Effect of Skeletal Muscle and Fat Mass on Muscle Strength in the Elderly
Abstract
1. Introduction
2. Materials and Methods
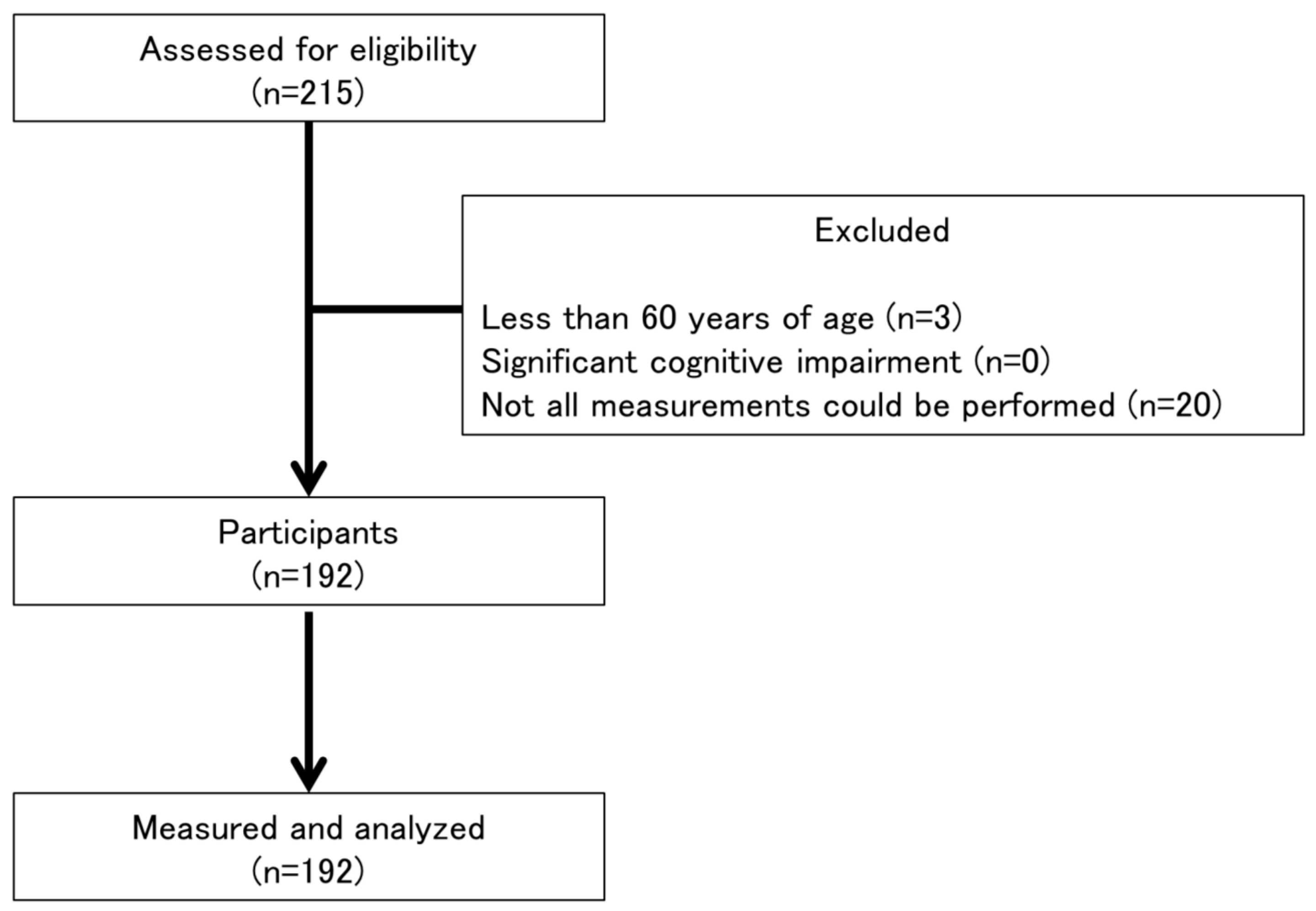
2.1. Participants
2.2. Body Mass Index (BMI)
2.3. Exercise Habits
2.4. SMM and FM
2.5. Grip Strength
2.6. Knee Extension Strength
2.7. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Kuh, D.; Bassey, E.J.; Butterworth, S.; Hardy, R.; Wadsworth, M.E.J. Grip strength, postural control, and functional leg power in a representative cohort of British men and women: Associations with physical activity, health status, and socioeconomic conditions. J. Gerontol. A Biol. Sci. Med. Sci. 2005, 60, 224–231. [Google Scholar] [CrossRef] [PubMed]
- Bijlsma, A.Y.; Meskers, C.G.M.; Van Den Eshof, N.; Westendorp, R.G.; Sipilä, S.; Stenroth, L.; Sillanpää, E.; McPhee, J.S.; Jones, D.A.; Narici, M.V.; et al. Diagnostic criteria for sarcopenia and physical performance. Age 2014, 36, 275–285. [Google Scholar] [CrossRef] [PubMed]
- Cesari, M.; Pahor, M.; Lauretani, F.; Zamboni, V.; Bandinelli, S.; Bernabei, R.; Guralnik, J.M.; Ferrucci, L. Skeletal muscle and mortality results from the InCHIANTI study. J. Gerontol. A Biol. Sci. Med. Sci. 2009, 64, 377–384. [Google Scholar] [CrossRef] [PubMed]
- Studenski, S.; Perera, S.; Wallace, D.; Chandler, J.M.; Duncan, P.W.; Rooney, E.; Fox, M.; Guralnik, J.M. Physical performance measures in the clinical setting. J. Am. Geriatr. Soc. 2003, 51, 314–322. [Google Scholar] [CrossRef] [PubMed]
- Janssen, I.; Baumgartner, R.N.; Ross, R.; Rosenberg, I.H.; Roubenoff, R. Skeletal muscle cutpoints associated with elevated physical disability risk in older men and women. Am. J. Epidemiol. 2004, 159, 413–421. [Google Scholar] [CrossRef] [PubMed]
- Schutz, Y.; Kyle, U.U.; Pichard, C. Fat-free mass index and fat mass index percentiles in Caucasians aged 18–98y. Int. J. Obes. Relat. Metab. Disord. 2002, 26, 953–960. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, Y.; Marcus, R.L.; Lastayo, P.C. Intramuscular adipose tissue and central activation in older adults. Muscle Nerve 2012, 46, 813–816. [Google Scholar] [CrossRef] [PubMed]
