2. Texture and/or Nutrition Enhanced and Traditional Cook-Fresh TMDs
3.3. Nutrition Adequacy
3.3.1. TMDs and Regular Diet
3.3.2. Texture and/or Nutrition Enhanced TMDs and Traditional Cook-Fresh TMDs
3.4. Nutrition Content of the Meal
3.4.1. TMDs and Regular Diet
3.4.2. Texture and/or Nutrition Enhanced TMDs and Traditional TMDs
3.5. Texture and Consistency
3.6. Quality Assessment
4. Discussion
4.1. Nutrition Intake and Meal Consumption
4.2. Nutrition Content
4.3. Meal Texture
4.4. Study Implications
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Clavé, P.; Shaker, R. Dysphagia: Current reality and scope of the problem. Nat. Rev. Gastroenterol. Hepatol. 2015, 12, 259. [Google Scholar] [CrossRef]
- Serra-Prat, M.; Hinojosa, G.; Lõpez, D.; Juan, M.; Fabré, E.; Voss, D.S.; Calvo, M.; Marta, V.; Ribõ, L.; Palomera, E.; et al. Prevalence of oropharyngeal dysphagia and impaired safety and efficacy of swallow in independently living older persons. J. Am. Geriatr. Soc. 2011, 59, 186–187. [Google Scholar] [CrossRef]
- Namasivayam, A.M.; Steele, C.M. Malnutrition and Dysphagia in Long-Term Care: A Systematic Review. J. Nutr. Gerontol. Geriatr. 2015, 34, 1–21. [Google Scholar] [CrossRef]
- Rothenberg, E.; Wendin, K. 7—Texture Modification of Food for Elderly People; Elsevier Ltd.: Amsterdam, The Netherlands, 2015; Volume 2, ISBN 9781782423522. [Google Scholar]
- Espinosa-Val, M.C.; Martín-Martínez, A.; Graupera, M.; Arias, O.; Elvira, A.; Cabré, M.; Palomera, E.; Bolívar-Prados, M.; Clavé, P.; Ortega, O. Prevalence, Risk Factors, and Complications of Oropharyngeal Dysphagia in Older Patients with Dementia. Nutrients 2020, 12, 863. [Google Scholar] [CrossRef]
- Wirth, R.; Pourhassan, M.; Streicher, M.; Hiesmayr, M.; Schindler, K. The Impact of Dysphagia on Mortality of Nursing Home Residents: Results From the nutritionDay Project. J. Am. Med. Dir. Assoc. 2018, 19, 775–778. [Google Scholar] [CrossRef]
- Martín, A.; Ortega, O.; Roca, M.; Arús, M.; Clavé Civit, P. Effect of a Minimal-Massive Intervention in Hospitalized Older Patients with Oropharyngeal Dysphagia: A Proof of Concept Study. J. Nutr. Health Aging 2018, 22, 739–747. [Google Scholar] [CrossRef]
- Baijens, L.W.J.; Clavé, P.; Cras, P.; Ekberg, O.; Forster, A.; Kolb, G.F.; Leners, J.C.; Masiero, S.; Mateos-Nozal, J.; Ortega, O.; et al. European society for swallowing disorders-European union geriatric medicine society white paper: Oropharyngeal dysphagia as a geriatric syndrome. Clin. Interv. Aging 2016, 11, 1403. [Google Scholar] [CrossRef] [PubMed]
- Robbins, J.; Nicosia, M.; Hind, J.A.; Gill, G.D.; Blanco, R.; Logemann, J. Defining Physical Properties of Fluids for Dysphagia Evaluation and Treatment. Perspect. Swallowing Swallowing Disord. 2002. [Google Scholar] [CrossRef]
- Garcia, J.M.; Chambers, E. Managing dysphagia through diet modifications. Am. J. Nurs. 2010. [Google Scholar] [CrossRef] [PubMed]
- Cichero, J.A.Y. Adjustment of Food Textural Properties for Elderly Patients. J. Texture Stud. 2016, 47, 277–283. [Google Scholar] [CrossRef]
- Steele, C.M.; Alsanei, W.A.; Ayanikalath, S.; Barbon, C.E.A.A.; Chen, J.; Cichero, J.A.Y.Y.; Coutts, K.; Dantas, R.O.; Duivestein, J.; Giosa, L.; et al. The Influence of Food Texture and Liquid Consistency Modification on Swallowing Physiology and Function: A Systematic Review. Dysphagia 2015, 30, 2–26. [Google Scholar] [CrossRef] [PubMed]
- Curran, J.; Groher, M.E. Development and dissemination of an aspiration risk reduction diet. Dysphagia 1990, 5, 6–12. [Google Scholar] [CrossRef] [PubMed]
- Bannerman, E.; McDermott, K. Dietary and fluid intakes of older adults in care homes requiring a texture modified diet: The role of snacks. J. Am. Med. Dir. Assoc. 2011, 12, 234–239. [Google Scholar] [CrossRef] [PubMed]
- Keller, H.H.; Chambers, L.; Niezgoda, H.; Duizer, L. Issues associated with the use of modified texture foods. J. Nutr. Health Aging 2012, 16, 195–200. [Google Scholar] [CrossRef] [PubMed]
- Colodny, N. Dysphagic independent feeders’ justifications for noncompliance with recommendations by a speech-language pathologist. Am. J. Speech Language Pathol. 2005. [Google Scholar] [CrossRef]
- Macqueen, C.E.; Taubert, S.; Cotter, D.; Stevens, S.; Frost, G.S. Which commercial thickening agent do patients prefer? Dysphagia 2003, 18, 46–52. [Google Scholar] [CrossRef]
- Eglseer, D.; Halfens, R.J.G.; Schols, J.M.G.A.; Lohrmann, C. Dysphagia in Hospitalized Older Patients: Associated Factors and Nutritional Interventions. J. Nutr. Health Aging 2018, 22, 103–110. [Google Scholar] [CrossRef]
- Miles, A.; Liang, V.; Sekula, J.; Broadmore, S.; Owen, P.; Braakhuis, A.J. Texture-modified diets in aged care facilities: Nutrition, swallow safety and mealtime experience. Australas. J. Ageing 2019. [Google Scholar] [CrossRef]
- Martineau, C. International Dysphagia Diet Standardisation Initiative: IDDSI Framework. Med. Mal. Metab. 2015. [Google Scholar] [CrossRef]
