Precision Medicine: The Role of the MSIDS Model in Defining, Diagnosing, and Treating Chronic Lyme Disease/Post Treatment Lyme Disease Syndrome and Other Chronic Illness: Part 2
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Methodology
2.3. Data Mining Procedure
2.4. Laboratory Testing
3. Results
3.1. MSIDS (Data Mining)
3.2. Patients Have More Than Lyme Disease: MSIDS Multifactorial Analysis
- Immune Dysfunction (positive ANA, RF, HLADR2, HLADR4: 145 (72.5%) participants had immune dysfunction, 13.5% had elevated IgM antibodies, and up to 85% had some form of immune deficiency:
- ○
- 20.6% had total IgG deficiency;
- ○
- 19.3% had IgM deficiency; and
- ○
- 15.9% had IgA deficiency.
85.5% had combined IgG subclass deficiencies 1–4 (see Table 2). - Inflammation (Elevated ESR, CRP, TGFB1, C3a, C4a, TNF, VEGF): 139 (69.5%) participants had markers of inflammation.
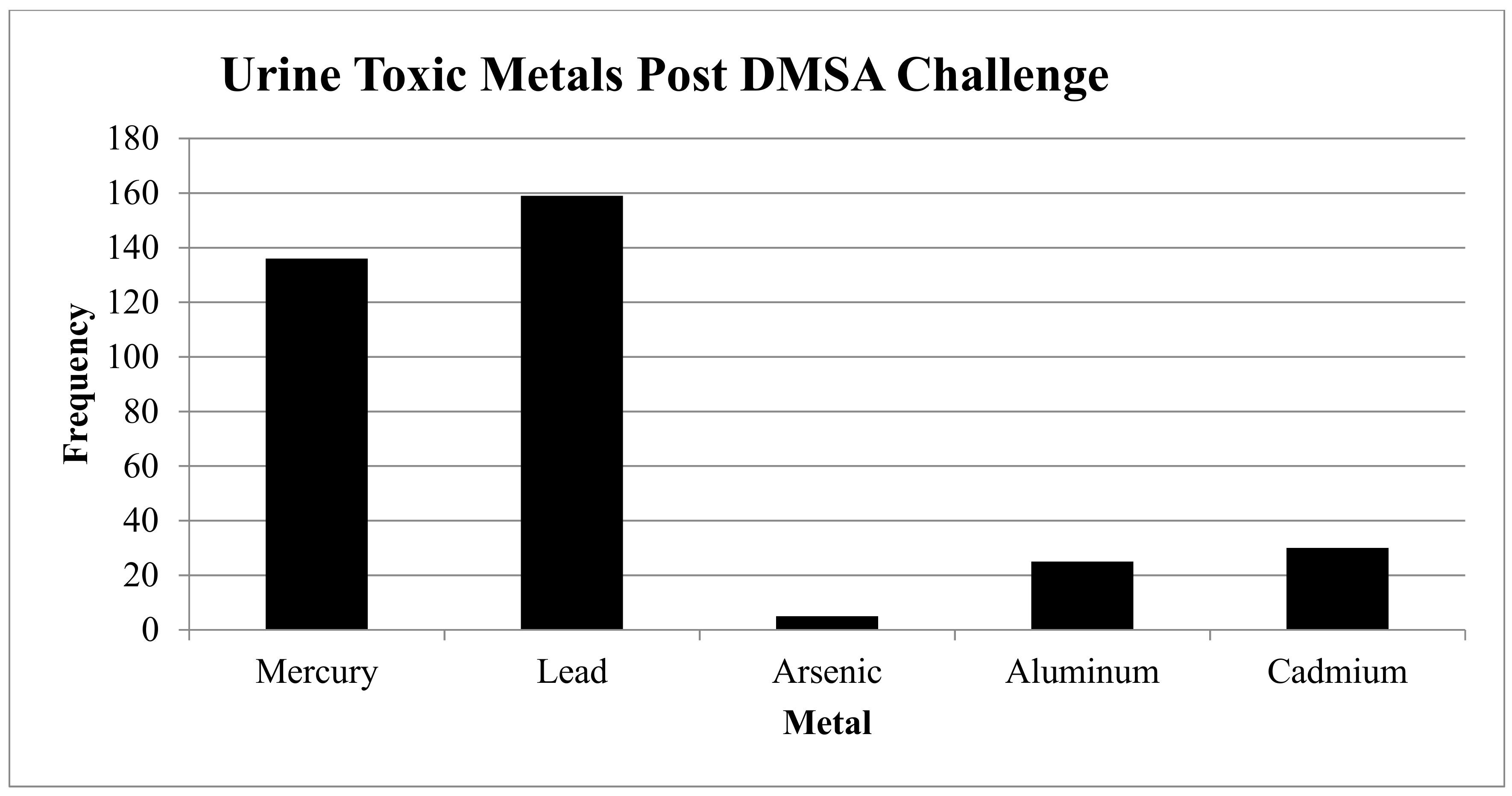
- Toxicity: See Figure 2.
- ○
- Heavy Metals: 169/185 (84.5%) had one or more elevated heavy metals using a 6-h urine DMSA challenge:
- ▪
- 159 (79.5%) had elevated lead levels (N = 73 were elevated, and N = 59 were very elevated);
- ▪
- 136 (68%) had elevated mercury levels (N = 77 were elevated, and N = 59 were very elevated);
- ▪
- 5 (2.5%) had elevated arsenic levels (N = 3 were elevated, and N = 2 were very elevated);
- ▪
- 25 (12.5%) had elevated aluminum levels (N = 25 were elevated); and
- ▪
- 30 (15%) had elevated cadmium levels (N = 26 were elevated, and N = 4 were very elevated).
- ○
- Mold: 30/42 (71.4%) had one or more elevated mold levels:
- ▪
- 13/25 (52%) had elevated aflatoxins;
- ▪
- 18/26 (69%) had elevated ochratoxins;
- ▪
- 20/26 (76.9%) had elevated trichothecenes;
- ▪
- 17/17 (100%) had elevated gliotoxins; and
- ▪
- 7/18 (38.9%) had other elevated mold (Stachybotrys exposure).
- ○
- Pesticides: 5 (2.5%) tested positive for pesticides*.
- Allergies: 163 (81.5%) of participants had allergies:
- ○
- 90 (45%) had food allergies;
- ○
- 43 (21.5%) had environmental allergies (e.g., seasonal allergies, allergy to animals, etc.);
- ○
- 7 (3.5%) had high IgE levels;
- ○
- 3 (1.5%) had high histamine levels (not all patients were tested for histamine sensitivity or Mast Cell Activation Disorder [MCAD]);
- ○
- 112 (56%) had drug allergies; and
- ○
- 12 (6%) had allergies categorized as “other”.
- Nutritional and Enzyme Deficiencies: 152 (76%) participants had one or more of these deficiencies. All patients were tested for mineral deficiencies, but only patients with poor nutritional intake were tested for amino acid and/or fatty acid deficiencies:
- ○
- 5 (2.5%) had amino acid deficiencies;
- ○
- 2 (1%) had fatty acid deficiencies;
- ○
- 36 (18%) had iodine deficiencies;
- ○
- 14 (7%) had copper deficiencies;
- ▪
- 3 (1.5%) had deficiencies in serum copper;
- ▪
- 6 (3%) had deficiencies in red blood cell [RBC] copper;
- ▪
- 5 (2.5%) had deficiencies in plasma copper;
- ○
- 31 (16%) had magnesium deficiencies;
- ▪
- 6 (3%) had deficiencies in serum magnesium;
- ▪
- 26 (13%) had deficiencies in RBC magnesium;
- ○
- 36 (18%) had zinc deficiencies;
- ▪
- 22 (11%) had deficiencies in serum zinc;
- ▪
- 7 (3.5%) had deficiencies in RBC zinc;
- ▪
- 7 (3.5%) had deficiencies in plasma zinc; and
- ○
- 105 (52.5%) had MTHFR mutations.
- Mitochondrial Dysfunction (defined by those who had positive responses to the following mitochondrial support supplements: ATP fuel (NT Factors, i.e., glycosylated phospholipids), Coenzyme Q10 (CoQ10), acetyl-l-carnitine, d-ribose): 15 (7.5%) had mitochondrial dysfunction.
- Psychological issues: 177 (88.5%) participants self-reported having at least one psychological problem:
- ○
- 154 (77%) had depression;
- ○
- 134 (67%) had anxiety;
- ○
- 4 (2%) had Obsessive Compulsive Disorder (OCD);
- ○
- 11 (5.5%) had Post Traumatic Stress Disorder (PTSD); and
- ○
- 9 (4.5%) had other psychological issues.
- Neurological Dysfunction: 190 (95%) had at least one of the following Neurological symptoms/disorders:
- ○
- 188 (94%) had neuropathy;
- ○
- 5 (2.5%) had Chronic Inflammatory Demyelinating Polyneuropathy (CIDP);
- ○
- 3 (1.5%) had Multiple Sclerosis;
- ○
- 2 (1%) had seizures; and
- ○
- 2 (1%) had other neurological issues (e.g., Parkinson’s symptoms).
- Endocrine Abnormalities: 195 (97.5%) had at least one of the following endocrine abnormalities:
- ○
- 121 (60.5%) had thyroid abnormalities;
- ○
- 144 (72%) had adrenal abnormalities;
- ○
- 82 (41%) had sex hormone abnormalities;
- ○
- 136 (68%) had vitamin D deficiencies;
- ○
- 3 (1.5%) had pregnenolone deficiencies; and
- ○
- 74 (37%) had DHEA abnormalities.
- Sleep Disorders: 196 (98%) had at least one of the following sleep disorders:
- ○
- 23 (11.5%) had Obstructive Sleep Apnea (OSA);
- ○
- 1 (0.5%) had Restless Leg Syndrome (RLS);
- ○
- 7 (3.5%) had Benign Prostatic Hyperplasia (BPH);
- ○
- 4 (2%) were in menopause;
- ○
- 2 (1%) had high adrenals;
- ○
- 1 (0.5%) had medication induced sleep problems; and
- ○
- 189 (94.5%) had other sleep problems, i.e., difficulties with insomnias, hypersomnias, circadian rhythm disorders (secondary to Lyme and tick-borne diseases).
- Autonomic Nervous System (ANS) Dysfunction/POTS: 83 (41.5%) had ANS dysfunction and/or POTS:
- ○
- 23 (11.5%) had mild POTS (1–10 mm Hg drop in BP, and/or 1–10-point increase in heart rate after standing);
- ○
- 41 (20.5%) had moderate POTS (11–29 mm drop in BP, and/or 11–29-point increase in heart rate after standing);
- ○
- 9 (4.5%) had severe POTS (30+ increase in heart rate standing);
- ○
- 19 (9.5%) had dysautonomia (e.g., gastroparesis, chronic constipation, bladder dysfunction, or dysfunction in temperature regulation); and
- ○
- 2 (1%) had ‘other’ (tremors and/or discoloration hands/feet).
- Gastrointestinal Dysfunction: 159 (79.5%) had one or more of the following gastrointestinal disorders:
- ○
- 10 (5%) had gluten sensitivity;
- ○
- 10 (5%) had celiac disease;
- ○
- 2 (1%) had colitis;
- ○
- 43 (21.5%) had Candidiasis;
- ○
- 15 (7.5%) had leaky gut;
- ○
- 35 (17.5%) had parasites;
- ○
- 17 (8.5%) had H. Pylori exposure;
- ○
- 37 (18.5%) had gastroesophageal reflux disease (GERD);
- ○
- 0% had a history of C. Difficile during treatment with dapsone; and
- ○
- 83 (41.5%) had ‘other’ gastrointestinal dysfunction (Irritable Bowel Syndrome [IBS]).
