How Effective Are Dietitians in Weight Management? A Systematic Review and Meta-Analysis of Randomized Controlled Trials
Abstract
:1. Introduction
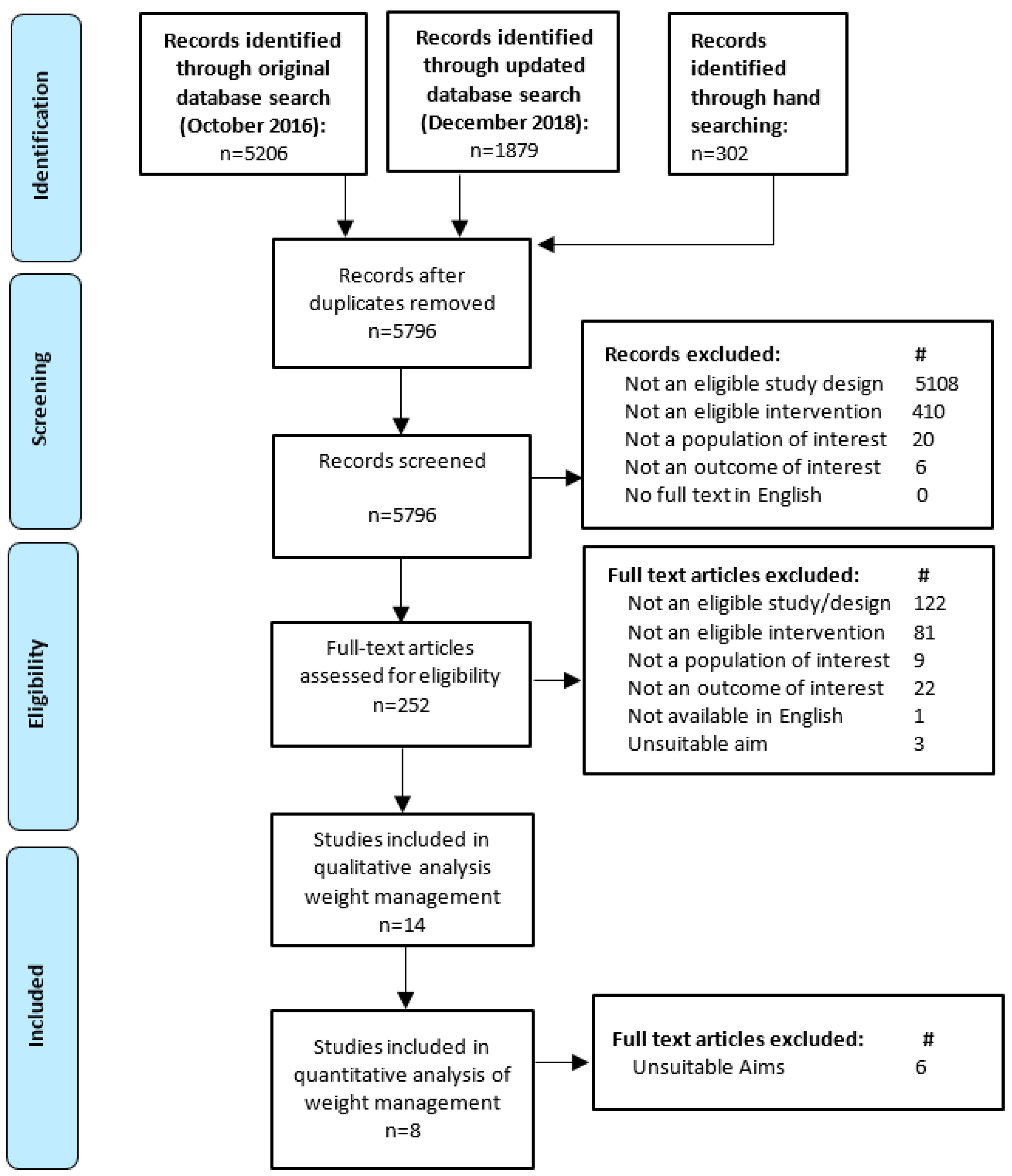
2. Materials and Methods
2.1. Systematic Review
2.2. Meta-Analyses
3. Results
3.1. Characteristics of Included Studies
3.2. Results of Included Studies
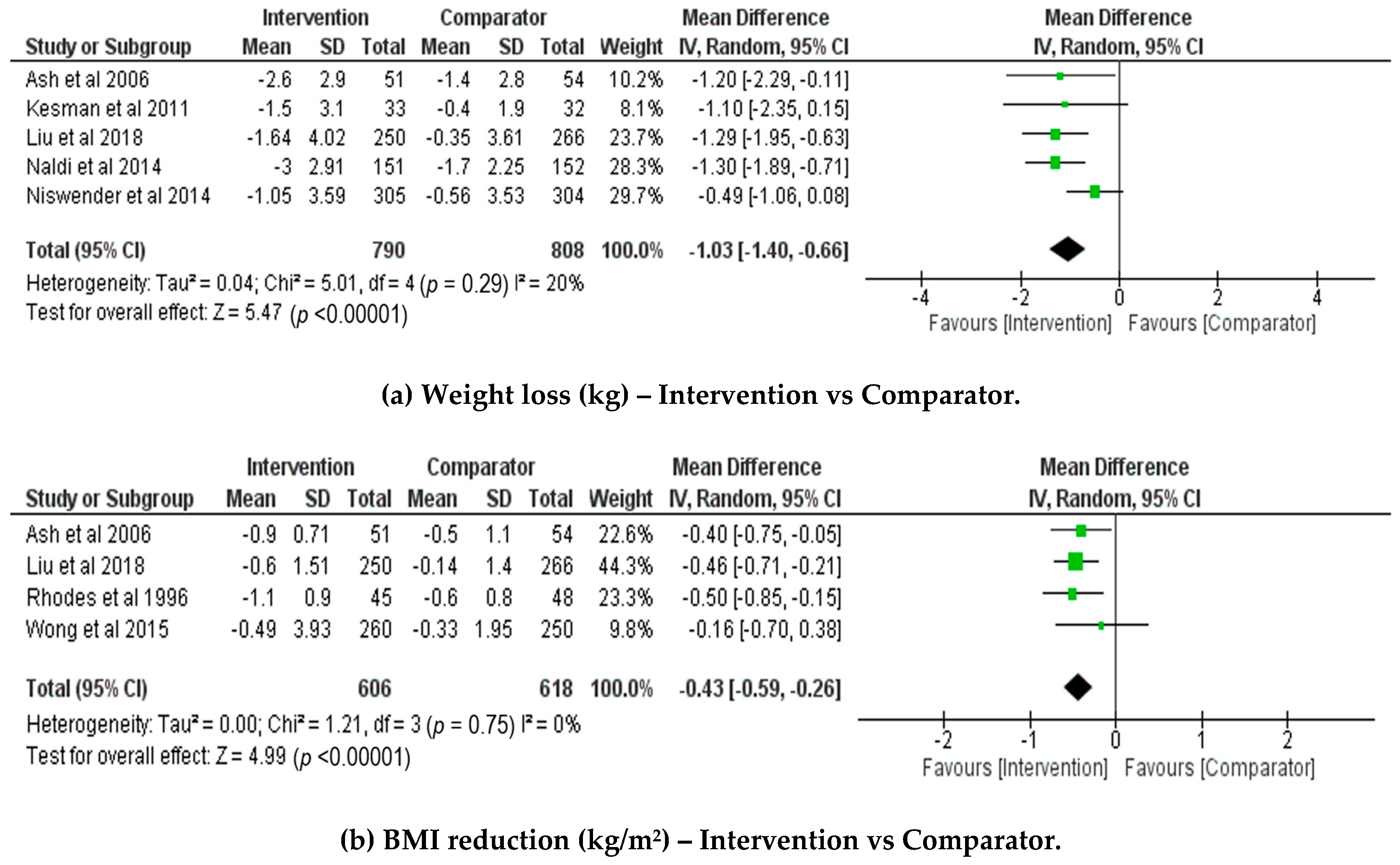
3.3. Meta-Analysis
4. Discussion
5. Limitations
6. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Roberto, C.A.; Swinburn, B.; Hawkes, C.P.; Huang, T.T.K.; Costa, S.A.; Ashe, M.; Zwicker, L.; Cawley, J.H.; Brownell, K.D. Patchy progress on obesity prevention: Emerging examples, entrenched barriers, and new thinking. Lancet 2015, 385, 2400–2409. [Google Scholar] [CrossRef]
- World Health Organization. Global Report on Diabetes; WHO: Geneva, Switzerland, 2016. [Google Scholar]
- Dietz, W.H.; Baur, L.A.; Hall, K.; Puhl, R.M.; Taveras, E.M.; Uauy, R.; Kopelman, P. Management of obesity: Improvement of health-care training and systems for prevention and care. Lancet 2015, 385, 2521–2533. [Google Scholar] [CrossRef]
- Bleich, S.N.; Bandara, S.; Bennett, W.; Cooper, L.A.; Gudzune, K.A. Enhancing the role of nutrition professionals in weight management: A cross-sectional survey. Obesity 2015, 23, 454–460. [Google Scholar] [CrossRef] [PubMed]
- Lacey, K.; Pritchett, E. Nutrition care process and model: Ada adopts road map to quality care and outcomes management. J. Am. Diet. Assoc. 2003, 103, 1061–1072. [Google Scholar] [PubMed]
- Raynor, H.A.; Champagne, C.M. Position of the academy of nutrition and dietetics: Interventions for the treatment of overweight and obesity in adults. J. Acad. Nutr. Diet. 2016, 116, 129–147. [Google Scholar] [CrossRef] [PubMed]
- BDA Obesity Specialist Group. Dietetic Obesity Management Interventions in Adults: Evidence Review & Clinical Application; British Dietetic Association: Birmingham, UK, 2018; pp. 1–102. [Google Scholar]
- National Institute for Clinical Excellence (NICE). Obesity: Guidance on the Prevention, Identification, Assessment and Management of Overweight and Obesity in Adults and Children; NICE: London, UK, 2006. [Google Scholar]
- Scottish Intercollegiate Guidelines Network (SIGN). Management of Obesity: A National Clinical Guideline; SIGN: Edinburgh, UK, 2010. [Google Scholar]
- Academy of Nutrition & Dietetics. Evidence Analysis Manual: Steps in the Academy Evidence Analysis Process; Research and Strategic Business Development, Academy of Nutrition & Dietetics: Chicago, IL, USA, 2012. [Google Scholar]
- Moller, G.; Andersen, H.; Snorgaard, O. A systematic review and meta-analysis of nutrition therapy compared with dietary advice in patients with type 2 diabetes. Am. J. Clin. Nutr. 2017, 106, 1394–1400. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; You, W.; Almeida, F.; Estabrooks, P.; Davy, B. The effectiveness and cost of lifestyle interventions including nutrition education for diabetes prevention: A systematic review and meta-analysis. J. Acad. Nutr. Diet. 2017, 117, 404–421.e436. [Google Scholar] [CrossRef] [PubMed]
