Ward-Based Non-Invasive Ventilation in Acute Exacerbations of COPD: A Narrative Review of Current Practice and Outcomes in the UK
Abstract
1. Introduction
2. Non-Invasive Ventilation in AECOPD
3. Development of Ward-Based NIV
4. Ward-Based NIV as an Alternative to Critical Care Admission
5. Ward-Based NIV in Patients with a ‘Do Not Intubate’ Order
6. ‘Real life’ Outcomes following NIV in the UK
7. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- National Institute for Health and Care Excellence. Chronic obstructive pulmonary disease in over 16s: diagnosis and management. Clinical guideline [CG101]. Available online: https://www.nice.org.uk/guidance/CG101 (accessed on 28 October 2018).
- Lozano, R.; Naghavi, M.; Foreman, K.; Lim, S.; Shibuya, K.; Aboyans, V.; Abraham, J.; Adair, T.; Aggarwal, R.; Ahn, S.Y.; et al. Global and regional mortality from 235 causes of death for 20 age groups in 1990 and 2010: A systematic analysis for the Global Burden of Disease Study 2010. Lancet 2012, 380, 2095–2128. [Google Scholar] [CrossRef]
- GBD 2016 Causes of Death Collaborators. Global, regional, and national age-sex specific mortality for 264 causes of death, 1980–2016: A systematic analysis for the Global Burden of Disease Study 2016. Lancet 2017, 390, 1151–1210. [CrossRef]
- Merinopoulou, E.; Raluy-Callado, M.; Ramagopalan, S.; MacLachlan, S.; Khalid, J.M. COPD exacerbations by disease severity in England. Int. J. Chron. Obstruct. Pulmon. Dis. 2016, 11, 697–709. [Google Scholar] [PubMed]
- Kim, V.; Aaron, S.D. What is a Copd Exacerbation? Current Definitions, Pitfalls, Challenges and Opportunities for Improvement. Eur. Respir. J. 2018, 52, 1801261. [Google Scholar] [CrossRef] [PubMed]
- Vogelmeier, C.F.; Criner, G.J.; Martinez, F.J.; Anzueto, A.; Barnes, P.J.; Bourbeau, J.; Celli, B.R.; Chen, R.; Decramer, M.; Fabbri, L.M.; et al. Global Strategy for the Diagnosis, Management, and Prevention of Chronic Obstructive Lung Disease 2017 Report: GOLD Executive Summary. Eur. Respir. J. 2017, 49, 1700214. [Google Scholar] [CrossRef] [PubMed]
- Roberts, C.M.; Stone, R.A.; Buckingham, R.J.; Pursey, N.A.; Lowe, D.; National Chronic Obstructive Pulmonary Disease Resources and Outcomes Project implementation group. Acidosis, non-invasive ventilation and mortality in hospitalised COPD exacerbations. Thorax 2011, 66, 43–48. [Google Scholar] [CrossRef] [PubMed]
- Bruno, C.M.; Valenti, M. Acid-base disorders in patients with chronic obstructive pulmonary disease: A pathophysiological review. J. Biomed. Biotechnol. 2012, 2012, 915150. [Google Scholar] [CrossRef]
- Hartl, S.; Lopez-Campos, J.L.; Pozo-Rodriguez, F.; Castro-Acosta, A.; Studnicka, M.; Kaiser, B.; Roberts, C.M. Risk of death and readmission of hospital-admitted COPD exacerbations: European COPD Audit. Eur. Respir. J. 2016, 47, 113–121. [Google Scholar] [CrossRef]