- Davis, L.A.; Spicer, M.T. Nutrition and Dysphagia in Older Adults. Top. Geriatr. Rehabil. 2007, 23, 211–219. [Google Scholar] [CrossRef]
- Niezgoda, H.; Project, M.-T.F. Issues and Challenges of Modified- Texture Foods in Long-Term Care: A Workshop Report. Ann. Long Term Care Clin. Care Aging 2012, 20, 22–27. [Google Scholar]
- Andersen, U.T.; Beck, A.M.; Kjaersgaard, A.; Hansen, T.; Poulsen, I. Systematic review and evidence based recommendations on texture modified foods and thickened fluids for adults (≥18 years) with oropharyngeal dysphagia. e-SPEN J. 2013. [Google Scholar] [CrossRef]
- Min, C.; Park, B.; Sim, S.; Choi, H.G. Dietary modification for laryngopharyngeal reflux: Systematic review. J. Laryngol. Otol. 2019, 133, 80–86. [Google Scholar] [CrossRef] [PubMed]
- Sodeman, W.A.; Sodeman, T.C. Diets. In Instructions for Geriatric Patients, 3rd ed.; Sodeman, W.A., Sodeman, T.C., Eds.; Saunders: Philadelphia, PA, USA, 2005; pp. 267–315. ISBN 978-1-4160-0203-1. [Google Scholar]
- Foeckler, P.; Henning, V.; Reichelt, J. Mendeley [Computer Program]; Version 1.19.4; Elsevier: London, UK, 2019. [Google Scholar]
- Microsoft Excel for Office 365 [Computer program]; Version 1902; Microsoft Corp: Redmond, WA, USA, 2019.
- Higgins, J.P.T.; Thompson, S.G.; Deeks, J.J.; Altman, D.G. Measuring inconsistency in meta-analyses. BMJ 2003, 327, 557–560. [Google Scholar] [CrossRef] [PubMed]
- Higgins, J.; Thompson, S.; Deeks, J.; Altman, D. Review article Statistical heterogeneity in systematic reviews of clinical trials: A critical appraisal of guidelines and practice. J. Health Serv. Res. Policy 2002, 7, 51–61. [Google Scholar] [CrossRef] [PubMed]
- Dekkers, O.M.; Vandenbroucke, J.P.; Cevallos, M.; Renehan, A.G.; Altman, D.G.; Egger, M. COSMOS-E: Guidance on conducting systematic reviews and meta-analyses of observational studies of etiology. PLoS Med. 2019, 16, e1002742. [Google Scholar] [CrossRef]
- The Nordic Cochrane Centre. Review Manager (Rev Man) [Computer Program]; Version 5.3; Copenhagen Nord. Cochrane Centre, Cochrane Collab: København, Danmark, 2014. [Google Scholar]
- Morgan, R.L.; Thayer, K.A.; Santesso, N.; Holloway, A.C.; Blain, R.; Eftim, S.E.; Goldstone, A.E.; Ross, P.; Guyatt, G.; Schünemann, H.J. Evaluation of the risk of bias in non-randomized studies of interventions (ROBINS-I) and the ‘target experiment’ concept in studies of exposures: Rationale and preliminary instrument development. Environ. Int. 2018, 120, 382–387. [Google Scholar] [CrossRef]
- Greenwood, D.C. Meta-analysis of observational studies. Mod. Methods Epidemiol. 2012, 173–189. [Google Scholar] [CrossRef]
- IOM. Institute of Medicine of the National Academies. Dietary Reference Intakes: The Essential Guide to Nutrient Requirements; National Academies Press: Washington, DC, USA, 2006; ISBN 0-309-65646-X. [Google Scholar]
- Adolphe, J.L.; Whiting, S.J.; Dahl, W.J. Vitamin fortification of pureed foods for long-term care residents. Can. J. Diet. Pract. Res. 2009, 70, 143–150. [Google Scholar] [CrossRef][Green Version]
- Beck, A.M.; Hansen, K.S. Meals served in Danish nursing homes and to meals-on-wheels clients may not offer nutritionally adequate choices. J. Nutr. Elder. 2010, 29, 100–109. [Google Scholar] [CrossRef]
- Cassens, D.; Johnson, E.; Keelan, S. Enhancing taste, texture, appearance, and presentation of pureed food improved resident quality of life and weight status. Nutr. Rev. 1996, 54, S51–S54. [Google Scholar] [CrossRef] [PubMed]
- Dahl, W.J.; Whiting, S.J.; Tyler, R.T. Protein content of pureed diets: Implications for planning. Can. J. Diet. Pract. Res. 2007, 68, 99–102. [Google Scholar] [CrossRef] [PubMed]
- de Sá, J.S.M.; Moreira, D.C.F.; Louvera Silva, K.A.; Morgano, M.A.; Quintaes, K.D.K.D.; de Sa, J.S.M.; Moreira, D.C.F.; Louvera Silva, K.A.; Morgano, M.A.; Quintaes, K.D.K.D.; et al. Consumption of oral hospital diets and percent adequacy of minerals in oncology patients as an indicative for the use of oral supplements. Clin. Nutr. 2014, 33, 655–661. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Durant, M. A comparison of Energy Provision by Diet Order in a Long-Term Care Facility. Can. J. Aging La Rev. Can. du Vieil. 2008, 27, 225–227. [Google Scholar] [CrossRef]
- Farrer, O.; Olsen, C.; Mousley, K.; Teo, E. Does presentation of smooth pureed meals improve patients consumption in an acute care setting: A pilot study. Nutr. Diet. 2016, 73, 405–409. [Google Scholar] [CrossRef]
- Finestone, H.M.; Foley, N.C.; Woodbury, M.G.; Greene-Finestone, L. Quantifying fluid intake in dysphagic stroke patients: A preliminary comparison of oral and nonoral strategies. Arch. Phys. Med. Rehabil. 2001, 82, 1744–1746. [Google Scholar] [CrossRef]
- Foley, N.; Finestone, H.; Woodbury, M.G.; Teasell, R.; Greene-Finestone, L. Energy and protein intakes of acute stroke patients. J. Nutr. Health Aging 2006, 10, 171–175. [Google Scholar]