- Mitchell, L.; Ball, L.; Ross, L.; Barnes, K.; Williams, L. Effectiveness of dietetic consultations in primary health care: A systematic review of randomized controlled trials. J. Acad. Nutr. Diet. 2017, 117, 1941–1962. [Google Scholar] [CrossRef] [PubMed]
- Higgins, J.P.T.; Green, S. Cochrane Handbook for Systematic Reviews of Interventions, Version 5.1.0. The Cochrane Collaboration, 2011. Available online: http://handbook.cochrane.org (accessed on 10 January 2019).
- Review Manager 5. RevMan, 5.3.5; The Nordic Cochrane Centre, The Cochrane Collaboration: Copenhagen, Denmark, 13 June 2014.
- Sulivan, L.; LaMorte, W. Interquartile Range (iqr). Available online: http://sphweb.bumc.bu.edu/otlt/mph-modules/bs/bs704_summarizingdata/bs704_summarizingdata7.html (accessed on 1 October 2018).
- Hozo, S.; Djulbegovic, B.; Hozo, I. Estimating the mean and variance from the median, range, and the size of a sample. BMC Med. Res. Methodol. 2005, 5, 13. [Google Scholar] [CrossRef] [PubMed]
- DerSimonian, R.; Kacker, R. Random-effects model for meta-analysis of clinical trials: An update. Contemp. Clin. Trials 2007, 28, 105–114. [Google Scholar] [CrossRef] [PubMed]
- Ash, S.; Reeves, M.; Bauer, J.; Dover, T.; Vivanti, A.; Leong, C.; O’Moore Sullivan, T.; Capra, S. A randomised control trial comparing lifestyle groups, individual counselling and written information in the management of weight and health outcomes over 12 months. Int. J. Obes. 2006, 30, 1557–1564. [Google Scholar]
- Delahanty, L.M.; Sonnenberg, L.M.; Hayden, D.; Nathan, D.M. Clinical and cost outcomes of medical nutrition therapy for hypercholesterolemia: A controlled trial. J. Am. Diet. Assoc. 2001, 101, 1012–1023. [Google Scholar] [CrossRef]
- Naldi, L.; Conti, A.; Cazzaniga, S.; Patrizi, A.; Pazzaglia, M.; Lanzoni, A.; Veneziano, L.; Pellacani, G. Diet and physical exercise in psoriasis: A randomized controlled trial. Br. J. Dermatol. 2014, 170, 634–642. [Google Scholar] [PubMed]
- Niswender, K.; Piletic, M.; Andersen, H.; Conradsen Hiort, L.; Hollander, P. Weight change upon once-daily initiation of insulin detemir with or without dietary intervention in overweight or obese insulin-naive individuals with type 2 diabetes: Results from the diet trial. Diabetes Obes. Metab. 2014, 16, 186–192. [Google Scholar] [PubMed]
- Rhodes, K.S.; Bookstein, L.C.; Aaronson, L.S.; Mercer, N.M.; Orringer, C.E. Intensive nutrition counseling enhances outcomes of national cholesterol education program dietary therapy. J. Am. Diet. Assoc. 1996, 96, 1003–1010. [Google Scholar] [CrossRef]
- Wong, M.C.; Wang, H.H.; Kwan, M.W.; Fong, B.C.; Chan, W.M.; de Zhang, X.; Li, S.T.; Yan, B.P.; Coats, A.J.; Griffiths, S.M. Dietary counselling has no effect on cardiovascular risk factors among chinese grade 1 hypertensive patients: A randomized controlled trial. Eur. Heart J. 2015, 36, 2598–2607. [Google Scholar] [PubMed]
- Liu, H.; Wang, L.; Zhang, S.; Leng, J.; Li, N.; Li, W.; Wang, J.; Tian, H.; Qi, L.; Yang, X.; et al. One-year weight losses in the tianjin gestational diabetes mellitus prevention programme: A randomized clinical trial. Diabetes Obes. Metab. 2018, 20, 1246–1255. [Google Scholar] [PubMed]
