Development and Use of the ‘SENS’-Structure to Proactively Identify Care Needs in Early Palliative Care—An Innovative Approach
Abstract
1. Introduction
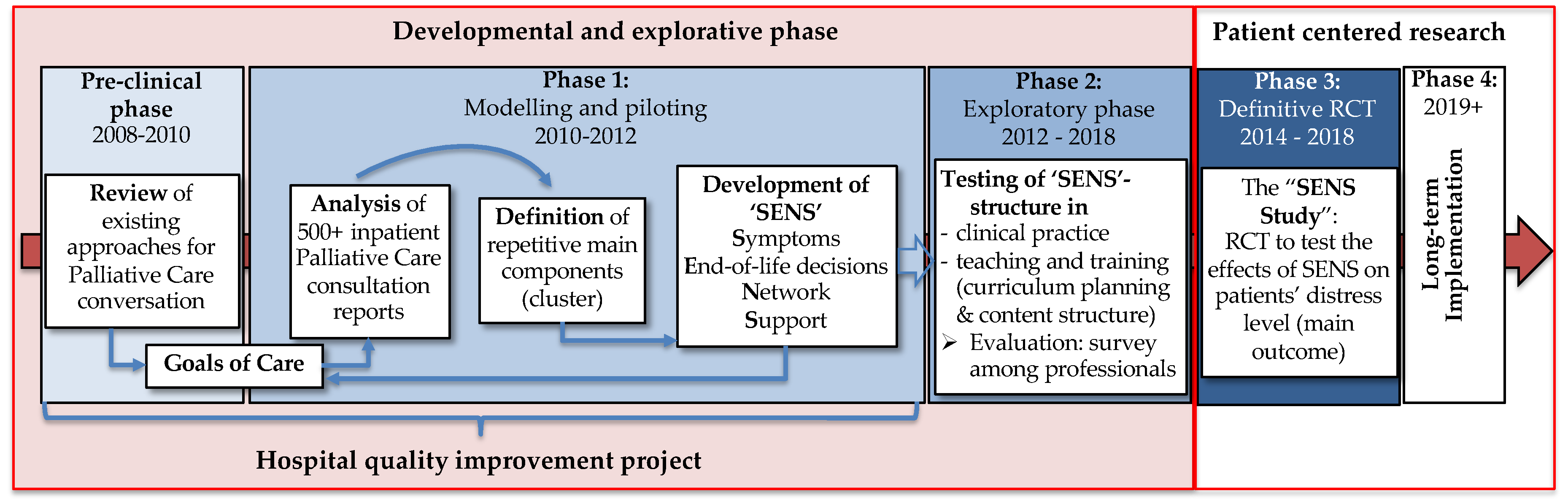
2. Materials and Methods
3. Results
3.1. Pre-Clinical Phase
3.2. Phase 1: Piloting and Modeling Phase
- Symptoms patients suffer from or worries they may have, including self-support strategies in case of a crisis, as well as carer empowerment in symptom crises;
- End-of-life decisions in regard to the future, including individual care and treatment preferences, potentially formulated as an advance directive;
- Network organization (private and professional), including assessment of current living circumstances and organization of support in case of an emergency not manageable at home/nursing home;
- Support for the carers to cope with the situation and to prevent overburdening.