- Yudkin, J.S. Inflammation, obesity, and the metabolic syndrome. Horm. Metab. Res. 2007, 39, 707–709. [Google Scholar] [CrossRef] [PubMed]
- Hardin, B.J.; Campbell, K.S.; Smith, J.D.; Arbogast, S.; Smith, J.; Moylan, J.S.; Reid, M.B. TNF-α acts via TNFR1 and muscle-derived oxidants to depress myofibrillar force in murine skeletal muscle. J. Appl. Physiol. 2008, 104, 694–699. [Google Scholar] [CrossRef] [PubMed]
- Goodpaster, B.H.; Theriault, R.; Watkins, S.C.; Kelley, D.E. Intramuscular lipid content is increased in obesity and decreased by weight loss. Metabolism 2000, 49, 467–472. [Google Scholar] [CrossRef]
- Abe, T.; Yaginuma, Y.; Fujita, E.; Thiebaud, R.S.; Kawanishi, M.; Akamine, T. Associations of sit-up ability with sarcopenia classification measures in Japanese older women. Interv. Med. Appl. Sci. 2016, 8, 152–157. [Google Scholar] [CrossRef] [PubMed]
- Bohannon, R.W. Test-retest reliability of hand-held dynamometry during a single session of strength assessment. Phys. Ther. 1986, 66, 206–209. [Google Scholar] [CrossRef] [PubMed]
- Reid, M.B.; Lännergren, J.; Westerblad, H. Respiratory and limb muscle weakness induced by tumor necrosis factor-α: Involvement of muscle myofilaments. Am. J. Respir. Crit. Care Med. 2002, 166, 479–484. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.; Kuk, J.L.; Davidson, L.E.; Hudson, R.; Kilpatrick, K.; Graham, T.E.; Ross, R. Role of exercise in reducing the risk of diabetes and obesity exercise without weight loss is an effective strategy for obesity reduction in obese individuals with and without Type 2 diabetes. J. Appl. Physiol. 2005, 99, 1220–1225. [Google Scholar] [CrossRef] [PubMed]
- Marcus, R.L.; Addison, O.; Kidde, J.P.; Dibble, L.E.; Lastayo, P.C. Skeletal muscle fat infiltration: Impact of age, inactivity, and exercise. J. Nutr. Health Aging 2010, 14, 362–366. [Google Scholar] [CrossRef] [PubMed]
| Variable | Mean ± SD |
|---|---|
| Age (years) | 73.7 ± 5.8 |
| Height (cm) | 151.5 ± 5.1 |
| Weight (kg) | 51.0 ± 8.0 |
| Underweight (BMI < 18.5), n (%) | 19 (10%) |
| Normal weight (18.5 ≤ BMI < 25.0), n (%) | 134 (70%) |
| Obese (25.0 ≤ BMI), n (%) | 39 (20%) |