- Garon, B.R.; Engle, M.; Ormiston, C. A randomized control study to determine the effects of unlimited oral intake of water in patients with identified aspiration. J. Neurol. Rehabil. 1997, 11, 139–148. [Google Scholar] [CrossRef]
- Germain, I.; Dufresne, T.; Gray-Donald, K. A novel dysphagia diet improves the nutrient intake of institutionalized elders. J. Am. Diet. Assoc. 2006, 106, 1614–1623. [Google Scholar] [CrossRef]
- Higashiguchi, T. Novel diet for patients with impaired mastication evaluated by consumption rate, nutrition intake, and questionnaire. Nutrition 2013, 29, 858–864. [Google Scholar] [CrossRef]
- Johnson, R.M.; Smiciklas-Wright, H.; Soucy, I.M. Nutrient intake of nursing-home residents receiving pureed foods or a regular diet. J. Am. Geriatr. Soc. 1995, 43, 344–348. [Google Scholar] [CrossRef] [PubMed]
- Karagiannis, M.J.P.; Chivers, L.; Karagiannis, T.C. Effects of oral intake of water in patients with oropharyngeal dysphagia. BMC Geriatr. 2011, 11, 9. [Google Scholar] [CrossRef] [PubMed]
- Keller, H.H.; Chambers, L.W.; Fergusson, D.A.; Niezgoda, H.; Parent, M.; Caissie, D.; Lemire, N. A mix of bulk and ready-to-use modified-texture food: Impact on older adults requiring dysphagic food. Can. J. Aging 2012, 31, 335–348. [Google Scholar] [CrossRef] [PubMed]
- Kennewell, S.; Kokkinakos, M. Thick, cheap and easy: Fortifying texture-modified meals with infant cereal. Nutr. Diet. 2007, 64, 112–115. [Google Scholar] [CrossRef]
- Massoulard, A.; Bonnabau, H.; Gindre-Poulvelarie, L.; Baptistev, A.; Preux, P.M.; Villemonteix, C.; Javerliat, V.; Fraysse, J.L.; Desport, J.C. Analysis of the food consumption of 87 elderly nursing home residents, depending on food texture. J. Nutr. Health Aging 2011, 15, 192–195. [Google Scholar] [CrossRef] [PubMed]
- McCormick, S.E.; Stafford, K.M.; Saqib, G.; Chroinin, D.N.; Power, D. The efficacy of pre-thickened fluids on total fluid and nutrient consumption among extended care residents requiring thickened fluids due to risk of aspiration. Age Ageing 2008, 37, 714–715. [Google Scholar] [CrossRef]
- Moreira, D.C.F.; de Sá, J.S.M.; Cerqueira, I.B.; Oliveira, A.P.F.; Morgano, M.A.; Quintaes, K.D. Evaluation of iron, zinc, copper, manganese and selenium in oral hospital diets. Clin. Nutr. 2014, 33, 808–814. [Google Scholar] [CrossRef]
- Nowson, C.; Sherwin, A.J.; McPhee, J.G.; Wark, J.D.; Flicker, L. Energy, protein, calcium, vitamin D and fibre intakes from meals in residential care establishments in Australia. Asia Pac. J. Clin. Nutr. 2003, 12, 172–177. [Google Scholar]
- Ott, A.; Senger, M.; Lötzbeyer, T.; Gefeller, O.; Sieber, C.C.; Volkert, D. Effects of a Texture-Modified, Enriched, and Reshaped Diet on Dietary Intake and Body Weight of Nursing Home Residents with Chewing and/or Swallowing Problems: An Enable Study. J. Nutr. Gerontol. Geriatr. 2019, 38, 361–376. [Google Scholar] [CrossRef]
- Philip, K.E.; Greenwood, C.E. Nutrient contribution of infant cereals used as fluid thickening agents in diets fed to the elderly. J. Am. Diet. Assoc. 2000, 100, 549–554. [Google Scholar] [CrossRef]
- Reyes-Torres, C.A.; Castillo-Martínez, L.; Reyes-Guerrero, R.; Ramos-Vázquez, A.G.; Zavala-Solares, M.; Cassis-Nosthas, L.; Serralde-Zúñiga, A.E.; Reyes-Torres, C.A.; Reyes-Guerrero, R.; Ramos-Vazquez, A.G.; et al. Design and implementation of modified-texture diet in older adults with oropharyngeal dysphagia: A randomized controlled trial. Eur. J. Clin. Nutr. 2019. [Google Scholar] [CrossRef] [PubMed]
- Rosenvinge, S.K.; Starke, I.D. Improving care for patients with dysphagia. Age Ageing 2005, 34, 587–593. [Google Scholar] [CrossRef] [PubMed]
- Sherwin, A.J.; Nowson, C.A.; McPhee, J.; Alexander, J.L.; Wark, J.D.; Flicker, L. Nutrient intake at meals in residential care facilities for the aged: Validated visual estimation of plate waste. Aust. J. Nutr. Diet. 1998, 55, 188–193. [Google Scholar]
- Taylor, K.A.; Barr, S.I. Provision of small, frequent meals does not improve energy intake of elderly residents with dysphagia who live in an extended-care facility. J. Am. Diet. Assoc. 2006, 106, 1115–1118. [Google Scholar] [CrossRef]
- Torrence, S.E. Pureed Diets in a Long-Term Care Setting: Does Use of Pre-Shaped Pureed Foods Increase Consumption? Northen Illinois University: DeKalb, IL, USA, 2011. [Google Scholar]
- Vivanti, A.P.; Campbell, K.L.; Suter, M.S.; Hannan-Jones, M.T.; Hulcombe, J.A. Contribution of thickened drinks, food and enteral and parenteral fluids to fluid intake in hospitalised patients with dysphagia. J. Hum. Nutr. Diet. 2009, 22, 148–155. [Google Scholar] [CrossRef]
- Vucea, V.; Keller, H.H.; Morrison, J.M.; Duncan, A.M.; Duizer, L.M.; Carrier, N.; Lengyel, C.O.; Slaughter, S.E. Nutritional quality of regular and pureed menus in Canadian long term care homes: An analysis of the Making the Most of Mealtimes (M3) project. BMC Nutr. 2017, 3, 80. [Google Scholar] [CrossRef]