3.3. Phase 2: Exploratory Phase
3.4. Phases 3 and 4: Patient-Centered Research and Implementation
4. Discussion
4.1. Impact of the Use of the ‘SENS’-Structure on Clinical Practice
4.2. Effects of the Initiation of a ‘SENS’-Based Conversation
4.3. Impact of ‘SENS’ on Financial Reimbursement of PC Interventions
4.4. Impact of ‘SENS’ on PC Education and Research
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Dixon, J.; Matosevic, T.; Knapp, M. The economic evidence for advance care planning: Systematic review of evidence. Palliat. Med. 2015, 29, 869–884. [Google Scholar] [CrossRef] [PubMed]
- Mitchell, G.K. The role of general practice in cancer care. Aust. Fam. Physician 2008, 37, 698–702. [Google Scholar] [PubMed]
- Rubin, G.; Berendsen, A.; Crawford, S.M.; Dommett, R.; Earle, C.; Emery, J.; Fahey, T.; Grassi, L.; Grunfeld, E.; Gupta, S.; et al. The expanding role of primary care in cancer control. Lancet Oncol. 2015, 16, 1231–1272. [Google Scholar] [CrossRef]
- Brinkman-Stoppelenburg, A.; Rietjens, J.A.; van der Heide, A. The effects of advance care planning on end-of-life care: A systematic review. Palliat. Med. 2014, 28, 1000–1025. [Google Scholar] [CrossRef] [PubMed]
- Bond, W.F.; Kim, M.; Franciskovich, C.M.; Weinberg, J.E.; Svendsen, J.D.; Fehr, L.S.; Funk, A.; Sawicki, R.; Asche, C.V. Advance care planning in an accountable care organization is associated with increased advanced directive documentation and decreased costs. J. Palliat. Med. 2018, 21, 489–502. [Google Scholar] [CrossRef] [PubMed]
- Zhang, B.; Wright, A.A.; Huskamp, H.A.; Nilsson, M.E.; Maciejewski, M.L.; Earle, C.C.; Block, S.D.; Maciejewski, P.K.; Prigerson, H.G. Health care costs in the last week of life: Associations with end-of-life conversations. Arch. Intern. Med. 2009, 169, 480–488. [Google Scholar] [CrossRef] [PubMed]
- Gomez-Batiste, X.; Martinez-Munoz, M.; Blay, C.; Espinosa, J.; Contel, J.C.; Ledesma, A. Identifying needs and improving palliative care of chronically ill patients: A community-oriented, population-based, public-health approach. Curr. Opin. Support. Palliat. Care 2012, 6, 371–378. [Google Scholar] [CrossRef] [PubMed]
- Temel, J.S.; Greer, J.A.; Muzikansky, A.; Gallagher, E.R.; Admane, S.; Jackson, V.A.; Dahlin, C.M.; Blinderman, C.D.; Jacobsen, J.; Pirl, W.F.; et al. Early palliative care for patients with metastatic non-small-cell lung cancer. N. Engl. J. Med. 2010, 363, 733–742. [Google Scholar] [CrossRef] [PubMed]
- Zimmermann, C.; Swami, N.; Krzyzanowska, M.; Hannon, B.; Leighl, N.; Oza, A.; Moore, M.; Rydall, A.; Rodin, G.; Tannock, I.; et al. Early palliative care for patients with advanced cancer: A cluster-randomised controlled trial. Lancet 2014, 383, 1721–1730. [Google Scholar] [CrossRef]
- Luckett, T.; Phillips, J.; Agar, M.; Virdun, C.; Green, A.; Davidson, P.M. Elements of effective palliative care models: A rapid review. BMC Health Serv. Res. 2014, 14, 136. [Google Scholar] [CrossRef]
- Weisbord, S.D.; Carmody, S.S.; Bruns, F.J.; Rotondi, A.J.; Cohen, L.M.; Zeidel, M.L.; Arnold, R.M. Symptom burden, quality of life, advance care planning and the potential value of palliative care in severely ill haemodialysis patients. Nephrol. Dial. Transplant. 2003, 18, 1345–1352. [Google Scholar] [CrossRef] [PubMed]
- Karam, C.Y.; Paganoni, S.; Joyce, N.; Carter, G.T.; Bedlack, R. Palliative care issues in amyotrophic lateral sclerosis: An evidenced-based review. Am. J. Hosp. Palliat. Care 2016, 33, 84–92. [Google Scholar] [CrossRef] [PubMed]
- Greutmann, M.; Tobler, D.; Colman, J.M.; Greutmann-Yantiri, M.; Librach, S.L.; Kovacs, A.H. Facilitators of and barriers to advance care planning in adult congenital heart disease. Congenit. Heart Dis. 2013, 8, 281–288. [Google Scholar] [CrossRef] [PubMed]
- Biondo, P.D.; Lee, L.D.; Davison, S.N.; Simon, J.E.; Advance Care Planning Collaborative Research; Innovation Opportunities Program. How healthcare systems evaluate their advance care planning initiatives: Results from a systematic review. Palliat. Med. 2016, 30, 720–729. [Google Scholar] [CrossRef] [PubMed]
- Block, S.D.; Billings, J.A. A need for scalable outpatient palliative care interventions. Lancet 2014, 383, 1699–1700. [Google Scholar] [CrossRef]
- Saunders, C. The treatment of intractable pain in terminal cancer. Proc. R. Soc. Med. 1963, 56, 195–197. [Google Scholar]
- Medical Research Council (MRC). A Framework for Development and Ecaluation of RCTs for Complex Interventions to Improve Health. 2000. Available online: https://mrc.ukri.org/documents/pdf/rcts-for-complex-interventions-to-improve-health/ (accessed on 20 May 2008).
