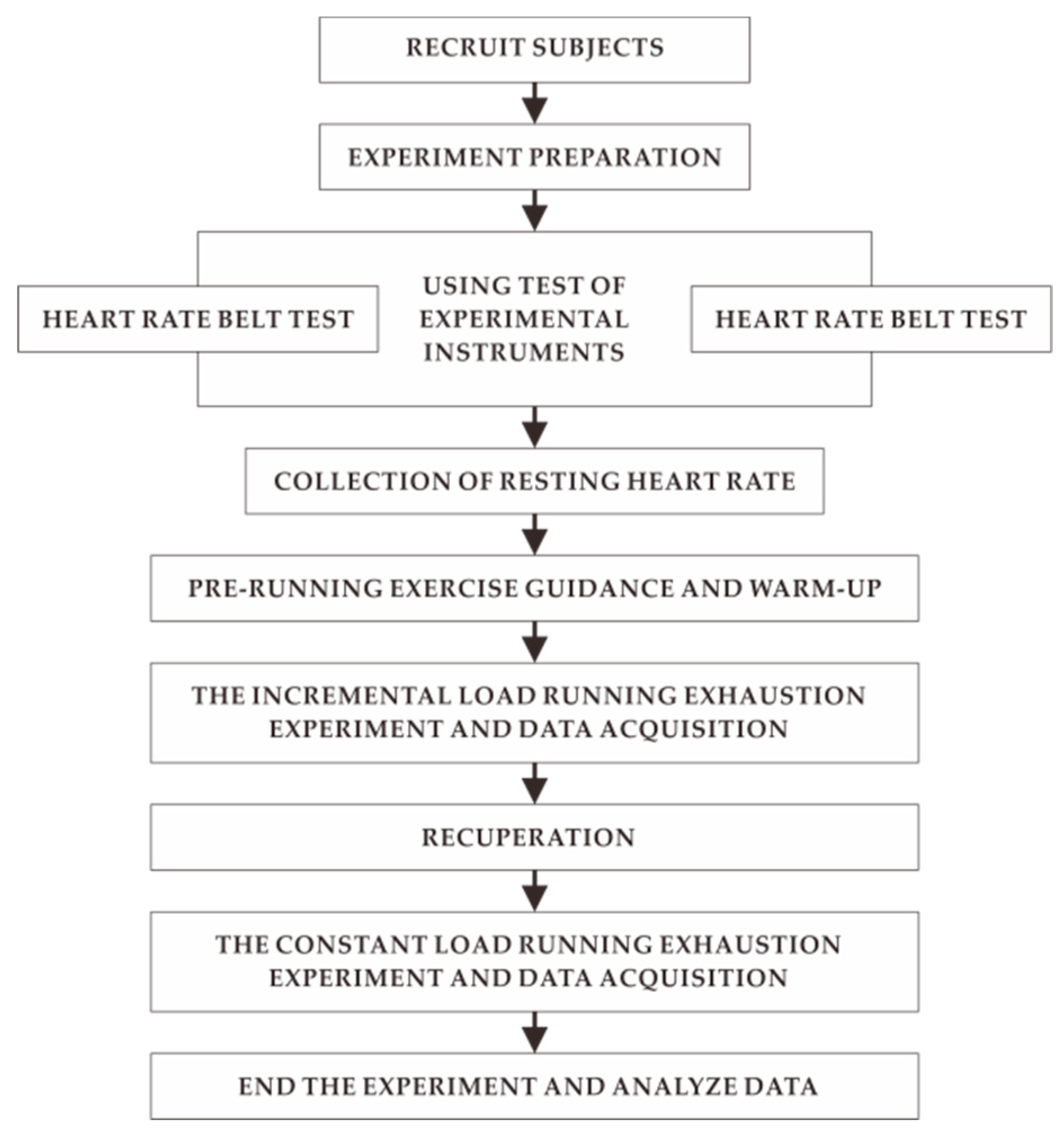
1. Introduction
With the improvement of socio-economic status, fitness is being considered as a lifestyle [
1]. Running is one of the most popular fitness regimens because it does not require equipment and can be practiced almost everywhere. Unfortunately, the annual incidence of injuries among runners is 40% to 50% [
2]. Inappropriate exercise intensity during running is one of the leading causes of injury. Even worse, excessive exercise leads to local muscular injuries [
3]. Through the monitoring and evaluation of exercise intensity and physical fatigue, scientific guidance during exercise is very useful and necessary to prevent injuries. To improve athletes’ performance and to cultivate talents in sports, the monitoring of training intensity and quantity in competitive sports requires precise and expensive scientific research instruments and one-to-one coaching with sports experts [
4]. This approach is not suitable for amateur runners. For them, a set of convenient and feasible detection methods needs to be developed to guide their running.