- Welch, P.K.; Dowson, M.; Endres, J.M. The effect of nutrient supplements on high risk long term care residents receiving pureed diets. J. Nutr. Elder. 1991, 10, 49–62. [Google Scholar] [CrossRef]
- Whelan, K. Inadequate fluid intakes in dysphagic acute stroke. Clin. Nutr. 2001, 20, 423–428. [Google Scholar] [CrossRef]
- Wright, L.; Cotter, D.; Hickson, M.; Frost, G. Comparison of energy and protein intakes of older people consuming a texture modified diet with a normal hospital diet. J. Hum. Nutr. Diet. 2005, 18, 213–219. [Google Scholar] [CrossRef]
- Zanini, M.; Bagnasco, A.; Catania, G.; Aleo, G.; Sartini, M.; Cristina, M.L.; Ripamonti, S.; Monacelli, F.; Odetti, P.; Sasso, L. A Dedicated Nutritional Care Program (NUTRICARE) to reduce malnutrition in institutionalised dysphagic older people: A quasi-experimental study. J. Clin. Nurs. 2017, 26, 4446–4455. [Google Scholar] [CrossRef]
- Sterne, J.A.C.; Sutton, A.J.; Ioannidis, J.P.A.; Terrin, N.; Jones, D.R.; Lau, J.; Carpenter, J.; Rücker, G.; Harbord, R.M.; Schmid, C.H.; et al. Recommendations for examining and interpreting funnel plot asymmetry in meta-analyses of randomised controlled trials. BMJ 2011, 343, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Roth, D.; Ritchie, C.; Burgio, K.; Locher, J.L. Reliability and Predictive Validity of Caloric Intake Measures from the 24-Hour Dietary Recalls of Homebound Older Adults. J. Am. Diet. Assoc. 2010, 5, 773–778. [Google Scholar] [CrossRef]
- Rodrigues-Fisher, L.; Bourguignon, C.; Good, B.V. Dietary Fiber Nursing Intervention: Prevention of Constipation in Older Adults. Clin. Nurs. Res. 1993. [Google Scholar] [CrossRef]
- Perry, H.M.; Horowitz, M.; Morley, J.E.; Patrick, P.; Vellas, B.; Baumgartner, R.; Garry, P.J. Longitudinal changes in serum 25-hydroxyvitamin D in older people. Metabolism 1999. [Google Scholar] [CrossRef]
- Morley, J.E. Undernutrition in older adults. Fam. Pract. 2012. [Google Scholar] [CrossRef] [PubMed]
- Lengyel, C.O.; Whiting, S.J.; Zello, G.A. Nutrient inadequacies among elderly residents of long-term care facilities. Can. J. Diet. Pract. Res. 2008, 69, 82–88. [Google Scholar] [CrossRef] [PubMed]
- Hubbard, G.P.; Elia, M.; Holdoway, A.; Stratton, R.J. A systematic review of compliance to oral nutritional supplements. Clin. Nutr. 2012, 31, 293–312. [Google Scholar] [CrossRef]
- Nieuwenhuizen, W.F.; Weenen, H.; Rigby, P.; Hetherington, M.M. Older adults and patients in need of nutritional support: Review of current treatment options and factors influencing nutritional intake. Clin. Nutr. 2010, 29, 160–169. [Google Scholar] [CrossRef]
- Wright, L.; Cotter, D.; Hickson, M. The effectiveness of targeted feeding assistance to improve the nutritional intake of elderly dysphagic patients in hospital. J. Hum. Nutr. Diet. 2008, 21, 555–562. [Google Scholar] [CrossRef]
- Gall, M.J.; Grimble, G.K.; Reeve, N.J.; Thomas, S.J. Effect of providing fortified meals and between-meal snacks on energy and protein intake of hospital patients. Clin. Nutr. 1998. [Google Scholar] [CrossRef]
- Cichero, J.A.Y.; Lam, P.; Steele, C.M.; Hanson, B.; Chen, J.; Dantas, R.O.; Duivestein, J.; Kayashita, J.; Lecko, C.; Murray, J.; et al. Development of International Terminology and Definitions for Texture-Modified Foods and Thickened Fluids Used in Dysphagia Management: The IDDSI Framework. Dysphagia 2017. [Google Scholar] [CrossRef] [PubMed]
- Sura, L.; Madhavan, A.; Carnaby, G.; Crary, M.A. Dysphagia in the elderly: Management and nutritional considerations. Clin. Interv. Aging 2012, 7, 287. [Google Scholar] [PubMed]
- Cichero, J.A.Y.; Steele, C.; Duivestein, J.; Clavé, P.; Chen, J.; Kayashita, J.; Dantas, R.; Lecko, C.; Speyer, R.; Lam, P.; et al. The Need for International Terminology and Definitions for Texture-Modified Foods and Thickened Liquids Used in Dysphagia Management: Foundations of a Global Initiative. Curr. Phys. Med. Rehabil. Rep. 2013. [Google Scholar] [CrossRef] [PubMed]
- Côté, C.; Giroux, A.; Villeneuve-Rhéaume, A.; Gagnon, C.; Germain, I. Is iddsi an evidence-based framework? A relevant question for the frail older population. Geriatrics 2020, 5, 82. [Google Scholar] [CrossRef] [PubMed]
- Wirth, R.; Dziewas, R.; Beck, A.M.; Clavé, P.; Hamdy, S.; Heppner, H.J.; Langmore, S.; Leischker, A.H.; Martino, R.; Pluschinski, P.; et al. Oropharyngeal dysphagia in older persons—From pathophysiology to adequate intervention: A review and summary of an international expert meeting. Clin. Interv. Aging. 2016, 11, 189–208. [Google Scholar] [CrossRef] [PubMed]
- Arahata, M.; Oura, M.; Tomiyama, Y.; Morikawa, N.; Fujii, H.; Minani, S.; Shimizu, Y. A comprehensive intervention following the clinical pathway of eating and swallowing disorder in the elderly with dementia: Historically controlled study. BMC Geriatr. 2017, 17, 1–11. [Google Scholar] [CrossRef]
- Ullrich, S.; Buckley, J.; Crichton, J.; Esterman, A. An Exploratory Study of the Mealtime Experience of Older People With Dysphagia. J. Aging Res. Clin. Pract. 2014, 3, 229–236. [Google Scholar]