- Elevated Liver Function Tests (LFTs): 148 (74%) had one or more of the following transient elevation in LFTs at some point during treatment
- ○
- 90 (45%) had elevated AST;
- ○
- 104 (52%) had elevated ALT;
- ○
- 36 (18%) had alkaline phosphatase;
- ○
- 47 (23.5%) had elevated T. Bilirubin; and
- ○
- 5 (2.5%) had ‘other’ (low albumin).
- Pain Syndromes: 185 (92.5%) had migratory pain, which other research has demonstrated is one of the hallmark symptoms of active Lyme disease [39].
- Deconditioning: 64 (32%) were disabled and/or in physical therapy (PT).
4. Discussion
4.1. Numerous Health Issues Confound Full Recovery
4.2. Neurocognitive Deficits in PTLDS and Lyme-MSIDS
4.3. The Role of Inflammation in Lyme Disease and MSIDS
4.4. Repairing Free Radical Damage: The 4 “R’s”: Replace, Repair, Rebalance, Re-inoculate the G.I. Microbiome
4.5. Important MSIDS Variables Determining Treatment Outcomes
4.6. Healthcare Opportunities Going Forward
The Role of Biofilms and “Persisters” in Chronic Disease
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Disclaimer
References
- Kugeler, K.J.; Farley, G.M.; Forrester, J.D.; Mead, P.S. Geographic Distribution and Expansion of Human Lyme Disease, United States. Emerg. Infect. Dis. 2015, 21, 1455–1457. [Google Scholar] [CrossRef] [PubMed]
- How Many People Get Lyme Disease? | Lyme Disease | CDC. Available online: https://www.cdc.gov/lyme/stats/humancases.html (accessed on 13 February 2018).
- Rosenberg, R. Vital Signs: Trends in Reported Vectorborne Disease Cases—United States and Territories, 2004–2016. MMWR Morb. Mortal. Wkly. Rep. 2018, 67. [Google Scholar] [CrossRef] [PubMed]
- Klempner, M.S.; Hu, L.T.; Evans, J.; Schmid, C.H.; Johnson, G.M.; Trevino, R.P.; Norton, D.; Levy, L.; Wall, D.; McCall, J.; et al. Two Controlled Trials of Antibiotic Treatment in Patients with Persistent Symptoms and a History of Lyme Disease. N. Engl. J. Med. 2001, 345, 85–92. [Google Scholar] [CrossRef] [PubMed]
- Johnson, L.; Wilcox, S.; Mankoff, J.; Stricker, R.B. Severity of chronic Lyme disease compared to other chronic conditions: A quality of life survey. PeerJ 2014, 2, e322. [Google Scholar] [CrossRef] [PubMed]
- Ward, B.W. Multiple Chronic Conditions Among US Adults: A 2012 Update. Prev. Chronic Dis. 2014, 11. [Google Scholar] [CrossRef] [PubMed]
- Public Information Office. Nearly 1 in 5 People Have a Disability in the U.S., Census Bureau Reports-Miscellaneous-Newsroom-U.S. Census Bureau. Available online: https://www.census.gov/newsroom/releases/archives/miscellaneous/cb12-134.html (accessed on 20 June 2018).
- Lyme Disease | 2017 Case Definition. Available online: https://wwwn.cdc.gov/nndss/conditions/lyme-disease/case-definition/2017/ (accessed on 21 May 2018).
- Stricker, R.B.; Johnson, L. Let’s tackle the testing. BMJ 2007, 335, 1008. [Google Scholar] [CrossRef] [PubMed]
- Surveillance Case Definitions for Current and Historical Conditions | NNDSS. Available online: https://wwwn.cdc.gov/nndss/conditions/ (accessed on 8 July 2018).
- Rudenko, N.; Golovchenko, M.; Grubhoffer, L.; Oliver, J.H. Updates on Borrelia burgdorferi sensu lato complex with respect to public health. Ticks Tick-Borne Dis. 2011, 2, 123–128. [Google Scholar] [CrossRef] [PubMed]
- Girard, Y.A.; Fedorova, N.; Lane, R.S. Genetic Diversity of Borrelia burgdorferi and Detection of B. bissettii-Like DNA in Serum of North-Coastal California Residents. J. Clin. Microbiol. 2011, 49, 945–954. [Google Scholar] [CrossRef] [PubMed]
- Margos, G.; Fedorova, N.; Kleinjan, J.E.; Hartberger, C.; Schwan, T.G.; Sing, A.; Fingerle, V. Borrelia lanei sp. nov. extends the diversity of Borrelia species in California. Int. J. Syst. Evol. Microbiol. 2017, 67, 3872–3876. [Google Scholar] [CrossRef] [PubMed]
- Molloy, P.J.; Telford, S.R.; Chowdri, H.R.; Lepore, T.J.; Gugliotta, J.L.; Weeks, K.E.; Hewins, M.E.; Goethert, H.K.; Berardi, V.P. Borrelia miyamotoi Disease in the Northeastern United States: A Case Series. Ann. Intern. Med. 2015, 163, 91–98. [Google Scholar] [CrossRef] [PubMed]
- Krause, P.J.; Carroll, M.; Fedorova, N.; Brancato, J.; Dumouchel, C.; Akosa, F.; Narasimhan, S.; Fikrig, E.; Lane, R.S. Human Borrelia miyamotoi infection in California: Serodiagnosis is complicated by multiple endemic Borrelia species. PLoS ONE 2018, 13, e0191725. [Google Scholar] [CrossRef] [PubMed]
- Myalgic Encephalomyelitis/Chronic Fatigue Syndrome (ME/CFS) | Myalgic Encephalomyelitis/Chronic Fatigue Syndrome (ME/CFS) | CDC. Available online: https://www.cdc.gov/me-cfs/index.html (accessed on 21 May 2018).
- Mozayeni, B.R.; Maggi, R.G.; Bradley, J.M.; Breitschwerdt, E.B. Rheumatological presentation of Bartonella koehlerae and Bartonella henselae bacteremias. Medicine 2018, 97. [Google Scholar] [CrossRef] [PubMed]
- Breitschwerdt, E.B. Did Bartonella henselae contribute to the deaths of two veterinarians? Parasit. Vectors 2015, 8, 317. [Google Scholar] [CrossRef] [PubMed]
- Maggi, R.G.; Mozayeni, B.R.; Pultorak, E.L.; Hegarty, B.C.; Bradley, J.M.; Correa, M.; Breitschwerdt, E.B. Bartonella spp. Bacteremia and Rheumatic Symptoms in Patients from Lyme Disease-endemic Region. Emerg. Infect. Dis. 2012, 18, 783–791. [Google Scholar] [CrossRef] [PubMed]
- Miklossy, J. Historic evidence to support a causal relationship between spirochetal infections and Alzheimer’s disease. Front. Aging Neurosci. 2015, 7, 46. [Google Scholar] [CrossRef] [PubMed]
- Allen, H.B. Alzheimer’s Disease: Assessing the Role of Spirochetes, Biofilms, the Immune System, and Amyloid-β with Regard to Potential Treatment and Prevention. J. Alzheimers Dis. 2016, 53, 1271–1276. [Google Scholar] [CrossRef] [PubMed]
- Readhead, B.; Haure-Mirande, J.-V.; Funk, C.C.; Richards, M.A.; Shannon, P.; Haroutunian, V.; Sano, M.; Liang, W.S.; Beckmann, N.D.; Price, N.D.; et al. Multiscale Analysis of Independent Alzheimer’s Cohorts Finds Disruption of Molecular, Genetic, and Clinical Networks by Human Herpesvirus. Neuron 2018, 99. [Google Scholar] [CrossRef] [PubMed]
- Brookmeyer, R.; Abdalla, N.; Kawas, C.H.; Corrada, M.M. Forecasting the prevalence of preclinical and clinical Alzheimer’s disease in the United States. Alzheimers Dement. J. Alzheimers Assoc. 2018, 14, 121–129. [Google Scholar] [CrossRef] [PubMed]
- Autoimmune Disease Statistics • American Autoimmune Related Diseases Association (AARDA) 2016. Available online: www.aarda.org/news-information/statistics/ (accessed on 21 May 2018).