- Lindenauer, P.K.; Dharmarajan, K.; Qin, L.; Lin, Z.; Gershon, A.S.; Krumholz, H.M. Risk trajectories of readmission and death in the first year after hospitalization for chronic obstructive pulmonary disease. Am. J. Respir. Crit. Care Med. 2018, 197, 1009–1017. [Google Scholar] [CrossRef]
- Osadnik, C.R.; Tee, V.S.; Carson-Chahhoud, K.V.; Picot, J.; Wedzicha, J.A.; Smith, B.J. Non-invasive ventilation for the management of acute hypercapnic respiratory failure due to exacerbation of chronic obstructive pulmonary disease. Cochrane. Database Syst. Rev. 2017, 7, CD004104. [Google Scholar] [CrossRef]
- Davidson, A.C.; Banham, S.; Elliott, M.; Kennedy, D.; Gelder, C.; Glossop, A.; Church, A.C.; Creagh-Brown, B.; Dodd, J.W.; Felton, T.; et al. BTS/ICS guideline for the ventilatory management of acute hypercapnic respiratory failure in adults. Thorax 2016, 71 Suppl 2, ii 1–35. [Google Scholar] [CrossRef]
- Wedzicha, J.A.; Ers, C.; Miravitlles, M.; Hurst, J.R.; Calverley, P.M.; Albert, R.K.; Anzueto, A.; Criner, G.J.; Papi, A.; Rabe, K.F.; et al. Management of COPD exacerbations: A European Respiratory Society/American Thoracic Society guideline. Eur. Respir. J. 2017, 49, 1600791. [Google Scholar] [CrossRef] [PubMed]
- Rochwerg, B.; Brochard, L.; Elliott, M.W.; Hess, D.; Hill, N.S.; Nava, S.; Navalesi, P.; Antonelli, M.; Brozek, J.; Conti, G.; et al. Official ERS/ATS clinical practice guidelines: Noninvasive ventilation for acute respiratory failure. Eur. Respir. J. 2017, 50, 1602426. [Google Scholar] [CrossRef] [PubMed]
- Ugurlu, A.O.; Sidhom, S.S.; Khodabandeh, A.; Ieong, M.; Mohr, C.; Lin, D.Y.; Buchwald, I.; Bahhady, I.; Wengryn, J.; Maheshwari, V.; et al. Where is noninvasive ventilation actually delivered for acute respiratory failure? Lung 2015, 193, 779–788. [Google Scholar] [CrossRef] [PubMed]
- Mukherjee, R.; Nenna, R.; Turner, A. Early ward-based acute noninvasive ventilation: A paper that changed practice. Breathe 2018, 14, 153–155. [Google Scholar] [CrossRef] [PubMed]
- Plant, P.K.; Owen, J.L.; Elliott, M.W. Early use of non-invasive ventilation for acute exacerbations of chronic obstructive pulmonary disease on general respiratory wards: A multicentre randomised controlled trial. Lancet 2000, 355, 1931–1935. [Google Scholar] [CrossRef]
- Confalonieri, M.; Gorini, M.; Ambrosino, N.; Mollica, C.; Corrado, A.; Scientific Group on Respiratory Intensive Care of the Italian Association of Hospital Pneumonologists. Respiratory intensive care units in Italy: a national census and prospective cohort study. Thorax 2001, 56, 373–378. [Google Scholar] [CrossRef]
- Dikensoy, O.; Ikidag, B.; Filiz, A.; Bayram, N. Comparison of non-invasive ventilation and standard medical therapy in acute hypercapnic respiratory failure: A randomised controlled study at a tertiary health centre in SE Turkey. Int. J. Clin. Pract. 2002, 56, 85–88. [Google Scholar]