- Imai, S.; Kozai, H.; Matsuda, M.; Hasegawa, G.; Obayashi, H.; Togawa, C.; Yamamura, T.; Watanabe, K.; Miyatani, S.; Yoshikawa, T.; et al. Intervention with delivery of diabetic meals improves glycemic control in patients with type 2 diabetes mellitus. J. Clin. Biochem. Nutr. 2008, 42, 59–63. [Google Scholar] [CrossRef] [PubMed]
- Johnston, H.J.; Jones, M.; Ridler-Dutton, G.; Spechler, F.; Stokes, G.S.; Wyndham, L.E. Diet modification in lowering plasma cholesterol levels. A randomised trial of three types of intervention. Med. J. Aust. 1995, 162, 524–526. [Google Scholar] [PubMed]
- Kesman, R.L.; Ebbert, J.O.; Harris, K.I.; Schroeder, D.R. Portion control for the treatment of obesity in the primary care setting. BMC Res. Notes 2011, 4, 346. [Google Scholar] [CrossRef] [PubMed]
- Loprinzi, C.L.; Athmann, L.M.; Kardinal, C.G.; O’Fallon, J.R.; See, J.A.; Bruce, B.K.; Dose, A.M.; Miser, A.W.; Kern, P.S.; Tschetter, L.K.; et al. Randomized trial of dietician counseling to try to prevent weight gain associated with breast cancer adjuvant chemotherapy. Oncology 1996, 53, 228–232. [Google Scholar] [CrossRef] [PubMed]
- Ramsay, L.E.; Ramsay, M.H.; Hettiarachchi, J.; Davies, D.L.; Winchester, J. Weight reduction in a blood pressure clinic. Br. Med. J. 1978, 2, 244–245. [Google Scholar] [CrossRef] [PubMed]
- Wolff, S.; Legarth, J.; Vangsgaard, K.; Toubro, S.; Astrup, A. A randomized trial of the effects of dietary counseling on gestational weight gain and glucose metabolism in obese pregnant women. Int. J. Obes. 2008, 32, 495–501. [Google Scholar] [CrossRef] [PubMed]
- Huang, M.C.; Hsu, C.C.; Wang, H.S.; Shin, S.J. Prospective randomized controlled trial to evaluate effectiveness of registered dietitian-led diabetes management on glycemic and diet control in a primary care setting in taiwan. Diabetes Care 2010, 33, 233–239. [Google Scholar] [CrossRef] [PubMed]
- Williams, L.; Hollis, J.; Collins, C.; Morgan, P. Can a relatively low-intensity intervention by health professionals prevent weight gain in mid-age women? 12-month outcomes of the 40-something randomised controlled trial. Nutr. Diabetes 2014, 4, e116. [Google Scholar] [CrossRef] [PubMed]
- Lamminpää, R.; Vehviläinen-Julkunen, K.; Schwab, U. A systematic review of dietary interventions for gestational weight gain and gestational diabetes in overweight and obese pregnant women. Eur. J. Nutr. 2018, 57, 1721–1736. [Google Scholar] [CrossRef] [PubMed]
- Tapsell, L.; Neale, E. The effect of interdisciplinary interventions on risk factors for lifestyle disease: A literature review. Health Educ. Behav. 2016, 43, 271–285. [Google Scholar] [CrossRef] [PubMed]
- Neve, M.; Morgan, P.; Collins, C.E. Effectiveness of web-based interventions in achieving weight loss and weight loss maintenance in overweight and obese adults: A systematic review with meta-analysis. Obesity Rev. 2010, 11, 306–321. [Google Scholar] [CrossRef] [PubMed]
- Hutchesson, M.J.; Rollo, M.E.; Krukowski, R.; Ells, L.; Harvey, J.; Morgan, P.J.; Callister, R.; Plotnikoff, R.; Collins, C.E. Ehealth interventions for the prevention and treatment of overweight and obesity in adults: A systematic review with meta-analysis. Obes. Rev. 2015, 16, 376–392. [Google Scholar] [CrossRef] [PubMed]