- Feder, H.M.; Johnson, B.J.B.; O’Connell, S.; Shapiro, E.D.; Steere, A.C.; Wormser, G.P.; Ad Hoc International Lyme Disease Group; Agger, W.A.; Artsob, H.; Auwaerter, P.; Dumler, J.S.; et al. A critical appraisal of “chronic Lyme disease”. N. Engl. J. Med. 2007, 357, 1422–1430. [Google Scholar] [CrossRef]
- Touradji, P.; Aucott, J.N.; Yang, T.; Rebman, A.W.; Bechtold, K.T. Cognitive Decline in Post-treatment Lyme Disease Syndrome. Arch. Clin. Neuropsychol. [CrossRef] [PubMed]
- Lerner, A.; Matthias, T. Changes in intestinal tight junction permeability associated with industrial food additives explain the rising incidence of autoimmune disease. Autoimmun. Rev. 2015, 14, 479–489. [Google Scholar] [CrossRef] [PubMed]
- Moutailler, S.; Valiente Moro, C.; Vaumourin, E.; Michelet, L.; Tran, F.H.; Devillers, E.; Cosson, J.-F.; Gasqui, P.; Van, V.T.; Mavingui, P.; et al. Co-infection of Ticks: The Rule Rather Than the Exception. PLoS Negl. Trop. Dis. 2016, 10, e0004539. [Google Scholar] [CrossRef] [PubMed]
- Nelder, M.P.; Russell, C.B.; Sheehan, N.J.; Sander, B.; Moore, S.; Li, Y.; Johnson, S.; Patel, S.N.; Sider, D. Human pathogens associated with the blacklegged tick Ixodes scapularis: A systematic review. Parasit. Vectors 2016, 9, 265. [Google Scholar] [CrossRef] [PubMed]
- Breitschwerdt, E.B. Bartonellosis: One Health Perspectives for an Emerging Infectious Disease. ILAR J. 2014, 55, 46–58. [Google Scholar] [CrossRef] [PubMed]
- Breitschwerdt, E.B. Bartonellosis, One Health and all creatures great and small. Vet. Dermatol. 2017, 28, 96-e21. [Google Scholar] [CrossRef] [PubMed]
- Nicolson, G.; Haier, J. Role of chronic bacterial and viral infections in neurodegenerative, neurobehavioural, psychiatric, autoimmune and fatiguing illnesses: Part 2. Br. J. Med. Pract. 2010, 3, 301–310. [Google Scholar]
- Simecka, J.W.; Ross, S.E.; Cassell, G.H.; Davis, J.K. Interactions of mycoplasmas with b cells: Antibody production and nonspecific effects. Clin. Infect. Dis. 1993, 17 (Suppl. 1), 176s–182s. [Google Scholar] [CrossRef]
- Furr, P.M.; Taylor-Robinson, D.; Webster, A.D. Mycoplasmas and ureaplasmas in patients with hypogammaglobulinaemia and their role in arthritis: microbiological observations over twenty years. Ann. Rheum. Dis. 1994, 53, 183–187. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.; Li, D.; Paulus, B.; Wilson, I.; Chadwick, V.S. High prevalence of mycoplasma pneumoniae in intestinal mucosal biopsies from patients with inflammatory bowel disease and controls. Dig. Dis. Sci. 2001, 46, 2529–2535. [Google Scholar] [CrossRef] [PubMed]
- Maeda, Y.; Kurakawa, T.; Umemoto, E.; Motooka, D.; Ito, Y.; Gotoh, K.; Hirota, K.; Matsushita, M.; Furuta, Y.; Narazaki, M.; et al. Dysbiosis Contributes to Arthritis Development via Activation of Autoreactive T Cells in the Intestine. Arthritis Rheumatol. 2016, 68, 2646–2661. [Google Scholar] [CrossRef] [PubMed]
- Parks, C.G.; Miller, F.W.; Pollard, K.M.; Selmi, C.; Germolec, D.; Joyce, K.; Rose, N.R.; Humble, M.C. Expert panel workshop consensus statement on the role of the environment in the development of autoimmune disease. Int. J. Mol. Sci. 2014, 15, 14269–14297. [Google Scholar] [CrossRef] [PubMed]
- Jomova, K.; Vondrakova, D.; Lawson, M.; Valko, M. Metals, oxidative stress and neurodegenerative disorders. Mol. Cell. Biochem. 2010, 345, 91–104. [Google Scholar] [CrossRef] [PubMed]
- Horowitz, R.I. Clinical Roundup: Selected Treatment Options for Lyme Disease. Altern. Complement. Ther. 2012, 18, 220–225. [Google Scholar] [CrossRef]
- Bransfield, R.C. Suicide and Lyme and associated diseases. Neuropsychiatr. Dis. Treat. 2017, 13, 1575–1587. [Google Scholar] [CrossRef] [PubMed]
- Ramesh, G.; Didier, P.J.; England, J.D.; Santana-Gould, L.; Doyle-Meyers, L.A.; Martin, D.S.; Jacobs, M.B.; Philipp, M.T. Inflammation in the Pathogenesis of Lyme Neuroborreliosis. Am. J. Pathol. 2015, 185, 1344–1360. [Google Scholar] [CrossRef] [PubMed]
- Horowitz, R. Why Can’t I Get Better? Solving the Mystery of Lyme and Chronic Disease, 1st ed.; St. Martin’s Press: New York, NY, USA, 2013; ISBN 978-1-250-01940-0. [Google Scholar]
- Cerar, T.; Strle, F.; Stupica, D.; Ruzic-Sabljic, E.; McHugh, G.; Steere, A.C.; Strle, K. Differences in Genotype, Clinical Features, and Inflammatory Potential of Borrelia burgdorferi sensu stricto Strains from Europe and the United States. Emerg. Infect. Dis. 2016, 22, 818–827. [Google Scholar] [CrossRef] [PubMed]
- Cimmino, M.A.; Trevisan, G. Lyme arthritis presenting as adult onset Still’s disease. Clin. Exp. Rheumatol. 1989, 7, 305–308. [Google Scholar] [PubMed]
- Kologrivova, E.N.; Baraulina, A.S.; Nechaeva, S.V.; Shestakova, I.I.; Stronin, O.V.; Vlasova, N.M.; Sal’nikova, O.M. Intensity of the production of rheumatoid factor in patients with different degrees of sensitization to Borrelia garinii antigens. Zh. Mikrobiol. Epidemiol. Immunobiol. 2005, 80–83. [Google Scholar]
- Fagan, D.L.; Slaughter, C.A.; Capra, J.D.; Sullivan, T.J. Monoclonal antibodies to immunoglobulin G4 induce histamine release from human basophils in vitro. J. Allergy Clin. Immunol. 1982, 70, 399–404. [Google Scholar] [CrossRef]
- Fallon, B.A.; Levin, E.S.; Schweitzer, P.J.; Hardesty, D. Inflammation and central nervous system Lyme disease. Neurobiol. Dis. 2010, 37, 534–541. [Google Scholar] [CrossRef] [PubMed]
- Pachner, A.R.; Steiner, I. Lyme neuroborreliosis: Infection, immunity, and inflammation. Lancet Neurol. 2007, 6, 544–552. [Google Scholar] [CrossRef]
- Whitmire, W.M.; Garon, C.F. Specific and nonspecific responses of murine B cells to membrane blebs of Borrelia burgdorferi. Infect. Immun. 1993, 61, 1460–1467. [Google Scholar] [PubMed]
- Clarkson, T.W.; Magos, L. The toxicology of mercury and its chemical compounds. Crit. Rev. Toxicol. 2006, 36, 609–662. [Google Scholar] [CrossRef] [PubMed]
- Cooper, G.S.; Parks, C.G.; Treadwell, E.L.; St Clair, E.W.; Gilkeson, G.S.; Dooley, M.A. Occupational risk factors for the development of systemic lupus erythematosus. J. Rheumatol. 2004, 31, 1928–1933. [Google Scholar] [PubMed]
- Gallagher, C.M.; Meliker, J.R. Mercury and thyroid autoantibodies in U.S. women, NHANES 2007-2008. Environ. Int. 2012, 40, 39–43. [Google Scholar] [CrossRef] [PubMed]
- Bransfield, R.C. The Psychoimmunology of Lyme/Tick-Borne Diseases and its Association with Neuropsychiatric Symptoms. Open Neurol. J. 2012, 6, 88–93. [Google Scholar] [CrossRef] [PubMed]
- Pietikäinen, A.; Maksimow, M.; Kauko, T.; Hurme, S.; Salmi, M.; Hytönen, J. Cerebrospinal fluid cytokines in Lyme neuroborreliosis. J. Neuroinflamm. 2016, 13, 273. [Google Scholar] [CrossRef] [PubMed]
- Patrick, L. Mercury toxicity and antioxidants: Part 1: Role of glutathione and alpha-lipoic acid in the treatment of mercury toxicity. Altern. Med. Rev. J. Clin. Ther. 2002, 7, 456–471. [Google Scholar]
- Manzel, A.; Muller, D.N.; Hafler, D.A.; Erdman, S.E.; Linker, R.A.; Kleinewietfeld, M. Role of “Western diet” in inflammatory autoimmune diseases. Curr. Allergy Asthma Rep. 2014, 14, 404. [Google Scholar] [CrossRef] [PubMed]
- Maintz, L.; Novak, N. Histamine and histamine intolerance. Am. J. Clin. Nutr. 2007, 85, 1185–1196. [Google Scholar] [CrossRef] [PubMed]
- Akin, C. Mast cell activation syndromes presenting as anaphylaxis. Immunol. Allergy Clin. N. Am. 2015, 35, 277–285. [Google Scholar] [CrossRef] [PubMed]
- Prasad, A.S.; Beck, F.W.J.; Bao, B.; Fitzgerald, J.T.; Snell, D.C.; Steinberg, J.D.; Cardozo, L.J. Zinc supplementation decreases incidence of infections in the elderly: Effect of zinc on generation of cytokines and oxidative stress. Am. J. Clin. Nutr. 2007, 85, 837–844. [Google Scholar] [CrossRef] [PubMed]
- Bralley, A.; Lord, R. Laboratory Evaluations in Molecular Medicine; IAMM: Norcross, GA, USA, 2001. [Google Scholar]
- Nicolson, G.; Settineri, R.; Ellithorpe, R. Lipid Replacement Therapy with a Glycophospholipid Formulation with NADH and CoQ10 Significantly Reduces Fatigue in Intractable Chronic Fatiguing Illnesses and Chronic Lyme Disease Patients. Int. J. Clin. Med. 2012, 3, 163–170. [Google Scholar] [CrossRef]
- Neustadt, J.; Pieczenik, S.R. Medication-induced mitochondrial damage and disease. Mol. Nutr. Food Res. 2008, 52, 780–788. [Google Scholar] [CrossRef] [PubMed]
- Nicolson, G. Lipid Replacement as an Adjunct to Therapy for Chronic Fatigue, Anti-Aging and Restoration of Mitochondrial Function. Available online: http://mytoprime.com/clinical_papers/lrt_jana_2003.html (accessed on 27 May 2018).
- Balakrishnan, N.; Ericson, M.; Maggi, R.; Breitschwerdt, E.B. Vasculitis, cerebral infarction and persistent Bartonella henselae infection in a child. Parasit. Vectors 2016, 9, 254. [Google Scholar] [CrossRef] [PubMed]
- Knapp, K.L.; Rice, N.A. Human Coinfection with Borrelia burgdorferi and Babesia microti in the United States. J. Parasitol. Res. 2015, 2015, 587131. [Google Scholar] [CrossRef] [PubMed]
- Hájek, T.; Pasková, B.; Janovská, D.; Bahbouh, R.; Hájek, P.; Libiger, J.; Höschl, C. Higher prevalence of antibodies to Borrelia burgdorferi in psychiatric patients than in healthy subjects. Am. J. Psychiatry 2002, 159, 297–301. [Google Scholar] [CrossRef] [PubMed]
- Finkel, M.J.; Halperin, J.J. Nervous System Lyme Borreliosis—Revisited. Arch. Neurol. 1992, 49, 102–107. [Google Scholar] [CrossRef] [PubMed]
- Berczi, I. The pituitary gland, psychoneuroimmunology and infectious disease. In Psychoneuroimmunology, Stress and Infection; Friedman, H., Klein, T., Friedman, A., Eds.; CRC Press: Boca Raton, FL, USA, 1996; pp. 79–109. [Google Scholar]
- Anisman, H.; Baines, M.G.; Berczi, I.; Bernstein, C.N.; Blennerhassett, M.G.; Gorczynski, R.M.; Greenberg, A.H.; Kisil, F.T.; Mathison, R.D.; Nagy, E.; et al. Neuroimmune mechanisms in health and disease: 2. Disease. Can. Med. Assoc. J. 1996, 155, 1075–1082. [Google Scholar]
- Berczi, I.; Baragar, F.D.; Chalmers, I.M.; Keystone, E.C.; Nagy, E.; Warrington, R.J. Hormones in self tolerance and autoimmunity: A role in the pathogenesis of rheumatoid arthritis? Autoimmunity 1993, 16, 45–56. [Google Scholar] [CrossRef] [PubMed]
- Milrad, S.F.; Hall, D.L.; Jutagir, D.R.; Lattie, E.G.; Czaja, S.J.; Perdomo, D.M.; Fletcher, M.A.; Klimas, N.; Antoni, M.H. Depression, evening salivary cortisol and inflammation in chronic fatigue syndrome: A psychoneuroendocrinological structural regression model. Int. J. Psychophysiol. 2017. [Google Scholar] [CrossRef] [PubMed]
- Haack, M.; Sanchez, E.; Mullington, J.M. Elevated Inflammatory Markers in Response to Prolonged Sleep Restriction Are Associated with Increased Pain Experience in Healthy Volunteers. Sleep 2007, 30, 1145–1152. [Google Scholar] [CrossRef] [PubMed]
- Lorton, D.; Lubahn, C.L.; Estus, C.; Millar, B.A.; Carter, J.L.; Wood, C.A.; Bellinger, D.L. Bidirectional Communication between the Brain and the Immune System: Implications for Physiological Sleep and Disorders with Disrupted Sleep. Neuroimmunomodulation 2006, 13, 357–374. [Google Scholar] [CrossRef] [PubMed]
- Greenberg, H.E.; Ney, G.; Scharf, S.M.; Ravdin, L.; Hilton, E. Sleep quality in Lyme disease. Sleep 1995, 18, 912–916. [Google Scholar] [PubMed]
- Dagan, Y.; Eisenstein, M. Circadian rhythm sleep disorders: Toward a more precise definition and diagnosis. Chronobiol. Int. 1999, 16, 213–222. [Google Scholar] [CrossRef] [PubMed]
- Mullington, J.M.; Hinze-Selch, D.; Pollmächer, T. Mediators of inflammation and their interaction with sleep: Relevance for chronic fatigue syndrome and related conditions. Ann. N. Y. Acad. Sci. 2001, 933, 201–210. [Google Scholar] [CrossRef] [PubMed]
- Parish, J.M. Sleep-related problems in common medical conditions. Chest 2009, 135, 563–572. [Google Scholar] [CrossRef] [PubMed]
- Vgontzas, A.N.; Papanicolaou, D.A.; Bixler, E.O.; Kales, A.; Tyson, K.; Chrousos, G.P. Elevation of Plasma Cytokines in Disorders of Excessive Daytime Sleepiness: Role of Sleep Disturbance and Obesity. J. Clin. Endocrinol. Metab. 1997, 82, 1313–1316. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Monco, J.C.; Seidman, R.J.; Benach, J.L. Experimental immunization with Borrelia burgdorferi induces development of antibodies to gangliosides. Infect. Immun. 1995, 63, 4130–4137. [Google Scholar] [PubMed]
- Jacob, G.; Robertson, D.; Mosqueda-Garcia, R.; Ertl, A.C.; Robertson, R.M.; Biaggioni, I. Hypovolemia in syncope and orthostatic intolerance role of the renin-angiotensin system. Am. J. Med. 1997, 103, 128–133. [Google Scholar] [CrossRef]
- Jordan, J.; Shannon, J.R.; Diedrich, A.; Black, B.K.; Robertson, D. Increased Sympathetic Activation in Idiopathic Orthostatic Intolerance: Role of Systemic Adrenoreceptor Sensitivity. Hypertension 2002, 39, 173–178. [Google Scholar] [CrossRef] [PubMed]
- Karas, B.; Grubb, B.P.; Boehm, K.; Kip, K. The postural orthostatic tachycardia syndrome: A potentially treatable cause of chronic fatigue, exercise intolerance, and cognitive impairment in adolescents. Pac. Clin. Electrophysiol. PACE 2000, 23, 344–351. [Google Scholar] [CrossRef]
- Rupprecht Tobias, A.; Elstner, M.; Weil, S.; Pfister, H.-W. Autoimmune-mediated polyneuropathy triggered by borrelial infection? Muscle Nerve 2008, 37, 781–785. [Google Scholar] [CrossRef] [PubMed]
- Staud, R. Autonomic dysfunction in fibromyalgia syndrome: Postural orthostatic tachycardia. Curr. Rheumatol. Rep. 2008, 10, 463–466. [Google Scholar] [CrossRef] [PubMed]
- Kanjwal, K.; Karabin, B.; Kanjwal, Y.; Grubb, B.P. Postural orthostatic tachycardia syndrome following Lyme disease. Cardiol. J. 2011, 18, 63–66. [Google Scholar] [PubMed]
- Raj, S.R. The Postural Tachycardia Syndrome (POTS): Pathophysiology, Diagnosis & Management. Indian Pac. Electrophysiol. J. 2006, 6, 84–99. [Google Scholar]
- Dysautonomia International: Postural Orthostatic Tachycardia Syndrome. Available online: http://www.dysautonomiainternational.org/page.php?ID=30 (accessed on 14 July 2018).