| Source | Method | Setting, Origin | Patient Characteristics | Interventions | Comparator/Control | Outcomes | Quality Assessment |
|---|---|---|---|---|---|---|---|
| Adolphe et al. (2009) [35] | Pre-post Experimental 8 weeks | LTC Canada | Residents ≥ 50 y consumed pureed diet daily Ex: Palliative Mean BMI (kg/m2) 25 | Vitamin-fortified pureed diets (lunch, dinner) n = 10 | Unfortified pureed diet n = 10 | -Vitamin intake (3-day weighed food record) -Adequacy (USA 2003) | Neutral |
| Bannerman and McDermott (2011) [14] | Observational Cross-sectional | 3 LTCs Scotland | Residents >60 y Ex: Nil by mouth, receiving artificial nutritional support, fluid restriction, acutely unwell, palliative Mean age (y) 88.1 ± 5.4 | Texture C–Thicker pureed: n = 11 Texture D–Minced/moist: n = 4 [UK national descriptors 2009] | Regular diet n = 15 | -Macronutrient and fluid intake (3-day weighed food record) -Adequacy (UK 1991) -Meal compliance -Role of snacks Snacks and fluid included | Neutral |
| Beck and Hansen (2010) [36] | Observational Cross-sectional | Meals-On-Wheels and LTCs Denmark | Diet orders from 10 Kitchens preparing meals for LTCs and Meals-On-Wheels | Chopped diet n = 9 Blended diet n = 6 | Regular diet n = 10 | -Nutrient content | Neutral |
| Cassen et al. (1996) [37] | Pre-post Experimental 16 days | LTC US | All residents consumed pureed diet Ex: discharged or passed away | 3D shaped pureed diet n = 18 | Unmodified pureed diet n = 18 | -Energy and protein intake (clinical record) -Meal consumption | Neutral |
| Dahl et al. (2007) [38] | Observational Cross-sectional | 8 LTCs Canada | Residents consumed pureed diet | None | Pureed diet n = 20 | -Energy and protein intake (3 to 5-day weighed food record) -Adequacy (USA 2003) -Nutrient content ONS and snacks included | Neutral |
| De Sa et al. (2014) [39] | Observational Cross-sectional | Oncology hospital Brazil | Oncology patients (admitted for surgery n = 83, intercurrence n = 58, chemotherapy n = 21, radiotherapy n = 1), mean stay 6.3 ±1.2 days) Mean age (y) 57 ± 15 | Regular + OFC n = 29 Soft + OFC n = 4 Blend + OFC n = 8 | Regular n = 97 Soft n = 6 Blend n = 19 | -Mineral adequacy (snacks included) -Meal consumption (plate wastage) -Supplement acceptance and content Snacks included | Neutral |
| Durant (2008) [40] | Observational Cross-sectional | LTC Canada | Diet orders from regular or pureed diets over 5-week menu | Pureed with thin fluids n = 9 | Regular diet n = 19 | -Nutrition content (duplicate meal trays) ONS excluded | Neutral |
| Farrer et al. (2016) [41] | Pre-post Experimental 2 weeks | Hospital Australia | All patients > 18 y consuming pureed diet in any acute care units (Cancer, n = 17, Parkinson n = 3, Cerebral palsy n = 3, Respiratory n = 10, Head trauma n = 5, Other n = 27) (reasons for pureed diet: dysphagia n = 40, comfort n = 6, temporary swallowing difficulty n = 18) 70% ≥ 65 y | Moulded pureed diet (Texture C) n = 27 | Unmodified pureed diet (Texture C) n = 38 | -Meal consumption (plate wastage) | Neutral |
| Finestone et al. (2001) [42] | Observational Cohort 21 days | Hospital Canada | Dysphagic patients admitted to hospital within 5-days of onset of first stroke (evaluated by SLT) Mean age (y) 66.1 ± 13.5 | Dysphagia diets (chopped/minced/pureed) + TF n = 6 | Started with enteral feed/IV fluids, progress to dysphagia diet +TF after 7–9 days n = 7 | -Fluid intake (2-day oral food and fluid intake record/5-day fluid balance sheets of enteral feed) -Fluid Adequacy (PENG 2004) Solid excluded | Positive |
| Foley et al. (2006) [43] | Observational Cohort 21 days (data collection at day 1, 7, 11, 14, 21) | Hospital neurological unit UK | Well-nourished acute stroke patients (evaluated by SLT) Mean age (y) 69 ±11.3 | Dysphagia diets n = 11–20 | Regular diet n = 25–48 Enteral feed n = 11–20 | -Nutrition intake (2-day of calorie counts by portion for oral diets and fluid balance sheets for enteral feed) -Adequacy (indirect calorimetry) | Neutral |
| Garon et al. (1997) [44] | RCT 1 year | Hospital stroke rehabilitation UK | Stroke patients with previously identified thin fluid aspiration by videofluoroscopy Mean age (y) 76.8 | TFs + free access of water n = 10 | TFs only n = 10 | -Fluid intake (Daily flowcharts) Solid excluded | Positive |
| Germain et al. (2006) [45] | RCT 12 weeks | LTC Canada | Residents aged 65–90 y admitted ≥3 m and had >7.5% weight loss in the last 3 m or BMI < 24 with dysphagia evaluated by RIC tool (Alzheimer n = 8, Dementia n = 6, Stroke =2, Parkinson n = 1) Ex. Cancer, chronic intestinal disease, terminally ill patients Mean age (y) 59 | Shaped minced, minced/pureed or pureed diet and consistency-controlled TFs using Bostwick consistometer (nectar, honey, pudding) n = 9 | Unmodified minced−70, minced−3 or pureed diet and honey level TF (consistency not systematically controlled) n = 8 | - Macro- and micronutrient intake (2-day weighed food record) -Adequacy Snacks and ONS included | Neutral |