- Ball, L.; Sladdin, I.; Mitchell, L.; Barnes, K.; Ross, L.; Williams, L. Quality of development and reporting of dietetic intervention studies in primary care: A systematic review of randomised controlled trials. J. Human Nutr. Diet. 2018, 31, 47–57. [Google Scholar] [CrossRef] [PubMed]
- Williams, R.L. Effectiveness of weight loss interventions—Is there a difference between men and women: A systematic review sex differences in men and women. Obes. Rev. 2015, 16, 171–186. [Google Scholar] [CrossRef] [PubMed]
- Joshi, M.; Royuela, A.; Zamora, J. Proper analysis in clinical trials: How to report and adjust for missing outcome data. BJOG Int. J. Obstet. Gynaecol. 2013, 120, 915–919. [Google Scholar] [CrossRef] [PubMed]
- Begg, C.; Cho, M.; Eastwood, S.; Horton, R.; Moher, D.; Olkin, I.; Pitkin, R.; Rennie, D.; Schulz, K.F.; Simel, D.; et al. Improving the quality of reporting of randomized controlled trials: The consort statement. JAMA 1996, 276, 637–639. [Google Scholar] [CrossRef] [PubMed]
- Moher, D.; Hopewell, S.; Schulz, K.F.; Montori, V.; Gotzche, P.; Devereaux, P.J.; Elbourne, D.; Egger, M.; Altman, D. Consort 2010 explanation and elaboration: Updated guidelines for reporting parallel group randomised trials. Br. Med. J. 2010, 340, 869. [Google Scholar] [CrossRef] [PubMed]
- Bell, M.L.; Fiero, M.; Horton, N.J.; Hsu, C.-H. Handling missing data in rcts; a review of the top medical journals. BMC Med. Res. Methodol. 2014, 14, 118. [Google Scholar] [CrossRef] [PubMed]
| Population: Adult patients who have received an individual dietetic consultation within a primary health care setting |
| Intervention: Individualized nutrition care for weight management provided by a dietitian in primary health care |
| Comparator: No intervention (including pre-intervention); usual care (where patients received usual care from another health professional or health program); and minimal care (nutrition-related print material, or a once-off general nutrition seminar) |
| Outcome: Change in any anthropometric measure: weight (including gestational weight gain), Body Mass Index, skinfolds, waist circumference, waist-to-hip ratio |
| Study design: Systematic reviews of Randomized Controlled Trials (RCTs) and RCTs |
| 1st Author Year, Country | Study Aim | Participants | Anthropometry Measures Other Metabolic Measures ITT or Per-Protocol Analysis # and Study End Point | INT ERVENTION: Dietary Advice, Consultation Number and Period; Total Time N Completed | Comparator N Completed | Risk of Bias | Mean (SD) Change in wt (kg), BMI (kg/m2), WC (cm) | ||
|---|---|---|---|---|---|---|---|---|---|
| Recruitment Criteria | Baseline Characteristics * | INT | CON | ||||||
| Studies citing decrease in weight or BMI as study aim or primary outcome variable | |||||||||
| Ash 2006 Australia [19] | Compare group wt reduction intervention to individual dietetic care and written information. | >18 y; BMI ≥ 27 kg/m2; OPC patients without cognitive impairment. | INT N = 65; 16M; 49F Age = 48 (13) y BMI = 34.2 (5.9) kg/m2 CON N = 54; 12M; 42F Age = 47 (14) y BMI = 35.8 (6.2) kg/m2 | Anthropometry: Wt, % body fat measured by trained researchers on bioelectrical impedance scales. BMI; WC (at umbilicus) Other metabolic: N/M # ITT (method unclear) used to analyze data at 6 months (N = 119) | Diet prescription aimed at wt loss of 0.5–1 kg/wk. 11 over 6 months; N/S. N = 49 | Written information N = 24 (Group education INT (N = 29) N/A) | Unclear | Wt b −2.6 (4.0) BMI −0.9 (1.1) WC b −4.8 (7.5) | Wt b −1.0 (3.4) BMI −0.4 (1.4) WC b −4.6 (7.2) |