| Regular exercise, n (%) | 127 (66.1%) |
| Upper limb SMM (kg) | 3.26 ± 0.65 |
| Upper limb FM (kg) | 2.17 ± 0.85 |
| Upper limb SMM/FM ratio | 1.76 ± 1.36 |
| Lower limb SMM (kg) | 10.62 ± 1.54 |
| Lower limb FM (kg) | 5.07 ± 1.54 |
| Lower limb SMM/FM ratio | 2.31 ± 1.04 |
| Grip strength (kg) | 22.1 ± 3.7 |
| Knee extension strength (kgf) | 19.9 ± 5.0 |
| Disease | |
| Hypertension, n (%) | 80 (42%) |
| Orthopedic disease, n (%) | 40 (21%) |
| Hyperglycemia, n (%) | 30 (16%) |
| Cardiovascular disease, n (%) | 10 (5%) |
| Diabetes mellitus, n (%) | 6 (3%) |
| Pulmonary disease, n (%) | 5 (3%) |
| Rheumatoid arthritis, n (%) | 3 (2%) |
| Renal disease, n (%) | 3 (2%) |
| Stroke, n (%) | 2 (1%) |
| Cancer, n (%) | 2 (1%) |
| Others, n (%) | 34 (18%) |
| Variable | β | SE | Standardized Beta | p-Value | R2 | Adjusted R2 |
|---|---|---|---|---|---|---|
| Grip strength | 0.358 | 0.351 | ||||
| Upper limb SMM | 3.889 | 0.384 | 0.691 | <0.001 | ||
| Upper limb FM | −1.137 | 0.294 | −0.269 | <0.001 | ||
| Knee extension strength | 0.231 | 0.227 | ||||
| Lower limb SMM | 1.563 | 0.207 | 0.481 | <0.001 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nonaka, K.; Murata, S.; Shiraiwa, K.; Abiko, T.; Nakano, H.; Iwase, H.; Naito, K.; Horie, J. Effect of Skeletal Muscle and Fat Mass on Muscle Strength in the Elderly. Healthcare 2018, 6, 72. https://doi.org/10.3390/healthcare6030072
Nonaka K, Murata S, Shiraiwa K, Abiko T, Nakano H, Iwase H, Naito K, Horie J. Effect of Skeletal Muscle and Fat Mass on Muscle Strength in the Elderly. Healthcare. 2018; 6(3):72. https://doi.org/10.3390/healthcare6030072
Chicago/Turabian StyleNonaka, Koji, Shin Murata, Kayoko Shiraiwa, Teppei Abiko, Hideki Nakano, Hiroaki Iwase, Koichi Naito, and Jun Horie. 2018. "Effect of Skeletal Muscle and Fat Mass on Muscle Strength in the Elderly" Healthcare 6, no. 3: 72. https://doi.org/10.3390/healthcare6030072
APA StyleNonaka, K., Murata, S., Shiraiwa, K., Abiko, T., Nakano, H., Iwase, H., Naito, K., & Horie, J. (2018). Effect of Skeletal Muscle and Fat Mass on Muscle Strength in the Elderly. Healthcare, 6(3), 72. https://doi.org/10.3390/healthcare6030072