| Higashiguchi (2013) [46] | Experimental Cohort 7 days | 17 hospital/LTC Japan | Inpatient with mastication difficulty under nutritional management and residents on TMDs with inadequate consumption (Stroke n = 19, Cancer n = 9, Heart failure n = 7, Fracture n = 5, Dehydration n = 4, Pressure ulcers, n = 3, Pneumonia n = 2, Anemia n = 2, COPD n = 2, Dementia n = 2, Diabetes n = 1, Parkinson n = 1, Other n = 17, None n = 2) (require total meal assistance n = 17, partial n = 6, none =34) Mean age (y) 81.6 ± 9.3 Mean BMI (kg/m2) 18.8 ± 0.34 | 3 days of nutrient -dense (Enzyme-infused) TMDs (enzyme was evenly infused into the ingredients to adjust the homogeneous softness of the meal instead of adding water for softening) Nutrients were not diluted and volume not increased n = 55 | 4 days of unmodified TMDs | -Macronutrient and sodium intake (mean meals weighed food record) -Meal consumption -Adequacy -Nutrient content (weighed plate wastage) Mean weight of the modified food is lower than unmodified | Positive |
| Johnson et al. (1995) [47] | Observational Cross-sectional | LTC US | 70 randomly selected female residents ≥65 y admitted for ≥6 m consuming food orally Mean age (y) 85 | Pureed, n = 20 | Regular diet, n = 31 | - Macro- and micronutrient intake (7-day food record by consumption monitoring system) -Adequacy (US 1989) -Nutrition content Snacks and ONS included | Neutral |
| Karagiannis et al. (2011) [48] | RCT 8 days | Hospital subacute units Australia | Patients ≥18 y aspirated on thin liquids with prescription of modified or TF diet by SLTs without chronic respiratory conditions or prior tracheostomy (Stroke n = 40, Dementia n = 11, Alzheimer n = 7, Parkinson n = 5, Cancer n = 10, Motor neuro disease n = 1, Huntington n = 1, accident n = 1) Mean age (y) 79.5 | TMDs (Pureed; Minced; Soft/Minced) + TF (Honey; Pudding; Nectar) + free access of water n = 42 | TMDs + TF n = 34 | -Fluid intake (Total daily oral liquid record) Solid excluded | Positive |
| Keller et al. (2012) [49] | Pre-post Experimental 9 m | Hospital and LTC Canada | All dysphagic residents fully consumed pureed or minced diets (Stroke, Parkinson, Dementia) Ex. enteral feed Facility avg. age 67 and 82 yr | 6 m of mix of 61% bulk and 39% shaped ready-to-use (reduced nutrients dilution and easier to chew and swallow) commercial TMDs n = 40 | 3 m of bulk commercial TMDs (unshaped, packaged in bulk) n = 40 | - Macro- and micronutrient intake (4 and 6-day of lunch/dinner food record by estimation) -Meal consumption (visual estimation of plate wastage) -Nutrient content Snacks and ONS excluded | Positive |
| Kennewell and Kokkinakos (2007) [50] | Observational Cross-sectional | 2 hospitals Australia | Dysphagic patients (unspecified aetiology) | Infant-cereal fortified minced/pureed diets | Unfortified minced/pureed diets | -Iron content -Meal consumption (plate wastage) -Meal compliance | Low |
| Massoulard et al. (2011) [51] | Observational Cross-sectional | 4 LTCs France | All residents with chewing and/or swallowing difficulties Mean age (y) 85.8 ± 9.3 Mean BMI (kg/m2) 26.8 ± 6.5 | Chopped n = 12 Mixed n = 26 | Regular diet n = 49 | -Nutrition intake (24 h weighed food record or staff reported survey) -Adequacy (France 2009) | Neutral |
| McCormick et al. (2008) [52] | Crossover Cohort 12 weeks | Geriatric hospital UK | Dysphagic patients who had were identified as at risk of aspiration and require TFs Mean age (y) 76 Mean BMI (kg/m2) 23.3 ± 0.4 | 6 weeks of pre-thickened TFs n = 11 | 6 weeks powder-thickened TFs (modified maize starch) n = 11 | -Macro- and micronutrient, fluid intake (Total daily oral liquid record) -Fluid adequacy (PENG 2004) Solid excluded | Positive |
| Miles, Liang et al. (2019) [19] | Observational Cross-sectional | 10 LTCs New Zealand | All residents ate in the dining room on the data collection day Ex. Residents ate outside the dining room Mean age (y) 78 | Pureed, n = 101 Minced and Moist, n = 99 Soft and Bite-sized, n = 100 [IDDSI, 2018] | Regular diet, n = 100 | -Meal consumption (visual estimation) -Nutrition content -Meal compliance -Snacks & Fortification | Neutral |
| Moreira et al. (2014) [53] | Observational Cross-sectional | Hospital Brazil | Diet orders from oncology patients | Blend, n = 6 Soft, n = 6 | Regular diet, n = 6 | -Supplement content | Neutral |
| Nowson et al. (2003) [54] | Observational Cross-sectional | 14 wards/dining rooms from LTCs Australia | Residents ate in the dining room on the data collection day Mean age (y) 82.9 ±9.5 | Pureed, n = 53 Soft/Minced, n = 48 | Regular diet, n = 114 | -Macro- and micronutrient intake (1-day food record by visual estimation of plate wastage) -Adequacy (Australia 1991) Snacks excluded | Neutral |