- Stiftung SanaCERT Suisse. Standard 25: [Palliative Care]. Available online: http://www.sanacert.ch/qualitaetsstandards/akutbereich/ (accessed on 21 November 2008).
- Federal Office of Public Health (FOPH). Framework concept for palliative care in switzerland. A basis for defining palliative care for the implementation of the «National Strategy for Palliative Care». In Proceedings of the Swiss Conference of the Cantonal Ministers of Public Health (CMH) and palliative ch (2014), Bern, Switzerland, 15 July 2014. [Google Scholar]
- Eychmueller, S. SENS is making sense—On the way to an innovative approach to structure Palliative Care problems. Ther. Umsch. 2012, 69, 87–90. [Google Scholar] [CrossRef]
- Qualitépalliative [Association for Quality in Palliative Care]. Available online: https://www.qualitepalliative.ch/das-label/?L=0 (accessed on 1 November 2018).
- Eychmueller, S.; Fliedner, M. Active Preparation for the End of Life with Patients with Advanced Cancer: A Way to Reduce Stress and Increase Self-Determination? Available online: http://www.nfp67.ch/de/projekte/modul-1-sterbeverlaeufe-und-versorgung/projekt-eychmueller (accessed on 1 October 2014).
- Sepulveda, C.; Marlin, A.; Yoshida, T.; Ullrich, A. Palliative Care: The World Health Organization’s global perspective. J. Pain Symptom. Manag. 2002, 24, 91–96. [Google Scholar] [CrossRef]
- World Health Organization (WHO). National Cancer Control Programmes: Policies and Managerial Guidelines, 2nd ed.; World Health Organization (WHO): Geneva, Switzerland, 2002. [Google Scholar]
- Levy, M.; Smith, T.; Alvarez-Perez, A.; Back, A.; Baker, J.N.; Beck, A.C.; Block, S.; Dalal, S.; Dans, M.; Fitch, T.R.; et al. Palliative Care Version 1.2016. J. Natl. Compr. Can. Netw. 2016, 14, 82–113. [Google Scholar] [CrossRef]
- Levy, M.H.; Back, A.; Benedetti, C.; Billings, J.A.; Block, S.; Boston, B.; Bruera, E.; Dy, S.; Eberle, C.; Foley, K.M.; et al. NCCN clinical practice guidelines in oncology: Palliative care. J. Natl. Compr. Can. Netw. 2009, 7, 436–473. [Google Scholar] [CrossRef]
- National Gold Standards Framework Centre. National Gold Standards Framework. Available online: http://www.goldstandardsframework.org.uk/advance-care-planning (accessed on 16 February 2014).
- Shaw, K.L.; Clifford, C.; Thomas, K.; Meehan, H. Review: Improving end-of-life care: A critical review of the gold standards framework in primary care. Palliat. Med. 2010, 24, 317–329. [Google Scholar] [CrossRef] [PubMed]
- Thomas, K.; Noble, B. Improving the delivery of palliative care in general practice: An evaluation of the first phase of the Gold Standards Framework. Palliat. Med. 2007, 21, 49–53. [Google Scholar] [CrossRef] [PubMed]
- Popescu, G.; Tschanz Cooke, K.; Leemann, H.; Zimmermann, J.; Popescu, R. SENS und Sensibility—Ambulante und stationäre Palliative Care in der Hirslanden Klinik Aarau (HKA) [SENS and Sensibility—outpatient and inpatient Palliative Care in the Hirslanden Hospital Aarau]. In Proceedings of the Nationaler Palliative Care Kongress, Biel, Switzerland, 26–27 November 2014; p. 29. [Google Scholar]
- Federal Office of Public Health (FOPH) and palliative.ch. Palliative Care: Quality of Life until the End—An Information Sheet for General Practitioners. 2014. Available online: http://www.palliative.ch/fileadmin/user_upload/palliative/fachwelt/informationsblatt/palliative_care_hausaerzte.pdf (accessed on 20 December 2019).