- Rooney, P.J.; Jenkins, R.T.; Buchanan, W.W. A short review of the relationship between intestinal permeability and inflammatory joint disease. Clin. Exp. Rheumatol. 1990, 8, 75–83. [Google Scholar] [PubMed]
- Knight, B.; Matthews, V.B.; Akhurst, B.; Croager, E.J.; Klinken, E.; Abraham, L.J.; Olynyk, J.K.; Yeoh, G. Liver inflammation and cytokine production, but not acute phase protein synthesis, accompany the adult liver progenitor (oval) cell response to chronic liver injury. Immunol. Cell Biol. 2005, 83, 364–374. [Google Scholar] [CrossRef] [PubMed]
- Horowitz, H.W.; Dworkin, B.; Forseter, G.; Nadelman, R.B.; Connolly, C.; Luciano, B.B.; Nowakowski, J.; O’Brien, T.A.; Calmann, M.; Wormser, G.P. Liver function in early Lyme disease. Hepatology 1996, 23, 1412–1417. [Google Scholar] [CrossRef] [PubMed]
- Zaidi, S.A.; Singer, C. Gastrointestinal and hepatic manifestations of tickborne diseases in the United States. Clin. Infect. Dis. 2002, 34, 1206–1212. [Google Scholar] [CrossRef] [PubMed]
- Shimizu, Y. Liver in systemic disease. World J. Gastroenterol. 2008, 14, 4111–4119. [Google Scholar] [CrossRef] [PubMed]
- Shadick, N.A.; Phillips, C.B.; Sangha, O.; Logigian, E.L.; Kaplan, R.F.; Wright, E.A.; Fossel, A.H.; Fossel, K.; Berardi, V.; Lew, R.A.; et al. Musculoskeletal and neurologic outcomes in patients with previously treated Lyme disease. Ann. Intern. Med. 1999, 131, 919–926. [Google Scholar] [CrossRef] [PubMed]
- Shadick, N.A. The Long-Term Clinical Outcomes of Lyme Disease: A Population-Based Retrospective Cohort Study. Ann. Intern. Med. 1994, 121, 560. [Google Scholar] [CrossRef] [PubMed]
- Clauw, D.J.; Chrousos, G.P. Chronic pain and fatigue syndromes: Overlapping clinical and neuroendocrine features and potential pathogenic mechanisms. Neuroimmunomodulation 1997, 4, 134–153. [Google Scholar] [CrossRef] [PubMed]
- Citera, M.; Freeman, P.R.; Horowitz, R.I. Empirical validation of the Horowitz Multiple Systemic Infectious Disease Syndrome Questionnaire for suspected Lyme disease. Int. J. Gen. Med. 2017, 10, 249–273. [Google Scholar] [CrossRef] [PubMed]
- Crane, J.D.; Ogborn, D.I.; Cupido, C.; Melov, S.; Hubbard, A.; Bourgeois, J.M.; Tarnopolsky, M.A. Massage Therapy Attenuates Inflammatory Signaling After Exercise-Induced Muscle Damage. Sci. Transl. Med. 2012, 4, 119ra13. [Google Scholar] [CrossRef] [PubMed]
- Moser, M.M.C. Treatment for a 14-Year-Old Girl with Lyme Disease Using Therapeutic Exercise and Gait Training | Physical Therapy | Oxford Academic. Available online: https://academic.oup.com/ptj/article/91/9/1412/2735165 (accessed on 27 May 2018).
- Nazıroğlu, M.; Akkuş, S.; Soyupek, F.; Yalman, K.; Çelik, Ö.; Eriş, S.; Uslusoy, G.A. Vitamins C and E treatment combined with exercise modulates oxidative stress markers in blood of patients with fibromyalgia: A controlled clinical pilot study. Stress 2010, 13, 498–505. [Google Scholar] [CrossRef] [PubMed]
- Wilder, R.L.; Crofford, L.J. Do infectious agents cause rheumatoid arthritis? Clin. Orthop. 1991, 36–41. [Google Scholar] [CrossRef]
- Alves, A.G.F.; Giacomin, M.F.d.A.; Braga, A.L.F.; Sallum, A.M.E.; Pereira, L.A.A.; Farhat, L.C.; Strufaldi, F.L.; Lichtenfels, A.J.d.F.C.; Carvalho, T.d.S.; Nakagawa, N.K.; et al. Influence of air pollution on airway inflammation and disease activity in childhood-systemic lupus erythematosus. Clin. Rheumatol. 2018, 37, 683–690. [Google Scholar] [CrossRef] [PubMed]
- Pfau, J.C.; Serve, K.M.; Noonan, C.W. Autoimmunity and Asbestos Exposure. Autoimmune Dis. 2014, 2014. [Google Scholar] [CrossRef] [PubMed]
- Steere, A.C.; Klitz, W.; Drouin, E.E.; Falk, B.A.; Kwok, W.W.; Nepom, G.T.; Baxter-Lowe, L.A. Antibiotic-refractory Lyme arthritis is associated with HLA-DR molecules that bind a Borrelia burgdorferi peptide. J. Exp. Med. 2006, 203, 961–971. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Yew, W.W.; Barer, M.R. Targeting Persisters for Tuberculosis Control. Antimicrob. Agents Chemother. 2012, 56, 2223–2230. [Google Scholar] [CrossRef] [PubMed]
- Barr, J. A Short History of Dapsone, or an Alternative Model of Drug Development. J. Hist. Med. Allied Sci. 2011, 66, 425–467. [Google Scholar] [CrossRef] [PubMed]
- Horowitz, R.I.; Freeman, P.R. The use of dapsone as a novel “persister” drug in the treatment of chronic Lyme disease/post treatment Lyme disease syndrome. J. Clin. Exp. Dermatol. Res. 2016, 7, 345. [Google Scholar] [CrossRef]
- WHO Model Prescribing Information: Drugs Used in Leprosy. Available online: http://apps.who.int/medicinedocs/en/d/Jh2988e/ (accessed on 27 May 2018).