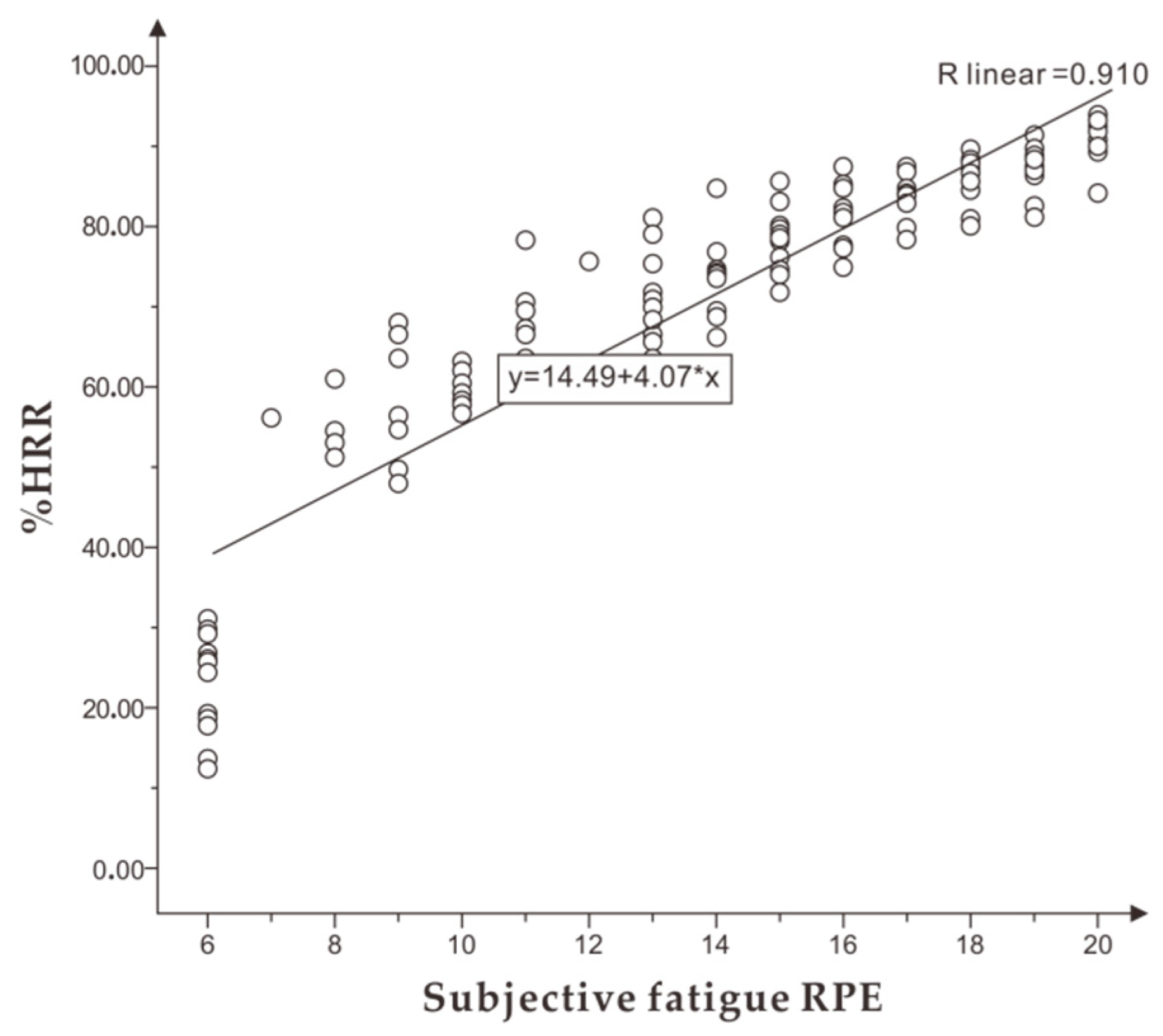
Exercise intensity and fatigue can be evaluated using subjective self-perception, objective physiological, and mechanical indicators [
5]. Self-perception is the self-evaluation [
6] of intensity and fatigue through the rating of perceived exertion (RPE) [
7]. The objective evaluation of exercise intensity is carried out by using direct indicators such as speed and force, and with the help of surface electromyography (sEMG) signals, percentage of maximum oxygen uptake (%VO
2max), percentage of reserve heart rate (%HRR), blood lactic acid (BLA), and creatine kinase (CK). Other physiological and biochemical indicators can indirectly evaluate exercise intensity. The objective evaluation of exercise fatigue is obtained with blood urea (BU), hemoglobin (HGB), testosterone (T), sEMG signals, and heart rate [