- Palliative Ostschweiz. Palliative Care—Professional Competencies Level A-C Based on‚ SENS; Palliative Ostschweiz: St. Gallen, Switzerland, 2015. [Google Scholar]
- Eychmueller, S.; Abbühl, L.S.; Bock, F.; Bueche, D.; Christen, S.; Gudat, H.; Marti, M.; Neuenschwander, H.; Obrist, S.; Pautex, S.; et al. Palliative Medicine Essentials—Fundamentals of Palliative Care; Verlag Hans Huber: Bern, Switzerland, 2015. [Google Scholar]
- Federal Statistical Office (BFS). Circular for Coders: 2018/Nr. 1. 2017, p. 11. Available online: https://www.bfs.admin.ch/bfs/de/home/aktuell/neue-veroeffentlichungen.assetdetail.4202459.html (accessed on 21 November 2018).
- Zwahlen, S.; Trelle, S.; Fliedner, M.C.; Juni, P.; Aebersold, D.; Aujesky, D.; Fey, M.; Eychmueller, S. A single early Palliative Care intervention—A randomized controlled trial. 2019; under submission. [Google Scholar]
- Fliedner, M.C.; Zambrano, S.; Schols, J.M.G.A.; Bakitas, M.; Lohrmann, C.; Halfens, R.J.G.; Eychmueller, S. A structured early palliative care intervention based on “SENS” can be confronting but reassuring: A qualitative study on the experiences of patients with advanced cancer. 2019; under review. [Google Scholar]
- Eychmueller, S.; Egger, M.; Streit, S.; Gross, C.; Kläui, H.; Gerber, A.; Trelle, S.; Cina, C.; Mitchell, G. The Bern Primary Palliative Care Trial (Bern-PPCT): A Cluster Trial of Palliative Needs Assessment and Care in General Practice. Available online: http://www.nfp74.ch/de/projekte/sektor-uebergreifende-versorgung/projekt-eychmueller (accessed on 17 July 2018).
- Walshe, C.; Caress, A.; Chew-Graham, C.; Todd, C. Implementation and impact of the Gold Standards Framework in community palliative care: A qualitative study of three primary care trusts. Palliat. Med. 2008, 22, 736–743. [Google Scholar] [CrossRef] [PubMed]
- Maas, E.A.; Murray, S.A.; Engels, Y.; Campbell, C. What tools are available to identify patients with palliative care needs in primary care: A systematic literature review and survey of European practice. BMJ Support. Palliat. Care 2013, 3, 444–451. [Google Scholar] [CrossRef]
- Smith, C.B.; Phillips, T.; Smith, T.J. Using the New ASCO Clinical Practice Guideline for Palliative Care Concurrent With Oncology Care Using the TEAM Approach. Am. Soc. Clin. Oncol. Educ. Book 2017, 37, 714–723. [Google Scholar] [CrossRef]
- Ferrell, B.R.; Temel, J.S.; Temin, S.; Alesi, E.R.; Balboni, T.A.; Basch, E.M.; Firn, J.I.; Paice, J.A.; Peppercorn, J.M.; Phillips, T.; et al. Integration of Palliative Care into Standard Oncology Care: American Society of Clinical Oncology Clinical Practice Guideline Update. J. Clin. Oncol. 2017, 35, 96–112. [Google Scholar] [CrossRef]
- Van den Block, L. The need for integrating palliative care in ageing and dementia policies. Eur. J. Public Health 2014, 24, 705–706. [Google Scholar] [CrossRef]
- Mehta, Z.; Giorgini, K.; Ellison, N.; Roth, M.E. Integrating palliative medicine with dementia care. Agging Well 2012, 5, 18. [Google Scholar]
- World Health Organization (WHO). Strengthening of Palliative Care as a Component of Comprehensive Care throughout the Life Course; World Health Assembly: Geneva, Switzerland, 2014. [Google Scholar]
- Greer, J.A.; Tramontano, A.C.; McMahon, P.M.; Pirl, W.F.; Jackson, V.A.; El-Jawahri, A.; Parikh, R.B.; Muzikansky, A.; Gallagher, E.R.; Temel, J.S. Cost analysis of a randomized trial of early palliative care in patients with metastatic nonsmall-cell lung cancer. J. Palliat. Med. 2016. [Google Scholar] [CrossRef]
- Smith, S.; Brick, A.; O’Hara, S.; Normand, C. Evidence on the cost and cost-effectiveness of palliative care: A literature review. Palliat. Med. 2014, 28, 130–150. [Google Scholar] [CrossRef]
| Common Goals of PC (Switzerland, 2014) | WHO 1 Definition of PC (2002) | NCCN 2 Guideline for PC (2009/2016) | GSF 3 (2014) | Theme | ‘SENS’-Structure Theme (Eychmueller, 2012) |
|---|---|---|---|---|---|
| Improving Self-help capacities | Self-effectiveness | Symptoms | |||