- Ma, B.; Christen, B.; Leung, D.; Vigo-Pelfrey, C. Serodiagnosis of Lyme borreliosis by western immunoblot: Reactivity of various significant antibodies against Borrelia burgdorferi. J. Clin. Microbiol. 1992, 30, 370–376. [Google Scholar] [PubMed]
- Lee, B.K.; Schwartz, B.S.; Stewart, W.; Ahn, K.D. Provocative chelation with DMSA and EDTA: Evidence for differential access to lead storage sites. Occup. Environ. Med. 1995, 52, 13–19. [Google Scholar] [CrossRef] [PubMed]
- Brewer, J.H.; Thrasher, J.D.; Straus, D.C.; Madison, R.A.; Hooper, D. Detection of mycotoxins in patients with chronic fatigue syndrome. Toxins 2013, 5, 605–617. [Google Scholar] [CrossRef] [PubMed]
- Horowitz, R. How Can I Get Better?: An Action Plan for Treating Resistant Lyme & Chronic Disease, 1st ed.; St. Martin’s Griffin: New York, NY, USA, 2017; ISBN 978-1-250-07054-8. [Google Scholar]
- Jopling, W.H. Side-effects of antileprosy drugs in common use. Lepr. Rev. 1983, 54, 261–270. [Google Scholar] [CrossRef] [PubMed]
- Krupp, L.B.; Hyman, L.G.; Grimson, R.; Coyle, P.K.; Melville, P.; Ahnn, S.; Dattwyler, R.; Chandler, B. Study and treatment of post Lyme disease (STOP-LD): A randomized double masked clinical trial. Neurology 2003, 60, 1923–1930. [Google Scholar] [CrossRef] [PubMed]
- Fallon, B.A.; Keilp, J.G.; Corbera, K.M.; Petkova, E.; Britton, C.B.; Dwyer, E.; Slavov, I.; Cheng, J.; Dobkin, J.; Nelson, D.R.; et al. A randomized, placebo-controlled trial of repeated IV antibiotic therapy for Lyme encephalopathy. Neurology 2008, 70, 992–1003. [Google Scholar] [CrossRef] [PubMed]
- Delong, A.K.; Blossom, B.; Maloney, E.L.; Phillips, S.E. Antibiotic retreatment of Lyme disease in patients with persistent symptoms: A biostatistical review of randomized, placebo-controlled, clinical trials. Contemp. Clin. Trials 2012, 33, 1132–1142. [Google Scholar] [CrossRef] [PubMed]
- Cameron, D.J.; Johnson, L.B.; Maloney, E.L. Evidence assessments and guideline recommendations in Lyme disease: The clinical management of known tick bites, erythema migrans rashes and persistent disease. Expert Rev. Anti-Infect. Ther. 2014, 12, 1103–1135. [Google Scholar] [CrossRef] [PubMed]
- Sapi, E.; Balasubramanian, K.; Poruri, A.; Maghsoudlou, J.S.; Socarras, K.M.; Timmaraju, A.V.; Filush, K.R.; Gupta, K.; Shaikh, S.; Theophilus, P.A.S.; et al. Evidence of in vivo Existence of Borrelia Biofilm in Borrelial Lymphocytomas. Eur. J. Microbiol. Immunol. 2016, 6, 9–24. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y. Persisters, persistent infections and the Yin–Yang model. Emerg. Microbes Infect. 2014, 3, e3. [Google Scholar] [CrossRef] [PubMed]
- Lewis, K. Persister cells, dormancy and infectious disease. Nat. Rev. Microbiol. 2007, 5, 48–56. [Google Scholar] [CrossRef] [PubMed]
- Feng, J.; Auwaerter, P.G.; Zhang, Y. Drug Combinations against Borrelia burgdorferi Persisters in vitro: Eradication Achieved by Using Daptomycin, Cefoperazone and Doxycycline. PLoS ONE 2015, 10, e0117207. [Google Scholar] [CrossRef] [PubMed]
- Feng, J.; Zhang, S.; Shi, W.; Zubcevik, N.; Miklossy, J.; Zhang, Y. Selective Essential Oils from Spice or Culinary Herbs Have High Activity against Stationary Phase and Biofilm Borrelia burgdorferi. Front. Med. 2017, 4. [Google Scholar] [CrossRef] [PubMed]
- Van Zyl, J.M.; Basson, K.; Kriegler, A.; van der Walt, B.J. Mechanisms by which clofazimine and dapsone inhibit the myeloperoxidase system: A possible correlation with their anti-inflammatory properties. Biochem. Pharmacol. 1991, 42, 599–608. [Google Scholar] [CrossRef]
- Tan, Z.S.; Beiser, A.S.; Vasan, R.S.; Roubenoff, R.; Dinarello, C.A.; Harris, T.B.; Benjamin, E.J.; Au, R.; Kiel, D.P.; Wolf, P.A.; et al. Inflammatory markers and the risk of Alzheimer disease: The Framingham Study. Neurology 2007, 68, 1902–1908. [Google Scholar] [CrossRef] [PubMed]
- Park, M.H.; Hong, J.T. Roles of NF-κB in Cancer and Inflammatory Diseases and Their Therapeutic Approaches. Cells 2016, 5, 15. [Google Scholar] [CrossRef] [PubMed]
- Pall, M.L. Common etiology of posttraumatic stress disorder, fibromyalgia, chronic fatigue syndrome and multiple chemical sensitivity via elevated nitric oxide/peroxynitrite. Med. Hypotheses 2001, 57, 139–145. [Google Scholar] [CrossRef] [PubMed]
- Tripathi, P.; Tripathi, P.; Kashyap, L.; Singh, V. The role of nitric oxide in inflammatory reactions. FEMS Immunol. Med. Microbiol. 2007, 51, 443–452. [Google Scholar] [CrossRef] [PubMed]
- Ramasamy, R.; Yan, S.F.; Schmidt, A.M. Receptor for AGE (RAGE): Signaling mechanisms in the pathogenesis of diabetes and its complications. Ann. N. Y. Acad. Sci. 2011, 1243, 88–102. [Google Scholar] [CrossRef] [PubMed]
- Crofts, C.A.P. Hyperinsulinemia: A unifying theory of chronic disease? Diabesity 2015, 1, 34. [Google Scholar] [CrossRef]
- Strle, K.; Drouin, E.E.; Shen, S.; El Khoury, J.; McHugh, G.; Ruzic-Sabljic, E.; Strle, F.; Steere, A.C. Borrelia burgdorferi stimulates macrophages to secrete higher levels of cytokines and chemokines than Borrelia afzelii or Borrelia garinii. J. Infect. Dis. 2009, 200, 1936–1943. [Google Scholar] [CrossRef] [PubMed]
- Lochhead, R.B.; Strle, K.; Kim, N.D.; Kohler, M.J.; Arvikar, S.L.; Aversa, J.M.; Steere, A.C. MicroRNA Expression Shows Inflammatory Dysregulation and Tumor-Like Proliferative Responses in Joints of Patients With Postinfectious Lyme Arthritis. Arthritis Rheumatol. 2017, 69, 1100–1110. [Google Scholar] [CrossRef] [PubMed]
- Dame, T.M.; Orenzoff, B.L.; Palmer, L.E.; Furie, M.B. IFN-γ Alters the Response of Borrelia burgdorferi-Activated Endothelium to Favor Chronic Inflammation. J. Immunol. 2007, 178, 1172–1179. [Google Scholar] [CrossRef] [PubMed]
- Mühlradt, P.F.; Quentmeier, H.; Schmitt, E. Involvement of interleukin-1 (IL-1), IL-6, IL-2, and IL-4 in generation of cytolytic T cells from thymocytes stimulated by a Mycoplasma fermentans-derived product. Infect. Immun. 1991, 59, 3962–3968. [Google Scholar] [PubMed]
- Soloski, M.J.; Crowder, L.A.; Lahey, L.J.; Wagner, C.A.; Robinson, W.H.; Aucott, J.N. Serum Inflammatory Mediators as Markers of Human Lyme Disease Activity. PLoS ONE 2014, 9, e93243. [Google Scholar] [CrossRef] [PubMed]
- Stich, R.W.; Shoda, L.K.; Dreewes, M.; Adler, B.; Jungi, T.W.; Brown, W.C. Stimulation of nitric oxide production in macrophages by Babesia bovis. Infect. Immun. 1998, 66, 4130–4136. [Google Scholar] [PubMed]
- Packer, L.; Tritschler, H.J.; Wessel, K. Neuroprotection by the metabolic antioxidant alpha-lipoic acid. Free Radic. Biol. Med. 1997, 22, 359–378. [Google Scholar] [CrossRef]
- Patrick, L. Toxic metals and antioxidants: Part II. The role of antioxidants in arsenic and cadmium toxicity. Altern. Med. Rev. J. Clin. Ther. 2003, 8, 106–128. [Google Scholar]
- Smith, J.P.; Stock, H.; Bingaman, S.; Mauger, D.; Rogosnitzky, M.; Zagon, I.S. Low-dose naltrexone therapy improves active Crohn’s disease. Am. J. Gastroenterol. 2007, 102, 820–828. [Google Scholar] [CrossRef] [PubMed]
- Cree, B.A.C.; Kornyeyeva, E.; Goodin, D.S. Pilot trial of low-dose naltrexone and quality of life in multiple sclerosis. Ann. Neurol. 2010, 68, 145–150. [Google Scholar] [CrossRef] [PubMed]
- Lie, M.R.K.L.; van der Giessen, J.; Fuhler, G.M.; de Lima, A.; Peppelenbosch, M.P.; van der Ent, C.; van der Woude, C.J. Low dose Naltrexone for induction of remission in inflammatory bowel disease patients. J. Transl. Med. 2018, 16, 55. [Google Scholar] [CrossRef] [PubMed]
- Younger, J.; Mackey, S. Fibromyalgia symptoms are reduced by low-dose naltrexone: A pilot study. Pain Med. 2009, 10, 663–672. [Google Scholar] [CrossRef] [PubMed]
- Lewis, K.N.; Mele, J.; Hayes, J.D.; Buffenstein, R. Nrf2, a guardian of healthspan and gatekeeper of species longevity. Integr. Comp. Biol. 2010, 50, 829–843. [Google Scholar] [CrossRef] [PubMed]
- Singh, K.; Connors, S.L.; Macklin, E.A.; Smith, K.D.; Fahey, J.W.; Talalay, P.; Zimmerman, A.W. Sulforaphane treatment of autism spectrum disorder (ASD). Proc. Natl. Acad. Sci. USA 2014, 111, 15550–15555. [Google Scholar] [CrossRef] [PubMed]
- Valenzano, D.R.; Terzibasi, E.; Genade, T.; Cattaneo, A.; Domenici, L.; Cellerino, A. Resveratrol prolongs lifespan and retards the onset of age-related markers in a short-lived vertebrate. Curr. Biol. 2006, 16, 296–300. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.M.; Wang, Z.R.; Hsieh, T.C.; Bruder, J.L.; Zou, J.G.; Huang, Y.Z. Mechanism of cardioprotection by resveratrol, a phenolic antioxidant present in red wine (Review). Int. J. Mol. Med. 2001, 8, 3–17. [Google Scholar] [CrossRef] [PubMed]
- Yadav, V.S.; Mishra, K.P.; Singh, D.P.; Mehrotra, S.; Singh, D.V.K. Immunomodulatory Effects of Curcumin. Immunopharmacol. Immunotoxicol. 2005, 27, 485–497. [Google Scholar] [CrossRef] [PubMed]
- Nicolson, G.L. Metabolic syndrome and mitochondrial function: Molecular replacement and antioxidant supplements to prevent membrane peroxidation and restore mitochondrial function. J. Cell. Biochem. 2007, 100, 1352–1369. [Google Scholar] [CrossRef] [PubMed]
- Thieben, M.J.; Sandroni, P.; Sletten, D.M.; Benrud-Larson, L.M.; Fealey, R.D.; Vernino, S.; Lennon, V.A.; Shen, W.-K.; Low, P.A. Postural orthostatic tachycardia syndrome: The Mayo clinic experience. Mayo Clin. Proc. 2007, 82, 308–313. [Google Scholar] [CrossRef]
- Watari, M.; Nakane, S.; Mukaino, A.; Nakajima, M.; Mori, Y.; Maeda, Y.; Masuda, T.; Takamatsu, K.; Kouzaki, Y.; Higuchi, O.; et al. Autoimmune postural orthostatic tachycardia syndrome. Ann. Clin. Transl. Neurol. 2018, 5, 486–492. [Google Scholar] [CrossRef] [PubMed]
- Fasano, A. Zonulin and its regulation of intestinal barrier function: The biological door to inflammation, autoimmunity, and cancer. Physiol. Rev. 2011, 91, 151–175. [Google Scholar] [CrossRef] [PubMed]
- Pianta, A.; Arvikar, S.; Strle, K.; Drouin, E.E.; Wang, Q.; Costello, C.E.; Steere, A.C. Evidence of the Immune Relevance of Prevotella copri, a Gut Microbe, in Patients with Rheumatoid Arthritis. Arthritis Rheumatol. 2017, 69, 964–975. [Google Scholar] [CrossRef] [PubMed]
- Rumah, K.R.; Linden, J.; Fischetti, V.A.; Vartanian, T. Isolation of Clostridium perfringens Type B in an Individual at First Clinical Presentation of Multiple Sclerosis Provides Clues for Environmental Triggers of the Disease. PLoS ONE 2013, 8, e76359. [Google Scholar] [CrossRef] [PubMed]
- Weiss, G.; Christensen, H.R.; Zeuthen, L.H.; Vogensen, F.K.; Jakobsen, M.; Frøkiær, H. Lactobacilli and bifidobacteria induce differential interferon-β profiles in dendritic cells. Cytokine 2011, 56, 520–530. [Google Scholar] [CrossRef] [PubMed]
- Foligne, B.; Nutten, S.; Grangette, C.; Dennin, V.; Goudercourt, D.; Poiret, S.; Dewulf, J.; Brassart, D.; Mercenier, A.; Pot, B. Correlation between in vitro and in vivo immunomodulatory properties of lactic acid bacteria. World J. Gastroenterol. 2007, 13, 236–243. [Google Scholar] [CrossRef] [PubMed]
- Kumar, H.; Lund, R.; Laiho, A.; Lundelin, K.; Ley, R.E.; Isolauri, E.; Salminen, S. Gut Microbiota as an Epigenetic Regulator: Pilot Study Based on Whole-Genome Methylation Analysis. mBio 2014, 5. [Google Scholar] [CrossRef] [PubMed]
- Ringel-Kulka, T.; Palsson, O.S.; Maier, D.; Carroll, I.; Galanko, J.A.; Leyer, G.; Ringel, Y. Probiotic bacteria Lactobacillus acidophilus NCFM and Bifidobacterium lactis Bi-07 versus placebo for the symptoms of bloating in patients with functional bowel disorders: A double-blind study. J. Clin. Gastroenterol. 2011, 45, 518–525. [Google Scholar] [CrossRef] [PubMed]
- Gill, H.S.; Rutherfurd, K.J.; Cross, M.L.; Gopal, P.K. Enhancement of immunity in the elderly by dietary supplementation with the probiotic Bifidobacterium lactis HN019. Am. J. Clin. Nutr. 2001, 74, 833–839. [Google Scholar] [CrossRef] [PubMed]
- Kawase, M.; He, F.; Kubota, A.; Harata, G.; Hiramatsu, M. Oral administration of lactobacilli from human intestinal tract protects mice against influenza virus infection. Lett. Appl. Microbiol. 2010, 51, 6–10. [Google Scholar] [CrossRef] [PubMed]
- Nearly Half a Million Americans Suffered from Clostridium Difficile Infections in a Single Year. Available online: https://www.cdc.gov/media/releases/2015/p0225-clostridium-difficile.html (accessed on 6 July 2018).