8]. sEMG and heart rate have the advantages of being non-invasive, real-time, and convenient, so they are widely used in the field of sports science and ergonomics.
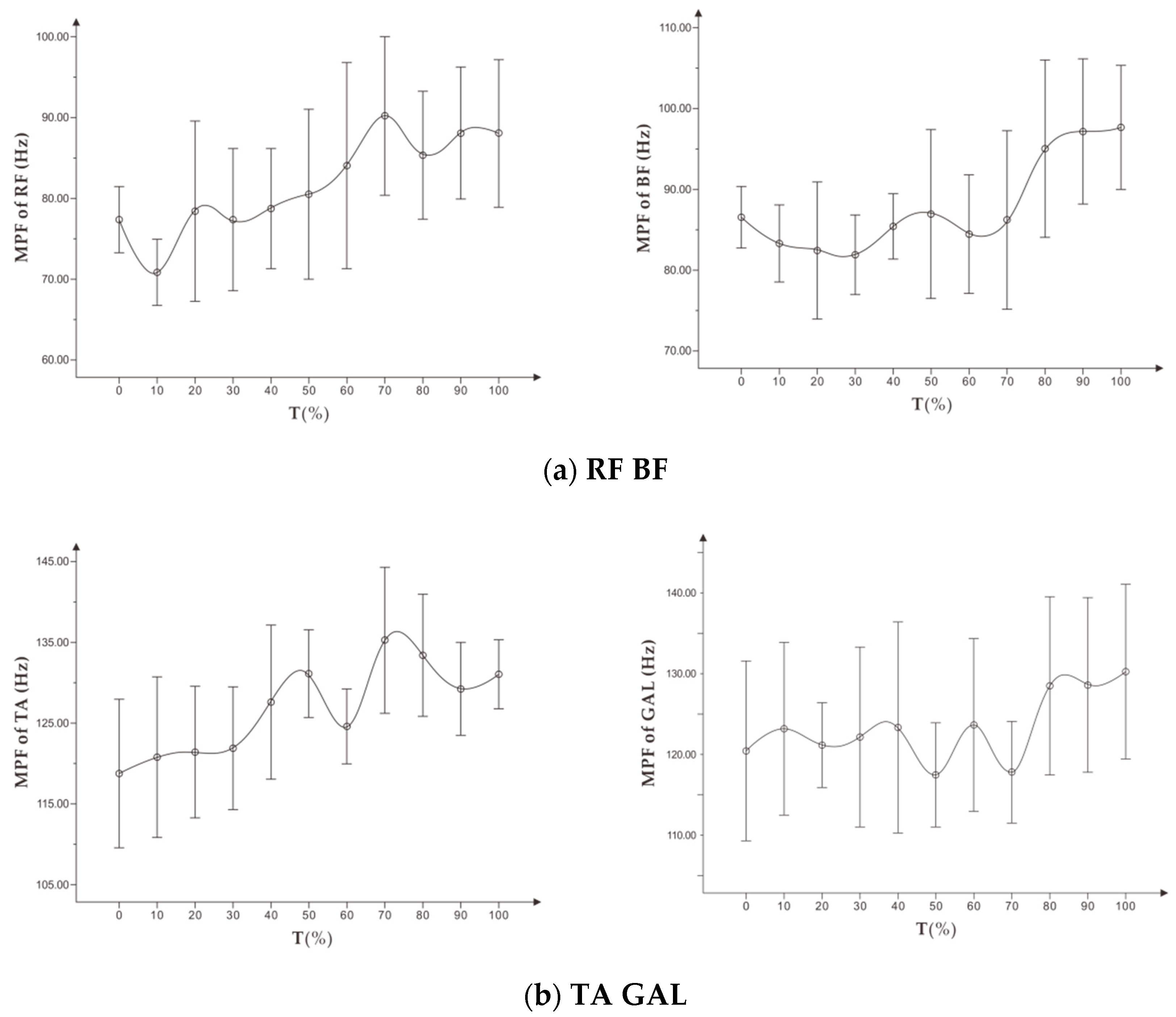
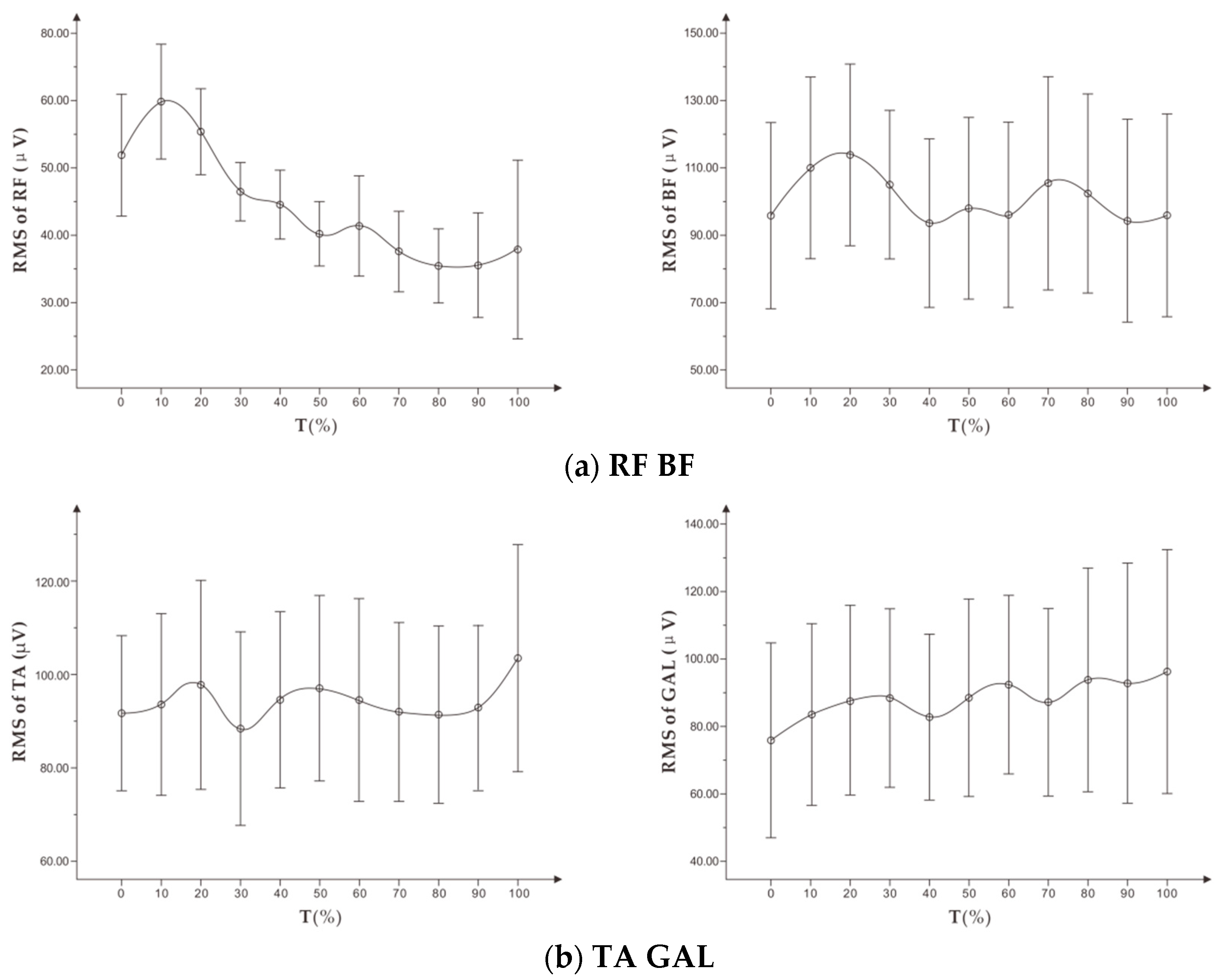
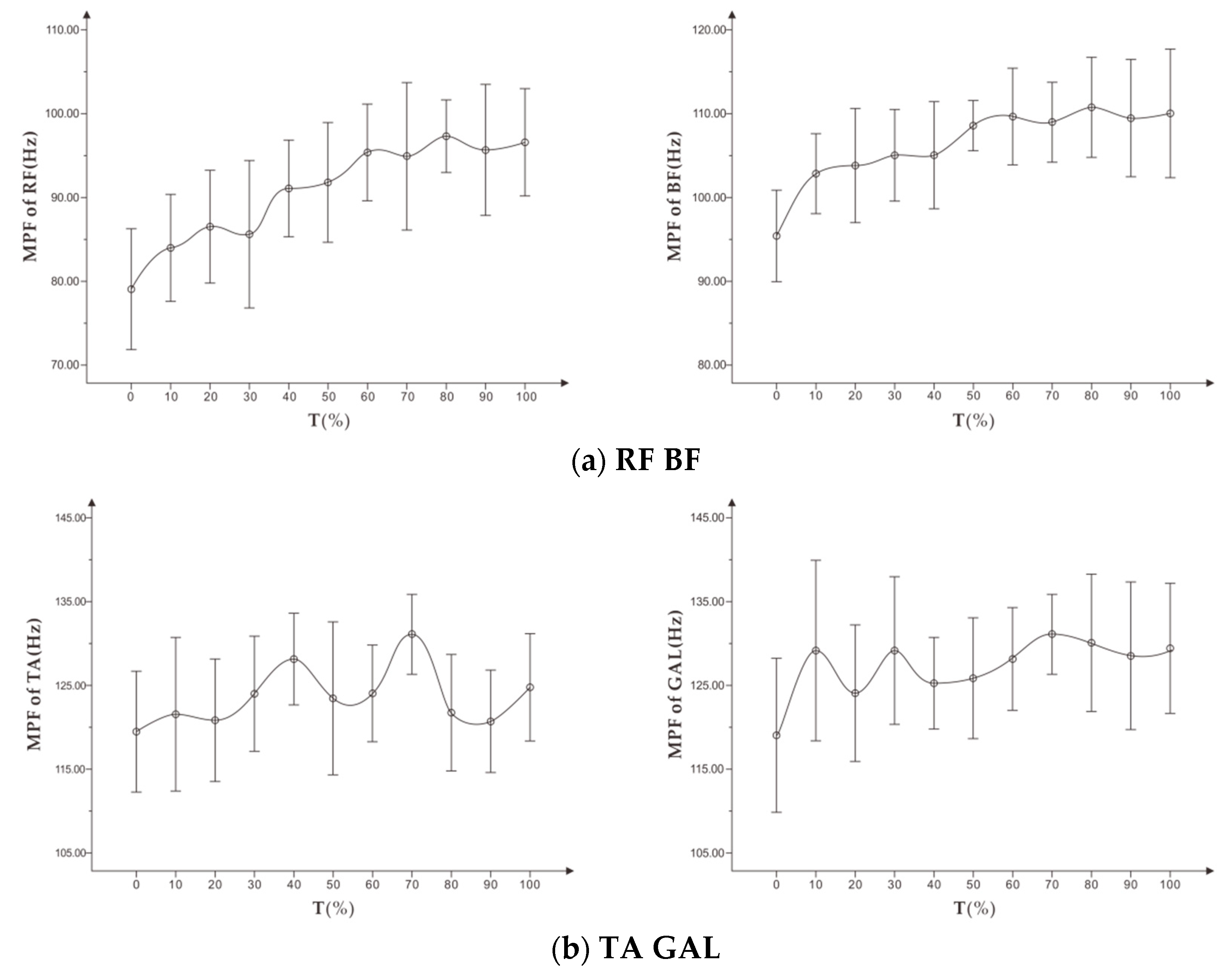
To study exercise-induced fatigue, especially localized muscle fatigue, sEMG technology uses the time and frequency domain indicators of the expanding signal. In the pedal exercise experiment with increasing load, Petrofsky et al. [
9] found that there is a significant linear correlation between the amplitude of EMG root mean square (RMS) and muscle exercise load. The RMS increases continuously within a certain load range, while the median frequency (MF) decreases during the entire exercise. Wang et al. [