- Fernández, J.G.; López-Campos, J.B.; Perea-Milla, E.L.; Pons, J.P.; Rivera, R.I.; Moreno, L.A. Non invasive ventilation for acute exacerbation of chronic obstructive pulmonary disease: A meta-analysis. Med. Clin. 2003, 120, 281–286. [Google Scholar]
- Scala, R.; Naldi, M.; Archinucci, I.; Coniglio, G. Non-invasive positive pressure ventilation in acute hypercapnic respiratory failure: Clinical experience of a respiratory ward. Monaldi. Arch. Chest Dis. 2004, 61, 94–101. [Google Scholar] [CrossRef]
- Collaborative Research Group of Noninvasive Mechanical Ventilation for Chronic Obstructive Pulmonary Disease. Early use of non-invasive positive pressure ventilation for acute exacerbations of chronic obstructive pulmonary disease: A multicentre randomized controlled trial. Chin. Med. J. 2005, 118, 2034–2040. [Google Scholar]
- Farha, S.; Ghamra, Z.W.; Hoisington, E.R.; Butler, R.S.; Stoller, J.K. Use of noninvasive positive-pressure ventilation on the regular hospital ward: Experience and correlates of success. Respir. Care 2006, 51, 1237–1243. [Google Scholar] [PubMed]
- Carrera, M.; Marin, J.M.; Anton, A.; Chiner, E.; Alonso, M.L.; Masa, J.F.; Marrades, R.; Sala, E.; Carrizo, S.; Giner, J.; et al. A controlled trial of noninvasive ventilation for chronic obstructive pulmonary disease exacerbations. J. Crit. Care 2009, 24, e7–e14. [Google Scholar] [CrossRef] [PubMed]
- McLaughlin, K.M.; Murray, I.M.; Thain, G.; Currie, G.P. Ward-based non-invasive ventilation for hypercapnic exacerbations of COPD: A ‘real-life’ perspective. QJM 2010, 103, 505–510. [Google Scholar] [CrossRef] [PubMed]
- Ciledag, A.; Kaya, A.; Akdogan, B.B.; Kabalak, P.A.; Onen, Z.P.; Seny, E.; Gulbay, B. Early use of noninvasive mechanical ventilation in patients with acute hypercapnic respiratory failure in a respiratory ward: A prospective study. Arch. Bronconeumol. 2010, 46, 538–542. [Google Scholar] [CrossRef]
- Khalid, I.; Sherbini, N.; Qushmaq, I.; Qabajah, M.R.; Nisar, A.; Khalid, T.J.; Wasfy, J.H. Outcomes of patients treated with noninvasive ventilation by a medical emergency team on the wards. Respir. Care 2014, 59, 186–192. [Google Scholar] [CrossRef] [PubMed]
- Olivieri, C.; Carenzo, L.; Vignazia, G.L.; Campanini, M.; Pirisi, M.; Della Corte, F.; Navalesi, P. Does noninvasive ventilation delivery in the ward provide early effective ventilation? Respir. Care 2015, 60, 6–11. [Google Scholar] [CrossRef]
- Fiorino, S.; Bacchi-Reggiani, L.; Detotto, E.; Battilana, M.; Borghi, E.; Denitto, C.; Dickmans, C.; Facchini, B.; Moretti, R.; Parini, S.; et al. Efficacy of non-invasive mechanical ventilation in the general ward in patients with chronic obstructive pulmonary disease admitted for hypercapnic acute respiratory failure and pH < 7.35: A feasibility pilot study. Intern. Med. J. 2015, 45, 527–537. [Google Scholar]