| Ott et al. (2019) [55] | Pre-post Experimental 12 weeks | 2 LTCs Germany | Residents diagnosed with chewing and/or swallowing receiving TMDs regularly (all patients had cognition impairment) Mean age (y) 86.5 ± 7.4 | 6 weeks of single level of reshaped TMDs and enriched with 600 kcal energy and 30 g protein n = 16 | 6 weeks of usual TMDs (completely pureed or partial soft food) n = 16 | -Energy and protein intake (3-day weighed food record) -Adequacy (German 2015) Snacks and ONS included | Neutral |
| Philip and Greenwood (2000) [56] | Observational Cross-sectional | Chronic-care hospital and LTC Canada | Residents/patients ≥65 y consumed ≥ 2 pureed entrees and/or TFs Ex: mixed texture diets, energy-controlled diets, specialized diets Mean age (y) 87.1 Mean BMI (kg/m2) 21.0 | Infant cereal fortified TFs with pureed diet n = 21 | Unfortified TFs with pureed diet Thin fluid with pureed diet n = 23 | -Macro-, micronutrient and fluid intake (7-day weighed food record) -Adequacy (USA 1989) -Nutrient content Snacks and fluid included | Neutral |
| Reyes-Torres et al. (2019) [57] | RCT 12 weeks | National institute Brazil | ≥65 yr with a caregiver and a confirmed diagnosis of oropharyngeal dysphagia, and consumed TMDs and TFs (evaluated by V-VST and EAT by dietitians) Ex: cancer, kidney/hepatic failure, terminally ill, high risk of aspiration, low oxygen saturation, enteral feed Mean age (y) 76 | Consistency modified and standardized TMDs and nectar or pudding level TFs (measured with Brookfield Viscometer) n = 20 | Unmodified pureed diet with one viscosity of TFs (consistency not systematically controlled) n = 20 | -Energy and protein intake (24 h recall) -Nutrient content | Positive |
| Rosenvinge and Starke (2005) [58] | Sequential observational with pre-post intervention | Hospital UK | Dysphagic patients from stroke, medicine, surgical, or geriatric wards (identified by SLTs) | TMDs + Pre-thickened TFs with education, n = 39 | TMDs + TFs, n = 16 | -Meal compliance | Neutral |
| Sherwin et al. (1998) [59] | Observational Cross-sectional | 6 LTC Australia | Residents ate in the dining room on the day of collection Mean age (y) 83.8 | Soft, n = 13 Homogenised, n = 26 | Regular diet, n = 36 | -Macronutrient and calcium intake (1-day weighed food record) -Adequacy Snacks excluded | Neutral |
| Taylor and Barr (2006) [60] | Crossover RCT 8 days | LTC Canada | Residents ≥65 y with dysphagia consumed TMDs (diagnosed by experienced swallowing team) Ex. enteral feed, medically unstable, diabetic diet Mean age (y) 85 ± 6.4 Mean BMI (kg/m2) 20.4 ± 3.4 | 4 days of isocaloric 5 meals pattern of minced/pureed diets n = 37 | 4 days of 3 meals pattern of minced/pureed diets n = 37 | -Energy and fluid intake (4-day weighed food record) -Nutrient content Snacks and fluids included | Neutral |
| Torrence (2011) [61] | Pre-post Experimental 15 days | LTC US | All residents consumed pureed diet (6 patients were on nutrition intervention program receiving ONS and/or fortification) Mean age (y) 85 Mean BMI (kg/m2) 27.6 | Pre-shaped pureed diet n = 10 | In-house made pureed diet n = 10 | -Meal consumption at breakfast, dinner and dessert (7.5-day weighed plate wastage) | Positive |
| Vivanti et al. (2009) [62] | Observational Cross-sectional | Hospital medical & neurosurgical wards Australia | Dysphagic patients consumed TMDs and TFs in for ≥5 days (diagnosed by SLTs) (Stroke n = 16, dementia n = 12, other n = 3) Mean age (y) 74.0 ± 16.2 Mean weight (kg) 62 ± 12.2 | TMDs + TFs + enteral/parenteral feed [UK national descriptors, 2002] n = 25 (36 days) | TMDs + TFs n = 25 (146 days) | -Fluid intake (7-day weighed plate wastage, observation and fluid balance charts) -Fluid Adequacy (PENG 2004) Snacks and fluids included | Neutral |
| Vucea et al. (2018) [63] | Observational Cross-sectional | 32 LTCs Canada | Diet orders from LTCs recruited for M3 study opened for ≥6 m had ≥50 residents over 65 y admitted for ≥1 m | Pureed diet n = 32 facilities | Regular diet n = 32 facilities | -Macro- and micronutrient content Snacks excluded | Neutral |
| Welch et al. (1991) [64] | Pre-post Experimental 6 m | LTC US | Residents consumed pureed diet and weighed below average or serum albumin/transferrin levels below normal values (identified from medical records) Mean age (y) 81 | Pureed diets with fortified high fiber cereals and commercial supplements n = 15 | Pureed diets with unfortified cereals n = 15 | -Macro- and micronutrient intake (3-day food record by staff) -Adequacy (USA 1980) -Supplement/fortification content Snacks and ONS included | Neutral |
| Whelan (2001) [65] | RTC 14 days | Hospital UK | Acute stroke dysphagic patients required syrup-consistency TFs (diagnosed by SLT or videofluoroscopy) Ex. diagnosis using only a screening tool Mean age (y) 72.3 ± 13.4 Mean weight (kg) 67.7 ± 12.7 | Pre-thickened TFs, n = 11 | Powder-thickened TFs, n = 13 | -Fluid intake (Total daily oral liquid record and fluid balance chart) -Adequacy (UK) -Nutrient content Solid excluded | Positive |