10] proposed that the mean power frequency (MPF) of the frequency domain indicator decreased with fatigue, while the change of the MPF indicator could vary in the dynamic fatigue process. Tamaki et al. [
11] proposed that the value of integrated EMG (iEMG) can better reflect the relationship between muscle fatigue and increase of exercise time. They also found that the frequency domain indicator (MPF) is more sensitive than the median frequency in measuring muscle fatigue.
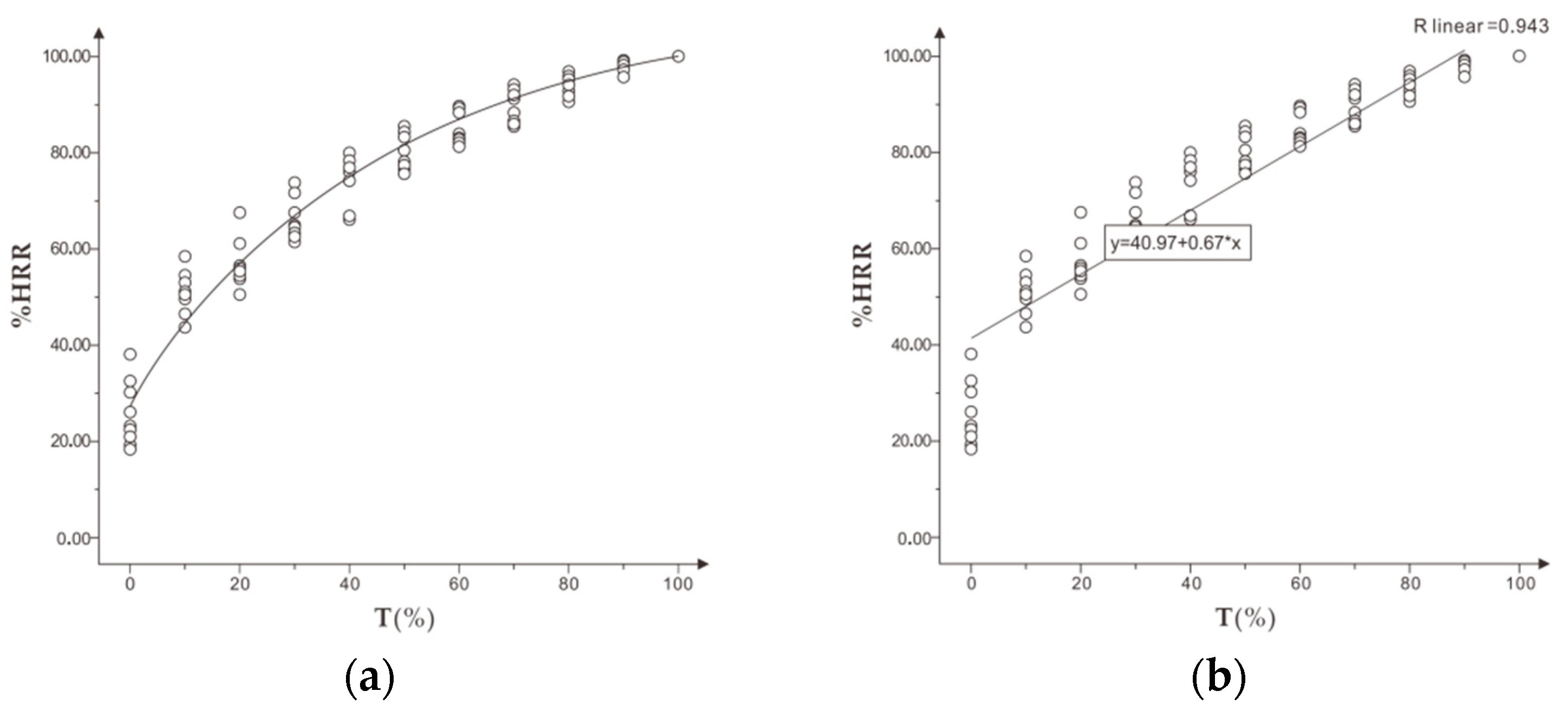
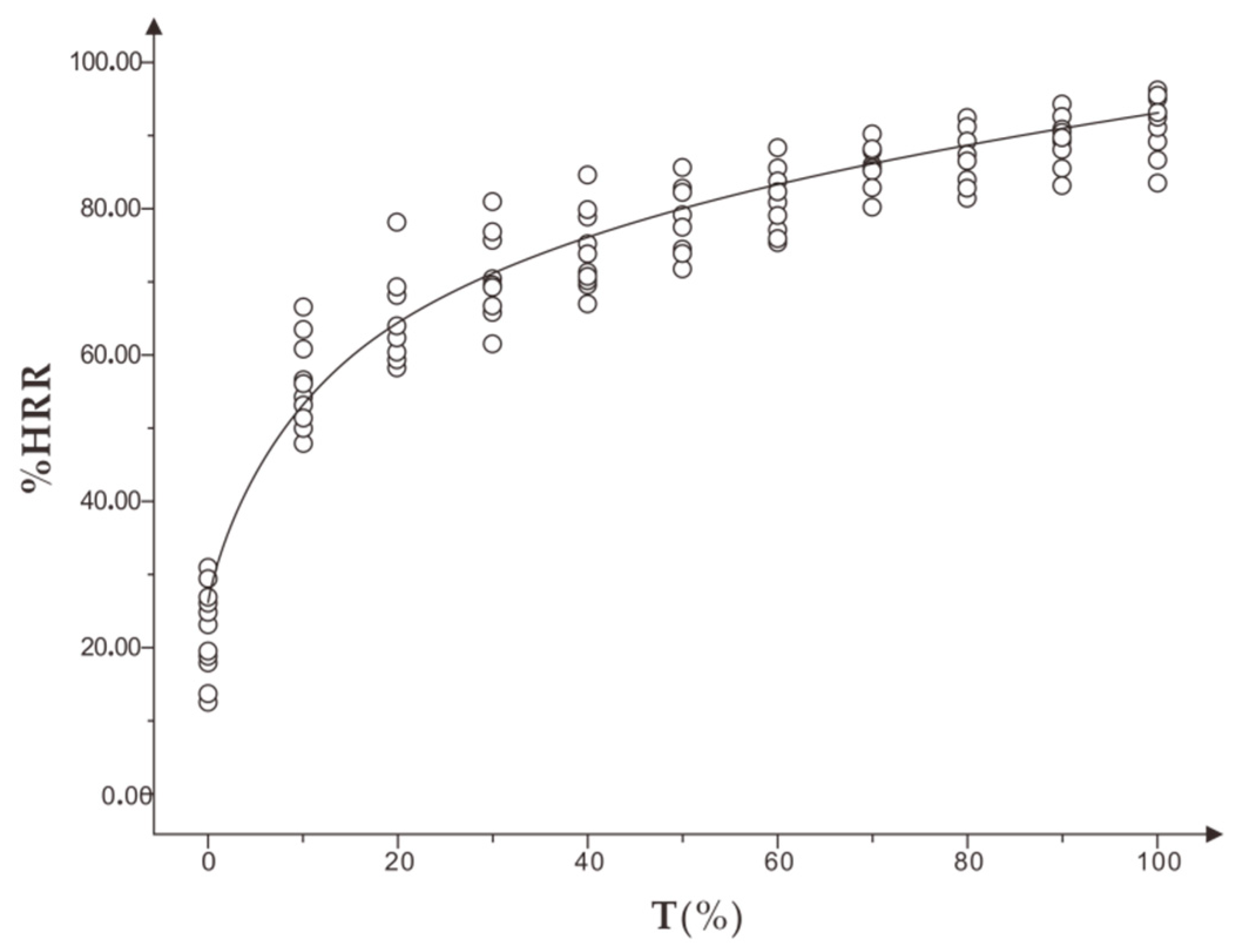
During training, coaches monitor athlete training using the principle that the heart rate of athletes increases with the increase of intensity