- Parker, K.; Perikala, V.; Aminazad, A.; Deng, Z.; Borg, B.; Buchan, C.; Toghill, J.; Irving, L.B.; Goldin, J.; Charlesworth, D.; et al. Models of care for non-invasive ventilation in the Acute COPD Comparison of three Tertiary hospitals (ACT3) study. Respirology 2018, 23, 492–497. [Google Scholar] [CrossRef]
- Dave, C.; Turner, A.; Thomas, A.; Beauchamp, B.; Chakraborty, B.; Ali, A.; Mukherjee, R.; Banerjee, D. Utility of respiratory ward-based NIV in acidotic hypercapnic respiratory failure. Respirology 2014, 19, 1241–1247. [Google Scholar] [CrossRef]
- Yalcinsoy, M.; Salturk, C.; Oztas, S.; Gungor, S.; Ozmen, I.; Kabadayi, F.; Oztim, A.A.; Aksoy, E.; Adıguzel, N.; Oruc, O.; et al. Can patients with moderate to severe acute respiratory failure from COPD be treated safely with noninvasive mechanical ventilation on the ward? Int. J. Chron. Obstruct. Pulmon. Dis. 2016, 11, 1151–1160. [Google Scholar] [CrossRef] [PubMed]
- Plant, P.K.; Owen, J.L.; Parrott, S.; Elliott, M.W. Cost effectiveness of ward based non-invasive ventilation for acute exacerbations of chronic obstructive pulmonary disease: Economic analysis of randomised controlled trial. BMJ 2003, 326, 956. [Google Scholar] [CrossRef] [PubMed]
- Patel, S.P.; Pena, M.E.; Babcock, C.I. Cost-effectiveness of noninvasive ventilation for chronic obstructive pulmonary disease-related respiratory failure in Indian hospitals without ICU facilities. Lung India 2015, 32, 549–556. [Google Scholar] [PubMed]
- Yeung, J.; Couper, K.; Ryan, E.G.; Gates, S.; Hart, N.; Perkins, G.D. Non-invasive ventilation as a strategy for weaning from invasive mechanical ventilation: A systematic review and Bayesian meta-analysis. Intensive Care Med. 2018, 44, 2192–2204. [Google Scholar] [CrossRef] [PubMed]
- Ozsancak Ugurlu, A.; Habesoglu, M.A. Epidemiology of NIV for Acute Respiratory Failure in COPD Patients: Results from the International Surveys vs. the “Real World”. COPD 2017, 14, 429–438. [Google Scholar] [CrossRef] [PubMed]
- Wilson, M.E.; Majzoub, A.M.; Dobler, C.C.; Curtis, J.R.; Nayfeh, T.; Thorsteinsdottir, B.; Barwise, A.K.; Tilburt, J.C.; Gajic, O.; Montori, V.M.; et al. Noninvasive Ventilation in Patients With Do-Not-Intubate and Comfort-Measures-Only Orders: A Systematic Review and Meta-Analysis. Crit. Care Med. 2018, 46, 1209–1216. [Google Scholar] [CrossRef]
- British Thoracic Society. BTS Reports: NIV Audit 2013. Available online: https://www.brit-thoracic.org.uk/document-library/audit-and-quality-improvement/audit-reports/bts-adult-niv-audit-report-2013/ (accessed on 28 October 2018).
- National Confidential Enquiry into Patient Outcome and Death. Inspiring Change. London. 2017. Available online: https://www.ncepod.org.uk/2017niv.html (accessed on 28 October 2018).