| Kesman 2011 USA [28] | Assess effectiveness of weight loss diet counselling in obese adults in medical primary care practice. | 18-75y; Mayo OPC; BMI ≥30 to <40 kg/m2; without: cancer, pregnancy, AN, BN, psychiatric illness or surgery, gastric bypass, wt loss Tx. | INT N = 33; 13M; 20F Age = 55 (9.4) y Weight = 97.6 (12.8) kg CON N = 32; 12M; 20F Age = 56.3 (10.7) y Weight = 98.8 (12.5) kg | Anthropometry: % Wt change (primary) Wt collected on digital scales by researcher (shoes off) Other metabolic: N/M ITT (LOCF) used to analyze data at 6 months (N = 65) # Per protocol analysis: 65 at baseline; 42 at 6 months | Portion control plate: ¼ protein ½ vegetables; ¼ starch/grain 4 over 6 months (60 mins face to face + 3 phone); > 60 mins N = 19 | Written information N = 23 | High | Wt −1.0 (3.0) | Wt −0.5 (3.6) |
| Naldi 2014 Italy [21] | Assess dietary intervention plus physical exercise for weight loss on improving psoriasis in overweight or obese adults. | 18–80 y; BMI ≥ 25 kg/m2; Hx chronic plaque psoriasis (PASI 10+); without: other psoriasis, weight loss Tx, pregnant/lactating, other chronic disease. | INT N = 151; 114M; 37F Age = 53 (16.7 IQR) y BMI = 30.8 (6.4IQR) kg/m2 CON N = 152; 101M, 51F Age = 53 (21 IQR) y BMI = 30.8 (6 IQR) kg/m2 | Anthropometry: Wt and WC collected by trained researchers. BMI. Other metabolic: N/M Primary: PASI score change ITT (LOCF) used to analyze data at 20 weeks (N = 303) # Per Protocol analysis: 303 at baseline; 282 at 20 weeks | Week 1 to 12: EI: 0.8 x RMR; week 13–20: 1.0 x RMR. Fat = 30% EI, Carbohydrate = 55% EI, Protein = 15% EI 5 over 20 weeks (20 min each); 100 min N = 137 | Control (15 min session advising wt reduction for psoriasis control) N = 145 | Unclear | Wt a,b,c −3.0 (4.5) WC a,b,c −3.0 (5.0) | Wt b,c −1.7 (3.0) WC a,b,c −2.0 (3.5) |
| Niswender 2014 Multinational [22] | Determine impact of modest dietary intervention on weight change for overweight T2DM adults initiating insulin. | >18 y; BMI = 25–45 kg/m2; T2DM > 6m poorly controlled on metformin (HbA1c 7–9%); without: insulin, pregnancy, wt-affecting medications or conditions. | INT N = 306; 153M; 152F Age = 58.2 (9.7) y BMI = 34.4 (5.4) kg/m2 CON N = 305; 158M, 143F Age = 56.5 (10.0) y BMI = 34.3 (5.6) kg/m2 | Anthropometry: Wt change (primary); BMI; WC. Measurements collected as part of study but details N/S Other metabolic: FPG, PPG, HbA1c ITT (LOCF) used to analyze data at 26 weeks (N = 611) # | Decrease caloric intake by 15%. 6 (3 face-to-face 3 by phone) over 22 weeks; N/S N = 246 | Minimal care (basic lifestyle advice from the local investigator) N = 242 | Unclear | Wt b −1.05 (3.59) WC b −1.79 (4.54) | Wt b −0.56 (3.53) WC b −1.02 (5.03) |