and fatigue. The percentage of heart rate reserve (%HRR) is included in the variable of resting heart rate of athletes. The difference in individual physical fitness can be better compared when monitoring and evaluating exercise intensity and exercise fatigue [
12].
Previous studies have focused on heart rate indicators, sEMG indicators, exercise intensity, and fatigue. Most of these studies focus on static exercise or short-term isokinetic exercise, which strictly control the experimental conditions. Until now, research on the intensity and fatigue of long-term exercise for non-athlete individuals remain limited. Considering the lack of scientific fitness knowledge and guidance, this study used MPF, RMS of sEMG signal and %HRR, combined with the RPE scale in the experimental design. Increasing load exhaustive exercise is the most widely used experiment to evaluate the VO
2 max. The %HRR has been highly correlated with %VO2max so it can replace %VO
2 max in the evaluation and guidance of exercise intensity [
13]. Therefore, an incremental load-running exhaustion experiment was designed to assess an appropriate personalized moderate running intensity using the %HRR interval and the mean personalized speed when the intensity is reached. In addition, a constant speed running exhaustion experiment was designed, in which the mean personalized speed, obtained in the incremental experiment, was used as the constant speed to explore the %HRR range at a moderate fatigue level. MPF and RMS were used to explore the localized muscle fatigue during the two experiments. The changing trend and correlation of each indicator in both experiments were observed, respectively. A suitable exercise intensity and fatigue degree for personalized running based on subjective and objective physiological information were further explored to provide real-time references for amateur runners.
5. Conclusions
First, this study showed that MPF and RMS indicators of sEMG signal during running could not conclusively identify the fatigue level of the targeted muscle. These indicators were not suitable for use as a reference for personalized running assessment.
Second, %HRR could effectively identify and evaluate the exercise intensity and fatigue of runners to a certain extent. It is suggested that the personalized running speed of different individuals with appropriate intensity could be measured in the form of incremental running speed according to the mean interval of %HRR of appropriate intensity 75%–80% HRmax, and the mean range of %HRR could be obtained from personalized running speed exercise in moderate fatigue, with a recommended range of 77%–85% HRmax. Individuals running at a personalized speed of appropriate intensity to moderate fatigue %HRR will achieve better fitness results and prevent excessive fatigue.
Lastly, the physiological indicators selected in this study are limited. In futures studies, wearable detecting techniques such as infrared sensing and respiratory telemetry can be implemented to identify the running status of runners, which could enable personalized scientific guidance for ordinary runners.