- Davies, M.; Allen, M.; Bentley, A.; Bourke, S.C.; Creagh-Brown, B.; D’Oliveiro, R.; Glossop, A.; Gray, A.; Jacobs, P.; Mahadeva, R.; et al. British Thoracic Society Quality Standards for acute non-invasive ventilation in adults. BMJ Open Respir. Res. 2018, 5, e000283. [Google Scholar] [CrossRef]
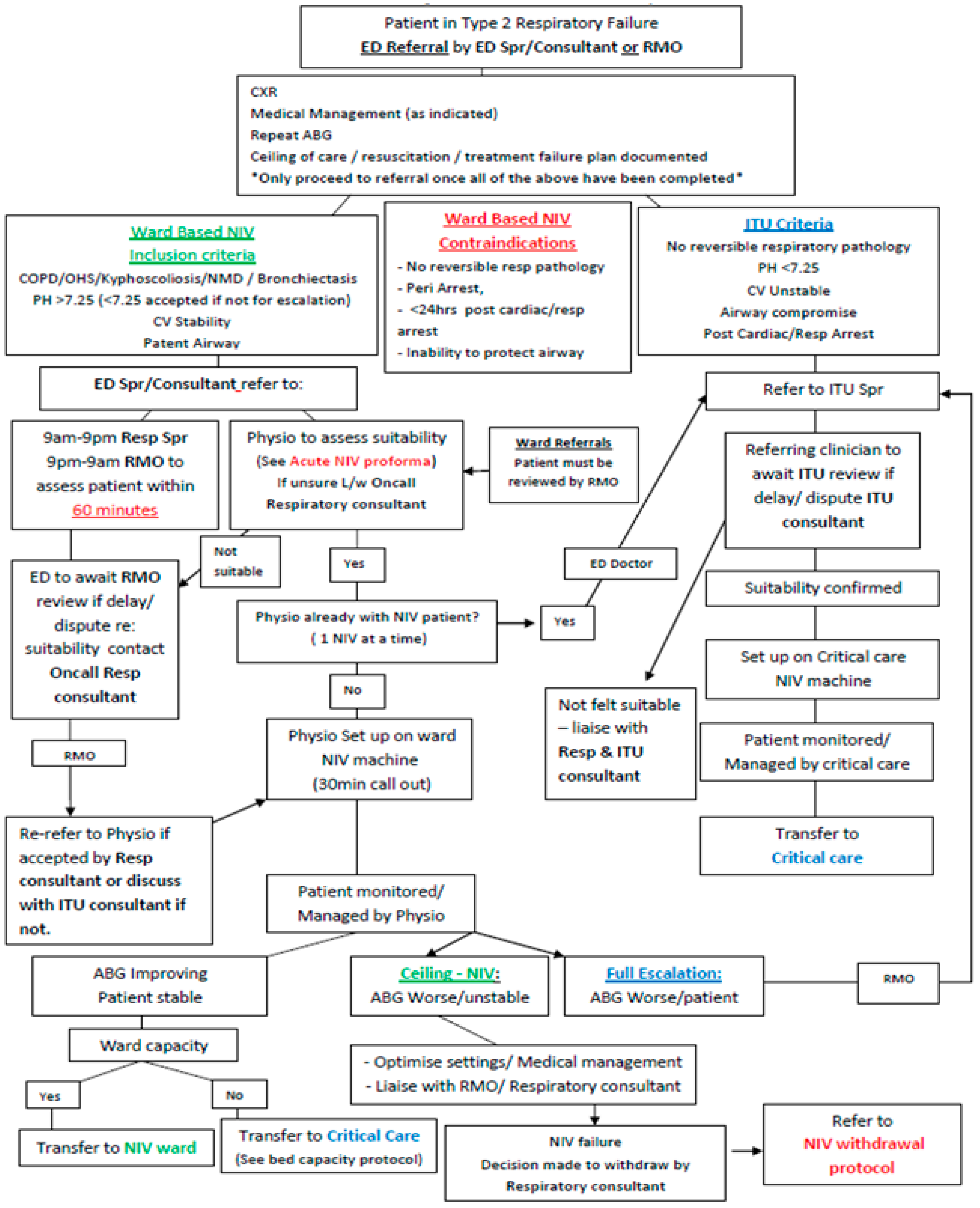
| 1 | Acute NIV should be offered to all patients who meet evidence-based criteria. Hospitals must ensure there is adequate capacity to provide NIV to all eligible patients. |
| 2 | All staff who prescribe, initiate, or make changes to acute NIV treatment should have evidence of training and maintenance of competencies appropriate for their role. |
| 3 | Acute NIV should only be carried out in specified clinical areas designated for the delivery of acute NIV. |
| 4 | Patients who meet evidence-based criteria for acute NIV should start NIV within 60 min of the blood gas result associated with the clinical decision to provide NIV and within 120 min of hospital arrival for patients who present acutely. |
| 5 | All patients should have a documented escalation plan before starting treatment with acute NIV. Clinical progress should be reviewed by a healthcare professional with appropriate training and competence within 4 h and by a consultant with training and competence in acute NIV within 14 h of starting acute NIV. |
| 6 | All patients treated with acute NIV should have blood gas analysis performed within 2 h of starting acute NIV; failure of these blood gas measurements to improve should trigger specialist healthcare professional review within 30 min. |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Trethewey, S.P.; Edgar, R.G.; Turner, A.M.; Mukherjee, R. Ward-Based Non-Invasive Ventilation in Acute Exacerbations of COPD: A Narrative Review of Current Practice and Outcomes in the UK. Healthcare 2018, 6, 145. https://doi.org/10.3390/healthcare6040145
Trethewey SP, Edgar RG, Turner AM, Mukherjee R. Ward-Based Non-Invasive Ventilation in Acute Exacerbations of COPD: A Narrative Review of Current Practice and Outcomes in the UK. Healthcare. 2018; 6(4):145. https://doi.org/10.3390/healthcare6040145
Chicago/Turabian StyleTrethewey, Samuel P., Ross G. Edgar, Alice M. Turner, and Rahul Mukherjee. 2018. "Ward-Based Non-Invasive Ventilation in Acute Exacerbations of COPD: A Narrative Review of Current Practice and Outcomes in the UK" Healthcare 6, no. 4: 145. https://doi.org/10.3390/healthcare6040145
APA StyleTrethewey, S. P., Edgar, R. G., Turner, A. M., & Mukherjee, R. (2018). Ward-Based Non-Invasive Ventilation in Acute Exacerbations of COPD: A Narrative Review of Current Practice and Outcomes in the UK. Healthcare, 6(4), 145. https://doi.org/10.3390/healthcare6040145