| Ramsay 1978 Scotland [30] | Compare efficacy of advice by diet sheet, doctor and dietitian on weight loss to reduce BP in BP clinic adults. | Age range NS; overweight on clinical judgment; no dietitian visit in 6 months prior, no special diet for medical reasons. | INT N = 15; Age = N/S BMI = N/S CON N = 20; Age = N/S BMI = N/S | Anthropometry: Wt change (primary) Measured by clinicians Other metabolic: DBP; SBP Per Protocol analysis: 67 at baseline; 49 at 12 months | 3.3 MJ diet prescribed by dietitian. At least one over 12 months N/S N = 15 | Minimal care (doctor advice to lose weight) N = 20 (Diet sheet N/A N = 14) | High | Wt a,c −5.10 (−15 to 0) | Wt c −2.15 (−9 to +5)c |
| Studies citing effect on weight or BMI as part of study aim | |||||||||
| Liu 2018 China [25] | Overall study aim: to assess extent to which a dietitian intervention can prevent T2DM development in normal wt and overweight women with GDM over 5 years. Aim of this paper: to analyze weight change results after first year of the study. | 24–49 y; GDM in preceding 4 years (diagnosed by OGTT using WHO criteria) without medications to influence BGLs; chronic disease; current or planned pregnancy. | INT N = 586 Age = 32.3 (3.4) y BMI < 24 kg/m2) (NW) = 57.3% BMI ≥ 24 kg/m2 (OW) = 42.7% CON N = 594 Age = 32.4 (3.6) y BMI < 24 kg/m2 (NW) = 57.3% BMI ≥ 24 kg/m2 (OW) = 42.7% | Anthropometry: Wt change, BMI; % body fat, WC. Other metabolic: N/A ITT (missing values imputed) (N = 1180) # Per Protocol analysis: 1180 at baseline; 930 at 12 months. | BMI < 24 kg/m2 Fat < 30% EI, Carbohydrate 55–65% EI, 20–30 g Fibre/day prescribed by dietitian + 5 day diet and written handbook. BMI ≥ 24 kg/m2: as above plus 10% calorie reduction to lost 5–10%wt. 6 over first 4 weeks N/S N = 460 | Usual care: Two diet education sessions on T2DM prevention. N = 470 | All Wt a −0.64 (3.29) BMI a −0.25 (1.22) NW Wt −0.10 (2.37) BMI −0.004 (0.88) OW Wt a −1.64 (4.02) BMI a −0.60 (1.51) | All Wt −0.07 (2.93) BMI −0.03 (1.13) NW Wt −0.15 (2.20) BMI −0.06 (0.85) OW Wt −0.35 (3.61) BMI −0.14 (1.40) | |
| Rhodes 1996 USA [23] | Compare effect of OPC dietitian with usual care on nutrition, BMI, and lipids in initial hypercholesterolemia management. | 30–65 y; LDL-C >4.14 mmol/L or >3.36 mmol/L + other risk factors; without: T2DM, pregnancy, liver conditions, triglycerides >2.82 mmol/L, lipid lowering meds in past 2/12. | INT N = 45; Age = 47.5 (9) y Weight = N/S BMI = 28.1 (4.2) kg/m2 CON N = 48 Age = 47.5 (9) y Weight = N/S BMI = 28.3 (4.3) kg/m2 | Anthropometry: BMI Method of collecting weight and height N/S Other metabolic: N/M Per Protocol analysis: 100 at baseline; 93 at 3 months | F ≤ 30% EI; Saturated Fatty Acids ≤ 10% EI, <300mg cholesterol. 3 over 7 weeks (Initial 60 min, reviews 30 min); 2 hours. N = 45 | Minimal care (10 minutes of advice from Physician/nurse) N = 48 | Unclear | BMI a,b −1.1 (0.9) | BMI b −0.6 (0.8) |