- Chronic Diseases Are Taxing Our Health Care System and Our Economy. Available online: https://www.statnews.com/2018/05/31/chronic-diseases-taxing-health-care-economy/ (accessed on 20 June 2018).
- Allen, H.B.; Shaver, C.M.; Etzler, C.A.; Joshi, S.G. Autoimmune Diseases of the Innate and Adaptive Immune System including Atopic Dermatitis, Psoriasis, Chronic Arthritis, Lyme Disease, and Alzheimers Disease. Immunochem. Immunopathol. 2015, 1, 1–4. [Google Scholar] [CrossRef]
- Alaedini, A.; Latov, N. Antibodies against OspA epitopes of Borrelia burgdorferi cross-react with neural tissue. J. Neuroimmunol. 2005, 159, 192–195. [Google Scholar] [CrossRef] [PubMed]
- Uhde, M.; Ajamian, M.; Li, X.; Wormser, G.P.; Marques, A.; Alaedini, A. Expression of C-Reactive Protein and Serum Amyloid A in Early to Late Manifestations of Lyme Disease. Clin. Infect. Dis. 2016, 63, 1399–1404. [Google Scholar] [CrossRef] [PubMed]
- Edmondson, D.A.; Barrios, C.S.; Brasel, T.L.; Straus, D.C.; Kurup, V.P.; Fink, J.N. Immune Response among Patients Exposed to Molds. Int. J. Mol. Sci. 2009, 10, 5471–5484. [Google Scholar] [CrossRef] [PubMed]
- Mangin, M.; Sinha, R.; Fincher, K. Inflammation and vitamin D: The infection connection. Inflamm. Res. 2014, 63, 803–819. [Google Scholar] [CrossRef] [PubMed]
- Sommer, C.; Kress, M. Recent findings on how proinflammatory cytokines cause pain: Peripheral mechanisms in inflammatory and neuropathic hyperalgesia. Neurosci. Lett. 2004, 361, 184–187. [Google Scholar] [CrossRef] [PubMed]
- Loggia, M.L.; Chonde, D.B.; Akeju, O.; Arabasz, G.; Catana, C.; Edwards, R.R.; Hill, E.; Hsu, S.; Izquierdo-Garcia, D.; Ji, R.-R.; et al. Evidence for brain glial activation in chronic pain patients. Brain J. Neurol. 2015, 138, 604–615. [Google Scholar] [CrossRef] [PubMed]
- Hutchinson, M. Glial Ties to Persistent Pain. Available online: https://www.the-scientist.com/?articles.view/articleNo/51172/title/Glial-Ties-to-Persistent-Pain/ (accessed on 28 May 2018).
- Lichtenstein, J.H.R.; Hsu, Y.-H.; Gavin, I.M.; Donaghey, T.C.; Molina, R.M.; Thompson, K.J.; Chi, C.-L.; Gillis, B.S.; Brain, J.D. Environmental Mold and Mycotoxin Exposures Elicit Specific Cytokine and Chemokine Responses. PLoS ONE 2015, 10, e0126926. [Google Scholar] [CrossRef]
- Yang, S.-N.; Hsieh, C.-C.; Kuo, H.-F.; Lee, M.-S.; Huang, M.-Y.; Kuo, C.-H.; Hung, C.-H. The Effects of Environmental Toxins on Allergic Inflammation. Allergy Asthma Immunol. Res. 2014, 6, 478–484. [Google Scholar] [CrossRef] [PubMed]
- Steinemann, A. National Prevalence and Effects of Multiple Chemical Sensitivities. J. Occup. Environ. Med. 2018, 60, e152. [Google Scholar] [CrossRef] [PubMed]
- Fitzgerald, K.N.; Brailley, J.A. Case Studies in Integrative and Functional Medicine; Metrametrix: Duluth, GA, USA, 2011; ISBN 978-0-9673949-5-4. [Google Scholar]
- Wu, G.; Fang, Y.-Z.; Yang, S.; Lupton, J.R.; Turner, N.D. Glutathione Metabolism and Its Implications for Health. J. Nutr. 2004, 134, 489–492. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Zhang, X.; Fang, X.; Niimi, M.; Huang, Y.; Piao, H.; Gao, S.; Fan, J.; Yao, J. Glutathione inhibits antibody and complement-mediated immunologic cell injury via multiple mechanisms. Redox Biol. 2017, 12, 571–581. [Google Scholar] [CrossRef] [PubMed]
- Kerstholt, M.; Vrijmoeth, H.; Lachmandas, E.; Oosting, M.; Lupse, M.; Flonta, M.; Dinarello, C.A.; Netea, M.G.; Joosten, L.A.B. Role of glutathione metabolism in host defense against Borrelia burgdorferi infection. Proc. Natl. Acad. Sci. USA 2018, 115, E2320–E2328. [Google Scholar] [CrossRef] [PubMed]
- Stephenson, J. CDC Report on Environmental Toxins. JAMA 2003, 289, 1230–1233. [Google Scholar] [CrossRef] [PubMed]
- Horowitz, R.I. Effects of Shifting the Acid-Base Balance Among Lyme Patients during Jarish Herxheimer Flares: A Small Prospective Study. In Proceedings of the 16th International Scientific Conference on Lyme Disease & Other Tick-Borne Disorders, Philadelphia, PA, USA, 15 November 2003. [Google Scholar]
- Theophilus, P.A.S.; Victoria, M.J.; Socarras, K.M.; Filush, K.R.; Gupta, K.; Luecke, D.F.; Sapi, E. Effectiveness of Stevia Rebaudiana Whole Leaf Extract Against the Various Morphological Forms of Borrelia Burgdorferi in vitro. Eur. J. Microbiol. Immunol. 2015, 5, 268–280. [Google Scholar] [CrossRef] [PubMed]
- Schillaci, D.; Napoli, E.M.; Cusimano, M.G.; Vitale, M.; Ruberto, A. Origanum vulgare subsp. hirtum essential oil prevented biofilm formation and showed antibacterial activity against planktonic and sessile bacterial cells. J. Food Prot. 2013, 76, 1747–1752. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.; Sturrock, A.; Weis, J.J. Intracellular localization of Borrelia burgdorferi within human endothelial cells. Infect. Immun. 1991, 59, 671–678. [Google Scholar] [PubMed]
- Montgomery, R.R.; Nathanson, M.H.; Malawista, S.E. The fate of Borrelia burgdorferi, the agent for Lyme disease, in mouse macrophages. Destruction, survival, recovery. J. Immunol. 1993, 150, 909–915. [Google Scholar] [PubMed]
- Girschick, H.J.; Huppertz, H.I.; Rüssmann, H.; Krenn, V.; Karch, H. Intracellular persistence of Borrelia burgdorferi in human synovial cells. Rheumatol. Int. 1996, 16, 125–132. [Google Scholar] [CrossRef] [PubMed]
- Livengood, J.A.; Gilmore, R.D. Invasion of human neuronal and glial cells by an infectious strain of Borrelia burgdorferi. Microbes Infect. 2006, 8, 2832–2840. [Google Scholar] [CrossRef] [PubMed]
- Krause, P.J.; Telford, S.R.; Ryan, R.; Conrad, P.A.; Wilson, M.; Thomford, J.W.; Spielman, A. Diagnosis of babesiosis: Evaluation of a serologic test for the detection of Babesia microti antibody. J. Infect. Dis. 1994, 169, 923–926. [Google Scholar] [CrossRef] [PubMed]
- Sexton, D.J.; Corey, G.R.; Carpenter, C.; Kong, L.Q.; Gandhi, T.; Breitschwerdt, E.; Hegarty, B.; Chen, S.M.; Feng, H.M.; Yu, X.J.; et al. Dual infection with Ehrlichia chaffeensis and a spotted fever group rickettsia: A case report. Emerg. Infect. Dis. 1998, 4, 311–316. [Google Scholar] [CrossRef] [PubMed]
- Maggi, R.G.; Mascarelli, P.E.; Havenga, L.N.; Naidoo, V.; Breitschwerdt, E.B. Co-infection with Anaplasma platys, Bartonella henselae and Candidatus Mycoplasma haematoparvum in a veterinarian. Parasit. Vectors 2013, 6, 103. [Google Scholar] [CrossRef] [PubMed]
- Lemieux, J.E.; Tran, A.D.; Freimark, L.; Schaffner, S.F.; Goethert, H.; Andersen, K.G.; Bazner, S.; Li, A.; McGrath, G.; Sloan, L.; et al. A global map of genetic diversity in Babesia microti reveals strong population structure and identifies variants associated with clinical relapse. Nat. Microbiol. 2016, 1, 16079. [Google Scholar] [CrossRef] [PubMed]
- Wormser, G.P.; Prasad, A.; Neuhaus, E.; Joshi, S.; Nowakowski, J.; Nelson, J.; Mittleman, A.; Aguero-Rosenfeld, M.; Topal, J.; Krause, P.J. Emergence of resistance to azithromycin-atovaquone in immunocompromised patients with Babesia microti infection. Clin. Infect. Dis. 2010, 50, 381–386. [Google Scholar] [CrossRef] [PubMed]
- Krause, P.J.; Spielman, A.; Telford, S.R.; Sikand, V.K.; McKay, K.; Christianson, D.; Pollack, R.J.; Brassard, P.; Magera, J.; Ryan, R.; et al. Persistent Parasitemia after Acute Babesiosis. N. Engl. J. Med. 1998, 339, 160–165. [Google Scholar] [CrossRef] [PubMed]
- Kuklina, E.M.; Glebezdina, N.S.; Nekrasova, I.V. Role of Melatonin in the Regulation of Differentiation of T Cells Producing Interleukin-17 (Th17). Bull. Exp. Biol. Med. 2016, 160, 656–658. [Google Scholar] [CrossRef] [PubMed]
- Monteleone, P.; Maj, M.; Beinat, L.; Natale, M.; Kemali, D. Blunting by chronic phosphatidylserine administration of the stress-induced activation of the hypothalamo-pituitary-adrenal axis in healthy men. Eur. J. Clin. Pharmacol. 1992, 42, 385–388. [Google Scholar] [CrossRef] [PubMed]
- Berczi, I. Pituitary hormones and immune function. Acta Paediatr. Suppl. 1997, 70–75. [Google Scholar] [CrossRef]
- Laaksonen, D.E.; Niskanen, L.; Punnonen, K.; Nyyssönen, K.; Tuomainen, T.-P.; Salonen, R.; Rauramaa, R.; Salonen, J.T. Sex hormones, inflammation and the metabolic syndrome: A population-based study. Eur. J. Endocrinol. 2003, 149, 601–608. [Google Scholar] [CrossRef] [PubMed]
- Shabsigh, A.; Kang, Y.; Shabsign, R.; Gonzalez, M.; Liberson, G.; Fisch, H.; Goluboff, E. Clomiphene citrate effects on testosterone/estrogen ratio in male hypogonadism. J. Sex. Med. 2005, 2, 716–721. [Google Scholar] [CrossRef] [PubMed]
- Leder, B.Z.; Rohrer, J.L.; Rubin, S.D.; Gallo, J.; Longcope, C. Effects of Aromatase Inhibition in Elderly Men with Low or Borderline-Low Serum Testosterone Levels. J. Clin. Endocrinol. Metab. 2004, 89, 1174–1180. [Google Scholar] [CrossRef] [PubMed]
- Abed, H.; Ball, P.A.; Wang, L.-X. Diagnosis and management of postural orthostatic tachycardia syndrome: A brief review. J. Geriatr. Cardiol. 2012, 9, 61–67. [Google Scholar] [CrossRef] [PubMed]
- Kaufmann, H.; Norcliffe-Kaufmann, L.; Palma, J.-A. Droxidopa in neurogenic orthostatic hypotension. Expert Rev. Cardiovasc. Ther. 2015, 13, 875–891. [Google Scholar] [CrossRef] [PubMed]
- Freeman, R.; Komaroff, A.L. Does the Chronic Fatigue Syndrome Involve the Autonomic Nervous System? Am. J. Med. 1997, 102, 357–364. [Google Scholar] [CrossRef]
- Kisand, K.E.; Prükk, T.; Kisand, K.V.; Lüüs, S.-M.; Kalbe, I.; Uibo, R. Propensity to excessive proinflammatory response in chronic Lyme borreliosis. APMIS 2007, 115, 134–141. [Google Scholar] [CrossRef] [PubMed]
- Younger, D.S.; Orsher, S. Lyme Neuroborreliosis: Preliminary Results from an Urban Referral Center Employing Strict CDC Criteria for Case Selection. Available online: https://www.hindawi.com/journals/nri/2010/525206/ (accessed on 28 May 2018).