| Wright et al. (2005) [66] | Observational Cross-sectional | Hospital elderly and neurology wards UK | All medically stable patients consumed TMDs or regular diet (reasons for TMDs: 80% dysphagia, 20% poor dental state; stroke n = 19, fall n = 8, other n = 3) Ex. < 60 y, nil by mouth, enteral feed, other therapeutic diets Mean age (y) 81.5 | Texture B–Smooth pureed, n = 10 Texture D- Minced/mashed, n = 9 Texture E- Soft, n = 11 [UK national descriptors, 2002] | Regular diet, n = 25 | -Nutrition intake (1-day weighed food record for main meals and food records) -Meal consumption (weighed plate wastage) -Adequacy (Schofieled equation 1985) -Supplements acceptance Snacks and ONS included | Neutral |
| Zanini et al. (2017) [67] | Pre-post experimental 6 m | 20 LTCs Italy | Dysphagic residents > 65 y with low comorbidity levels (diagnosed by a physician or reported in medical records) Ex. high level of comorbidity, medically unstable, terminally ill, chronic or cancer disease, sever dysphagia (DOSS ≥ 2), enteral feed Mean age (y) 79.72 ± 12.31 | 6 m of personalised levels of density, viscosity, texture and particle size TMDs n = 401 | 6 m of unmodified TMDs n = 401 | -Nutrient content -Meal consumption (visual estimation) | Positive |
| Studies | Outcomes |
|---|---|
| Cassen et al. [37] | 15% ↑ intake with 16-day of 3 D moulded pureed intervention |
| de Sa et al. [39] | Main meal consumption rate: blended–75.3%, soft–74.2% and regular diet–79.7% Main meal with provision of ONS: blended–78.0%, soft–68.9% and regular diet–74.2% Consumption of ONS at morning and afternoon tea: blended–82.6 vs. 100%, soft–84.7 vs. 96.8% and regular diet–81.9 vs. 58.3% |
| Farrer et al. [41] | ↑ %patients consuming from <25% to >75% in 2-week of moulded pureed p = 0.03 NS ↓ in plate wastage with moulded compared to non-moulded pureed (160 g vs. 286 g, p = 0.09) |
| Higashiguchi [46] | Enzyme-infused TMDs showed a slightly ↑ consumption compared to unmodified TMDs (69.6% vs. 68.7%, p > 0.05) |
| Keller et al. [49] | NS ↑ by using mix of cMTF and rMTF compared to cMTF (p = 0.1) |
| Kennewell and Kokkinakos [54] | NS ↑ with infant cereal fortified pureed |
| Miles et al. [19] | % patient consumed the full meal: pureed–59%, minced & moist–55%, soft–52% and regular diet–43% |
| Nowson et al. [54] | % patient consumed the full meal: pureed–74.1 (19.8) %, soft–76.7 (24.5) % and regular diet–82.2 (16.9)% |
| Torrence [61] | Significant ↑ consumption of breakfast (p = 0.007), dinner (p = 0.017) and dessert (p = 0.005) with pre-shaped pureed |
| Wright et al. [66] | 13% (n = 4) of the patients on TMDs completed the full meal |
| Zanini et al. [67] | 83.4% and 12.3% of the texture-individualised TMDs were fully or partially consumed respectively 4.1% of the meals were not eaten |
| Studies | Meal Compliance Outcomes |
|---|---|
| Bannerman and McDermott [14] | Unmodified vegetables Unrecognizable meal (mix blend) High-risk foods, e.g., pineapple, tomatoes Mixed consistency, e.g., lumps, crumbs Texture modification was not used on snacks |
| Cassens et al. [37] | Improved texture of pureed foods after using enhancer and thickener |
| Kennewell and Kokkinakos [50] | Improved food texture consistency after fortification the pureed foods with fortified infant cereal |
| Miles et al. [19] | 60% (n = 6) and 33% (n = 1) facilities met IDDSI criteria for pureed and minced and moist diets respectively. None of the facilities provided appropriate soft and bite-sized diet. |
| Rosenvinge and Starke [58] | TFs consistency sig. ↑ at 2nd audit (64.1% vs. 48.4%, p < 0.05) No sig. differences in compliance with dietary modifications (82.5% vs. 78.7%) Sig. ↑ compliance with the quantity of food/fluids was given (35.3% vs. 68.8%, p < 0.05), 52.4% of the time patient was fed more than specified |
| Wright et al. [66] | 13% (n = 4) patients in TMDs received correct food delivery |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wu, X.S.; Miles, A.; Braakhuis, A. Nutritional Intake and Meal Composition of Patients Consuming Texture Modified Diets and Thickened Fluids: A Systematic Review and Meta-Analysis. Healthcare 2020, 8, 579. https://doi.org/10.3390/healthcare8040579
Wu XS, Miles A, Braakhuis A. Nutritional Intake and Meal Composition of Patients Consuming Texture Modified Diets and Thickened Fluids: A Systematic Review and Meta-Analysis. Healthcare. 2020; 8(4):579. https://doi.org/10.3390/healthcare8040579
Chicago/Turabian StyleWu, Xiaojing Sharon, Anna Miles, and Andrea Braakhuis. 2020. "Nutritional Intake and Meal Composition of Patients Consuming Texture Modified Diets and Thickened Fluids: A Systematic Review and Meta-Analysis" Healthcare 8, no. 4: 579. https://doi.org/10.3390/healthcare8040579
APA StyleWu, X. S., Miles, A., & Braakhuis, A. (2020). Nutritional Intake and Meal Composition of Patients Consuming Texture Modified Diets and Thickened Fluids: A Systematic Review and Meta-Analysis. Healthcare, 8(4), 579. https://doi.org/10.3390/healthcare8040579