| Wong 2015 China [24] | Compare DASH diet and dietitian counselling with usual care on BP, fasting lipid profile, and BMI. | 40–70 y; newly diagnosed grade I hypertension; without: medical conditions requiring dietary control, antihypertensive medication; | INT N = 281; 131M, 150F Age = 55.4 (5.6) y BMI = 24.17 (2.83) kg/m2 CON N = 275; 142M, 133F Age = 54.9 (5.2) y BMI = 24.23 (3.06) kg/m2 | Anthropometry: BMI (secondary). Wt measured by clinicians in indoor clothing and height on a wall-mounted stadiometer. Other metabolic: SBP, DBP (primary) ITT (LOCF) used to analyze data at 12 months (N = 556) # | DASH diet goals for food groups. 1 x 25 min over 6 months 25 min N = 243 | Usual care (physician) N = 242 | Unclear | BMI b −0.49 (3.93) | BMI −0.33 (1.95) |
| Studies citing weight gain prevention as study aim | |||||||||
| Loprinzi 1996 USA [29] | Can dietitian counselling prevent wt gain in women receiving adjuvant systemic chemotherapy for resected breast cancer. | 26–57 y; Women on chemotherapy post breast resection; without: special diet needs, wt >20% below IBW, conditions/medications causing wt gain or fluid retention. | INT N = 54; 54F Age = 43 y BMI = N/S CON N = 53; 53F Age = 43 (26–57) y BMI = N/S | Anthropometry: Wt change (primary). Measurements collected as part of study but details N/S Other metabolic: N/M Per protocol analysis: 109 at baseline; 107 at 6 months | Diet to limit wt gain to 5lb or less. 3 over 6 months; N/S N = 54 | Usual care (physician/nurse advice to prevent weight gain) N = 53 | High | Wt c,d +2.0 | Wt c,d +3.5 |
| Wolff 2008 Denmark [31] | Investigate if obese women can restrict GWG and pregnancy-induced increases in insulin, leptin, and glucose. | 19–45 y; BMI ≥ 30 kg/m2; singleton pregnancy; non-smokers; without complications affecting foetal growth. | INT N = 23; 23F Age = 28.7 (4) y BMI = 34.9 (4) kg/m2 CON N = 27; 27F Age = 30.7 (5) y BMI = 34.6 (3) kg/m2 | Anthropometry: GWG (primary): Wt measured by researchers at 36 weeks gestation— self-reported wt at conception Other metabolic: Fasting: Insulin; glucose, OGTT Per Protocol analysis: 66 at baseline; 50 at 36 weeks gestation | Total Energy Requirement = Basal Metabolic Rate X 1.4; Carbohydrate = 50–55% EI; Protein = 15–20% EI; Fat = 30% EI. 10 x 60 min over 24 weeks; 10 hours. N = 23 | Control (no advice on diet) N = 27 | High | Wt a +6.6(5.5) | Wt +13.3 (7.5) |
| Studies that measured weight or BMI without stating them in study aim or as primary or secondary outcome variables | |||||||||
| Delahanty 2001 USA [20] | To compare impact of cholesterol lowering protocol by dietitian with physician advice. | 21–65 y; cholesterol 5.2–8.84mmol/L; without: dietitian contact in 12 months, medical conditions/meds influencing lipids. | INT N = 45; 30M, 15F Age = 49 (10) y Weight = 79.6 (15.4) kg CON N = 45; 30M, 15F Age = 49 (9) y Weight = 83.2 (15.0) kg | Anthropometry: Wt change measured to nearest 0.1 kg by trained researchers Other metabolic: N/M ITT (method unclear) used to analyze data at 6 months (N = 90) # | NCEP cholesterol lowering protocol. 4 over 6 months (90 min in first 3 months and 30 min in months 4 to 6. 2 hours. N = 44 | Usual care (physician advice) N = 44 | Unclear | Wt a −1.9 (21.2) | Wt 0 (8.08) |