- Horowitz, R.I.; Freeman, P.R. Improvement of common variable immunodeficiency using embryonic stem cell therapy in a patient with lyme disease: A clinical case report. Clin. Case Rep. 2018, 6, 1166–1171. [Google Scholar] [CrossRef] [PubMed]
- Tunev, S.S.; Hastey, C.J.; Hodzic, E.; Feng, S.; Barthold, S.W.; Baumgarth, N. Lymphoadenopathy during Lyme Borreliosis Is Caused by Spirochete Migration-Induced Specific B Cell Activation. PLoS Pathog. 2011, 7, e1002066. [Google Scholar] [CrossRef] [PubMed]
- Schutzer, S.E.; Coyle, P.K.; Belman, A.L.; Golightly, M.G.; Drulle, J. Sequestration of antibody to Borrelia burgdorferi in immune complexes in seronegative Lyme disease. Lancet 1990, 335, 312–315. [Google Scholar] [CrossRef]
- Coyle, P.K.; Schutzer, S.E.; Deng, Z.; Krupp, L.B.; Belman, A.L.; Benach, J.L.; Luft, B.J. Detection of Borrelia burgdorferi-specific antigen in antibody-negative cerebrospinal fluid in neurologic Lyme disease. Neurology 1995, 45, 2010–2015. [Google Scholar] [CrossRef] [PubMed]
- Blum, L.K.; Adamska, J.Z.; Martin, D.S.; Rebman, A.W.; Elliott, S.E.; Cao, R.R.L.; Embers, M.E.; Aucott, J.N.; Soloski, M.J.; Robinson, W.H. Robust B Cell Responses Predict Rapid Resolution of Lyme Disease. Front. Immunol. 2018, 9. [Google Scholar] [CrossRef] [PubMed]
- Hypoglycemia-Symptoms and Causes-Mayo Clinic. Available online: https://www.mayoclinic.org/diseases-conditions/hypoglycemia/symptoms-causes/syc-20373685 (accessed on 11 July 2018).
- Whalen, K.A.; McCullough, M.L.; Flanders, W.D.; Hartman, T.J.; Judd, S.; Bostick, R.M. Paleolithic and Mediterranean Diet Pattern Scores Are Inversely Associated with Biomarkers of Inflammation and Oxidative Balance in Adults. J. Nutr. 2016, 146, 1217–1226. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.; Barnes, G.T.; Yang, Q.; Tan, G.; Yang, D.; Chou, C.J.; Sole, J.; Nichols, A.; Ross, J.S.; Tartaglia, L.A.; et al. Chronic inflammation in fat plays a crucial role in the development of obesity-related insulin resistance. J. Clin. Investig. 2003, 112, 1821–1830. [Google Scholar] [CrossRef] [PubMed]
- Peppa, M.; Vlassara, H. Advanced glycation end products and diabetic complications: A general overview. Horm. Athens Greece 2005, 4, 28–37. [Google Scholar] [CrossRef]
- Sullivan, P.G.; Brown, M.R. Mitochondrial aging and dysfunction in Alzheimer’s disease. Prog. Neuropsychopharmacol. Biol. Psychiatry 2005, 29, 407–410. [Google Scholar] [CrossRef] [PubMed]
- Ljungman, M.; Hanawalt, P.C. Efficient protection against oxidative DNA damage in chromatin. Mol. Carcinog. 1992, 5, 264–269. [Google Scholar] [CrossRef] [PubMed]
- Niki, E. Lipid peroxidation products as oxidative stress biomarkers. BioFactors 2008, 34, 171–180. [Google Scholar] [CrossRef]
- Fenga, C.; Gangemi, S.; Teodoro, M.; Rapisarda, V.; Golokhvast, K.; Docea, A.O.; Tsatsakis, A.M.; Costa, C. 8-Hydroxydeoxyguanosine as a biomarker of oxidative DNA damage in workers exposed to low-dose benzene. Toxicol. Rep. 2017, 4, 291–295. [Google Scholar] [CrossRef] [PubMed]
- Martindale, J.L.; Holbrook, N.J. Cellular response to oxidative stress: Signaling for suicide and survival. J. Cell. Physiol. 2002, 192, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Cohen, B.H.; Gold, D.R. Mitochondrial cytopathy in adults: What we know so far. Cleve. Clin. J. Med. 2001, 68, 625–626, 629–642. [Google Scholar] [CrossRef] [PubMed]
- Bennett, M.J.; Powell, S.; Swartling, D.J.; Gibson, K.M. Tiglylglycine excreted in urine in disorders of isoleucine metabolism and the respiratory chain measured by stable isotope dilution GC-MS. Clin. Chem. 1994, 40, 1879–1883. [Google Scholar] [PubMed]
- Naviaux, R.K. Mitochondrial DNA disorders. Eur. J. Pediatr. 2000, 159 (Suppl. 3), S219–S226. [Google Scholar] [CrossRef]
- Fetherolf, M.M.; Boyd, S.D.; Taylor, A.B.; Kim, H.J.; Wohlschlegel, J.A.; Blackburn, N.J.; Hart, P.J.; Winge, D.R.; Winkler, D.D. Copper-zinc superoxide dismutase is activated through a sulfenic acid intermediate at a copper ion entry site. J. Biol. Chem. 2017, 292, 12025–12040. [Google Scholar] [CrossRef] [PubMed]
- Jahnen-Dechent, W.; Ketteler, M. Magnesium basics. Clin. Kidney J. 2012, 5, i3–i14. [Google Scholar] [CrossRef] [PubMed]
- Chung, H.R. Iodine and thyroid function. Ann. Pediatr. Endocrinol. Metab. 2014, 19, 8–12. [Google Scholar] [CrossRef] [PubMed]
- Foster, M.; Samman, S. Zinc and Regulation of Inflammatory Cytokines: Implications for Cardiometabolic Disease. Nutrients 2012, 4, 676–694. [Google Scholar] [CrossRef] [PubMed]
- Auld, D.S.; Bergman, T. Medium- and short-chain dehydrogenase/reductase gene and protein families: The role of zinc for alcohol dehydrogenase structure and function. Cell. Mol. Life Sci. 2008, 65, 3961–3970. [Google Scholar] [CrossRef] [PubMed]
- Hao, Q.; Maret, W. Aldehydes release zinc from proteins. A pathway from oxidative stress/lipid peroxidation to cellular functions of zinc. FEBS J. 2006, 273, 4300–4310. [Google Scholar] [CrossRef] [PubMed]
- Halperin, J.J.; Heyes, M.P. Neuroactive kynurenines in Lyme borreliosis. Neurology 1992, 42, 43–50. [Google Scholar] [CrossRef] [PubMed]
- Albrecht, J.; Zielińska, M.; Norenberg, M.D. Glutamine as a mediator of ammonia neurotoxicity: A critical appraisal. Biochem. Pharmacol. 2010, 80, 1303–1308. [Google Scholar] [CrossRef] [PubMed]
- Ytrebø, L.M.; Sen, S.; Rose, C.; Ten Have, G.A.M.; Davies, N.A.; Hodges, S.; Nedredal, G.I.; Romero-Gomez, M.; Williams, R.; Revhaug, A.; et al. Interorgan ammonia, glutamate, and glutamine trafficking in pigs with acute liver failure. Am. J. Physiol.-Gastrointest. Liver Physiol. 2006, 291, G373–G381. [Google Scholar] [CrossRef] [PubMed]
- Albrecht, J.; Norenberg, M.D. Glutamine: A Trojan horse in ammonia neurotoxicity. Hepatology 2006, 44, 788–794. [Google Scholar] [CrossRef] [PubMed]
- Mittal, V.V.; Sharma, B.C.; Sharma, P.; Sarin, S.K. A randomized controlled trial comparing lactulose, probiotics, and L-ornithine L-aspartate in treatment of minimal hepatic encephalopathy. Eur. J. Gastroenterol. Hepatol. 2011, 23, 725–732. [Google Scholar] [CrossRef] [PubMed]
- Takuma, Y.; Nouso, K.; Makino, Y.; Hayashi, M.; Takahashi, H. Clinical trial: Oral zinc in hepatic encephalopathy: Clinical trial: Oral zinc in hepatic encephalopathy. Aliment. Pharmacol. Ther. 2010, 32, 1080–1090. [Google Scholar] [CrossRef] [PubMed]
- Noseworthy, M.D.; Bray, T.M. Zinc deficiency exacerbates loss in blood-brain barrier integrity induced by hyperoxia measured by dynamic MRI. Proc. Soc. Exp. Biol. Med. Soc. Exp. Biol. Med. N. Y. N 2000, 223, 175–182. [Google Scholar] [CrossRef]
- Irani, V.; Guy, A.J.; Andrew, D.; Beeson, J.G.; Ramsland, P.A.; Richards, J.S. Molecular properties of human IgG subclasses and their implications for designing therapeutic monoclonal antibodies against infectious diseases. Mol. Immunol. 2015, 67, 171–182. [Google Scholar] [CrossRef] [PubMed]
- Pieringer, H.; Parzer, I.; Wöhrer, A.; Reis, P.; Oppl, B.; Zwerina, J. IgG4- related disease: An orphan disease with many faces. Orphanet J. Rare Dis. 2014, 9, 110. [Google Scholar] [CrossRef] [PubMed]
- Widhe, M.; Ekerfelt, C.; Forsberg, P.; Bergström, S.; Ernerudh, J. IgG subclasses in Lyme borreliosis: A study of specific IgG subclass distribution in an interferon-gamma-predominated disease. Scand. J. Immunol. 1998, 47, 575–581. [Google Scholar] [PubMed]
- Pausa, M.; Pellis, V.; Cinco, M.; Giulianini, P.G.; Presani, G.; Perticarari, S.; Murgia, R.; Tedesco, F. Serum-resistant strains of Borrelia burgdorferi evade complement-mediated killing by expressing a CD59-like complement inhibitory molecule. J. Immunol. 2003, 170, 3214–3222. [Google Scholar] [CrossRef]
- Cunningham-Rundles, C. The many faces of common variable immunodeficiency. Hematol. Educ. Program Am. Soc. Hematol. Am. Soc. Hematol. Educ. Program 2012, 2012, 301–305. [Google Scholar] [CrossRef]
- Ombrello, M.J.; Remmers, E.F.; Sun, G.; Freeman, A.F.; Datta, S.; Torabi-Parizi, P.; Subramanian, N.; Bunney, T.D.; Baxendale, R.W.; Martins, M.S.; et al. Cold urticaria, immunodeficiency, and autoimmunity related to PLCG2 deletions. N. Engl. J. Med. 2012, 366, 330–338. [Google Scholar] [CrossRef] [PubMed]