| × | × | × | Physical | ||
| × | × | Educational and informational needs | |||
| × | × | Spiritual | |||
| × | × | Quality of life | |||
| × | Personal (e.g., inner journey) | ||||
| × | Late (e.g., death rattle, agitation) | ||||
| Promoting Self-determination | × | × | Self determination | End-of-life decisions | |
| × | Benefits and risks of (anticancer) treatment | ||||
| × | Emotional (e.g., fears, relationships) | ||||
| × | Dying issues | ||||
| Ensuring Safety in sometimes life-threatening situations | × | Out of hours—emergency | Network | ||
| Safety | |||||
| × | × | Psychosocial | |||
| × | Cultural factors affecting life | ||||
| Assuring Support for the encumbered family | × | × | Support of family | Support of the carer | |
| × | Afterwards (bereavement period) |
| ‘SENS’—Theme and Definition | Potential Assessment Questions: To Identify Patients’ and Their Family’s Priorities It is Essential to Ask Questions that will Help the Patient to Focus |
|---|---|
| Symptom management: The best possible way to treat the symptoms and to self-empower the patient for self-help in dealing with the symptom. |
|
| End-of-life decisions and expectations: Step-by-step and self-regulated decision-making, definition of personal preferences, and preventive planning for potential complications. |
|
| Network-organization: Professional (including out-of-hours support) and private care network. |
|
| Support of the carers: support system for family members, including in the bereavement phase, and for the involved professional carers. |
|
| Variables | Number | Percentage | |
|---|---|---|---|
| Gender (N = 55) | Male | 18 | 31.6% |
| Female | 37 | 64.9% | |
| Unknown | 2 | 3.5% | |
| Years in profession (N = 56) | Professional years in general (mean) | 19.4 | |
| Years working in PC (mean) | 7.6 | ||
| Profession (N = 57) | Physician | 26 | 45.6% |
| Nurse | 28 | 49.1% | |
| Pastoral carer | 2 | 3.5% | |
| Psycho(onco)logist | 1 | 1.8% | |
| Main place of work (N = 57) | Hospital-based general PC service | 7 | 13.0% |
| Hospital-based special PC service | 21 | 38.9% | |
| General practitioner | 9 | 16.7% | |
| Home care | 12 | 22.2% | |
| Nursing home | 6 | 11.1% | |
| Hospice | 2 | 3.7% | |
| ‘SENS’-Themes | Involved Professionals (Examples) |
|---|---|
| Symptoms | Physician, nurse, physiotherapist, psychologist, pastoral care worker, dietician, music or art therapist. |
| End-of-life decisions | Physician, nurse, pastoral care worker, psychologist. |
| Network-organization | Nurse, social worker, general practitioner, volunteer service. |
| Support of the carer | Physician, nurse, psychologist, spiritual counselor, social worker. |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fliedner, M.C.; Mitchell, G.; Bueche, D.; Mettler, M.; Schols, J.M.G.A.; Eychmueller, S. Development and Use of the ‘SENS’-Structure to Proactively Identify Care Needs in Early Palliative Care—An Innovative Approach. Healthcare 2019, 7, 32. https://doi.org/10.3390/healthcare7010032
Fliedner MC, Mitchell G, Bueche D, Mettler M, Schols JMGA, Eychmueller S. Development and Use of the ‘SENS’-Structure to Proactively Identify Care Needs in Early Palliative Care—An Innovative Approach. Healthcare. 2019; 7(1):32. https://doi.org/10.3390/healthcare7010032
Chicago/Turabian StyleFliedner, Monica C., Geoffrey Mitchell, Daniel Bueche, Monika Mettler, Jos M. G. A. Schols, and Steffen Eychmueller. 2019. "Development and Use of the ‘SENS’-Structure to Proactively Identify Care Needs in Early Palliative Care—An Innovative Approach" Healthcare 7, no. 1: 32. https://doi.org/10.3390/healthcare7010032
APA StyleFliedner, M. C., Mitchell, G., Bueche, D., Mettler, M., Schols, J. M. G. A., & Eychmueller, S. (2019). Development and Use of the ‘SENS’-Structure to Proactively Identify Care Needs in Early Palliative Care—An Innovative Approach. Healthcare, 7(1), 32. https://doi.org/10.3390/healthcare7010032