| Huang 2010 Taiwan [32] | Are T2DM patients who receive dietitian consultations more likely to follow glycaemic control diet. | 30–70 y; diagnosed T2DM; without pregnancy, dialysis, amputation, blindness, cancer or cardiovascular disease. | INT N = 75; 29M, 46F Age = 56.6 (8.0) y BMI = 25.7 (3.2) kg/m2 CON N = 79; 38M, 41F Age = 56.9 (7.5) y BMI = 27.0 (4.7) kg/m2 | Anthropometry: BMI changeWeight and height measurement technique N/S Other Metabolic: FPG; HbA1c (primary) Per protocol analysis: 39 dropouts and only analyzed the 154 analyzed at 12 months | Avoid excessive EI. Carbohydrate = 50–60% EI; Protein = 15–20% EI; Fat = 25–30% EI. 4 (30–60 mins each) over 12 months. 2–4 hours N = 57 | Usual care (summary of basic dietary principles by nurses) N = 58 | High | BMI 3.3 (1.2) | BMI 0.2 (1.5) |
| Imai 2008 Japan [26] | Investigate effect of individual dietetic counselling on glycaemic control in T2DM patients | 36–80 y; diagnosed T2DM; without: significant comorbidity. | INT N = 29; 13M, 16F Age = 62.0 (10.9) y BMI = 23.8 (4.1) kg/m2 CON N = 30; 14M, 16F Age = 64.3 (10.7) y BMI = 23.6 (2.9) kg/m2 | Anthropometry: BMI change Weight and height measurement technique N/S Other Metabolic: FPG; HbA1c (primary) ITT not stated but no dropouts reported: 77 at baseline and 12 months | General diet advice. 12 (20–30 mins each) over 12 months; 4–6 hours. N = 29 | Usual care (brief advice by Dr/Nurse) N = 30 (Food provision INT N/A N = 18) | High | BMI −0.3 (1.98) | BMI 0.3 (1.51) |
| Johnston 1995 Australia [27] | Compare efficacy of three diet and lifestyle interventions in lowering plasma lipids. | 24–81 y; BMI > 20 kg/m2; TC 5.5–8.0 mmol/L; without: T2DM, Coronary Artery Disease, uncontrolled hypertension, pregnancy, appetite suppressants, lipid lowering meds. | INT N = 44; 13M, 16F Age = 56 (N/S) y BMI = 24.2 (22.7–26.4 IQR) kg/m2 CON N = 47; 14M, 16F Age = 56 (N/S) y BMI = 25.1 (22.3–26.3IQR) kg/m2 | Anthropometry: Wt change Measurements collected as part of study but details N/S Other metabolic: N/M Per protocol analysis: 179 at baseline; 131 at 6 months | Diet change strategies: food planning, cooking methods, recipe modification. 3 over unstated period; N/S N = 44 | Minimal care (written information) N = 47 (Group counselling INT N/A N = 40) | High | Wt b,c −1.0 (−3.0 to 0.0) | Wt c −1.0 (−2.0 to +1.0) |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Williams, L.T.; Barnes, K.; Ball, L.; Ross, L.J.; Sladdin, I.; Mitchell, L.J. How Effective Are Dietitians in Weight Management? A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Healthcare 2019, 7, 20. https://doi.org/10.3390/healthcare7010020
Williams LT, Barnes K, Ball L, Ross LJ, Sladdin I, Mitchell LJ. How Effective Are Dietitians in Weight Management? A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Healthcare. 2019; 7(1):20. https://doi.org/10.3390/healthcare7010020
Chicago/Turabian StyleWilliams, Lauren T., Katelyn Barnes, Lauren Ball, Lynda J. Ross, Ishtar Sladdin, and Lana J. Mitchell. 2019. "How Effective Are Dietitians in Weight Management? A Systematic Review and Meta-Analysis of Randomized Controlled Trials" Healthcare 7, no. 1: 20. https://doi.org/10.3390/healthcare7010020
APA StyleWilliams, L. T., Barnes, K., Ball, L., Ross, L. J., Sladdin, I., & Mitchell, L. J. (2019). How Effective Are Dietitians in Weight Management? A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Healthcare, 7(1), 20. https://doi.org/10.3390/healthcare7010020