- Hausmann, O.; Warnatz, K. Immunodeficiency in adults a practical guide for the allergist. Allergo J. Int. 2014, 23, 261–268. [Google Scholar] [CrossRef] [PubMed]
- Dorward, D.W.; Fischer, E.R.; Brooks, D.M. Invasion and cytopathic killing of human lymphocytes by spirochetes causing Lyme disease. Clin. Infect. Dis. 1997, 25 (Suppl. 1), 2S–8S. [Google Scholar] [CrossRef]
- Elsner, R.A.; Hastey, C.J.; Olsen, K.J.; Baumgarth, N. Suppression of Long-Lived Humoral Immunity Following Borrelia burgdorferi Infection. PLoS Pathog. 2015, 11, e1004976. [Google Scholar] [CrossRef] [PubMed]
- Kaufman, D.L.; Kogelnik, A.M.; Mozayeni, R.B.; Cherry, N.A.; Breitschwerdt, E.B. Neurological and immunological dysfunction in two patients with Bartonella henselae bacteremia. Clin. Case Rep. 2017, 5, 931–935. [Google Scholar] [CrossRef] [PubMed]
- Breitschwerdt, E.; Sontakke, S.; Hopkins, S. Neurological manifestations of Bartonellosis in immunocompetent patients: A composite of reports from 2005–2012. J. Neuroparasitol. 2012, 3, 1–15. [Google Scholar] [CrossRef]
- Bossou, Y.M.; Serssar, Y.; Allou, A.; Vitry, S.; Momas, I.; Seta, N.; Menotti, J.; Achard, S. Impact of Mycotoxins Secreted by Aspergillus Molds on the Inflammatory Response of Human Corneal Epithelial Cells. Toxins 2017, 9, 197. [Google Scholar] [CrossRef] [PubMed]
- Strausbaugh, L.J.; Caserta, M.T.; Mock, D.J.; Dewhurst, S. Human Herpesvirus 6. Clin. Infect. Dis. 2001, 33, 829–833. [Google Scholar] [CrossRef]
- Office of HIV/AIDS and Infectious Disease Policy, A.S. for H. (ASH) Report of Other TBDS and Co-Infections Subcommittee. Available online: https://www.hhs.gov/ash/advisory-committees/tickbornedisease/reports/other-tbds-2018-5-9/index.html (accessed on 21 May 2018).
- Christian, L.M.; Glaser, R.; Porter, K.; Malarkey, W.B.; Beversdorf, D.; Kiecolt-Glaser, J.K. Poorer self-rated health is associated with elevated inflammatory markers among older adults. Psychoneuroendocrinology 2011, 36, 1495–1504. [Google Scholar] [CrossRef] [PubMed]
- Allen, H.B.; Morales, D.; Jones, K.; Joshi, S. Alzheimers Disease: A Novel Hypothesis Integrating Spirochetes, Biofilm, and the Immune System. J. Neuroinfectious Dis. 2016, 7. [Google Scholar] [CrossRef]
- Costerton, J.W.; Stewart, P.S.; Greenberg, E.P. Bacterial biofilms: A common cause of persistent infections. Science 1999, 284, 1318–1322. [Google Scholar] [CrossRef] [PubMed]
- Parsek, M.R.; Singh, P.K. Bacterial Biofilms: An Emerging Link to Disease Pathogenesis. Annu. Rev. Microbiol. 2003, 57, 677–701. [Google Scholar] [CrossRef] [PubMed]
- Hoa, M.; Syamal, M.; Schaeffer, M.A.; Sachdeva, L.; Berk, R.; Coticchia, J. Biofilms and chronic otitis media: An initial exploration into the role of biofilms in the pathogenesis of chronic otitis media. Am. J. Otolaryngol. 2010, 31, 241–245. [Google Scholar] [CrossRef] [PubMed]
- Al-Mutairi, D.; Kilty, S.J. Bacterial biofilms and the pathophysiology of chronic rhinosinusitis. Curr. Opin. Allergy Clin. Immunol. 2011, 11, 18–23. [Google Scholar] [CrossRef] [PubMed]
- Costerton, J.W.; Montanaro, L.; Arciola, C.R. Biofilm in implant infections: Its production and regulation. Int. J. Artif. Organs 2005, 28, 1062–1068. [Google Scholar] [CrossRef] [PubMed]
- Cushion, M.T.; Collins, M.S.; Linke, M.J. Biofilm formation by Pneumocystis spp. Eukaryot. Cell 2009, 8, 197–206. [Google Scholar] [CrossRef] [PubMed]
- Miyaue, S.; Suzuki, E.; Komiyama, Y.; Kondo, Y.; Morikawa, M.; Maeda, S. Bacterial Memory of Persisters: Bacterial Persister Cells Can Retain Their Phenotype for Days or Weeks After Withdrawal From Colony–Biofilm Culture. Front. Microbiol. 2018, 9. [Google Scholar] [CrossRef] [PubMed]
- Wormser, G.P. Should Patients Infected with Borrelia Burgdorferi No Longer Be Referred to as Having Lyme Disease? Am. J. Med. 2018. [Google Scholar] [CrossRef] [PubMed]
- Abrahamian, F.M. Consequences of delayed diagnosis of Rocky Mountain spotted fever in children—West Virginia, Michigan, Tennessee, and Oklahoma, May–July 2000. Ann. Emerg. Med. 2001, 37, 537–540. [Google Scholar] [CrossRef] [PubMed]
- Hirsch, A.G.; Herman, R.J.; Rebman, A.; Moon, K.A.; Aucott, J.; Heaney, C.; Schwartz, B.S. Obstacles to diagnosis and treatment of Lyme disease in the USA: A qualitative study. BMJ Open 2018, 8, e021367. [Google Scholar] [CrossRef] [PubMed]

| 1. Infections | Laboratory tests for the presence of Borrelia spp., Babesia spp., Bartonella spp., Rickettsia spp., etc. |
| 2. Immune Dysfunction | Laboratory tests for autoimmune markers (ANA, RF), HLA status, immunoglobulin levels, and subclasses |
| 3. Inflammation | Laboratory tests for markers of inflammation, i.e., ESR, CRP, TGFB1, C3a, C4a, and/or VEGF |
| 4. Toxicity | Laboratory tests for heavy metals, mold toxins, pesticides, etc. |
| 5. Allergies | Laboratory tests for IgE levels, food and environmental allergies, histamine, etc. |
| 6. Nutritional and Enzyme Deficiencies | Laboratory tests for amino acids, fatty acids, mineral levels (serum, plasma, red blood cell) |
| 7. Mitochondrial Dysfunction | Clinical evaluation of response to mitochondrial support (NT Factors, CoQ10, l-carnitine), evaluation of mtDNA mutations, etc. |
| 8. Psychological Dysfunction | Clinical evaluation for evidence of depression, anxiety, OCD, PTSD, etc. |
| 9. Neurological Dysfunction | Clinical examination, EMG, Small fiber biopsy, MRI brain, etc. |
| 10. Endocrine Abnormalities | Laboratory evaluation of hormone levels (thyroid, adrenal, sex hormones, Vitamin D) and hormone precursors (DHEA-S, pregnenolone) |
| 11. Sleep Disorders | Clinical evaluation (diet, medication), sleep studies, laboratory evaluation of hormone levels, etc. |
| 12. Autonomic Nervous System Dysfunction | Tilt table testing with or without small fiber biopsies and autonomic/electrodiagnostic testing (EMG), clinical evaluation sitting/standing BP/heart rate |
| 13. Gastrointestinal Dysfunction | Endoscopy, colonoscopy, clinical/laboratory evaluation (celiac markers, H. pylori), Comprehensive Digestive Stool Analysis (CDSA) for bacteria (C. difficile), ova and parasites, Candida, etc. |
| 14. Elevated Liver Enzymes | Laboratory evaluation of AST, ALT, Alkaline phosphatase, total bilirubin, etc. |
| 15. Pain Syndromes | Clinical evaluation, EMG, small fiber biopsy, laboratory markers for autoimmune disease (anti-myelin antibodies), etc. |
| 16. Deconditioning | Clinical evaluation and need for physical therapy |
| N * | Low | Normal | High | ||||
|---|---|---|---|---|---|---|---|
| Frequency | Percent | Frequency | Percent | Frequency | Percent | ||
| IgA | 170 | 27 | 15.88 | 139 | 81.76 | 4 | 2.35 |
| IgM | 171 | 33 | 19.30 | 115 | 67.25 | 23 | 13.45 |
| IgG | 175 | 36 | 20.57 | 131 | 74.86 | 8 | 4.57 |
| SubClass1 | 163 | 45 | 27.61 | 115 | 70.55 | 3 | 1.84 |
| SubClass2 | 164 | 30 | 18.29 | 126 | 76.83 | 8 | 4.88 |
| SubClass3 | 164 | 51 | 31.10 | 112 | 68.29 | 1 | 0.61 |
| SubClass4 | 164 | 14 | 8.54 | 142 | 86.59 | 8 | 4.88 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Horowitz, R.I.; Freeman, P.R. Precision Medicine: The Role of the MSIDS Model in Defining, Diagnosing, and Treating Chronic Lyme Disease/Post Treatment Lyme Disease Syndrome and Other Chronic Illness: Part 2. Healthcare 2018, 6, 129. https://doi.org/10.3390/healthcare6040129
Horowitz RI, Freeman PR. Precision Medicine: The Role of the MSIDS Model in Defining, Diagnosing, and Treating Chronic Lyme Disease/Post Treatment Lyme Disease Syndrome and Other Chronic Illness: Part 2. Healthcare. 2018; 6(4):129. https://doi.org/10.3390/healthcare6040129
Chicago/Turabian StyleHorowitz, Richard I., and Phyllis R. Freeman. 2018. "Precision Medicine: The Role of the MSIDS Model in Defining, Diagnosing, and Treating Chronic Lyme Disease/Post Treatment Lyme Disease Syndrome and Other Chronic Illness: Part 2" Healthcare 6, no. 4: 129. https://doi.org/10.3390/healthcare6040129
APA StyleHorowitz, R. I., & Freeman, P. R. (2018). Precision Medicine: The Role of the MSIDS Model in Defining, Diagnosing, and Treating Chronic Lyme Disease/Post Treatment Lyme Disease Syndrome and Other Chronic Illness: Part 2. Healthcare, 6(4), 129. https://doi.org/10.3390/healthcare6040129





